Abstract
A new preconcentration procedure named “Magnetic textile solid phase extraction” was developed. A piece of nonwoven textile (fabric) was used for the immobilization of copper phthalocyanine dye and employed for the adsorption of crystal violet and safranin O from large volumes of sample. The bound dye was eluted into small volume of methanol. The limit of detection and limit of quantification values for safranin O were 2.66 µg/L and 8.87 µg/L, while for crystal violet these values were 2.17 µg/L and 7.23 µg/L, respectively. Linearity in the range 5–100 µg/L was observed. In order to prepare magnetically responsive adsorbent, an iron-based standard staple was inserted in the textile material using an office stapler. The developed procedure can be easily adapted for the detection of large variety of analytes in biochemistry, biotechnology, environmental technology, and forensic analysis.
Introduction
Analysis of various types of biologically active compounds, inorganic and organic pollutants, and radioisotopes in biological and environmental matrices often requires preconcentration of the target compounds from large volumes of solutions and/or suspensions. Currently, various types of solid phase extraction (SPE) have been employed to isolate and preconcentrate desired components from a sample matrix; SPE is an excellent alternative to the liquid–liquid extraction. The standard column SPE is usually used for separation and preconcentration of analytes from milliliter-sized volumes of solution. In order to extract target analytes from large sample volumes, magnetic solid-phase extraction (MSPE) employing magnetically responsive adsorbents was developed in 1999 [1]. This approach has several advantages over traditional solid phase extraction, namely: (a) it avoids time-consuming and tedious on-column SPE procedures, (b) it provides a rapid and simple analyte separation that avoids the need for centrifugation or filtration steps, (c) the magnetic adsorbents may have high selectivity, even when complex biological and environmental samples are analyzed, (d) MSPE can also be used for samples containing suspended solids, (e) since the majority of sample impurities are diamagnetic, they do not interfere with magnetic particles during the magnetic separation step, and (f) automation of the whole process is possible, which leads to rapid, selective, sensitive, and repeatable methods for routine analyte determinations. For these reasons, MSPE is now a very useful and widely used preconcentration procedure for both organic and inorganic species [2].
Other procedures utilizing a magnetic stir bar or other mixing element covered with an adsorbent layer, namely stir bar sorptive extraction (SBSE) [3], rotating-disk sorptive extraction [4], and stir-rod sorptive extraction [5] have been developed recently. Also stir membrane extraction procedure, using an iron wire to enable magnetic stirring of the membrane during the extraction process, has been utilized for analyte preconcentration [6].
In this paper, we describe a new, low-cost extraction and preconcentration procedure named “Magnetic textile solid phase extraction” (MTSPE) where appropriate textile (fabric) is used as a carrier for the immobilization of an appropriate affinity ligand. In order to prepare magnetically responsive adsorbent, an iron-based standard staple was inserted in the textile material using an office stapler. Two important dyes, namely crystal violet (a well-known topical antiseptic) and safranin O (a biological stain used in histology and cytology) were preconcentrated on nonwoven textile with immobilized copper phthalocyanine dye.
Materials and methods
Reagents and materials
Nonwoven fabric (Funky Felt Sheet, 9 × 12 inch, white acrylic felt) was obtained from a local stationery; its thickness was 1.5 mm and mass was ca. 35 mg/cm2 (350 g/m2). Reactive copper phthalocyanine dye (Ostazin turquoise V-G; C.I. Reactive Blue 21) was purchased from Spolek pro chemickou a hutni vyrobu, Czech Republic. Crystal violet (Basic Violet 3; C.I. 42555; mol. wt. 408.0) and methanol were from Lachema, Czech Republic, while safranin O (Basic Red 2; C.I. 50240; mol. wt. 350.8) was from Sigma, USA. NaCl, NaOH, HCl and anhydrous Na2CO3 were supplied by Lachner, Czech Republic. A regular office stapler and iron-based standard staples were used for magnetic modification.
Preparation of magnetic adsorbent material
The nonwoven fabric sheet was cut into 1 × 1 cm squares. The textile squares were dyed with reactive phthalocyanine dye as follows. Approximately 100 pieces of textile squares were suspended in 300 mL of water and mixed with 2 g of Ostazin turquoise V-G (C.I. Reactive Blue 21) and 18 g of sodium chloride. The mixture was warmed to 70℃ and 15 min later, 15 g of anhydrous sodium carbonate was added. The mixture was stirred at 70℃ for 4 h and then left overnight at ambient temperature without mixing. The blue textile squares were thoroughly washed with water and the remaining free color was removed using an extraction with methanol. The extracted squares were air dried and stored in a dry place before use. The average mass of one dyed textile square (area 1 cm2) was 35.4 ± 2 mg. In order to prepare magnetically responsive squares, an iron-based standard staple was inserted in the textile material using an office stapler.
Adsorption of dyes on copper phthalocyanine dyed textile
All following experiments were performed with one half of the standard copper phthalocyanine dyed textile square with the area 0.5 cm2 and mass 17.7 ± 1 mg.
Influence of initial pH value of dye solution on adsorption efficiency was studied in a batch system using one piece of dyed textile incubated in 10 mL of 100 mg/L dye solution of particular pH (3–9) for 3 h. Absorbance of dye solution before (Ai) and after adsorption (Af) was measured by UV-VIS spectrophotometer (Cintra 20; GBC Scientific Equipment, Australia) at wavelengths 420 nm and 456 nm for safranin O and crystal violet, respectively. The adsorption efficiency (E) was calculated using the following formula
Time dependence and kinetic studies were carried out as described previously [7,8]. One piece of dyed textile was incubated in 10 mL of 50 mg/L dye solution for 0–360 min. Experimental data were evaluated using pseudo-first-order and pseudo-second-order kinetic models.
The adsorption of crystal violet and safranin O on phthalocyanine dyed textile was tested in a batch system at room temperature. One piece of dyed textile was incubated in 10 mL of dye solution (pH 7) with concentration ranging between 0 and 200 (safranin O) or 250 (crystal violet) mg/L on a rotator mixer (Dynal, Norway, 27 rpm) for 3 h. The equilibrium adsorption data were analyzed, as described previously [7,8], using nonlinear regression analysis (Microsoft Excel, function Solver-add) by Langmuir and Freundlich adsorption isotherm models. Fitting to each isotherm model was assessed on the basis of standard error of estimate (SEE) values.
Magnetic textile solid phase extraction
In a typical procedure, magnetically responsive phthalocyanine dyed textile 1 × 1 cm square was inserted into 100 mL of distilled water with a trace amount of planar organic dye tested (0.5–10 µg) and incubated for 3 h (200 rpm) at room temperature by means of either a standard horizontal shaker or (due to the presence of magnetic staple) a magnetic stirrer. Then, the square with adsorbed dye was taken out from solution (e.g. using a permanent magnet placed on the outer wall of the vessel, or by a magnetic stirring bar retriever), and dried using paper towel. The adsorbed dye was extracted from textile square into 2 mL of methanol during shaking on automatic rotator for 30 min at room temperature. This methanol extract was measured spectrophotometrically at maximum wavelength specific to each of the dyes tested, namely 590 nm for crystal violet and 528 nm for safranin O. The absorbance of the released copper phthalocyanine dye from textile square (the blank) was subtracted from the absorbance value of each sample. Dye concentrations were determined from calibration curves.
The relative recovery (R) of dyes after the adsorption and elution processes was calculated using the following formula, where mi (µg) is the mass of dye used in the experiment, Cr (µg/mL) is the concentration of eluted dye, and the constant corresponds to the volume of the elution agent (2 mL).
Results and discussion
Preparation of magnetic textile affinity adsorbent
This new magnetic solid phase extraction procedure is based on the use of low-cost, easily available material, namely textile. Textile (fabric) is produced in huge quantities, using several technologies and various raw materials [9]. Incorporation of a piece of the iron-based wire (e.g. a steel staple) into the piece of textile enables to prepare magnetically responsive material (Figure 1). Magnetically modified pieces of textile (fabric) can be moved using appropriate laboratory magnetic stirrers in a similar way as magnetic stirring bars. At the end of the extraction process the piece of textile can be separated magnetically, as shown in Figure 1. The whole process of magnetic textile solid phase extraction is shown in Figure 2.
(Top) Textile squares prepared from nonwoven textile. From left to right, native textile, copper phthalocyanine dyed textile, and dyed textile with inserted iron-based standard staple. (Bottom) Textile adsorbent in water sample and its removal using a permanent magnet. Scheme of magnetic textile solid phase extraction.

Textile material enables immobilization of various affinity ligands. Reactive copper phthalocyanine dye was easily immobilized on small squares of acrylic felt, using modified procedure taken from the textile dyeing industry. Immobilized copper phthalocyanine, due to its specific molecular structure, exhibits high affinity to selected low molecular weight organic compounds such as polyaromatic hydrocarbons [10], heteropolyaromatic dyes [11], and triphenylmethane dyes [12]. That is why crystal violet (a triphenylmethane dye) and safranin O (a heteropolyaromatic dye) were selected as model analytes to test the new, textile-based solid phase extraction procedure. Crystal violet is known for its antibacterial, antifungal, and anthelmintic properties. Together with another triphenylmethane dye—malachite green it was often employed in aquacultures; due to their potential carcinogenic properties their use has been banned in many countries. Nevertheless, there are still countries where triphenylmethane dyes are routinely used; that is why simple detection of these dyes in water samples is necessary [13]. Safranin O was used as a model analyte with a different structure. The chemical structures of both tested dyes are shown in Figure 3.
Chemical structures of crystal violet (left) and safranin O (right).
Study of the adsorption process
The preliminary experiments with native nonwoven textile have shown that there is no binding of the tested dyes from diluted solutions. The adsorption of crystal violet and safranin O on copper phthalocyanine dyed textile was studied in the standard way. pH value is an important parameter significantly affecting the adsorption processes [14]. As can be seen from Figure 4(a), the initial pH of dye solutions significantly influenced the adsorption efficiency. In the case of crystal violet adsorption, the lowest efficiency was observed at low pH; with increasing pH values, the adsorption efficiency increased. The highest dye removal was found to be at pH 7–9. Adsorption of safranin O exhibited a similar trend, with the lowest efficiency at low pH and maximum efficiency at pH higher than 5. It is obvious that neutral and alkaline pH values are more convenient for adsorption of both tested dyes. Previous studies also indicated that change of ionic strength has negligible effect on the adsorption process [11,12].
(a) Dependence of initial pH of crystal violet (▪) and safranin O (Δ) solutions (100 mg/L, 10 mL) on adsorption efficiency. (b) Time dependence of crystal violet and safranin O adsorption on phthalocyanine-dyed textile (conditions: room temperature; total volume 10 mL; dye concentration 50 mg/L). (c) Langmuir (…..) and Freundlich (_._._) adsorption isotherm models describing adsorption of crystal violet and safranin O. Ce – concentration of unbound dye in equilibrium (mg/L); qe – amount of dye adsorbed on unit mass of adsorbent in equilibrium (mg/g). (d) Time dependence of crystal violet (dark columns) and safranin O (light columns) recovery from 100 mL of solutions containing 2.5 µg of each dye. (e) Dependence of crystal violet and safranin O recovery (expressed as recovered mass of dye) on the amount of dye present in 100 mL of solution. (f) Dependence of crystal violet and safranin O relative recovery on the amount of dye present in 100 mL of solution.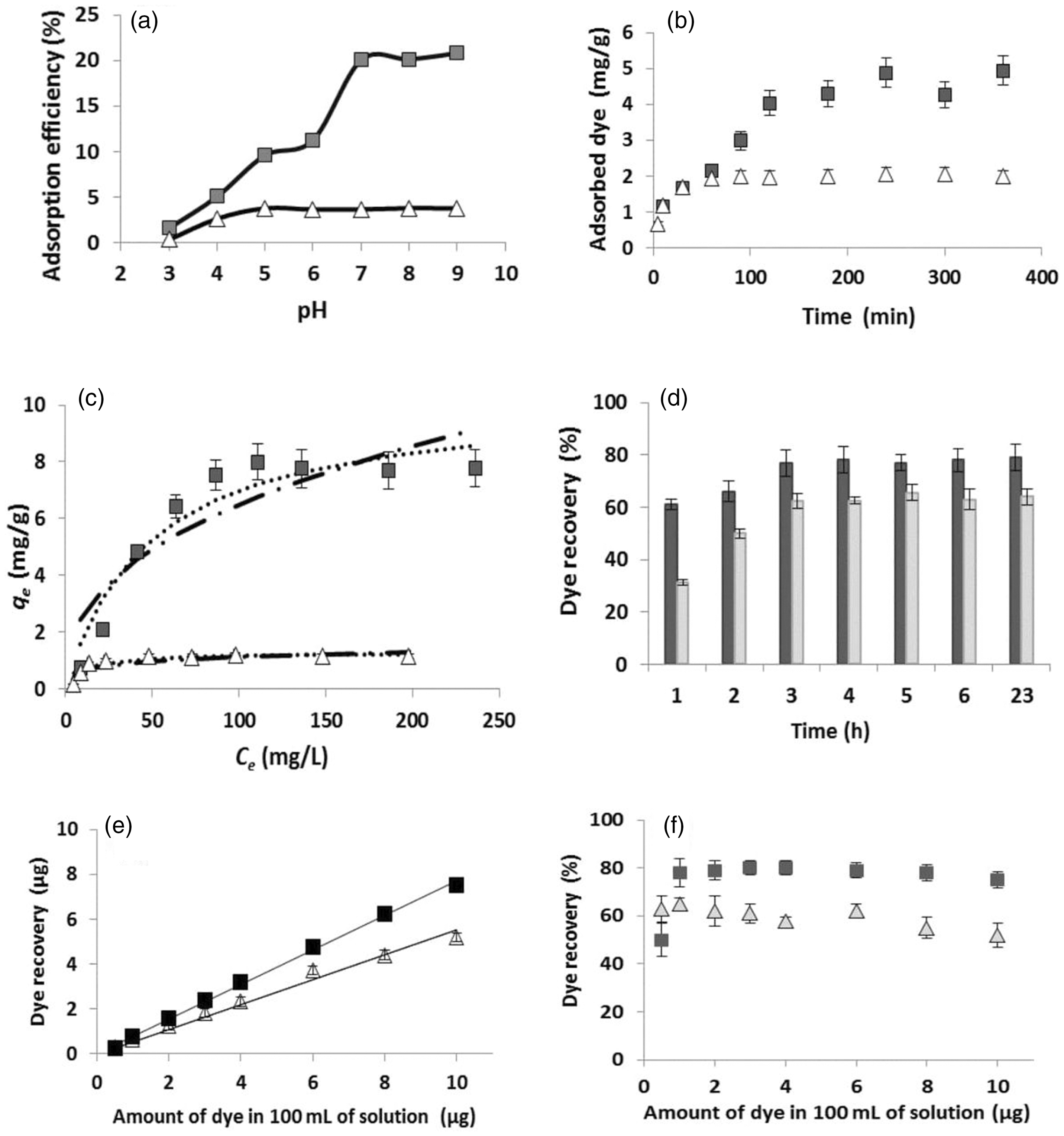
Figure 4(b) demonstrates the time necessary to reach the adsorption equilibrium for both dyes at pH 7; it is evident that safranin O needs 120 min, while crystal violet 180 min. Hence, the incubation time for the study of adsorption isotherms was set to 3 h.
Adsorption parameters obtained from the Langmuir and Freundlich isotherm models.
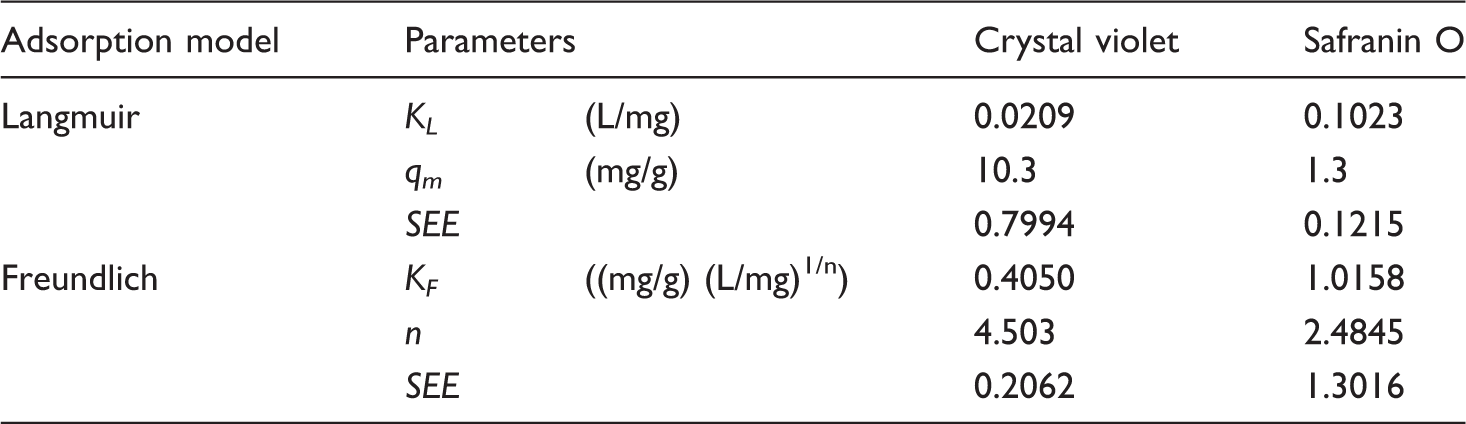
KL: Langmuir constant; qm: maximum adsorption capacity; KF and n: Freundlich constants.
Kinetic parameters obtained from pseudo-first-order and pseudo-second-order models.
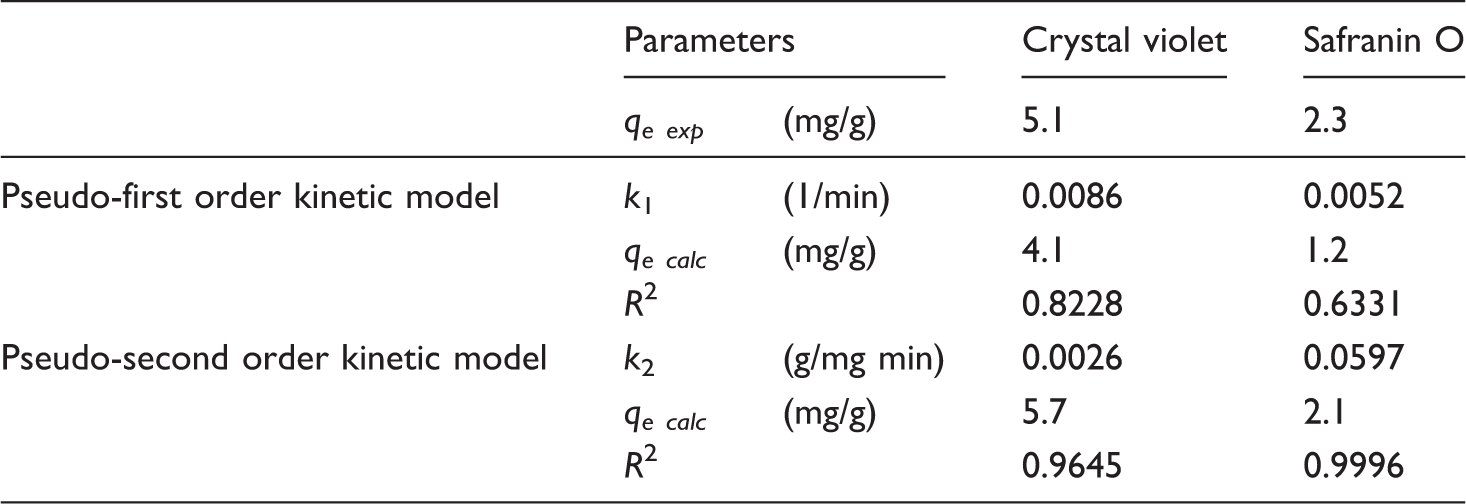
Magnetic textile solid phase extraction
Magnetic textile solid phase extraction enables simple preconcentration of target analytes. Figure 4(d) shows the result of the extraction and subsequent elution of tested dyes from 100 mL of water containing 2.5 µg of each dye. Extraction time varied from 1 h to 23 h, elution with 2 mL of methanol took 30 min. It can be seen from Figure 4(d) that 3 h extraction enabled maximum dye recovery from 100 mL of solution.
Figure 4(e) demonstrates the dependence of the dye recovery (combination of extraction and elution processes, expressed as recovered mass of dye) on the amount of dye in 100 mL of solution (concentration range 5–100 µg/L; pH 7), using 3 h extraction time. For both dyes the dependence is linear in the whole concentration range, with very high correlation coefficients (R2 = 0.9978 for crystal violet and 0.9850 for safranin O). The same data were used to demonstrate the dependence of the relative dye recovery on the amount of dyes in 100 mL of solution. The relative dye recovery was always higher than 50%, the highest value was ca. 80% (Figure 4(f)).
Limit of detection (LOD), limit of quantification (LOQ), and linearity were determined using the standard procedures [15]. The absorbance blank values were 0.00875 ± 0.001299 for safranin O determination (measured at 528 nm), and 0.03175 ± 0.0034551 for crystal violet determination (measured at 590 nm). Taking into account the parameters of the whole preconcentration process (2 mL of extractant, 100 mL total sample volume, minimum relative recovery set up to 50%), the LOD and LOQ values for safranin O were 2.66 µg/L and 8.87 µg/L, while for crystal violet these values were 2.17 µg/L and 7.23 µg/L, respectively. Linearity in the range 5–100 µg/L was observed for both dyes.
MTSPE can also be used to detect target analytes in natural water samples. It has to be taken into account that this procedure can only preconcentrate soluble analytes, not the analyte fraction adsorbed on particulate impurities present in natural water samples.
MTSPE can be compared with the original procedure called magnetic solid phase extraction (MSPE). In MSPE, surface modified magnetic particles or magnetically responsive composite particles are used as carriers for immobilization of appropriate affinity ligand. In many cases, the preparation of magnetic carriers requires a multistep procedure [16]. On the contrary, pieces of magnetic textile as carriers can be prepared in an extremely simple and rapid way.
Due to the extremely low cost of the magnetic textile adsorbent, its regeneration and reuse for analytical applications is not expected. New pieces of adsorbent should be used for preconcentration of target analytes present in studied samples.
Conclusions
This proof of concept paper clearly shows that appropriate textile (fabric) material can be easily modified into the magnetic form and used for efficient affinity ligand immobilization. The developed “Magnetic textile solid phase extraction” can be easily adapted for the detection of large variety of analytes in biochemistry, biotechnology, environmental analysis, and forensic analysis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Ministry of Education, Youth and Sports of the Czech Republic (Projects LD14075 and LO1305) and by the Ministry of the Interior of the Czech Republic (project No. VI20162019017). This work was also carried out in the frame of the COST Action CA16101 entitled “MULTI-modal Imaging of FOREnsic SciEnce Evidence - tools for Forensic Science”.


