Abstract
In this paper, polyaniline/attapulgite (PANI/APT)-supported nanoscale zero-valent iron (nZVI) composites were synthesized by liquid-phase chemical reduction method and used for the removal of two kinds of dyes. The structure of as-prepared nZVI/PANI/APT was characterized by various test methods. The removal property and degradation mechanism for azo (alizarin yellow R, methyl red, chrome black T, methyl orange) and non-azo (methylene blue, rhodamine B) dyes in aqueous solution were investigated. The presence of PANI/APT can decrease the aggregation of nZVI particles with maintenance of reactivity and improving adsorption capacity for degradation azo dye. The experiment results showed that the removal property of the composite materials on azo dyes is obviously better than that on non-azo dyes. The varying removal efficiencies of dyes depend on the different degradation mechanisms. Azo dyes removal by nZVI/PANI/APT was mainly due to the reductive cleavage of the N = N of nZVI, while non-azo dyes removal mainly contributes to the adsorption of PANI/APT. The study demonstrated that nZVI/PANI/APT has potential applications for the removal of azo dyes from wastewaters.
Introduction
Remediation of dye wastewater has aroused much attention owing to the fact that they are severe harmful to the environment and human health (Robinson et al., 2001). Most of the dyes and pigments can lead to serious environmental and health problems due to their high toxicity, chemical stability, difficult biodegradation, and possible carcinogenicity (Cheng et al., 2010). Nowadays, coexistence of various dyes in industrial wastewater is a serious environmental problem. Thus, the simultaneous removal of diverse organic dyes from wastewater has a particular significance to both pollution control and remediation. Among various dyes, azo dyes (−N=N– unit as the chromophore) from leather, textile, or dyestuff industries are the major source of dye wastewater pollution. However, the complex structures and metabolic intermediates of azo dyes make them more difficult to degrade (Park and Choi, 2003). Therefore, much concern should be paid to treat the industrial wastewater containing azo dyes before discharge. Up to now, many methods involving physical, chemical, and biological methods for the destruction of dyes have been applied in treating the dye wastewater (Chen et al., 2013; Jin et al., 2015). Physical treatments like adsorption have the advantages of low cost and easy operation; however, it may produce large quantities of solid wastes. Biological degradation is a good alternative way for the removal of dyes and organic pollutants. But traditional biological processes are expensive, ineffective, and probably produce harmful intermediates (Garg et al., 2003; Li et al., 2013). Recently, nanoscale zero-valent iron (nZVI) have attracted great attention for the removal of heavy metal ions and organic pollutants from wastewater (Shaibu et al., 2014; Quan et al., 2014) due to high reducing capacity, large specific surface area, and good surface degradation ability (Jabeen et al., 2011). In most studies, the nZVI main mechanism was the reductive degradation. However, nZVI particles without any modification tend to aggregate into larger ones because of their high surface energy or react with surrounding media to form oxide layers on the particle surface to a significantly decrease in the reactivity of nZVI (Choe et al., 2000). To prevent the aggregation and oxidation of nZVI particles, nZVI particles are supported or modified on substrates to change nZVI particle surface characteristics (Uzum et al., 2009; Sun et al., 2013). Various types of supported or modified nZVI have been used for the removal of dyes and heavy metals, such as kaolinite-supported nZVI, bentonite-supported nZVI, meerschaum-supported nZVI, polymeric resin-supported, and nZVI polyacrylamide-stabilized (Chen et al., 2011; Fu et al., 2015; Shi et al., 2011; Sun et al., 2013). Researchers have reported that modified nZVI showed greater reactivity on the remediation of various kinds of pollutants compared to nonmodified nZVI. Clays with porous structure and high surface area, such as kaolin, bentonite, and zeolite, were evaluated as support material for nZVI to remove pollutants. Attapulgite (APT) is a naturally occurring hydrated magnesium aluminum silicate clay mineral with nanorod-like crystal morphologies and a rich number of pores. With its special crystal structure, APT has been widely used as adsorbents, carriers, colloidal agents, and thixotropic agent in many fields, such as in nanocomposites, catalysts, coatings, and environmental protection. Among them, the application of APT as adsorbents is especially focused (Liu et al., 2014, 2015; Xue et al., 2011). APT has already been put into use as a mechanical support to disperse and stabilize engineered nanoparticles due to its porous structure and large specific surface area (He and Yang, 2013). Unfortunately, the natural APT mineral is usually low in adsorption capacity and difficult to reuse. Polyaniline (PANI), with linear macromolecule chains, has abounded amine and imine functional groups, which exhibit good adsorption for various pollutants and electrical conductivity (Fan et al., 2015; Xu et al., 2015). Up to now, there are few reports on the utilization of organic conductive polymers to modify the surface of nZVI particles for improving reactivity.
In our previous study, we used of polyaniline/attapulgite (PANI/APT) composite as a support could effectively decrease the aggregation of zero valent iron (ZVI), therefore enhanced their reactivity and stability in removing Cr(VI) (Lu et al., 2014), where the simultaneous adsorption and degradation demonstrated that this was a promising remediation method for heavy metal ions. Also, APT has been put into use as a mechanical support to disperse ZVI. The strong anticorrosion properties and excellent conductivity of PANI play an important role in the improvement the reaction efficiency and lifetime of iron particles. The PANI film in the ZVI/PANI/APT composite has a positive adsorption effect towards the contaminants. It is speculated that organic conductive polymer covered the surface of the ZVI, which is transferring contaminants to its surface owing to the adsorption of PANI/APT and facilitating the electron transfer between the iron particles. However, little information is available regarding the support of nZVI on the surface of PANI/APT to significantly improve their aggregation and reactivity, and even use of PANI/APT-supported nZVI to remove dyes in aqueous solution has not been reported. In this work, we synthesized PANI/APT-supported nZVI composites by liquid-phase reduction method. The structure of as-prepared nZVI/PANI/APT was characterized and utilized for the removal of azo dyes (alizarin yellow R (AYR) chrome black T, methyl red, and non-azo dyes (methylene blue (MB) rhodamine B) from aqueous solution. The study explores the possibility for the simultaneous removal of dyes using PANI/APT-supported nZVI and understanding its mechanism for the removal of different structure of dyes. The effects of main parameters including pH of solution, initial concentration of dye on the removal efficiency and adsorption isotherms of dyes were also assessed. In addition, the research also investigates whether nZVI/PANI/APT improves nZVI aggregation and results in significantly increased reactivity. The results demonstrated that nZVI/PANI/APT has potential applications for the removal of azo dyes from wastewaters.
Experimental details
Materials
Attapulgite (99%, APT) (JC-J503) was purchased from Jiangsu Xuyu Nanomaterials Science and Technology Co. Ltd., China. Aniline (An) was purified before use. FeSO4·7H2O was provided by Tianjin Kaixin Chemical Reagent Co. Ltd., China. Dyes (alizarin yellow R, methyl red, chrome black T, methyl orange, methylene blue, and rhodamine B) were offered by Shanghai Zhongtai Chemical Reagent Co. Ltd., China.
Sample preparation
The PANI/ATP composites were synthesized by the chemical oxidative polymerization of aniline (An) monomer. The detailed process is described as follows: 1.0 g of ATP was added into a 250-mL flask containing 100 mL of HCl (0.5mol·L−1) and 1.5 mL of aniline (An), and then the mixed solution was treated with ultrasound for 30 minutes. After full mixing, a solution of ammonium persulphate (APS) in aqueous HCl (0.5mol·L−1) was added at once (molar ratio aniline/APS = 1/1) to the above solution and vigorously stirred for 30 minutes and kept for 4 hours. The solid material was filtered and washed with distilled water repeatedly. The resulting PANI/ATP composite was dried in vacuum oven at 60°C.
PANI/ATP-supported nZVI composite samples were prepared using liquid-phase reduction method; 1.0 g of APT and 1.5 mL of aniline (An) were added into a 250-mL flask containing 100 mL of HCl (0.5 mol·L−1), and subsequently, different amounts (2.5–3.5 g) of FeSO4·7H2O was introduced into the mixed solution under stirring to adsorb Fe2+ onto APT. After full mixing, a solution of ammonium persulphate (APS) in aqueous HCl (0.5mol·L−1) was added at once (molar ratio aniline/APS = 1/1). The suspension was vigorously stirred for 30 minutes and kept for 4 h. Lastly, the new solution of NaBH4 (molar ratio FeSO4·7H2O/NaBH4 = 1/2) was added drop-wise into the stirred mixture and stirred for 20 minutes under N2 atmosphere. The derived products were filtrated and washed thoroughly with distilled water and alcohol, and then the resulting nZVI/PANI/APT was put in a vacuum drying oven at 60°C.
Characterization
Fourier transforms infrared concerning composite materials was obtained by using Fourier transform infrared spectroscope (FT-IR, NEXQS670, USA) using KBr pellets. Morphology and structure of composite materials were observed by scanning electron microscope (SEM, JSM-6701F, Japan) and transmission electron microscope (TEM, JEM-1200EX, USA). The XPS data were obtained by a V. G. ESCA Laboratory 210 photoelectron spectrometer with an Mg Ka source and the spectra were acquired with 30 eV pass energy. X-ray diffraction patterns were collected on a X-ray diffractometer (XRD, D/MAX–2400X, Japan) using Cu Kα radiation (λ = 0.15406 Å) in a 2θ range of 10–90° at room temperature. Spectrophotometry was used for the measurement of the concentration of dyes in solution at specific adsorption wave on a 7230G spectrophotometer (Shanghai Precision Scientific Instrument Co. LTD., China), respectively. The pH measurements were conducted with a glass electrode (PHS-3D Model pH meter, China).
Batch adsorption experiments
60 mg·L−1 of dye solution (AYR or MB) 50 mL and 0.05 g of nZVI/PANI/APT (1.0 g·L−1) was added in the conical flask, which was immediately sealed and solution pH was adjusted to the desired value by negligible amount of HCl and NaOH. Mixed solution at their initial pH level was stirred with 300 r/min for 180 minutes at 298 K. They were then filtered through 0.45 µm membranes to measure the residual concentration of dye. The concentration of dye was measured using a UV-Spectrophotometer. The removal capacity and the removal efficiency of dye were calculated using equations (1) and (2)


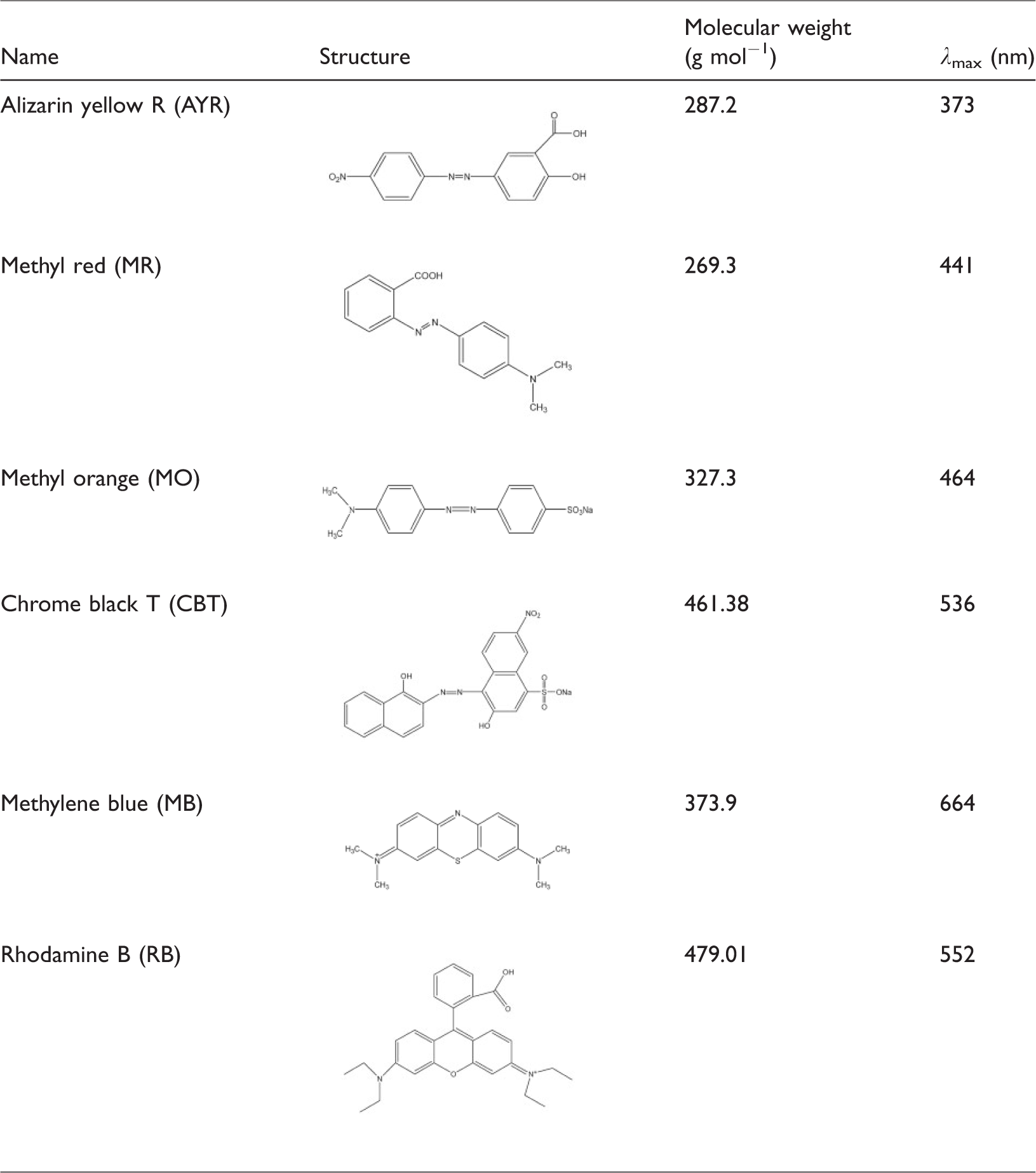
General characteristics of the studied dyes.
The concentrations of Fe2+ in the solution were measured by the phenanthroline spectrophotometric method (Yi et al., 2015). The filtrate after leaching of nZVI/PANI/APT is injected into a 50-mL sample bottle, and an appropriate amount of sample is taken during the analysis. Then, the buffer solution (10% hydroxylamine hydrochloride solution) and phenanthroline solution are added to develop color 5–10 minutes, measured absorbance at 510 nm.
Results and discussion
Characterization
The surface morphology images of the nZVI/PANI/APT composites were evaluated by SEM and TEM. As shown in (Figure 1(a)), SEM images clearly indicate that the resulting nZVI/PANI/APT composite exhibits rough and porous surface with a rod-like structure. The nanoparticles that adhered to the surface of rod-like APT are generally spherical in shape and have diameters within 50 nm. However, nZVI accumulated into a bulky nanocluster, which can be attributed to nZVI and APT wrapped with polyaniline intertwined together. TEM images (Figure 1(b)) revealed that the nanostructure of composite materials was mainly composed of nanometer rods of APT. nZVI coated on PANI sheets was well dispersed on the surface of APT and no obvious aggregation was observed, and the size of the particles was in the range of several nanometers. The results demonstrated that PANI/APT as a support material is effective in reducing the aggregation and accelerating the dispersion of nZVI, which was consistent with the observation of PANI/APT used for ZVI (Lu et al., 2014).

(a) Scanning electron microscope and (b) transmission electron microscope images of nZVI/PANI/ATP.
The XRD pattern of nZVI, APT, and nZVI/PANI/APT was presented in Figure 2. The nZVI showed a weak and broad peak at about 2θ = 44.6°, which were indexed to the (110) planes and consistent with the standard XRD data for bcc iron (0) (JCPDS no. 06–0696). The presence of iron oxide with weak intensities in the XRD pattern of the nZVI particles (2θ = 55°, 62.8°) was due to the surface oxidation of nZVI. The APT displayed diffraction peaks at 2θ = 8.28°, 19.7°, 26.8°, and 31.2°, which is consistent with the standard peak of APT the XRD patterns in Figure 2(b) (Mu and Wang, 2016). For the sample of nZVI/PANI/APT (Figure 2(c)), nZVI peak becomes invisible owing to the high intensity of PANI and APT peaks. It can be seen that characteristic peaks at 2θ = 8.28° were consistent with the standard peak of APT (Liu et al., 2012), and the peeks displayed at 2θ = 20.76° correspond to PANI (Xu et al., 2014), which were assigned to the amorphous structure of organic matrix. And the peaks at approximately 2θ = 26.71°, 34.79° confirmed the characteristics of iron oxides, indicating that nZVI particles were incompletely covered with PANI/APT, which led to the surfaces of the ZVI particles being oxidized. However, as shown in Figure 2(d), the peaks of nZVI/PANI/APT changed slightly after being used to remove AYR. The peaks originated from iron oxide were intensified, and the new peak at 2θ = 15.82° also represented iron oxide. The result may be attributed to that iron oxides formed as a result of nZVI/PANI/APT reducing the azo dye, where nZVI as the electron donor provided the electron and the azo dyes accepted the electron to form the hydrazo (–N(H)–N(H)–) derivative as an intermediate (Fu et al., 2014; Nam and Tratnyek, 2000).
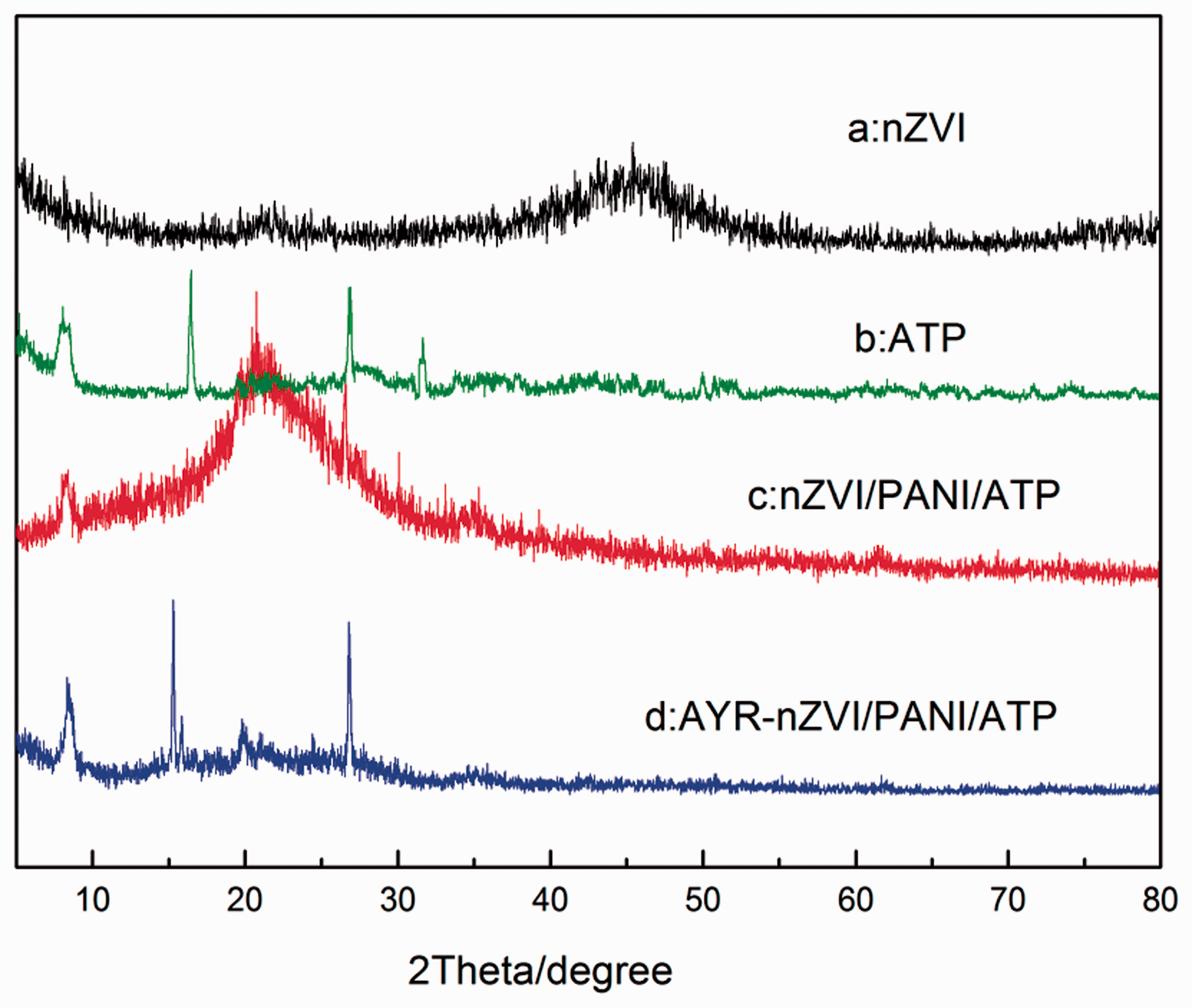
X-ray diffractometer image of nZVI and nZVI/PANI/ATP before and after removal alizarin yellow R.
The FT-IR spectrums of nZVI/PANI/APT composite before and after the removal of AYR were revealed in Figure 3. The FT-IR spectrum of the nZVI/PANI/APT consisted of –NH– (3392 cm−1), quinoid peak (1579 cm−1), benzenoid peak (1490 cm−1), C–N (1297 cm−1), and C–H (1133 cm−1, 798 cm−1), which can be indicated the backbone structure of PANI (Xu et al., 2013). And the peaks appearing at 3447, 1670, 1374, and 1022 cm−1 can be attributed to the existence of APT (Yan et al., 2012). The broad peak at 2923 cm−1 corresponds to the stretching vibration of hydrogen bond (adsorption of water on nZVI surface). In addition, the bands at 583 cm−1 corresponding to Fe–O stretch for the existing iron oxides (Jin et al., 2015) were observed in Figure 6(a) and (b), which indicates that the part of the nZVI in nZVI/PANI/APT has been oxidized. Furthermore, new bands at 1671, 1341, and 1242 cm−1 correspond to C=O, –NO2, C–O stretching vibrations (Ayed et al., 2010), illustrating that AYR are still adsorbed by the composite material after decomposing into small molecules.

FT-IR spectra of nZVI/PANI/ATP: (a) before the removal of alizarin yellow R and (b) after the removal of alizarin yellow R.
The Fe 2p XPS spectra of nZVI/PANI/APT composites before (a) and after (b) the removal of azo dyes were presented in Figure 4. The main peaks at 707.5, 711.60, and 725.45 eV correspond to Fe0, Fe3O4, and Fe2O3, respectively (Li et al., 2015; Lu et al., 2014). It is known that a small amount of nanometer iron was oxidized into Fe3O4 and Fe2O3 due to not coated PANI, which agrees with the IR spectra. And the adsorption peak intensity of 711.60 and 725.45 eV has no obvious change, which proved that the amount of iron oxide without change on the removal reaction for azo dyes. The peak intensity of 705.26 eV (Fe0) at line b relative to the line a decreased significantly, indicating the amount of zero valent iron reduced due to the removal reaction caused. The changes of the main element content before and after the removal of AYR were studied to obtain the same removal mechanism as Fe was reduced (Table 2).

Fe 2p X-ray photoelectron spectra of nZVI/PANI/ATP: (a) before removal alizarin yellow R and (b) after the removal of alizarin yellow R.
Element composition of nZVI/PANI/APT.

Removal of dyes on nZVI/PANI/APT
Influence of pH on the removal of AYR and MB
The pH of solution is an important factor to effect the removal property of composite materials (Fan et al., 2015). The hydrogen ion concentration (pH) primarily affects the degree of ionization of the dyes and the surface properties of the adsorbent. The effect of pH on removal efficiency of nZVI/PANI/APT was shown in Figure 5. It was indicated that the removal efficiency of dyes was dependent on varying pH conditions. Acidic condition favored AYR removal, the removal of AYR increased as pH value decreased, and the maximum efficiency was observed at pH = 3 (99.56% after 10 minutes). The possible reason for that is more dissolved Fe2+ and electrons are generated from the nZVI/PANI/APT under the acidic conditions, which is a crucial step for azo dyes (Chen et al., 2013; Xin et al., 2010). And the increase in the concentration of H+ was favorable to the formation of [H] in the reaction solution, which can speed the removal of AYR (Sun et al., 2013). Moreover, the removal of AYR was restrained at alkaline pH range because OH− would enhance significantly the formation of the iron hydroxide occupying the reactive sites to inhibit the reaction of dyes (Bokare et al., 2008; Zhang, 2003). The result demonstrated that nZVI of the composite material plays a key role in the removal of AYR. However, the opposite rule for MB removal was shown in Figure 5. The highest removal efficiency of MB was 30% at pH 11 after 30 minutes. This can be attributed to the effect of pH on the charge of reactive group within the adsorbents, which in turn makes them more effective at adsorbing MB in alkaline pH value. At low pH, the surfaces of the composite materials are positively charged, according to the principle of same charges repel, which can weaken the removal properties of the positively charged MB cations by the remover through electrostatic forces of attraction. With increasing pH of the solution, fracturing of Si–O–Si of APT occurred in the role of proton by the formation of Si–OH groups, which lead to an increase in the number of active sites and the removal capacity of MB (Zhang et al., 2014). And the number of composite materials of Si–OH groups is certain; therefore, the removal efficiency of MB remains constant when the pH was above 9. A similar observation was reported by Salman et al. (2012). In order to further study the influence of pH on the removal of dyes, the changes of pH of the solution before and after the removal of dyes were measured. It is found that the pH of AYR solution was increased from 2.6 to 3.2, and the pH of MB solution remained the same. The results indicated that the acidic environment was beneficial for the removal of AYR, with the consumption of a certain amount of H+. However, H+ did not participate in the removal process of MB.

The influence of pH on the removal of alizarin yellow R and methylene blue.
The Fe2+ content in the solution after the removal of AYR and MB
On the basis of the content of Fe2+ in the solution after the removal of AYR and MB, as shown in Figure 6, it was observed that the contents of Fe2+ increased rapidly at the initial stage of the removal of AYR and then became gently above 60 minutes. Meanwhile, the content of Fe2+ remained much lower after the removal of MB. It is attributed to the nZVI that plays an important role in the removal of AYR dyes, which dissolved Fe2+ obtained from the degradation process that nZVI was oxidized after the removal of AYR. And there was almost no chemical reduction in the removal of MB.

The Fe2+ content after the removal of alizarin yellow R and methylene blue in the solution.
Influence of initial concentration of dye and adsorption isotherms
Effect of initial concentrations of AYR and MB solution was carried out as presented in Figures 7 and 8 by using different concentrations of AYR and MB at three temperatures. The results indicated that the removal capacity of dyes on nZVI/PANI/APT increases sharply in the initial concentration and then gradually becomes flat until it reaches a fixed value.

Adsorption isotherms of alizarin yellow R on nZVI/PANI/ATP.

Adsorption isotherms of methylene blue on nZVI/PANI/ATP.
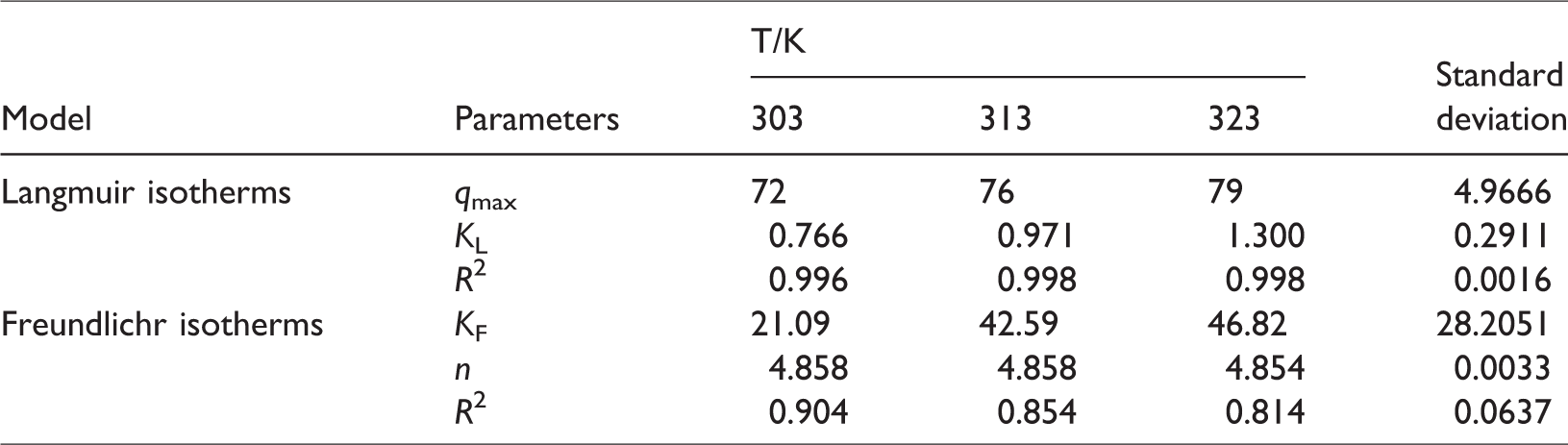
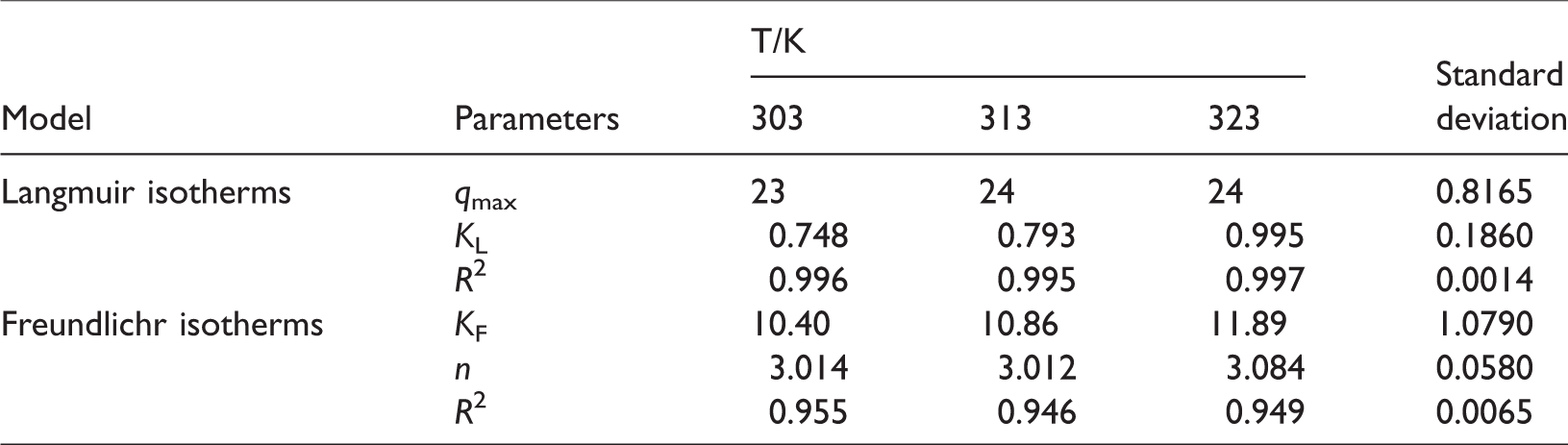
The results of the AYR and MB concentration dependent study were subjected to evaluate by means of Langmuir and Freundlich adsorption isotherm models. The Langmuir equation (equation (3)), which is valid for monolayer sorption on a surface containing a finite number of sites, predicts a homogeneous distribution of sorption energies. The Freundlich equation (equation (4)) was basically empirical in nature and deduces that the adsorption occurs on heterogeneous surfaces.


Isotherm parameters for the adsorption of alizarin yellow R on nZVI/PANI/APT.
Isotherm parameters for the adsorption of methylene blue on nZVI/PANI/APT.
Reusability and stability of nZVI/PANI/APT
Regeneration and stability of the nZVI/PANI/APT composite for the removal of dyes from wastewater are also important properties for the evaluation of its practical usage performance. In this work, regeneration and recycling of the nZVI/PANI/APT composite are attained by water washing and oscillation treatment. After five times, the removal efficiency of AYR dropped to 48%, and the removal efficiency of MB almost unchanged. This is due to when AYR is removed, the consumption of nZVI gradually increases with the increase of cycle usa; in contrast, nZVI on the MB removal is not consumed. It is from another aspect indicated that the degradation of AYR by nZVI/PANI/APT and adsorption of MB on PANI/APT.
NZVI particles tend to aggregate and easy to form oxide layers on the particle surface, resulting in reducing lifetime and reactivity. Thus, Figure 11 shows nZVI/PANI/APT placed in different storage time for the removal of ARY to evaluate the activity of composite material. It can be noted from Figure 9 that nZVI/PANI/APT exposed in the air maintains high catalytic activity after 45 days, and the removal efficiency exceeded 94%. It is further illustrated that conductive polymer PANI played an important role in maintaining the reactivity of nZVI. PANI coating nZVI particles not only prevent nZVI from forming Fe3O4 and Fe2O3 on the particle surface, but also contribute to the electron transfer between nZVI particles and contaminants to increase the activity of nZVI/PANI/APT. This confirms that the presence of PANI/APT as support materials caused dispersion and stabilization of nZVI particles, leading to enhanced reactivity of nZVI.

The influence of storage time on the removal of AYB dyes.
The removal of two kinds of dyes on nZVI/PANI/APT or PANI/APT
To further investigate the different removal performances for dyes on ZVI/PANI/APT, the removal efficiencies of a serious of dyes using nZVI/PANI/APT and PANI/APT are presented in Figure 10. Iron content was the same in all experiments performed when using PANI/APT-supported nZVI materials. It was found that the removal efficiencies of azo dyes (AYR, MR, CBT, and MO) using ZVI/PANI/APT was greater than that of non-azo dyes (MB and RB). The removal efficiencies of non-azo dyes were about 20%–30%, while the removal of azo dyes exceeded 90%. In addition to, the removal efficiency of azo dyes using nZVI/PANI/APT was distinctly superior to that of using PANI/APT. This confirms that the nZVI were used for reductive degradation of the azo dye, PANI/APT as support materials caused chemical adsorption of dyes, which indicates that PANI/APT-supported nZVI system has synergetic effect responsible for enhanced azo dye removal.

The removal of different dyes on nZVI/PANI/ATP or PANI/ATP.
Relevant mechanisms
Figure 11 illustrates the UV–Vis spectra of AYR before and after the reaction with nZVI/PANI/APT, using a standard solution containing an initial concentration of 100 mg·L−1, where the band at 400 nm originated from the –N=N– bond of AYR (Chen et al., 2009), and bands at 249, 303 nm related to aromatic rings were observed. As shown in Figure 11, the band at 400 nm was disappeared completely when the nZVI/PANI/APT was added to the solution containing 100 mg·L−1 AYR. This indicates that the degradation of AYR was reduced by the nZVI/PANI/APT, and then nZVI was first oxidized to provide electron and then the dye molecule accepted electron to form intermediate just as the relevant literatures described (Fu et al., 2014; Nam and Tratnyek, 2000). It implies that the chromophoric structure –N=N– of AYR is cleavaged commonly. However, the absorbance bands at 298 nm are weakened but not disappeared in the end, which suggests that the bonds benzene amino group and phenolic group cannot be completely degraded in the experiment. The intermediate products of AYR degradation by Fe0 decomposed into small molecules may proceed in a longer time or combine with Fenton reaction (Fenglian et al., 2014). Further detailed work for the AYR decolorization procedure is needed to investigate. But the color of azo dyes degrease remarkably by using PANI/APT-supported nZVI. Figure 12 illustrates the UV–Vis spectra of MB before and after the reaction with nZVI/PANI/APT; it was observed that all the characteristic peaks only decreased in intensity. It implied that nZVI did not react with the molecule of non-azo dyes and PANI/APT as support materials caused chemical adsorption of dyes. The adsorption mechanism is inferred that the adsorption of MB on PANI and APT, respectively. The first is that the amino group of PANI can be protonated, which can form hydrogen bond with the N atom of MB frame to achieve the purpose of chemical adsorption, under the condition of weak acid or alkaline. Meanwhile, there is a large specific surface area and a negative charge on APT, so the cation-exchange capacity can be carried out with the cationic dye, which forms a hydrogen bond with the adsorption nucleus.

UV–visible spectral of nZVI/PANI/ATP: (a) before the removal and (b) after reaction with nZVI/PANI/ATP.

UV–visible spectral of methylene blue: (a) before reaction with nZVI/PANI/APT and (b) after reaction with nZVI/PANI/APT.
On the basis of the results analyzed above, relevant mechanisms of the nZVI/PANI/APT composite toward for the degradation of azo dyes (e.g., AYR) are proposed. The first step, AYR is adsorbed onto PANI/APT surface and the oxide layer of nZVI; the second step, its chemical reduction by nZVI nanoparticles, which led to a breakage of the –N=N– bond; finally, the intermediates are decomposed into small molecules by the reaction of H+. In summary, the possible mechanism can be described as follows


Conclusions
The composite PANI/APT-supported nZVI was synthesized by sodium borohydride reduction method. Among them, APT as dispersant role support material can disperse nZVI to prevent the aggregation, and PANI as a stabilizer can effectively prevent the oxidation of nZVI by oxygen in the air and accelerate electron transfer between nZVI particles and contaminants. Therefore, the reactivity of nZVI is greatly increased and the cost is reduced while using PANI and APT in the composite materials. And in this study, nZVI/PANI/APT has been used to effectively remove azo dyes compared with non-azo dyes. The varying removal efficiencies of dyes depend on the different degradation mechanisms. The removal of azo dyes by nZVI/PANI/APT was breakage of the N=N bond and the formation of new degradation products during the removal of non-azo dyes was only chemical adsorption. Therefore, the removal efficiencies of non-azo dyes were about 20%–30% while the removal of azo dyes exceeded 90%, which demonstrated that nZVI/PANI/APT has potential applications in the removal of azo dyes from wastewaters.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NSFC) (51763015, 51503092), the Foundation of Key Laboratory of Clay Mineral Applied Research of Gansu Province, Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences (CMAR-04), and the Foundation for Innovation Groups of Basic Research in Gansu Province (no. 1606RJIA322).
