Abstract
A novel monomeric amine sodium montmorillonite swelling inhibitor: N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide was obtained from diethanolamine, 1-bromotetradecane, and 1, 2-dibromoethane. N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide was characterized by means of Fourier transform infrared, 1H NMR spectroscopy, elemental analysis, linear swelling tests, particle size distribution tests, X-ray diffraction, and thermogravimetric. Linear swelling tests showed that the swelling height of sodium montmorillonite in 1.0 wt% N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide solution was only 2.0 mm after 16 h (fresh water was 5.0 mm). Particle size distribution tests exhibited that the median diameter and mean particle size of sodium montmorillonite in 1.0 wt% N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide solution obviously increased to 16.1 and 85.4 µm, respectively (fresh water was 8.1 and 21.8 µm). In thermogravimetric tests, in comparison with pure sodium montmorillonite, the decrease of water content in sodium montmorillonite/N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide indicated N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide expelled the water molecules out of the interlayer, which was beneficial to wellbore stability. Fourier transform infrared spectra of certain concentration N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide/sodium montmorillonite indicated the successful physical adsorption and interaction between N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide with sodium montmorillonite. In addition, the results of X-ray diffraction tests showed the obtained 1.0 wt% N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide solution could remarkably reduce the interlayer distance of wet sodium montmorillonite (from 1.94 to 1.37 nm).
Introduction
Wellbore instability has a tremendously adverse impact on drilling operations such as stuck pipe, tight hole, bit balling, etc. (Balaban et al., 2015; Jain et al., 2015; Rossi et al., 2003). Over the past decades, wellbore stabilizer, namely sodium montmorillonite (Na-MMT) swelling inhibitor or shale inhibitor, has been employed to relieve or solve the problems of wellbore instability (Anderson et al., 2010; Boek et al., 1995; Kelland, 2009). Initially, simple inorganic salts Na-MMT swelling inhibitor was utilized to prevent clay-related damage. However, as soon as the inorganic salts are depleted or displaced by fresh water, the shale will again hydrate and swell (Salles et al., 2013; Wilson and Wilson, 2014). To combat the performance-related issues associated with inorganic salts, owing to the low hydration energy and hydrated volume, various monomeric amines have been applied in the oilfield with local success (Fink, 2011). However, the poor resistance of temperature, strong odors, toxicity, and low level of Na-MMT swelling inhibition has limited the application of these monomeric amines (Gomez and Patel, 2013; Xuan et al., 2013).
Surface-active monomer, which contains hydrophobic tail or head group, has been widely used in biological simulations, cereals, and water soluble hydrophobically associating polymers. In petroleum industry, surfactants are widely used in enhanced oil recovery and drilling fluids. Surfactants could adsorb onto the surface of Na-MMT owing to silanol groups and negative charges on the Na-MMT, thereby suppressing the Na-MMT swelling (Shadizadeh et al., 2015). Suter et al. (2011) have devised a set of “rule-based” design criteria for clay-swelling inhibitors; they proposed that the ideal cationic inhibitor should possess a water soluble, hydrophobic backbone and have primary diamine or mono-quaternary amine functionality. In the past years, cationic and nonionic surfactants have been developed as Na-MMT swelling inhibitor. However, to our best knowledge, Gemini surfactant monomeric amine Na-MMT swelling inhibitor has seldom reported.
In the present work, a novel monomeric amine Na-MMT swelling inhibitor, which contains hydrophobic backbone and quaternary amine as well as alcohol functionality, was obtained from diethanolamine, 1-bromotetradecane, and 1,2-dibromoethane (Figure 1). The monomeric amine N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide (DTHDB) was characterized by FT-IR, 1H NMR spectroscopy, elemental analysis, linear swelling tests, particle distribution tests, thermogravimetric (TG), and X-ray diffraction (XRD). Evaluation results demonstrated that DTHDB was a potential Na-MMT swelling inhibitor.
Preparation of DTHDB. DTHDB: N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide.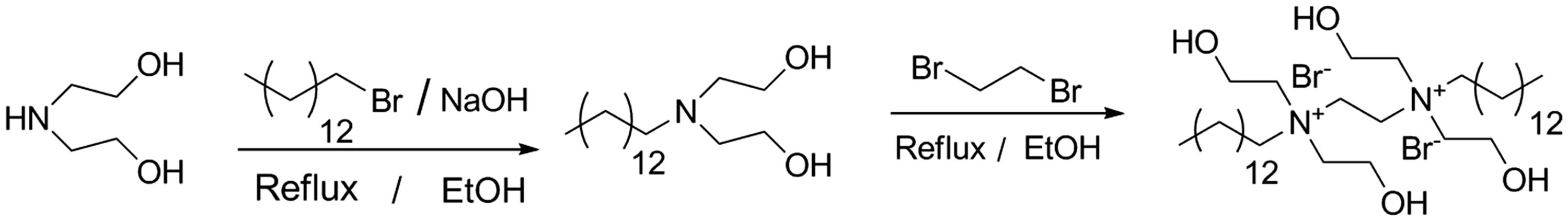
Experimental
Material
Diethanolamine, 1-bromotetradecane, 1,2-dibromoethane, tetrabutyl ammonium bromide (TBA), and K2SiO3 were purchased from Aladdin Reagent Co., Ltd, Shanghai, China; ethanol, KCl, choline chloride, sodium hydroxide, ether, and anhydrous magnesium sulfate were purchased from Kelon Chemical Reagent Factory Co, Chengdu, China. All the chemicals used were analytical grade chemical and used without further purification. TAAC (quaternary ammonium salts) and a diquat with smaller carbon chain was prepared based on the reported methods (Pu et al., 2015, 2016). Na-MMT with cation exchange capacity of 81.3 mmol/100 g was obtained from Xia Zijie Bentonite Technology Co., Ltd. The elemental composition of the raw sample was Si, 58.7 wt%; Al, 20.2 wt%; Na, 46 wt%; Fe, 8.1 wt%; Mg, 3.6 wt%; K, 3.1 wt%; Ca, 0.8 wt%; Ti, 0.9 wt%.; 5# white oil was purchased from Shanghai Lubricant Co., Ltd, Shanghai, China.
Synthesis
Preparation of 2,2′-(propylazanediyl) diethanol
A total of 27.73 g 1-bromotetradecane (0.10 mol) was dissolved in 150 ml EtOH, when the temperature rose to 70–85℃, 10.51 g diethanolamine (0.10 mol) was added into the flask and kept stirred under argon for 48 h. After cooling to room temperature (rt), 4.0 g sodium hydroxide was added and the mixture was stirred for another 1 h, ethanol was removed by evaporation on a rotavapor. Deionized water (150 ml) and ether (100 ml) were added and the mixture separated into two layers. Water layer was extracted with ether several times and extracts were combined. The ether solution was washed with water five times and then dried with anhydrous magnesium sulfate. The ether was removed under reduced pressure. Product isolated in the form of colorless liquid did not require further purification. Yield: 73.50%. IR (KBr, cm−1): 3358, 2960, 2925, 1466, 1377, 1149, 1045, 721; 1H NMR (400 MHz, DMSO) δ 5.99–6.31(m, 1H), 4.28 (s, 2H), 3.40 (t, J = 5.8 Hz, 4H), 2.48 (t, J = 6.4 Hz, 4H), 2.41 (t, 2H), 1.36 (t, 2H), 1.24 (s, 20 H), 0.86 (t, J = 6.8 Hz, 3H). Anal. calcd for C18H39NO2:C 70.33, H 12.82, N 5.12; found C 70.12, H 12.67, N 5.08.
Preparation of DTHDB
Into a 50 ml, round bottomed flask, equipped with reflux condenser and a positive flow of nitrogen, was placed preparation of 2,2′-(propylazanediyl)diethanol (0.2 mol), 1,2-dibromoethane (0.08 mol), and absolute EtOH (20 ml). The mixture was refluxed at 60℃ for 24 h. After slowly being cooled to rt, crude sample was concentrated under reduced pressure. The resulting production was precipitated with petroleum ether (five times) and placed under a vacuum to remove trace solvent. Resulting product was redissolved in acetone solution and then cooled to −10℃ in a dry ice bath for 24 h (Coneski et al., 2012; Wynne et al., 2011). The product was isolated in middle yield as a white powder. Yield: 36.62%. IR (KBr, cm−1): 3266, 2930, 2856, 1648, 1400, 1075, 721; 1H NMR (400 MHz, DMSO) δ 3.95–3.97(m, 8H), 3.63 (s, 4H), 3.68 (s, 4H), 3.42–3.45 (m, 8H), 3.25 (t, 8H), 1.29 (t, J = 5.8 Hz, 36H), 0.89 (t, J = 6.4 Hz, 6H). Anal. calcd for C38H82Br2N2O4:C 55.59, H 10.08, N 8.72; found C 55.63, H 10.06, N 8.75.
Fresh water-based mud preparation
Fresh water-based mud (4 w/v%) was prepared as followed: dissolve 40 g sodium bentonite and 2.4 g Na2CO3 into 1 l fresh water, and stir for 20 min at a high speed of 10,000 r/min. The suspension was left for 16 h at rt for following use.
Fresh water-based mud (1 w/v%) was prepared as followed: dissolve 10 g sodium bentonite and 0.6 g Na2CO3 into 1 l fresh water, and stir for 20 min at a high speed of 10,000 r/min. The suspension was left for 16 h at rt for following use.
Characterization
The FT-IR spectra of products were acquired via a WQF-520 Fourier transform infrared spectrometer in the optical range of 4500–400 cm−1. 1H NMR spectra were performed on a Bruker AV-400 nuclear magnetic resonance spectrometer with chemical shift values in ppm downfield from TMS using DMSO as solvents.
Linear swelling tests
The linear swelling tests were carried out in laboratory by using CPZ-2 swelling apparatus (Dongying, China). 10 ± 0.01 g, 40 mesh Na-MMT which was dried at 100 ± 3℃ for 4 h was pressed tightly into a sample tube by hydraulic press under 10 MPa for 5 min. Then, the sample tube was fixed in the chamber of the swelling apparatus. The change in height of the Na-MMT was recorded over time through the sensor.
Particle size distribution tests
Inhibitors with certain concentrations were added to 4 w/v% fresh water-based mud suspension and stir for 20 min at a high speed of 10,000 r/min, then, the particle distribution tests were measured by laser diffraction technique (HORIBA, Japan).
TG analysis tests
DTHDB/Na-MMT samples were prepared as described in “Particle size distribution tests” section. TG analysis tests were performed on an STA-409 simultaneous TGA instrument (NETZSCH, Germany) under a flowing nitrogen atmosphere at a scan rate of 15℃/min from 30 to 600℃ (pure Na-MMT was from 30 to 400℃). Temperature was reproducible to ± 3℃.
XRD analysis tests
Inhibitors with certain concentrations were added to 1 w/v% fresh water-based mud suspension and stirred for 2 h, then, centrifuged for 30 min and the precipitations collected. X-ray diffraction tests were performed on an X Pert PRO MPD diffract meter.
Results and discussion
Linear swelling tests
The levels of swelling differ from shale to shale and the type of drilling fluids (Patel, 2009), and linear swelling test is often utilized to quantify drilling fluids–shale interaction though there are still clear drawbacks (Oort et al., 2016).
As shown in Figure 2, swelling height increased dramatically during the initial 600 min in fresh water 1.0 wt% TBA, 1.0 wt% diquat, and 1.0 wt% choline chloride solutions and followed by a relatively tardier increase within the rest test time (14 h), except DTHDB and 5# white oil. Indicating that DTHDB provided a nice capacity to inhibitive the hydration and swelling of clay. Swelling height of Na-MMT in any concentration of DTHDB was found to be significantly lower than that of other cases at each time period. The final swelling height of Na-MMT in 1.0 wt% DTHDB and 5# white oil was 2.0 and 0.02 mm after 16 h, respectively. The reasons why the swelling height of Na-MMT in DTHDB was lower than that of other cases were as follows: First, owing to the low molecular weight of DTHDB, it could enter the clay interlayer to compress the diffuse electric double of clay and prevent the hydration expansion of clay. Second, ascribed to the adsorption group hydroxyl and ammonium group, DTHDB would compete with free water for adsorption sites, thus replace the water adsorbed onto the surface of clay. What’s more, the most important was that DTHDB could modify the hydrophilic clay surface to a hydrophobic one, thereby preventing water entering the clay layer. The experiment also verified the common sense that white oil owns the best Na-MMT swelling inhibitive property.
Swelling height of Na-MMT in different inhibitor solutions and 5# white oil. Na-MMT: sodium montmorillonite.
Particle size distribution tests
Laser diffraction technique (HORIBA, Japan) was utilized to study the influence of different inhibitors on 4 w/v% fresh water-based mud particle size distribution, and the results are shown in Figure 3.
Particle size distribution tests with different inhibitors solutions: (a) the initial 50 µm and (b) the whole particle size distribution (µm).
The median diameter and mean particle size of fresh-water based mud were 8.1 and 21.8 µm, respectively. The addition of inhibitors changed the particle size obviously with the sequence of 1.0 wt% TBA < 1.0 wt% KCl < 1.0 wt% diquat < 1.0 wt% K2SiO3 < 1.0 wt% choline chloride < 0.5 wt% DTHDB < 0.7 wt% DTHDB < 1.0 wt% DTHDB. KCl, commonly used as Na-MMT swelling inhibitor, had a high performance in inhibiting Na-MMT hydration owing to the appropriate cationic size and low hydration (Boyd, 2012). The hydroxyl and ammonium group could guarantee the adsorb of choline chloride could compress the diffusion double layer of Na-MMT, and in that way contributed to the aggregation of Na-MMT. With there were hydrophobic long carbon chains on the DTHDB molecule, it could drive water out of crystal layer, reduce the interlayer repulsion force, and make the clay particles become larger. The combined adsorption group and the hydrophobic long carbon chains of DTHDB could drive water out of Na-MMT crystal layer, reduce the interlayer repulsion force, and finally make the clay particles larger. The median diameter and mean particle size of Na-MMT in 1.0 wt% DTHDB solution obviously increased to 16.1 and 85.4 µm.
TG analysis tests
The thermal analysis was carried out to probe the thermal stability and the microenvironment of Na-MMT/inhibitor. As we all know, Na-MMT contains water due to the hydrated Na+ intercalated inside the clay layers (Branda et al., 2010). The TG profiles of pure Na-MMT, pure DTHDB, and Na-MMT with certain DTHDB are exhibited in Figure 4.
TG analysis tests of pure Na-MMT, DTHDB, and Na-MMT/DTHDB. DTHDB: N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide; Na-MMT: sodium montmorillonite.
TG curve of pure Na-MMT showed two step weight losses on the thermal treatment in the range of 30–600℃, and the rapid mass loss between 30 and 190℃ was attributed to the desorption of free water, strongly and weakly bound adsorbed water in the interlayer (Caglar et al., 2013; Letaief et al., 2008; Salles et al., 2008; Xie et al., 2016). The second part was attributed to dehydroxylation ranging from 400 to 600℃. While for DTHDB modified Na-MMT, there were three main steps for the thermal decomposition curves. To begin with, in the range of 30–190℃, the gentle weight decreased corresponding to the evaporated mount water in the interlayer of Na-MMT as well as the decomposition of little mount DTHDB. The second step weight loss in the temperature range of 190–375℃ was due to the thermal decomposition of existing organic DTHDB in the interlayer. The third step mass loss in the range of 375–600℃ was attributed to the decomposed positive ions and the dehydroxylation of hydroxyl groups in cationic structures (Caglar et al., 2015; Ren et al., 2013).
Because of free water and adsorbed water influences the stability of clay, therefore the first step of thermal decomposition was mainly focused. For Na-MMT, the loss of free water (40–125℃) was 2.31 g/g, whereas in the case of Na-MMT/DTHDB, when the DTHDB concentration was 0.5 wt%, the mass loss from dehydration was 2.16, and when the DTHDB concentration increased to 1.0 wt%, the mass loss was 2.08. In comparison with pure Na-MMT, Na-MMT/ DTHDB showed lower mass loss in the range of 30–190℃, implying that less free water existed in the interlayer. These results indicated that DTHDB could expel free water out of Na-MMT, which was beneficial to clay stability, but 0.5 wt% showed little impact on bound adsorb water.
The second step of thermal decomposition ranging from 190 to 375℃ also played a significant role in analyzing the difference between Na-MMT and organically modified Na-MMT. Since DTHDB degraded at about 300℃, the intercalation of the DTHDB into the Na-MMT interlayer appeared to improve the thermal stability, corresponding to higher degradation temperature of Na-MMT/DTHDB around 375℃.
XRD analysis tests
The XRD was used to determine the variation in the interlayer spacing (d-spacing) between Na-MMT sheets. As is known, the Na-MMT interlayer space, i.e. basal spacing d (001) of Na-MMT, is obviously affected by the balanced ions in the interlayer space. Figure 5 shows the wet XRD patterns for the d (001) diffraction peaks of pure wet Na-MMT and wet Na-MMT/ DTHDB.
X-ray diffraction patterns of Na-MMT and modified Na-MMT at different DTHDB concentrations (wt%). (a) dry samples and (b) wet samples. DTHDB: N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide; Na-MMT: sodium montmorillonite.
The XRD patterns of Na-MMT and Na-MMT modified by DTHDB are shown in Figure 5. For the XRD of dry Na-MMT itself, the appearance of a reflection around 6.93° was indicative of regular repetitions of silicate layers, and the corresponding interlayer spacing was 1.28 nm. After modification by DTHDB, the reflections were shifted to lower values, indicating the enlargement of the interlayer spacing. In the case of Na-MMT/DTHDB, when the concentration was 0.5, 0.7, and 1.0 wt%, the interlayer spacing increased to 1.38, 1.44, and 1.46 nm, respectively, indicating the entrance of DTHDB in the interlayer.
In the wet samples, the interlayer distance of pure wet Na-MMT was 1.94 nm, which indicated that the interlayer ions of Na-MMT were sodium ions. After being intercalated by different concentration DTHDB, the diffraction bands were shifted to a higher diffraction angle, which was corresponding to a lower interlayer distance, indicating the decrease of interlayer water (Caglar, 2012; Li et al., 2010). The entrance of DTHDB can increase the spacing of interlayer, and the decrease of the interlayer water can make the interlayer spacing weak. However, the final results were the reduction of the interlayer spacing.
There are numerous hydroxyl and quaternary ammonium groups on the DTHDB, large amounts of hydrogen bondings were formed between the terminal hydroxyl groups and siloxane of clay, which were the driving force for adsorption. Besides, the hydrophobic long chain of DTHDB could prevent water enter the internal of Na-MMT; thus, resulting in a perfect inhibition performance for DTHDB. The interlayer distance of Na-MMT/1.0 wt% DTHDB was 1.37 nm; the phenomenon confirmed that DTHDB effectively inhibited the hydration swelling of Na-MMT.
FT-IR analysis
Na-MMT/ DTHDB precipitations (as described in “XRD analysis tests” section) were washed three times with ethanol to remove the unabsorbed DTHDB, and then the precipitations were dried at 105℃ and powdered.
Figure 6 presents the FT-IR spectra of Na-MMT, DTHDB, and Na-MMT with certain concentration of DTHDB. As shown in Figure 6, the pure Na-MMT shows absorption band at 1000 cm−1, which corresponding to the Si–O stretching vibration of tetrahedral sheet, at 3615 and 1637 cm−1 are attributable to O–H stretching and bending vibrations from both the hydration water and the structural Si–OH, at 773 cm−1 corresponding to the Mg (Li)–(OH) vibration. All spectra display typical absorption bands of Na-MMT (Ren et al., 2013; Kaur et al., 2011).
FT-IR spectrum of Na-MMT, DTHDB, and Na-MMT/DTHDB. DTHDB: N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide; FT-IR: Fourier transform infrared; Na-MMT: sodium montmorillonite.
For the pure DTHDB, the bands at 3266, 1440, 1075 cm−1 corresponding to O–H stretching vibration; C–H bending vibration of –CH3; C–N stretching vibration, respectively. The bands at 2930 and 2856 cm−1 were attributed to stretching vibration and bending vibration of –CH2.
In the case of Na-MMT/DTHDB, the spectrum shows both typical absorption bands of pure Na-MMT and pure DTHDB. Some peaks position assigned to DTHDB were observed without any changes and the intensity intensified with the increasing of DTHDB concentration, such as 2930, 2856, 1440, and 1075 cm−1, which indicated the successful physical adsorption of DTHDB onto Na-MMT. The peaks (1637 and 3615 cm−1) corresponding to the physisorbed water of Na-MMT were weaker when compared to that in the spectrum of the pure Na-MMT, indicating a significant decrease of interlayer water in Na-MMT. Why there was a decrease of interlayer water in Na-MMT/DTHDB? We believed that the two main reasons contributed to it: (1) It is usually considered that there is a competition between hydroxyl and water molecules for the adsorption sites on the Na-MMT. The adsorption seems to be based on ion–dipole interactions between the exchangeable cations of Na-MMT and the hydroxyl functional groups of DTHDB. Therefore, DTHDB rather than water molecules will absorb onto the Na-MMT. (2) The hydrophobic long chains of DTHDB would exclude the water out of Na-MMT interlayer and prevent the entry of other water into the layer. The weakening or disappearance of the shoulder peak at 1000 and 1637 cm−1 corresponding to Si–O–Si and Si–OH bending vibration appears to indicate the hydrogen bonding interaction between hydroxyl groups and O or OH on siloxane surface (Sarier et al., 2010; Xuan et al., 2013)
Inhibition mechanism analysis
Through the inhibition evaluation of DTHDB on Na-MMT via several methods, based on the reported Na-MMT swelling inhibition mechanism analysis (Ferreira et al., 2016; Zhong et al., 2011), a possible inhibition mechanism of DTHDB for Na-MMT swelling might be expected to emerge (Figure 7). The interaction or physical adsorption between DTHDB and Na-MMT might be explained by the multiple driven forces including electrostatic interaction between the quaternary ammonium groups of DTHDB and the negative clay, meanwhile the high adhesive energy of hydrogen bonding groups and so forth. Positive charged DTHDB will adsorb in interlayer of clay instead of water, which would lead to the collapse of the diffuse double layer and further resulting the occurrence of Na-MMT swelling inhibition (Al-Hashmi and Luckham, 2010; Jain et al., 2014; Muppalla and Jewrajka, 2012). What’s more, DTHDB could convert the hydrophilic Na-MMT surface to a hydrophobic one, it will be harder for water to get close to Na-MMT surface, and thus inhibit the Na-MMT swelling.
Inhibition mechanism analysis of DTHDB on Na-MMT. DTHDB: N1,N2-ditetradecy-N1, N1, N2, N2-tetrakis (2-hydroxyethyl) ethane-1,2-diaminium bromide; Na-MMT: sodium montmorillonite.
Conclusions
In summary, a novel monomeric amine shale inhibitor (DTHDB) was prepared by diethanolamine, 1-bromotetradecane, and 1,2-dibromoethane. Traditional inhibition evaluation experiments indicated that the inhibition property of DTHDB was superior to KCl, K2SiO3, and some quaternary ammonium salts Na-MMT swelling inhibitor. The wet XRD patterns showed that after being intercalated by DTHDB, the diffraction band of Na-MMT was shifted to a higher diffraction angle, corresponding to a lower interlayer distance (from 1.94 to 1.37 nm), indicating the decrease of interlayer water in the interlayer. FT-IR spectra of Na-MMT/DTHDB showed the successful incorporation of DTHDB in Na-MMT due to the interactions between the alcohol polar functional groups and the exchangeable cations of Na-MMT as well as H-bonding, van der Waals interactions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Major State Basic Research Development Program of China (973 Program, contract grant number: 2013CB228003) and National High Technology Research and Development Program of China (863 Program, contract grant number: 2012AA091502) for their financial support.
