Abstract
In this study, the effects of temperature, pH, and biochar under cadmium stress on the adsorption characteristics of atrazine in soils in northeast China were studied by batch adsorption method. In the atrazine–Cd(II) coexistence system, the adsorption of atrazine by the soils reached equilibrium within 24 h, but there were some differences in sorption capacities of the three types of soil and the order of adsorption is albic soil > black soil > saline-alkaline soil. With the concentration of atrazine increased, the adsorption capacity of atrazine in the three types of soil gradually increased, the upward trend became more obvious with the ambient temperature of the solution decreased. The adsorption kinetics curves of atrazine in the three types of soil conform to the pseudo-second-order kinetic model and the adsorption isotherm follows the Langmuir model. When atrazine and Cd(II) coexist in soils, the decrease in atrazine adsorption in the soil may be due to the competitive interaction between the two chemicals. Cd(II) occupies part of the adsorption site of atrazine, thus saturating the active site in soils. Since atrazine is a weakly alkaline pesticide, the lower the pH of the soil, the higher the affinity of atrazine for the soil. After adding biochar to the soil, the functional groups in biochar can form π bond with atrazine, which promotes the fixation of atrazine in the soil. The results show that the prevention of atrazine and cadmium leaching can be achieved by appropriately adjusting the pH, temperature, clay content, and organic matter of the soils.
Introduction
The adsorption of pollutants on soil is an important environmental behavior. The physical and chemical properties of the soil determine the migration and fate of pollutants in the environment, and the feasibility of contaminated soil remediation technologies (Ahmed et al., 2018; Smaranda et al., 2016). Among the heavy metals and organic pollutants, cadmium and atrazine are the most common pollutants in soils (Duan et al., 2016; Yang et al., 2015). Atrazine is a potential carcinogen, teratogenic agent, and endocrine disruptor. Atrazine is a kind of organic pollutant which exists in soil for a long time because of its long-term biological accumulation and not easy to be naturally degraded (Cheyns et al., 2012; Khromonygina et al., 2004; Weber et al., 2013). With the increase of the application amount of atrazine, the adsorption of atrazine by soils reached saturation, and the non-adsorbed atrazine will pollute surface water and groundwater through surface runoff and leaching (Alsharekh et al., 2018). Cadmium is a major heavy metal pollutant in the environment. With the development of mining, smelting, and electroplating technology, a large amount of cadmium-containing wastewater is discharged into rivers, causing pollution to the soil and water environment. Pesticides and fertilizers, the use of plastic films, wastewater irrigation, and sludge fertilization are also important sources of cadmium pollution in the soil. Cadmium deposition into soils can affect crop yield and soil quality (Guo et al., 2017; Lian et al., 2015). When atrazine and cadmium coexist in the soil, it is difficult to fix them in the soil at the same time due to the different physical and chemical properties of metals and organic compounds (Salazar-Ledesma et al., 2018). Therefore, it is necessary to study the adsorption characteristics of atrazine in cadmium-contaminated soil. At present, it is found that the adsorption relationship between organic pollutants and heavy metals in soil has been widely concerned by scholars (Wei et al., 2018). Si et al. (2013) observed that the addition of lead increased the adsorption of glyphosate on the two soil types. This was because Pb2+ can replace H+ in the soil and reduce the pH value of the solution, leading to less negative charge on glyphosate molecules and promoting the adsorption of glyphosate in the soil. Meanwhile, it is found that the adsorption of lead and glyphosate is closely related to the soil type, and the adsorption of lead and glyphosate was the largest in red soil. This observation is attributed to higher contents of iron and aluminum oxides and a lower pH value in the red soil than in the other soils. Wei et al. (2018) found that the co-dissolution of heavy metals Cr and atrazine leads to the competitive adsorption of atrazine in the soils, in which the adsorption of atrazine by soil was greatly affected by the specific surface area and porosity of the soil, while the adsorption of Cr(III) by soil was greatly affected by pH and electrostatic adsorption.
However, the actual soil environment may be polluted by both atrazine and Cd(II), due to the widespread use of them. Therefore, it is of great significance to study the adsorption of atrazine and Cd(II). In the current study, there were few reports on the adsorption of atrazine on soils under the stress of heavy metal Cd(II) (Sun et al., 2006; Ke‐Bin et al., 2010). Therefore, this experiment studied the co-adsorption characteristics of Cd(II) and atrazine on the three types of soil, and discussed their potential interaction mechanisms and environmental implications. This study provided a basis for the dynamics of atrazine residue in soils, and pre-uses pH, organic matter, and other factors to promote the adsorption behavior of atrazine in soil containing Cd(II), thus providing a feasible method for preventing atrazine leaching.
Materials and methods
Sample collection and basic physical and chemical properties
The test samples were taken from Yongji city, Daan city, and Gongzhuling city of Jilin province (depth 0–20 cm). After drying at 288 K for about two weeks, all soil samples were ground into 100 mesh. The physical and chemical properties of the three types of soil are shown in Table 1. The silt content ratio of albic clay and black clay was higher and the sand content ratio of saline-alkaline soil was higher. Soil particle composition directly affects its retention of water, gas, and heat, and thus its adsorption of atrazine. Biochar was pyrolysis of corn straw in muff furnace (450°C, 2 h), cooled and screened with 60 mesh, with a pH of 9. The total organic carbon in soils was measured by TOC analyzer. The cation exchange capacity of the soil was measured by replacing the cation in the soil with the neutral salt BaCl2 aqueous solution. The pH meter was used to determine the pH value of samples. The particle size composition of soils was measured by sedimentation method (Taha SM et al., 2014).
Physical and chemical properties of the test soils.

Test design
In the adsorption kinetics experiment of cadmium in soil, 2 g of soil samples and 10 ml of Cd(II) solution with a concentration of 20 mg/l were mixed in a centrifugal tube and placed in a shaking bath (temperature: 298 K, speed: 120 r/min). The working hours are 10, 20, 30, 60, 120, 240, 360, 480, 720, and 1440 min, respectively (Zhang R et al., 2014). After the shock, put the centrifuge tube into the centrifuge (speed was 10,000 r/min, time was 10 min). Cd(II) concentration was determined by flame atomic absorption spectrophotometer after filtration of supernatant.
In the adsorption kinetics experiment, atrazine–Cd mixed solution was prepared, in which atrazine concentration was 5 mg/l and Cd(II) concentration was 20 mg/l. The mixing solution of 2 g soil and 10 ml atrazine–Cd(II) solution was put into a centrifuge tube for 10, 20, 30, 60, 120, 240, 360, 480, 720, and 1440 min, respectively, at a temperature of 298 K. After the end of the shaking, the centrifuge tube was put into a centrifuge and centrifuged at a speed of 10,000 r/min for 10 min. After the supernatant was filtered, the concentration of atrazine in the supernatant was determined by HPLC (Sun et al., 2019).
In the isothermal adsorption experiment, 1, 2, 5, 10, 20, and 30 mg/l atrazine–Cd(II) mixed solution was prepared, and the concentration of Cd(II) in atrazine solution was all 20 mg/l. Then 2 g albic soil, black soil, and saline-alkaline soil were suspended in 10 ml atrazine mixture with different concentrations. The centrifuge tube was placed in a 298 K water bath vibrating screen and oscillated for 24 h. The rest were referred to the adsorption kinetics experiment. The concentration of atrazine in the supernatant was determined by HPLC.
In the thermodynamic experiment, the temperature of the water bath was set as 288, 298, and 308 K respectively, and the oscillation time was 24 h. The remaining operations were simulated by adsorption isotherm test.
In the pH factor experiment, atrazine mixed solution which of Cd(II) concentration of 20 mg/l and atrazine concentration of 5 mg/l was prepared. Then 2 g of soils were mixed with 10 ml of atrazine in a centrifugal tube. The HCl and NaOH of 1 mol/l were used to adjust the pH value of atrazine solution to 3.0, 5.0, 7.0, 9.0, and 11.0, respectively (Sawai et al., 2015). The centrifuge tube was placed in a 298 K water bath vibrating screen and oscillated for 24 h. The rest were referred to the isothermal adsorption experiment.
In the biochar factor experiment, atrazine mixed solution which of Cd(II) concentration of 20 mg/l and atrazine concentration of 5 mg/l was prepared. Biochar of 0.05, 0.25, 0.5, 0.75, and 1% was added to the soil. Then 2 g of soils were mixed with 10 ml of atrazine in a centrifugal tube. The centrifuge tube was placed in a 298 K water bath vibrating screen and oscillated for 24 h. The rest were referred to the isothermal adsorption experiment. The concentration of Cd(II) was determined by flame atomic absorption spectrophotometer and the concentration of atrazine in the supernatant was determined by HPLC.
All tests were performed in triplicate.
In order to describe the adsorption characteristics of atrazine in the soil under heavy metal stress, the experimental data of adsorption kinetics were fitted by the pseudo-second-order kinetic model (1) and Elovich model (2)


The atrazine adsorption capacity was established according to equation (3)

Langmuir (4) and Freundlich (5) models were used to fit the adsorption isotherm data


Thermodynamic parameters of the adsorption process were obtained using the following equations



Results and discussion
Adsorption kinetics of Cd(II) on soils
The adsorption kinetics curves of heavy metal Cd(II) in the three types of soil are shown in Figure 1. As can be seen from Figure 1(a), the adsorption capacity of Cd(II) in saline-alkaline soil, black soil, and albic soil was 723, 62, and 527 mg kg, respectively. As can be seen from Figure 1(b), the heavy metal Cd(II) (concentration of 20 mg/l) was completely absorbed by the three types of soil for about 720 min. At each time point, Cd(II) had the highest adsorption rate in alkaline soil (saline-alkaline soil), followed by neutral soil (black soil), and the lowest adsorption rate in acidic soil (saline-alkaline). In the current study, it can be seen that pH was a major factor affecting the equilibrium time of metal adsorption (Abate and Masini, 2005). As for the adsorption of Cd(II) by soil, it can be preliminarily determined that the residue of heavy metal Cd is larger in alkaline soil, which is consistent with the results of study by Huang et al. (2009).

The adsorption of heavy metal cadmium in the three types of soil (conditions: 2 g of soils, volume of 10 ml Cd solution at a concentration of 20 mg/l, shaking bath at 298 K and 120 r/min, pH = 6).
Adsorption kinetics of atrazine on the soils
The critical concentration range of heavy metal cadmium poisoning in crop soil was 15.63–20.19 mg/kg (Zhao, 2008). Therefore, in this study, Cd(II) with a concentration of 20.00 mg/kg was added into the soil as the background value. The adsorption kinetics curves of atrazine in the three types of soil are shown in Figure 2(a). As can be seen from Figure 2(a), the capacity of atrazine adsorbed in albic, black, and saline-alkaline soils was 14.36, 12.76, and 7.85 mg/kg, respectively. The adsorption kinetics curves of atrazine in the three soils under Cd stress are shown in Figure 2(b). As can be seen from Figure 2(b), the adsorption quantity of atrazine increased with time, until the basic balance (1440 min). The maximum adsorption capacity of atrazine in albic soil, black soil, and saline-alkaline was 14.16, 11.96, and 7.30 mg/kg. The maximum adsorption capacity and equilibrium time of atrazine in soil were compared with the maximum adsorption capacity and equilibrium time of atrazine in soil under Cd(II) stress, as shown in Table 2. The order of adsorption was albic soil > black soil > saline-alkaline soil. The adsorption rate of atrazine was the highest within 60 min. The adsorption rate of atrazine in albic soil was 38.21% of the total adsorption rate, atrazine in black soil was 26.41%, and atrazine in saline-alkaline was 55.14%, respectively. With the increase of adsorption time, the adsorption rate decreases gradually until equilibrium. This may be due to the fact that with the increase of the contact time between the soil and atrazine, the concentration of atrazine gradually decreases, the adsorption sites in the soil decrease, and eventually the adsorption gradually becomes saturated. Compared with the single atrazine adsorption test, the adsorption capacity of atrazine in soil was reduced after adding Cd(II), which may be caused by Cd(II) occupying a part of the adsorption sites in the soils (Wei et al., 2018).

Adsorption kinetics curves of atrazine in the three types of soil (a) (conditions: 2 g of soils, volume of 10 ml Cd solution, shaking bath 298 K and 120 r/min, pH = 7.2); adsorption kinetics curves of atrazine in the three types of soil under heavy metal cadmium stress (b) (conditions: 2 g of soils, volume of 10 ml atrazine–Cd solution, shaking bath at 298 K and 120 r/min, pH = 6).
Maximum adsorption capacity and equilibrium time of atrazine in soils.

Elovich equation is widely used to describe intra-particle diffusion mechanism. The pseudo-second-order kinetics model can describe not only the internal diffusion mechanism but also the external diffusion mechanism (Witek-Krowiak et al., 2011). The adsorption parameter fitting of atrazine by Elovich equation and pseudo-second-order kinetic model is shown in Table 3. As can be seen from Table 3, both Elovich and pseudo-second-order kinetics model were suitable for describing the adsorption kinetics curves of atrazine in the three types of soil containing Cd, and the fitting effect of pseudo-second-order kinetics model was better. This suggests that the relationship between soils and atrazine was controlled by multiple mechanisms, including adsorption comprehensive control of diffusion, dissolution, mineralization, and other mechanisms (Huang and Zhang, 2015).
The adsorption constants of pseudo-second-order kinetics model and Elovich model.

Adsorption isotherms of atrazine on the soils under Cd stress
The adsorption isotherms of atrazine on albic soil, black soil, and saline-alkaline soil under the stress of Cd(II) are shown in Figure 3. As can be seen from Figure 3, with the increase of atrazine concentration, the adsorption degree of atrazine in soils increases gradually until it reaches the limit value corresponding to its maximum capacity. The content of soil organic matter of albic, black, and saline-alkaline soil was about 14.54, 30.10, and 6.8 g/kg and CEC was 22.63, 22.32, and 15.56 mol/kg, respectively. The pH value, TOC, and CEC can affect the adsorption of Cd(II) in soils, and low pH is conducive to the adsorption of Cd(II) in soils (Liang et al., 2017; Sharma et al., 2015). Saline-alkaline soil has the lowest organic matter and strong alkalinity. Black soil is neutral but rich in organic matter. Albic clay is acidic soil. The maximum adsorption capacity of atrazine was 32.72 mg/kg for albic soil, 29.28 mg/kg for black soil, and 20.45 mg/kg for saline-alkaline soil. No Cd(II) concentration was detected in the supernatant after centrifugation, indicating that the heavy metal Cd(II) in the mixture had been completely adsorbed by the three types of soil.

Adsorption isotherms of atrazine in the three types of soil under heavy metal Cd stress (conditions: 2 g of soils, volume of 10 ml atrazine–Cd solution, 24 h contact time at 298 K and 120 r/min, pH = 6).
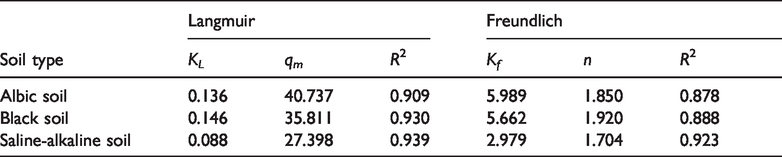
The adsorption parameter fitting of atrazine is shown in Table 4. As can be seen from Table 4, both Freundlich and Langmuir nonlinear adsorption isotherms can describe the adsorption curves of atrazine in three soils under the stress of heavy metal cadmium, but Langmuir fits the isotherm better (R2 > 0.909). It is concluded that the adsorption process includes single layer adsorption and chemical adsorption in addition to physical adsorption (Fiorentin et al., 2010). The KL in Table 4 is the intensity factor. It can be concluded that the larger the KL value is, the stronger the binding capacity of the sediment with heavy metal ions is (Dabbagh et al., 2015; Xiuhong et al., 2018). The qm in Table 4 is the maximum adsorption amount, which is close to the experimental results.
Fitting parameters of atrazine adsorption isotherms in the three types of soil.
Adsorption thermodynamics of atrazine on the soils under Cd stress
The fitting data of adsorption thermodynamics of atrazine in the three types of soil are shown in Table 5. It can be seen from Table 5 that the thermodynamic curves can be fitted by Langmuir and Freundlich models, and the Langmuir model had a good fitting effect. The parameter qm in the Langmuir model decreases with the increase of temperature, at 288 and 308 K, albic, black, and saline-alkaline soil of qm was reduced by 11.27, 11.19, and 40.97%, respectively.
Adsorption simulation equation parameters for different temperatures (conditions: 2 g of soils, volume of 10 ml atrazine–Cd solution, 24 h contact time at 288 and 308 K and 120 r/min, pH = 6).

The adsorption thermodynamic parameters of atrazine on the three types of soils are shown in Table 6. As shown in Table 6 adsorption thermodynamic parameters, the Gibbs free energy was less than 0 (ΔG < 0) and the enthalpy change was less than 0 (ΔH < 0). Therefore, the adsorption process of atrazine in soil is a spontaneous exothermic reaction. The spontaneity of the adsorption process proves that atrazine is easy to be absorbed by soil during application. The exothermic process of atrazine adsorption by the three kinds of soil was that atrazine changes from aqueous phase to solid phase and releases a lot of heat at the same time (Wei et al., 2018). In addition, the water solubility of atrazine in soils increases gradually with the increase of temperature; the higher the solubility, the stronger the interaction between atrazine and aqueous solution, and the more difficult it was to separate atrazine from aqueous solution (Yang et al., 2018). With the increase of temperature, the reaction moves toward endothermic direction and the adsorption capacity is weakened, which was consistent with the study of Yue et al. (2017). It was further proved that the adsorption of atrazine on the soil surface was more favorable at 288 K.
Adsorption thermodynamic parameters of atrazine in the three types of soil.

Effect of pH on atrazine adsorption on the soils under Cd stress
The change of soil atrazine adsorption in the presence of Cd(II) was studied by adjusting the pH value of atrazine background solution. As can be seen in Figure 4, the adsorption capacity of atrazine to soil decreases significantly with the increase of pH value. The results showed that atrazine was highly pH dependent. When the pH value is 3.0, atrazine has better adsorption capacity in the three types of soil. The adsorption capacities of atrazine in albic, black soil, and saline-alkaline soil were 14.52, 12.99, and 7.56 mg/kg, respectively. Atrazine is a weak alkaline pesticide with a protein kinase A (pKa) of 1.64. When the pH value of the soil is close to pKa, most of atrazine exists in the form of cations (Da Silva et al., 2019), the number of protons in the solution increases, and the protonation is enhanced. The adsorption capacity of atrazine is better when the ion exchange is the main adsorption mechanism. With the increase of pH value, atrazine gradually existed in the form of molecules, and the adsorption mechanism of atrazine was mainly carried out by molecular state and protonized carboxyl group. Sufficient exchangeable cations were required for protonation, and insufficient cations result in reduced adsorption rate. In addition, pH value of the soil solution increased, more polar groups in the soil was deprotonized, which can bind with metal ions on the inside and outside surfaces, thus enhancing the binding ability of soil to Cd(II). After Cd(II) occupied a certain adsorption site in the soil, the adsorption capacity of atrazine decreased gradually. In the experiment, the change of pH value had the largest effect on the adsorption capacity of atrazine in albic soil, while the effect on saline-alkaline soil was not obvious, which may be related to the pH of albic soil themselves.

Effects of different pH values on the adsorption of atrazine under heavy metal Cd stress (conditions: 2 g of soils, volume of 10 ml atrazine–Cd solution, 24 h contact time at 298 K and 120 r/min, pH values were 3.0, 5.0, 7.0, 9.0, and 11.0).
Effect of biochar on atrazine adsorption on the soils under Cd stress
The effect of adding different proportions of biochar on the atrazine adsorption in the three soils is shown in Figure 5. With the increase of the proportion of biochar, the adsorption capacity of atrazine by Cd(II) in the soil gradually increased. The maximum adsorption amounts of atrazine in albic soil, black soil, and saline-alkaline soil were 17.80, 15.93, and 9.60 mg/kg. The Cd2+ of 20 mg/l was completely absorbed by soils. The addition of biochar can increase the soil’s adsorption capacity for atrazine. The reason may be that biochar can increase the porosity and specific surface area of the soils, improve the cation exchange capacity of the soil, and the functional groups of biochar can form π bonds (Ge et al., 2014), improving soil cohesion. In addition, biochar can be combined with heavy metal Cd(II) to form insoluble complexes through various mechanisms such as electrostatic adsorption, ligand exchange and surface functional group complexation, so that Cd(II) is quickly and completely absorbed in solution (Zhao et al., 2018). Adding a certain proportion of biochar can inhibit the adsorption of atrazine by changing the pH value, but the experimental results show that biochar has a more significant promoting effect (Zhao et al., 2018)

Effects of different biochar contents on the adsorption of atrazine under heavy metal Cd stress (conditions: 2 g of soils, volume of 10 ml atrazine–Cd solution, 24 h contact time at 298 K and 120 r/min, pH = 6, biochar addition of 0.05, 0.25, 0.5, 0.75, and 1%).
Conclusions
In this study, the adsorption characteristics of atrazine in albic soil, black soil, and saline-alkaline soil under the stress of heavy metal Cd were studied. According to the parameters of experimental evaluation, we verified that the adsorption of atrazine in soil was inhibited under the stress of heavy metal Cd, and the adsorption amount was significantly reduced. In the adsorption kinetics test, the adsorption process reached equilibrium after 720 min of contact, and the fitting effect of the pseudo-secondary model was the best. Langmuir model has the best fitting effect on adsorption isothermal data, and the maximum adsorption amounts of atrazine in albic soil, black soil, and saline-alkaline soil were 32.72, 29.28, and 20.45 mg/kg, respectively. Thermodynamic parameters showed that the adsorption process is a spontaneous and disordered exothermic process. Under the stress of heavy metal cadmium, the adsorption amount of atrazine in soil decreased. It can be inferred that the interaction between atrazine and cadmium in the soil leads to the increase of fluidity of atrazine in the soil. Therefore, it is necessary to improve the adsorption capacity of atrazine in the soil by reducing the pH value of the soil, such as adding an appropriate amount of alkaline biochar to the soil, so as to provide a basis for the dynamics of atrazine residue in soil and further provide a scientific basis for the remediation of atrazine contaminated soil in northeast China.
Footnotes
Acknowledgments
The authors thank anonymous reviewers for their comments on this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Education department of Jilin province (JJKH20200337KJ), The National Natural Science Foundation (31672051), The National Key Basic Research Program (2016YFD0200203), and The National Key Projects (2017YFC0504200).
