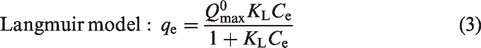Abstract
The capacity and underlying mechanism of hydrochars derived from commercial
Keywords
Introduction
In developing countries like Vietnam, the concurrent expansion of industrialization and population growth has led to a significant increase in water pollution in recent years. Certain types of organic dyes have been recognized as major contaminants and frequently detected in water bodies due to the fact that they are recalcitrant to be degraded by microbes (Zhang et al., 2013). Methylene blue (MB), which is widely used in textiles, printing, dyeing, dyestuff manufacturing and food plants, is one of the dyes that cause environmental problems (Anastopoulos and Kyzas, 2014). Hence, how to effectively remove MB from wastewater has become an increasingly critical issue (Mohanty et al., 2006).
To date, a number of methods such as electrochemical techniques, membrane separation, photo-degradation, flocculation-coagulation, ozonation, anaerobic treatment, adsorption, etc. have been developed to remove MB from industrial effluent (Cengiz et al., 2012; Mohammadi et al., 2011; Wang, 2017). Of them, adsorption is considered the most common approach ( Padmapriya et al., 2019a, 2019b; Yagub et al., 2014). In the past, activated carbon was often used as the preferential sorbent in this regard; however, high pyrolysis temperatures including activation steps in the manufacture make activated carbon, in general, a high-cost material (Zhang et al., 2015). Thus, there is a need for the development of novel adsorbents via a relatively simple, green, and robust means for effective use in dealing with MB contamination in aqueous solutions.
Recent attention has been drawn to hydrochar, an emerging environmentally friendly adsorbent that can be derived from hydrothermal carbonization (HTC) of organic wastes including agricultural refuses, forest residues, and animal husbandry manures (Fang et al., 2015, 2017; Wang et al., 2018b). HTC is a viable technology for biomass conversion (Libra et al., 2011; Oliveira et al., 2013), through which the biomass is heated at relatively low temperatures (180–350°C) and decomposed in a closed reactor under autogenous pressure (Fang et al., 2017; Jain et al., 2015; Libra et al., 2011). The formation of the products, i.e. hydrochars, is thus a result of hydrolysis, condensation, decarboxylation, and dehydration (Berge et al., 2011; Sevilla and Fuertes, 2009b; Xiao et al., 2012). Although the specific surface area of hydrochar prepared in this way is typically lower than that of biochar—another less-expensive carbonaceous sorbent that is also considered an environmentally sustainable alternative to activated carbon—hydrochar may exhibit higher adsorption capacity toward the given contaminants as compared with biochar and even activated carbon because of its relatively abundant oxygenated functional groups (e.g. carboxylic, phenolic, hydroxyl) on the surface (Elaigwu and Greenway, 2016; Jain et al., 2016). Therefore, a considerable amount of investigations on the potential use and pertaining mechanisms of hydrochar in removing various contaminants from the aqueous phase has been undertaken lately (Islam et al., 2017; Sun et al., 2015; Xue et al., 2012). For example, Sun et al. (2011) demonstrated that for the given polar and nonpolar organic contaminants (e.g. bisphenol A, 17α-ethinylestradiol (EE2) and phenanthrene), hydrochar possessed higher uptake capacity than biochar under otherwise identical conditions; also, the study of Tran et al. (2017c) pointed out that increases in oxygenated groups on the surface of hydrochar resulted in electrostatic attraction, rather than π–π interaction, as the primary mechanism underlying the adsorptive interaction between hydrochar and methylene green, a cationic dye-like MB.
Notably, hydrochar synthesized using different feedstocks may possess different kinds and varying abundances of surface functional groups (Libra et al., 2011). In addition, with an effort to promote the adsorption capacity of hydrochars, several chemical agents have been applied to modify hydochar surface structure. Nitric acid (Güzel et al., 2017; Jin et al., 2018; Shim et al., 2001), sulfuric acid (Jiang et al., 2003), hydrogen peroxide (Huang et al., 2018; Wang et al., 2018a; Xue et al., 2012), potassium hydroxide (Sun et al., 2015), ozone (Valdés et al., 2002), and phosphoric acid (Chen et al., 2017) are the reagents frequently used to enhance oxygen content on the adsorbent surface. Although such a chemical modification process may also affect the surface area and pore size distribution of the adsorbent (Güzel et al., 2017), when a synthetic hydrochar is gone through such advanced oxidation processes at room temperature and converted to the so-called “oxidized-hydrochar,” enhancement in its adsorption performance in large part owing to the further increasing oxygenated moieties on the material surface can be expected (Xue et al., 2012). Yet, to the best of our knowledge, the adsorption of MB on oxidized-hydrochar has not been extensively researched.
In this study, two kinds of hydrochars were produced using commercial
Materials and methods
Feedstock and chemical
OP was collected from a local market at Taoyuan City, Taiwan. Upon collection, OP was washed with tap water and deionized water (DI-water) to remove any water-soluble impurities and adhering dirt. After washing, OP was dried at 80°C for 48 h and crushed to 0.074–0.105 mm particles using an electric grinder. All chemicals are analytical grade with the purity more than 95%. Commercial D-glucose was purchased from Sigma-Aldrich (Darmstadt, Germany). The basic dye, MB (Type: cationic; C.A.S: 7220–79-3; chemical formula: C16H18ClN3S.xH2O (x = 2–3); Mw: 319.86 g/mol (anhydrous basic); λmax: 665 nm; color index: basic blue 9) was chosen as the adsorbate. MB was purchased from Fisher Scientific, and the solution was prepared by dissolving MB in DI-water (Pelekani and Snoeyink, 2000).
Preparation and modification of hydrochars
Hydrochars were prepared following a typical HTC process (Mumme et al., 2011). In brief, approximately 15 g of the precursor was added in a 150-mL Teflon-lined autoclave-containing 110 mL of DI-water. After a 24-h HTC process at 190°C with a heating rate of 8°C/min, the brown precipitate was separated by filtration. The obtained hydrochar particles were washed with 0.1 M HCl and then repeatedly rinsed with DI-water until the filtrate reached a neutral pH. The particles were then dried at 105°C for 24 h, ground, and sieved to a size ranging 0.074–0.105 mm. The samples were stored in airtight bottles and used as pristine-hydrochars without any further treatments (Tran et al., 2017c). Hereinafter, hydrochars derived from OP and D-glucose were designated as OPH and GH, respectively.
For the production of oxidized-hydrochars, different concentrations of nitric acid (HNO3) were used as the modification reagents. One gram of the GH or OPH was added to 30 mL of 30, 50, and 70% (w/w) of HNO3 solution and reacted for 4 h. After the oxidation process, the samples were washed with DI-water and then dried overnight at 105°C. The modified samples were referred as mGH and mOPH, respectively (Figure S1).
Adsorbent characterization
The pristine and oxidized-hydrochars were characterized with the elemental composition, specific surface area, identification of functional groups on the surface, quantification of oxygen-containing functional groups, and the point of zero charge (PZC).
The main elements (% carbon, oxygen, and nitrogen) were measured using X-ray photoelectron spectrometer techniques (Thermo Scientific, UK) with high-resolution spectra binding energies. Meanwhile, the morphology of hydrochar samples was also examined using scanning electron microscopy (SEM; JSM-7600F, JEOL). The pore characteristics of the samples were determined by N2 adsorption at 77 K using a surface area analyzer (ASAP 2020/Tristar 3000, Micromeritics Instrument Corp.). The specific surface area was calculated following the multipoint N2-Brunauer–Emmett–Teller (BET) adsorption method. The volume and surface area for the micro-pores were obtained using the t-plot method. The functional groups on the samples were examined using Fourier transform infrared spectroscopy (JASCO FT/IR-4600, JASCO International Co., Ltd.) in the wavenumber range of 4000–500 cm−1. Oxygen-containing functional groups were quantified using the Boehm titration method (Boehm, 1966). According to the report, based on the drift methods, the pH value of the sample at the PZC (pHpzc) was determined (Tran et al., 2016).
Adsorption study
The MB concentrations in the solution were determined at the wavelength of 665 nm by a UV–vis spectrophotometer (Genesys 10S UV–VIS, Thermo Scientific) (Figure S2). The adsorbed amount of MB in equilibrium, qe (mg/g), was calculated by the mass-balance equation
The adsorption isotherms were conducted in centrifuge tubes containing 25 mL of MB with various initial concentrations (100–1000 mg/L) at pH 7.0 and 0.05 g of the selected hydrochar sample at 10, 30, and 50°C. The tubes were placed in a reciprocating shaker with 150 r/min equilibrated for 24 h. The equilibrium concentrations of adsorbate lay in the range 20–80% of initial concentrations to obtain the more accuracy parameter values. The solution pH was set as 7.0 that could avoid H+ or OH– interference during the adsorption process. The equilibrated amount of MB on the adsorbent was determined based on equation (1). The effect of pH of initial dye solutions (2.0–11.0) was also investigated. The Freundlich (1906) and Langmuir (1918) models presented in equations (2) and (3) were used to describe the adsorptive behavior of MB onto the hydrochars
Results and discussion
Morphological and textural properties of hydrochars
The SEM images for the pristine and oxidized-hydrochars are illustrated in Figure 1. The sizes of these hydrochar particles ranged from several hundreds of nanometers to several micrometers. Notably, the pristine-hydrochars derived from D-glucose were spherical particles with a relatively more uniform size and smooth surface, whereas hydrochars prepared from OP displayed fragment shapes and porous structures. These morphological results are similar to other studies (Sevilla and Fuertes, 2009a; Tran et al., 2017c).

SEM images of (a)
Interestingly, the SEM images of the oxidized-hydrochars are not significantly different from those of pristine-hydrochars, as the shape of mGH also clearly exhibited micrometer-sized spheres (Figure 1(b)), and mOPH showed the non-spherical shape with irregular surface and fragmentation connection (Figure 1(d)). This indicated that HNO3-modification did not markedly destruct hydrochar surface.
Table 1 shows that the BET surface area of samples ranged from 4.49 to 34.06 m2/g. This is expected because hydrochars have been shown to possess relatively low surface area as compared with biochars as well as activated carbons. In addition, Table 1 clearly shows that surface modification resulted in the oxidized hydrochars having lower BET surface area and limited pore volume, which is similar to the results of biochar and activated carbon modification (Gokce and Aktas, 2014). Because the pore volumes were similar between pristine and oxidized-hydrochars, the obtained result was attributed to the blocking of the narrow pores by HNO3 treatment (Boehm et al., 1982). Moreover, the increases in pore size after the oxidation process seemed to reflect this blocking effect.
Textural properties of the hydrochar samples.

Chemical properties of hydrochars and oxidized-hydrochars
Elemental composition
Table 2 summarizes the elemental composition of the surface of the synthetic hydrochars. The carbon contents of the hydrochars are roughly ranged from 70 to 79%, consistent with that reported by others (Fang et al., 2017; Sun et al., 2014). The carbon content decreases after HNO3 treatment, whereas the oxygen and nitrogen contents increase. This might be due to (i) the corresponding increase in oxygen-containing functional groups involving carboxylic, phenolic, and nitro moieties on the surface (Fang et al., 2015; Kim et al., 2011) and (ii) increase in NO2 or NO3 groups resulting directly from HNO3 (Trompowsky et al., 2005). It could be concluded that nitric acid oxidation could increase the oxygen and nitrogen content on the surface of hydrochars.
Surface elemental compositions of the hydrochar samples.

Surface polarity ((O + N)/C) was determined from the atomic ratio of (O + N) and C.
In this study, O/C ratios representing the degree of hydrophilicity were regarded as one of critical factors to determine the adsorptive amount (Jin et al., 2018). The mGH possesses the highest O/C ratio about 0.3. Hence, it is expected that mGH would generate the higher adsorptive amount for MB.
Surface functionality
The images of FTIR for before and after MB-adsorption on the pristine and oxidized-hydrochars are shown in Figure 2. The 3200–3650 cm−1 represents the OH group, which corresponds to an associated peak of the hydroxyl group from phenols, alcohols, and absorbed water (El-Hendawy, 2003). Because the hydrochars might generate dehydration and decarboxylation reaction during the HTC process, the peaks located in 1500–1600 cm−1 resulted from aromatic C=C ring (Fang et al., 2017; Libra et al., 2011). All adsorbents with stronger peaks at 1650–1750 cm−1 indicated the C=O bond on the surfaces. The peak around 1137 cm−1 was attributed from the C–O stretching vibrations, which was consistent with carboxyl O=C–O, epoxy C–O–C, and alkoxy C–O bonds, respectively. It seems that the functional groups for O–H stretching, C–H stretching, aromatic C=C ring, and C=O stretching, C–O stretching, and aromatic C–H bending significantly increased after HNO3 activation.

FTIR spectra of the synthesized hydrochar samples before and after adsorption: (a) GH and mGH; (b) OPH and mOPH.
In addition, acidic and basic surface functionalities determined using the Boehm titration methods (Boehm, 1966) are summarized in Table 3. It can be seen that the HNO3 treatment processes lead to an increase in carboxyl, lactone, and phenolic hydroxyl groups on the oxidized-hydrochars. This is consistent with the data shown in Figure 2 and Table 2. These oxygen-containing functional groups played an important role in the hydrochar–MB interaction.
The acidic and basic groups of the hydrochar samples determined by the Boehm titration.

MB adsorption
The adsorption isotherms for MB on the adsorbents conducted at pH 7.0 are illustrated in Figure 3, and the relative adsorption parameters are listed in Table 4. In Figure 3, the isotherms exhibited the concave-downward curves, representing the high affinity between the adsorbate and adsorbents. Further, the R2 values obtained from the isotherms revealed that the equilibrium data fitted the Langmuir model well, and the maximum adsorption capacities of MB followed the decreasing order of GH < OPH < mOPH < mGH (Table 4).

Adsorption isotherms of GH, mGH, OPH, and mOPH for MB at 30°C. (Experimental conditions: initial MB concentrations ranging from 100 to 1000 mg/L, initial solution pH: 7.0 and contact time: 24 h).
Corresponding isotherm parameters for MB adsorption onto the hydrochar samples.

The effects of pH (2–11) on MB adsorption on the all hydrochars at 30°C are shown in Figure 4(a). In Figure 4(a), the data indicates that the amounts of adsorbed MB are the function of solution pH. Interestingly, the hydrochar samples, especially in the oxidized-hydrochars, seemed to remain the capacity of MB-adsorption at pH as low as 2.0. Values for PZC of the tested adsorbents are shown in Figure 4(b) according to the method in literature (Tran et al., 2016). The higher PZC values often coincide with a lower density of acidic groups on the surface. These data, together with those in Figure 3, indicate that the maximum MB-adsorption capacity on the hydrochars could fit the order for the surface charge density of acidic functional groups, i.e. GH< OPH< mOPH< mGH, suggesting that the ion exchange might have been one of the adsorption mechanisms for MB–hydrochar interactions. However, the hydrochars generating the adsorption amount for MB in low pH solution represented the other adsorption mechanisms needed to be considered.

(a) The pH dependence on adsorption capacity; (b) point zero charge of the hydrochar samples. (Experimental conditions: initial (MB): 620 mg/L, contact time: 24 h, temperature: 30°C and solution pH: 7.0).
In Table 4, the adsorptive amounts of MB onto hydrochars increase with the increasing temperatures for each adsorbent at 10, 30, and 50°C. The adsorptive amount of MB increased with the temperature, implying that the van der Waals force was not the primary adsorption mechanism (Güzel et al., 2014; Tran et al., 2017b). In addition, the Q0max values on the oxidized-hydrochars were found four to fivefold higher than those on the counterpart hydrochars at 30°C. Therefore, it can be clearly concluded again that the HNO3-modified process could effectively enhance the MB removal. The reason was attributed to the test oxidant that can alter the surface functional groups of adsorbents. Similar results for other carbonaceous materials with respect to dye adsorption have been reported in literature (Güzel et al., 2017; Ncibi et al., 2007).
The maximum adsorption capacities of MB on the oxidized-hydrochars compared with carbonaceous materials reported in literature are listed in Table 5 (Ai and Li, 2013; Fang et al., 2015; Gokce and Aktas, 2014; Güzel et al., 2014; Hao et al., 2014; Islam et al., 2017; Qian et al., 2018; Sun et al., 2013). Although the Q0max values in this study are lower than those from activated carbons, the cost for the hydrochars and oxidized-hydrochars is far less than that for activated carbons. As a result, the oxidized-hydrochars can still be regarded as the potential candidates for effective MB removal.
Comparison of adsorption capacities of various adsorbents for MB removal.

Adsorption mechanisms of MB on the oxidized-hydrochar
The adsorption mechanisms of aromatic dyes onto carbonaceous materials normally include (i) van der Waals force, (ii) pore filling, (iii) electrostatic force, (iv) hydrogen bond, (v) π–π interaction, and (vi) n–π interaction (Tran et al., 2017a, 2017c). However, given the differential surface characteristics among carbonaceous sorbents, the primary interactions between MB and hydrochars may be significantly different from biochars and activated carbons. The van der Waals force mainly involves physical adsorption. The oxidized-hydrochars do not possess the high specific surface area. Thus, van der Waals force is not an important mechanism for the MB adsorption. The pore filling easily occurs in the porous carbonaceous sorbents. The mechanism is not suitable for the MB adsorption on the oxidized-hydrochar with less micro-pore and low BET specific surface area.
The adsorbate MB (formula: C16H18ClN3S) is an aromatic dye that contains an amino group (R2N+), possibly exhibiting positive charge. The surface of oxidized-hydrochar with rich acid groups could attract the MB molecules by electrostatic force. The result can be demonstrated through the total acidic groups in Table 3. The adsorption mechanisms regarding to electrostatic force and hydrogen bond are indicated in Figure 5. Moreover, the oxygen and nitrogen content potentially determined the hydrogen bond between the adsorbate and adsorbent. The hydrogen bond might play a role in the adsorption of MB on the hydrochars.
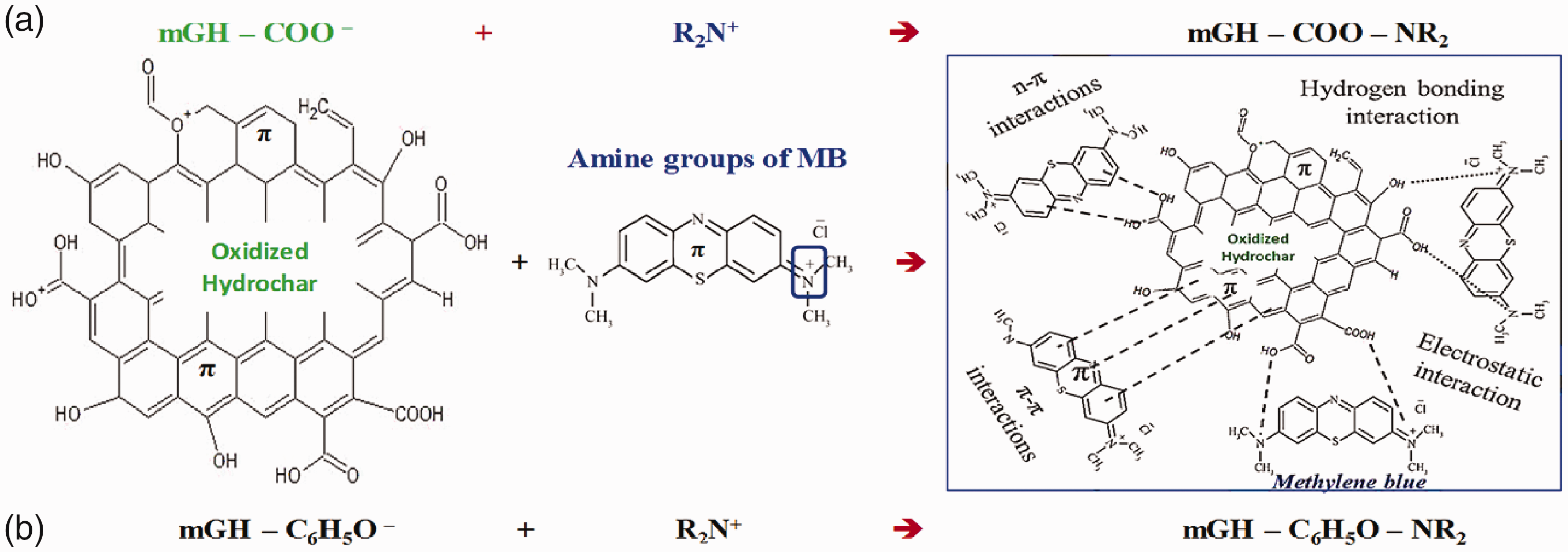
Proposed adsorption mechanisms for MB removal onto oxidized hydrochars.
As for the π–π interaction, it can be defined that the π bond on the surface of adsorbent might generate the interaction with that in the adsorbate. The investigators have presented that the π–π interaction can be neglected for the adsorption of dye on the carbonaceous material (Tran et al., 2017c). In this study, the aromatic dye MB possesses the unsaturated bonds. Moreover, the oxidation process by nitric acid can enhance the π bond resulting from aromatic C=C ring and C=O on the oxidized-hydrochars. Thus, the π–π interaction was regarded as a potential adsorption mechanism between MB and oxidized-hydrochars. The n–π interaction can be regarded as the interaction between the electron-donor and electron-acceptor. The lone electron pair (electron-donor) can generate the interaction with the π bond (electron-acceptor). The O and N atoms can provide the lone electron pair. It has been known that the surfaces of oxidized-hydrochars possess the rich unsaturated bonds to form the π bond. The n–π interaction is the potential adsorption mechanism.
In this study, the adsorption mechanisms were divided into three groups. The van der Waals force and pore filling were less-important for the MB adsorption on the hydrochars or oxidized-hydrochars. The electrostatic force and hydrogen bond were the critical adsorption mechanisms regarding to the MB adsorption on the hydrochars and oxidized-hydrochars. The π–π interaction and n–π interaction for the MB adsorption on the hydrochars and oxidized-hydrochars indicated the inconsistent result. The two mechanisms are less important for the MB adsorption on the hydrochars. When the oxidation process enhanced the oxygen-containing or nitrogen-containing functional groups and unsaturated bonds on the surface of oxidized-hydrochars, the π–π interaction and n–π interaction became the potential adsorption mechanisms.
Conclusions
Hydrochar products derived from D-glucose and OP were successfully modified with nitric acid to produce the oxidized-hydrochars. While having low BET-specific surface area, mGH and mOPH were enriched with more surface carboxylic and lactonic functional groups than GH and OPH. When these hydrochars were used as sorbents for MB adsorption, it was found that the adsorption of MB onto the hydrochar samples strongly depended on the level of these oxygen-containing moieties. On the basis of these observations, the electrostatic force might be the primary mechanism controlling the adsorption of MB onto the hydrochars, whereas hydrogen bonding and other pathways played minor roles. This study demonstrated that the modified hydrochar is promising new adsorbents, which could constitute a significant contribution to the search for cheaper adsorbent material for effective removal of MB from wastewater or within using to solve an environmental pollutant problem.
Supplemental Material
Supplemental material for Effect of nitric acid oxidation on the surface of hydrochars to sorb methylene blue: An adsorption mechanism comparison
Supplemental Material for Effect of nitric acid oxidation on the surface of hydrochars to sorb methylene blue: An adsorption mechanism comparison by Duy H Nguyen, Hai Nguyen Tran, Huan-Ping Chao and Chu-Ching Lin in Adsorption Science & Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology and the Environmental Protection Administration in Taiwan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.