Abstract
The carbon dioxide sorption process at coal seams is very important for understanding the trapping mechanisms of carbon capture and storage. The gas retention capacity of coal seams can be estimated using indirect methods based on the adsorption/desorption isotherms obtained in the laboratory. However, the gas sorption capacity can be overestimated or underestimated depending on the sample preparation. This work evaluates different sample preparations and their theoretical adsorption capacity using coal samples from the Cambui coal field (Parana Basin), southern Brazil. Experiments using a thermogravimetric balance were done to calculate the theoretical adsorption capacity, while sample characterization was done through immediate analysis, elementary analysis, and mineralogical studies. The sample preparations used in this work were powder, pellets, and fragments. While the powder form presents an average behavior, without any experimental complication, the pellet is extremely sensitive to any variation in the sample preparation, such as fractures, and the fragment requires a much longer experiment time than the other sample preparations, being impracticable for some cases.
Keywords
Introduction
Climate and environmental changes generated by pollutants resulting from fossil fuel combustion have been seriously discussed lately. One of the alternative ways to reduce the carbon dioxide (CO2) concentration in the atmosphere is by storing the gas in coal seams. Although the technology necessary for the storage process in coal seams is too expensive, these costs can be reduced by applying the enhanced coal-bed methane (ECBM) technology. The ECBM methodology makes it possible to recover methane and store CO2 in the coal matrix by CH4/CO2 exchange sorption (Busch and Gensterblum, 2011).
Possible sites for geological sequestration of CO2 include unminable coal seams, depleted oil and gas reservoirs, abandoned and sealed mines, saline aquifers, oceans, etc. The revenues from this ECBM process can, to a large extent, offset the cost of CO2 sequestration. Understanding and quantifying the coal–gas interaction is, therefore, an important area of research (Harpalani et al., 2006).
One approach to sequestering CO2 is to inject it into deep, unminable coal seams. A particular advantage of coal seam sequestration is that coal seams can store several times more CO2 than the equivalent volume of a conventional gas reservoir because coal has a large surface area. Another advantage of coal seams is that not only does such a process sequester CO2, but methane is displaced which can be recovered and sold to help offset costs. This process is known as ECBM recovery or ECBM. Advanced Resources International and their partners are using the only long-term, multiwell ECBM projects that exist in the world today to evaluate the viability of storing CO2 in deep, unminable coal seams. The two existing ECBM pilots are located in the San Juan Basin in northwest New Mexico and southwestern Colorado. The knowledge gained from studying these projects is being used to verify and validate gas storage mechanisms in coal reservoirs and to develop a screening model to assess CO2 sequestration potential in other promising coal basins of the US (Reeves and Koperna, 2008).
The interaction of CO2 with the coal matrix under in situ gas pressure and temperature conditions is a prerequisite for the application of CO2–ECBM and can be understood through high pressure sorption isotherms (Zhang et al., 2014). The adsorption isotherm expresses the ratio between the quantity of adsorbed adsorbate (the component that will be adsorbed) and the quantity of desorbed adsorbate in a solid–gas equilibrium (Dreisbach and Losch, 2002).
A variety of factors affect the capacity of coal to store gas, among which the most common are the moisture content, mineral content and composition, temperature, depth, porosity, natural fracturing, and sample particle size (Aziz et al., 2011; Gray, 1987; Mavor et al., 1992). During experimental analysis, laboratories usually take coal samples in powder form. After grinding, the coal samples are reduced to a small particle size to decrease the time required to achieve saturation. Moisture-equilibrated coal samples have generally shown an increase in adsorption capacity with increasing rank, while for dry coals, the reverse trend has also been reported (Harpalani et al., 2006). Various studies have reported that the gas sorption capacity of coal is affected by mineral matter. By comparing ash content to Langmuir volume, a negative correlation was commonly observed and accepted (Crosdale et al., 1998; Faiz et al., 1992; Ma et al., 2014; Yee et al., 1993). One interpretation of this effect was that mineral matter has less surface area compared to the microporous organic constituents of coal (Clarkson and Bustin, 1996; Gan et al., 1972). However, it should be noted that all of these studies on the relation between minerals and gas sorption capacity only analyzed the mineral content in relation to gas sorption capacity. Moreover, these previous studies ignored the possibility that mineral occurrence and mineral composition may impact gas sorption capacity (Deng et al., 2015). According to the literature, the grain size of coal used in adsorption testing affects the measured gas storage capacity and the use of finely ground coal samples in sorption testing leads to a risk of overstating the sorption capacity of the coal (Aziz et al., 2011; Mavor et al., 1992). Zhang et al. (2014) studied the CO2 adsorption in different particle sizes and found that the Langmuir volume decreases with increasing coal particle size for CO2 (Zhang et al., 2014). Therefore, the gas sorption capacity can be overestimated or underestimated depending on the particle size. All of the above factors contribute to the variability of sorption behavior of coals, making it important to study this on a case-by-case basis. The gas retention capacity of a reservoir at in situ temperature and pressure conditions can be estimated from a sorption isotherm. Detailed knowledge of the sorption isotherms for methane and CO2 is a required input parameter for methane production simulations from ECBM operations. This information is also needed to estimate the coal seam CO2 storage capacity and the overall economics of the entire process. Gas sorption isotherms in coal are often modeled using the Langmuir approach, as it usually exhibits excellent correlation with the experimental sorption data (Busch et al., 2003; Prinz et al., 2001; Yee et al., 1993).
There is no work on the preparation of different samples of coals to understand the quality of gas sorption data at high pressure for CO2. Therefore, the objective of this paper is to show the evolution of the new forms of preparation of samples of coal, analyzing the effectiveness, the time of analysis, and the theoretical adsorption capacity of coal samples. The experiments were done using a magnetic suspension balance using different sample preparations: powder, pellet, and fragment forms. Complementary coal characterization was also done using X-ray diffraction (XRD) analysis, immediate analysis (moisture, ash, and volatile material), and elementary analysis (C, H, N, and S).
Materials and methods
The coals of the Figueira deposit belong to the Parana Basin (Rio Bonito Formation) and are classified as highly volatile bituminous coals. The full coal seam of Cambui has the following characteristics in the subsurface area where the samples were collected (Mine-07/Well F07–55): the full coal seam is 0.85 m thick and the coal contained in the layer is 0.61 m thick.
Mineral matter and elementary analysis
The determination of the total carbon percentage, hydrogen percentage, total nitrogen percentage, and total organic carbon percentage was carried out on a TruSpec® elementary analyzer from LECO (Ramos et al., 2015). These analyses were done according to ASTM D-5373 (2008). The total sulfur percentage was determined using a model SC-632 elementary analyzer from LECO. These analyses were done according to ASTM D-4239 (2008). The moisture, ash, volatile material, and fixed carbon contents of the samples were determined according to ABNT NBR 8289 (1983), ABNT NBR 8290 (1983), ABNT NBR 8293 (1983), and ABNT NBR 8299 (1983), respectively.
XRD
The sample preparation methodology was the powder method, which consists in sample preparation for total analysis followed by sample preparation for clay fraction analysis (Alves, 1987). After preparation, the analyses were performed on a Bruker D8 Advance diffractometer with a copper tube operating at a voltage of 40 kV, current of 30 mA, 2-theta scan of 3°–70°, step of 0.015°, and count time of 0.2 s.
Sample preparation for adsorption isotherm
The sample preparation can be divided into three different steps: fragment, powder, and pellet. These steps were used in the preparation of the three different series that were carried out with the same pieces from Cambui coal field.
Fragment sample: Initially, a coal piece from Cambui coal field was divided into smaller ones. Afterward, three pieces of fragments with dimensions of approximately 5 mm (close to 200 mg each) were collected for the PTGA analysis of representative manner. Before analysis, the fragments were heated in an oven at 110°C for 120 min to remove their moisture (ABNT NBR 8293:1983, 1983; Clarkson and Bustin, 2000; Joubert et al., 1973; Krooss et al., 2002; Ozdemir et al., 2003).
Powder sample: After carrying out the adsorption experiment with the fragment, the three small pieces with dimensions of approximately 5 mm were passed through a series of sieves and only the material with dimensions smaller than 0.25 mm (approximately 160 mg) was heated in the oven at 110°C for 120 min to remove its moisture (ABNT NBR 8293:1983, 1983; AS1038.3-1989, 1989; Clarkson and Bustin, 2000; Joubert et al., 1973; Krooss et al., 2002; Ozdemir et al., 2003).
Pellet sample: The first step was the standardization of the pellets. For that, pellets were made with pressures of 1, 2, 3, and 4 kN and amounts of coal varying between 200 and 220 mg using a hydraulic press apparatus (Specac, 15 t). It was observed that the pellets pressed at 1 and 2 kN broke during the realization of the experiments when they were submitted to the CO2 pressures, while the pellets pressed with 3 and 4 kN showed the expected behavior, remaining intact. With this, the preparation of all pellets was standardized to a pressure of 3 kN for 60 s.
After finishing the adsorption experiments on the powders, the powder samples were pressed according to the methodology above and each pellet sample, with 13 mm of diameter, 1 mm of depth, and close to 150 mg of mass, was heated in the oven at 110°C for 120 min before the PTGA analysis. Figure 1 represents the sample preparation process used for the adsorption experiments and shows that three different pieces of the same raw sample were separated. Subsequently, the respective Langmuir volumes were obtained for each of the methodologies.
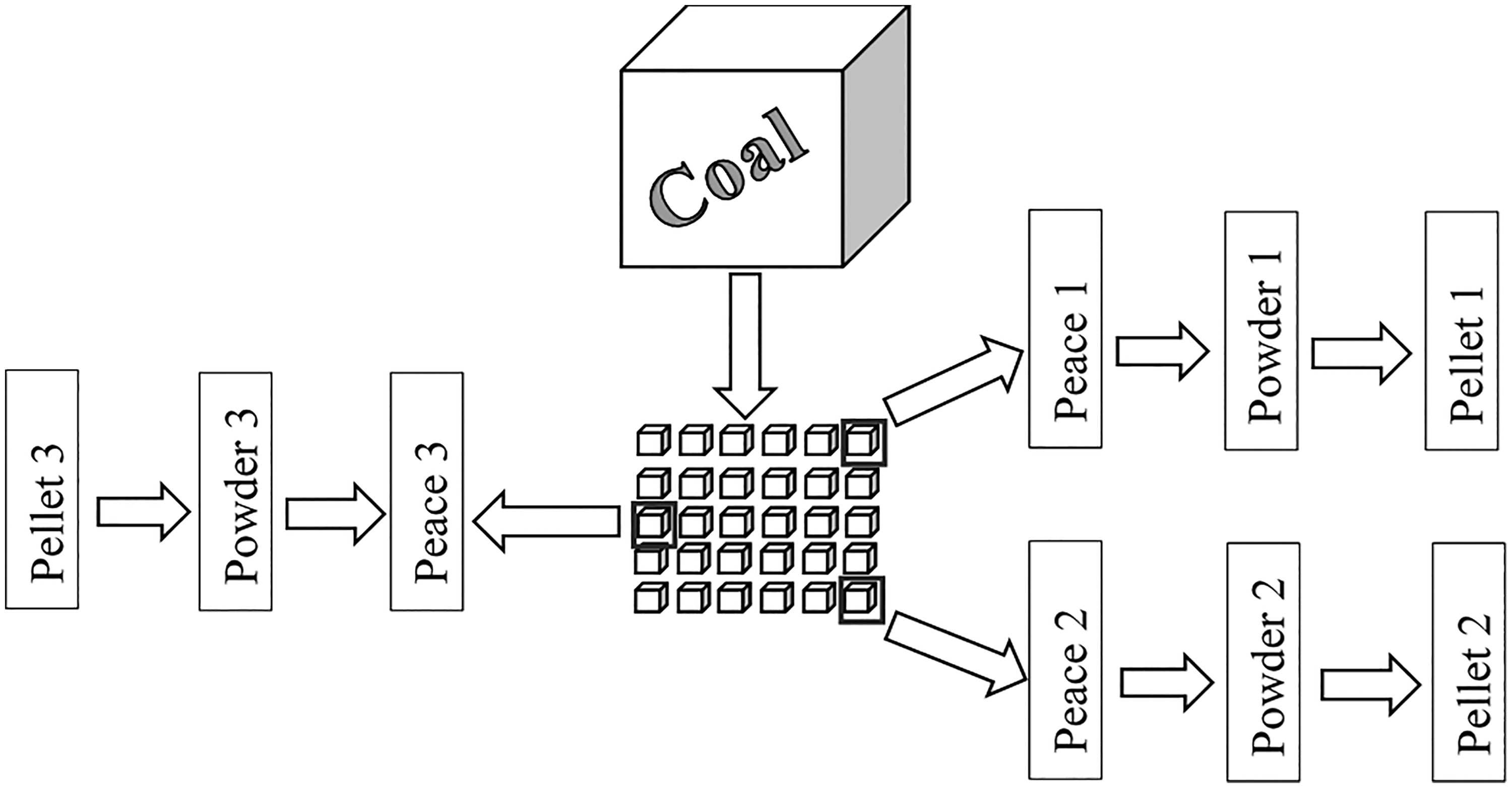
Drawing representing the sample preparation process used for the adsorption experiments.
Adsorption measurements
Three different techniques can be used to setup an adsorption isotherm: manometric, volumetric, or gravimetric adsorption measurement. Both manometric and volumetric apparatuses are equipment with a reference volume used for calculating the adsorbed mass. While manometric apparatus uses only the pressure variation for calculating the adsorbed mass, the volumetric apparatus uses both pressure and volume variation for this calculation. The gravimetric technique measures the mass variation due to the gas adsorption on the surface of solids with better reproducibility and precision than the volumetric and manometric methods (Dreisbach and Losch, 2002).
For gravimetric adsorption measurement, a magnetic suspension balance is used. Three different vertical positions of the suspension magnet (Figure 2), which can be manually or automatically controlled, correspond to three different measuring positions:

Universal sorption-measuring apparatus with a magnetic suspension balance: (a) zero point position, (b) sorption measurement, and (c) density measurement.
A. Zero point: the permanent magnet alone is in a freely suspended state, allowing the balance to be tared and calibrated.
B. Measuring point 1: the sample and sample container are lifted up and their total mass is weighed.
C. Measuring point 2: the sinker is raised together with the rest of the system and both masses are weighed together. By subtracting the first measuring point value from the second, the mass of sinker is given and, consequently, the gas density can be calculated. The magnetic suspension offers the possibility of lowering the suspension magnet in a controlled way to a second stationary position a few millimeters below the measuring position (Dreisbach and Losch, 2002).
The different sample adsorption experiments were executed in a Rubotherm PTGA magnetic suspension balance (MSB Standard Version HPII), following a methodology that has already been applied in other papers (Dreisbach and Losch, 2002).
The first part of the experiment is done with an inert gas, namely helium (99.999%, White Martins). The test consisted of putting the sample in different pressure environments and checking the mass variation. For this part of the experiment, a pressure variation between 0 and 50 bar was used and the time between each pressure change was the time taken until mass stabilization. The second part of the experiment was done with CO2 (99.99%, White Martins). Again the mass variation was checked for different pressure values. As for helium, the pressure was kept between 0 and 50 bar and the time for each pressure step was the time taken until mass stabilization. All adsorption experiments using PTGA were done at a constant temperature close to 25.5°C, controlled by a thermostat and a water bath, aiming to understand the different sample preparation effect under the same experiment conditions.
The measuring parameters in gravimetric experiments with a magnetic suspension balance are the measured quantities: balance reading (Δm), pressure (P), and temperature (T). Due to the buoyancy effects acting on the adsorbent and the components of the balance holding the sample, this measure is equal to the difference (Dreisbach and Losch, 2002)
This equation expresses m(P,T), the adsorbed mass, as a function only of the mass difference Δm(P,T), gas density ρ(P,T), volume of the balance components holding the sample (VSC), and sample volume V. Using the converted adsorbed mass value, it is possible to calculate the adsorption volume with equation (2) (Da Silva et al., 2004)
The stored volume Varm(P,T) is a function of the adsorbed mass calculated before, the gas molar mass (M), and the constants that follow standard conditions (T0 = 20°C; P0 = 1 bar) of measurement from the Brazilian National Petroleum Agency (ANP). Using the stored volume, a volume isotherm can be constructed as a function of each pressure. Together with the isotherms, it is possible to find the Langmuir volume, which represents the theoretical adsorption capacity, of each sample preparation by plotting P/Varm(P,T) × P (Da Silva et al., 2004).
Results and discussion
XRD
Through the arrangement of the peaks found in the X-ray diffractograms of the samples (Pellet II and Pellet III), it is possible to identify a large amount of amorphous material, making the background of the spectrum quite high. But, in the Pellet II sample, it is possible to determine that the major minerals are quartz (SiO2) and kaolinite Al2Si2O5(OH)4. The results of the analysis of the Pellet III sample showed the minerals quartz (SiO2), kaolinite Al2Si2O5(OH)4, and illite (K, H3O)(Al, Mg, Fe)2(Si, Al)4O10. These results are in agreement with data from the literature, which mentions clays, sulfides, carbonates, and quartz as the most common minerals in the coals (Speight, 2005). It should be noted that similarities in the identified minerals (XRD) were observed for the Pellet II and Pellet III samples (Figure 3). However, we can verify the presence of the mineral illite in the Pellet III sample. Because coal samples have a low mineral content and a high content of organic matter, identification and direct quantification of the minerals by XRD were hampered by the interference of the organic matter present (Yan et al., 2016). Another observed fact is the heterogeneity of the Pellets II and III samples and the difference that can occur in the minerals found. The XRD spectrum for Pellet I shown in Figure 3 is similar to Pellet II.

XRD analysis of Cambui coal field samples: Pellet I, Pellet II, and Pellet III.
Immediate and elemental analysis
Some of the main factors proven to have a significant influence on the Langmuir volume are the mineral matter, moisture, and volatile material. When the coal composition has high contents of moisture and volatile material, the adsorption capacity decreases, while the adsorption capacity can vary with the quantity and type of mineral matter. However, when the organic matter content is higher, the adsorption capacity of coal also increases (Busch and Gensterblum, 2011). With the aim of characterizing the Cambui coal field, the immediate analysis results, showing that it is composed of moisture (4.04 ± 0.05%), ash (16.92 ± 0.03%), volatile material (34.93 ± 0.10%), and fixed carbon (44.11 ± 0.07%), as well as the elementary analysis results, showing a composition of carbon (63.96 ± 0.14%), hydrogen (3.80 ± 0.03%), and nitrogen (1.54 ± 0.17%), are in accordance with the literature values found (Gomes, 2002). The total sulfur content found was 5.76 ± 0.004%, in correspondence with other coals from Paraná Basin, which have values of sulfur content between 3 and 8% (GöThe, 1989).
The carbon content is within the expected range for low-rank Brazilian coals. The hydrogen and nitrogen contents are low in the sample, as expected. On the other hand, the total sulfur contents presented significant variation in the sample. Sulfur contents between 3 and 8% are also verified (GöThe, 1989). It should be noted that, from the point of view of validation of the proposed methods, more differentiated sulfur contents are interesting because they suggest a varied mineralogical composition. On the other hand, mineral matter was not determined quantitatively, because the high quantity of organic matter masked the measurement.
CO2 adsorption isotherm analysis
After the geochemical characterization and XRD, adsorption isotherms were done in three different series, all of them with the pressure ranging from 0 to 50 bar and a constant temperature of 25.5°C. The results of adsorption experiments for the first series of fragment, powder, and pellet are shown in Figure 4(a), through an isotherm plot, and in Figure 4(b).

(a) Adsorption isotherms of CO2 and (b) Langmuir equation, both for the first series of experiments.
In both Figure 4(a) and (b), it is possible to notice two different experiments for pellet form. This was necessary because the first experiment (Pellet I) resulted on higher adsorption values than expected, so another pellet was made with the same material of the first one and a second experiment was done (Pellet Ia). The second result was closer to that for the powder one, as expected before. The reason for the difference between the pellet tests was probably the presence of fractures on the surface of the Pellet I, which created preferential paths via which the gas could be adsorbed, leading to the result being overstated.
According to Gray (1987) the coal has a natural dual porosity structure that consists of the coal matrix and some fractures. More than 95% of the gas occurs as adsorbed gas in the sorption space of the abundant micropores. The gas migrates across the micropore system and closely spaced fractures surrounding the coal matrix, creating preferred flow paths for the CO2. Probably, after powder pressing, fractures were formed inside the pellet, like the ones on the surface, and contributed to the ability of the coal structures to adsorb gas (Gray, 1987; Pinkun and Yuanping, 2013). Figure 5 shows the difference between the two pellets done together with a mark over the fracture of the first pellet.

(a) Pellet Ia after the experiment and (b) Pellet I after the experiment.
Still in Figure 4(a) and (b), it can be seen that the adsorption is significantly lower for the fragment than for the pellet and powder. This difference can be correlated with the time of the experiment. In this first series of experiments, it was proposed that the time between each pressure change was constant at around 3 h for all the samples. However, because the fragment is less porous than the powder or pellet form, it takes more time for the gas to diffuse inside its matrix and, consequently, to be adsorbed. Therefore, the fragment experiment did not reach the maximum adsorption at each pressure, presenting a lower Langmuir volume at the end of the analysis.
In order to demonstrate that the fragment takes a long time to stabilize the mass of gas adsorption at each of the pressures during the experiment, Figure 6 presents a part of the first series experiment. In this plot it is shown the adsorbed mass as a function of time at 2 bar for powder, pellet, and fragment. The equations close to each curve are a linear model for the last points, which, supposedly, should match the stabilization part of the curve, resulting in an angular coefficient as close to zero as possible. Through each equation and a close look at the curves it is possible to notice that both pellet and powder samples were stable after less than 100 min at 2 bar; however, the fragment was not, confirming the theory that the adsorption process is slower for the fragment form.

Plot showing mass stabilization as a function of time for each of the different sample preparations at a pressure of 2 bar.
After the results of the first series of experiments, it was decided that for a better comparison between the methods of sample preparation it would be necessary to wait until complete stabilization of each pressure was achieved, which would imply a different experiment time for the three samples. The second series was done with this concept and the results of adsorbed volume were closer between the different sample forms than on series one. The results of this second series are shown in Figure 7(a) and (b).

(a) Adsorption isotherms of CO2 and (b) Langmuir equation, both for the second series of experiments.
Following the same method as for the second series, during the third series each pressure was applied until complete stabilization was achieved, and thus different experiment times were required for each sample preparation. The results from this series agree with the results from the second series, because in this case the fragment, powder, and pellet presented closer adsorption values. As can be seen, when removing the influence of time on the experiment, analyzing each pressure until complete stabilization is achieved, the adsorbed volume for each sample preparation is almost the same, confirming the developed theory. The similarity between the powder, fragment, and pellet sample preparations can be seen from the isotherms in Figure 8 too.

(a) Adsorption isotherms of CO2 and (b) Langmuir equation, both for the third series of experiments.
At the same time, although the different sample preparations result in close adsorbed volume results, powder and pellet experiments required only about 2–3 h for total stabilization at each pressure, obtaining total analysis times of 14–21 h, while for the fragment samples it took approximately 160 h to complete an analysis, with a stabilization time for each pressure ranging from 14 to 33 h. These data show that time is a much more significant variable for an experiment using fragments than for one using the other two forms of samples.
Using the equation found with the second plot from each series (P/Varm × P), it is possible to calculate the Langmuir volume from each experiment. All these results are put together in Table 1 to simplify the comparison between the data acquired.
Main results acquired for the three different sample preparations in the three series.

The results shown in Table 1 agree with the conclusions drawn from the previous plots. The first series presented an invalid value of Langmuir volume for the fragment, namely 41.84 cm³/g, due to the insufficient stabilization time step between pressures. Pellet I examined in this series resulted in a Langmuir volume of 69.44 cm³/g, which is too high when compared to the value for powder of 50.00 cm³/g. However, fractures were found in Pellet I and the experiment was repeated (Pellet Ia), resulting in an adsorption value closer to that of the powder, namely 55.25 cm³/g. The results of the second series showed that, as in first series, the values of the Langmuir volume for Powder II and Pellet II were relatively close: 49.02 and 47.39 cm³/g, respectively. Still, the difference from first series was that, when the stabilization time was achieved for each pressure, the Langmuir volume of Fragment II, 49.50 cm³/g, was the same as that of the powder, which is the most common sample preparation found in the literature and was used as a reference value.
The three ways of preparing Series 2 and 3 samples analyzed showed that the adsorption isotherms can be obtained from any of the preparation forms (powder, pellet, or fragment), if the sample has been left long enough at each experimental pressure point to allow its total stabilization. However, in the case of fragments, for each pressure point, the stabilization of the adsorbed gas mass ranged from 14 to 33 h, giving a mean total time of around one week for each sample. On the other hand, experiments with powder samples took about one day to provide results, while for the pellet samples, the results were obtained in 18 h on average, demonstrating a faster data acquisition.
Moreover, the results of the adsorption isotherms obtained in Series 2 and 3 showed a small variation within the respective series and the variation of results between Series 2 (47.39–49.50 cm3/g) and Series 3 (38.02–40.48 cm3/g) occurred due to the heterogeneity composition between samples.
The Langmuir isotherms satisfactorily represented the adsorption data found for this work. Therefore, the Langmuir monolayer mechanism can represent CO2 adsorption for pressures of up to 6 MPa. The maximum CO2 adsorption capacity of these coals ranged from 38 to 70 cm3/g. However, the projected CO2 storage capacity for the coals and mentioned by Saghafi et al. (2007) in their work ranged from 38 to 53 m³/t under the conditions “as received” being in accordance with the mentioned literature for different samples of coals at temperatures up to 27°C according to Table 2.
Study of adsorption properties in coal samples from the Sydney Basin, Australia (Saghafi et al., 2007).

Conclusions
In this study, a comprehensive set of CO2 adsorption data was presented, including measurements on samples such as fragments, powder, and pellets, to improve the understanding of carbon capture and storage mechanisms. The adsorption analyses shown in the results suggest that, for the different sample preparations demonstrated here, accurate measurements are obtained that allow reliable estimates of gas storage in coal layers. The values obtained through second and third series proved the similarity between the results of Langmuir volume for the different sample preparation, ranging from 47.39 to 49.50 and 47.39 to 49.50 cm3/g, for Series 2 and Series 3, respectively.
Besides that, the first series showed that the correct stabilization time for the adsorbed mass of CO2 is essential to obtain precise and reliable results, especially for the fragment form. Due to this time particularity, it becomes almost impracticable to perform experiments with this sample preparation, once that the each test might take almost a week. On the other hand, both powder and pellet form did not present this time problem, finishing each experiment with less than a day.
In practice, due to shorter time, it is most suitable to perform the experiments on powder and pellet samples than on fragment and, in addition, both pellet and powder form present smaller variation on the results, once that both methods of preparation enable the homogenization of the sample before performing the tests. However, at last, the different results from Pellet I and Ia, on first series, showed that the pellet is extremely sensitive to any variation in the sample preparation, such as fractures on the surface, which can overestimate the Langmuir volume. Because of that, it can be considered simpler and more reliable to perform experiments with powder, even if it requires a special sample container.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


