Abstract
Pb(II) can cause a hazardous effect on ecosystem and public health due to its high biotoxicity. A polyvinylidene fluoride-type membrane bearing both poly(amino phosphonic acid) and poly(amino carboxylic acid) functional groups was fabricated for the purpose of Pb(II) removal from the aqueous solutions. The adsorption behaviors of the fabricated chelating membrane toward Pb(II) were studied by the series of static and continuous adsorption experiments. When the pH, adsorption equilibrium time, initial Pb(II) concentration, and temperature were 5.1, 300 min, 1.0 mmol g−1, and 298 K, respectively, Pb(II) uptake of the membrane was 1.1 mmol g−1. The presence of coexisting metal ions and complexing reagents decreased the Pb(II) uptake. The adsorption kinetic and isotherm adsorption followed pseudo-second-order equation and Langmuir model, respectively; this adsorption process showed a spontaneous and exothermic feature. The bed depth service time and Thomas models were suitable for describing obtained breakthrough curves.
Keywords
Introduction
Water pollution caused by metal pollutants has received tremendous attention due to the increases in their discharged volumes and emission amounts (Cao et al., 2017; Qi et al., 2017). Pb(II) being one of the most toxic metal pollutants, its concentration in discharged wastewater has been strictly limited, for example the discharged concentration of total lead in China is strictly limited within 0.2 mg l−1 according to the published Chinese regulation of “Emission standard of pollutants for electroplating (GB 21900–2008),” while the discharge limit for lead by USEPA is 0.005 mg l−1 (Arbabi et al., 2015; Asuquo et al., 2017; Qi et al., 2017). Up to present, numerous techniques such as chemical precipitation, coagulation, biological treatment, ion exchange and adsorption, solvent extraction, and membrane separation have been developed for removing this pollutant from aqueous solutions (Abo-Farha et al., 2009; Akar et al., 2012; Baharuddin et al., 2015; Chen et al., 2009; Ghassabzadeh et al., 2010; Kang et al., 2011). Compared with other aforementioned methods, the membrane separation technology has been developed rapidly in the past few decades; for instance nanofiltration, reverse osmosis, forward osmosis, polymer-enhanced ultrafiltration (UF), and electrodialysis have been employed extensively for the treatment of heavy metal-containing wastewater (Al-Rashdi et al., 2013; Baharuddin et al., 2015; Barakat and Schmidt, 2010; Farahani and Vatanpour, 2018; Mikulasek and Cuhorka, 2016; Nemati et al., 2017; Salih and Al-Alawy, 2018; Zhao and Liu, 2018). In view of operating convenience, compared with the membrane techniques mentioned above, microfiltration (MF) and UF techniques exhibit the merits of needless strict pretreatment, high permeation flux, low pressure loss, and inexpensive investment, so they will be more competent for the practical application in metal removal (Swaminathan et al., 2013; Wang et al., 2018). However, the aforesaid two membrane techniques are undesirable for the removal of the dissolvable heavy metals because of the membrane pore size being larger than hydration diameters of metal ions and the insignificant affinities between the membrane’s matrix and metal ions. Thus, the modification for these two membranes should be needed to enhance the performance for the capture of metal pollutants.
To take the efficiency of chelating resin for the capture in heavy metals via an adsorption process into view, the modified MF and UF membranes bearing the chelating groups will be applicable for the removal of heavy metals. The chelating groups of diethylenetriamine pentaacetic acid (DTPA) and ethylenediamine tetra(methylenephosphonic acid) (EDTMPA) deserve to be employed on account of their excellent affinities to metal ions (Song et al., 2015; Wang et al., 2017). Polyvinylidene fluoride (PVDF) is one of the extensively used polymer for the preparation of MF and UF membranes due to the excellent chemical, mechanical, and heat resistance properties (Munirasu et al., 2017; Yan et al., 2017). The PVDF-type membrane simultaneously bearing DTPA and EDTMPA complexing groups will be expected to be competent for removal of heavy metal pollutants from industrial effluents.
The aim of this research is to offer an effective pathway for the removal of metal pollutants from electroplating effluents via the adsorption process. Herein, a PVDF-type chelating membrane bearing DTPA and EDTMPA complexing groups was fabricated and employed to remove Pb(II) from the aqueous solution. The morphology, chemical components, and groups of the fabricated membrane were characterized by field emission scanning electron microscope (FE-SEM), X-ray photoelectron spectrometer (XPS), and nuclear magnetic resonance (NMR) spectra, respectively. The effects of pH, initial concentration of Pb(II), temperature, contact time, thickness of membrane stack, as well as flow rate on Pb(II) adsorption were evaluated. For evaluating the interferences of coexisting substances in lead electroplating effluent, the effects of three coexisting metal ions (Cd(II), Ni(II), and Fe(III)) and three complexing reagents (citrate, nitrilotriacetic acid (NTA), and ethylene diamine tetraacetic acid (EDTA)) on the Pb(II) uptake were assessed. The adsorption kinetics and adsorption isotherms of Pb(II) adsorption were investigated at the presence of above coexisting cations and complexing reagents. The breakthrough curves of the membrane were determined by continuous filtration tests, and the obtained data were analyzed by the bed depth service time (BDST) and Thomas models. In addition, the reusability of this chelating membrane was evaluated.
Materials and methods
Materials
The PVDF powders were provided by Chen Guang Co., Ltd (Chengdu, China); the average relative molecular mass of this polymer is 400,000. Polyvinylpyrrolidone (PVP) was employed as a pore former, and dimethyl sulfoxide (DMSO) and tetrahydrofuran (THF) as solvents. EDTMPA, tetrabutyl orthotitanate (TBOT), DTPA, and 3-aminopropyltrimethoxysilane (APTMS) were used for incorporation of EDTMPA and DTPA chelating groups into the membrane matrix. Other reagents including Pb(NO3)2, Cd(NO3)2·4H2O, Ni(NO3)2·6H2O, Fe(NO3)3·9H2O, citrate, NTA, EDTA, absolute ethanol, sodium acetate, glacial acetic acid, hydrochloric acid (36.5 wt%), and sodium hydroxide were purchased from Jingchun Scientific Co., Ltd (Shanghai, China). All above reagents were analytical grade and used as received. The stock solution of Pb(II) (100 mmol l−1) and those of coexisting cations and complexing reagents with a concentration of 20 mmol l−1 were prepared by dissolving weighed amounts of Pb(NO3)2 and the corresponding cation-containing salts and complexing reagents in deionized water. The Pb(II) working solutions were prepared by diluting the stock solution to appropriate volumes.
Preparation of the chelating membrane
First, TBOT (4.7 ml) and APTMS (0.5 ml) were dropwise added to 10 ml THF, and then the mixture was magnetically stirred for 24 h at room temperature to obtain the solution containing TBOT–APTMS colloids (denoted as solution A). Second, 2.0 g EDTMPA was dissolved in 30 ml of ethanol/water mixed solution with a volume ratio of 1:2, pH of this mixture was adjusted to 6.0 with 5 mol l−1 NaOH; this obtained solution was denoted as B. Then, the solution A was dropwise added to solution B, and the mixed solution was constantly stirred for 24 h at room temperature, so a suspension of dispersed microparticles for anchoring EDTMPA groups was obtained. These microparticles were collected by centrifugation at a rate of 3000 r min−1 for 10 min, followed by washing using absolute ethanol three times and DMSO two times. The collected microparticles (denoted as EDTMPA–TBOT–APTMS) were adequately dispersed in 10 ml of DMSO. Third, 1 g DTPA was added to 18 ml DMSO solution, where the temperature of the DMSO was previously heated to 363 K. After the dissolution of DTPA, other 10 ml DMSO containing the dispersive EDTMPA–TBOT–APTMS microparticles was also added in this solution. Then, the mixed solution was slowly heated to 393 K with a rate of 1 K min−1. Subsequently, the solution was stirred at this temperature for 30 min to obtain a colloidal solution containing both DTPA and EDTMPA chelating groups (also denoted as EDTMPA–TBOT–APTMS–DTPA). After that, the colloidal solution was cooled down to 353 K, and 4.8 g PVDF and 0.53 g PVP were successively added into this solution. The casting solution was magnetically stirred for 4 h at 353 K. Lastly, the PVDF chelating membrane was prepared via a phase inversion technique with the clean glass plates as casting supports and deionized water as the coagulation bath. The fabricated chelating membrane was cleaned with deionized water and kept for subsequent characterization and adsorption experiments. The preparation procedure of the chelating groups is shown in Figure 1.
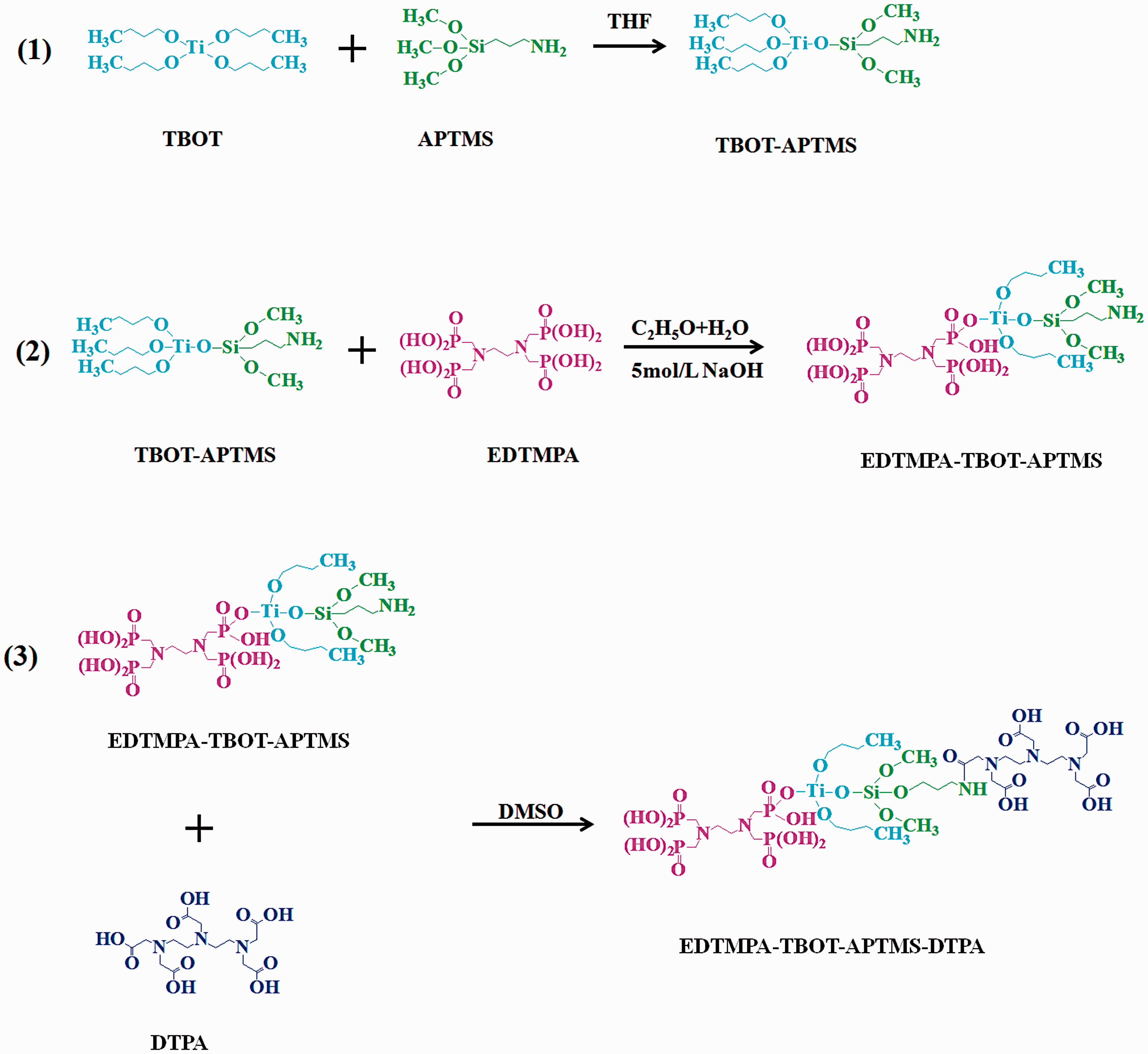
Synthetic illustration of the EDTMPA–TBOT–APTMS–DTPA polymer. APTMS: 3-aminopropyltrimethoxysilane; DMSO: dimethyl sulfoxide; DTPA: diethylenetriamine pentaacetic acid; EDTMPA: ethylenediamine tetra(methylenephosphonic acid); TBOT: tetrabutyl orthotitanate; THF: tetrahydrofuran.
Characterization of the chelating membrane
A SUPRA55 FE-SEM (Zeiss, Germany) was employed to monitor the surface and sectional morphologies of the chelating membrane. An XPS (ESCALAB 250XI, ThermoFisher Scientific, USA) with 150 W monochromated Al Kα as the radiation source was used to characterize the elements of the membrane; the C 1s peak from adventitious carbon at 284.8 eV was employed to calibrate the binding energy. The 13C and 31P solid-state NMR spectra of the membrane were identified using a BRUKER AVANCE III 400 solid-NMR spectrometer (Karlsruhe, Germany). Moreover, the pure water flux, ion exchange capacity, porosity, mean pore size, point of zero charge (pHpzc), tensile strength, and swelling property of the membrane were measured. The detailed information of above characterizations and tests was reported in the supplementary material.
Static adsorption experiments
All adsorption experiments were performed in triplicate and the average value was recorded. pH values were adjusted to a range of 1–7 by a buffer solution consisting of 0.3 mol l−1 acetic acid and 0.2 mol l−1 sodium acetate; the effect of pH on the adsorption of Pb(II) was evaluated at 298 K with keeping the initial Pb(II) concentration at 1.0 mmol l−1 and the contact time as 5 h. The determined optimum pH value was employed for all the other adsorption processes. For the single Pb(II) system, adsorption kinetics and the influence of contact time were studied at 288, 298, and 308 K with 0.1 g of chelating membrane in 200 ml of Pb(II) solution; the initial concentration of Pb(II) was 1.0 mmol l−1. At the different time intervals (i.e. 5, 10, 30, and 60 min), the concentration of Pb(II) was determined by an atomic absorption spectrophotometer (AA6800, Shimadzu, Japan). The adsorbed amount of Pb(II) at time t (qt, mmol g−1) by the chelating membrane was obtained as follows: qt = [(c0 − ct)×V ]/M, where c0 and ct (mmol l−1) are the Pb(II) concentration in the aqueous phase at initial time and time t, respectively. M (g) the dry weight of the chelating membrane, and V (l) is the volume of the aqueous solution (Wang et al., 2016).
Isotherm adsorption tests were performed at different initial Pb(II) concentrations (0.1–5 mmol l−1) with 0.5 g l−1 addition of the membrane. After the equilibrium, the residual Pb(II) was measured and the equilibrium adsorption capacity of the membrane was calculated. Adsorption kinetic experiments and isotherm adsorption studies with regard to the existence of Fe(III), Ni(II), Cd(II), citrate, NTA, and EDTA were carried out at 298 K by the same method as that of the single Pb(II) system. All concentrations of coexisting substances and Pb(II) were 1.0 mmol l−1.
Continuous filtration adsorption experiments
The continuous experiments with effluent circulating back to influent were performed using a single-pass mode device at 298 K, the detailed process was illustrated in the previous report (Song et al., 2014), and the membranes with ∼230 µm in thickness and ∼55 mm in diameter were sealed layer by layer. The flow rate of influent was controlled by manually adjusting the pressure taps in inlet and outlet, and the temperature of influent was maintained at 298 K using a heat exchanger. The effective filtration area per membrane was 24 cm2. Before each test, the membrane stack module was cleaned using deionized water at 298 K for 30 min, keeping inlet pressure at 0.10 MPa and outlet pressure at 0.003 MPa. During the adsorption filtration, Pb(II)-containing influent with initial concentration of 1 mmol l−1 and pH of 5.1 was pumped through the membrane stack. One milliliter of effluent was withdrawn periodically to determine the residual Pb(II) concentration. Influences of flow rate (0.5, 0.75, and 1.0 cm min−1) and thickness of membrane stack (0.7, 0.9, and 1.2 mm) on the Pb(II) uptake were evaluated.
As effluent was not circulated back to influent, the breakthrough curves for the single Pb(II) and six coexisting systems were measured at 298 K with the abovementioned single-pass membrane setup. Influents were pumped through three membrane stacks with thicknesses of 0.7, 0.9, and 1.2 mm with a flow rate of 0.75 cm min−1. The concentration of Pb(II) at the outlet was measured at 5, 10, 30, and 60 min. The transmembrane pressure (TMP, i.e. the difference between the inlet pressure and the outlet pressure) was monitored. The obtained breakthrough data were analyzed using the BDST and Thomas models.
The BDST model described by equation (1) was used to predict the relationship between membrane thickness (Z) and service time (Farooq et al., 2013)
Thomas model was also considered and the rate driving force in accordance with a second-order reversible reaction kinetics is assumed. The nonlinear form of this model is depicted by equation (2) (Ghasemi et al., 2011)
Desorption experiments
According to the effect of pH on Pb(II) adsorption and the previous reports (Wang et al., 2017), the lower pH value is not favored on the uptake of Pb(II). Through experimental estimation, the 0.5 mol l−1 H2SO4 solution was used as the elution agent in the desorption experiments. The membrane with a dry weight of ∼0.1 g loading Pb(II) was immersed in 200 ml of the elution solution which was repeatedly employed for the desorption of Pb(II). As the adsorbed Pb(II) was eluted from the surface of membrane, the chelating membrane was immersed into 1 mmol l−1 of Pb(II) solution for the uptake of this metal, the readsorption process after desorption was the same as the static adsorption experiment. To examine the reusability of the chelating membrane, the adsorption/desorption processes for the same membrane were repeated 10 times. The amounts of adsorbed and eluted Pb(II) during these tests were determined. The desorption efficiency (DE) was calculated as follows: DE = (q1/q2)×100%, where q1 is the desorbed amount of Pb(II) from the membrane (mmol g−1) and q2 is the adsorbed amount of Pb(II) on the membrane at equilibrium (mmol g−1) (Min et al., 2012; Wang et al., 2016).
Results and discussion
Characterization of the chelating membrane
FE-SEM
Morphologies of the PVDF chelating membrane are shown in Figure 2. Figure 2(a) and (b) displays the surface morphology of the chelating membrane before and after Pb(II) adsorption; Figure 2(c) and (d) presents the sectional morphology of the chelating membrane before and after Pb(II) adsorption. As demonstrated by Figure 2(a), a uniform microporous structure can be observed. The determined mean pore size of the chelating membrane is 0.14 ± 0.02 µm, which will be helpful for the solution permeating through this membrane. In addition, some small particles (labeled by green circles) are seen in the surface and the microporous interior of the membrane; they can be indexed to colloids containing the chelating groups. As shown in Figure 2(c), two kinds of micrographs including finger-like pores near the surface skin (labeled by red rectangle) and sponge-like microstructures in inner side (described using red circle) are identified. Compared with the surface and sectional morphologies of the chelating membrane before Pb(II) adsorption, the change in SEM micrographs after the Pb(II) adsorption (Figure 2(b) and (d)) can be ignored.

FE-SEM morphologies of the chelating membrane: (a) surface morphology before Pb(II) adsorption, (b) surface morphology after Pb(II) adsorption, (c) sectional morphology before Pb(II) adsorption, and (d) sectional morphology after Pb(II) adsorption.
In addition, measured pure water flux, ion exchange capacity, porosity, tensile strength, and swelling property of the fabricated chelating membrane are 1399 ± 26 l m−2 h−1, 0.63 ± 0.03 mmol g−1, 58 ± 1%, 1.07 ± 0.12 MPa, and 70 ± 1%, respectively.
XPS analysis
The XPS measurements were performed to examine the chemical components of the membrane and to further ascertain the uptake of Pb(II); the obtained results are displayed in Figure 3. For the full XPS spectrum of the chelating membrane before the Pb(II) adsorption (Figure 3(a)), two peaks at 286 and 688 eV can be assigned to C and F elements in the PVDF chain. Apart from the characteristic peaks of PVDF, peaks for O 1s (531 eV), Si 2p (101 eV), P 2p (133 eV), N 1s (400 eV), and Ti 2p (458 eV) are also observed, which indicates that the chelating groups containing microparticles are blended into the PVDF matrix. Therefore, the DTPA and EDTMPA chelating groups are successfully incorporated into the PVDF membrane. After the Pb(II) adsorption (Figure 3(b)), except for abovementioned seven peaks for C 1s, F 1s, O 1s, Si 2p, P 2p, N 1s, and Ti 2p, the peaks of Pb 4f are detected. High-resolution XPS spectra for P 2p before Pb(II) adsorption and Pb 4f after Pb(II) adsorption are displayed in Figure 3(c) and (d). Compared with Figure 3(c), the peaks at 139 eV for Pb 4f7/2 and 144 eV for Pb 4f5/2 can be clearly identified in Figure 3(d), but these two peaks cannot be observed in Figure 3(c), thereby suggesting the Pb(II) uptake of the chelating membrane. Additionally, the fact of Pb(II) adsorbed by the chelating membrane is mainly exhibited by Pb 4f7/2 and Pb 4f5/2 and these two peaks correspond to the Pb(II) chemical state of Pb2+ (Qi et al., 2017).

XPS spectra of the chelating membrane: (a) full XPS spectrum before Pb(II) adsorption, (b) full XPS spectrum after Pb(II) adsorption, (c) P 2p before Pb(II) adsorption, and (d) Pb 4f after Pb(II) adsorption.
NMR analysis
The measured 13C and 31P solid-state NMR spectra of the chelating membrane before and after Pb(II) adsorption are shown in Figure 4. For the 13C NMR spectrum (Figure 4(a)), the peaks at 44.6 and 121.8 ppm can be attributed to –CH2– and –CF2– carbons of the PVDF chain (Tony et al., 2012). The peaks at 18–23 ppm can be assigned to –CH3 and –CH2– groups of APTMS and TBOT. The signals at ∼31.5 ppm are assigned to –C–PO(O)2 group of EDTMPA and the peaks at 53.0–56.5 ppm can be identified to carbons of –CH2N– group in EDTMPA and DTPA. In addition, the peaks at 176–184 ppm are corresponding to the carboxylic group of DTPA (Kao et al., 2008; Zhao et al., 2011). Therefore, it can be ascertained that the model poly(amino phosphonic acid) and poly(amino carboxylic acid) chelating groups were blended to PVDF matrix successfully. After Pb(II) adsorption, the positions of –C–PO(O)2, –CH2N–, and carboxylic groups (Figure 4(c) to (e)) are slightly shifted to their low field, which can be ascribed to the complexing interaction between Pb(II) and the chelating groups.

Solid-state NMR spectra: (a) 13C NMR, (b) 31P NMR, (c) –C–PO(O)2 group, (d) –CH2N– group, and (e) –COOH group: (1) before Pb(II) adsorption and (2) after Pb(II) adsorption.
As to the 31P NMR spectra (Figure 4(b)), before the Pb(II) adsorption, two signals at 5.8 and 15.6 ppm are detected, corresponding to P of EDTMPA; the shoulder peak appearing at ∼15.6 ppm can be attributed to the combination between EDTMPA and TBOT (Wang et al., 2017; Zheng et al., 2016). After the uptake of Pb(II), the above two peaks shift from 5.8 to 8.8 ppm and from 15.6 to 18.6 ppm, which could be an indication that P atoms of phosphonic acid groups are chelated by Pb(II).
As the matter of above results, we confirm that poly(amino phosphonic acid) and poly(amino carboxylic acid) groups are incorporated into the PVDF membrane, and thereby achieving the capture of Pb(II) from aqueous solutions.
Effects of various variables
Effects of pH, initial Pb(II) concentration, contact time, and temperature
The variation of Pb(II) adsorption by the chelating membrane with the increase in pH value was assessed (Figure 5(a)); it is explicit that Pb(II) uptake of the membrane is strongly dependent on the change of pH. The Pb(II) uptake increases with the increase in pH (from 1.0 to 5.1) and then it decreases as the pH value is higher than 5.1. This pH value is excellently consistent with the determined pHpzc of the membrane (pHpzc = 5.1). At pH < 5.1, the carboxylic and phosphonic acid functional groups of the chelating membrane will be protonated, the decrease of Pb(II) adsorption with pH decrease is probably due to the electrostatic repulsion between the surface net charge of adsorbent and Pb(II); also, the competition between Pb(II) and H+ for the adsorption sites will become dominant. As pH increases higher than 5.1, a part of Pb(II) can transform from Pb2+ to other forms such as Pb(OH)+ and Pb(OH)2, thereby resulting in the abatement of Pb(II) uptake. In this research, the pH of 5.1 was adopted for all the following adsorption experiments.

Effects of pH, initial Pb(II) concentration, contact time and temperature. (a) pH (c0(Pb(II)) = 1 mmol l−1; membrane addition = ∼0.5 g l−1; T = 298 K; t = 300 min) and initial Pb(II) concentration (membrane addition = ∼0.5 g l−1; T = 298 K; t = 300 min; pH = 5.1) and (b) contact time (c0(Pb(II)) = 1 mmol l−1; membrane addition = ∼0.5 g l−1; T = 298 K; pH = 5.1) and temperature (c0(Pb(II)) = 1 mmol l−1; membrane addition = ∼0.5 g l−1; pH = 5.1).
Also, indicated by Figure 5(a), as the initial Pb(II) concentration varies from 0.1 to 5 mmol l−1, the Pb(II) uptake at 298 K increases from 0.14 to 1.22 mmol g−1; this can be on account of the increase in Pb(II) concentration accelerating the diffusion of Pb(II) from solution to the membrane surface. However, the removal efficiency of Pb(II) decreases from 71 to 12%. Thus, the small concentration of Pb(II) will be beneficial to the removal of this metal.
Pb(II) uptakes of the chelating membrane as a function of time at 288, 298, and 308 K three temperatures are presented in Figure 5(b); the Pb(II) uptake reduces with the increase in temperature. From this point of view, it can be inferred that this adsorption of Pb(II) is an exothermic process. Also, it can be seen that there are two stages (rapid adsorption and slow adsorption) occurring in the Pb(II) adsorption process. At the rapid stage (from 0 to 120 min), Pb(II) easily interacts with the chelating membrane because of the presence of abundant active sites on the surface of the chelating membrane. On the other hand, in the slow adsorption process with time ranging from 120 to 420 min, the adsorption rate of Pb(II) decreases because most of the active sites are occupied.
Effects of flow rate and membrane thickness
The effect of flow rate on Pb(II) adsorption by chelating membrane was carried out by varying the flow rate from 0.5 to 1.0 cm min−1, while keeping membrane stack thickness at 0.9 mm. The effect of flow rate on the Pb(II) uptake of the membrane is depicted in Figure 6(a). When the flow rate increases from 0.5 to 0.75 and 1.0 cm min−1, the uptake of Pb(II) reduces from 1.43 to 1.29 and 1.13 mmol g−1, consistent with the previous reports (Song et al., 2014). This can be attributed to the fact that at a low flow rate, Pb(II) will have abundant time to diffuse into the pores of the membrane and can be helpful to the metal capture by EDTMPA and DTPA chelating groups; on the other hand, at a high flow rate, the chelating reaction between Pb(II) and the PVDF chelating membrane is insufficient because Pb(II) can rapidly permeate through the membrane. Therefore, the adsorption of Pb(II) decreases with the increasing flow rate. Thus, the efficiency in Pb(II) adsorption can be assumed to be controlled by the diffusion limitation of Pb(II) entering to the surface and inner pores of the membrane. The low flow rate will elapse the retention time of Pb(II), which is advantageous to the uptake of this metal; conversely, the high flow rate decreases the retention time and retards the adsorption of Pb(II).

Effects of flow rate and thickness of membrane stack: (a) flow rate (c0(Pb(II)) = 1 mmol l−1; thickness of membrane stack = 0.9 mm; T = 298 K; pH = 5.1) and (b) thickness of membrane stack (c0(Pb(II)) = 1 mmol l−1; flow rate = 0.75 cm min−1; T = 298 K; pH = 5.1).
In addition, the adsorption performance of chelating membrane was tested at different membrane stack thickness at a flow rate of 0.75 cm min−1. As shown in Figure 6(b), the effect of the membrane stack thickness is remarkable; as the thickness of the membrane stack increases from 0.7 to 0.9 and 1.2 mm, the equilibrium time for the Pb(II) uptake on the chelating membrane is extended. This can be assigned to the increase in the surface area and the abundant adsorption sites accompanied by the increase in thickness of membrane stack. It can be observed that as the time ranges from 0 to 300 min, the thick membrane stack will be helpful to guarantee the low Pb(II) concentration in effluent; however, the uptake of Pb(II) decreases to some extent. This may be due to the membrane weight increasing with the increase of thickness of membrane stack.
Effects of the coexisting cations and complexing reagents
Three cations of Fe(III), Ni(II), and Cd(II), and three complexing reagents including citrate, NTA, and EDTA were selected to elucidate the influence of coexisting substances on the Pb(II) adsorption by the chelating membrane. The Pb(II) uptake of the membrane reduces with the concentrations of Fe(III), Ni(II), and Cd(II) increasing from 0 to 5 mmol l−1 (Figure 7(a)). The coexistence of the abovementioned three coexisting cations shows a negative effect on Pb(II) adsorption, and the interference of them follows the order of Fe(III) < Ni(II) < Cd(II). During the adsorption process, these three coexisting cations compete with Pb(II) to occupy the active sites for complexation, resulting in the decrease of Pb(II) uptake. When the concentration of Fe(III), Ni(II), and Cd(II) is 1 mmol l−1, Pb(II) uptake of the membrane decreases by 19, 28, and 40%, further confirming that Cd(II) shows the most notable interference on the Pb(II) adsorption, followed by Ni(II), and last by Fe(III). Despite the interference of above three coexisting cations, the Pb(II) uptake of the chelating membrane still dominates the adsorption process.

Effects of coexisting cations and complexing reagents on Pb(II) adsorption: (a) coexisting cations (b) complexing reagents: (c0(Pb(II)) = 1 mmol l−1; t = 300 min; membrane addition = ∼0.5 g l−1; T = 298 K; pH = 5.1).
The influences of citrate, NTA, and EDTA with different concentrations (from 0 to 5 mmol l−1) on the Pb(II) uptake of the chelating membrane are shown in Figure 7(b). Similar to that of the coexisting cations, the increase in the concentrations of these three complexing reagents also causes the reduction of Pb(II) uptake. This can be explained that much more Pb(II) will be complexed by these three complexing reagents as their concentrations increase, and the complexed form of Pb(II) reduces this metal uptake of the membrane to some extent. As citrate, NTA, and EDTA coexist with Pb(II) at the concentration of 1 mmol l−1, Pb(II) uptake of the membrane decreases by 32, 47, and 69%, respectively. The negative effects of the three complexing reagents on the Pb(II) uptake are in the order: citrate < NTA < EDTA. Among the three complexing reagents, EDTA exhibits the most interference on the Pb(II) adsorption. It should be pointed out that although the coexisting cations and complexing reagents disturb the uptake of Pb(II), the fabricated PVDF chelating membrane retains the ability for the capture of Pb(II). Thus, this chelating membrane will be competent for recycling Pb(II) from the solution.
Comparison of Pb(II) adsorption
For the purpose of comparison, other two PVDF-type chelating membranes only bearing DTPA (for DTPA–APTMS/PVDF membrane) or EDTMPA (for EDTMPA–TBOT/PVDF membrane) were also employed to capture Pb(II). The detailed information of these two membranes can be referred in our previous studies (Song et al., 2015; Wang et al., 2017). At the same experimental condition (c0(Pb(II)) = 1 mmol l−1, t = 300 min, membrane addition = ∼0.5 g l−1, T = 298 K, pH = 5.1), the Pb(II) uptake of the fabricated PVDF chelating membrane and those of two abovementioned membranes are 1.1, 0.38, and 0.74 mmol g−1, respectively. Pb(II) uptake of the chelating membrane simultaneously bearing DTPA and EDTMPA groups is 2.8 and 1.4 times larger than those of DTPA–APTMS/PVDF and EDTMPA–TBOT/PVDF membranes.
By comparison with previous reported adsorbents (Table 1), the fabricated PVDF chelating membrane shows an excellent performance in Pb(II) adsorption. In this regard, the fabricated PVDF chelating membrane will deserve to be recommended as a candidate for the removal of metal pollutants from the aqueous solution.
Comparison of Pb(II) uptake for different adsorbents.

ATMPZ: amino tris (methylenephosphonic acid); CNT: carbon nanotubes; CS: chitosan; EDTA: ethylene diamine tetraacetic acid; GONF: hybrid graphene oxide based inverse spinel nickel ferrite; PAC: powder activated charcoal; PVA: polyvinyl alcohol; PVDF: polyvinylidene fluoride; TNT: titanate nanotubes.
Studies of adsorption kinetics and adsorption isotherm
The adsorption kinetics and adsorption isotherms experimental data of the chelating membrane toward Pb(II) at the presence of Fe(III), Ni(II), Cd(II), citrate, NTA, and EDTA were investigated at 298 K (Figure 8). The variation of Pb(II) adsorption versus time was analyzed by the pseudo-first-order and pseudo-second-order models (Adrián et al., 2017; Kalantari et al., 2015; Singh et al., 2016) (see Supplementary material). The analyzed results are shown in Figure S1 and Table S1; the pseudo-second-order parameters are also presented in Table 2. In addition, two error parameters, i.e. the root mean square error (RMSE) and Chi square (χ2) were obtained (shown in Table S1). From Table S1, it is known that the pseudo-second-order model (the coefficient of determination, R2 > 0.99) is more suitable than the pseudo-first-order model for the description of Pb(II) adsorption process. Therefore, the chemisorption may play a more important role than physisorption for the Pb(II) adsorption (Yakout and Borai, 2013).

Adsorption kinetics and adsorption isotherms for the Pb(II) uptake by the chelating membrane: (a) adsorption kinetics of the cation coexisting systems; (b) adsorption kinetics of the complexing reagent coexisting systems: (c0(Pb(II)) = c0(Fe(III)) = c0(Ni(II)) = c0(Cd(II)) = c0(citrate) = c0(NTA) = c0(EDTA) = 1 mmol l−1; T = 298 K; pH = 5.1; membrane addition = ∼0.5 g l−1); (c) adsorption isotherms of the cation coexisting systems; and (d) adsorption isotherms of the complexing reagent coexisting systems: (t = 300 min; c0(Fe(III)) = c0(Ni(II)) = c0(Cd(II)) = c0(citrate) = c0(NTA) = c0(EDTA) = 1 mmol l−1; T = 298 K; pH = 5.1; membrane addition = ∼0.5 g l−1). EDTA: ethylene diamine tetraacetic acid; NTA: nitrilotriacetic acid.
Analyzed parameters of the pseudo-second-order and Langmuir models for the chelating membrane toward Pb(II) at 298 K.

EDTA: ethylene diamine tetraacetic acid; NTA: nitrilotriacetic acid; RMSE: root mean square error.
Compared with the Pb(II) uptake of the single Pb(II) system, the coexisting Fe(III), Ni(II), and Cd(II) show an interference on the uptake of this metal (Figure 8(a)); their interferences are in the trend of Cd(II) > Ni(II) > Fe(III). Similarly, the coexisting citrate, NTA, and EDTA also exhibit the negative effects on the Pb(II) uptake (Figure 8(b)), the interferences of them follow the sequence: citrate < NTA < EDTA.
By comparison of k2, it can be concluded that the competitive adsorption between Pb(II) and the three coexisting cations accelerates the adsorption process, due to the large values of k2 for Pb(II)–Fe(III), Pb(II)–Ni(II), Pb(II)–Cd(II) systems. In addition, for the complexing reagent coexisting systems, k2 follows the ascending sequence of single Pb(II) < Pb(II)–citrate < Pb(II)–NTA < Pb(II)–EDTA, indicating the negative effects of these three complexing reagents on the Pb(II) uptake; among them, the detrimental effect of EDTA is most considerable.
The adsorption isotherm data at equilibrium for seven systems (single Pb(II), Pb(II)–Fe(III), Pb(II)–Ni(II), Pb(II)–Cd(II), Pb(II)–citrate, Pb(II)–NTA, and Pb(II)–EDTA) are shown in Figure 8(c) and (d). The obtained data were analyzed using Freundlich, Langmuir, and Dubinin-Radushkevich (D–R) adsorption isotherm models (Ammar et al., 2015; Asuquo et al., 2017; Liu and Liu, 2008; Min et al., 2012; Zhao et al., 2015) and shown in Figures S2 and S3. The analyzed RMSE and χ2 are summarized in Table S2; also, the analyzed parameters of Langmuir model are shown in Table 2. By comparison of R2, the Langmuir model is more suitable than Freundlich and D–R models for describing the isotherm adsorption process of Pb(II) uptake. The data in Table S2 show that the calculated maximum adsorption capacity (qm) by the Langmuir model is slightly greater than the experimental value (qe) because some active sites on the membrane surface are not occupied. The obtained Freundlich parameter of 1/n related to the adsorption intensity is smaller than 1, indicating the easy occurrence of the Pb(II) adsorption; the mean free energy (E) of adsorption derived from D–R model is in the range of 12.8–17.2 kJ mol−1, suggesting the chemisorption nature of the chelating membrane toward Pb(II).
Additionally, the Langmuir parameter (KL) associated with the adsorption affinity for the above three cation coexisting systems follows the order of Pb(II)–Fe(III) > Pb(II)–Ni(II) > Pb(II)–Cd(II), suggesting the interferential effect of Cd(II) higher than those of Fe(III) and Ni(II). Also, in view of the parameter KL, it can be deduced that the interferences of three complexing reagents on the Pb(II) adsorption follow the trend of EDTA > NTA > citrate. The result is very similar to that of adsorption kinetics study: the coexistence of abovementioned cations and complexing reagents reduces the uptake of Pb(II); among them, the detrimental effects of Cd(II) and EDTA are more considerable than other cations and complexing reagents.
Three thermodynamic parameters (standard free energy change (ΔG°), standard enthalpy change (ΔH°), and standard entropy change (ΔS°)) were also calculated to reveal the adsorption characteristic of Pb(II) by the chelating membrane; the calculated equation was described in the supplementary material. In this work, the Langmuir equilibrium constant (KL) was used to calculate ΔG°, ΔH°, and ΔS° (Liu, 2009; Liu and Xu, 2007; Liu and Liu, 2008). The analyzed results are listed in Table S3. It can be seen that the values of ΔG°, ΔH° for the single Pb(II) and Pb(II)–Fe(III), Pb(II)–Ni(II), Pb(II)–Cd(II), Pb(II)–citrate, Pb(II)–NTA, and Pb(II)–EDTA six coexisting systems are negative, which validates the spontaneous and exothermic feature of Pb(II) adsorption by the chelating membrane. The low and negative value of ΔS° indicates a slight decrease in randomness at the solution/membrane interface. The presence of aforementioned coexisting substances does not change the thermodynamic characteristics of Pb(II) adsorption. In addition, it can be inferred that Pb(II) adsorption process is a chemisorption according to the value of ΔH° (from −74.02 to −82.70 kJ mol−1), and the adsorption is an enthalpy controlled process due to ΔH° > TΔS° (Adrián et al., 2017).
Experimental parameters of Pb(II) adsorption in continuous experiments at 298 K.

EDTA: ethylene diamine tetraacetic acid; NTA: nitrilotriacetic acid.
In summary, based on FTIR, XPS, and NMR characterizations, data analyses of pseudo-second-order equation and (D–R) isotherm model as well as the thermodynamic parameter calculation, the mechanism for Pb(II) adsorption by the fabricated chelating membrane may be interpreted as follows. First, as Pb(II) is close to the surface of chelating membrane, the chelating groups of the membrane will repel away the combined inorganic ions (such as Na+ and Ca2+) because the chelating groups show a stronger affinity toward Pb(II) than the conventional inorganic ions; at the same time, the unoccupied orbitals of this metal ion will prepare to accept the donored electrons by chelating groups. Then, the coordinating interaction between Pb(II) and the chelating membrane occurs, thus the chemisorption action dominates the uptake process of Pb(II) from the solution.
Analysis of breakthrough curves
The breakthrough curves were obtained at different thicknesses of the membrane stack (Z = 0.7, 0.9, and 1.2 mm) for the single Pb(II) and Pb(II)–Fe(III), Pb(II)–Ni(II), Pb(II)–Cd(II), Pb(II)–citrate, Pb(II)–NTA, and Pb(II)–EDTA six coexisting systems (Figure 9), with keeping the concentrations of all substances at 1 mmol l−1 and the flow rate at 0.75 cm min−1. The determined TMP values of membrane stacks are 0.26 MPa (Z = 0.7 mm), 0.35 MPa (Z = 0.9 mm), and 0.48 MPa (Z = 1.2 mm), so TMP rises with the increase in thickness of the membrane stack. Among the abovementioned seven systems with the same membrane stack thickness, the variations for operating pressure and TMP are inconspicuous and can be ignored. Pb(II) uptake capacity (qexp) of the chelating membrane was obtained through the mass of Pb(II) in the membrane stack divided by the mass of membrane stack; the mass of Pb(II) in the membrane stack was calculated by numerical integration of the breakthrough curve. The length of the mass transfer zone (Zm) is expressed as follows: Zm = Z (1−tb/te), where tb (the breakthrough time) is the time at ct/c0 = 0.05, te (the exhaustion time) is the time at ct/c0 = 0.95 (Ghasemi et al., 2011). These obtained experimental parameters are listed in Table 3. As shown in Table 3, when the thickness of membrane stack increases, tb and te are extended, Zm becomes longer and this can be due to the much available adsorption sites in the membrane stack. Thus, it may be implied that complete exhaustion of the membrane stack will need a long time as the thickness of membrane stack increases. Additionally, the Pb(II) uptake (qexp) decreases with the increase in the membrane stack thickness, indicating that the amount of Pb(II) uptake is not directly proportional to the increasing thickness of membrane stack because the available adsorption sites in membrane stack are incompletely utilized. Moreover, compared with the single Pb(II) system (Z = 0.7 mm), qexp for three cation coexisting systems with the same membrane stack thickness reduces and follows the order of Pb(II)–Cd(II) > Pb(II)–Ni(II) > Pb(II)–Fe(III), corresponding to the decreases of 13.4, 7.1, and 4.2%. For the three complexing reagents coexisting systems, the trend of qexp bears a very close resemblance to that of the cation coexisting systems. The Pb(II) uptake of the membrane decreases by 7.3% with the presence of citrate, and 16.8% for NTA, 24.9% for EDTA. Unambiguously, Cd(II) and EDTA show the more noticeable interferences on Pb(II) uptake than other two cations and two complexing reagents.

Breakthrough curves of chelating membrane toward Pb(II): (a) single Pb(II) system; (b) Pb(II)–Fe(III) system; (c) Pb(II)–Ni(II) system; (d) Pb(II)–citrate system; (e) Pb(II)–Cd(II) system; (f) Pb(II)–NTA system; and (g) Pb(II)–EDTA system: c0(Pb(II)) = c0(Fe(III)) = c0(Ni(II)) = c0(Cd(II)) = c0(citrate) = c0(NTA) = c0(EDTA) = 1.0 mmol l−1; T = 298 K; flow rate = 0.75 cm min−1; pH = 5.1.
Herein, the measured S-like breakthrough curves were analyzed using the BDST and Thomas models (analyzed parameters reported in Table 4). Based on the coefficient of determination (R2), it can be confirmed that both models are suitable for describing the breakthrough process. Compared with q0 (parameters of Thomas model) of the single Pb(II) system, those of other six coexisting systems decrease, showing the interferences of the coexisting cations and complexing agents, in which Cd(II) and EDTA exhibit a remarkable interference on the Pb(II) uptake. By comparison of Ka derived from BDST model for the six coexisting systems and the same thickness of membrane stack, the higher Ka for the single Pb(II) system suggests that thin thickness of this membrane stack can avoid the breakthrough (Li et al., 2016). Under the same conditions, for the abovementioned six coexisting systems, a high thick membrane stacks would be required to avoid the breakthrough, and the thickness of membrane stacks should be progressively increased following the order: Pb(II)–Fe(III) < Pb(II)–Ni(II) < Pb(II)–citrate < Pb(II)–Cd(II) < Pb(II)–NTA < Pb(II)–EDTA.
Parameters of the BDST and Thomas models for the chelating membrane toward Pb(II) at 298 K.
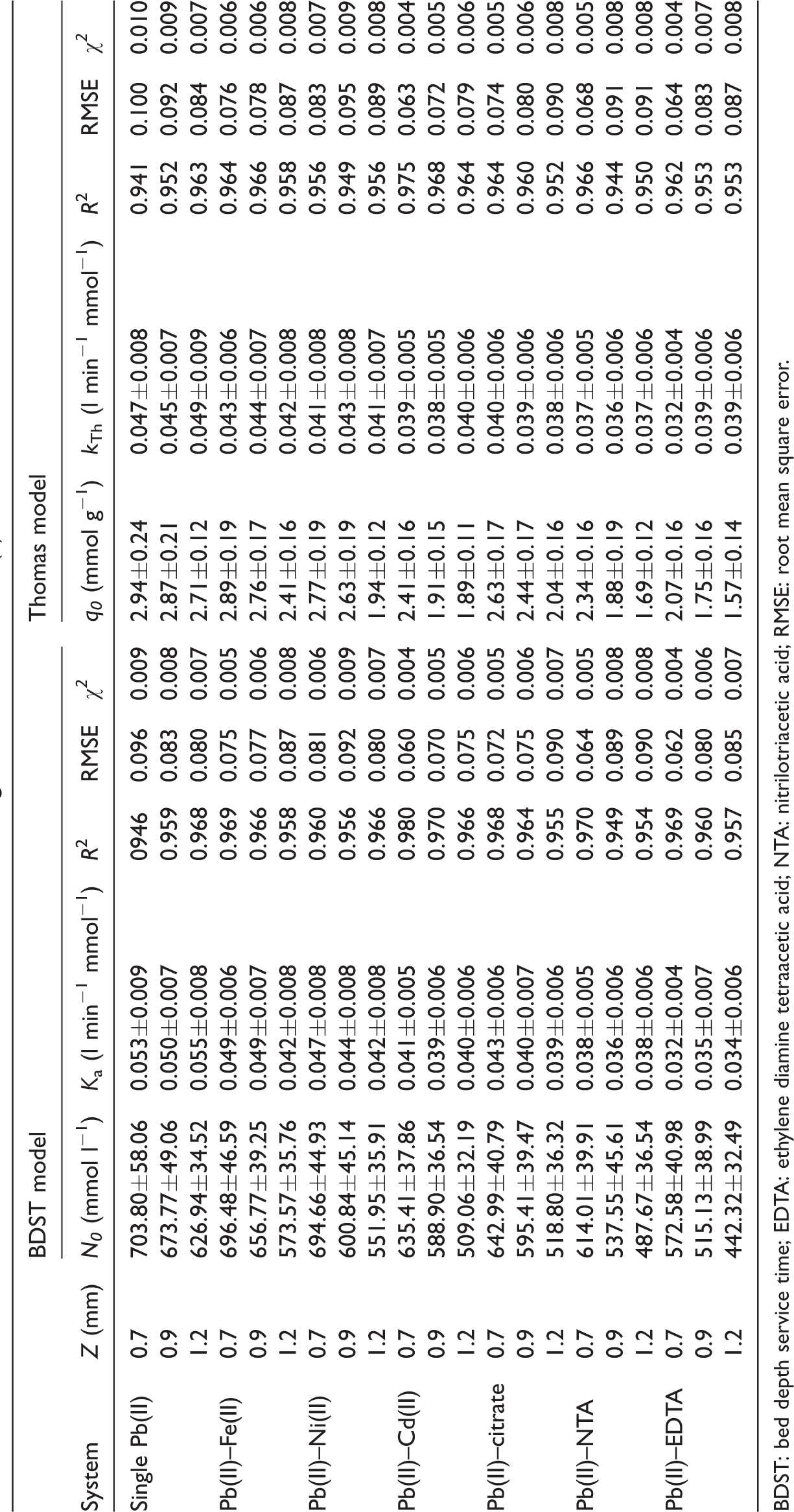
BDST: bed depth service time; EDTA: ethylene diamine tetraacetic acid; NTA: nitrilotriacetic acid; RMSE: root mean square error.
Regeneration of the chelating membrane
Taking the practical application into account, the reusability of fabricated PVDF chelating membrane is essential, thus the adsorption/desorption test was carried out. Herein, the 0.5 mol l−1 H2SO4 solution was repeatedly used to regenerate the adsorption spent chelating membrane. Figure 10 depicts the Pb(II) adsorption by the membrane over 10 adsorption/desorption processes. During these processes, the Pb(II) adsorption capacity and DE of the membrane vary slightly. After 10 adsorption/desorption cycles, the Pb(II) uptake of the membrane is well maintained, and this metal uptake still exceeds 1 mmol g−1; moreover, DE of the membrane is higher than 95%. Based on this fact, the PVDF chelating membrane can be repeatedly used to recycle Pb(II) from the aqueous solution.

Reusability of the fabricated PVDF-type chelating membrane.
Conclusions
The fabricated PVDF chelating membrane bearing poly(amino phosphonic acid) and poly(amino carboxylic acid) groups demonstrates a high capture efficiency in Pb(II) via an adsorption process. The Pb(II) uptake decreases with the presence of Fe(III), Ni(II), Cd(II), citrate, NTA, and EDTA; the interferential effects of Cd(II) and EDTA are more remarkable than those of other coexisting cations and complexing reagents. The-pseudo-second-order model and Langmuir model are suitable for the descriptions in the adsorption kinetics and adsorption isotherms. The breakthrough process can be described by the BDST and Thomas models. Additionally, the fabricated PVDF chelating membrane showed an excellent regenerative performance. With respect to the results derived from this study, the fabricated PVDF chelating membrane can be considered as a candidate employed for the removal of metal pollutants from aqueous solutions.
Footnotes
Authors’ contributions
LS conceived and designed the experiments; XiW, PL, and XuW performed the static adsorption experiments; MJ and FL performed the continuous filtration adsorption experiments; XiW analyzed the data and wrote the paper. All authors read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hebei Provincial Natural Science Foundation of China (Grant No. B2016203012) and the Science and Technology Support Key Research and Development Program of Qinhuangdao, China (Grant No. 201703A012).


