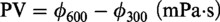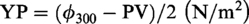Abstract
In this work, a series of ammonium–lauric salts (ALS) was prepared with lauric acid and amines as small molecular shale swelling inhibitor. The inhibitors were screened by the linear expansion test first, and the result shows that the inhibitor prepared by lauric acid and diethylenetriamine with the mole ratio of 2:1 (ALS-2) displays excellent inhibition effect on the hydration expansion of bentonite. The inhibition of ALS-2 to bentonite was fully evaluated by various methods in the following work, including clay linear swelling test and particle distribution measurement. The results show that ALS-2 has superior performance to inhibit the hydration swelling and dispersion of bentonite, and the swelling rate of bentonite in 0.5% ALS-2 was reduced to 29.7%. In water-based drilling fluid, ALS-2 is compatible with the conventional additives, and it can improve the lubricity of the mud cake obviously after aged under 120°C. Besides, it can control the particle size of bentonite in water. The inhibition mechanism of the ammonium–lauric salts was discussed in detail through physical adsorption, scanning electron microscopy, X-ray diffraction, thermogravimetric analysis, and Fourier transform infrared spectroscopy.
Introduction
The shale oil/gas exploration is developing rapidly in recent years. From the beginning, during the drilling, the stability of shale plays a very important role, and the stability control also will affect the drilling costs greatly (Bol, 1986; Lal, 1999). Consequently, a high level of shale inhibition should be concerned during drilling operations. Generally, the ways to inhibit hydration and expansion of clay in shale include the following aspects: the inhibitors can adsorb on the clay surface through electrostatic interaction and form a hydrophobic shell surface on the shale particle, thereby inhibiting the penetration of water into the interlayers (Steiger and Leung, 1992); the coupling force between the clay particles increases through hydrogen bonds, anchoring effect, etc. thus making the interlayer space impermeable for water molecules (Qiu et al., 2011). Inorganic salts have been used as clay expansion inhibitors in the early time, such as ammonium chloride (NH4Cl), potassium chloride (KCl), etc. which are cheap, highly effective shale inhibitors for well-drilling timeframes, but they may easily lead to flocculation under high concentration, and the inhibition is not durable (Van Oort, 2003). Recently, polyammonium has been developed as a kind of excellent clay expansion inhibitor, but these polymers adsorb on the surface of rock, block the formation, and then reduce the permeability of reservoir. Besides, the high toxicity of cationic polymers limits their use either (Deng et al., 2012; Ni et al., 2016; Zhong et al., 2013). Some small molecular ammonium salts, as inhibitors for shale hydration, can avoid such defect above and exhibit ideal inhibitions without any poisonous or hazardous materials. And the use of water-based drilling fluid can avoid the cost of oil contaminated drilled cuttings disposal (Wang et al., 2007; Zhang et al., 2015, 2007). In this work, ammonium–lauric salts (ALSs) were prepared and the properties as clay expansion inhibitors were evaluated through such experiments as linear expansion test, antiswelling test, and particle distribution (Lv et al., 2014; Zhong et al., 2013). Also, the compatibility of the ammonium salt with traditional drilling fluid additives was evaluated. In addition, the inhibition mechanism was discussed in detail.
Materials and methods
Materials and reagents
Diethylenetriamine, triethylenetetramine, and tetraethylenepentamine were purchased from Sinopharm Chemical Reagent Co. Ltd. Triethanolamine and lauric acid were purchased from Tianjin Kemiou Chemical Reagent Co. Ltd, China. Modified starch and xanthan gum were supplied by Yangzhou Oilfield Chemical Co., Ltd, China. Bentonite was obtained from Changqing Bentonite Group Co. Ltd, China, and the chemical composition (wt%) is SiO2 60.54, Al2O3 23.93, FeO 0.23, Fe2O3 4.29, MgO 2.56, CaO 2.78, K2O 1.45, and others 4.22. The main mineral component in bentonite ore is montmorillonite, and impurity minerals are feldspar, quartz, and cristobalite.
Preparation of ALS
A series of ALSs was synthesized using lauric acid and organic amines (diethylenetriamine, triethylenetetramine, tetraethylenepentamine, triethanolamine) as raw materials, and a certain amount of acid and amine was dissolved in water with different ratios, respectively (Chen et al., 2017). The reaction of lauric acid and diethylenetriamine is shown in Figure 1 as an example.

The reaction of lauric acid and diethylenetriamine.
Swelling inhibition
The hydration swelling of bentonite was tested by an NP-01 shale expansion instrument (Haitongda, Co., Ltd, Qingdao, China), in accordance with Chinese Petroleum and Natural Gas Industry Standards SY/T6335-1997. Then antiswelling property of ALS on clay was evaluated in accordance with Chinese Petroleum and Natural Gas Industry Standards SY/T5971-1994.
Drilling fluid properties evaluation experiments
Four percent (m/m) bentonite was dispersed in 350 ml water containing certain amount of inhibitor (Cai et al., 2014; Zhang et al., 2014). After stirred for 20 min, aged for 16 h at room or high temperature, the rheological properties and filtration of the fluid samples were measured using a model ZNN-D6S viscometer (Haitongda, Co., Ltd, Qingdao, China), including apparent viscosity (AV), plastic viscosity (PV), yield point (YP), dynamic plastic ratio (YP/PV), API filtration (FL), and friction coefficient (tg) (Chen and Chen, 2006). The AV, PV, and YP were calculated from 300 and 600 r/min readings using the following formulas from petroleum and natural gas industry standards for field testing of drilling fluids (API RP 13B-1-2009)
Particle distribution test
The size distribution of the particles was measured by LS-13320 laser particle size analyzer based on the light scattering principle (Beckman Coulter, Inc., USA) using equipment operating procedure under the pump speed of 50%.
Characterization
After the 4% bentonite was dispersed in 0.5% inhibitor solutions for 24 h, the bentonite was separated and dried at 105°C for thermogravimetric analysis (TGA), scanning electron microscopy (SEM), Fourier transform infrared (FT-IR) spectroscopy analysis, X-ray diffraction (XRD), and specific surface areas (BET method). The surface morphology of the sample under study in the absence and presence of inhibitors was investigated using a digital microscope imaging scanning electron microscope (Model SU6600, serial no. HI-2102-0003) at an accelerating voltage of 20.0 kV. Samples were attached on the top of an aluminum stopper by means of carbon conductive adhesive tape. All micrographs of the specimen were taken at 5000 times magnification. TGA experiments were performed on a TGA/DSC 1/1600 thermal analysis machine (METTLER TOLEDO, Inc., Switzerland) at a ramp of 10°C/min from 50 to 300°C under a flow of nitrogen. The bentonite powder was pressed into KBr pellets for FT-IR analysis. FT-IR absorption was recorded by a NEXUS FT-IR spectrometer (Thermo Nicolet Corporation), scanning from 4000 to 400 cm−1, with a 4 cm−1 resolution in transmission. XRD patterns were collected on a Rigaku D/MAX-2500 diffractometer with a 2θ range of 5–80° using CuKα radiation (λ = 0.154 nm). The specific surface area and pore size distribution of the samples were measured by Micromeritics ASAP2010 Physical Adsorbent. The samples were pretreated at 180°C for 3 h and processed by BET model.
Results and discussion
Preparation and swelling inhibition
The series of ALSs was prepared from lauric acid and organic amines (diethylenetriamine, triethylenetetramine, tetraethylenepentamine, triethanolamine) as raw materials, through dissolving a certain amount of acid and amine in water with different ratios, respectively. Then the inhibition of 0.5% ALS was evaluated by linear swelling (Table 1). The linear swelling rate of bentonite is impacted by the type of amine and mole ratio of the functional groups. The linear expansion rate of bentonite in ALS-2 solution is much lower than that of the others within 90 min; therefore, ALS-2 was chosen to be studied in detail. As mole ratio of functional group is 1:3, the number of cationic adsorption points is lower than 1:2. As mole ratio of functional group is 1:1, the number of cationic adsorption point increases but the electrostatic force between carboxyl anion and negatively charged clay surface is enhanced, which is not beneficial to the adsorption of inhibitor on the surface, so the inhibition effect of bentonite is weakened.
Name and inhibitory activity of ammonium–lauric acid salts.
Then centrifugal separation was used to determine the antiswelling property of ALS-2 to bentonite, in accordance with Petroleum and Natural Gas industry Standard of China SY/T5971-1994. The results show that swelling rate of ALS-2 presents a tendency of decline first and then increase with the increase of concentration (Figure 2). And the swelling rate was reduced by ALS-2 to the lowest, 29.7%, under the concentration of 0.5%.

The swelling rate varies of ALS-2 with the concentration.
Effect on water-based drilling fluid
The effect of ALS-2 on water-based drilling fluids was evaluated in accordance with API RP 13B-1-2009 (Table 2). Compared with the nonadditive control mud at room temperature, AV, PV, and YP of the mud treated with 0.5% ALS-2 increase slightly, and FL increases dramatically. It is expected that an excess of ALS can result in flocculation due to electrostatic interaction. However, ALS-2 in xanthan gum/modified starch treated with water-based drilling fluids shows well viscosity increasing and filtration reducing abilities. After aged for 16 h at 120°C, the rheological property of the mud was evaluated accordingly. The results show that the viscosity of the drilling fluids treated with ALS-2 is higher than that without inhibitors. ALS-2 showed well friction reducing abilities in modified starch treated with drilling fluid. Besides, it should be noticed that the lubricity of the mud cake of the ALS-2-modified drilling fluid has improved obviously after aged under 120°C, which may be attributed to the hydrophobic chain of lauric group.
The effects of ALS-2 to the rheological performance of drilling fluids as measured in accordance with API RP 13B-1-2009 at different temperature.

ALS-2: lauric acid and diethylenetriamine with the mole ratio of 2:1.
Particle size distribution test
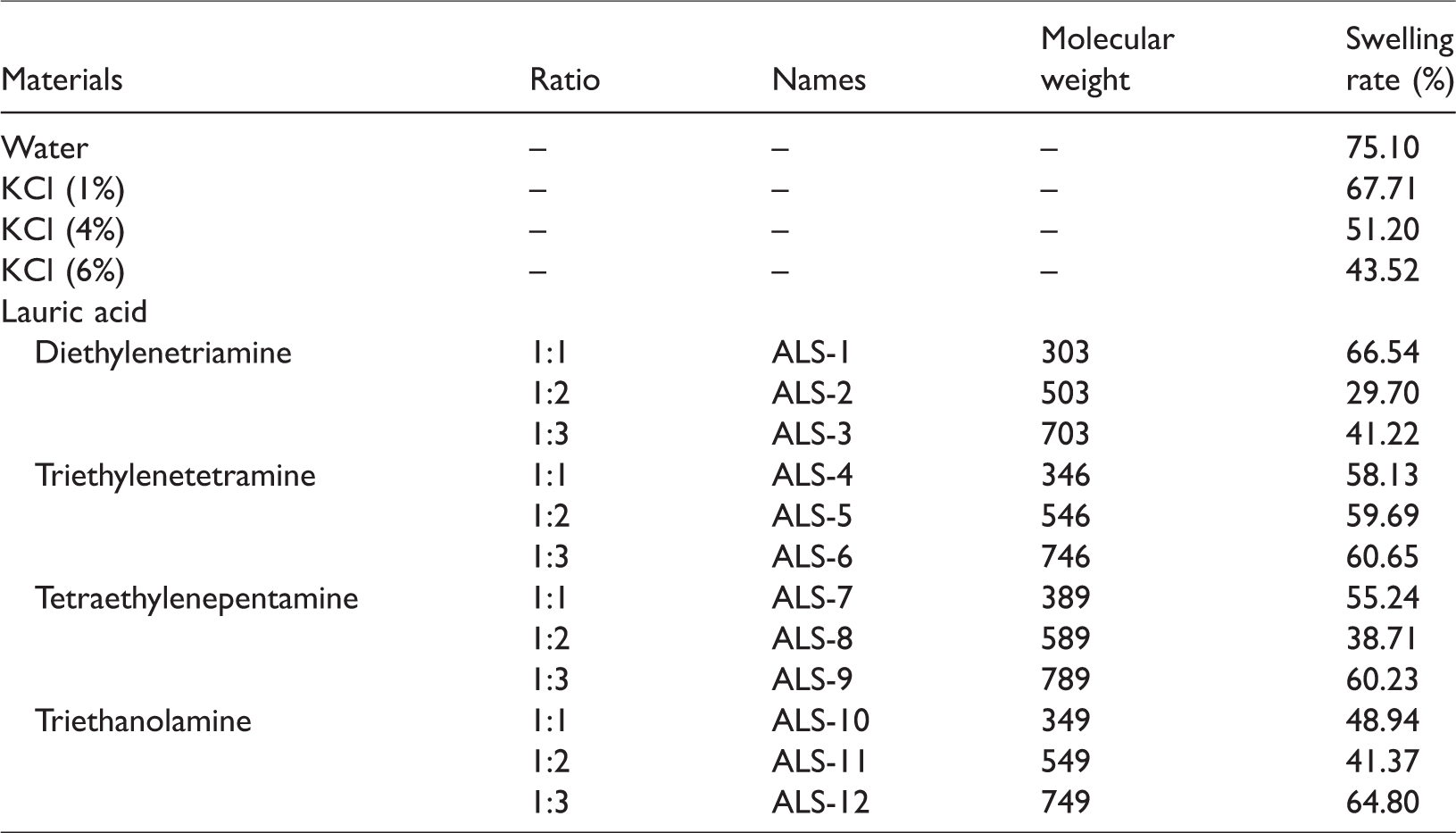
Bentonite is easy to hydrate and disperse to smaller particles, which will increase the viscosity during the drilling process. The effect of ALS-2 on the bentonite particle size in water was investigated (Figure 3 and Table 3). It shows that the bentonite particle size distribution is quite different when treated by different ways. Compared to the unhydrated control sample, the average size of hydrated bentonite particles is reduced from 33.26 to 11.06 µm. And it should be noticed that it is different when ALS-2 was added before and after the hydration. When ALS-2 was added before the virgin bentonite was immersed into water, after 16 h, the average size of clays changed slightly, even it is slightly larger than that of virgin bentonite. When 0.5% ALS-2 was added into the hydrated bentonite suspension, the particle size grows up and is much larger than that of the blank suspension, possibly due to the agglomeration of ALS-2 by the electrostatic interaction. The results indicate that ALS-2 can not only inhibit the swelling and dispersion of bentonite, but also an excess of ALS can result in flocculation due to electrostatic interaction.
Effect of 0.5% ALS-2 on particle size distribution of bentonite.
Effect of 0.5% ALS-2 on particle size of bentonite.

ALS-2: lauric acid and diethylenetriamine with the mole ratio of 2:1.
Textural properties of bentonite treated with water and ALS-2.

ALS-2: lauric acid and diethylenetriamine with the mole ratio of 2:1; BET: ▪.
Physical adsorption characterization
Specific surface areas (BET method) and other textural properties of bentonite samples treated with different ways are summarized in Table 4. For bentonite treated with 0.5% ALS-2, the surface areas, the average pore diameters, and the cumulative pore volumes are quite smaller than that of bentonite treated with water. For example, BET area of bentonite treated with 0.5% ALS-2 is 48.0253 m2/g, and BET area of bentonite treated with water is 61.4085 m2/g, which may be due to the swelling and dispersion of bentonite from large aggregation to smaller lattices. In 0.5% ALS-2 solution, the swelling and dispersion of bentonite were inhibited, and bentonite maintains in large particles and shows relatively small BET area, which shows the effect of inhibition activity of ALS-2 to the hydration expansion of bentonite.
SEM
The SEM was used to evaluate the morphology of bentonite particles treated in different ways. Figure 4(a) shows SEM image of the bentonite treated with 0.5% ALS-2 solution, Figure 4(b) shows SEM image of the bentonite treated with tap water, and Figure 4(c) shows SEM image of the bentonite without any treatment. From the three graphs, it was found that the particles dispersed and changed to smaller particles when immersed in water. It is obvious that the particle size of ALS-2-treated sample is similar to that of the virgin sample, but much larger than that of tap water-treated sample. In other words, ALS-2 can inhibit the hydration, swelling, and dispersion of bentonite, probably by the electrostatic interaction (Qiu et al., 2011).

SEM pictures of bentonite treated with ALS-2 solution (0.5%ALS-2) (a), bentonite treated with water (b), and virgin bentonite (c).
XRD
The XRD was used to evaluate the layer spacing of bentonite treated with water and 0.5% ALS-2 solution. In Figure 5, diffraction peak of bentonite treated with 0.5% ALS-2 is similar with bentonite-treated water, and no new peak was found for the ALS-2-treated bentonite, which suggests that the absorption of ALS-2 has little effect on the crystalline structure of bentonite, and ALS-2 does not crystallize on bentonite. According to the Bragg equation, 2(d) sinθ = λ (d is the interlayer spacing, nm; θ is the diffraction angle 2θ in the XRD spectrum; λ is the wavelength of CuKα radiation, 0.154 nm), the diffraction angle 2θ in the XRD spectrum can be concluded as follows: the interlayer distance of interlayer spacing is 0.3347 nm after treatment with water, and the interlayer spacing is d = 0.3340 nm after treatment with ALS-2 solution, which shows that ALS-2 inhibited the hydration expansion of bentonite and made bentonite maintain in large particles tightly.
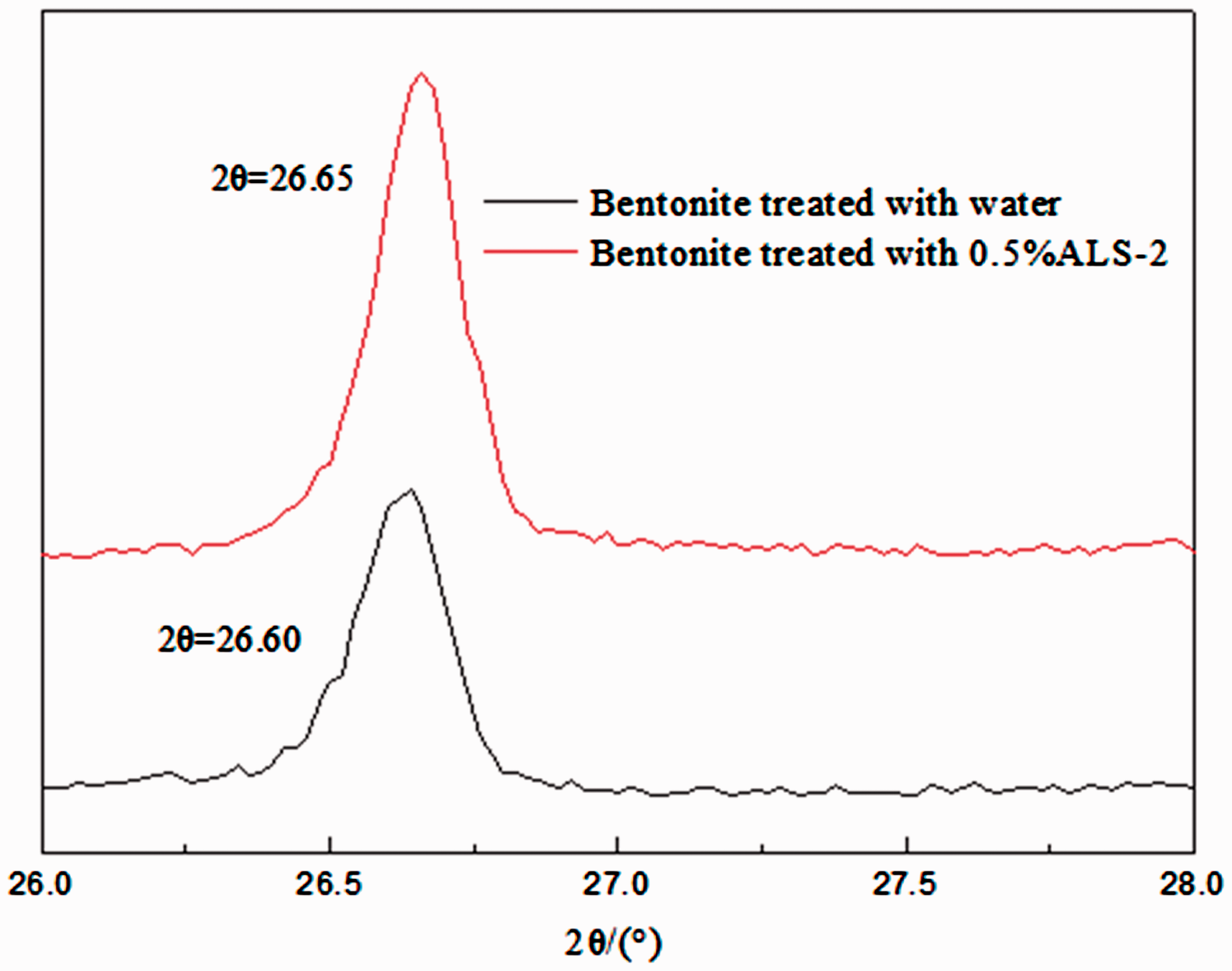
XRD patterns of bentonite treated with water and 0.5%ALS-2.
TGA
The TGA measures the mass loss of a bentonite sample as a function of temperature (Figure 6). Obvious mass loss steps are observed in the curves of bentonite treated in tap water and ALS-2 solution. And the thermal degradation of the ALS-2-treated bentonites differs from that of the control bentonite between 50 and 300°C significantly. The weight loss of bentonite treated with tap water is 1.6% from 50 to 100°C, while the weight loss of ALS-2-modified bentonite is no more than 1.0%. And the weight loss of bentonite treated with tap water is 3.6% within 300°C, while the weight loss of ALS-2-modified bentonite is only 2.4%. It can be concluded that ALS-2 can prevent water molecule permeating into the clay interlayer effectively.

The TGA of the bentonite modified by water and 0.5%ALS-2 solution. ALS-2: lauric acid and diethylenetriamine with the mole ratio of 2:1.
FT-IR analysis
The FT-IR spectroscopy was used to discuss the interaction between bentonite and ammonium–lauric molecules (Figure 7). Compared with sample treated with water, the FT-IR spectrum of bentonite treated with ALS-2 shows absorption band at 2919 and 2850 cm−1, corresponding to the sp3 C–H stretching band of ammonium–lauric molecules. The FT-IR spectrum of bentonite treated with ALS-2 shows absorption band at 1629 cm−1, corresponding to the sp2 C = O stretching band. And 3210 cm−1 is the characteristic absorption, due to inhibitor adsorbs on the bentonite surface by hydrogen bonding. The FT-IR spectrum intensity of the bentonite treated with ALS-2 appears to weaken at 3440 cm−1, which indicates ammonium–lauric molecule can limit water molecules penetrating into clay interlayer. The bands corresponding to Al–Al–OH, Al–Fe–OH, and Al–Mg–OH bending vibrations are observed at 918, 876, and 843 cm−1, respectively. A complex band at 1048 cm−1 is related to the stretching vibrations of Si–O bonds, while the peaks at 525 and 468 cm−1 are due to Al–O–Si and Si–O–Si bending vibrations, respectively. The peak at 622 cm−1 is assigned to coupled Al–O and Si–O out-of-plane vibrations (Madejova et al., 2002). The bending of Si–O–Si in lattice of bentonite at 1040 cm−1 does not change much, which suggests ammonium–lauric inhibitor has little effect on the crystalline structure of bentonite. Based on the data and the analysis above, it can be deduced that ammonium may absorb on bentonite by ion exchange, laurate may absorb on bentonite surface to form a hydrophobic film, as shown in Figure 8, which can prevent water molecule permeating into the clay interlayer effectively.

FT-IR of the bentonite treated with water and ALS-2 solution. ALS-2: lauric acid and diethylenetriamine with the mole ratio of 2:1.

A molecular model for the interaction of ALS and bentonite.
Conclusions
In this work, a series of ALSs was prepared using lauric acid and amines as shale swelling inhibitor. The inhibition properties of ALS-2 to the swelling of bentonite were evaluated by linear expansion tests, etc. The results indicate that ALS-2 shows high inhibition to hydration and swelling of bentonite, and the swelling rate in 0.5% ALS-2 was reduced to as low as 29.7%. ALS-2 is compatible with the conventional additives in water-based drilling fluids, and it can improve the lubricity of the mud cake obviously after aged under 120°C. Besides, it can control the particle size of bentonite changing slightly. The inhibition mechanism of ALS-2 to bentonite should be due to electrostatic interaction and surface modification. The inhibitor adsorbs on the clay surface, and its hydrophobic group contacts with the free water, which prevents free water from entering the clay layer and then inhibits its hydration expansion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the grants from National Science Foundation of China (50874092), Scientific Research Program Funded by Shaanxi Provincial Education Department (17JS114) and Xi'an Science and Technology Planning Project (2017081CGRC044[XASY005]).