Abstract
One of the main challenges of the ammonia-based CO2 capture process is how to further reduce the regeneration energy consumption. An antisolvent crystallization method was proposed to strengthen the crystallization process of carbonized ammonia, and heating the crystal products instead of rich solution can greatly reduce renewable energy consumption. The main component of the crystal product was NH4HCO3 analyzed by X-ray diffraction. Therefore, it is very important to study the thermodynamic properties of ammonium bicarbonate in the ternary system of NH3–H2O–ethanol. In this paper, the solubility curves under different temperature and solvent compositions were determined by static method and the mathematical model of solubility was established. The effects of the addition of ammonium carbamate which was produced in the initial stage of the CO2 absorption process and the addition of ammonia on the solubility in ternary system of NH4HCO3–H2O–ethanol were studied. The dissolution heat was also calculated in the end. The research of this article has a guiding significance for the antisolvent method to strengthen the crystallization of carbonized ammonia of ammonia-based carbon capture technology. It is helpful to further strengthen the crystallization process of low carbonized ammonia and improve the crystallization yield.
Introduction
The artificial emission of a large number of CO2 is one of the main causes of global climate change, the concentration of CO2 in the atmosphere has increased by 43% from the industrial revolution, and the pressure of emission reduction is huge (Espinal et al., 2013). The most potential way for large-scale and effective emission reduction of CO2 is CO2 capture and sequestration determined by coal-based energy structure of China. The fast absorption, simple process, and large processing ability are the advantages of chemical absorption, but the disadvantages such as high energy consumption and serious corrosion of equipment also exist at the same time (Rochelle, 2009). The main factor that limits the large-scale industrial application is the high cost of carbon capture (Joos et al., 2016). The key to cut down the cost of carbon capture is to reduce the regeneration energy consumption of rich solution. For the main absorption system of organic amine, the energy consumption of regeneration mainly includes the energy required by the CO2 desorption process and the latent heat of water in the solution (Zhao et al., 2015). In order to reduce the latent heat of water evaporation in the solution, at present, researches at home and abroad mainly focus on the development of anhydrous or less water absorbent, new types of ionic liquids, and phase transformation absorbent (Chen et al., 2016).
In the field of absorbent development, ammonia absorbent mixed with PZ from University of Texas (Du et al., 2017), new types of ionic liquids and ammonia from Australian CSIRO (Li et al., 2015, 2016; Sun et al., 2015; Zhu et al., 2014), and alkali metal adsorbent from Korean KIER (Lee et al., 2016) are the representatives of first generation and the second generation of commercialized absorption technologies. Fang et al. (2014, 2015) were devoted to the research of the new type of amine-based absorbent, more than 20 kinds of high efficient mixed amine absorbents were developed. Tang et al. (2016) were mainly engaged in thermodynamics and kinetics-related studies of alcamines and mixed absorbent. Zhang et al. (2016) and Yu et al. (2016) have developed a new phase transformation absorbent, where the energy consumed by the latent heat of water in traditional absorbent is saved. Li et al. (2017) and Dai et al. (2016) exploited ionic absorbent with low volatility, low viscosity, and high absorbency, which will reduce the volatilization of absorbents during regeneration. Jiang et al. (2017) and Wang et al. (2016) were engaged in the research and development of high performance anhydrous oxide adsorption materials. Luo et al. (2018) proposed a promising renewable absorbent for simultaneous removal of CO2/SO2/NOx, which made a foundation for following research in this field. Yu et al. (2016) and Wei et al. (2015) were committed to the research of CO2 capture technology for dry removal of flue gas by potassium-based absorbent. However, little research has been done on the carbonized ammonia in the process of CO2 capture process. Because of its high carbon dioxide loading, nonoxidative degradation, low price, and the potential for joint removal, CO2 capture by ammonia has drawn lots of attentions, but the large-scale industrial application was limited by high energy consumption of the regeneration process. Strube et al. (2011) found the formation of ammonium salts in the experimental CO2 capture with a solvent mixture of aqueous ammonia and ethanol (EAA). And they thought if CO2 can be captured in the form of ammonium salts, energy requirements are greatly reduced. Based on this research, an antisolvent crystallization method was proposed to strengthen the crystallization of carbonized ammonia and heating the crystal products instead of rich solution can greatly reduce renewable energy consumption, by this way the blank of the research on reducing energy consumption was made up in ammonia recovery process (Gao et al., 2015; Zhang et al., 2016). The main component of the crystal product which is produced by adding ethanol into the carbonized ammonia was NH4HCO3 analyzed by XRD (Gao et al., 2015). Therefore, it is very important to study the thermodynamic properties of ammonium bicarbonate in the ternary system of NH3–H2O–ethanol which cannot be found anywhere else. In this paper, the solubility curve and the metastable zone in the ternary system of NH3–H2O–antisolvent were determined by laser dynamic method, and the mathematical model of solubility was established.
Materials and methods
Reagents
Analytical ammonium bicarbonate and ammonia solution (AR) were purchased from Tianjin Recovery Fine Chemicals Institute (China), and ethanol absolute (analytically pure) was purchased from Harbin Chemicals Institute and used without further purification. Deionized water and sulfuric acid (0.05 mol/l) were supplied from Harbin Zhongjia Chemical Reagent Co., Ltd.
Experimental method
The accuracy of data measured by static method is very high, which is suitable for the method of this research. The equipment diagram is shown in Figure 1. A mixed solvent of different solvents is placed in the glass jacket crystallizer, the super thermostatic water bath is set at a specific temperature, and the agitator turns on at the same time. When the temperature of the mixed solvent is in accordance with the set temperature, the excess ammonium bicarbonate is put into the mixed solvent for stirring for 24 h and the agitator is closed. Titrating (Zhang et al., 2016) the upper clear solution to make sure the amount of ammonium bicarbonate in solution when the liquid appears distinct stratification. In order to improve the efficiency and accuracy of measurement, first of all, the measurement points need to be measured according to the above method. Each test point was repeated at least three times. In the reasonable range of

Equipment diagram of reaction system.
Molar fraction solubility 

Solubility model
Aiming at solving the problem of solid–liquid equilibrium for the solute in binary solvent system, the CNIBS/Redlich–Kister equation was proposed to calculate the solubility of solutes in binary solvent system (Buchowski, 1986; Domańska et al., 1987). The expression is as follows
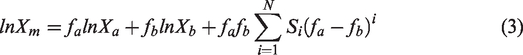

Further simplifying equation (4), equation (5) can be obtained as follows
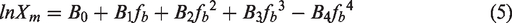
Dissolution heat
The determination of heat of dissolution (Ulrich and Omar, 2006) is usually based on the solubility method and the equation is as follows

Discussion and results
Effect of solvent composition on solubility
According to the theory of static electricity (Huang, 1983), by adding of a polar soluble organics to an electrolyte solution, the hydrogen bonds or other molecular forces between organic polar molecules and water molecules make them close to each other, resulting in an increase in the amount of water molecules interacting with organic polar molecules. Thus, the solubility of electrolytes in mixed solvents is reduced and electrolytes precipitate in the form of solid and this process is called antisolvent crystallization. As mentioned above, in the system of electrolyte–nonelectrolyte–water, there are static electricity competition among particles, such as electrostatic interaction between nonelectrolyte molecules and water molecules, and between ions and water molecules. Therefore, the quantity of nonelectrolyte molecules is the main factor affecting the competition of static electricity. The amount of nonelectrolyte, that is the amount of antisolvent increases, and the interaction between the antisolvent molecule and the water molecule is dominant. As shown in Figure 2, the solubility of ammonium bicarbonate decreases with the increase of antisolvent content in the mixed solvent. With the increase of the content of antisolvent in the mixed solvent, the solubility of ammonium bicarbonate decreases with the temperature, and the solubility of ammonium bicarbonate tends to be 0 when the molar fraction of antisolvent exceeds 0.6. At the initial stage of adding antisolvent, the solubility changes obviously with the antisolvent dose. When the molar fraction is bigger than 0.3, the solubility changes slowly. It is helpful for us to choose the suitable amount of antisolvent in this process.

The change of solubility with solvent components at different temperatures.
Effect of temperature on solubility
The kinetic energy of the particles in the solution which is composed of electrolyte–anelectrolyte–water can be changed by temperature, and then the electrostatic force between the particles is changed which influences the solubility. As shown in Figure 3, the solubility of ammonium bicarbonate in the mixed solvent increases with the increase of temperature, and the trend slows down with the increasing amount of antisolvent in the mixed solvents. The main reason is that the influence of temperature is not dominant when the antisolvent dose is large, this time the electrostatic force between particles is mainly controlled by the amount of antisolvent. When the temperature is below 15°C which is the relative low temperature, the solubility curve does not change obviously with the temperature, and the influence of temperature becomes weaker with the increase of the amount of antisolvent. When the temperature is above 15°C which is the relative high temperature, the solubility is influenced evidently by temperature. The solubility curves in mixed solvents with different molar fraction are approximately parallel lines.
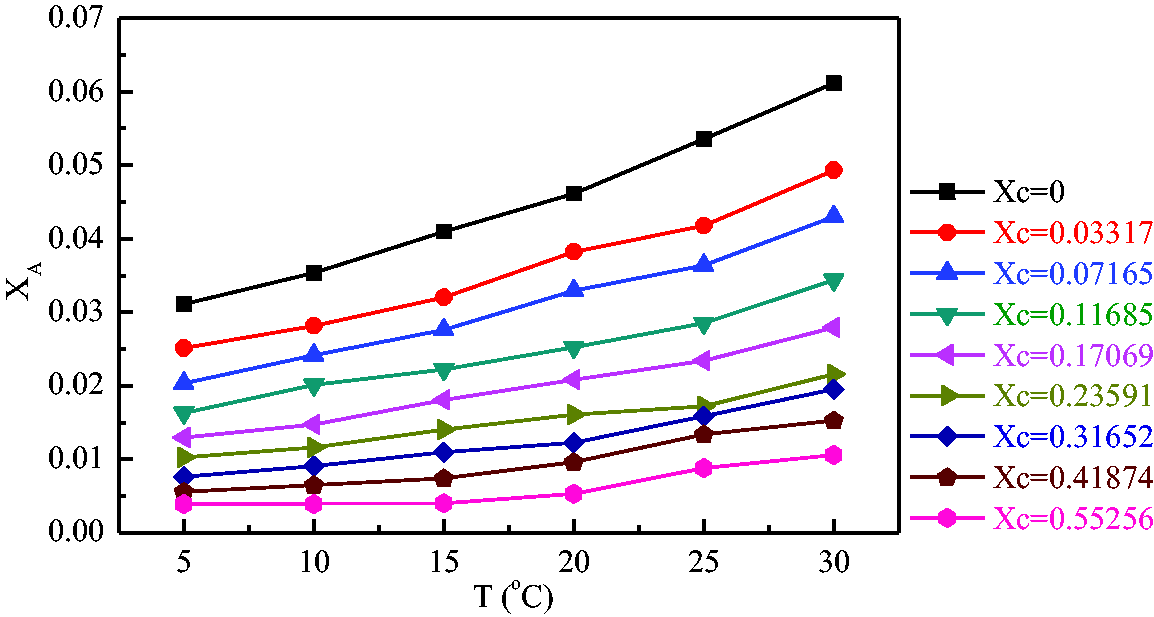
The change of solubility with temperature at different solvent components.
Effect of the existence of ammonium carbamate
Some researchers found that in the early stage of absorption, the concentration of NH2COO− almost accounted for two-thirds of all carbon ions (Yeh and Bai, 1999). With the further carbonation reaction, the concentration of NH2COO− gradually decreases, but there are still one-sixth amount of ammonium bicarbonate in the traditional process before the rich solution enters the desorption tower. Therefore, this part mainly focuses on the influence of ammonium carbamate content on the solubility of ammonium bicarbonate when the amount of ammonium carbamate is one-sixth of ammonium bicarbonate. As shown in Figure 4, the solubility curve with ammonium carbamate is lower than that without ammonium carbamate, and the influence of adding ammonium carbamate in the mixed solvent is weakened with the increasing amount of antisolvent.

The effect of the existence of ammonium carbamate on the solubility in mixed solvent.
The addition of ammonium carbamate to electrolyte–anelectrolyte–water system increases the amount of electrolytes. Therefore, water molecules with high permittivity were gathered around ionized ions which are generated by ionization process of ammonium carbamate, which resulted in the further reduction of free water, thus the solubility of ammonium bicarbonate was further reduced. As ammonium carbamate is dissolved in the antisolvent, the effect of ammonium carbamate on the antisolvent molecules is becoming more and more prominent when the amount of the antisolvent is increased, which leads to a reduction in its effect on water molecules. Therefore, the effect of ammonium carbamate on the solubility of ammonium bicarbonate shows a downward trend when the content of antisolvent increases. It can be clearly seen in Figure 5 that when
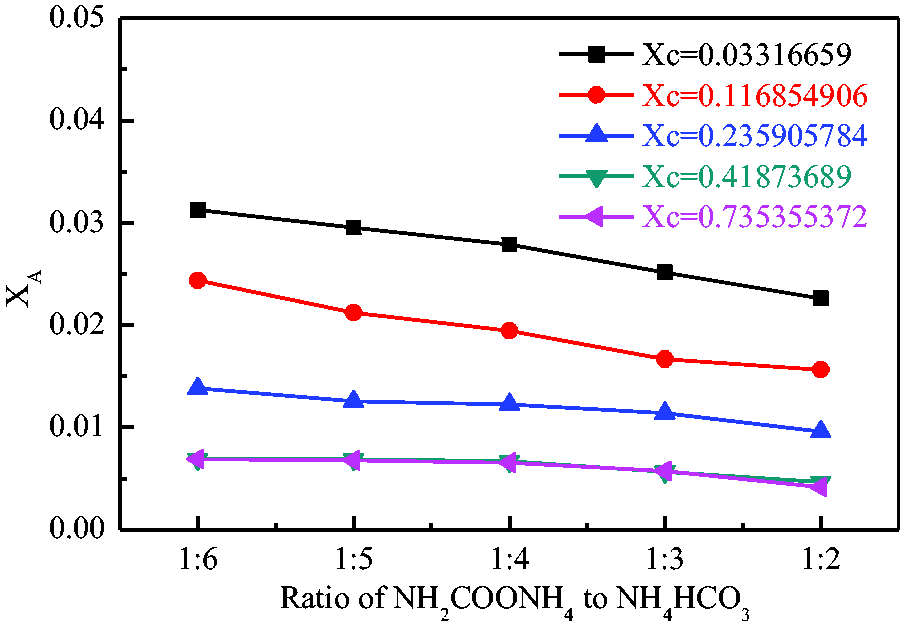
The solubility in mixed solvent changing with the ratio of NH2COONH4 to NH4HCO3.
Effect of the existence of ammonia
In the carbonized ammonia, there are still a certain amount of ammonia, and the content of ammonia in the carbonized ammonia changes with C load of the carbonized ammonia. In Figure 6, we can see that the solubility of ammonium bicarbonate with ammonia is higher than that without ammonia at different temperatures, and this trend is consistent with the increase of

The effect of the existence of ammonia on the solubility in mixed solvent.

Solubility changing with the amount of ammonia.
Solubility correlation equation under different working conditions
The practical objective of fitting the solubility data lies in the prediction of solubility data of the ammonium bicarbonate in different solvent composition (Dong et al., 2000). In this work, the CNIBS/R–K model with high prediction accuracy is verified. The correlation formula (5) is adopted to calculate the model parameters and they are shown in Table 1. The comparison between solubility data and experimental results is shown in Figure 8. It can be seen that the prediction accuracy of the model can meet the industrial needs.

The fitting results of solubility with the change of temperature in binary solvent.
Calculation of solubility by using CNIBS/R–K model.

CNIBS/R-K: Redlich-Kister; RMSD: Root-Mean-Square Deviation.
Dissolution heat
According to equation (6), in Figure 9, the slopes can be used to calculate the dissolution heat, and the calculated results are shown in Table 2. For crystals, dissolution heat is the energy required to destroy the crystal lattice structure, so the dissolution heat can be used to measure the amount of crystal cohesive energy. The large dissolution heat means the energy required to destroy the crystal lattice structure is large, and the energy required to form a crystal lattice is large for the crystallization process. As shown in Table 2, as the value of

The analysis of dissolution heat in binary solvent.
Results of dissolution heat in binary solvent.

Conclusion
An antisolvent crystallization method to strengthen the crystallization of carbonized ammonia was put forward which can improve the absorption efficiency by ammonia with low carbonized ratio recycling and regeneration of crystal products instead of rich solution. The thermodynamic properties of ammonium bicarbonate in the ternary system of NH3–H2O–antisolvent were discussed in this paper, the solubility curve under different temperature and solvent compositions was determined by static method, and the mathematical model of solubility was established. The effects of the amount of ammonium carbamate and ammonia on solubility in the ternary system were studied. In the antisolvent–water binary solvent system, the dissolution heat of ammonium bicarbonate was calculated ranging from 16.41 to 29.17 kJ/mol. The basic research in this paper has a guiding significance for the antisolvent method to strengthen the crystallization of carbonized ammonia of ammonia-based carbon capture technology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank International Technical Cooperation Projects of People’s Republic of China for support of the work: New Generation of Coal Conversion and Power Technology Facing the Sino-US Advanced Coal Technical Cooperation (2010DFA24580-501) and Fundamental Research Funds for the Central Universities (Grant NO.HIT.NSRIF.201174).
