Abstract
Tectonic deformation can cause significant changes in the physical and chemical structures of coal by damaging the macrostructure and macromolecular composition. For thorough research on the coal tectonic deformation impacts on gas adsorption capacity, this paper collected and summarized the parameters of experimental adsorption isotherms and coal macerals, conducted proximate and ultimate analyses, and systematically discussed the adsorption properties of different structures of coals and the influence of temperature and pressure on coal adsorption. Furthermore, the semi-quantitative relationships between the structural parameters of coal and its methane adsorption capacity are explored. The results show that (1) due to different tectonic stresses, the molecular and porous structures of different types of tectonic coal exhibit significant differences (e.g. sample N25, which is in a fault zone, has the highest methane adsorption capacity), and (2) coal methane adsorption capacity decreases along with increasing temperature. At a pressure of 12 MPa, primary coal (N32) showed Langmuir volume (VL) values of 15.38, 9.58, and 7.86 cm3/g and Langmuir pressure (PL) values of 3.82, 2.07, and 1.81 MPa at temperatures of 30°C, 50°C, and 70°C, respectively. (3) The Langmuir volume appears to have a linear relationship with parameters ID1/IG, Al/OX, and A-factor, as well as a parabolic curve relationship with
Introduction
Tectonically deformed coals, which is an important coal type in Chinese coal mining, affect the safety of coal mine exploration and coalbed methane production. The properties of higher porosity and lower permeability make this type of coal capable of keeping higher gas pressure. Tectonization is an important factor in the formation of coal seams (Zhai et al., 2018). Coal methane adsorption capacity is influenced by pore structure (Hou et al., 2017), temperature (Pan et al., 2012), pressure, coal composition (Crosdale, 1998; Mohanty et al., 2018), chemical properties (Shan et al., 2018), etc. Meanwhile, ash, fixed carbon, moisture, vitrinite content, vitrinite reflectance, and temperature have also been previously selected as independent variables to predict VL (Langmuir volume) (Feng et al., 2014). Sorption capacity shows a weak positive correlation with coal rank and a negative correlation with temperature (Weniger et al., 2012). Liu et al. (2018) applied Raman spectroscopy in coals ranging from bituminous C to anthracite, analyzed the effect of coal macromolecular structure on methane adsorption, and concluded coal macromolecular structure had prominent effects on methane adsorption, and a U-shape relationship was observed between VL and Raman structural parameters (G-D1 and AD1/AG).
The methane and carbon dioxide sorption capacity of tectonically deformed coals (TDCs) and primary structure coals is different, as the composition of TDCs will have been physically and chemically deformed under the movement of tectonic stress in previous geological periods (Wang et al., 2017). Previous studies showed that strong-brittle deformed coal has a higher adsorption capacity than weak-brittle deformed coal (Pan et al., 2012). The effect of structure characteristics of deformed coal on methane adsorption was investigated. The results showed that the main factor affecting methane adsorption capacity at the brittle deformation stage is the macromolecular structure of coal, while at the ductile deformation stage, both macromolecular structure and pore structure affect methane adsorption capacity (Guo et al., 2017). There are numerous adsorption models, such as the Langmuir model, the D-R model, and the dual sorption model, which are used for fitting methane and subcritical CO2 sorption data. The model with the best correlation coefficient (R2) is used to represent each group of adsorption or desorption data (Wang et al., 2014). To date, some experimental attempts have been made to investigate the relation between structural defects and deformation (Hou et al., 2012; Jiang et al., 2010; Ju et al., 2004; Pan et al., 2015). The role of structural defects and their influence on the macromolecule are further discussed using quantum chemistry calculations (Han et al., 2017). Tectonic deformation damages the macromolecular structures of coal. Evidently, high shearing stress leads to strong ductile deformation, while brittle deformation is related to the breaking of a series of bonds as a result of tensile stress and direct bond breakage (Li et al., 2017). Although much effort has been made to understand kerogen structure, it is essential to validate these structures by comparing simulated adsorption isotherms before analyzing gas adsorption mechanisms.
Previously, the gas adsorption behaviors and methane adsorption capacity of different structures of coals were mainly investigated separately for the molecular simulation studies in the literature. Although many papers have been devoted to understanding the effect of deformation on gas adsorption capacity (Meng et al., 2016), to the best of our knowledge, the microscopic adsorption mechanisms of CH4 on TDCs with different chemical structures have not been investigated, and our work aims to gain insight into this topic. A series of samples from one location that provided a range from structurally unaffected samples to highly deformed samples were collected. The ductile deformation mechanisms of coal were investigated in TDC samples using structural characterization methods and by examining the evolution of the molecular structure. The adsorption behaviors of CH4 were investigated by isothermal adsorption experiments. This study will give insight into the mechanisms of tectonic deformation on coal adsorption.
Samples and experiments
Samples
To avoid influence of other factors that may affect coal deformation and analyze the methane adsorption of different structures of coals, several coal samples (N20, N24-2, N25, N26-1, N27, and N32) were collected from the face of the No. 6 coalbed in the Qinan Coal Mine in the Huaibei Coalfield of China, following Chinese Standard Method GB/T 482-2008. The sampling position is located in the No. 6125 coal face, where there is a thrust fault. Sample N25 was collected from the hanging wall of the fault, while samples N24-2, N26-1, and N27 were collected from the footwall (Figure 1). More details on samples are given elsewhere (Li et al., 2017). We carried out a proximate analysis for these samples using China’s national standards for proximate analysis of coal (GB/T 212–2008). The results from the proximate analysis and elemental analysis of the coal samples are listed in Table 1.

The distribution of sample collection locations.
Key measured parameters of coal samples.
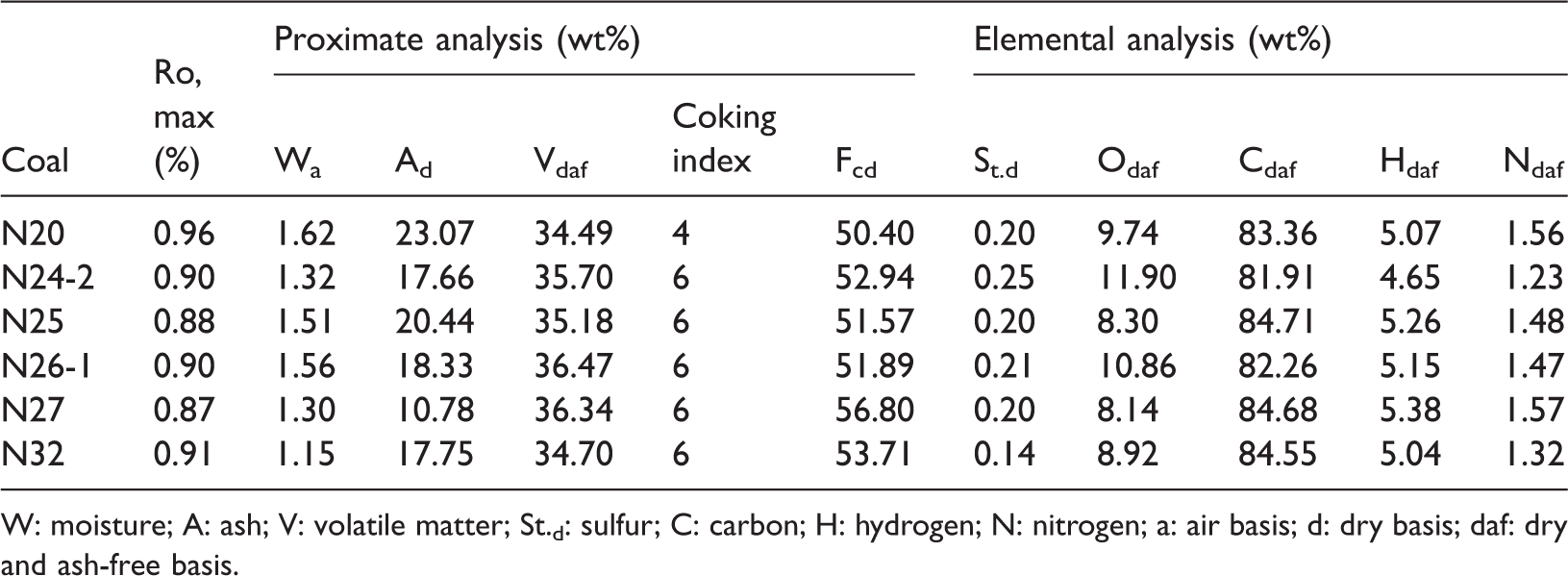
W: moisture; A: ash; V: volatile matter; St.d: sulfur; C: carbon; H: hydrogen; N: nitrogen; a: air basis; d: dry basis; daf: dry and ash-free basis.
Isothermal adsorption experiment
Four blocks of coal samples were used, all maintained at equilibrium moisture. Isothermal adsorption experiments were performed at a maximum pressure of 12 MPa and a temperature of 30°C. Meanwhile, isothermal adsorption experiments were also conducted on sample N32 at temperatures of 50°C and 70°C, at a pressure of 12 MPa. The purity of adsorbent methane was 99.99%. The high-pressure adsorption testing system was a HPVA200. In this study, the testing procedure was performed in accordance with the Chinese Petroleum and Natural Gas Standard of the “Determination of the methane isothermal adsorption quantity in coal - Capacity method of dry basis” SY/T 6132–2013.
Results and discussion
Ductile and brittle deformation characterization
The organic fraction of coal is mainly composed of the maceral groups, vitrinite, inertinite, and liptinite. The main maceral in sample N20 is desmocollinite. The types of inertinite are predominately semifusinite and inertodetrinite. Liptinite is dominitely sporinite. There are also some microcracks in sample N20 (Figure 2(a)). The macerals of sample N24-2 are predominantly telocollinite, telinite, and semifusinite. Vitrinite exhibits microfolding and bending characteristics under deformation, which indicates that the sample is affected by ductile deformation (Figure 2(b)). The macerals in sample N25 are dominantly desmocollinite, semifusinite, and barkinite with few liptinite (Figure 2(c)). There are some microcracks in sample N26-1, which occurs in desmocollinite and semifusinite (Figure 2(d)). The macerals in sample N27 are mostly desmocollinite and inertodetrinite. These are interlaced with clay minerals, which demonstrate bending shapes (Figure 2(e)). The macerals in sample N32 are desmocollinite and semifusinite (Figure 2(f)). Macroscopic characteristics of the coal samples are shown in Figure 3.

Macerals feature derived from TDCs.

Hand pictures derived from TDCs.
Adsorption properties of different structures of coals
Figure 4 shows the changes of the adsorption capacities of the TDCs at 30°C. The methane isothermal adsorption curves have similar trends, in which the methane adsorption capacity increases with increasing pressure. The methane adsorption capacity increases rapidly at low pressures and then more gradually as it reaches saturation. At a constant temperature and low pressure, methane adsorption showed an almost-linear relationship with increasing pressure. Methane adsorption then reached a saturation point, and after this point, no further increases were observed with further increases in pressure in most of the samples. However, in samples N20, N24, and N27, methane adsorption decreased slowly after the saturation point. The Langmuir volume value of samples N20, N24, N25, N26, N27, and N32 are 12.54, 16.02, 18.86, 13.56, 15.69, and 15.68 cm3/g at 30°C, respectively. Due to different tectonic stresses, the molecular and porous structures of different types of tectonic coal exhibit significant differences. Sample N25, which is in a fault zone, has the highest methane adsorption capacity.

Adsorption isotherms of different types tectonically deformed coal at 30°C.
Influences of temperature and pressure on coal adsorption
These curves are similar under different temperatures. Figure 5(a) shows that the adsorption capacity of coal is closely related to the temperature. The adsorption isotherms indicate that the temperature has a significant influence on coal methane adsorption capacity, which has been previously demonstrated (Pan et al., 2012). The methane adsorption volume increases with increasing pressure for all coal samples. At a certain pressure, the methane adsorption volume decreases as the temperature increases, which follows the ideal gas law. This outcome indicates methane adsorption capacity decreases along with increasing temperature. At a pressure of 12 MPa, primary coal (N32) showed Langmuir volume (VL) values of 15.38, 9.58, and 7.86 cm3/g and Langmuir pressure (PL) values of 3.82, 2.07, and 1.81 MPa at temperatures of 30°C, 50°C, and 70°C. At high pressure, the rate of increase of adsorption volume decreases rapidly. The discrepancy of adsorption curves among different temperatures is mainly triggered by temperature. As temperature decreases, the rate of change of adsorption capacity increases.

(a) Adsorption isotherms of N32 (undeformed coal) at 30°C, 50°C, 70°C and (b) the relationship between temperature (T) and adsorbed gas content (V).
The gas content at each pressure has been plotted as a function of temperature (Figure 5(b)). The curves are flat above 30°C. There is a linear relationship between temperature and adsorbed gas content at a given pressure (i.e. 0.49, 2.03, and 4.35 MPa) above approximately 30°C. At the lowest temperature, the adsorbed gas content is slightly elevated for all pressures. With the same pressure, gas content at 30°C is much greater than at 50°C and 70°C. This result indicates that temperature obviously influences the gas content at high pressure (Crosdale et al., 2008).
Influence of structural deformation on coal adsorption
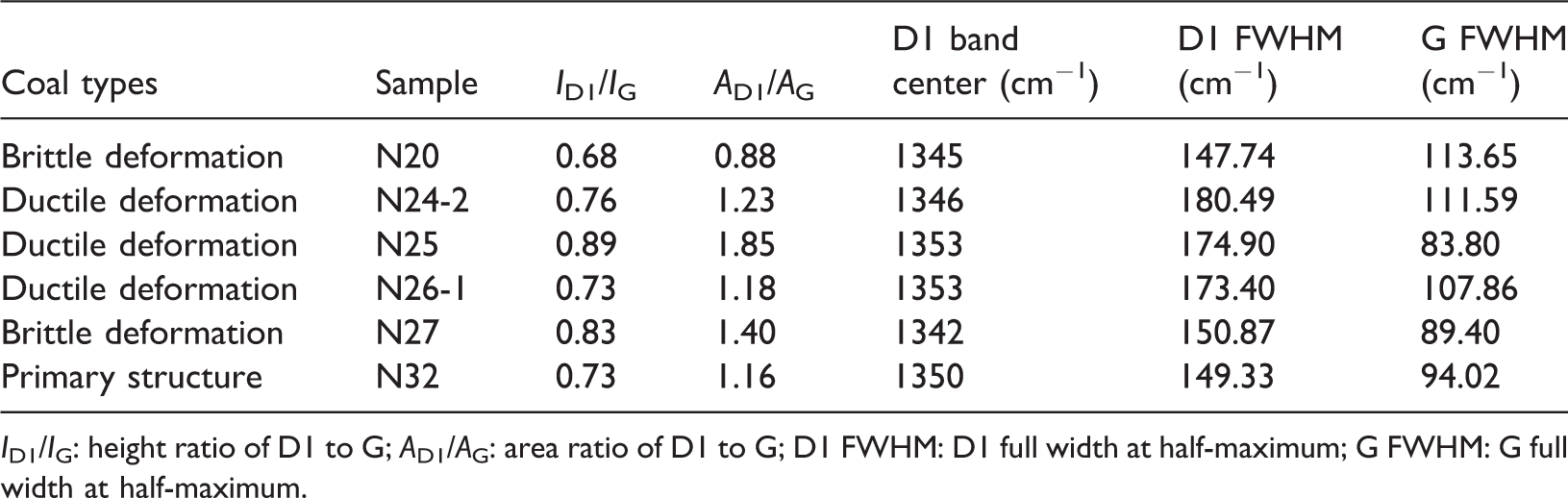
The Raman spectra of coal are commonly divided into first-order and second-order regions. Generally, there are two peaks, a defect peak (D peak) and a graphite peak (G peak) (Pan et al., 2017). G peak can be attributed to the in-plane vibration of carbon atoms in graphene sheets (Li et al., 2019). D1 FWHM means crystallite sizes from D1 full width at half-maximum (Seong and Boehman, 2013). The D1 FWHM parameter of the ductile deformation coal (N24-2, N25, N26-1) is greater than that of the brittle deformation coal (N20, N27), as a result of its high rate of defects in the graphitic structures, even though the differences relative to the G FWHM are not statistically significant. The N24-2 sample has the highest D1 FWHM value and a higher G FWHM value. An increase in ID/IG indicates the growth of basic graphene structural units (BSUs). The correlation between the Raman structural parameter ID1/IG and the Langmuir volume (VL) is evident (Figure 6(a)). This relationship indicates that graphitized coal and its defects can greatly enhance the methane adsorption capacity, as the intensity ratio of the D and G bands (ID/IG) can be used to indicate the crystallite dimension (Seong and Boehman, 2013) (Table 2).

(a) Structural parameter ID1/IG with Langmuir volume in different types of TDCs and (b) structural parameter
Parameters obtained from Raman spectra of the coals.
The apparent aromaticity (

Vitrinite reflectance with Langmuir volume in different types of coals (Yao et al., 2011).
As shown in Figure 8, the ratio of CHal/(CHal+C = C) (i.e. the A-factor) (Chen et al., 2012), which reflects the hydrocarbon-generating potential, increases along with the Langmuir volume (VL). This factor displays a strong linear relationship (

Structural parameters Al/OX and A-factor with Langmuir volume in different types of TDCs.
Conclusions
Due to different tectonic stresses, the molecular and porous structures of different types of tectonic coal exhibit significant differences. Sample N25, which is in a fault zone, has the highest methane adsorption capacity. At a pressure of 12 MPa, primary coal (N32) showed Langmuir volume (VL) values of 15.38, 9.58, and 7.86 cm3/g and Langmuir pressure (PL) values of 3.82, 2.07, and 1.81 MPa at 30°C, 50°C, and 70°C, respectively. The Langmuir volume appears to have a linear relationship with parameters ID1/IG, Al/OX, and A-factor, as well as a parabolic curve relationship with
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) gratefully acknowledge the financial support of the National Natural Science Foundation of China (Grant No. 41702167 and No. 41972169), the Natural Science Foundation of Jiangsu Province (Grant No. BK20181362 and No. BK20160243) and a project funded by the priority academic program development of Jiangsu Higher Education Institutions.
