Abstract
Intermolecular forces that act between moisture and the atoms of the coal structure have a significant influence on methane adsorption- and desorption-induced deformation in coal. After analyzing the porous characteristics and existing forms of moisture in coal, both the adsorption-induced swelling and the desorption-induced shrinkage deformation experiments were carried out under the conditions of varying moisture content, constant temperature, and variable equilibrium pressure. Both the swelling and shrinkage volumetric strains with different coal moisture contents were fitted by Langmuir-type equations in which the fitting coefficients were functions of the moisture content. It was found that there is a lag between the swelling curve and the corresponding shrinkage curve, and a variable known as the hysteresis rate was defined to illustrate this characteristic. A mathematical model of swelling and shrinkage deformation that considers the effect of moisture content was established based on the experimental results and analysis.
Introduction
The swelling and shrinkage (volumetric change) of coal during methane adsorption and desorption is a well-known phenomenon (Pan and Connell, 2007). Experiments using either strain gauges or optical methods have shown that coal swells with the adsorption of carbon dioxide (CO2), methane (CH4), and other gases (Pan and Connell, 2007), conversely, coal shrinks during the desorption of these gases.
Many researchers have studied coal swelling and shrinkage. For example, Lin and Zhou (1986) conducted a series of experiments using coal samples of different ranks, strengths, and porosities. They found that the deformation path follows the Langmuir equation when coal swells after gas adsorption, while the coal shrink after gas desorption follows the exponential equation. The amount of deformation primarily depends on the metamorphism, strength, and porosity of the coal. Reucroft and Patel (1986) completed dilatometry studies on three coal samples in various gaseous environments. The carbon contents of the three samples were 65.8, 78.3, and 83.8% dry free ash. They found that the sample with the lowest carbon content demonstrated the maximum length increase (0.5%) during exposure to a CO2 atmosphere. Levine (1996) summarized a series of measurements of adsorption-induced coal swelling. The results showed that the swelling behavior followed the same form as the adsorption isotherm, which, in this case, is explained using a Langmuir-like equation. In the work of George and Barakat (2001), volumetric strains of New Zealand coal samples were measured for CO2, CH4, N2, and helium adsorption. Although the magnitude of swelling varied for different coal types, the swelling isotherms showed similar trends with a maximum value of approximately 15.0 MPa. Chikatamarla et al. (2004) measured gas-induced swelling by H2S, CO2, CH4, and N2 on four coal samples with pressures up to 5.0 MPa. Their measurements showed that the larger the adsorption capacity, the greater the deformation. The relationship of volumetric strain and pressure could be described using a Langmuir-like equation, and the volumetric strain was approximately linearly proportional to the amount of adsorbed gas. Karacan (2007) used scanning electron microscopy (SEM), photographic X-ray, and computerized tomography (CT) images of a polished coal sample section to identify the microlithotypes and microstructures. They found that the CO2 sorption-associated swelling and volumetric strains in consolidated coal under constant effective stress were heterogeneous processes that depend on the lithotypes. Durucan et al. (2009) performed matrix deformation experiments on a range of different rank coal subjected to carbon dioxide and methane sorption at typical in situ reservoir pressures under unconfined conditions. It was found that matrix swelling due to CO2 adsorption displayed a positive correlation with coal rank, resulting in a significant decrease in permeability. Liang et al. (2013) studied coal deformation under low pressure after adsorption and desorption. They discovered that the deformation curve of coal samples at the same gas pressure includes three stages, which were a rapid growth stage, a slow growth stage, and a balance stage. The adsorption inflation deformation of coal appears as anisotropy, and strain in the vertical and parallel directions of the bedding is consistent. Cao et al. (2013) and Zhang et al. (2014) performed experiments on intact coal and briquette coal under different gas pressures and found that the swelling and shrinkage deformation during adsorption–desorption follow the Langmuir equation for the two kinds of samples. The relationship between the adsorption strain and equilibrium gas pressure can be described by the Langmuir equation for the two kinds of samples, while the relationship between the desorption strain and the initial gas pressure follows the power function. Liu et al. (2016) studied the anisotropy characteristics of coal shrinkage/swelling and its impact on coal permeability evolution with CO2 injection. It was found that application of isotropic deformation in permeability model can overestimate the permeability loss compared to anisotropic deformation. Zhou et al. (2017) observed meso-structural deformations of coal during three cycles of methane adsorption–desorption using a method combining SEM-EDS and micro-CT scan. They indicated that irreversible damage in coal occurred and accumulated during methane adsorption–desorption cycles, with the deformation capacity of coal decreasing with an increasing number of cycles. Nie et al. (2018) proposed CT and a coal particle tracking method to quantitatively calculate the strain of coal particulates in view of the pixel gray-level matrix of CT image. The results show that the spatiotemporal distribution of coal mesoscopic deformation is inhomogeneous during the process of methane adsorption.
The previously discussed studies significantly advanced the scientific understanding of the deformation mechanism of coal during gas adsorption and desorption. However, these studies mainly focused on the swelling behavior aspects, and few studies have reported on the shrinkage behavior. It is known that degassing coal seams can lead to volumetric shrinkage. Moreover, it has been shown that the presence of moisture can directly affect the gas diffusion properties of in situ coal seams (Busch et al., 2003; Chen et al., 2018; Guo et al., 2015; Hossein and Faruc, 2007; Li and Nie, 2006; Pan et al., 2010; Wang et al., 2017; Xie and Chen, 2007; Zhang and Ma, 2008; Zhang et al., 2012; Zhang and Sang, 2008; Zhao et al., 2011), and the effect of moisture on reduction of adsorbed volume is more prominent in CH4 compared to that of CO2. To date, many laboratory studies of gas-induced swelling in coal have used dry material or samples in which the moisture content was not closely controlled. Therefore, it is necessary to understand the swelling and shrinkage characteristics of coal under moisture-saturated conditions. Recognizing this, some recent studies have considered the effect of moisture on gas-induced swelling behavior.
Recently, Van et al. (2009) reported on the swelling measurements of two European coals in CO2, CH4, and Ar at pressures of up to approximately 8 MPa. Experiments were completed on both air-dried materials and samples that were moistened by exposure to a 97% relative humidity atmosphere. The results suggested that moisture tends to compete for adsorption sites on the coal, implying that gas-induced swelling is suppressed in the presence of moisture. Kiyama et al. (2011) also found that the CO2-induced volumetric swelling in constrained samples of Japanese bituminous coal was somewhat less in the presence of moisture. reported on the gas-induced swelling behavior of four moist Australian coals and found that the presence of moisture significantly reduced the amount of additional swelling by the gas compared to that in dry coals. However, the degree to which the swelling of the coals was affected by moisture depended on the rank of the coal. It was also found that, proportionally, CH4-induced swelling was affected by the presence of moisture more seriously than CO2-induced swelling. Although moist coal swelled less in CO2 or CH4 than in dry coals, if the swelling due to moisture is included, the total swelling is more than that induced by the corresponding gas in the dry coal. These results are consistent with the results of gas adsorption measurements on moist coal that generally show a substantially reduced adsorptive deformation capacity in the presence of moisture. However, insufficient data are currently available to make quantitative predictions.
To bridge the gap discussed above, this research examines the swelling and shrinkage behavior of the tectonically deformed coal with different moisture contents at a constant temperature.
The physical structure of coal and the occurrence of moisture
The flow and transport processes of methane in coal seams are quite complex, due to the extremely intricate and heterogeneous of coalbed. Coal exhibits multiscale heterogeneity that is often characterized by a distinctive matrix structure involving macropores and micropores (Gan et al., 1972; Unsworth et al., 1982), which can be assumed to follow a bimodal pore size distribution. Coal can be viewed as the solid, organic microporous material containing micropores, interconnected macropores, and uniformly distributed fractures, i.e. cleats. Pores exist in the coal matrix, while cracks consist of a natural fracture network surrounding the coal matrix. Most methane adsorption occurs within the microporous matrix, which has a large internal surface area (Yi et al., 2009). As shown in Figure 1, histological examination indicated that micropore distribution and connectivity in coal are not uniform, but interconnected open micropores and disconnected closed micropores are also present (Hossein and Faruc, 2007). The spatial range of the open micropores is called effective pore space where gases and liquid can flow in and out, while the closed micropores are called ineffective pore space where gases and liquid cannot be transported. The effective pore space is the main determinant of the adsorption capacities, and regular methods can only measure the effective pore space.

An indication of pore structure. A—coal matrix, B—crack, C—open micropore, and D—closed micropore.
There are two kinds of moisture in coal according to the existing state, which are free moisture and combined moisture (Xie, 2002; Zhang and Zhang, 2012). Free moisture consists of the outer and inherent moisture. The outer moisture is adhered to the external surface of the coal particles and large pores of diameters greater than 10−7 m. This part of moisture is mechanically combining with coal matrix and relating to the external conditions and the particle size, yet they do not have direct relationship with coal quality. The inherent moisture adsorbed or condensed in the capillaries or voids that distribute on the surface of coal particles and diameters less than 10−7m. This part of moisture combines with coal physically and chemically and relates to the coal species. The inherent moisture cannot be lost at room temperature. The greater the internal surface area of coal is and the more small capillaries are present, the higher is the inherent moisture of the coal. Generally, inherent moisture is in a gaseous state under the inner vapor pressure in the coal and is typically the main cause for gas adsorption. The percentage of chemically combined moisture with minerals is very low, and this kind of moisture can be lost only at higher temperatures. Therefore, it is unnecessary to consider combined moisture in the coal industry analysis, and the effect of combined moisture on coal adsorption/desorption is negligible.
In the actual formation conditions, a coal reservoir is least saturated by gas and moisture and varying degrees of liquid moisture. In other words, outer and inherent moisture occur concurrently in coal. Therefore, in this paper, it is more realistic to consider the effects of free moisture (includes the outer and inherent moisture) on the deformation of coal during methane adsorption/desorption.
Sorption-induced deformation experiments
Experimental device
The experimental device used in the investigation is self-designed and self-made, and the test system schematic is shown in Figure 2. The main performance parameters of the test system are listed in the Table 1. Tightness is a very important factor that affects test results for gas experimental equipment (Fang et al., 2009). Helium is used to test the system tightness at a constant temperature of 30°C before the experiments. The gas pressure is kept at an initial value of 9.577 MPa for 19 h, which fully satisfied the test requirements.

The experimental device. 1—CH4, 2—circulating moisture bath, 3—high pressure cell, 4—coal sample, 5—strain gauge,6—computer, 7—strain monitoring device, 8—vacuum pump, 9—acquisition card, 10—vacuum gauge, 11—pressure transducer, 12—inlet valve, 13—pressure reducing valve, 14—dryer, 15—vacuum pumping valve, and 16—exhaust valve.
Main performance parameters of the test system.

Experimental samples
The tectonically deformed coal is not only a sign of geotectonic movement, but also a typical geological body of gas. Almost all the coal and gas outburst are related with the structure of tectonically deformed coal (Zhang et al., 2007). Due to the tectonic stress, there were obvious differences among the damage degree, morphological characteristics, and physical properties of coal seams (Tang et al., 2004). The coal seam suffered a severe damage, especially the textures and structures are destroyed, is called “soft layer” (Wu, 1989), and the coal in the soft layer is called tectonic soft coal (Gao et al., 2013). On the other hand, the coal seam without damage or suffered slight damage is called “hard coal” (Tang et al., 2005). Observations showed that the coal and gas outburst happened first in the tectonic soft coal and with the thick of soft layer increase, the risk of coal and gas outburst increased (Lin and Zhou, 1986). Therefore, deeply development related to gas properties study of tectonic soft coal has important significance to understand the mechanism of coal and gas outburst, and the coal samples in this paper were taken from the soft layer in the eighth coal seam of the Yuyang Mining Company in Chongqing, China, which had coal and gas outburst tendentiousness. The industrial analysis results and pore characteristics of the samples are summarized in Table 2.
Properties of the coal sample.

daf: dry free ash.
However, the strength of tectonic soft coal is very low, as shown in Table 2, the firmness coefficient is only 0.16. It can be twist into powder by hand easily. The tectonic soft coal is too soft to process into intact coal samples. In view of this, some scholars put forward by pressure molding coal sample (also called briquette or shaped coal) instead of intact coal to develop related research (Dutka et al., 2013, 2012; Hu et al., 2012; Otsuka et al., 1982; Skoczylas, 2012; Sobczyk, 2011; Yin et al., 2009; Zhang et al., 2014). Recently, Norbert et al. (2014) suggested, in required situations, the use of coal briquette as a research material. A comparison of the sorption and deformation behavior between intact coal and briquette has been carried out in the previous work (Zhang et al., 2014). A strain trend comparison is shown in Figure 3, for illustrating the intact coal’s fungibility. It is in view of the briquette’s properties being similar to the intact samples, this paper uses briquette as the research object. Although different mechanics characteristics between intact coal and briquette are indeed existed, such as the intact coal is almost transverse isotropic due to the existence of the bedding, while the briquette is isotropic material due to the deformations of the radial and axial are approximately the same. Furthermore, the strain magnitude of the two samples is different.

Diagram of kinetic characteristic curves of adsorption/desorption-induced strain of intact coal and briquette (Zhang et al., 2014). (a) Intact coal and (b) Briquette.
As shown in Figure 4(a), a simple and self-made device is applied to make the briquette samples. The making process of the briquette samples is indicated as follows: (1) Grinding the massive soft coal with grinding equipment while screening the coal particles sized in the range of 0.18–0.25, (2) mixing approximately 31 g of coal particles with 10 ml distilled water while packing the mixture into the mold, (3) taking the mold immediately under the jack and putting the load sensors between the mold and jack, and (4) regulating the hydraulic pump slowly and observing the reading data of load sensor in real time to make sure the briquetting pressure is kept at 50 ± 0.1 kN for 15 min. The prepared briquette sample is shown in Figure 4(b). The moisture content of briquette sample reaches 10%, thus the strain gauge could not be conglutinated firmly under the original conditions. Therefore, the briquettes are putting in an environment at room temperature for a certain period of time (at least 4 h according to experience) before the experiment until the humidity is fit for conglutinating the strain gauge.

Briquette processing device and briquette sample: (a) Briquette processing device and (b) Briquette sample. 1—load sensors, 2—mold, 3—hydraulic pump, 4—jack, 5—loading mechanism, 6—digital display meter, and 7—electronic balance.
Deformation and moisture content measurement
The strains of the coal samples are measured using 5 mm one-way strain gauges. The test is required to complete a full cycle of adsorption/desorption process continually for the briquettes with different moisture content. Therefore, the vessel is designed to fit only one sample, with two strain gauges attached to the sample that were aligned radially and vertically.
After sealing the high-pressure cell, it was heated to a constant temperature of 30°C. Apparently, some moisture was lost during the “warm-up” period due to the evaporation. Typically, each sample will keep to this experimental condition for 12 h to reach the moisture content equilibrium before the test was started. In this process, a small amount of shrinkage was observed due to moisture loss (Day et al., 2008).
Before starting the measurements, the cell needs to be evacuated to degas the sample. It should be noted that this procedure removes a significant amount of moisture from the briquette. Hence, for the moist samples, the moisture content was controlled by the evacuating time (for at least 5 min) which aimed to remove air in the system and part of the moisture in the samples. For the dry sample, it was baked at 30°C under vacuum for at least 12 h before the experiment.
Experiments were run with 99.99% pure CH4 by increasing the pressure in a series of steps to a maximum pressure (approximately 8.8 MPa). At each gas pressure, both of the strain and pressure readings were permitted to stabilize before moving to the next pressure step. Once the maximum pressure was reached, the cell was depressurized in stages. After allowing the samples to attain equilibrium, the strains and pressures were monitored prior to progressing to the next pressure level. After the adsorption/desorption process finishing, the sample was removed immediately and measured by electronics with a precision of 0.01 g. Later, the sample was dried for more than 4 h at 105°C in a drying box to ensure that the free moisture was completely lost. Then, the residual mass was weighed and the moisture content was calculated.
According to the manufacturer specifications of the apparatus capacity, the equilibrium criterion of pressure was as follows: the changes of pressures <0.001 MPa/h; the equilibrium criterion of strain equals the changes of strain <3 × 10−6/h.
Results and analysis
Experimental results
For a particular coal sample, volumetric strain was adopted hereafter for evaluating the swelling and shrinkage characteristics of samples. The same with the standard rock mechanics (Durucan et al., 2009), the volumetric strain calculated by the strain perpendicular to the bedding plane and twice of the strain parallel to the bedding plane as

It should be noted that in the present investigation, as the common sense in geological science, the coal swelling strain was defined as negative deformation, while the shrinkage strain was positive deformation.
All of the experimental results are shown in Figure 5, including the adsorption and desorption volumetric strains at various equilibrium pressures with different moisture contents. However, the desorption data were lost at the moisture content of 1.66% due to the strain gauge failure. At a constant temperature and each moisture content, both of the volumetric strains in the adsorption and desorption processes decreased with the increase of the equilibrium pressure. However, the decreasing rate decreased with further increase of the equilibrium pressure. The volumetric strain would reach to a balance state with the increase of the equilibrium pressure. The trend in the cases of adsorption curves of high moisture content could be seen already as was shown in Figure 5(e) to (g). The volumetric deformation isotherms of adsorption and desorption indicated that the pressure and moisture content have significant influences on the methane adsorption and desorption deformation capacity of the briquette samples.
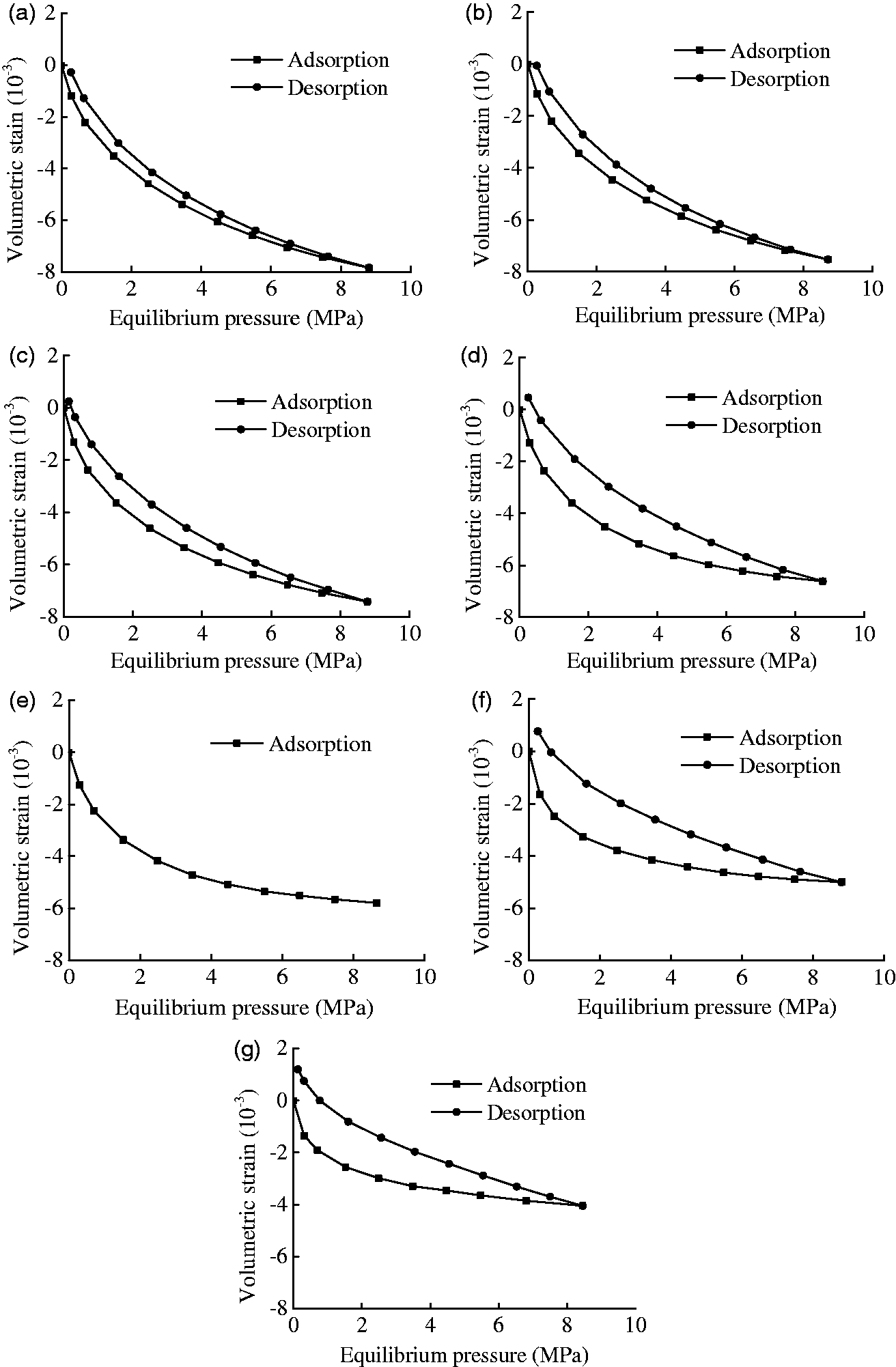
Measured volumetric strains of coal samples with different moisture contents. (a) Moisture content is 0.00%, (b) moisture content is 0.38%, (c) moisture content is 0.75%, (d) moisture content is 1.42%, (e) moisture content is 1.66%, (f) moisture content is 2.66%, and (g) moisture content is 3.13%.
Many studies verified that the CH4-induced swelling behavior could be modeled using a Langmuir-type equation in the following form (Cao et al., 2013; Lin and Zhou, 1986; Zhang et al., 2014)

Lin and Zhou (1986) suggested that the CH4-induced shrinkage behavior had the form of an exponential equation, while Cao et al. (2013) proposed a power function model. Most of the research indicated that shrinkage behavior did not match the standard Langmuir equation. In the present test, the difference existed in the residual strain when the equilibrium pressure was zero. A fitting equation of the shrinkage strain was established as

The fitting coefficients of the experimental curves in Figure 5 by equations (2) and (3) are listed in Table 3. The fitting curves are plotted in Figure 6, together with the experimental data. The fitting accuracy was very good because all of the fitting variances R2 were greater than 0.988. It could be found in Table 3 that when the coal sample’s moisture content increased from 0 to 3.13%, the swelling characteristic parameters (
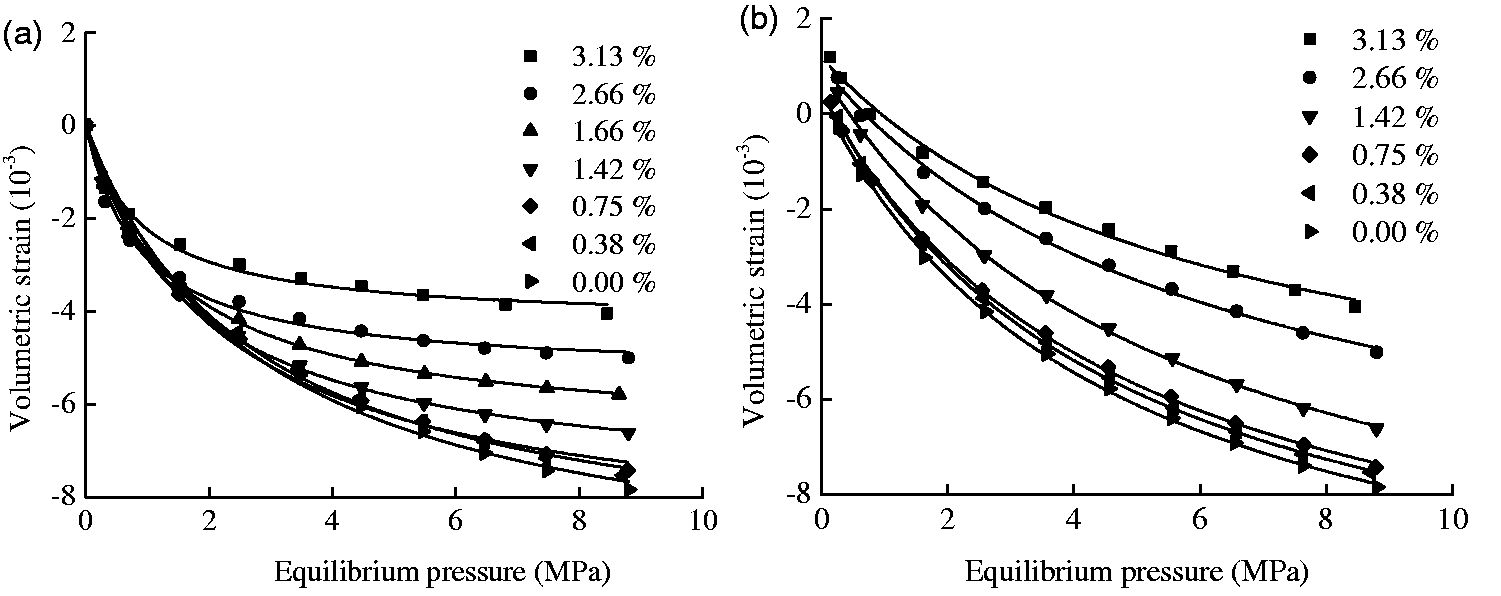
The strain curves of coal samples with different moisture contents. The line plots represent the modified model results. (a) Swelling and (b) shrinkage.
Fitting coefficients.

Influence of moisture content
The swelling and shrinkage of the coal samples with different moisture contents at various equilibrium pressures are shown in Figure 6(a) and (b). Both of the curves indicated that the moisture content significantly influences the CH4-induced deformation capacity of the coal. The volumetric strain of coal with a high moisture content was less than that of coal with a low moisture content at a constant equilibrium pressure, which indicated that the moisture content would reduce the adsorption and desorption deformation capacity of coal. For instance, at an equilibrium pressure of 7 MPa, the swelling deformations were −7.23, −6.98, −6.89, −6.30, −4.77, and −3.77 × 10−3 and the shrinkage deformations were −7.10, −6.99, −6.71, −5.91, −4.32, and −3.53 × 10−3 for a moisture content of 0.00, 0.38, 0.75, 1.42, 2.66, and 3.13%, respectively. At an equilibrium pressure of 8 MPa, the swelling strains were −7.50, −7.23, −7.11, −6.46, −4.84, and −3.83 × 10−3 and the shrinkage deformations were −7.49, −7.28, −7.09, −6.31, −4.65, and −3.82 × 10−3 for a moisture content of 0.00, 0.38, 0.75, 1.42, 2.66, and 3.13%, respectively. These results indicated that with a pressure increase from 7 to 8 MPa, the swelling strain would increase by 3.8, 3.6, 6.2, 2.6, 1.4, and 1.4%. Meanwhile, with a pressure decreasing from 8 to 7 MPa, the shrinking strain decreased by 5.1, 5.3, 5.4, 6.3, 7.2, and 7.5% for the coal samples with a moisture content of 0.00, 0.38, 0.75, 1.42, 2.66, and 3.13%.
Hysteresis of swelling and shrinkage deformation
Related studies showed that adsorption/desorption isotherms exhibited obviously hysteresis, that is the desorption isotherms lag was relative to adsorption isotherms (Ma et al., 2012). As seen from Figure 5, the swelling and shrinkage deformation curves also would exhibit hysteresis characteristics, but the characteristics were different from those of the adsorption/desorption isotherms. The hysteresis mentioned hereafter indicated that the swelling deformation lags corresponded to shrinkage deformation. To illustrate this appearance, a variable referred to the hysteresis rate was defined as

The hysteresis rate versus equilibrium pressure curves for different moisture contents are shown in Figure 7. The following phenomenon could be observed: (1) The hysteresis rate increased when the equilibrium pressure decreased, and low pressures correspond to a large hysteresis rate value. In other words, the hysteresis of the isothermal adsorption/desorption deformation curves was more significant at a lower equilibrium pressure. (2) The hysteresis rate increased when the moisture content increased at a certain equilibrium pressure. This indicated that the moisture content significantly affects the hysteresis of the isothermal adsorption/desorption deformation curves.

Hysteresis rate with different moisture contents.
Mechanism of moisture influences on gas-induced deformation
The deformation mechanism of gas-induced swelling in coal can be explained by three aspects (Cao et al., 2013; He et al., 1996; Lin and Zhou, 1986; Liu et al., 2010; Zhang et al., 2014). First, at low pressures, the methane molecules entered into the micropores and microfractures with a size greater than or equal to the mean free path of methane gas molecules. The surface energy of the micropores and microfractures was reduced due to the methane adsorption and resulting in an increased thickness of the surface layer. Concurrently, the micropore and microfracture volume increased with the presence of free methane. Second, at high pressures, the methane molecules wedge into the fractures with sizes greater than or equal to the gas molecular size and promote the formation and propagation of micro cracks, which resulted in coal expansion. Third, a certain compression deformation under the methane pressure occurred in the coal matrix, and this deformation segment offseted part of the coal swelling. It was apparent that the higher the methane pressure is, the more obvious the compression deformation is. However, in these experiments, compression deformation was relatively small compared to the gas-induced swelling.
Li and Nie (2006) investigated the coal adsorption characteristic of moisture and indicated that the presence of moisture may affect the performance of coal on gas adsorption in three ways. First, part of the external moisture combined with the coal surface by wetting action and it occupied a certain amount of the adsorptive space. The corresponding effective area of coal gas absorption was decreased, and the gas adsorption decreased. Second, due to the moisture vapor pressure, a spot of inherent moisture would exist in the small pores in a gaseous state. There was competitive adsorption between the moisture vapor molecules and the methane molecules on the same active competition center, thus resulting in the decrease of methane adsorption. Third, moisture blocked the methane molecules from passing into the pore channel. The effects of these three factors decreased the coal adsorption capacity and reduced the gas-induced swelling capacity.
The softened property of the moisture would also affect the coal deformation capacity. The wetting action of moisture weakened the link between coal particles, and the mechanical properties of the coal changed from good to bad; pressure was also produced by the moisture in pores and cracks. As a result, the elastic modulus of the coal decreased, and its deformation ability was enhanced (Yan et al., 2002), leading to an increase in the compressive deformation of the coal matrix in the role of free methane pressure. Thereby, part of the swelling in the adsorption process was offset, and shrinkage in desorption process was promoted.
Additionally, the deformation characteristics of coal were also affected by the existence of moisture through the change of pore distribution. As discussed in the first part of this paper, the coal pore distribution and connectivity were not uniform and included interconnected open pores and disconnected closed pores. As shown in Figure 6, the coal particles were simplified as a square model containing only one open pore (a micropore) without considering the methane adsorption in the coal. For dry coal (Figure 8(a)), methane gas entered the open pore due to the seepage and diffusion effects, at which time the coal matrix exhibited a certain compression deformation under the methane pressure. Thus, the coal particles showed shrinkage deformation. The pore volume also increased due to internal gas pressure. This deformation offseted part of the shrinkage deformation of coal particles. Comprehensive performance for coal sample particle shrinkage deformation occurred; apparently, the shrinkage deformation of coal particles contained only part of the compression deformation of the coal matrix. For the saturated coal (Figure 8(b)), it was difficult for methane gas to enter the original open pore due to the blocking effect of the moisture. Therefore, the open pore was essentially transformed into a closed pore, reducing the effective space of the coal and decreasing the effective porosity. The shrinkage deformation of the coal particle included the compression deformation of the coal matrix and the compression deformation of the pore. Obviously, the compression deformation of coal saturated with moisture was greater than the deformation of dry coal under the same equilibrium pressures. Therefore, the effective porosity of coal tended to be lower when the moisture content was higher; the coal sample was more prone to produce compression deformation, thereby reducing the adsorption swelling capacity and promoting the shrinkage capacity of the coal.
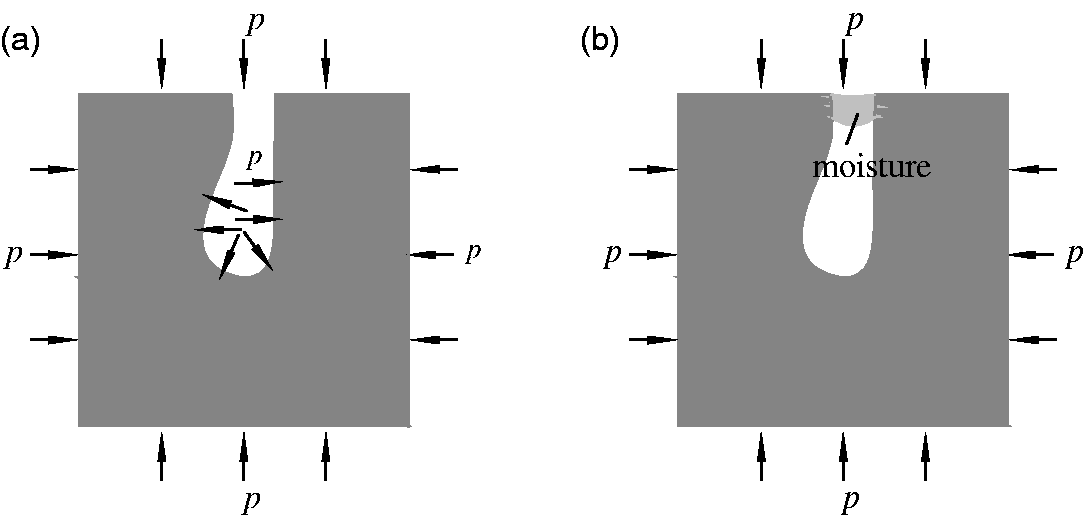
Schematic of the force applied to a coal particle. (a) Dry coal and (b) moist coal.
In the adsorption process, the coal adsorption capacity decreased due to the existence of moisture coupled with the softening effect and the change of the coal pore distribution. The compression effect of the coal matrix was enhanced by free methane, which partly offseted the swelling deformation, resulting in a decreased strain rate of coal swelling and allowing the swelling to easily achieve equilibrium under the same equilibrium pressures. Then, the rate manifested as the swelling characterization data
Deformation models considering moisture content
According to the foregoing analysis, moisture has a significant effect on the adsorption/desorption-induced swelling/shrinkage deformations. The swelling and shrinkage deformations follow equations (2) and (3) very well, but the unique variable in the two equations is only the equilibrium pressure without moisture. Therefore, the model construction section tries to establish deformation model that can reflect the effects of both moisture and gas pressure, so as to accurately describe the deformation characteristics of coal samples under different moisture content and different pressure conditions.
Model construction
The swelling and shrinkage deformations follow equations (2) and (3) very well. The unique variable in the two equations is the equilibrium pressure. According to Table 4, the coefficients in equations (2) and (3) are related to moisture content and could be fitted by linear or exponential curves in advance, as shown in Figure 9.
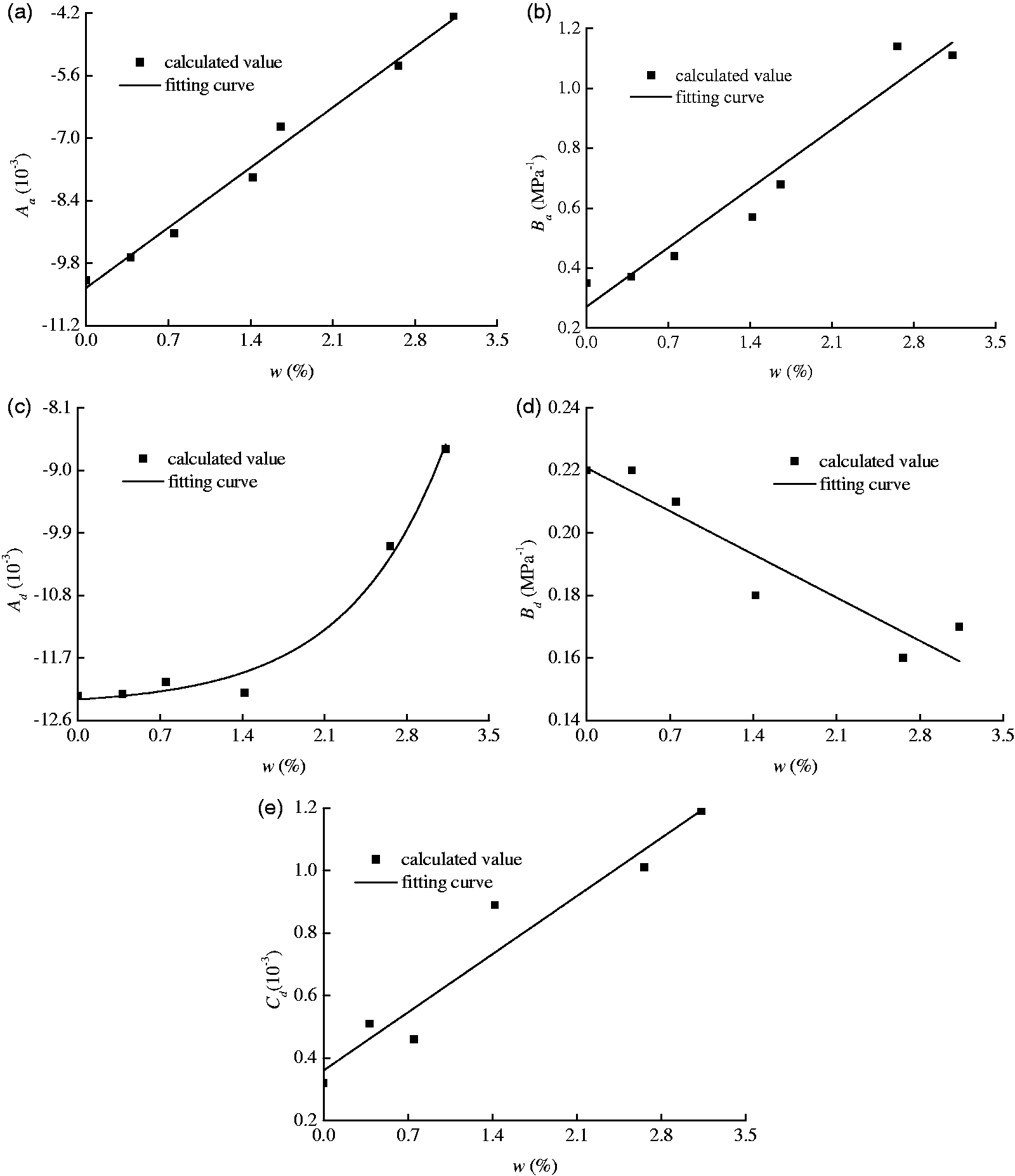
Relationship of the fitting coefficients with moisture content. (a)
Fitting relationships between the parameters and the moisture.

During adsorption, the swelling deformation indicated by equation (2) and its coefficients 
The coefficient 
During desorption, the shrinkage deformation was indicated by equation (3), and the coefficient 
The coefficient 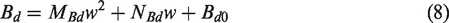
The coefficient 
Substituting equations (5) and (6) into equation (2), the mathematical model of the swelling deformation, considering the effect of moisture content, is rewritten as

Similarly, by substituting equations (7) to (9) into equation (3), the mathematical model of the shrinkage deformation, considering the effect of moisture content, can be written as
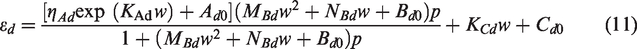
Model validation
In order to validate the reliability of the proposed deformation models, the model results and experimental results are compared as shown in Figure 10. The results showed that the model results exhibit a good calculation accuracy and the deformation models could describe the relation between moisture content and features of adsorption- and desorption-induced deformation of coal.
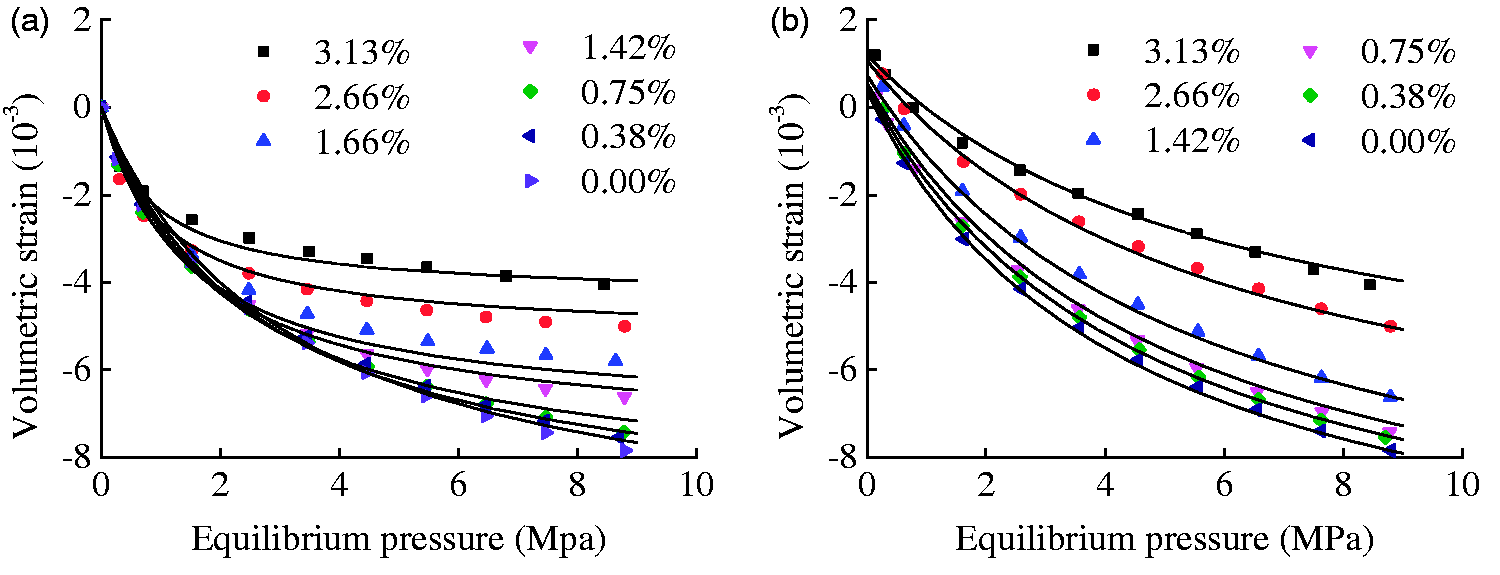
The results of model validation. The line plots represent the model results. (a) Swelling and (b) shrinkage.
Conclusions
The swelling behavior of coal samples with different moisture contents obeyed the Langmuir-type equation Moisture had a significant influence on the CH4-induced deformation capacity of coal. The swelling capacity exhibited an inverse relationship with the moisture content, while shrinking capacity exhibited appositive relationship with the moisture content. At a constant equilibrium pressure and increasing moisture content, coal swelling capacity decreased, and the swelling strain values increased. As the coal shrinking capacity increased, the residual strain values, the swelling strain, and the deformation rate would increase. The swelling and shrinkage strain curves exhibited significant hysteresis, which means that the swelling strain curve lags related to the corresponding shrinkage strain curve. The lower the equilibrium pressure was, the more obvious the hysteresis was; the higher the moisture content was, the more obvious the hysteresis was. Moisture affected the CH4-induced swelling and shrinkage capacity of coal, mainly in three ways: the reduction of the adsorption/desorption capacity of coal, a softening effect on the mechanical properties of coal, and changes in the pore distribution of coal to reduce the effective porosity. Because of the high gas pressure, the shrinkage was greater than the swelling after the methane was completely desorbed. It was clear that typical residual strain occurs when the coal sample completely desorbs the methane, which is larger and has a higher moisture content. Based on the best-fit relationships between the deformation characterization data and the moisture content, the mathematical model of swelling and shrinkage strains considering the effect of moisture was established. This mathematical model accurately showed the deformation features of coal adsorption/desorption at low and high pressures.
Footnotes
Acknowledgements
We thank the reviewers and editors for their constructive suggestions while proofreading this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (51704147, 51404046, 51604116), the Foundation of Liaoning Educational Committee (LJYL005), the Basic Research of Frontier and Application of Chongqing (cstc2016jcyjA0085), and the Chongqing Postdoctoral Research Project (Xm2017117).
