Abstract
The study of oil sorbents with high oil sorption capacity is of great significance for oil–water separation, especially for oil spillage on seawater. In this work, we report polysulfone, poly(lactic acid), and polysulfone/poly(lactic acid) nanoporous fibrous mats prepared by one-step electrospinning process used as oil sorbents for oil–water separation. The morphology, porous structure, oleophilicity and hydrophobicity, oil sorption capacity, and oil retention of these nanoporous fibrous mats are investigated in detail. It is noticeable that the motor oil and linseed oil sorption capacity of polysulfone and polysulfone/poly(lactic acid) fibrous mats have little difference, but the motor oil sorption capacity (147.8 g g−1) of polysulfone is approximately 2.95 times as much as that (50.1 g g−1) of poly(lactic acid). The linseed oil sorption capacity (131.3 g g−1) is approximately 3.74 times than that (34.6 g g−1) of poly(lactic acid). Besides, they all exhibit quick oil sorption speed and high buoyancy. It is suggested that polysulfone and polysulfone/poly(lactic acid) nanoporous fibrous mats have a better oil sorption capacity than poly(lactic acid) nanoporous fibrous mat, which is contributed to volume expansion, highly micrometer porous structure among fibers, and specific surface area of fiber.
Keywords
Introduction
Oil spill accidents during oil transportation and storage will cause huge environment pollution, especially occurrence of oil spill at sea. The spilled oil not only results in loss of energy, but also affects huge population of marine birds and shellfish. Therefore, oil spill cleanup has become an urgent problem to solve. There are many methods for treating oil spills, such as mechanical collection (Add and Keller, 2006), in situ burning (Buist et al., 2011), bioremediation (Prince et al., 2013), dispersion (Roulia et al., 2003), and use of sorption materials (Wang and Lin, 2013). Among these methods, using oil sorption materials is one of the most popular methods for its efficiency, economy, and easy oil recovery.
An ideal oil sorbent material should have oleophilicity and hydrophobicity, high oil sorption capacity, good oil–water selectivity, good oil retention ability, and buoyancy. (Li et al., 2014; Pakravan et al., 2012; Yoshida et al., 2015) Currently, organic synthetic materials, inorganic mineral products, and organic natural materials are the main three types of oil sorbent materials (Choi and Moreau, 1993). Among of these, nonwoven polypropylene (PP) fibrous mat has been widely used as oil sorbent due to their oleophilicity and hydrophobicity, and good oil/water selectivity (Rengasamy et al., 2011). Many research showed that the porosity, pore size among fibers, and the voids in the interior of fibers have great effect on oil sorption capacity of fibrous sorbent (Abdullah et al., 2010; Wang et al., 2012). However, the pore size among fibers of fibrous sorbent is related to the fiber diameter. Large fiber diameter results in large pore size among fibers and low oil sorption capacity (Choi and Cloud, 1992). Therefore, it is important to make a fibrous sorbent which has small fiber diameter and high porosity.
Electrospinning is an effective technique for producing fibers with diameters ranging from micrometer to nanometer (Reneker and Chun, 1996; Wu et al., 2012). The electrospun fibrous mats have high porosity and small pore size among fibers due to their micrometer and nanometer scale fiber diameter, which make them have better oil sorption capacity and oil-water selectivity than conventional PP fibers (Lin et al., 2012b, 2012a). Zhu et al. (2011) first reported electrospun fibrous mats used as fibrous sorbent for oil sorption with an oil sorption capacity of 146 g g−1 which was five times higher than commercial PP nonwoven fabric. After that there are many reports about electrospun fibrous sorbents for oil sorption. They mainly focus on the properties of electrospun fibrous mats for oil sorption capacity, such as surface morphology, pore size among fibers, porosity, diameter of fibers, surface tension, and oleophilicity–hydrophobicity (Alazab et al., 2016; Antonio et al., 2014; Li et al., 2015; Zhang et al., 2016). But the property of electrospun fibrous mats for oil retention ability has rarely been reported. High oil retention can keep oil encapsulated in the sorbent and is helpful for transferring the oil from the pollution area.
In the current study, the main objectives were to investigate the use of electrospun nanoporous fibrous mats made of polysulfone (PSF) and poly(lactic acid) (PLA) as oil sorbents. The effect of the fiber diameter and the pore structure of electrospun nanoporous fibrous mats on the oil sorption capacity and oil retention capacity are, respectively, evaluated.
Materials and methods
Materials
PLA (Wm=1.0 × 105) was purchased from Zhejiang Hai Zheng Biological Materials Co., Ltd, Zhejiang, China. PSF (Wm=75,000), dichloromethane (DCM), and N,N-dimethylacetamide (DMAC) were all purchased from Sigma-Aldrich, Inc., New Jersey, USA. All of the chemicals were of analytical grade and used without further purification.
Electrospinning
PSF was dissolved in a binary solvent of DMAC/DCM (with weight ratio of 9:1) at room temperature for 4 h stirring to obtain 20 wt% solution. PLA and PSF/PLA (with weight ratio of 1:1), respectively, were dissolved in a binary solvent of DCM/DMAC (with weight ratio of 9:1) at room temperature for 4 h stirring to obtain 8 wt% solutions. Each solution was loaded into a 5 ml syringe and discharged at a collector with a rotation speed of 6.2 m min−1. PSF, PSF/PLA, and PLA nanoporous fibrous mats were electrospun on the drum located 10 cm in front of the needle tip (0.6 mm inner diameter) with 1 ml h−1 flow rate and a 13 kV voltage was provided. All the experiments were carried out at 20°C. The PSF nanoporous fibrous mats were electrospun at (45 ± 5)%, but the PSF/PLA and PLA nanoporous fibrous mats were electrospun at (65 ± 5)%.
Characterization
The morphologies of the electrospun fibers were examined by a field emission scanning electron microscope (S-4800, Hitachi Ltd, Japan). The fiber diameter was measured by Image processing software (Image-Pro Plus 5.0).
The Brunauer–Emmett–Teller surface area and pore width distribution of the fibers were measured at 77 K by a nitrogen adsorption–desorption Micromeritics ASAP-2020 analyzer (Micromeritics Co., USA).
The through-pore size and through-pore size distribution of each fibrous mat was measured by Porometer 3G (Quantachrome Ltd, USA).
The viscosity of the oils was measured with a viscometer C-PTD 180/AIR/QC (Anton Paar Co., Ltd, Austria) at 20°C.
The porosity of all fibrous mats was calculated by the following equation
The wetting properties of static contact angle of the fibrous mats were measured with Krüss DSA 100 (Krüss Company, Germany) apparatus. The volume of droplets used for static contact angle was 6 µl.
Oil sorption test
The oil sorption capacities of electrospun nanoporous fibrous mats were tested as following. A 20 ml of oil was poured into a beaker containing 200 ml water. Approximately 0.1 g of mat was then placed onto the oil surface. After every interval time, the oil-containing mats were removed and weighted to access their sorption capacities after natural draining for 20 s. The oil sorption capacities of mats were obtained from the following equation
Oil retention test
After the mats adsorbed oil for 1 h, the oil retention of mats was tested by weighting the weight of oil natural draining from the mats at every interval time. The oil retention of mats was obtained from the following equation
Results and discussion
Morphology and pore structure of nanoporous fibrous mats
Figure 1 shows the photographs of nanoporous fibrous mats. As can be seen, highly porous structure on the surface of PSF/PLA and PLA fibers (Figure 1(d) and (f)) is clearly observed, but the pores on the surface of PSF fibers are not obvious (Figure 1(b)). The average diameters of the PSF, PSF/PLA, and PLA fibers are 496 ± 155.7, 1029 ± 232.6, and 1137 ± 322.1 nm, respectively. The pore formation could be owing to phase separation and breath figure induced by the rapid evaporation of the solvent (Casper et al., 2004; Fan et al., 2017; Liu and He, 2017; Qi et al., 2009). The high volatile solvent in the polymer fluid jet evaporates into the air rapidly during electrospinning resulting in the surface temperature of the jet decrease rapidly, which causes the water vapor around the jet condense into water droplets on the jet surface. Then the pore formed after the water droplets evaporated. This pore formation mechanism is called breath figure. In addition, the solvent volatiles from inside to the surface of the jet, which result in a fast phase separation induced by thermal instability of the jet and then yields a polymer-rich phase and a solvent-rich phase. After the solvent diffuses out, the concentrated polymer-rich phase solidifies into the matrix and the solvent-rich phase is converted into the pores. Therefore, the more volatile the solvent, the more likely to produce thermal phase separation and breath figure, which result in more obvious porous structure. In this research, the DCM and DMAC blending solvent was used to prepared PLA and PSF electrospinning solution. The boiling point (40°C) of DCM is much lower than DMAC (166°C), so that the more ratio of DCM in the blending solvent, the porous structure on the surface of electrospun fibers is more obvious. The ratio of DCM in PLA solution is much higher than that in PSF solution, which results in obviously intensive porous structure on the surface of PLA fibers and not obviously tiny holes on the surface of PSF fibers.

SEM images of nanoporous fibrous mats: (a) and (b) for PSF, (c) and (d) for PSF/PLA, (e) and (f) for PLA.
Figure 2(a) shows the N2 adsorption–desorption isotherms of the obtained fibers. The significant increase in adsorption under a relative pressure (P/P0) of >0.8 is contributed to the mesopores and macropores within or among the fibers (Chen et al., 2013a, 2013b). PSD curves derived from the BJH method (Figure 2(b)) reveal that the pore sizes of the obtained fibers are in the range of 2–60 nm. The pore structural parameters of the obtained fibers are shown in Table 1. The specific surface areas of the PSF, PSF/PLA, and PLA fibers are 21.331, 13.8176, and 11.6719 m2 g−1, and the total pore volumes are 0.1433, 0.1467, and 0.0743 cm3 g−1, respectively. The average pore width of the obtained fibers is all less than 50 nm. The cumulative pore volume is mainly from the pores of <50 nm as shown in Figure 2(b), namely the mesopores on the surface of fibers, which mainly contributed to the total pore volume.

(a) Nitrogen adsorption–desorption isotherms of obtained fibers and (b) pore size distribution derived from BJH method. PLA: poly(lactic acid); PSF: polysulfone.
Pore structure of nanoporous fibers testing by BET method.

BET: Brunauer–Emmett–Teller; PLA: poly(lactic acid); PSF: polysulfone.
Fibers were deposited directly on a grounded metallic rotating roller to form PSF, PSF/PLA, and PLA nanoporous fibrous mats. The pore size descriptions between fibers of these mats were measured by a manually operated pore size analyzer, as shown in Figure 3, and the mean flow pore sizes of these mats were 1.9, 2.8, and 3.66 µm, respectively, as shown in Table 2. It is well known that the pore size of fibrous mat is related to the fiber diameter and packing density. With the fiber diameter increased, the pore size of samples increased, but the porosity decreased. It can be seen from Table 2, among the three kinds of samples, the PSF has the smallest pore size and the highest porosity (84.6%). The PLA has the biggest pore size and the lowest porosity (73.8%). However, all of them have micrometer scale pore size and high porosity due to the sub/micro-meter diameter.

Through-pore size distributions of nanoporous fibrous mats. PLA: poly(lactic acid); PSF: polysulfone.
Structure parameters of nanoporous fibrous mats.

PLA: poly(lactic acid); PSF: polysulfone.
Oleophilicity and hydrophobicity of nanoporous fibrous mats
On one hand, the oleophilicity and hydrophobicity of nanoporous fibrous mats affect the oil removal efficiency. On the other hand, it is easier to collect fibrous sorbents with oleophilic and hydrophobic characteristics during oil spills cleanup operation because they can be disposed appropriately in order to avoid second surrounding contamination. Therefore, the oleophilic and hydrophobic properties of electrospun fibrous mats are investigated. The contact angles for water on the PSF, PSF/PLA, and PLA fibrous mats are similar which are (130.3 ± 1.2)°, (132.8 ± 1.5)°, and (131.6 ± 0.8)°, respectively, and for oil are all 0°, as shown in Figure 4. It is suggested that these samples all showed good and similar hydrophobicity. Because of PSF and PLA are all hydrophobic and oleophilic macromolecular polymers, and all have mesopores on the surface of fibers or the micrometer scale through pores among the fibers. The porous structure traps a large number of air within and among the fibers, which result in the surface free energy of fibrous mats reduced. So that all these fibrous mats exhibit good hydrophobicity by minimizing the interfacial tension which results in the water droplets keeping spherical shape (Wu et al., 2012). Therefore, these fibrous mats could be a potential oil sorption material due to their excellent hydrophobicity and oleophilicity.

Contact angle of nanoporous fibrous mats: (a) for PSF, (b) for PSF/PLA, (c) for PSF/PLA, (d) photograph of water and oil droplets on the nanoporous fibrous mats.
Oil sorption capacity and oil retention of nanoporous fibrous mats
The water sorption of PSF, PSF/PLA, and PLA fibrous mats can be neglected because of their high hydrophobicity. To evaluate the oil sorption capacity and oil–water selectivity, each sample (0.1 g) was dropped into the as-prepared artificial oil–water bath, and then weighed after draining for 20 s at every interval time until 60 min. To investigate the effect of high porous structure on the oil sorption capacity and oil retention of the PSF, PSF/PLA and PLA fibrous mats for motor oil and linseed oil are measured and shown in Figure 5. The sorption capacities of the PSF, PSF/PLA, and PLA fibrous mats for motor oil are 147.8, 119.8, and 50.1 g g−1, respectively. The linseed oil sorption capacities of these mats indicate a similar tendency that is 131.3, 127.9, and 34.6 g g−1, respectively. Obviously, the oil sorption capacities of PSF and PSF/PLA are similar and are much more than that of PLA. It is noticeable that the motor oil sorption capacity of PSF is approximately 2.95 times as much as that of PLA, while its linseed oil sorption capacity is approximately 3.74 times than that of PLA. Even if the PLA fibers have high porous structure, the oil sorption of PLA fibrous mats is the lowest.

Oil sorption capacities of nanoporous fibrous mats for 1 h. PLA: poly(lactic acid); PSF: polysulfone.
Oil sorption mechanism for a fibrous porous sorbent is controlled significantly by adsorption and capillary action (Singh et al., 2013; Zhu et al., 2011). The oil was adsorbed by physical trapping on the fiber surface due to the adhesion of atom, ions, or molecules of oil to a surface, thus creating an oil film on the fiber surface. Subsequently, the oil fills the voids among the fibers, and then diffuses into the internal pore structure of the fibers by means of capillary (Choi and Cloud, 1992; Choi and Moreau, 1993). Capillary action is a surface phenomenon wherein a liquid flows into narrow spaces without any assistance. Therefore, the oil sorption capacity of the fibrous sorbents mainly depends on oil properties, fiber properties, and voids within and among fibers (Sarbatly et al., 2016; Singh et al., 2013). The PSF fibrous mat has high oleophilicity, the biggest specific surface of fibers and smallest through-pore size due to its sub-micrometer scale diameter, which result in excellent oil sorption capacity. However, the PLA fiber has porous structure, but the pore size is less than 60 nm, thus the oil hardly enter into the intra-fiber pores by means of capillary due to their high viscosity, therefore its oil sorption capacity does not increase. On the other hand, the specific surface area of PLA fiber is lowest and the through-pore size of PLA fibrous mat is the biggest, which result in relatively weaker adsorption action. Additionally, the PLA fibrous mat has the lowest porosity which reduces the storage volume for adsorbed oil. But the structure of PLA fibrous mat is tight so that its volume cannot be expanded when the oil is adsorbed into the voids, as shown in Figure 6(c) and (f). Therefore, the PLA has the lowest oil sorption capacity. Although the porosity of PSF/PLA fibrous mat is lower than that of PSF, the volume expanded obviously after contact with oil for 1 h, as shown in Figure 6(e), which results in relative high oil sorption capacity. Once the oil fills into the voids among the fibers, the space not only in the through-pores from up to under of the mats but also between the fiber layers is enlarged, due to the weak force between fibers is easily broken, which result in volume of mats expanded, as shown in Figure 6(a) to (e), and the oil sorption capacity increasing. It is suggested that the volume expansion of the fibrous sorbents has great effect on oil sorption capacity.

Photographs of the nanoporous fibrous mats before and after contact with oil for 60 min: (a) and (d) for PLA, (b) and (e) for PSF/PLA, (c) and (f) for PLA, (g) represents oil–water separation of samples for 60 min.
Figure 5 shows the tendency of maximum oil sorption capacity of PSF and PLA that is motor oil > linseed oil. It can be seen from Table 3 that the viscosity of motor oil is larger than linseed oil. With increasing of oil viscosity, the oil can be more likely to adhere to the surface and voids among the fibers, which make the adsorbed oil remain in rather than flow out from the voids among fibers in the process of draining. Figure 7 shows the oil sorption capacities of nanoporous fibrous mats changed with contact time. It can be obviously observed that the oil sorption capacities of PSF and PSF/PLA increase with increasing contact time, especially in the initial 10 min. But the oil sorption capacity of PLA is almost the same after 10 min. In the initial 10 min, the rapid sorption capacity is mainly produced by adsorption and capillary action, but the volume expansion of sorbents has important effect to oil sorption capacity after 10 min. It is consistent with the oil-sorption mechanism which has been analyzed above. After 60 min, all the fibrous mats are almost immersed in the oil layer, forming a down-heave below the oil–water interface, as shown in Figure 6(g), which exhibits high buoyancy and good oil–water separation property due to the low volume density, good oleophilic and hydrophobic property, and high porosity.

Oil sorption capacities of nanoporous fibrous mats versus contact time.
Properties of the studied oils at room temperature (20°C).
High oil retention ability is important to keep oil encapsulated in the sorbent so that the sorbent can be relocated from the water to a designated area without losing the oil into the surrounding so that it avoids second contaminant. To investigate the oil retention of the nanoporous fibrous mats, the oil retention of PSF, PSF/PLA, and PLA for motor oil and linseed oil during natural draining was measured, as shown in Figure 8. It can be seen that the oil retention quickly decreases in the initial 10 min for all samples, and the decreasing rate slows down after 10 min, especially for PLA decreases slightly from 10 to 60 min. This tendency is almost opposite to the oil sorption capacity as discussed above. Sixty minutes later, the oil retention of PSF, PSF/PLA, and PLA decreases by 42.04, 38.02, and 53.69%, respectively, for motor oil, and decreases by 62.17, 56.42, and 50.61%, respectively, for linseed oil, which means almost half parts of adsorbed linseed oil are lost. But the PSF and PSF/PLA have relative better oil retention for motor oil. It is suggested that the high porosity of nanoporous fibrous mats is not beneficial to oil retention, but the higher oil viscosity brings larger oil retention.
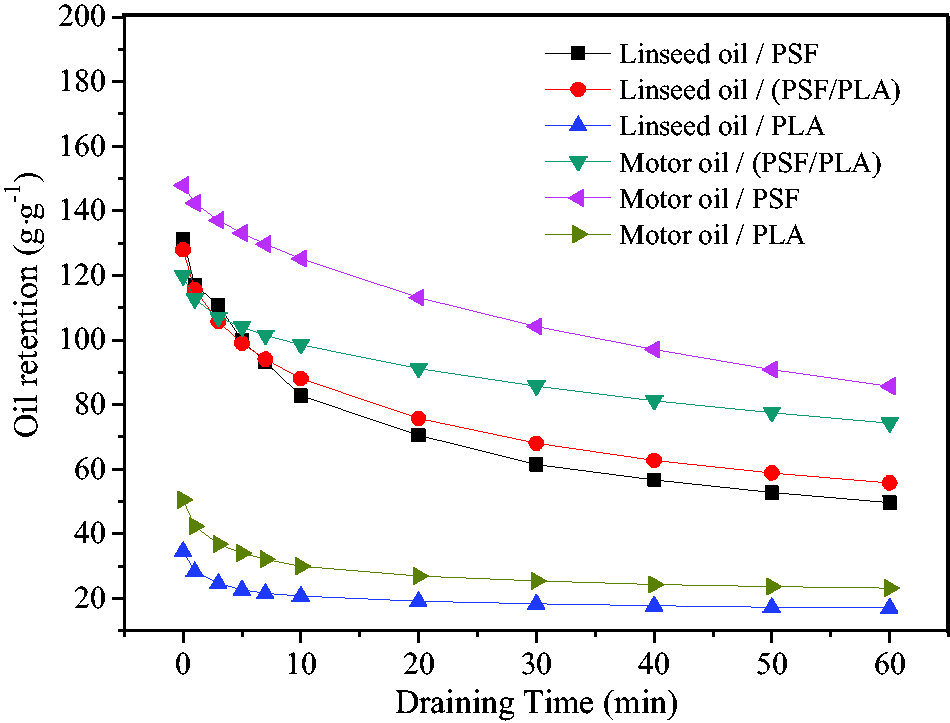
Oil retention of nanoporous fibrous mats during natural draining.
Conclusion
The PSF, PLA, and PSF/PLA nanoporous fibrous mats were prepared by one-step electrospinning process at room temperature. It is noticeable that the motor oil and linseed oil sorption capacities of PSF and PSF/PLA have small difference, but the motor oil sorption capacity (147.8 g g−1) of PSF is approximately 2.95 times as much as that (50.1 g g−1) of PLA. The linseed oil sorption capacity (131.3 g g−1) is approximately 3.74 times than that (34.6 g g−1) of PLA. Besides, they all exhibit quick oil sorption speed and good oil–water separation property as well as high buoyancy. It is suggested that the oil sorption capacity is a result of chemical composition of fibers, high micrometer porous structure among fibers, and specific surface area which relates to fiber diameter. In addition, the volume expansion of sorbents has a great effect to oil sorption capacity. But the mesoporous on the surface of fibers is not valid for increasing oil sorption capacity. The high porosity is not beneficial to oil retention, whereas the higher oil viscosity brings larger oil retention. Therefore, the PSF and PSF/PLA nanoporous fibrous mats would be a good oil sorbent for removing oil from water. However, the range of pore size on the surface of fiber which has effect to oil sorption will be studied in our next work.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding of the financial support from the Science and Technology Project of Jiangsu Province and Suzhou City in China (BE2017676, SS201725, SYG201827), Jiangsu Qinlan Project in China, and National Natural Science Foundation of China (11372205).



