Abstract
With the rapid development of the chemical industry, oil–water emulsion separation has received increasing attention worldwide. In this study, waste wool was used as the raw material. The ratios of polyacrylonitrile and wool keratin were first adjusted to control the fiber diameter of the membrane. Then, micro-nanostructures were constructed on the membrane surface by adding SiO2 and zeolitic imidazolate framework-8. The zeolitic imidazolate framework-8 on the fiber surface could absorb tiny oil droplets to achieve efficient separation of oil–water emulsions. The prepared membrane has a high flux (>390 L m−2 h−1), high separation efficiency (>95%), good chemical resistance, and reusability. The water contact angle and oil contact angle are 0° and the underwater oil contact angle is 143° of the PAN/wool keratin/SiO2/ZIF-8 membrane. This research can be used in the study of fiber membrane and oil–water separation.
Introduction
Water is an important and indispensable resource for human life and the ecological environment. However, due to the rapid development of the social economy and chemical industry, much oil and industrial wastewater are discharged into nature, which causes waste and pollution of water resources, creates serious ecological and environmental problems, and even affects the life and health of human beings. How to separate the oil–water mixtures to obtain pure water resources has become an urgent problem to solve. 1 To solve these problems, traditional methods include gas separation, gravity separation, 2 centrifugal separation, electric separation, and absorption separation, which have the disadvantages of high input, high energy consumption, and low separation efficiency, and can easily cause secondary pollution.3–5 Membrane separation technology is regarded as an efficient method in oil–water separation owing to its advantages of low energy consumption, simple operation, and high efficiency. 6 Among the many methods of preparing membranes, the electrospinning method is capable of preparing nanofibers with controlled diameters,7–12 and the nonwoven structure formed by nanofibers can bring a large number of pores and a high specific surface area.13,14 Oil–water separation membranes are divided into hydrophobic and hydrophilic separation membranes. 15 Highly hydrophobic composite nanofiber membranes are used effectively for the separation of oil–water mixtures. 16 Usually, hydrophobic separation membranes choose to pass oil during the separation process and the absorbed oil tends to contaminate or even clog the membrane, leading to a decrease in separation capacity. 17 However, the density of water is typically greater than that of oil. Hydrophilic separation membranes can form a water barrier between the water and the membrane, thus achieving the effect of retaining oil droplets and overcoming the disadvantages of hydrophobic membranes.18,19 Therefore, it is very important to fabricate a super-hydrophilic/underwater oleophobic membrane for the efficient separation of oil–water emulsions with a low cost, simple preparation, and which is environment-friendly.
Wool keratin (WK) is a renewable and green source with excellent biocompatibility and biodegradability; non-toxic and environmentally friendly, and contains a large number of hydrophilic groups such as amino and hydroxyl groups, which is beneficial to improve the hydrophilicity of as-prepared materials. Keratin nanofiber membranes for oil–water separation have rarely been reported. Polyacrylonitrile (PAN) is often used in electrospinning to produce nanofibers because of its good spinnability and high resistance to chemical reagents.20,21 The addition of SiO2 nanoparticles to the spinning solution enhances the hydrophilicity of the material. Zeolitic Imidazolate Framework-8 (ZIF-8), a type of metal–organic framework (MOF) material, has high porosity, large specific surface area, low density, and tunable structural and chemical properties, and is also used in oil–water separation membranes due to its excellent hydrothermal stability and ease of preparation. ZIF-8 is inherently oleophilic and the fact that it protrudes on the surface of the fiber membrane can help to capture the oil droplets.22–24
In this article, we tried to reuse the waste wool and prepare an electrospun membrane for efficient separation of oil and water by combining it with ZIF-8. The as-prepared membranes were characterized by scanning electron micrography (SEM), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), X-ray photoelectron spectroscopy (XPS), the Brunauer–Emmett–Teller (BET) method, and contact angle measurements. PAN/wool keratin/SiO2/ZIF-8 (PWSZ) membranes exhibited excellent super hydrophilic/underwater oleophobicity, high flux (greater than 390 L m−2 h−1), high separation efficiency (greater than 95%), good chemical resistance, and reusability. In conclusion, this simple-to-prepare, environment-friendly, and low-cost fiber membrane has great potential for application in industrial and marine oily wastewater.
Experiment
Reagents
Wool keratin was produced in the laboratory. PAN (Mw = 150,000) and 2-methylimidazole were purchased from Adamas. Zn(NO3)2 6H2O and sodium dodecyl sulfate (SDS) were purchased from Sinopharm Chemical Reagent Co. Ltd (Shanghai, China). SiO2 nanoparticles (hydrophilic, d = 7–40 nm) were purchased from Aladdin Chemical Ltd (Shanghai, China). N, N-Dimethylformamide (DMF) and organic reagents (n-hexane, n-heptane, petroleum ether, kerosene, and dichloroethane) and anhydrous ethanol were purchased from Shanghai Titan Technology Co. Ltd (Shanghai, China). All chemicals were of analytical grade and were used directly without further purification.
Preparation of Keratin Powder
Five grams of waste wool was pulverized after washing and degreasing, then put into 100 mL of an aqueous solution containing urea (8 M) and L-cysteine (0.165 M), adjusted to pH 10.5 with NaOH, and treated by shaking at 75°C for 5 h. After filtration, the dialysate was dialyzed with deionized water using a dialysis bag (molecular weight cut-off 8000–14,000 Da) at room temperature for 3 days, and the deionized water was changed three times a day. Afterward, the pH of the dialysate was adjusted to 4.0–4.5 with acetic acid and a large amount of keratin was precipitated. The mixture was centrifuged and freeze-dried at −60°C for 3 days to obtain keratin powder, which was stored in a refrigerator at 4°C.
Synthesis of ZIF-8 Crystals 25
The ZIF-8 was fabricated based on recent report. 26 0.908 g of Zn (NO3)2 6H2O was added to 100 mL of methanol to form a zinc nitrate hexahydrate solution. A 4.54 g aliquot of 2-methylimidazole was added to 100 mL of methanol to form a 2-methylimidazole solution, which was stirred well at room temperature. Zinc nitrate hexahydrate solution was slowly added dropwise to the 2-methylimidazole solution; the mixture gradually became milky white and was magnetically stirred at room temperature for 12 h. The resulting ZIF-8 was collected by centrifugation (10,000 rp/m, 5 min), washed with anhydrous ethanol three times and dried under vacuum at 60°C overnight.
Preparation of Electrospinning Solution
PAN/WK solution: PAN and WK were slowly added to DMF (10 mL), with 10 wt% or 15 wt% of PAN and WK at a mass ratio of 9:1, 8:2, and 7:3 (e.g. 10 wt% of PAN and WK at mass ratios of 9:1, i.e. 1 g PAN (0.9 g) and WK (0.1 g)/10 mL DMF), respectively. The mixture was stirred at 80°C for 12 h to form a homogeneous solution. The membrane obtained from the preparation was named the PW membrane.
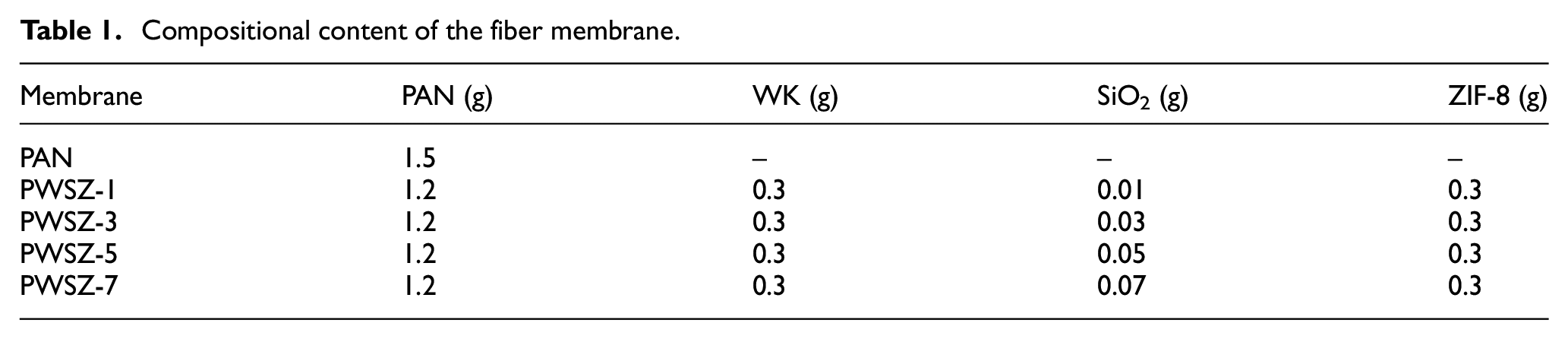
PAN/WK/SiO2/ZIF-8 spinning liquid: PAN and WK were slowly added to DMF (10 mL) and stirred well, after which SiO2 nanoparticles (0.01, 0.03, 0.05, and 0.07 g) were added and the loading amount of ZIF-8 was 30% of spun substrate (PAN and WK). The as-prepared membrane was named PWSZ-x (where x is the content of SiO2, e.g. PWSZ-1 with 0.01 g of SiO2).
Preparation of Fiber Membrane
Different spinning liquids were electrospun onto aluminum foil paper under a positive voltage of 18 kV and a negative voltage of 2 kV, respectively, with a distance of 15 cm from the spinneret to the collector, a flow rate of 1.0 mL/h, a rotational speed of 300 r/min, and a spinning time of 10 h. The spinnerets were used for the spinning of aluminum foil paper. The PWSZ membrane was prepared by conventional electrospinning as shown in Figure 1. The compositional content of the fiber membrane is shown in Table 1.
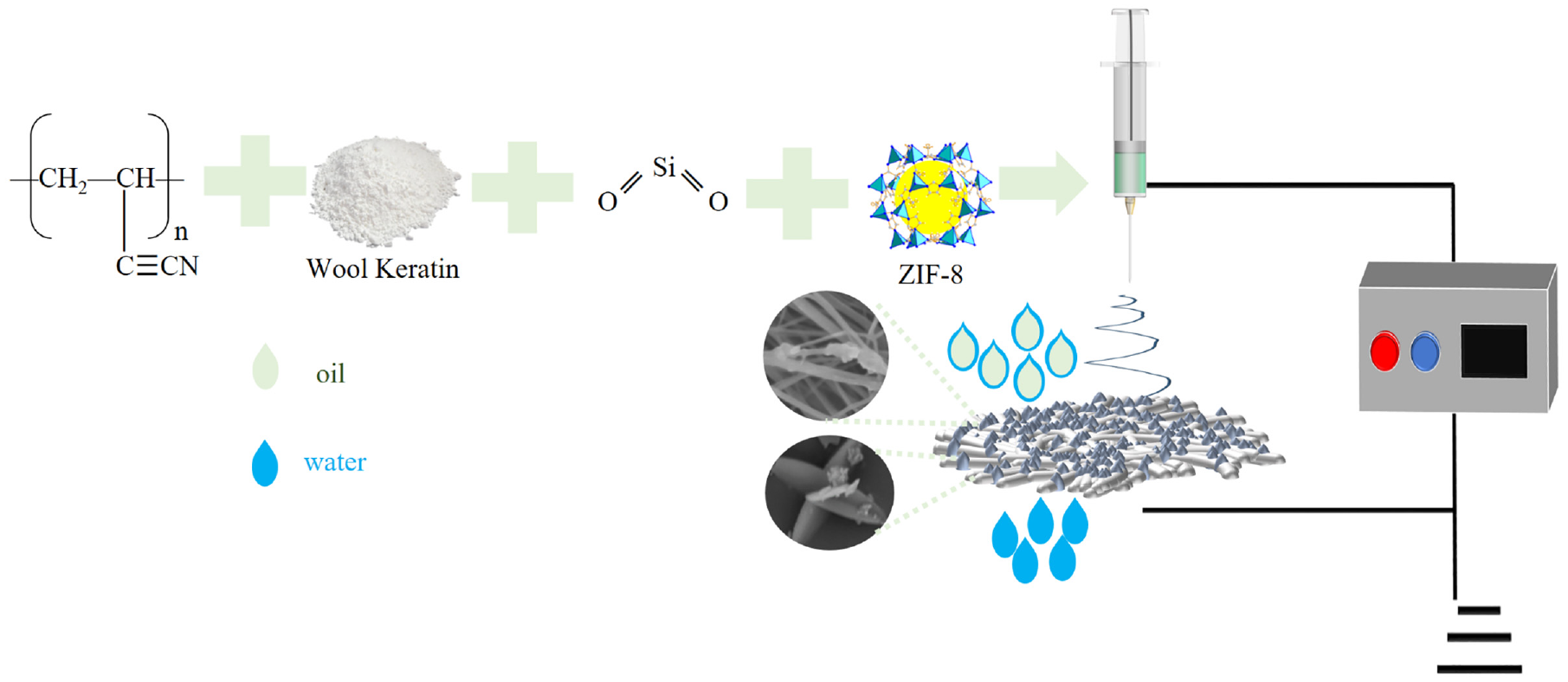
Preparation of PWSZ electrospun fiber membranes and its application in separating oil–water emulsions. Schematic illustration.
Compositional content of the fiber membrane.
Preparation of Oil-in-Water Emulsions
To prepare O/W emulsions containing surfactants, first, 20 mg of SDS was dissolved in 99 mL of deionized water (DI) water, followed by the addition of 1 mL of organic solvent. Subsequently, the resulting solutions were stirred for 10 min with a homogenizer at 12,000 rp/m to obtain a series of surfactant-stabilized oil-in-water emulsions, including n-hexane in water (Hex/W), n-heptane in water (Hep/W), petroleum ether in water (Pe/W), kerosene in water (K/W), and dichloroethane in water (DCM/W).
Oil-in-Water Emulsion Separation Test
The composite membrane was fixed in an oil–water separation unit with an effective area of 12.56 cm2. The sand core in the device was replaced by an experimentally prepared membrane. The diagram of the oil–water separation testing device is shown in Supplemental Figure S2. Different emulsions were poured into the unit to allow free permeation of the composite membrane under gravitational conditions. The membrane was cleaned with ethanol and deionized water after each separation cycle. The membrane flux (J) and separation efficiency (R) were calculated as follows:

Here, V represents the filtrate volume (L), S represents the effective area (m2), and t represents the separation time (h):

Here, m0 (g) represents the total mass of membrane before the separation and m1 (g) represents the total mass of membrane after the separation.
Characterization
The surface morphology and microstructure of the fiber membranes were observed using a scanning electron microscope (TM-3030; Hitachi, Ltd., Japan). The chemical functional groups of the materials were characterized by FTIR spectroscopy (Spectrum Two; PerkinElmer, USA). The contact angles were measured at room temperature using a contact angle analyzer (DSA30; Kruss, Germany), and the average contact angle values were obtained by randomly selecting three positions from the same sample. The crystal structures were characterized using an X-ray diffractometer (Bruker D8, Germany). The specific surface areas of samples were evaluated by the BET method (Autosorb iQ, Quantachrome, USA China), at the temperature of liquid nitrogen (77 K). X-ray photoelectron spectroscopy (XPS Escalab 250 Xi; Thermo Fisher Scientific, USA) was used to characterize the surface elemental composition of PWSZ membranes. The droplet sizes of the emulsion and filtrate were measured using a Malvern Zetasizer Nano ZS (Malvern, UK) instrument using dynamic light scattering (DLS) technology.
Results and Discussion
Morphological Analysis
It can be observed from Figure 2(a)–(f) that WK can be blended with PAN to prepare fiber membranes. A small number of WK particles are attached to the fibers, and all the produced fiber membranes have no bead-like structure and a smooth and continuous surface. Under the condition of the same solute type, the average diameter of the fiber becomes larger as the mass fraction of the solute increases, because the viscosity of the spinning mixing solution increases; the macromolecules within the spinning solution are entangled with each other, which inhibits the traction refinement of the fibers; and a thicker needle is required for spinning.

(a, c, e) Scanning electron micrographs (SEM) image of fiber membranes with PAN and WK mass ratios of 9:1, 8:2, 7:3, PAN and WK mass fractions of 10%, respectively, (b, d, f) SEM image of fiber membranes with PAN and WK mass ratios of 9:1, 8:2, 7:3, and PAN and WK mass fractions of 15%, respectively. (g) SEM image of ZIF-8 crystals, and (h) SEM image of the PWSZ-5 fiber membrane.
Under the condition that the electrospinning solution contains the same mass fraction of solute, the average diameter of the fibers shows a decreasing trend with the increase in the content of WK in the spinning mixing solution, because when the content of keratin is higher, the viscosity of the solution is smaller, and when the keratin content is more than 30%, it is difficult to spin the fibers and the fiber membrane obtained by collection is easily brittle and cracked. Owing to the uneven thickness of the fiber membrane prepared using PAN: WK = 9:1, the stability of the 7:3 electrospinning solution was poorer. The ratio of PAN: WK = 8:2, with 15 wt% was chosen as the optimal conditions for the subsequent experiments.
Figure 2(g) shows the crystal structure of ZIF-8, which exhibits an obvious petaloid structure with a rougher and more angular surface morphology. Figure 2(h) shows that the addition of SiO2 and ZIF-8 to form the fiber membranes does not change the basic structure of the fibers, which is more difficult than that of the pure PAN and PW membranes; SiO2 particles and ZIF-8 are uniformly distributed on the fiber membrane; ZIF-8 crystals do not show obvious agglomeration phenomenon; the smooth fiber surface appears to have a rough structure, which helps to block the oil droplets on the membrane surface and it also helps to increase the hydrophilicity of the fiber membrane, and also improves the underwater oleophobicity of the PWSZ membranes, which can achieve efficient oil–water separation.
FT-IR, XRD, and EDS
The positions of the characteristic peaks on the wool fiber and keratin are shown in Figure 3(a). Some characteristic peaks of the protein structure appear in both, indicating that the extracted keratin has the same molecular structure as that of the original wool. The strong absorption peak near 3283 cm−1 is attributed to the N-H stretching vibration (amide A). A strong absorption peak at 1638 cm−1 should be assigned to the C=O (amide I). A medium-strong peak at 1524 cm−1 is ascribed to C-N stretching and N-H in-plane bending vibrations (amide II). The weak band at 1238 cm−1 is related to C-N and C-O stretching vibrations (amide III). 25
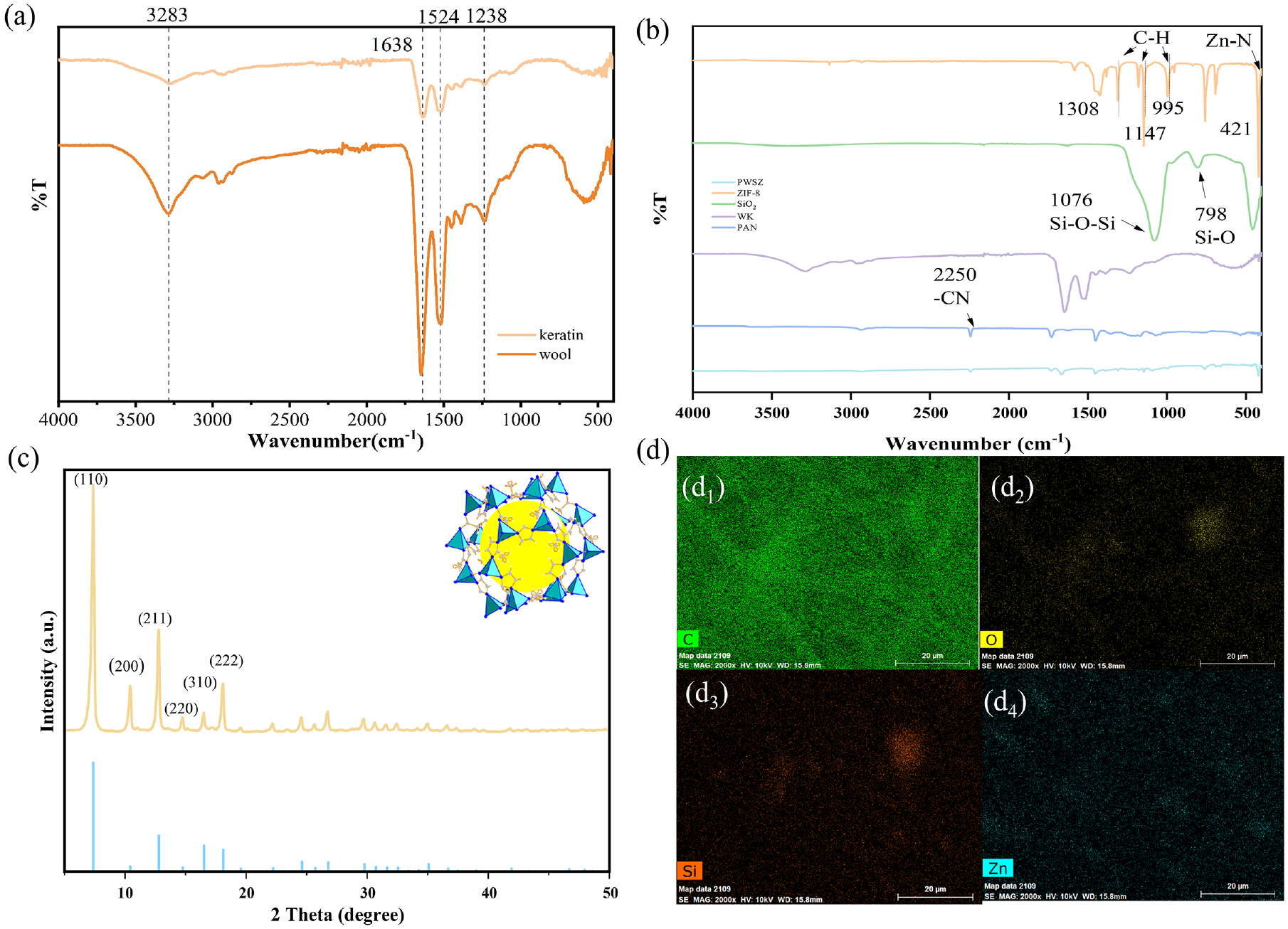
(a) FTIR spectra of wool fiber and keratin; (b) FTIR spectra of PAN, WK, SiO2, and PWSZ membrane; (c) XRD patterns of simulated and real preparation and synthesis of ZIF-8 crystals; and (d) EDS mapping image of PWSZ membrane, including C O Si Zn elements.
To gain insight into chemical groups of the as-prepared membranes after electrospinning, PWSZ membrane, ZIF-8 crystal, PAN powder, and SiO2 nanoparticles were characterized by FTIR spectra (Figure 3(b)). As seen in Figure 3(b), the peaks at 1308, 1147, and 995 cm−1 are caused by the stretching vibration of the ZIF-8 imidazole ring C-H and the band at 421 cm−1 for pronounced Zn–N stretching, which is consistent with what was stated in Hu et al., 27 indicating that ZIF-8 has been prepared. For the FT-IR spectra of SiO2 nanoparticles, the obvious absorption band at 1076 cm−1 is typical for the Si–O–Si bending vibration. This result is consistent with the literature.28–30 The absorption band at 798 cm−1 is the Si–O stretching vibration, which is also found in that of as-prepared membranes. For the FTIR spectra of PAN powder, the typical absorption band at 2250 cm−1 is in accordance with the –CN group stretching vibration. 20 In the PWSZ membrane, with all the above typical absorption bands, all the characteristic peaks of SiO2, ZIF-8, WK, and PAN appear in the spectra curves of the obtained PWSZ fiber membranes.
Figure 3(c) shows the XRD analysis of the ZIF-8 crystals, which confirms the formation of the ZIF-8 crystalline phase. It can be seen that there are obvious diffraction characteristic peaks at 2θ = 7.38°, 10.43°, 12.78°, 14.74°, 16.50°, and 18.08°, which match with the standard pattern and correspond to (110), (200), (211), (220), (310), and (222) planes, respectively. 31
BET
The separation performance of the membrane is largely determined by the specific surface area and pore volume. The results of BET test data show that the specific surface area of the PWSZ membrane is 162.637 m2/g and the pore volume is 0.1414 cm3/g. From the adsorption isotherm and pore size distribution graphs, as seen in Figure 4, it can be seen that the adsorption isotherm is similar to type IV, with a small number of micropores present and most of them are mesoporous structures, concentrated at 3.862 nm. The large specific surface area of the PWSZ membrane is conducive to the formation of an oil layer on the surface of the membrane, improving the separation efficiency.
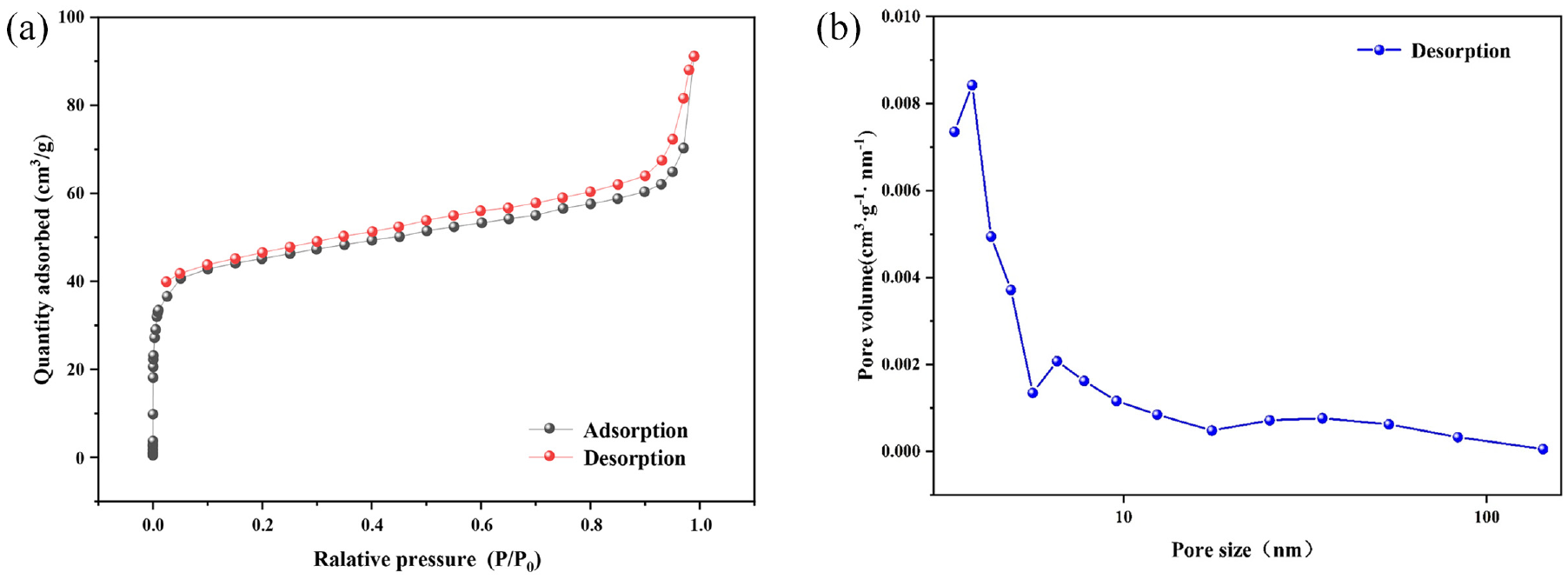
(a) N2 adsorption/desorption isotherms of PWSZ at 77K and (b) the distribution of pore sizes of PWSZ.
XPS
The XPS spectra of the PWSZ membrane are shown in Figure 5(a), which correspondingly show peaks of C1s, O1s, N1s, and Zn2p, indicating the presence of C, N, O, and Zn elements in PWSZ membrane. In Figure 5(b), the peaks at 285.85 and 284.8 eV are related to C-N and C–C, respectively. In Figure 5(c), the peaks at 399.01 and 398.9 eV are related to Zn–N and –C=N, respectively, showing that the presence of Zn–N indicates the presence of ZIF-8. 32 In Figure 5(d), the peaks at 533.1 and 531.2 eV are related to C–OH and C=O, respectively. The Zn2p spectrum in Figure 5(e) has peaks at 1044.49 eV (Zn 2p1/2) and 1021.23 eV (Zn 2p3/2), which characterize the binding of Zn2+ ions and the N atom in ZIF-8. In Figure 5(f), the PWSZ membrane at 100.77 eV is related to SiO2.
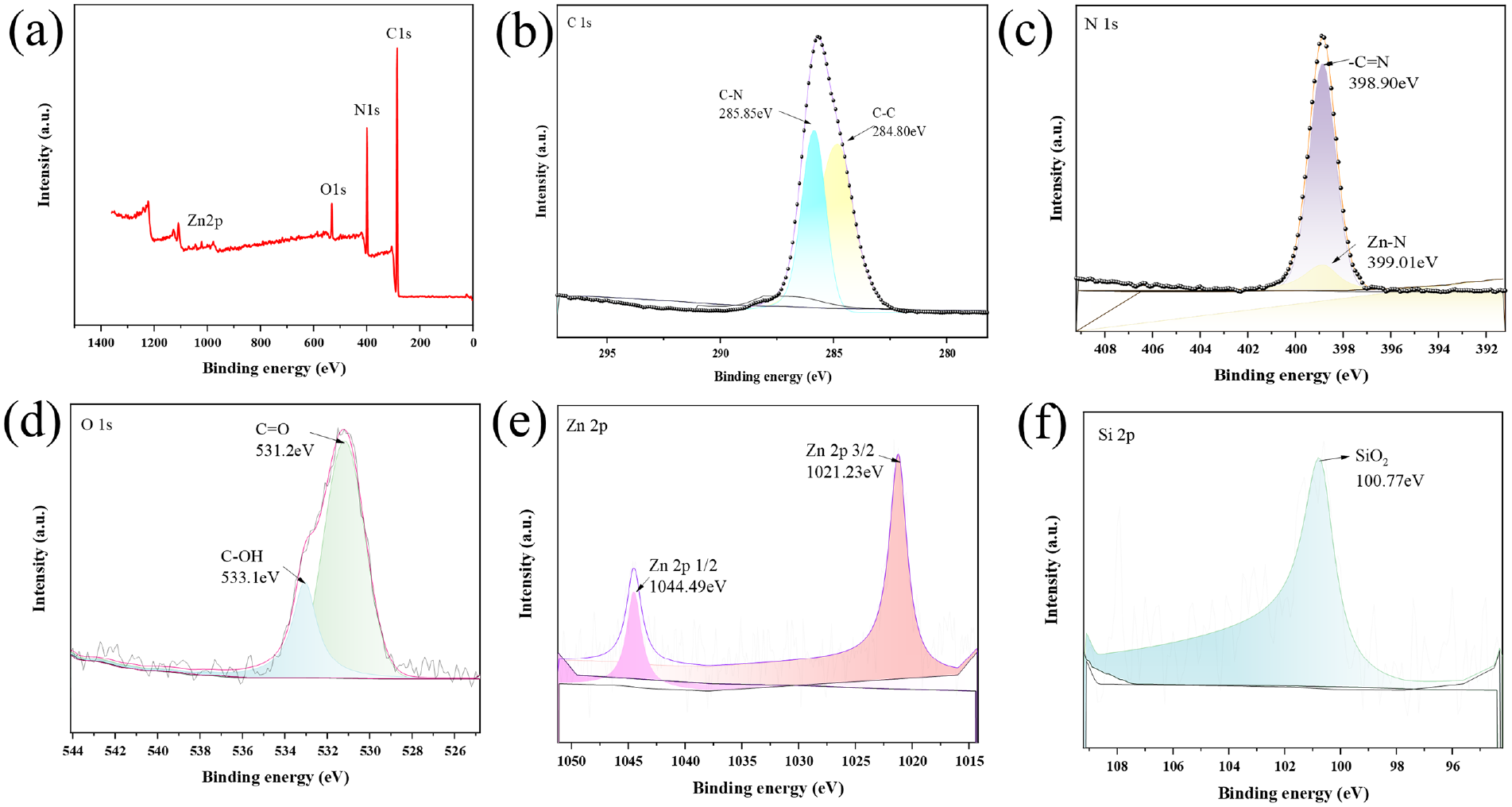
(a) XPS wide spectra and high-resolution curves of (b) C 1s, (c) N 1s, (d) O 1s, (e) Zn 2p, and (f) Si 2p of the PWSZ membrane.
Wetting Properties of Fiber Membranes
Figure 6(a) shows that the water contact angle (WCA) of the PAN membrane in the air is about 56°. Figure 6(b) shows that the WCA of the PW membrane in the air is about 36°. The decrease in the contact angle indicates that the hydrophilicity of the blended electrospun membranes of keratin and PAN has been improved owing to the presence of hydrophilic groups, such as hydroxyl and amino groups in the structure of WK, 25 which improves the hygroscopicity and hydrophilicity of the PAN fiber. Figure 6(c) and (d) show the WCA and the oil contact angle (OCA) in the air of the PWSZ membrane, respectively. When a water droplet (2 μL) comes into contact with the surface of the membrane, it diffuses and rapidly penetrates the membrane. The WCA in the air is close to 0°. When the contact angle of the oil droplets (5 μL) was measured, the contact angle of the oil in the air was also close to 0°. The PWSZ fiber membrane is highly amphiphilic to water and oil in air. The wettability mainly depends on the surface roughness and chemical composition. Compared with the PAN membrane, the increased surface roughness and the addition of SiO2 and WK make the PWSZ membrane more hydrophilic. 33 When the membrane was submerged in water, the PWSZ membrane exhibited oleophobicity with an underwater OCA of 143°. Owing to the rough structure of the fiber membrane surface as well as the super-amphiphilicity, the ZIF-8 petaloid structure on the surface captured the oil droplets and formed a water/oil/solid composite interface, which provides the basis for applications in oil–water separation.
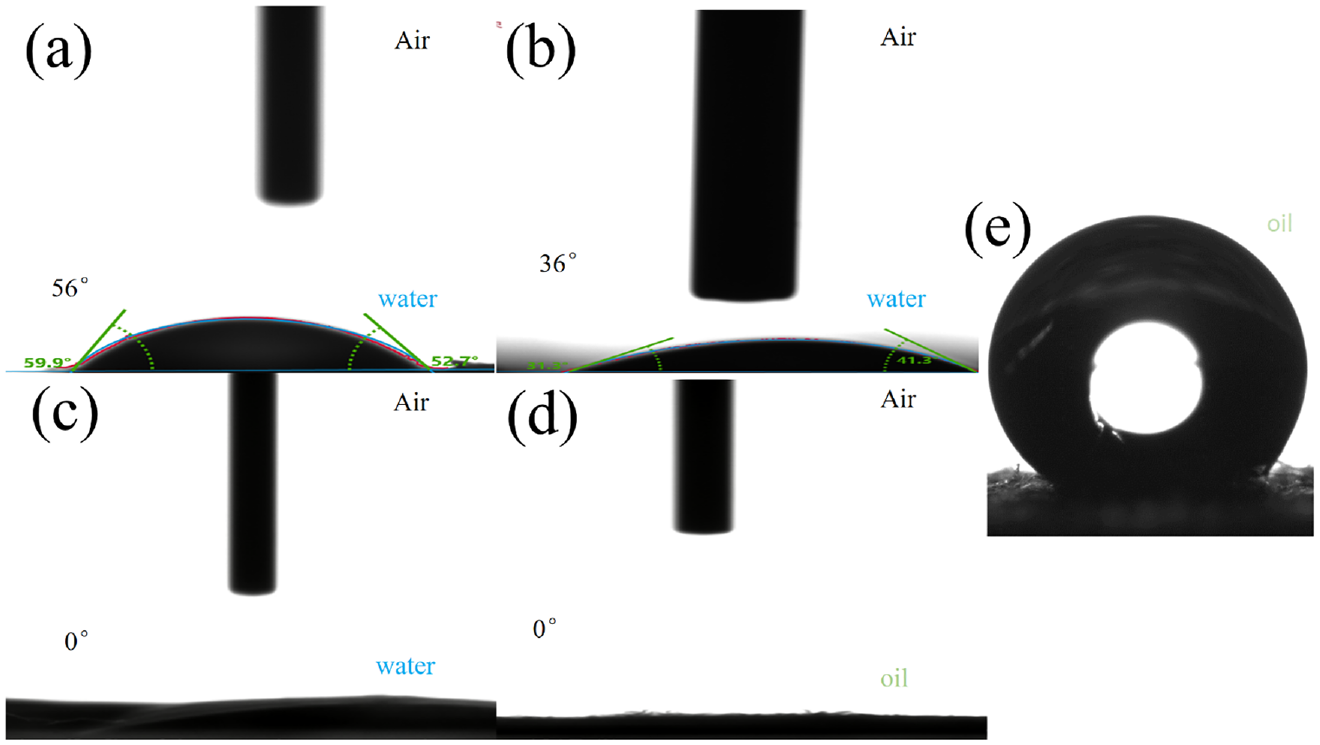
(a) Water contact angle (WCA) in the air for the PAN membrane, (b) WCA in the air for the PW membrane, (c) WCA in the air for the PWSZ membrane, (d) oil contact angle (OCA) in the air for the PWSZ-5 membrane, and (e) underwater oil contact angle (UWOCA) for the PWSZ-5 membrane.
Application of PWSZ Fiber Membrane in Separation of Oil/Water Emulsion
Figure 7(a) shows the underwater OCA plots of the PAN and PWSZ membranes with different SiO2 contents. Figure 7(b) shows the flux and separation efficiency of the PAN and PWSZ membranes for the water-in-hexane emulsion. The OCAs are 121°, 130°, 136°, 143°, and 146°, respectively. The plots of the results of the underwater OCA tests and the Figure 6(a)–(d) fiber membrane wettability plots show that PWSZ membranes have excellent super-amphiphilicity and underwater oleophobicity compared to PAN membranes. As the mass of SiO2 increased, the corresponding membrane separation efficiency increased and the flux decreased, probably because the surface of the membrane became rough, the flow rate slowed down, and the flux decreased. When the SiO2 was added up to 0.05 g, the separation efficiency reached 97.8% and the trend of the increase was not obvious after that, and the flux reached 392 L m−2 h−1. Considering the fabrication cost and performance of the membrane-separated emulsion, the PWSZ-5 membrane was optimal. We also tested whether keratin would have an effect on the separation efficiency and flux. Figure 7(c) shows that the addition of keratin enables the separation efficiency to be improved and the flux to be slightly decreased, because a large number of hydrophilic groups in keratin can help in the formation of a denser water layer in the fiber membranes when oil droplets encounter the water layer, which is more easily intercepted, and then captured by the ZIF-8 quickly. The improved separation efficiency suggests that keratin plays a crucial role in the PWSZ membrane.
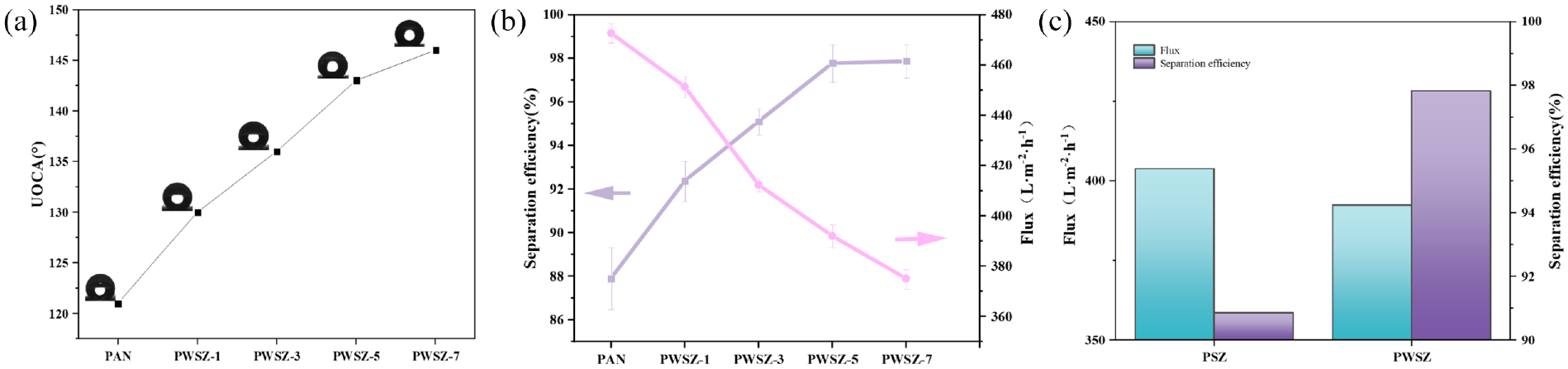
(a) Oil contact angle plots of PAN, PWSZ-x (x = 1, containing SiO2 0.0 × g) underwater (n-hexane for oil), (b) PAN, PWSZ-x (x = 1, containing SiO2 0.0× g) flux and separation efficiency for n-hexane-in-water emulsion, and (c) flux and separation efficiency before and after addition of keratin to PWSZ.
Figure 8(a) and (b) shows the separation efficiency and flux plots of the PAN and PWSZ fiber membranes in different oil-in-water emulsions. In different oil-in-water emulsions, the separation efficiencies of the prepared PWSZ fiber membranes for oil-in-water emulsions were higher than those of PAN, in which the separation efficiencies reached more than 95%, whereas the separation efficiency of the PAN membrane reached a maximum of 87.9%. It is worth noting that in the separation of oil-in-water ether Pe/W, the flux of the PWSZ membrane was significantly larger than that of the PAN membrane, which increased from 440 to 798 L m−2 h−1. The ZIF-8 particles on the surface of the PWSZ membrane were able to capture tiny oil droplets, realizing high-efficiency and high-throughput separation of different water-in-oil systems. Figure 8(c) shows the corrosion resistance of the PWSZ membrane. The flux and separation efficiency of the PWSZ membrane was slightly reduced in the saline solution, but the decrease in the alkaline and acid solutions was obvious because the ZIF-8 crystals were not resistant to corrosion. ZIF-8 crystals are not acid-resistant under the condition of acid solution. SiO2 nanoparticles easily produce slow chemical reactions in NaOH solution. Meanwhile, keratin is not alkali-resistant, which makes the fibers softer and more brittle, making the pore size larger, so the flux is higher and the separation efficiency is decreased. It can still maintain more than 90% and the flux is above 380 L m−2 h−1, which shows that the PWSZ membrane has good chemical corrosion resistance and can meet the separation environment under different pH conditions. The reusability of the fiber membrane is also important; as shown in Figure 8(d), the PWSZ membrane still maintains a high flux (330 L m−2 h−1) and separation efficiency (96.3%) after 10 cycles. Figure 9(a) shows a diagram of the device for oil–water separation. Figure 9(b) and (c) shows that the filtrate is clear and transparent compared to the original emulsion before separation. Figure 9(d) and (e) and Supplemental Figure S1 show particle size analyses of Hex/W, Hep/W, DCM/W, Pe/W, and K/W emulsions before and after separation.
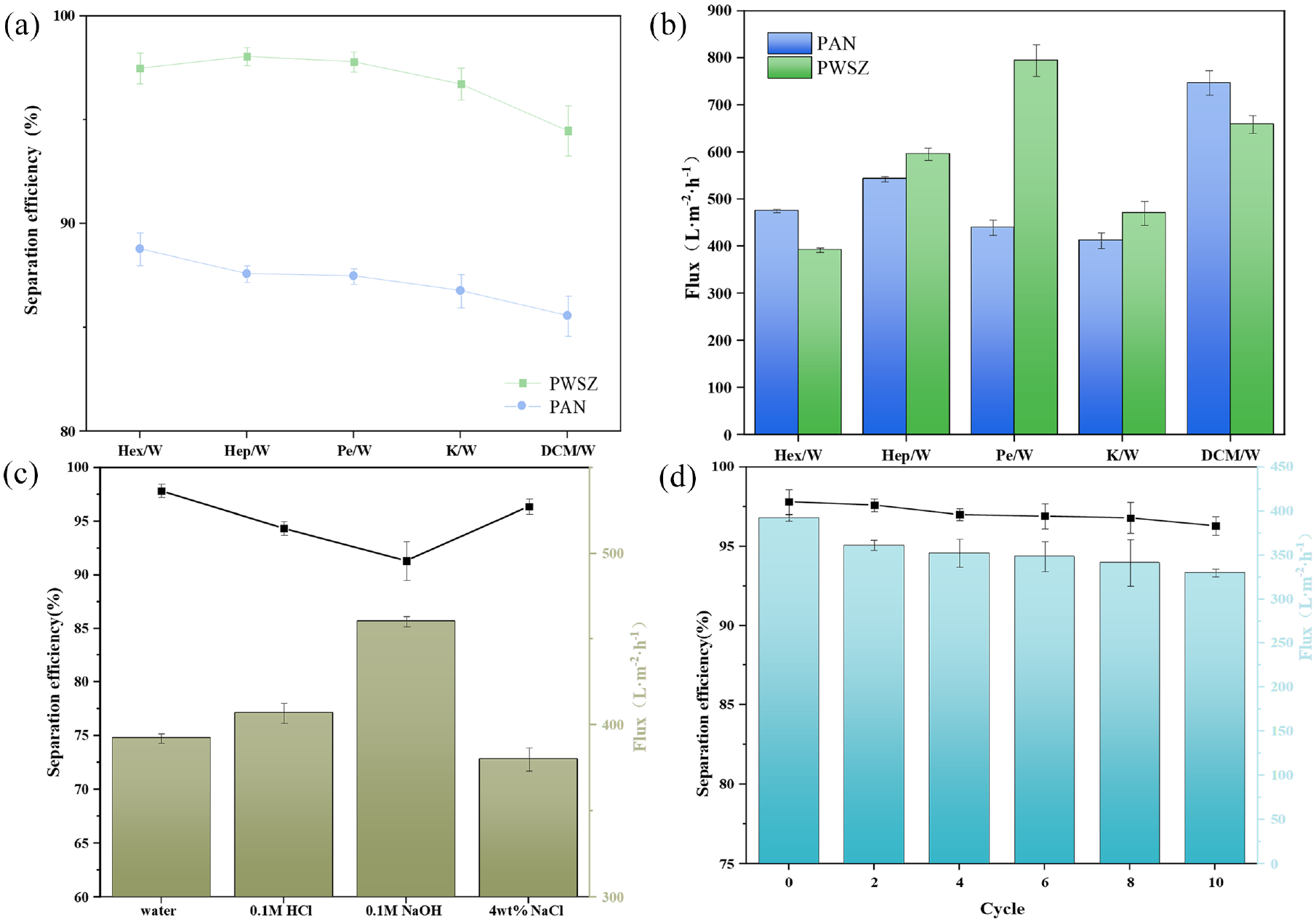
(a, b) Separation efficiency and flux plots of PAN, PWSZ-5 fiber membranes in different oil-in-water emulsions (Hex/W; Hep/W; Pe/W; K/W; DCM/W); (c) chemical resistance of PWSZ-5 membrane, flux, and separation efficiency of water-in-hexane emulsion in deionized water, 0.1 mol/L HCl, 0.1 mol/L NaOH, and 4 wt% NaCl environments, respectively; and (d) cyclic separation performance of PWSZ membrane for water-in-hexane emulsion.

(a) Diagram of the oil–water separation unit; (b) optical microscope view of the water-in-hexane emulsion; (c) optical microscope view of the water-in-hexane filtrate; the inset in the upper-right corner shows a photograph of the emulsion and the filtrate; (d) particle size analysis of Hex/W emulsions before separation; and (e) particle size analysis of Hex/W emulsions after separation.
Table 2 shows the comparison of the oil–water separation performance of the PWSZ membrane with other modified materials reported in the literature. Obviously, the membrane fluxes perform better than in reports in the literature.
Comparison of the oil–water separation performance of the PWSZ membrane with other modified materials reported in the literature.
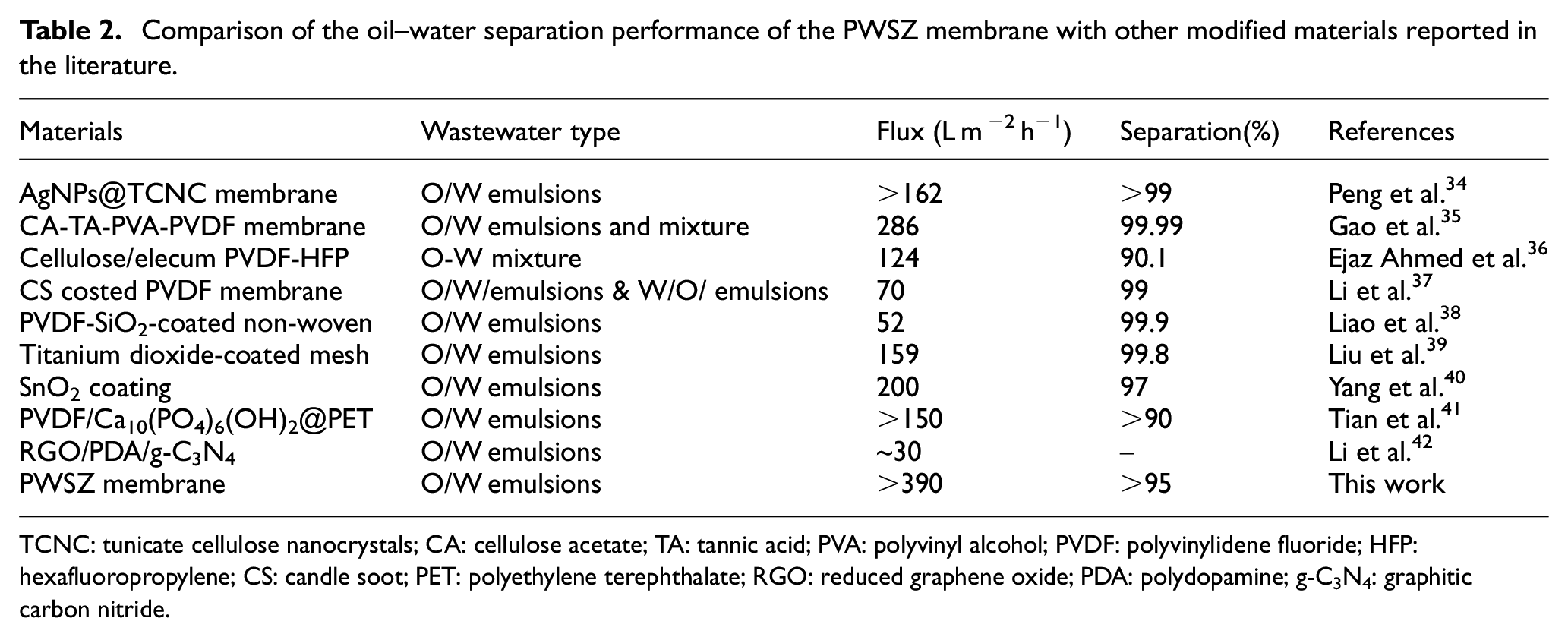
TCNC: tunicate cellulose nanocrystals; CA: cellulose acetate; TA: tannic acid; PVA: polyvinyl alcohol; PVDF: polyvinylidene fluoride; HFP: hexafluoropropylene; CS: candle soot; PET: polyethylene terephthalate; RGO: reduced graphene oxide; PDA: polydopamine; g-C3N4: graphitic carbon nitride.
Mechanism Analysis
Figure 10(a) and (b) shows the mechanism analysis. It has been reported that24,43–46 the positive charge on the surface of ZIF-8 enables it to absorb the negatively charged sulfate groups on the surface of oil droplets, which promotes the absorption and aggregation of tiny oil droplets through electrostatic attraction. In addition, owing to the hydrophobic and lipophilic nature of ZIF-8, the interactions between the hydrophobic chains of ZIF-8 and the oil droplets can also lead to the adsorption of the oil droplets on the surface of ZIF-8, which contributes to droplet trapping. The ZIF-8, due to its excellent hydrophobicity and high porosity, can act as a thin “oil layer” to capture oil droplets. At the same time, oil droplets encountering the “water layer” of the hydrophilic fiber membrane are intercepted and bounced back to be captured by the ZIF-8 surface bumps. As oil capture continues, the surface bumps form aggregated oil droplets, and the thickness of the “oil layer” increases to form large oil droplets, which then detach from the surface owing to buoyancy. After the capture, aggregation, and separation, the oil-in-water emulsion was demulsified and layered separation was achieved.
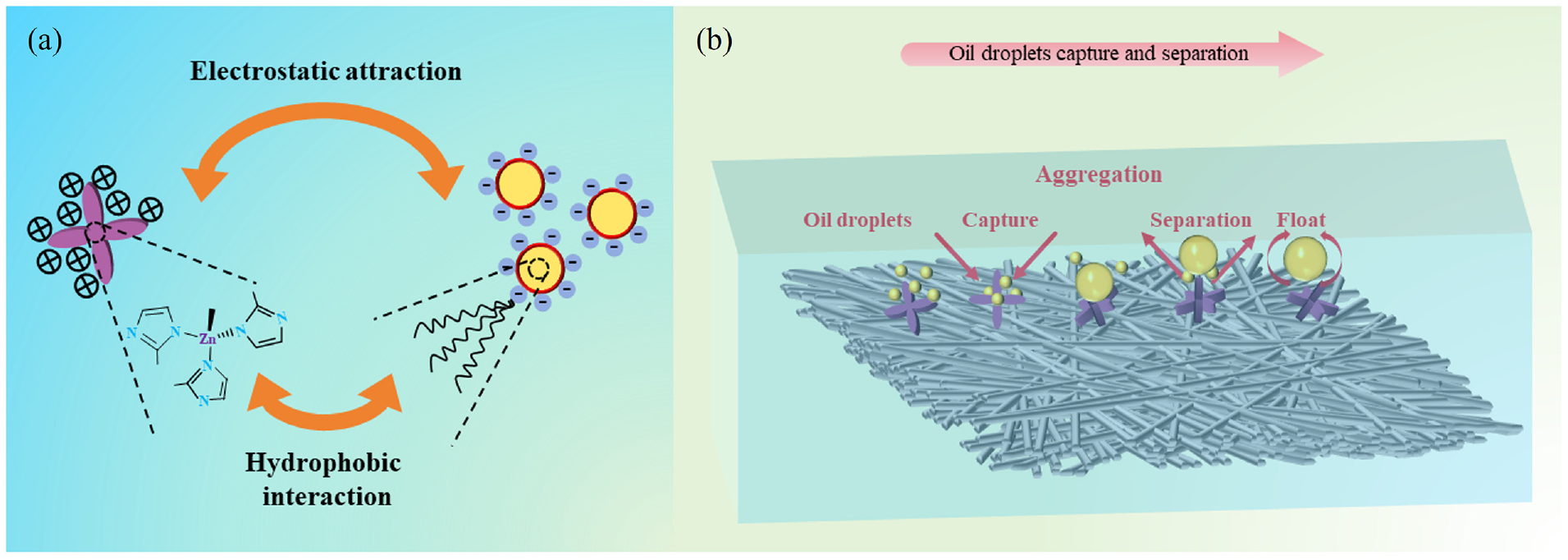
(a) Proposed mechanism for the capture of oil droplets by ZIF-8. (b) Schematic illustration of oil-capture and detachment processes on the PWSZ membrane.
Conclusion
In conclusion, we report a super-hydrophilic/underwater oleophobic PWSZ fiber membrane regenerated from waste wool, which is easy to prepare, has good chemical corrosion and reusability, exhibits high flux for various oil-in-water emulsions (Hex/W, Hep/W, Pe/W, K/W and DCM/W) (greater than 390 L m−2 h−1) and high separation efficiency (greater than 95%). Converting waste wool into oil–water emulsion separation membranes by combining it with other materials is a green and sustainable concept, which we believe can realize the transformation of waste to treasure and at the same time contribute to the realization of the sustainable development of nature and provide an experimental and theoretical basis for the treatment of complex oil–water emulsions in real environments.
Supplemental Material
sj-docx-1-aat-10.1177_24723444241288296 – Supplemental material for Polyacrylonitrile/Wool Keratin/SiO2/Zeolitic Imidazolate Framework-8 Electrospun Fiber Membrane for Highly Efficient Separation of Oil–Water Emulsions
Supplemental material, sj-docx-1-aat-10.1177_24723444241288296 for Polyacrylonitrile/Wool Keratin/SiO2/Zeolitic Imidazolate Framework-8 Electrospun Fiber Membrane for Highly Efficient Separation of Oil–Water Emulsions by Yuwen Zhu, Chengang Zhao and Rong Li in AATCC Journal of Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
