Abstract
This paper aims to investigate the uptake of two basic dyes (methylene blue [MB] and brilliant cresyl blue [BCB]) using the formaldehyde-treated algae
Introduction
Overpopulation, urbanization, and industrial development are the leading causes of environmental pollution. Pharmaceutical, cosmetic, agricultural, and textile industries are the main polluters because their wastewater effluents are discharged without or with poor treatment (Raj et al., 2020). The wastewater effluents from the textile industry are a mixture of recalcitrant pollutants such as surfactants, disinfectants, and high concentrations of dyes (Paździor et al., 2017). Dyes are applied in biology and medicine for microbiological and histological stains. Water that is not clear is associated with poor quality or polluted water. A low dye concentration (1 mg L−1) is enough to be visually perceived (Malik et al., 2007) and may also present a risk to human health because some dyes are carcinogenic and mutagenic. Therefore, its regulation has been recently considered by the Zero Discharge of Hazardous Chemicals Program, which is an organization dedicated to eliminating hazardous chemicals in the leather, textile, and synthetic sectors (ZDHC, 2022).
The World Health Organization suggests drinking water should ideally have no visible color, or that levels of color below 15 TCU (True Color Units) are often acceptable to consumers (World Health Organization, 2022). The problem of color in water can be assumed as an aesthetic situation because water that is not transparent can be perceived as dirty water (Naseem et al., 2019). Furthermore, the loss of water transparency causes a decrease in the sunlight penetration, thus affecting the photosynthetic function of plants (Garg et al., 2004) and to a decrease in the amount of dissolved oxygen available to aquatic flora and fauna (Lakshmi et al., 2009). Therefore, color affects the balance of aquatic ecosystem and can lead to species turnover (U.S. Environmental Protection Agency, 1974).
The treatment of wastewater from the textile industry is one of the main environmental concerns today. Organizations such as Environmental, Health, and Safety (EHS) propose in their guidelines biological and physicochemical methods as the predominant control option for the treatment of the color parameter (EHS, 2007). Some biological materials, such as bacteria, fungi, yeasts, and algae, have demonstrated their ability to remove various synthetic dyes (Ali, 2010). These materials can break the bound of the chromophoric group by enzymatic action and remove the color (Satheesh Babu et al., 2015). The advantages of biological methods include that they are economically viable, environmentally friendly, and generate less sludge volume compared to other techniques, while their principal disadvantage is the transition from laboratory scale to real scale (Bhatia et al., 2017). Some bacteria have been successfully employed to eliminate different dyes. For example, the
The most applied physicochemical methods for dye removal are adsorption, ion exchange, oxidation process, and irradiation. In adsorption, the dye is adsorbed on the surface of the material through chemical or physical interactions. Various waste materials have been used as an adsorbent for dye removal. For example, sawdust was used to remove brilliant cresyl blue (BCB; Elhami et al., 2011) and methylene blue (MB; Garg et al., 2004). In addition, different types of resins were applied to remove basic dyes such as safranin T, Nile blue, and BCB (Iyim et al., 2008) and cationic dyes: brilliant green, methyl violet, MB, phenosafranine, and BCB (Çinar et al., 2018) using ion exchange.
Physicochemical techniques have a high dye removal efficiency, are easy to use, nevertheless require high production and operating costs, regeneration of the material is not always possible, and produce sludge or by-products.
Marine macroalgae such as
Experimental
Dyes
Methylene Blue C.I. 52,015 (C16H18ClN3S) and BCB C.I. 51,010 ((C17H20ClN3O)2·ZnCl2) were procured from Merck and Fagalab, respectively, as a dye source, their chemical structures were drawn with ChemDraw professional software (Figure 1(a) and (b), respectively).

Chemical structure of (a) brilliant cresyl blue and (b) methylene blue (ChemDraw professional software).
Preparation of biosorbent
Characterization techniques
pHPZC
The pHPZC of the raw and brown algae FV-FORMALD was determined using 0.01 M NaCl as the background electrolyte solution; the pH of the working solutions was fixed between 1 and 12 using 0.1 M NaOH or 0.1 M HCl. First, the initial pH of each solution was measured. Subsequently, solutions were put in contact with the algae at 120 rpm at room temperature for 1440 min. The solutions were decanted, and the pH values were measured using a Hanna instruments (model HI2020-01) pH meter.
Fourier-transform infrared spectroscopy
Fourier-transform infrared (FTIR)
Scanning electron microscopy and EDS
The surface morphologies of the raw (FV) and modified seaweed (FV-FORMALD) were observed by scanning electron microscopy (SEM) using a JEOL JSM-IT300 electron microscope operating at 20 kV, and the superficial composition was estimated using an EDS system coupled to the electron microscope.
Brunauer–Emmett–Teller surface area (SBET)
Brunauer–Emmett–Teller (BET) surface area and pore size were measured in a Nova 2200e equipment (Quantachrome). FV-FORMALD was pretreated under vacuum at 393 K for 16 h. The adsorption isotherm of N2 onto FV-FORMALD was performed at 77 K. The BET theory and the BJH method were used to calculate the surface area and the pore size.
Sorption studies
Effect of contact time
Adsorption kinetics of both dyes (MB and BCB) was performed as follows: 0.01 g of FV-FORMALD and 10 mL aliquot of dye solution (1000 mg L−1) were contacted for different time's intervals (5–1440 min) in a rotating incubator at 150 rpm at room temperature. After each contacted time, the samples were centrifuged and analyzed by UV-Vis spectrophotometry (Thermo Scientific, Evolution 220) at a wavelength of 664 and 635 nm for MB and BCB, respectively.
Effect of dye concentration
To study the effect of the initial dye concentration on the adsorption capacity of the algae, different MB and BCB dye concentrations were produced from 100 to 1000 mg L−1. Then, 0.01 g of FV-FORMALD samples were added to 10 mL of each dye solution and kept in contact for 360 min, under the same conditions mentioned above.
Effect of pH
The initial pH medium's effect on MB and BCB adsorption was explored in the range of 2–8 for both dyes. Different dye concentrations were prepared (100–1000 mg L−1). pH was adjusted by adding 0.1 M HCl or 0.1 M NaOH. The conditions were the same as for the sorption isotherm study.
Adsorbent concentration effect
The effect of FV-FORMALD algae concentration on the dye sorption was evaluated. An aliquot of 10 mL of each dye (1000 mg L−1) was put in contact with the following amounts of biomass: 1 g L−1 to 10 g L−1 for 360 min, at room temperature, and pH = 4 for both dyes.
Effect of temperature
A volume of 30 mL of each dye at different concentrations (100–1000 mg L−1) was placed in contact with 0.03 g of biomass for 360 min, 150 rpm agitation, and at the following temperatures: 300.15, 313.15, and 330.15 K.
Kinetic and isotherm models
The adsorption capacity (





The equilibrium data obtained from concentration, pH, and temperature effects mentioned above were fitted to the linear and non-linear Langmuir and Freundlich models. For linear form of Langmuir isotherm (equation (7)), the parameters were obtained from the intercept and slope of the plot of 



where
Error functions analysis
In order to confirm the accuracy of the measurement results and a good fit of the experimental adsorption data with the proposed kinetic and isotherm models, four error functions such as the average percentage error (ARE), nonlinear chi-square test (χ2) (Al-Ghouti and Da’ana, 2020), residual root mean square error (RMSE), normalized standard deviation (Δ(%)) (Rahman and Sathasivam, 2015) were calculated with the equations given below:

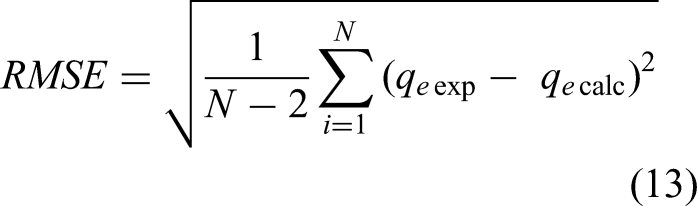

where
Reusability study
The reuse of the FV-FORMALD biosorbent and the recovery of MB and BCB are very important to assess the sustainability and cost-effectiveness of adsorbent. For this purpose, three adsorption/desorption cycles were carried. Many researchers have used HCl as the main eluent of MB dye (Hidayat et al., 2022; Momina et al., 2020; Tehubijuluw et al., 2021). In this case, 0.1 M HCl solution was used as eluent to desorb MB and BCB dye from the adsorbent surface.
For the adsorption step, 50 mL of each dye at the initial concentration of 1000 mg L−1 was placed in contact with 50 mg of FV-FORMALD and stirred at room temperature for 6 h. The mixture was then centrifuged to obtain the adsorbent, and the residual concentration was measured by UV-vis spectrophotometric analysis. The quantity of MB or BCB adsorbed is calculated using equation (1). For desorption process, the loaded FV-FORMALD was added into 50 mL of 0.1 M HCl solution followed by continuous stirring at room temperature for 6 h. Next, the sample was centrifuged and the residual concentration of MB or BCB was measured. The quantity of dyes desorbed 
where
The regenerated FV-FORMALD was washed severally with deionized water and dried at 60°C for 8 h before the next cycle of adsorption took place. Three cycles of consecutive adsorption–desorption studies were carried out.
Real colored wastewater treatment
The waste generated in our experiments was used as real colored wastewater. The sample has a blue color and pH = 2.4. A 10 mL aliquot of the wastewater was placed in contact with 0.01 g of FV-FORMALD at room temperature for 6 h. The sample was scanned in a UV-Vis spectrophotometer from 380 to 750 nm before and after the adsorption process.
Results and discussions
pHPZC
The determination of the adsorbent's zero-point charge is useful because the dye's adsorption depends on the solution's pH. Figure 2 shows that pHPZC = 7.2 for raw

pHPZC determination of raw
Fourier-transform infrared spectroscopy
Fourier-transform infrared spectra of FV-FORMALD were recorded before and after adsorption of MB and BCB within the range of 650–4000 cm−1 (Figure 3(a)–(c)). The vibrational spectra of FV-FORMALD before dye biosorption showed a broadband between the range 3200–3500 cm−1, representing the O-H and -NH groups of the carbohydrates and protein present in the algae. The peak at 3300 cm−1 corresponds to O-H vibrations of cellulose, pectin, and lignin, the peak at 2920 cm−1 represents (C-H) of alkyl groups (Gokulan et al., 2019), a peak around 1600 cm−1 could be attributed to the C=O stretch of the carboxylic or aldehyde functions. The peak at 1025 cm−1 could be related to the C–O stretching vibration of carboxylic acids, alcohol, or phenol. After MB biosorption, the peaks shifted to 3275, 1595, and 1025 cm−1, respectively, suggesting that these functional groups on the surface of the algae interacted with the dye. In addition, two new signals were identified, at 1330 cm−1 (C–N stretching) and at 884 cm−1 (C–H bending of arene rings), which identify the presence of the MB molecule (Salazar et al., 2017). After BCB biosorption, the peaks shifted to 3290, 1590, and 1010 cm−1, respectively, indicating that the hydroxyl, amine, and carboxyl, are the main groups responsible for the biosorption of the two dyes (Table 1).

FTIR of (a) FV-FORMALD (b) FV-FORMALD-MB and (c) FV-FORMALD-BCB.
FTIR bands before and after the adsorption process.

Scanning electron microscopy/EDS
Figure 4 shows the micrographs and respective EDS analysis of raw and treated algae

SEM images and EDS analysis of (a) and (b) raw seaweed
Brunauer–Emmett–Teller surface area (SBET)
The adsorption–desorption isotherm of N2 on the FV-FORMALD sample is shown in Figure 5. As can be seen in this figure, the shape of isotherm allows it to be classified as a type IV isotherm (IUPAC classification), this type of isotherm is characteristic of mesoporous materials (Barton et al., 1999).

Adsorption–desorption isotherm of N2 on the FV-FORMALD.
The results of surface area, pore volume, and mean pore diameter were calculated by BET and BJH methods and are shown in Table 2. The surface area of FV-FORMALD is similar with those of other algae species, slightly higher than of surface of the
Results surface area, pore volume, and mean pore size for different algae materials.

Effect of contact time
Figure 6(a) and (b) depicts the adsorption kinetics of BCB and MB dyes, respectively, by the FV-FORMALD and their modeling with the PFO and PSO order nonlinear equations. It was observed that BCB and MB were rapidly removed during the first 30 min; after this time, the adsorption capacity gradually reached equilibrium; this behavior is due to the that during the initial biosorption phase, dye particles are more present in the solution and the actives sites in algae are unoccupied and available (Silva et al., 2019). Adsorption equilibrium was reached in around 360 min for both dyes.
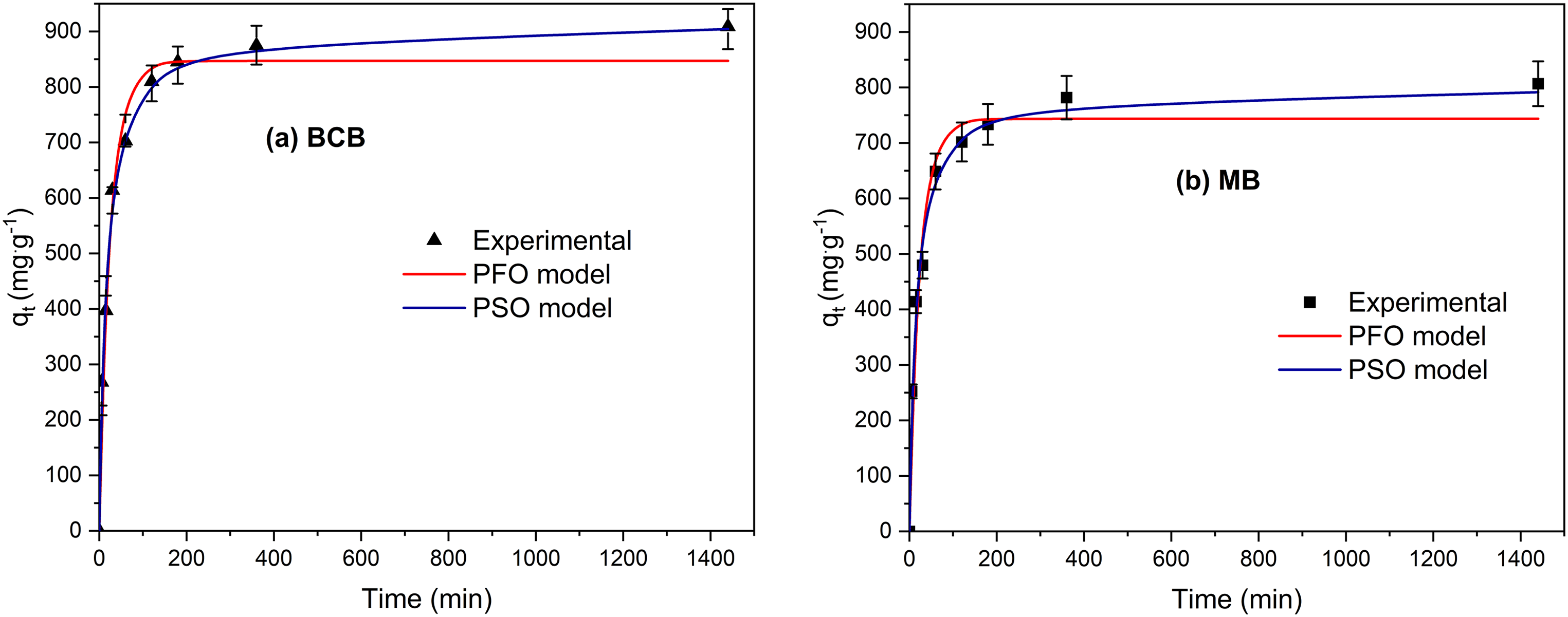
Kinetic modeling of (a) BCB and (b) MB according to pseudo-first order and pseudo-second order nonlinear models by FV-FORMALD. (
Kinetic parameters were calculated utilizing linear and nonlinear PFO and PSO models. Results are depicted in Table 3. For MB, PFO linear and nonlinear models show values of
Kinetic parameters of MB and BCB biosorption onto

Error function data of the kinetic models.

Effect of dye concentration
The isotherm study was performed to describe the interaction between adsorbate and adsorbent surface, calculate the maximum biosorption capacity of adsorbent, and describe the dynamic equilibrium of the adsorption system. Figure 7(a) and (b) shows adsorption isotherms for BCB and MB, respectively, and the fit to Langmuir and Freundlich nonlinear isotherm models. Table 5 presents the parameters of the Langmuir and Freundlich isotherm obtained from their linear and nonlinear forms by fitting the experimental data.
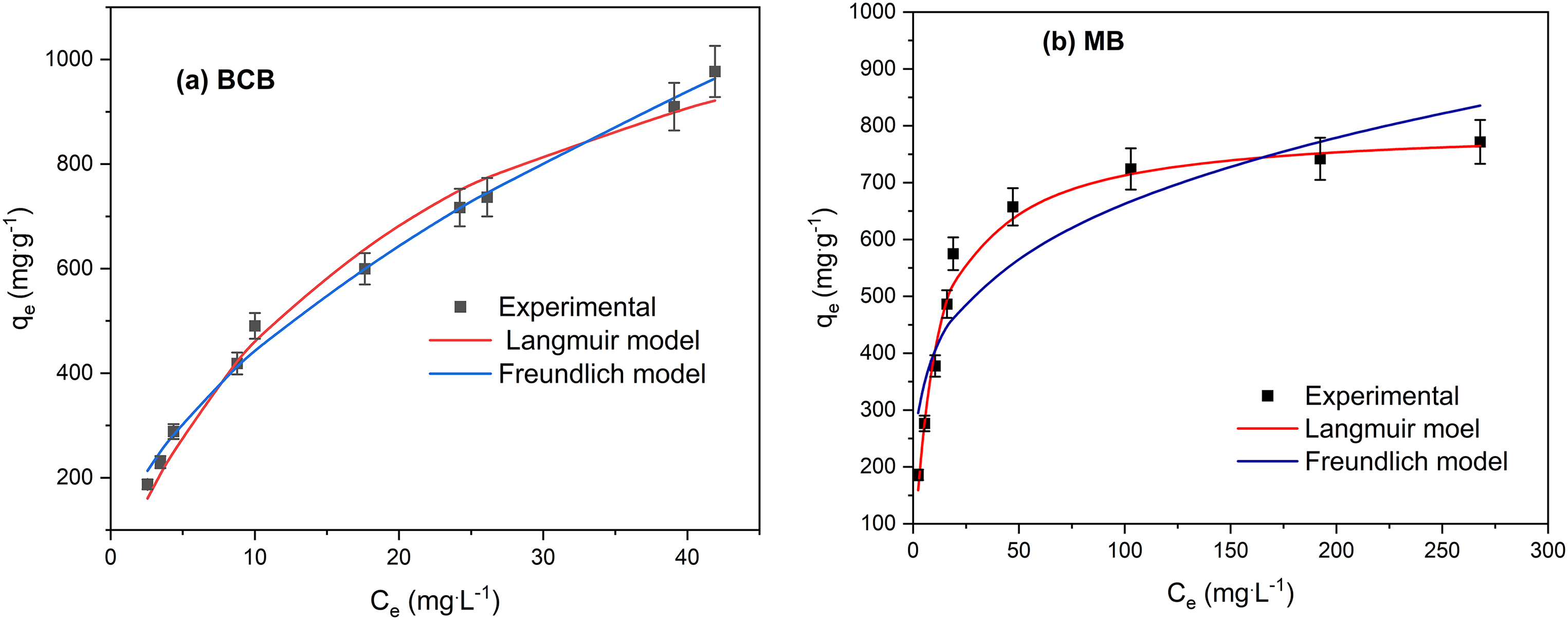
Fitting data for Langmuir and Freundlich nonlinear isotherms for (a) BCB and (b) MB adsorption onto FV-FORMALD (
Isotherm parameters obtained by linear and nonlinear model for methylene blue (MB) and brilliant cresyl blue (BCB) biosorption onto

Error function data of the isotherm models.

For MB adsorption, both, linear and nonlinear forms of the Langmuir isotherm model fitted better than the Freundlich isotherm, given the
For BCB adsorption, as shown in Table 5, the
It can be assumed that both models are suitable to describe the adsorption of BCB dye onto FV-FORMAD. However, the values of the error functions are lower for the nonlinear Freundlich model, suggesting that this model better fits to the adsorption data. The Freundlich isotherm model assumes a heterogeneous surface, reversible adsorption, and multilayer formation (Özcan et al., 2004). Values of the parameter n between 1 and 10 indicate a favorable adsorption process (Lebron et al., 2021). Also,
Based on the BCB and MB isotherms shapes and the results are shown above, it can be assumed that the Langmuir model describes the biosorption of MB onto FV-FORMALD, while Freundlich better describes the biosorption of BCB.
pH effect
Figure 8 shows the adsorption isotherms of BCB and MB fitted by Langmuir and Freundlich isotherms at different pH values. It can be seen that for all pH values, the same isotherm shapes are observed for both dyes.
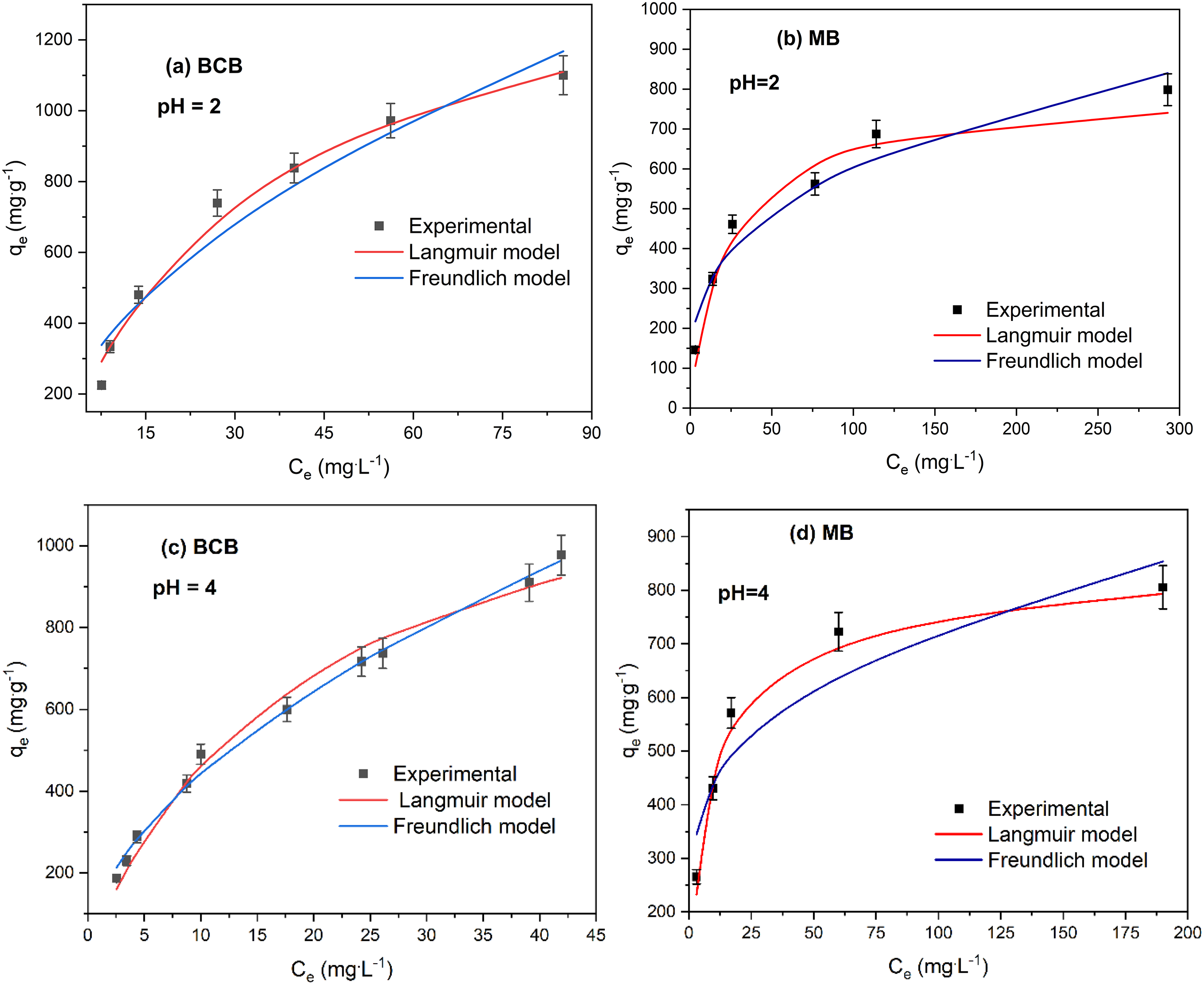
Effect of pH on the modeling of Langmuir and Freundlich nonlinear isotherms for MB and BCB biosorption onto FV-FORMALD (
In the whole range studied (pH 2–8), the MB biosorption capacity increased with increasing solution pH values (Figure 8(b), (d), (e), and (h)), which also means that under low pH conditions, low adsorption capacity of MB was achieved. This occurred when the pH is acidic (low value); the electrostatic repulsion between the positive surface of the adsorbent and the positive charge of the cationic dyes (Figure 1(b)) inhibits dye adsorption, and excess of hydrogen ions can compete with the dye molecules for adsorption sites. Conversely, when the pH increases, the surface of the biosorbent becomes negatively charged due to the deprotonation of the surface (Pang et al., 2017), which facilitates the adsorption of MB. Other authors have reported similar behavior for the adsorption of cationic dyes onto other algae species (Caparkaya and Cavas 2008; Guarín et al. 2018; Liang et al. 2017). In other hand, in Figure 8, the effect of the pH solution on BCB adsorption is also shown. There is a minimal decreased in the adsorption capacity of BCB form pH = 2 to pH = 4 (Figure 8(a) and (c)), it could be said that in the whole studied range, there is no significant difference between the adsorption capacity of BCB by changing the pH of the solutin.
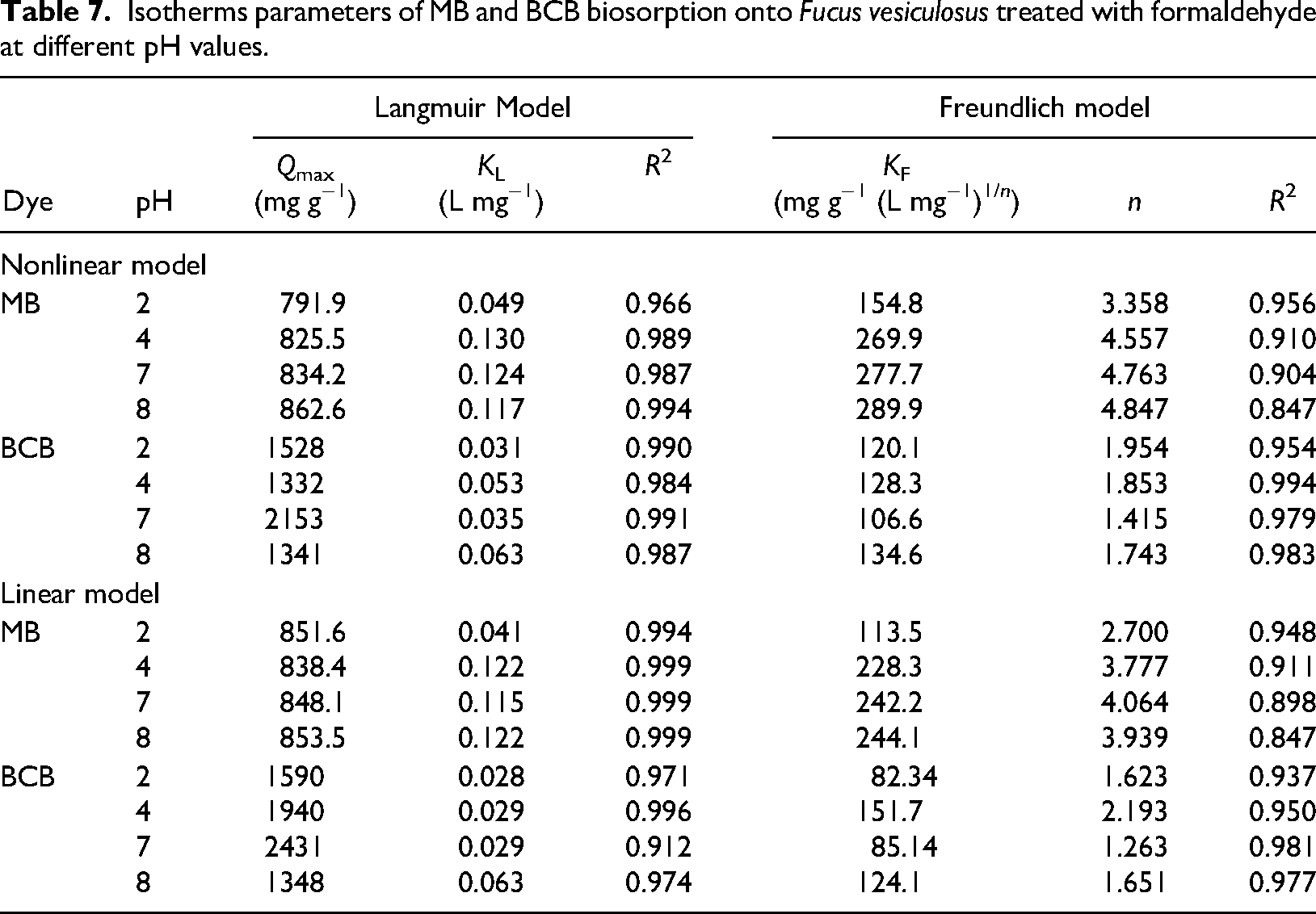
The results of the linear and nonlinear modeling of the Langmuir and Freundlich isotherm models are presented in Table 7. For the linear and nonlinear Langmuir models,
Isotherms parameters of MB and BCB biosorption onto
Error function data of the isotherm models at different pH values.

For BCB biosorption, when the pH increased from 2 to 4,
Adsorbent concentration effect
The effect of the concentration of the brown algae

Effect of FV-FORMALD concentration on the percentage removal of MB and BCB (
Temperature effect
The effect of temperature on the biosorption capacity of MB and BCB onto FV-FORMALD was performed by an isotherm study. The experiments were carried out at 300.15, 313.15, and 330.15 K. As can be seen in Figure 10, the biosorption process of BCB dye continues fitting to both nonlinear isotherm models. However, experimental data are closer to the Freundlich model curve than the Langmuir curve, and the shape of the isotherm is similar for the three values of temperatures (Figure 10(a), (c) and (e)), where no significant change is observed in the adsorption capacity (qe). Figure 10(b), (d), and (f) shows the fitting isotherm curves of MB at 300.15, 313.15, and 330.15 K, respectively. It can be observed that the experimental curves are best fitted by the Langmuir model, with a slight increase in the adsorption capacity with increasing temperature.

Temperature influence on BCB and MB uptake by the brown algae
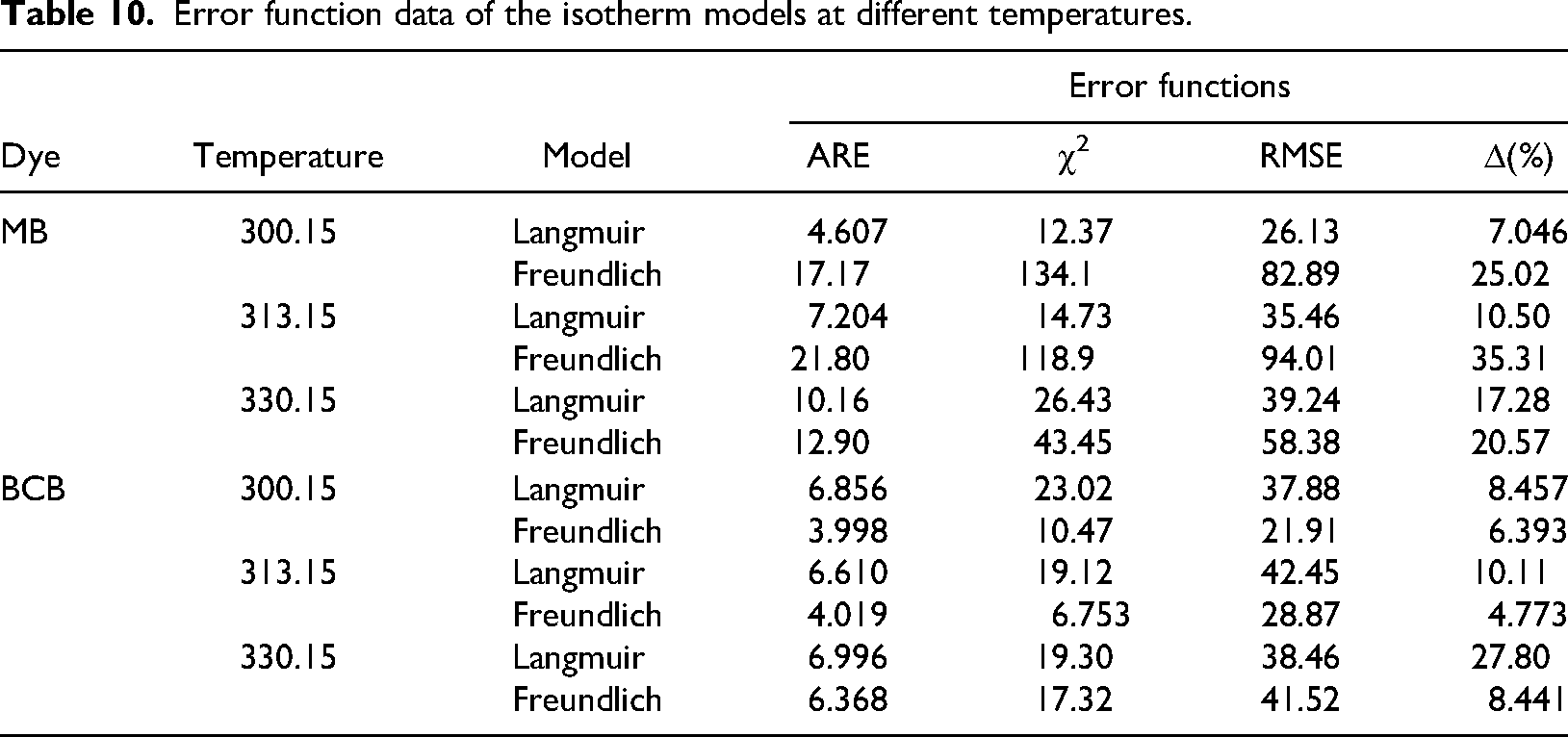
The Langmuir and Freundlich parameters obtained with the linear and nonlinear model for MB and BCB biosorption are shown in Table 9. For BCB adsorption, although the values of
Isotherms parameters of MB and BCB biosorption onto

Error function data of the isotherm models at different temperatures.
Thermodynamic of Mb and BCB adsorption
According to Tran (2022), the constant KL from the Langmuir model, in correct units (Lmol−1), is appropriate for calculating the thermodynamics parameters of the solid-liquids adsorption process. The results obtained from the temperature effect were used to calculate 
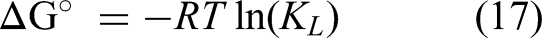
The thermodynamics parameters of the adsorption of both dyes are shown in Table 11. For both dyes, ΔG° presents negatives values for the three studied temperatures, which means that the adsorption of MB and BCB onto FV-FORMALD occurs spontaneously (Al-Ma’abreh et al., 2024). When ΔH° < 0, it indicates that the adsorption process is exothermic in nature (Al-Dalahmeh et al., 2024). ΔH° = −10.873 and −29.322 kJ.mol−1 for MB and BCB, respectively. Therefore, the adsorption of both dye is an exothermic process. Finally, ΔS° is positive for MB dye indicating a higher randomness at the interface of the dye and FV-FORMALD (Medhat et al., 2021). Whereas, for the BCB dye, ΔS° = −10.892 kJ.mol−1K−1, the negative value indicates a lower randomness at the interface of BCB dye and the adsorbent (Egbosiuba et al., 2020).
Thermodynamic parameters of MB and BCB adsorption by

Comparison of the maximum adsorption capacities Q m of MB and BCB onto different adsorbents
Table 12 compares the
Comparison of the maximum adsorption capacity of MB and BCB onto different adsorbents.

Although there are not many studies of the removal of BCB dye. In this study, the
Reusability study
Reuse of adsorbents has been identified as an important parameter for economically and environmental valuable application. The result of the adsorption–desorption cycles of MB onto FV-FORMALD is shown in Figure 11(a). The figure shows that high adsorption capacities (
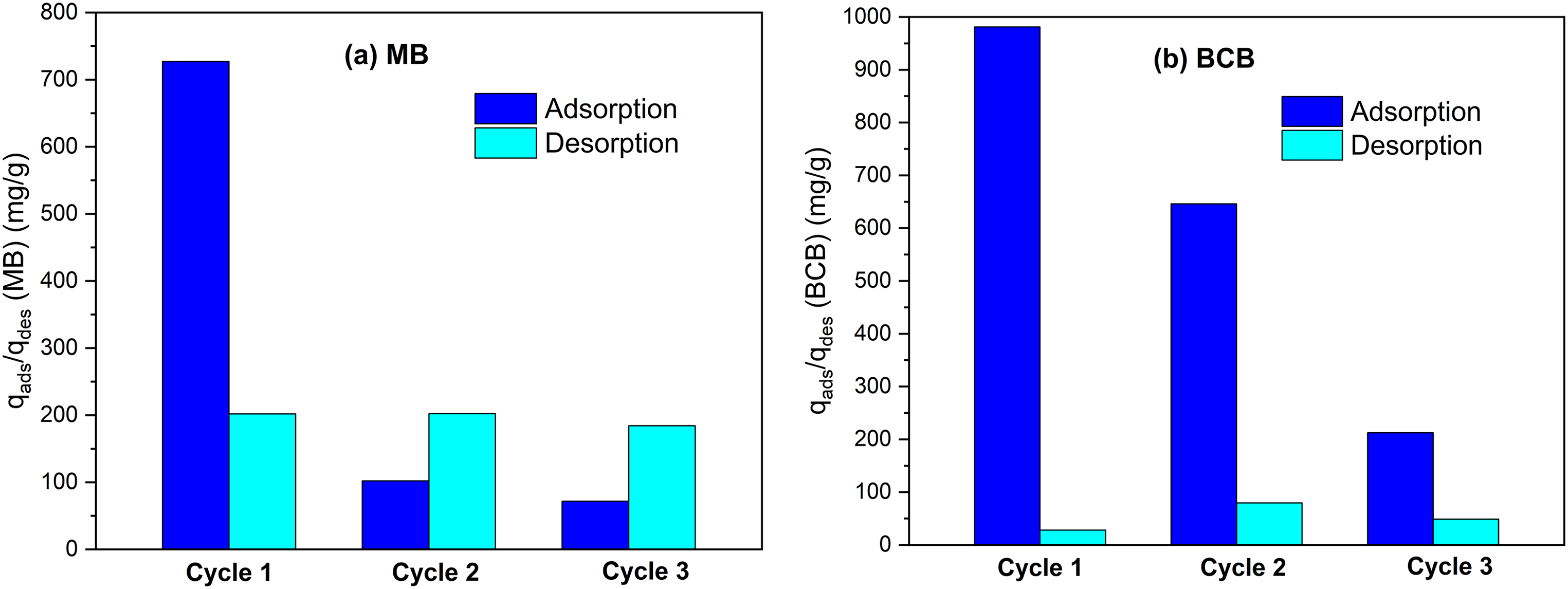
Adsorption/desorption cycles of (a) MB and (b) BCB onto FV-FORMALD using 0.1 M HCl as eluent (
Real colored wastewater treatment
The waste generated from this research was treated with the FV-FORMALD biosorbent. The UV–Visible spectra of wastewater before and after the adsorption is shown in Figure 12. The maximum absorbance of the colored wastewater corresponds at λmax = 655 nm. After adsorption, the absorbance was reduced from 1.129 to 0.089 a.u, indicating a removal efficiency of 92%. According to Sepulveda and Santana (2013), the λmax = 655 nm could be attributed to basic blue 3, another cationic dye. Therefore, it can be assumed that the algae treated with formaldehyde can be used for the treatment of wastewater containing other catatonics dyes such as basic blue 3 efficiently, besides of MB and BCB removal.
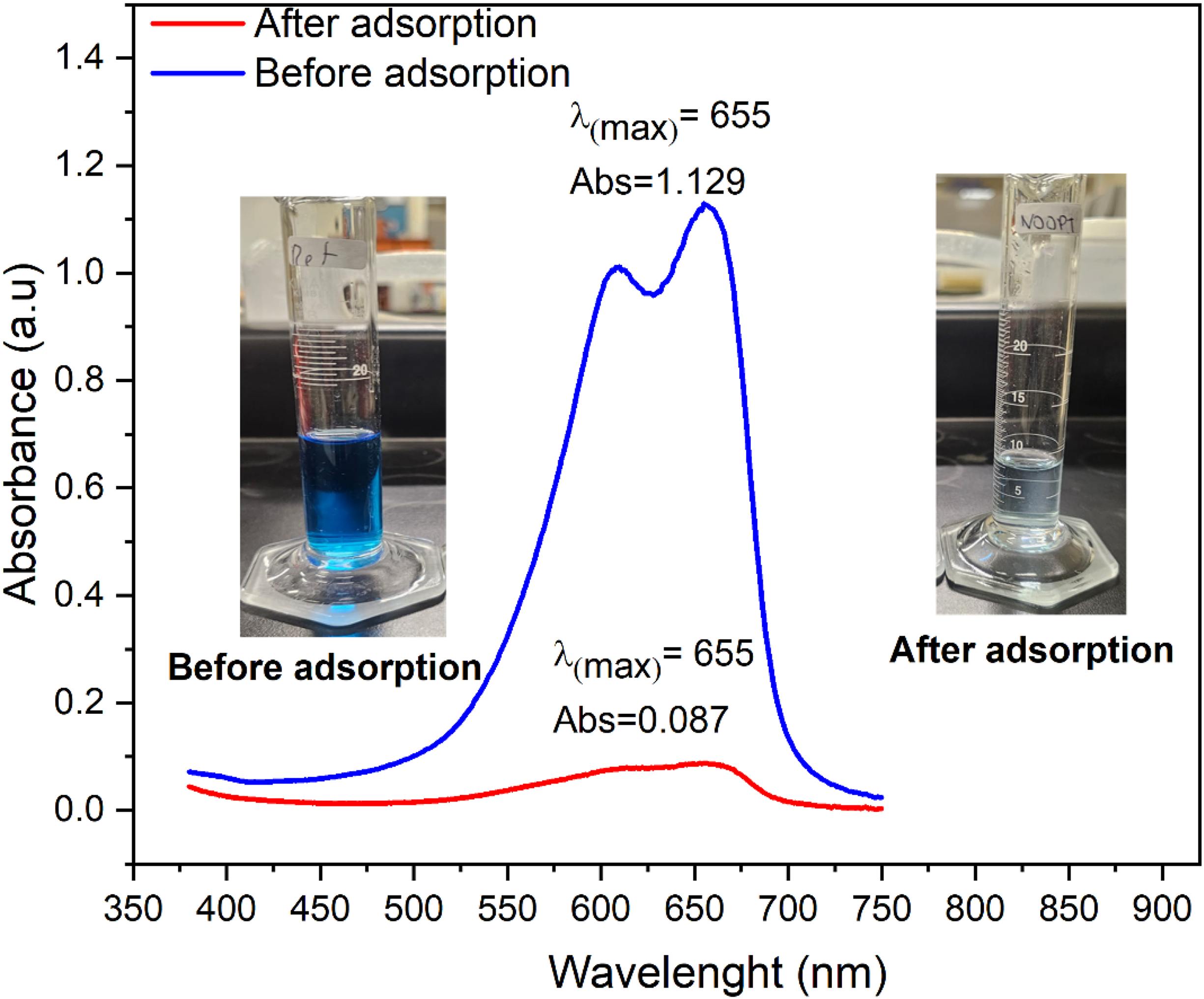
Adsorption of real colored wastewater onto FV-FORMALD.
Conclusions
The brown algae
The fit to the Freundlich model and the adsorption/desorption study suggest a physical mechanism of BCB biosorption. The maximum adsorption (
A chemical adsorption mechanism of MB is suggested as a result of the adjustment to the Langmuir model, the kinetics, FTIR, and the adsorption/desorption results. The maximum adsorption capacity (
The optimal conditions for MB biosorption onto algae were at 313.15 K, adsorbent concentration = 2 g L−1, and pH = 8. While for BCB biosorption, the optimal conditions were at 300.15 K, pH 4, and adsorbent concentration = 1 g L−1. The adsorption of both dyes was exothermic and spontaneous.
Considering the high adsorption capacities of both dyes compared to other biosorbents, its reusability for at least three cycles, its successfully application in the treatment of the real wastewater, its cost–benefit compared to more complex and expensive methods, the brown algae
Footnotes
Acknowledgements
The authors thank the Congreso Nacional de Ciencia y Tecnología (CONACyT) for supporting the national master's scholarship. Rogelio Morán Elvira from IER-UNAM for BET characterization. CONAHCYT CF-2023-I-698, CF 2023-I-775, LNC 2023-33, and DGAPA PAPIIT IT100821.
Credit author statement
All authors have read and agreed to the published version of the manuscript.
Data availability statement
The datasets generated and used to support the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
