Abstract
Dyes and pigments have been used in many industries for colorization purpose but they pose hazards to the environment and end users of water. Therefore, it is important to remove this pollutant from waste water before their final disposal. This study aimed to investigate the removal of methylene blue by cost effective, ecofriendly, high-efficiency bio-sorbent from activated coffee husk. The process was carried out using chemical activation (H3PO4) process. Fourier transform infrared spectroscopy and surface analyzer (Brunauer-Emmett-Teller) were used to characterize the adsorbent. The specific surface area adsorbent was obtained as 28.54 m2/g. The maximum removal efficiency was obtained as 96.9% at pH of 5, initial dye concentration of 20 mg/L, adsorbent dosage of 0.8 g/50 mL, for contact time of 50 min and 30°C temperature on the activation surface of coffee husk. Langmuir model was found to fit the equilibrium data for methylene blue adsorption with 6.82 mg/g at 30°C. The adsorption process follows the pseudo-second-order model. Thermodynamics analysis showed that the adsorption of methylene blue on to the activated coffee husk was a spontaneous and endothermic process. The experimental data obtained in the present study proved that coffee husk is a suitable bio-sorbent in removal of cationic dyes.
Introduction
There are often huge amounts of colored waste water produced in industries which use dyes to report a desired color to their products such as rubber, paper, plastics and textile are released into natural streams with adverse impacts to the environment and to human health. Apart from unpleasant esthetic aspects, the existence of dyes in natural streams can cause serious harm to the aquatic life by increasing toxicity and chemical oxygen demand, and by obstructing photosynthetic spectacles through declining of light dispersion (Bulut and Aydin, 2006). Correspondingly, some dyes, such as methylene blue (MB), can cause injuries to humans and animals by direct contact (eye burns), inhalation or ingestion (nausea, vomiting, mental confusion and others) (Hameed et al., 2007).
MB has been found to be the most suitable substrate in synthetic textile fiber industries where it is widely used. However, such extensive use often causes problems in the form of colored waste water, and adequate treatment is required for its removal before discharged into water bodies. MB has complex chemical structure; it is tough to fading to exposure light and water and is therefore very difficult to be removed from waste water by commonly used techniques.
Recently, many conventional methods have been used to treat textile effluents, such as membrane separation, ion exchange, electrochemical, precipitation, rivers osmosis, Fenton-reagent method, microbial degradation and fungal decolorization (Kyzas et al., 2013). Since the dye molecule is a very stable not degradable material, it could not be absolutely treated by a conventional method and will produce a secondary pollutant as by-product. However, dyes dissociate in aqueous solutions into MB cations and chloride ions. It is tending to be absorbed into solid, such as activated carbons.
Adsorption process using activated carbons has been extensively suggested and used for the removal of both organic and inorganic contaminants from aqueous solution effluents. However, commercially available activated carbons are expensive, and in recent years, a great deal of effort has been put into the proposal and usage of low-cost adsorbents prepared from naturally occurring materials and wastes for removal of dyes from waste water. Agricultural wastes are the principal raw materials being considered for this purpose because they are renewable, usually available in large volume and potentially less expensive than other materials to produce a variety of types of adsorbents. In recent years, adsorption has gained wide attention and popularity in textile waste water treatment (Kousha et al., 2012). Adsorption is a simple method, easy to handle, able to function at very low concentration treatment, safe and cost effective to remove pollutant in textile waste water compared to the previous methods (Velmurugan et al., 2011).
Coffee husk is one of the largest agro-industrial processing wastes in Ethiopia which consists of a lignin (18%), cellulose (45%) and hemi-cellulose (28%) (Oliveira et al., 2007). Processing of coffee generates significant amounts of agricultural wastes. Coffee husks (CH), comprised of dry outer skin, pulp and parchment, are the major residual from the handing and processing coffee, represent about 25.5% of coffee weight (Hayashi et al., 2000). The production of coffee in Ethiopia in the last five years ranged from 211,000 to 288,000 metric tons (Amamo, 2014). This represents an average of over 72,000 metric tons of coffee husk being produced every year.
To our best information, there is no profitable use for this type of reside and its disposal constituents a major environmental problem in Ethiopia. Even though combustion of coffee husks is a common practice in farms area, it has already been verified that major problems are prone to happen, such as fouling, agglomeration and excessive emissions, due to low melting point of the ash of burnet coffee husks and significant amount of volatile organic matter present in the husks (Saenger et al., 2001). Furthermore, since sustainability development should be prioritized, the development of techniques for giving additional value and reusing this type of residual should be required.
The present study aims to study the use of chemically treated coffee husk as low adsorbent for the removal of MB from aqueous solution through batch mode adsorption technique. The adsorption isotherm, adsorption kinetics and adsorption thermodynamics of the activated coffee husk have been also studied.
Methodology
Raw material and chemicals
Dry coffee husk was collected from local coffee agro processing unit, Bahir Dar, Ethiopia. The coffee husks were obtained from a dry processed coffee after de-hulling. MB stock solution was prepared with distilled water, and working solutions (20–100 mg/L) was prepared by diluting the stock solution prior to batch adsorption experiment. All laboratory-grade reagents H3PO4, NaOH and HCl were used without purification.
Bio-sorbent preparation
Coffee husk was washed with distilled water (approximately 250 mL of water per gram of coffee husk) to remove dirt and color, and dried at 105°C for 1 h in a convection oven. Dried coffee husk was grounded into granules, sieved to 200 µm particle sizes. Dried and sieved powder was impregnated with H3PO4 (50% v/v) for 24 h. After the reaction subsided, activated powder was carbonized at a temperature of 400–600°C with Muffle Furnace (Nabertherm B180) for an activation holding time of 30–90 min to develop porosity and high adsorption efficiency. Then, the activated powder was neutralized with excess of distilled water and was dried at 105°C, and kept in a plastic bag for the subsequent experiment and adsorbent characterization.
Preparation of dye solution
The physical state of MB dye is a powdered solid and the solution was prepared by adding 0.5 g of dye into 1 L of distilled water and well mixed. The maximum wavelength (540 nm) is obtained after scanning the dye solution using Ultraviolet-visiblespectrometry (UV/VIS) (Lamda 35 Ferkin Elmer).The known dye concentrations were prepared and their absorbency values were measured from UV/VIS spectrometer. A linear calibration curve was obtained from absorbency values versus with their respective concentrations of dye solution (20, 40, 60, 80 and 100 mg/L), as shown in Figure 1.

Calibration curve of MB solution at different concentrations.
Bio-sorption studies
Batch adsorption experiments of MB on to the activated coffee husk were conducted in a set of 250 mL conical flasks containing 100 mL of MB solution. The flasks were covered in an isothermal water bath shaker and agitated with a speed of 120 r/min at 300 K until equilibrium was reached. The batch adsorption experiment was investigated with different experiment parameters such as initial concentration of MB (20, 40, 60, 80 and 100 mg/L), solution pH (1, 4, 6, 7, 8 and 10), adsorbent dosage (0.4, 0.6, 0.8, 1.0 and 1.2 g) and contact time (20, 30, 40, 50 and 60 min). The initial pH of the solution was adjusted with 1 M HCl or 1 M NaOH before adding the adsorbent. At the end of each experiment, small amount of supernatant solution was withdrawn at predetermine time and filtered with Whatman filter paper grade 42. Then, 5 mL of the solution was taken from the Erlenmeyer flasks and the concentration of MB was determined with UV spectrophotometer at 540 nm. The amount of MB adsorbed was determined by taking the difference between the initial dye concentration and the concentration of the solution at the time of sampling. All determinations were performed in a total of three replicates per experiment and the average values were reported. Percentage of adsorption and adsorption capacity at equilibrium of MB ion were determined by equations (1) and (2), respectively (Bhattacharyya and Gupta, 2008). The sorption capacity qt (mg/g) at time t was obtained from equation (3) (Meroufel et al., 2013)
Analysis and characterization
The dye concentration has been measured by using UV/VIS spectrometer (Lamda 35 Ferkin Elmer) at a wavelength of 540 nm. The physicochemical characterization like moisture content (ASTM 2867-99), ash content (AC) (ASTM D1102-84), volatile matter (ISO 562) and fixed carbon (percentage carbon) were determined by ASTM D3172. The specific surface area and pore size of the adsorbent were analyzed by Brunauer-Emmett-Teller (BET) surface analyzer (NOVA 4000e). Fourier transform infrared (FT-IR) (JASCO FTIR 4100) spectra were used to record KBr pressed pellets powder of the absorbent in the range of 4000–400 cm−1.
Results and discussion
Characterization of bio-adsorbent
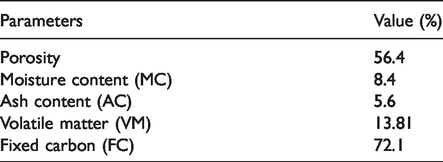
Proximate analysis was performed for coffee husk to analyze physiochemical characteristics as presented in Table 1. The moisture content of the coffee husk was found to be 8.4% which is slightly less than the value reported by Demirbas (2009). The AC showed the presence of the amount of inorganic constituent and obtained as 5.6%. The AC in the coffee husk may be due to the presence of residual inorganic compounds in the adsorbent, which was found to be 0.25% to 12.40% for agricultural products (Avelar et al., 2010). The volatile matter was obtained as 13.8% which is lower than 25% as previous report for coffee husk (Olakunle et al., 2017). The effluence of calcined temperature on moisture and AC of activated coffee husk is presented in Figure 2. As carbonization temperature increases from 400°C to 600°C, the moisture content was reduced from 10.2 to 7.2%, with the activation time increases from 30 to 90 min, the AC was dramatically raised from 1.25 to 8.2%. This is due to the fact that AC increases when biomass carbonized with high temperature for a long activation time. And the entire moisture content cannot evolve at low calcination temperature. The high value of fixed carbon indicates that there is high graphitization grade and low number of functional groups. Therefore, it can be deduced that the lower value of AC would have better for adsorptions of MB dye.
Proximate analysis of coffee husk.
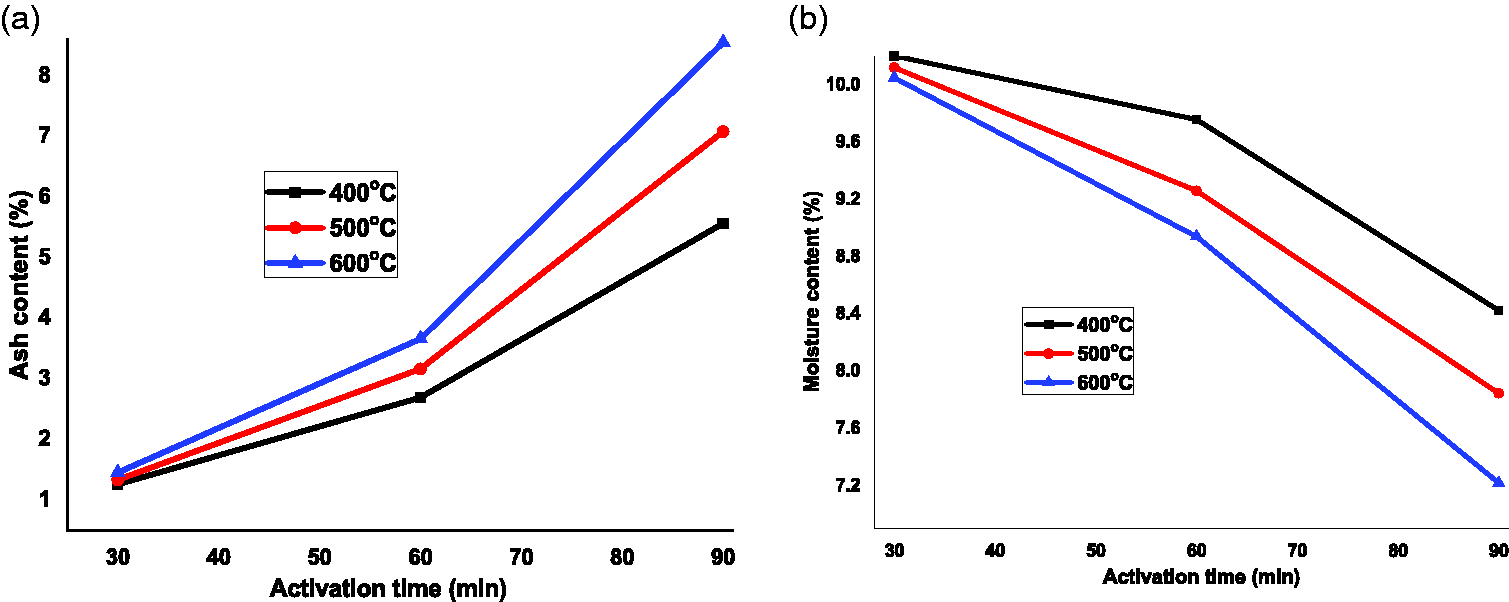
Effect of calcination temperatures on (a) ash and (b) moisture content determinations.
Surface area and porosity analysis
The textural property of the prepared coffee husk adsorbent was characterized with surface area and pore size analyzer and the tabular results are presented in Table 2. It has been observed that the surface area for the prepared adsorbent from coffee husk was higher as compared to the previous report by Oliveira et al. (2009) which is 1.7m2/g. which is 1.7 m2/g. In other study, the surface area of activated coffee husk was found to be 2.06 m2/g, which has been used to remove copper and lead from waste water (Shukla and Pai, 2005). Table 3 shows that the comparative value of proximate analysis with some of the low-cost adsorbent materials which were synthesized as activated carbon.
Textural properties of activated coffee husk adsorbent.

Proximate value of activated carbon prepared from different biomasses.

MC: moisture content; FC: fixed carbon.
The vibrational and bending spectral functional groups of activated coffee husk were studied before and after adsorption using FTIR spectrometer, as shown in Figure 3. The spectra showed peaks typical for hydroxide functional groups at the range of 3255–3504 cm−1 suggests that the presence of free hydroxyl groups from carboxylic acids, phenols, alcohols and other hydroxyl ion containing organics which is present with cellulose and lignin. This result is similar with previous study where the peak at 3421.57 cm−1 revealed that there are free hydroxide groups within the adsorbent surface (Li et al., 2007). The adsorption peaks at 2952, 1650, 1425, 1228 and 870 cm−1 assign due to the stretching bond of –CH2, C–C, C = C, C–O and C–N, respectively, while, after MB adsorption on to AC, other additional peaks were observed at 1640, 1450, 1100 and 650 cm−1 attributed to N = O, C–N, C = O and C–S, respectively. It can be seen that bending shape of adsorbent surface before adsorption is greater than the surface of adsorbent after adsorption. This is due to the presence and the interaction of MB molecule with functional group of the adsorbent. It can also be observed that the bending shape of the adsorbent before adsorption has high transmittance value than after adsorption. This attributes to the reason that all the active site adsorbates are occupied by the MB molecule after adsorption. The obtained results ensure that the surfaces of adsorbents were occupied by the dye molecules, and due to this, transmittance has been decreased after adsorption. This is because the number of ions is adsorbed high on the adsorbent surface, and the ability of light to transfer through the surface of adsorbent is low (Nasuha et al., 2010).

FT-IR spectra of activated coffee husk before and after adsorption.
Effect of adsorbent dosage
The effect of the adsorbent dose at different calcination temperatures (400, 500 and 600°C) on the percentage of MB removal was carried out in the range of 0.4–1.2 g at a fixed pH of 5, temperature of 30°C, contact time of 50 min and initial dye concentration of 20 mg/L, as shown in Figure 4(a). It has been shown that the percentage removal of MB increased from 69.68% to 94.10% as adsorbent dosage increases from 0.4 to 1.2 g. This attributes due to the increases of the available sorption surface of the adsorbent and the presence of free adsorption sites. Similar results have been reported in literature (Barka et al., 2012) for the removal of reactive dye from aqueous solution by adsorption on to hydroxyapatite adsorbent. However, the removal efficiency does not increase with the increase dosage due to the saturations of active site on the adsorbent surface in solution (Jiang et al., 2010). Therefore, in this study, the maximum removal adsorption occurred at 0.8 g adsorbent dosage for adsorbents calcined 600°C temperature.

Effect of (a) dosage and (b) contact time for the removal of methylene blue dye.
Effect of contact time
To explore the minimum time required for maximum performance of adsorption process at three calcination temperatures (400, 500, 600°C) with contact time varies from 20 to 100 min at a minimum dosage of 0.8 g/50 mL, pH of 5 and temperature of 30°C with initial dye concentration of 20 mg/L. As shown in Figure 4(b), it has been observed that the removal efficiency of MB increased with increasing contact time up to 50 min. But further increasing the contact time was not effective for maximum removal efficiency. The maximum removal efficiency was recorded as 92.021% at 600°C. From this result, it can be described that most vacant surface sites are available for adsorption during the initial stage and the remaining vacant surface sites are hard to be employed due to repulsive forces between the MB molecules on to the solid phase (Fu et al., 2015). The declined adsorption rate was observed due to the decreasing number of free sites on to the adsorbent (Abd El-Latif and Ibrahim, 2009) and the possible formation of MB monolayer on the adsorbent surface (Chowdhury and Saha, 2012; Liang et al., 2010).
Effect of solution pH
The effect of solution pH on adsorption was studied at pH of 1, 4, 5, 6, 7, 8 and 10 with activated coffee husk at 30°C temperature and 20 mg/L MB concentration as shown in Figure 5(a). It has been observed that when the initial pH solution was increased from 1 to 5, the removal performance increased from 87% to 97% and reached maximum level at pH of 5. Then, the efficiency started to decline from 96 to 89% with pH increases from 5 to 10. Thus, it can be concluded that the percentage removal of MB is lower in basic media and high in acidic media. This is due to less concentration of H+ ion competing with the MB dye cations for adsorption sites. The better adsorption at low pH may be due to the presence of large number of hydrogen ions in the liquid–solid phase. In the basic medium, the positive charged surface sorbent attributed to oppose the adsorption of the cationic adsorbate. When the pH of MB solution is slowly increased, the surface acquires a negative charge, thereby resulting in an increased adsorption of MB, which is due to an increase in the electrostatic attraction between positively charged dye and negatively charged adsorbent (Malik, 2003). Also, the higher adsorption of MB at low pH values may be explained by the competition of excess H+ ions with the dye for cation for active adsorption (Pehlivan et al., 2006).

Effect of (a) pH of solution and (b) initial MB concentration for the removal of methylene blue dye.
Effect of initial dye concentration
The effect of initial dye concentration at 20 mg/L, 40 mg/L, 60 mg/L, 80 mg/L and 100 mg/L adsorption on activated coffee husk adsorbents was evaluated. As it can be seen from Figure 5(b), the percentage removal of MB molecules removed decreases with an increase in initial dye concentration. This may be due to the saturation of available active sites on the adsorbent and the formation of condensation polymerization formation in the adsorption process. At a temperature of 30°C, pH of 5, 0.8 g adsorbent dosage and 50 min contact time, the percentage removal of MB dye is decreased from 95.01% to 82.73%. At low MB concentration, there will be unoccupied active sites on the adsorbent surface, and when the initial dye concentration increased, most of the vacant active surface of the adsorbent is going to be occupied (Dula et al., 2014). Similarly, another study reported using untreated coffee husk for the removal of MB recorded as 82% of removal for 100 mg/L dye concentration, pH of 8.0, and contact time of 3 h at 30°C temperature (Oliveira et al., 2008).
Adsorption isotherm studies
Langmuir and Freundlich isotherms were used to analyze equilibrium data of solute between adsorbent and solution (Oguntimein, 2015). The parameters obtained from these models provide important information on adsorption mechanism and surface properties of the adsorbent. The Langmuir parameters have been determined by transforming the Langmuir equation (4) into the linear form as given by equation (5)


The values of Qm and Klhave been computed from the intercept and slope of the Langmuir plot of 
Type of separation factor, R of Langmuir isotherm.


Adsorption isotherm for the removal of MB on coffee husk adsorbent: (a) Langmuir and (b) Freundlich.
Kl is the Langmuir constant (L/mg) related to the affinity of binding sites and the free energy of sorption and Co is the highest initial MB concentration (mg/L).The calculated RL value for coffee husk adsorbent was 0.109 which exists in the range of 0, an indicating favorability of adsorption. From Figure 7, it is shown that the RL value gradually decreases as the concentrations of MB increased, showing that the higher initial concentrations of MB may improve the adsorption process. The applicability of Freundlich isotherm adsorption has been analyzed with the same set of experimental data and presented in Figure 6(b). The Freundlich parameters have been determined by transforming the Freundlich equation (7) into linear form, as mentioned equation (8)



Plot of separation factor versus dye concentration.
Kf and n are the Freundlich adsorption isotherm constants, which indicate the capacity and intensity of the adsorption, respectively (Demiral et al., 2008). The Freundlich constants were determined from the slope and intercept of a plot of
Langmuir and Freundlich isotherm parameters.

Adsorption kinetics studies
The kinetics of MB adsorption on coffee husk adsorbent was analyzed by using pseudo-first-order and pseudo-second-order kinetics models (Khaled et al., 2009). For the conformity between experimental data and model, predicted values were expressed in correlation coefficient. The relative higher value is the more applicable model to the kinetics of MB adsorption on to adsorbent.
The pseudo-first-order rate has been expressed by equation (9), and applying boundary condition t = 0 to t = t and qt = 0 to qt = qt, it becomes equation (10)




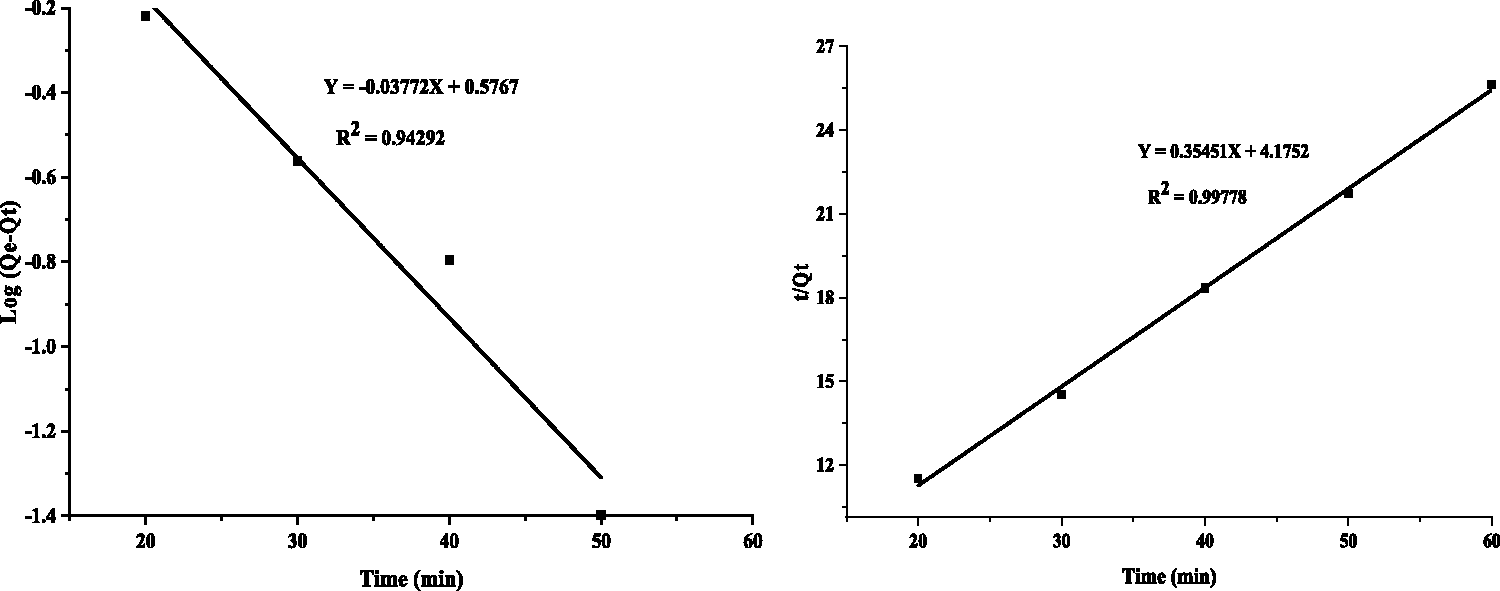
Adsorption kinetics of MB: (a) pseudo-first-order and (b) pseudo-second-order rates.
The value of k2 (g/mg min) and qe was calculated from slope and intercept of the graph
Pseudo-first-order and pseudo-second-order adsorption of MB over activated coffee husk.
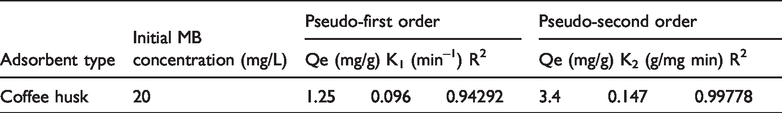
The correlated coefficient, R2 of the pseudo-second-order kinetic model is greater than that of the pseudo-first-order kinetics models. Thus, batch adsorption experiment for the removal of MB by using activated coffee husk adsorbent follows the pseudo-second-order kinetics model. The kinetics and mechanism of MB adsorption on commercially activated carbon and indigenously prepared activated carbons from coconut shell, bamboo dust, rice husk, straw and ground nut have been studied. The kinetics of adsorption was found to be first order with regard to intra-particles diffusion rate (Kannan and Sundaram, 2001).
Thermodynamics adsorption
The thermodynamics parameters of adsorption of MB on to coffee husk were derived from the experimental data obtained at 298.15, 303.15 and 308.15 K to deduce the nature and thermodynamics feasibility of the adsorption process. The standard free energy change (ΔGo), Enthalpy change (ΔHo) and Entropy change (ΔSo) were determined using equations (13) to (15) (Zenasni et al., 2013)



Qe is the MB concentration adsorbed on to the coffee husk (mg/L), R is the universal gas constant (8.314 J/mole) and Ce is the MB concentration at equilibrium in solution (mg/L). The values of ΔH and ΔS were determined from the slope and intercept of the plot of lnKd versus 1/T (Demiral et al., 2008), as shown in Figure 9. Gibb’s free energy value comes out to be positive which suggests that it is a non-spontaneous reaction. The small value of Gibb’s free energy shows higher adsorption potential. Positive value of ΔHo suggests that the process is endothermic in nature (Meroufe et al., 2013). The results in Table 7 have been shown at low temperature, the values of ΔG° was small, at this condition high amount of MB was adsorbed over activated coffee husk.

Plot of lnKd against 1/T for adsorption of MB.
Thermodynamics parameters.

The adsorption capacity of AC from coffee husk for the removal of MB was compared with some reference adsorbate particles as reported from literature. The values of adsorption capacity are present in Table 8. The experimental data of the present investigation had high adsorption capacity as compared with reference adsorbent.
Comparison of adsorption capacity of activated coffee husk with various adsorbents.

Conclusion
The coffee huskis exhibited a great potential as low-cost adsorbent for effective removal of MB from aqueous solution. Physiochemical characterization revealed that the hydroxyl and carboxylic group played an important role in the adsorption of MB. The optimum coffee husk dosage was 0.8 g and higher MB adsorption capacity was obtained in solution of pH 5 and 50 min contact time. The adsorption equilibrium data obeyed the Langmuir isotherm model; meanwhile, the pseudo-second-order model was determined to better fit for kinetics data as compared to pseudo-first-order model. The thermodynamic parameter values showed that the MB adsorption by activated coffee husk was a spontaneous and endothermic process. At last, it can be concluded that the activated coffee husk adsorbent is used as a low-cost, effective alternative adsorbent for the removal of MB from textile waste water. It is recommended that this economical bio-adsorbent can be applied in industries to optimize the treatment costs of their waste water treatment plant.
Footnotes
Acknowledgments
The authors would like to thank the Faculty of Chemical and Food Engineering, Bahir Dar Institute of Technology, for their support and access to all necessary materials for the successful completion of the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



