Abstract
This research evaluated the biosorption potential of the 134% Yellow Reafix BR2 dye by the malt bagasse. Tests were conducted at batch conditions, under controlled agitation, pH, and temperature. The biosorbent was characterized through scanning electron microscopy and Fourier transform infrared spectroscopy, before and after biosorption. Malt bagasse presented a point of zero charge at 6.75. In the process variables evaluation, there was a greater biosorption potential in acidic pH, without a significant influence of size on the biosorbent particles. The equilibrium time was achieved in 360 min, with approximately 93% removal at the evaluated temperatures. The experimental data were best represented by the pseudo-second-order model. Biosorption was characterized as spontaneous and endothermic, with indicative of physical. Considering the equilibrium, the Langmuir isotherm was the one that best fit the experimental data, with a maximum biosorption capacity of 68.75 mg g–1 (at 303 K and pH 2). The removal suffered no significant interference with the presence of another dye nor with the increase of ionic strength. Thus, the malt bagasse shows potential to be used as a biosorbent in treatment systems of industrial effluents.
Introduction
Water resources are essential for the maintenance of life and biodiversity on our planet. However, several anthropogenic activities contribute to the degradation of water quality, especially due to the incorrect arrangement of sewers and disposals. The industrial activity corresponds to 22% of the consumption of fresh water. Consequently, large volumes of wastewaters are generated. Currently, several legislations are in force to ensure the correct disposal of these effluents. However, globally, over 80% of wastewaters are estimated to still be disposed of without proper treatment (WWAP, 2017).
Textile industries, for example, form the share of industries with the greatest problems associated with effluents due to their complex composition and elevated coloration (Ge et al., 2012; Iqbal and Nisar, 2015; Hethnawi et al., 2017). The textile industrial process basically involves the steps of assembly, fixation, and final treatment to dye fibers and fabrics. Retention of dyes to the fibers occurs in the assembly and fixation steps. Otherwise, in the final treatment step, fiver washing is conducted for removing excessive and hydrolyzed dyes which, however, were not fixed to fibers. Therefore, this step generates large volumes of effluents that carry colorful inorganic salts and chemical compounds used throughout the process (Bilal et al., 2016a; Iqbal et al., 2019; Noreen et al., 2017).
Dyes are organic compounds that have chromophore groups in their structure and, therefore, display coloration in response to light absorption. Within the synthetic class, there are the reactive, disperse, acidic, and basic dyes, which differ from each other regarding the chemical structure and the method by which they are fixed to fibers. Among these, reactive dyes are the most used due to their high solubility in water, formation of covalent bonds with fibers (which provides more stability to the color), and their use in the dyeing of cotton, which is the major fiber used in the world (Guaratini and Zanoni, 2000; Kimura et al., 1999; Marin, 2013).
Textile effluents have in their composition suspended solids, metals, chlorides, aromatic compounds, and toxic substances. Such a complex composition, in addition to the high coloration, generates risks to human beings, aquatic creatures, and biodiversity when the effluent is incorrectly arranged (Nouren et al., 2017). When released in water bodies, the color can impair the photosynthesis of plants since it interferes in the transmission of sunlight. The biological oxidation of dyes consumes the oxygen dissolved in the environment, impairing the respiratory activity of aquatic organisms (Acar et al., 2015; Aksu and Tezer, 2005; Chen et al., 2014). In addition, the contamination of water supply networks, in the long term, aggravates the mutagenic, carcinogenic, and pathogenic potential in human beings (Iqbal, 2016; Iqbal et al., 2019; Saha et al., 2012).
Synthetic dyes are substances developed to resist the discoloring caused by the action of light and chemical agents, as well as by the exposure to high temperatures and to the enzymatic action resulting from the use of detergents. Consequently, dye biodegradation is a slow process (Aksu and Tezer, 2005; Bilal et al., 2016b). The textile effluents are generally treated by physical and chemical processes, including chemical oxidation, photocatalysis, membrane filtration, electroflocculation, among others (Babu and Murthy, 2017; Colpini et al., 2014; Iqbal et al., 2015; Nagel-Hassemer et al., 2012; Zodi et al., 2013). However, with these treatments, the effluent often does not reach the standards allowed for disposal, and sometimes they can become economically unviable due to operating conditions and to the high cost of raw materials required (Aksu and Tezer, 2005; Bilal et al., 2016a).
In the search for viable alternatives, biosorption has been widely disseminated and evaluated by several researchers (Fiorentin et al., 2010; Fontana et al., 2016b; Kini et al., 2014; Nguyen et al., 2016; Shoukat et al., 2017; Tahir et al., 2018). The biosorption process consists in the bonding of a species dissolved in a liquid phase (sorbate) to a solid material of natural origin – biomass (biosorbent). This process is based on different mechanisms, namely, physical adsorption, chemical adsorption, ion exchange, complexation, chelation, and microprecipitation. The bonding mechanism between species depends on the chemical nature of the pollutant (ionic charge, size, and species), of the biomass type (its preparations and specific surface properties), in addition to the environmental working conditions. The application of this technique is more viable when the biosorbent is abundant, has low cost, and requires no previous treatment (Aksu and Tezer, 2005; Fiorentin et al., 2015).
Several materials, such as industrial residues and by-products, have been used as biosorbents (Akar et al., 2009; Angin, 2014; Geçel et al., 2016; Reis et al., 2018). Malt bagasse, for example, is a by-product generated on a large scale in processes of beer fermentation. The estimate is that for every 100 kg of malt used, 110 to 120 kg of malt bagasse are produced, which are commonly intended for animal treatment due to its high nutritional value (Bronchier and Carvalho, 2009). However, the exceeding quantity of this by-product generates the need for new options for disposal. The proposed alternative is the application of the malt bagasse as biosorbent in the removal of pollutants. Thus, the by-product of the brewing industry is reallocated to a new process, such as biosorption, which may become more attractive and economically viable.
Therefore, this study aims at contributing to the soaring literature on the theme based on the biosorption evaluation of reactive yellow dye by the malt bagasse, with investigation of the best removal conditions (pH, particle size, contact time, and temperature), kinetic and equilibrium studies, evaluation of interference on removal, and characterization of biomass before and after biosorption.
Materials and methods
Preparation of solutions and calibration curve
Aqueous solutions were prepared from the dissolution of the 134% Yellow Reafix B2R dye (YR-B2R) – sorbate (empirical formula C28H19ClN11Na5O19S6; molecular weight of 1160.50 g mol–1) in distilled water. This is a reactive dye, whose chemical structure is shown in Figure 1. The pH of solutions was adjusted by adding aliquots of acid and basic solutions (HCl and NaOH).

Chemical structure of reactive yellow dye (TOXNET – Toxicology Data Network, 2012).
The calibration curve was obtained from the standard solutions of dye (0–100 mg L–1), and the corresponding absorbance was obtained by ultraviolet-visible spectroscopy (UV-Vis) (Femto, 800XI) at a 410 nm wavelength.
Biomass preparation and characterization
The biosorbent used in the trials – the malt bagasse – was provided by a Microbrewery in Ponta Grossa/PR. The biomass was dried at 303.15 ± 2 K in an air-circulating oven with air renewal, being later ground in a macro shredder mill. The sieving was carried out in an electromagnetic stirrer for 10 min, with the granulometric range (16.20), (42.48), (500.635) mesh, corresponding to the mean particle sizes of 0.912, 0.323, and 0.0225 mm, respectively.
Point of zero charge test (pHpcz) was performed to evaluate the charge on the biosorbent surface. It was determined from samples containing 50 mL of sodium chloride (NaCl) solution (0.01 M), with the pH adjusted from 1 to 12 ± 0.1 and 0.10 g of biosorbent. Samples were maintained for 24 h at 130 rpm and 303.15 ± 2 K. After this period, the final pH was determined, and data were plotted for pHpcz determination.
The malt bagasse characterization was evaluated before and after biosorption, based on the scanning electron microscopy, which was carried out in an electron microscope (Tescan, Vega 3 LMU) equipped with an energy-dispersive detector (EDS) (Oxford, AZTec Energy X-Act). Analyses were performed with the previously dried (303.15 K) and nonsieved biosorbent. Samples were covered with a thin deposition of a mixture of gold and palladium on the biomass surface; in addition, micrographs were obtained at a voltage of 10.0 kV.
Fourier transform infrared spectroscopy (FTIR) (Bruker, Vertex 70v) was also used for bagasse evaluation, with a transmittance in the spectral range from 4000 to 400 cm–1, resolution of 4 cm–1, and 128 scans. Samples of malt bagasse before and after the biosorption (previously dried at 303.15 K and nonsieved) were prepared with KBr discs and then subjected to spectral analysis.
Batch biosorption trials
To improve operating conditions, preliminary tests were carried out to investigate the effect of the solution pH and particle size on the removal of the YR-B2R dye. For this, the trial conditions of the biosorption process initially studied were pH (1 to 12 ± 0.1) and biosorbent particle size (0.912 to 0.0225 mm, in addition to nonsieved samples).
Batch trials were carried out based on the preparation of samples containing 0.30 g of malt bagasse and 50 mL of the YR-B2R solution at 75 mg L–1. The dye and biosorbent in the aqueous solution were kept in contact in a rotating shaker, with a 130 rpm rotation at 303.15 ± 2 K. After contact, the samples were removed and centrifuged at 3000 rpm for 10 min. Dye concentrations were measured using the UV-Vis spectrophotometer at 410 nm.
Equation (1) was used to determine the amount of dye biosorbed by the biomass (
Kinetic study
To determine the contact time and characterize the biosorption equilibrium, a kinetic test was performed, in triplicate. The samples, with the dye solution at 75 mg L–1 and pH 2 ± 0.1, were placed in the shaker at 130 rpm. At predetermined time intervals, samples were removed and subsequently centrifuged and analyzed in UV-Vis. The trials were conducted at temperatures of 303.15, 313.15, and 323.15 ± 2 K.
Equilibrium study
The equilibrium test was aimed at quantifying the dye removal ability of the malt bagasse and at determining the system behavior. The experiment was held in triplicate with 0.30 g of biosorbent, 50 mL of YR-B2R solution at different concentrations (from 12.5 to 900 mg L−1), and pH 2 ± 0.1. Samples were kept in the shaker at 303.15 ± 2 K, and, after a contact period equivalent to the equilibrium time (360 min), they were centrifuged and subsequently subjected to UV-Vis readings.
Interference test on removal
Interference by other dye
Standard solutions of YR-B2R and 250% Violet Solimax 4BLE (VS-4BLE) dyes (both at 75 mg L–1 and pH 2 ± 0.1) were mixed in proportions of 50%, 80/20%, and 20/80% (reactive yellow/violet) for final mixture concentration at 75 mg L–1. The removal was evaluated in triplicate through contact between 50 mL of these mixture solutions and 0.30 g of biomass for 1 h in the shaker, at 303.15 ± 2 K and 130 rpm. Samples were centrifuged, and the interference analysis was conducted through the scanning of solutions in UV-Vis spectrophotometer in the range of 300 to 800 nm (scanning resolution of 5 nm), before and after biosorption. Removal percentage was determined by the difference between the areas below the curves, obtained from scans of the solutions.
Effect of ionic strength
To evaluate the effect caused by the presence of ions on the biosorption process, NaCl, potassium chloride (KCl), and sodium nitrate (NaNO3) were used. Stock solutions of each salt were diluted in 50 mL of YR-B2R solution (75 mg L–1, pH 2 ± 0.1), generating samples with salt concentrations from 0.01 to 1 mol L–1. An amount of 0.30 g of biosorbent was added to these samples, in triplicate, which was then kept in the shaker for 1 h (130 rpm, 303.15 ± 2 K). After centrifugation, the supernatant was subjected to readings in UV-Vis, and the removal was evaluated.
Results and discussion
Biomass characterization
Point of zero charge (pHpzc)
The pHpzc is an important factor to be evaluated because it reflects the surface charge of a biosorbent material, being defined as the pH value at which the external and internal net charge of the functional groups on the biosorbent surface is neutral and does not contribute to the solution pH; hence, the number of positive and negative sites is the same (Chham et al., 2018; Cueva-Orjuela et al., 2017; Shoukat et al., 2017).
In the case of anionic dyes such as YR-B2R, the removal is favored when pH < pHpzc where the surface of the material becomes positively charged. The pHpzc is determined from the intersection of the curve carrying ΔpH = (pHinitial–pHfinal) as a function of the pHinitial with the abscissa axis (Chham et al., 2018; Shoukat et al., 2017). Figure 2 shows the relationship between pHinitial and ΔpH for malt bagasse. We found that pHpzc was 6.75 – indicating that, if the pH of the solution is below this value, the surface of the material is positively charged. Whereas if the pH of the solutions is above the pHpzc, the surface of the material is negatively charged.
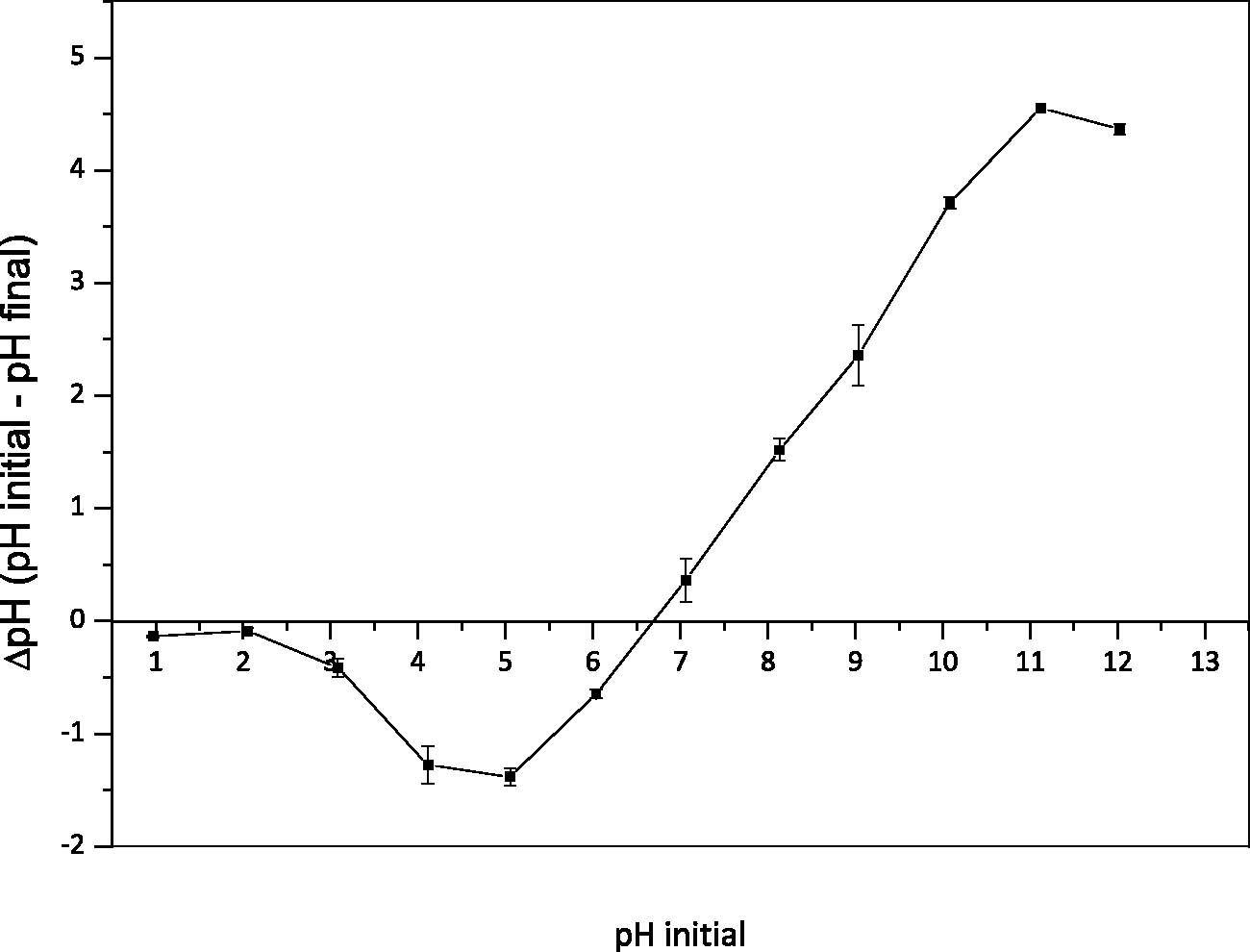
Point of zero charge of malt bagasse (conditions: 50 mL of NaCl solution at 0.01 M, pH range of 1 to 12 ± 0.1, 0.1 g biosorbent dose, 24 h contact time at 303.15 ± 2 K and 130 rpm).
Similar results were also found by the researchers Brito et al. (2010), Cardoso et al. (2011), and Scheufele (2014), who used residual materials such as Brazil nuts shell (pHpzc = 6.57), cupuassu shell (pHpzc = 5.92), and sugarcane bagasse (pHpzc = 5.90), respectively, in the treatment of effluents.
Scanning electron microscopy
Biomass micrographs before and after the yellow dye biosorption are shown in Figure 3(a) and (b), respectively. In Figure 3(a), one can observe that the malt bagasse has a rough surface, with ordered layers that show the fibrous nature of the material. This morphology favors the adherence of species, such as compounds and ions, to the irregular surface of the biomass (Boniolo et al., 2010; Paredes-Doig et al., 2014). Figure 3(b) shows the filling of the surface with the YR-B2R dye after biosorption.

SEM of the malt bagasse (a) before and (b) after biosorption (conditions: biosorbent dried at 303.15 K and not sieved, 500× magnification, tension 10 kV).
EDS spectra shown in Figure 4 have the representative bands of the elements in it. The elements nitrogen, calcium, potassium, and iron are not within the significance interval determined by the EDS equipment used. However, according to the results, the biomass surface before biosorption basically contains carbon and oxygen, characteristic of fibrous materials, in addition to the elements calcium, potassium, and iron, which may have been leached after the biosorption process, as observed in Figure 4(b). This same spectrum shows the elements nitrogen and chloride, which can result from the dye structure and the operation of the samples’ pH adjustment. One can also verify a characteristic band of silicon, which can be an indication of impurities in the adsorbate.

EDS of malt bagasse surface (a) before and (b) after biosorption (conditions: biosorbent dried at 303.15 K and not sieved, tension 10 kV).
FTIR analysis
Infrared spectra of malt bagasse, before and after biosorption, are shown in Figure 5. The same peaks are observed for both spectra. The band located at 3388 cm–1 is indicative of groups -OH and -NH. Differently, the peak located at 1666 cm–1 is the strain rate for the carboline group (C=0) at 1527 cm–1, which indicates the presence of aromatic rings or stretching -N = N- and at 1027 cm–1, for stretching C-O in carboxylic acids, alcohols, phenols, and esters. For these wavelengths, the malt bagasse containing YR-B2R (Figure 5(b)) had an increased absorption of these peaks. As for peaks located between 2916 and 2861 cm–1, there were no significant changes in absorption when spectra were compared before and after biosorption. These peaks indicate C-H vibrations of the methyl and methylene groups (Fontana et al., 2016a; Geçel et al., 2016; Pavia et al., 2010; Scheufele, 2014).

FTIR spectra (a) before and (b) after biosorption of YR-B2R by malt bagasse (conditions: biosorbent dried at 303.15 K, resolution 4 cm–1, 128 scans, spectral range from 4000 to 400 cm–1).
Infrared spectra of malt bagasse (before biosorption) present characteristics similar to several natural fibers such as the stretches -OH (3388 cm–1), C-H in methyl and methylene groups (2916 and 2861 cm–1), and C-O (1027 cm–1), characteristic of lignocellulosic materials. Similarly, the peak located at 1527 cm–1 indicates the presence of hydroxycinnamic alcohols, showing the presence of lignin in the natural fiber (Miranda, 2009; Scheufele, 2014; Zhang et al., 2013).
According to Nascimento et al. (2014), the infrared analysis contributes to revealing the bonding mechanisms associated with the biosorption. Adsorption mechanism, for example, is identified from covalent compounds present in the biosorbent material that can act as adsorption sites (Pavia et al., 2010). From the groups identified in the malt bagasse (Figure 5(a)), these binding sites can be promoted by the -OH (3388 cm–1), CH (2916 and 2861 cm–1), and CO (1027 cm–1). In particular, the CO group of carboxylic acids acts as the main binding site in the adsorption of reactive dyes since they have a higher reactivity than hydroxyls due to their structure that allows the charge resonance (Machado et al., 2011; Scheufele et al., 2018). However, more effective conclusions about the removal mechanism should be made based on the FTIR of the biosorbent under different forms of pretreatment and operating conditions so that physicochemical interactions responsible for the removal are better understood (Saeed et al., 2010; Santos, 2016; Scheufele et al., 2018).
Textile dyes classified as reactive have as main characteristic the presence of azo groups (chromophore groups responsible for staining), sulfonate groups (conveying anionic character and guaranteeing dye solubility), and functional groups (promoting covalent bonds with cellulosic fibers) (Guaratini and Zanoni, 2000; Kiernan, 2001; Kimura et al., 1999). For the latter, the reactive groups are vinylsulfone and chlorotriazine. In the case of the reactive yellow dye (chemical structure shown in Figure 2), the correlated reactive groups can be identified. In the infrared spectra of malt bagasse after biosorption (Figure 5(b)), there is an indication of the presence of these reactive groups, as well as at 3388 cm–1 with the stretching -NH, which is a primary amine present in monochlorotriazine groups, and C = C vibrations at 1666 cm–1 typical of vinyl radicals, of the vinylsulfone groups. The peak located at 1527 cm–1, in turn, reflects the azo characteristic of the dye, with stretching -N = N- (Holler et al., 2009; Pavia et al., 2010; Scheufele, 2014; Zhang et al., 2013).
Process variables effect on dye removal
Influence of pH on removal
The pH is an important parameter in characterization of biosorption process, as it can influence the surface charge of the biosorbent, the ionization of dye molecules, and even interfere on the removal (Gad and Daifullah, 2007; Wang et al., 2009). Trial results to evaluate the influence of this parameter on the biosorption of YR-B2R (303.15 ± 2 K, 24 h) are shown in Figure 6. We verified that, with the pH decrease, there was an increase in removal, with a capacity of 10.73 mg g–1 in pH 2 ± 0.1. From pH 7 ± 0.1 on, the removal was ineffective and null. Hence, pH 2 ± 0.1 was adopted as an operation condition.

Removal of dye YR-B2R by malt bagasse in function of the pH (conditions: 50 mL of YR-B2R at 75 mg L–1, 0.30 g biosorbent dose, pH range of 1 to 12 ± 0.1, 24 h contact time at 303.15 ± 2 K and 130 rpm).
Such fact can be explained by its relation with the electrostatic interaction between dye and biomass surface. The pH decrease raises the number of protons in the solution and, consequently, the number of negatively charged sites on the surface decreases. Reactive dyes have anionic character due to the presence of a sulfonate group (–SO3–) in their structure; therefore, the interaction with the cationic surface is favored (Fiorentin et al., 2010; Kimura et al., 1999; Módenes et al., 2011). Comparing this result to the pHpzc evaluation (Figure 2), we verified that, in fact, the removal was favored when the solution pH was lower than the pHpzc of the malt bagasse (pHpzc = 6.75). In contrast, for ranges higher than the pH of the biosorbent, the removal capacity is null, which may indicate that the biosorption of the YR-B2R dye is strongly influenced by ion–ion interactions (Al-Degs et al., 2008; Scheufele, 2014).
In the work carried out by Said et al. (2013) on the removal of the reactive yellow 2 and reactive blue 4 dyes from modified sugarcane bagasse, the removal was also effective in acidic pH (pH = 1.9 and 2.1, respectively). In contrast, Reis et al. (2018) obtained better percentages of removal in basic pH (pH = 10) when using the malt bagasse in the removal of malachite green dye; nonetheless, this is a dye of cationic nature.
Influence of particle size on removal
Removal in function of different particle sizes of malt bagasse was evaluated through the contact with the YR-B2R solution at 303.15 ± 2 K for 24 h, which showed the granulometry had no significant influence on biosorption. The trial was carried out using 0.922, 0.323, 0.0225 mm of particle size ranges; besides, only ground malt bagasse samples (nonsieved) were used. After biosorption, the removal percentage obtained was 78.41%, 77.05%, 77.78%, and 85%, respectively. In studies conducted by Fontana et al. (2016a, 2016b), the size of malt bagasse particles (0.12 to 2 mm) also did not influence the removal of direct orange and lead (II) pollutants, respectively.
Differences between removal percentages on granulometric ranges were not significant; however, when comparing them with the nonsieved samples, an increase in removal was observed. This fact can be explained by the diversity of granulometric ranges the sample without sieving had which, in addition to including correlated ranges, also included larger, smaller, and thinner unspecified particle sizes. Therefore, we chose to eliminate the sieving stage for subsequent tests.
Removal percentage was similar between specific particle sizes (0.922 to 0.0225 mm) and was around 77.75% with a variation of only ± 0.685%. However, when compared to the sample of nonsieved malt bagasse, there was a 7.25% increase in removal. This fact can be explained by the presence of particles smaller than 0.0225 mm in the nonsieved sample. As Khattri and Singh (2009) explain, the removal capacity is directly proportional to the total surface exposed and inversely proportional to the particle diameter. Thus, a sample containing a larger number of smaller particles provides greater available area and, consequently, greater removal. Therefore, nonsieved biosorbent samples were used for the subsequent tests.
Kinetic study
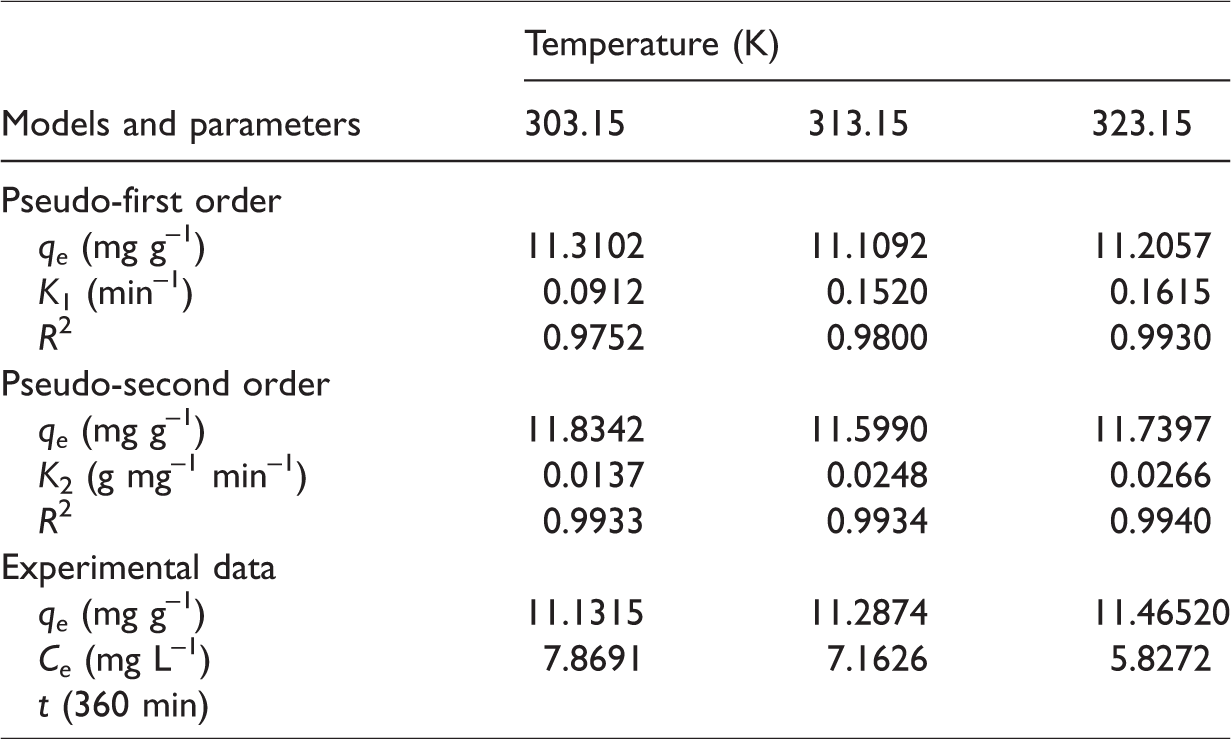
The result of quantity of the YR-B2R dye removed by malt bagasse at temperatures of 303.15, 313.15, and 323.15 ± 2 K in function of time can be observed in Figure 7. We verified the removal was similar for the three temperatures under study. At first, the biosorption was fast, reaching a removal of 8.25, 9.72, and 9.96 mg g–1 in the first 15 min. This fact contributes for the application of the technique in systems in which reasonable levels of removal are required. After this period, the biosorption occurred more slowly until the equilibrium was reached after 360 min, with the removal of 11.13, 11.28, and 11.47 mg g–1 that corresponds to 93%, 90%, and 92% of removal, respectively.

Effect of contact time on biosorption at different temperatures (conditions: 50 mL of YR-B2R at 75 mg L–1, 0.30 g biosorbent dose, pH 2 ± 0.1, 0–1440 min contact time at 130 rpm, temperature range 303–323.15 ± 2 K).
The rapid removal at the beginning, followed by a slower rate, can be explained by the availability of sites on the biomass surface. Throughout the biosorption, these sites are occupied by the dye, and consequently, the surface becomes saturated, which reflects on the decreased removal rate until equilibrium is reached (Cruz et al., 2004; Debrassi et al., 2011). Such behavior was also identified by other researchers who studied the biosorption of reactive dyes by malt bagasse, such as Zanette et al. (2015), Fontana et al. (2016a), and Reis et al. (2018), who found an equilibrium time of 240, 150, and 60 min in the removal of reactive blue, direct orange, and cationic green dye, respectively.
As the kinetic behavior for the temperatures evaluated was similar, the use of lower temperatures implies a lower energy expenditure without impairing the efficiency. For this reason, the temperature of 303.15 ± 2 K was adopted for further trials.
Bisorption kinetics
To evaluate the bisorption kinetics and determine the dye removal rate, we used the kinetic models of pseudo-first and pseudo-second order. The pseudo-first-order model is defined by equation (2).

The pseudo-second-order model is shown by equation (3).

The adjustment of the kinetic models to the experimental data was performed from the mathematical software Origin 6.0 Demo. From the adjustment, we verified the pseudo-second-order model was better adjusted to the data, as shown in Figure 8. The determination coefficient (

Kinetic models of pseudo-first and pseudo-second order adjusted to the experimental data of removal at different temperatures.
Kinetic parameters and determination coefficients of adjustment of kinetic models.
Adjustment of the pseudo-second-order model also allows to determine the activation energy and the constant of the Arrhenius equation, based on equation (4).

Activation energy, besides providing an estimate of the minimum energy for removal, also characterizes the type of interaction, which can be physical or chemical. Physical biosorption involves weaker bonding forces between sorbate and biosorbent surface with
Thermodynamic study
Kinetic evaluation at different temperatures allows the obtainment of thermodynamic parameters, such as enthalpy, entropy, and Gibbs free energy. The linear van’t Hoff equation provides the relation for determining these parameters as a function of temperature, as expressed in equation (5).


Thus, Δ
Thermodynamic parameters evaluated at temperatures 303.15, 313.15, and 323.15 ± 2 K for the biosorption of YR-B2R by malt bagasse can be observed in Table 2.
Thermodynamic parameters of biosorption of reactive yellow dye by malt bagasse, valued at different temperatures.

According to the results, the biosorption was favored by the temperature because it occurred through a spontaneous process, with Δ
Equilibrium study
The concentration of reactive yellow dye in fluid phase (

Experimental results of biosorption of YR-B2R dye by malt bagasse (conditions: 50 mL of YR-B2R at 12.5 to 900 mg L–1, 0.30 g biosorbent dose, pH 2 ± 0.1, 360 min contact time at 303.15 ± 2 K and 130 rpm).
The models commonly used to represent favorable processes are the Langmuir and Freundlich isotherms. The Langmuir isotherm assumes biosorbent surface as homogeneous and a single-layer sorption. In this process, only one molecule can be accommodated in each site, and the interaction energy between adsorbed molecules is negligible (Abdi et al., 2017; Ruthven, 1984). The relationship that describes the Langmuir isotherm is represented by equation (8):

The Freundlich isotherm is the equilibrium model that characterizes heterogeneous systems and assumes a multilayer process, without a prevision for saturation (Chen et al., 2014; Farooq et al., 2010; Geankoplis, 2003). The equation of the model is given by equation (9):

The adjustment of isotherms to the experimental data was performed from the mathematical software Origin 6.0 Demo. Table 3 shows the parameters and determination coefficients obtained. We verified that Langmuir isotherm was the model with the best adjustment, with an
Determination coefficients and parameters of isotherm models.
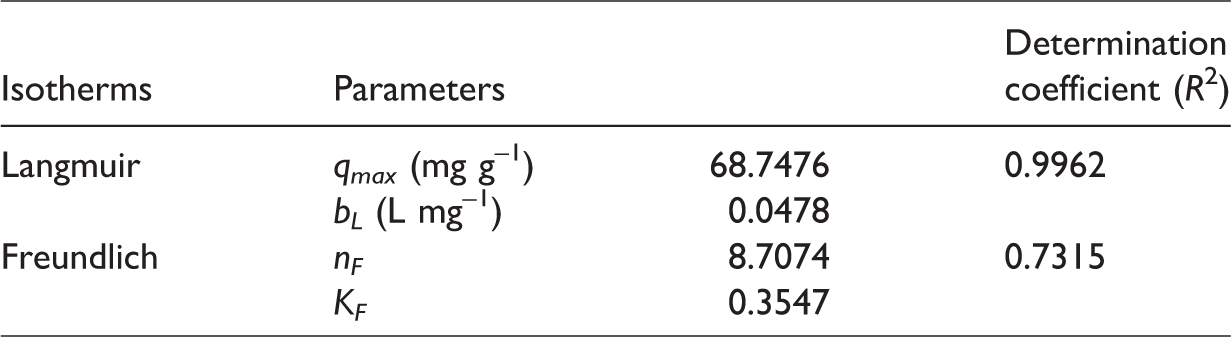
The maximum biosorption capacity (
Maximum biosorption capacity of dye by different types of biosorbents.

Interference test
The efficiency and ability of biosorption processes depend not only on biosorbent properties but also on the effluent composition (Kratochvil and Volesky, 1998). In industrial processes, the textile effluent can have a variety of other dissolved or suspended compounds that have the potential to interfere on the removal. These compounds can be acids, bases, salts, metal ions, and other dyes, deriving from the industrial process. Presence of metal ions such as Na+ (sodium ion) and K+ (potassium ion) in textile effluents raises the ionic strength of the solution and, consequently, influences the biosorption (Fontana et al., 2016a; Maurya et al., 2006).
This study evaluated the interferences caused by the 250% VS-4BLE dye in different proportions of the mixture with reactive yellow dye. Evaluations were also performed by adding NaCl, KCl, and NaNO3 salts to the solution, at different concentrations (0.01–1 mol L–1), in the YR-B2R/malt bagasse system.
Interference by other dye
The UV-Vis spectra of solutions before and after biosorption are shown in Figure 10. According to the results, the absorbance values were lower after biosorption for all cases due to the high percentage of dye removal from the solution.

Interference by mixture of dyes evaluated by UV-Vis spectra before and after biosorption of solutions of (a) YR-B2R at 75 mg L–1, (b) VS-4BLE at 75 mg L–1, and (c) 80% YR-B2R/20% VS-4BLE; 50% YR-B2R/50% VS-4BLE, and 20% YR-B2R/80% VS-4BLE (conditions: 50 mL of mixture of dyes, 0.30 g biosorbent dose, pH 2 ± 0.1, 1 h contact time at 303.15 ± 2 K and 130 rpm).
From the UV-Vis spectra, we could visualize the wavelength characteristic of each dye, even when mixed in different proportions, as in the case of the yellow dye with 410 nm wavelength (Figure 10(a)) and the violet dye at 520 nm (Figure 10(b)). The same wavelengths can be identified in the mixtures 80% YR-B2R/20% VS-4BLE and 20% YR-B2R/80% VS-4BLE, respectively (Figure 10(c)), where there is a predominance of dyes in each mixture. In the 50% YR-B2R/50% VS-4BLE solution, one can see the highest absorbance peaks are around 420 nm and 510 nm. After biosorption, we verified that even the diversification in the tonality of the samples did not interfere in the removal of the dyes, as the low values of absorbance indicate the absence of sorbates.
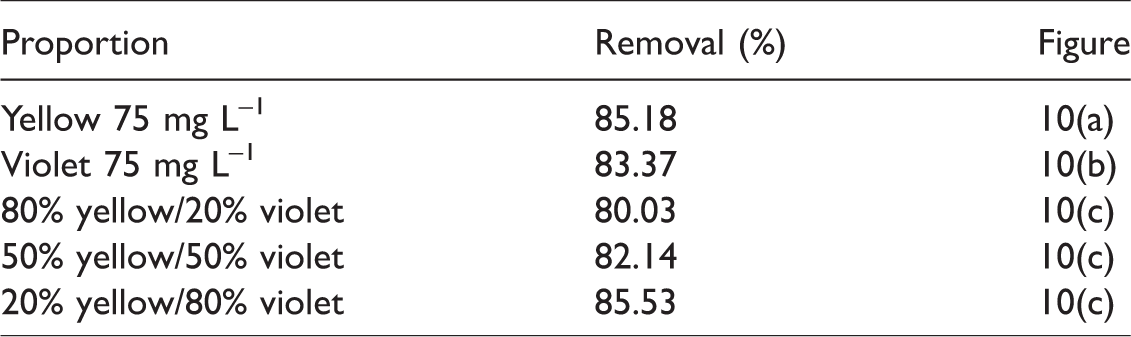
Removal percentages for the biosorption of separated dyes (Figure 10(a) and (b)) and at different proportions (Figure 10(c)) were obtained based on the integral of the areas under the curves formed by UV-Vis spectra and are shown in Table 5. This procedure was conducted with the mathematical software Origin 6.0 Demo. Based on the results, we concluded the mixture of dyes exercised no interference on biosorption because the removal only varied from 80.03% to 85.8% between samples with only one dye species and samples with different proportions. This indicates that malt bagasse has great biosorption potential of textile dyes until a concentration of 75 mg L–1.
Removal percentage of yellow and violet dyes, separated and combined in different proportions.
Effect of ionic strength
The effect of salt concentration on the biosorption of reactive yellow dye by the malt bagasse can be observed in Figure 11, in which we can verify the same behavior for all salts. In other words, the increased concentration of these compounds in the solution caused a decrease in removal rates. The removal was 11.13 mg g–1 in the absence of salts and 9.36, 9.40, and 7.80 mg g–1 in the presence of 1 mol L–1 of the NaCl, KCl, and NaNO3, respectively.

Effect of concentration of the salts NaCl, KCl, and NaNO3 in the biosorption of YR-B2R dye (conditions: 50 mL of YR-B2R solution at 75 mg L–1 with salt concentrations from 0.01 to 1 mol L–1, 0.30 g biosorbent dose, pH 2 ± 0.1, 1 h contact time at 303.15 ± 2 K and 130 rpm).
In the fact explained by Al-Degs et al. (2008), when electrostatic strengths between adsorbate and biosorbent are attractive, an increase in the ionic strength causes a decreased biosorption capacity. Inversely, if the interaction between biosorbent and dye is repulsive, the increase in ionic strength favors the removal. Therefore, we can suppose that the interaction between malt bagasse and reactive yellow dye is of attractive nature since the concentration of salts caused a decrease in removal rates.
The increase in ionic strength causes a competition between species in the solution (Esmaeli et al., 2013). Therefore, we can suppose the addition of cationic species in the solution, such as Na+ and K+, can prevent dye biosorption to the biosorbent surface due to interaction with the anionic part of the dye molecule (SO3–). On the other hand, the decreased removal percentage can also be attributed to anionic species in the solution – Cl– and NO3– – which can possibly compete with the anionic groups of the dye structure during biosorption (Al-Degs et al., 2008; Maurya et al., 2006).
Results indicate the interference by ionic species in biosorption was not significant for lower salt concentrations, in which removal had a decrease of only 0.11, 0.27, and 0.25 mg g–1 (corresponding to 0.87%, 2.20%, and 1.69%) after the addition of NaCl, KCl, and NaNO3, respectively. For higher concentrations, however, we found lower tolerance to salt concentration considering the malt bagasse. Researchers who evaluated the effect of ionic strength in its biosorption processes have also identified the influence of these compounds in higher concentrations (Fontana et al., 2016a; Maurya et al., 2006).
Conclusions
This study aimed at evaluating the biosorption of the Yellow Reafix B2R dye using the malt bagasse as biosorbent. Based on the parameters evaluated, we verified the dye removal was effective in acidic medium and suffered no significant influence of granulometry. Equilibrium time of the system was achieved after 360 min of contact. The pseudo-second-order model had the best adjustment in biosorption kinetics. The activation energy obtained suggested a process of physical biosorption. The Langmuir model had the best adjustment to the equilibrium data obtained, and the maximum biosorption capacity was quantified at 68.75 mg g–1. Based on thermodynamic parameters, the removal was evaluated as spontaneous, endothermic, and with the disorder increase on liquid/solid interface throughout biosorption. The interference of VS-4BLE and the effect of ionic strength were not significant. These facts are indicatives of the good performance of the malt bagasse, given the presence of other components in solution.
Thus, we verified the malt bagasse is a low-cost biosorbent and that, under favorable conditions, it shows a high potential in the treatment of effluents with dyes.
Footnotes
Acknowledgements
The authors thank the Schultz Bier microbrewery for the donation of the malt bagasse, the UTFPR – Ponta Grossa Campus for the structure provided for conducting this research, and the Tutorial Education Program of Chemical Engineering of UTFPR (PET-EQ –
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
