Abstract
Partially reduced graphene oxide-Fe3O4 composite was prepared through in situ co-precipitation and used as an efficient adsorbent for removing Pb(II) from water. The composites were characterized by X-ray diffraction, high-resolution transmission electron microscopy, X-ray photoelectron spectra, Fourier transformation infrared, Raman spectrometer, N2 adsorption–desorption, vibrating sample magnetometer, and zeta potential analyses. The impacts of pH, contact time, adsorbent dosage, temperature, and foreign substances on Pb(II) adsorption performance were investigated. The adsorption mechanism, kinetics, and thermodynamics were analyzed. The results indicate that Fe3O4 is homogeneously anchored inside the thin graphene sheets, with a particle size of 15–20 nm, resulting in a very low remanence and coercivity. The composite shows excellent and efficient adsorption performance toward aqueous Pb(II): adsorption equilibrium was reached in 10 min with the adsorption percent and quantity of 95.77% and 373.14 mgċg−1, respectively, under a condition of pH = 6, adsorbent dosage 250 mgċL−1, and Pb(II) initial concentration 97.68 mgċL−1, with the subsequent magnetic separation taking only 10 s. The adsorption performance is dependent on adsorbent dosage. A lower dosage favors a higher adsorption quantity, implying a strong adsorptive potential for partially reduced graphene oxide-Fe3O4. The adsorption quantity reached 777.28 mgċg−1, given the dosage 100 mgċL−1. The adsorption is monolayer chemisorption, the whole process of which is controlled by chemisorption and liquid film diffusion. In terms of thermodynamics, the adsorption is an exothermic and spontaneous process.
Introduction
With the rapid development of the modern industry, various heavy metals like Pb(II), Cd(II), Cu(II), Hg(II), and Cr(VI) were discharged into the receiving water, causing serious environmental contamination. Pb(II) is one of the most toxic heavy metals, which is generated by mining, electroplating, dyeing, battery, textile, explosive, and other industries. Lead poisoning can bring serious risks to kidney, liver, blood, nerve, and reproductive systems, causing symptoms as anemia, chronic headache, dysentery, amentia, etc. For the sake of ecological stability and public safety, it is necessary to eliminate Pb(II) from water in a simple, efficient, and environmentally friendly way.
Various methods including chemical precipitation, ion exchange, membrane separation, flocculation, electrolysis, and adsorption (Canivet et al., 2014; Gdula et al., 2017; Manisha, 2013) were employed for treating heavy metal pollution. Among these methods, adsorption technique is considered to be one of the most promising strategies because of its safe, low-cost, simple, and efficient merits. Many types of adsorbents including polymers (Ge et al., 2015; Karthik and Meenakshi, 2015), oxides (Rahimi et al., 2015), carbonaceous materials (Dehghani et al., 2015; Hajati et al., 2015; Tao et al., 2015), mineral (Arancibia-Miranda et al., 2015), and biomass (Ding et al., 2016; Komkiene and Baltrenaite, 2016) have been extensively tested in removing lead from water. As for iron oxides, Rajput et al. (2016) synthesized magnetic Fe3O4 by chemical co-precipitation, the maximum Langmuir adsorption capacities were 34.87 (Cr6+) and 53.11 (Pb2+) mgċg−1 at 45℃, respectively. Nalbandian et al. (2016) synthesized α-Fe2O3 nanofibers with a diameter of 23 nm, reaching an equilibrium adsorption capacity of 90.9 mgċg−1 chromate (
Graphene, a single-layered carbon sheet with a hexagonal packed two-dimensional lattice structure, has attracted researchers' interest widely ever since its discovery by Geim and Novoselov (2004). Graphene has unique optical, electrical, magnetic, and mechanical properties, with extremely high theoretical specific surface area up to 2630 m2 g−1. Graphene oxide (GO) has the same basic framework as graphene, which can be synthesized via the oxidization and exfoliation of graphite. It is a potential adsorbent of heavy metals, due to its high specific surface area and various oxygen containing groups (carboxyl, hydroxyl, carbonyl, epoxy groups) in its structure. Unfortunately, the stacking and aggregation of GO sheets caused by the strong π–π interplanar attraction weakens its adsorption property. Moreover, the complete and efficient recovery of GO from water are remaining to be a challenge, due to the high hydrophilicity and dispersibility of GO in water. In view of the above-mentioned facts, preventing aggregation and improving recovery of GO from water via modification of GO are of primary importance to promoting practical application of GO-based adsorbents. Consequently, magnetic graphene (Cui et al., 2015; Guo et al., 2014; Yang et al., 2012; Zhang et al., 2013) was developed as a promising alternative for efficient adsorbent.
However, as reported by previous studies (Cui et al., 2015; Guo et al., 2014; Kumari et al., 2015; Yang et al., 2012; Yu et al., 2016), the graphene-related Pb(II) adsorbents has following drawbacks need to be overcome: (1) Low efficiency caused by long adsorption time; (2) low adsorption capacity remains to be improved. In this work, first, we prepared GO using new modified Hummer's method developed by our group (Bian et al., 2015), second, we prepared partially reduced graphene oxide-Fe3O4 (PRGO-Fe3O4) through in situ growth of Fe3O4 between GO sheets, thereby restricting the aggregation and stacking of the GO sheets to achieve superior adsorption property. The experimental results present that PRGO-Fe3O4 exhibit excellent and efficient adsorption property toward aqueous Pb(II). The as-prepared PRGO-Fe3O4 was characterized in detail by various techniques, subsequently, the adsorption performance, mechanism, thermodynamics, and kinetics toward aqueous Pb(II) were systematically explored.
Experimental
Chemicals and materials
Natural flake graphite (325 meshes, Qingdao Meilikun Co., Ltd., China), KMnO4 (Tianjin wind ship chemical reagent Co., Ltd., China), K2FeO4 (AR, Hubei CSW Chemistry Co., Ltd., China), H2SO4 (98 wt%, AR, Beijing chemical works, China), H2O2 (30 wt%, AR, Tianjin Huadong Reagent Co., China), FeCl3·6H2O (AR, Beijing Chemical Reagent Co., China), FeSO4·7H2O (AR, Beijing Chemical Reagent Co., China), Pb(NO3)2 (AR, Aladdin reagent Co., Ltd, China), NH3·H2O (25 wt%, AR, Beijing Chemical Works, China), hydrochloric acid (38 wt%, AR, Beijing Chemical Works, China), NaOH (AR, Beijing Chemical Works, China), EtOH (AR, Tianjin Yongda Chemical Reagent Co., Ltd, China). Deionized water was used throughout the overall experiments.
Materials preparation
Preparation of GO
GO was one-pot synthesized by oxidizing natural flake graphite based on new modified of Hummers method developed by our group (Bian et al., 2015). In a typical experiment, natural flake graphite (10 g), KMnO4 (6 g), and boric acid (0.01 g) were dispersed in concentrated sulfuric acid (100 mL), stirred for 1.5 h under a temperature of 5℃ in an ice bath. Afterward, another KMnO4 (5g) was slowly added, then the reaction vessel was transferred into a water bath under 35℃ and stirred for another 3 h to complete deep oxidation. Next, deionized water (250 mL) was dropped into the vessel, and the temperature elevated to 95℃, under which kept for 15 min, making the suspension turned brown, enabling the exfoliation and oxidation of the flake graphite. Subsequently, H2O2 (12 mL) was dropped into the brown suspension to resolve the residual oxidants and intermediates. Finally, the as-obtained suspension was centrifuged under 10,000 rpm for 20 min, washed with 1 mol·L−1 HCl and deionized water repeatedly to remove the residues, obtaining the terminal GO hydrogel.
Preparation of PRGO-Fe3O4
GO hydrogel (13.22 mgċmL−1, 8.76 mL) was ultrasonicated for 5 min, then a mixed solution with the molar ratio Fe2+/Fe3+ 0.55 was slowly added into the reaction vessel, followed by stirring for 15 min. Afterward, 25 wt% of ammonia solution was slowly dropped into the reaction vessel to complete the coprecipitation. Finally, the as-obtained sample was washed with ethanol for 3∼5 times, centrifuged and freeze-dried to obtain PRGO-Fe3O4.
Characterization
The morphology of the samples was observed using field-emission scanning electron microscopy (FESEM; JEOL, JSM-6701F) and high-resolution transmission electron microscopy (HRTEM; JEOL, JEM-2100). The phases of the samples were analyzed by X-ray diffraction (XRD; PANalytical, X'Pert PRO, Cu-Ka radiation). The functional groups in the samples were analyzed using Fourier transformation infrared spectroscopy (FTIR; Bruker IFS66V). Raman spectra were measured using Raman spectrometer (RS; Renishaw). The surface elemental compositions of the samples before and after adsorption were analyzed using X-ray photoelectron spectra (XPS; Kratos Axis Supra, Al-Ka radiation). N2 adsorption–desorption tests were carried out under specific surface and pore size analyzer (Micromeritics; ASAP 2020 V3.04 G). The pore size was calculated based on Kelvin equation. The magnetization curve was measured under a vibrating sample magnetometer (VSM, LakeSnore). The pH of the PRGO-Fe3O4 hydrogel was monitored by pH meter (Shanghai Leici Co., Ltd.; PHS-3E), zeta potential measured by micro-electrophoresis meter (Shanghai Zhongchen Co., Ltd.; China).
Adsorption property
In all, 0.15615 g of Pb(NO3)2 was dissolved in 1000 mL deionized water to obtain a solution with Pb(II) concentration of 97.6844 mgċL−1. A total of 20 mL of the as-prepared Pb(II) solution was transferred into a conical flask with a capacity of 50 mL, the pH was adjusted to the target value using 0.1 M HCl or NaOH. A certain dosage of PRGO-Fe3O4 was added into the Pb(II) solution, then the flask was shook under a certain temperature and a fixed rotation speed of 230 rpm in a shaker. After completing a certain contact time, PRGO-Fe3O4 was separated from Pb(II) solution using an NdFeB permanent magnet to obtain a clear solution. The concentration of the residue Pb(II) in the clear solution was measured by flame atomic absorption spectrophotometer (PerkinE Co., Ltd.; AA800).
The adsorption percent R
i
(%) and adsorption quantity Q
i
(mg g−1) were determined by following two equations
Results and discussions
Characterization
Phase analysis
Figure 1 shows the XRD patterns of GO and PRGO-Fe3O4. The broad diffraction peaks are indicative of small grain size. The peaks at 2θ = 30.3 °, 35.7 °, 43.2 °, 53.4 °, 57.4 °, 62.7 °, and 74.4 ° can be assigned to the (220), (311), (400), (422), (511), and (440) reflections, respectively, corresponding to the cubic reverse spinel crystal structure of Fe3O4 with cell constant a = 8.39 Å (JCPDS card No. 19-0629). While the peaks at 2θ = 10.4 ° and 21.3 ° are characteristic of GO and reduced GO, respectively (Cui et al., 2015), suggesting that GO was partially reduced during the coprecipitation process.
XRD patterns of GO and PRGO-Fe3O4. XRD: X-ray diffraction; GO: graphene oxide; PRGO: partially reduced graphene oxide.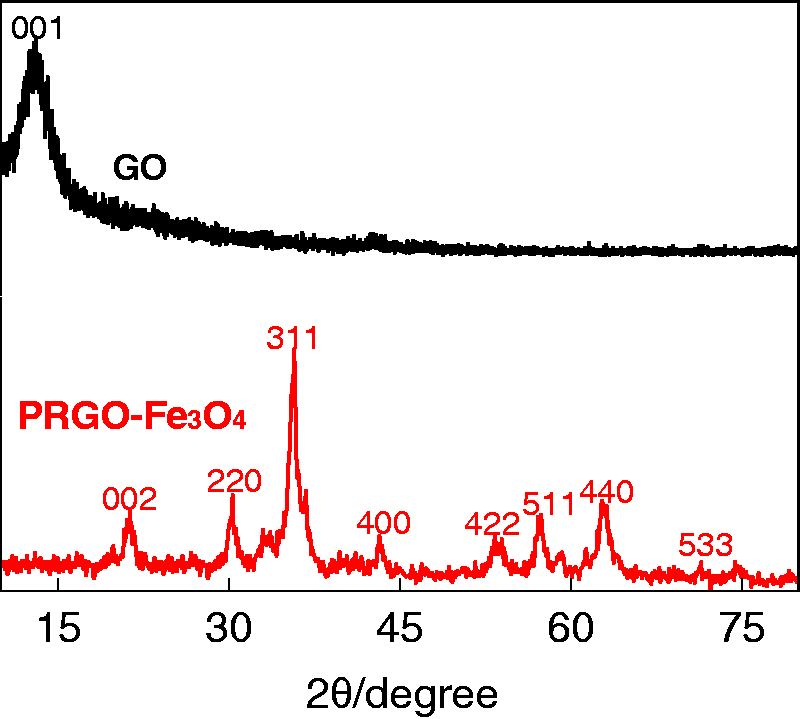
FTIR analysis
As shown in Figure 2, for PRGO-Fe3O4, the peaks at 3425 cm−1, 1633 cm−1, 1399 cm−1, and 1113 cm−1 arise from the stretching vibration of O–H, C=C, COO−, and C–O, respectively. Besides, the peak at 590 cm−1 is attributed to the stretching vibration of Fe-O from Fe3O4 (Cui et al., 2015; Guo et al., 2014; Kumari et al., 2015; Peng et al., 2015; Yu et al., 2016; Zhang et al., 2013). It should be noted that PRGO-Fe3O4 has the characteristic peaks of both GO and Fe3O4, confirming the existence of the two phases in PRGO-Fe3O4.
FTIR spectra of GO, Fe3O4 and PRGO-Fe3O4. Fourier transformation infrared; GO: graphene oxide; PRGO: partially reduced graphene oxide.
Morphology analysis
HRTEM was performed on the as-prepared PRGO-Fe3O4 to observe the features of morphology and structure in nanometer domain. Figure 3(a) shows that graphene exhibits a crumpled, nearly transparent lamellar structure, demonstrating that the graphene sheets are rather thin. Fe3O4 particles are nearly spherical, with a diameter of 15–20 nm, being wrapped and homogeneously anchored on the graphene sheets. It is clear that the Fe3O4 nanoparticles anchored on the graphene surface may act as nanoscale spacers to prevent the restacking of the graphene sheets, guaranteeing excellent adsorption property. In addition, since the Fe3O4 particles are wrapped inside the graphene sheets, agglomeration, oxidation, and corrosion by acidic media were effectively prevented, preserving the magnetic property. In Figure 3(b), the clear lattice fringes with interplanar distances of 0.25 nm correspond to the (311) planes of the cubic Fe3O4, corroborating a single crystalline nature of Fe3O4. The bright dots in the Selected area electron diffraction (SAED) pattern (inset) come from Fe3O4, while the rings from graphene, indicating a polycrystalline nature.
TEM images of PRGO-Fe3O4. (a) TEM image and (b) HRTEM image (inset: SAED pattern). PRGO: partially reduced graphene oxide. TEM: transmission electron microscopy; PRGO: partially reduced graphene oxide; high-resolution transmission electron microscopy.
Raman analysis
In Figure 4, Raman spectra of GO and PRGO-Fe3O4 display two prominent peaks at 1345 cm−1 and 1578 cm−1, which correspond to the well-documented D band and G band (Kumari et al., 2015; Zhang et al., 2013), respectively. It is widely known that the G band corresponds to the sp2 carbons, while the D band is associated with structural defects, amorphous carbon, or edges that can break the symmetry and selection rule. The values of ID/IG are 1.003 and 0.932 for GO and PRGO-Fe3O4, respectively, demonstrating that the ratio of sp2 carbon in PRGO-Fe3O4 is higher than that in GO, implying that GO was reduced partially during the coprecipitation process. This is consistent with the conclusion drawn from XRD.
Raman spectra of GO and PRGO-Fe3O4. GO: graphene oxide; PRGO: partially reduced graphene oxide.
XPS analysis
XPS measurements were conducted to elucidate the surface compositions. In Figure 5(a), the Fe2p peaks of both PRGO-Fe3O4 and PRGO-Fe3O4-Pb (PRGO-Fe3O4-Pb denotes Pb(II) adsorbed PRGO-Fe3O4) under 724.9 eV come from Fe3O4. The Pb4f peak of PRGO-Fe3O4-Pb under 143.7 eV comes from the Pb(II) adsorbed on the surface. Figure 5(b) shows the C1s spectrum of GO, in which the peaks at 288.4 eV, 286.9 eV, and 284.8 eV correspond to C=O, C–O, and C–C (Bian et al., 2015; Li et al., 2012), respectively. Figure 5(c) presents the C1s spectrum of GO-Fe3O4, in which appeared the same three peaks of C=O, C–O, and C–C as in GO. It is noteworthy that, for GO, the main peak of C1s is C–O, while for PRGO-Fe3O4, it is C–C. This is due to the detachment of O atoms chemically bonded to C atoms caused by the reduction of GO. Moreover, the quantitative analysis given by XPS presents, the molar percent of O attached to graphene in GO and PRGO-Fe3O4 are 32.04% and 27.21%, respectively, another proof of the partial reduction of GO, which is consistent with the results given by XRD and RS. The two peaks at 711.7 eV and 724.9 eV in Figure 5(d) correspond to Fe2p3/2 and Fe2p1/2, come from Fe3O4. The two peaks at 138.9 eV and 143.7 eV in Figure 5(e) correspond to Pb4f7/2 and Pb4f5/2, come from Pb(II) adsorbed on the surface.
X-ray photoelectron spectra. (a) Survey spectra of GO, PRGO-Fe3O4, and PRGO-Fe3O4-Pb; (b) C1s of GO; (C) C1s of PRGO-Fe3O4; (d) Fe2p of PRGO-Fe3O4; (e) Pb4f of PRGO-Fe3O4-Pb. GO: graphene oxide; PRGO: partially reduced graphene oxide.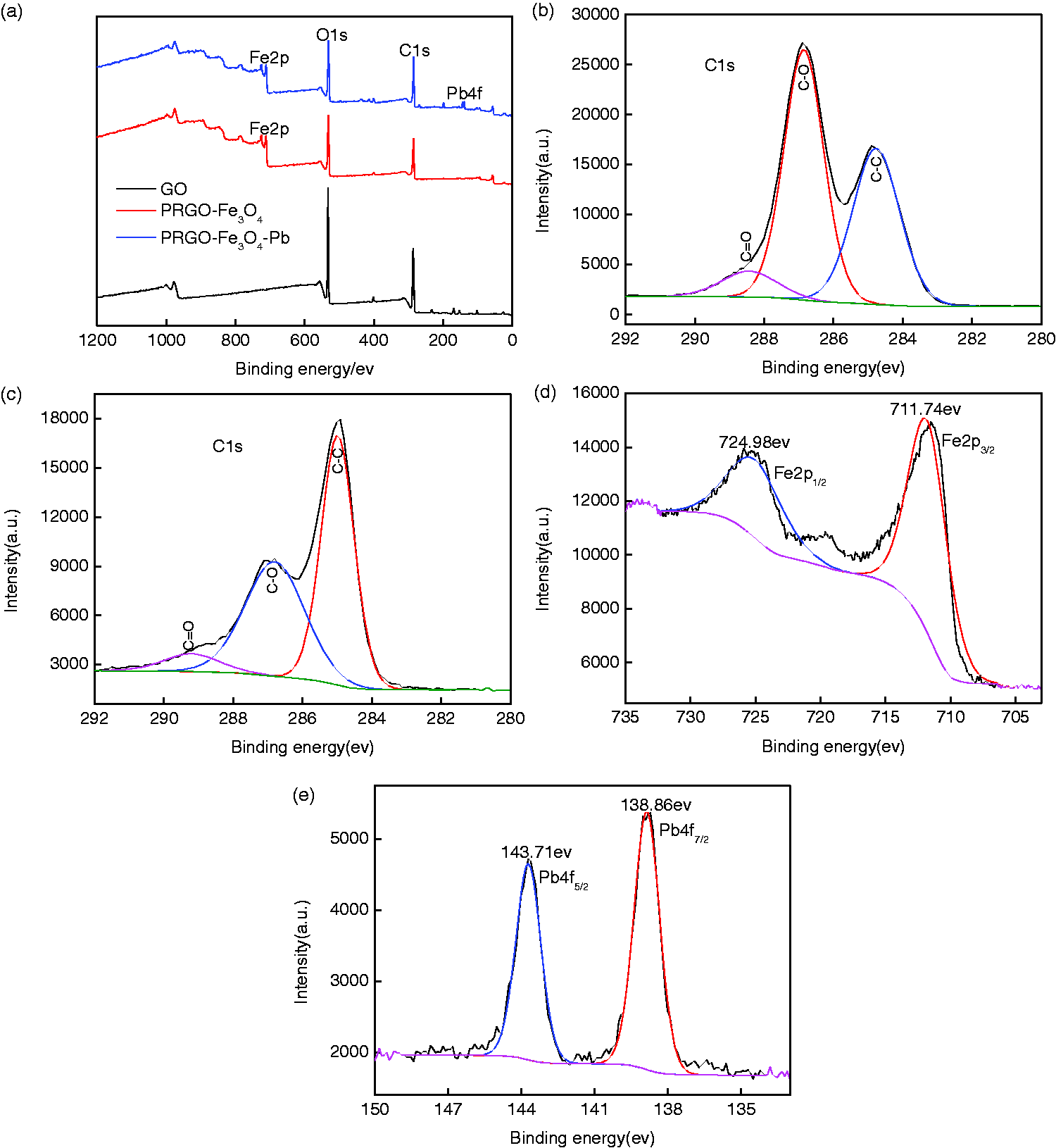
Surface area and pore distribution analyses
According to the N2 adsorption–desorption tests result and the calculation based on Kelvin equation (Dollimore and Heal, 1964), we got the pore size distribution form adsorption curve, as shown in Figure 6(b). Figure 6(a) shows the N2 adsorption curve is type IV characterized by its shape, indicating that pore size has an upper limit. Figure 6(b) shows the increasing rate of the quantity of adsorbed N2 slow down with the increase of the pore size, indicating the weakening of the capillary condensation due to the increase of the pore size.
(a) N2 adsorption–desorption curves; (b) pore size distribution form adsorption curve.
Thermogravimetric analysis (TGA)
The content of each component in the PRGO-Fe3O4 can be determined facilely with TGA. Figure 7 shows the TGA curves of GO and PRGO-Fe3O4, which were performed in argon atmosphere at a heating rate of 10℃/min. Clearly, there are three weight loss processes for both the two materials. The slight weight loss below 130℃ can be ascribed to the evaporation of adsorbed water molecules. With increasing temperature, a significant mass drop within the temperature range from 130℃ to 350℃ was observed in the TGA curve of GO, which can be attributed to the removal of labile oxygen functional groups. Further increase of the temperature leads to weight losses for both materials, which ascribed to the bulk pyrolysis of the carbon skeleton (Li et al., 2012; Peng et al., 2015; Raghubanshi et al., 2017; Yu et al., 2016). For PRGO-Fe3O4, an abrupt weight loss was observed when temperature exceeds 840℃, which is probably due to the decomposition of Fe3O4. According to the mass loss in PRGO-Fe3O4, we confirm that about 40 wt.% of Fe3O4 was deposited on the surface of graphene.
TG curves of GO and PRGO-Fe3O4. TG: thermogravimetry; GO: graphene oxide; PRGO: partially reduced graphene oxide.
Magnetic property and zeta potential
Figure 8 shows that the saturation magnetization, remanence, and coercivity are 22.11 emu g−1, 2.46 emu g−1, and 68.36 Oe, respectively. Due to the low remanence and coercivity, PRGO-Fe3O4 can be efficiently separated from water within only 10 s, as shown by the inset, which is desirable to practical application.
Magnetization curve of PRGO-Fe3O4 (inset at the upper left corner: magnification of hysteresis loop, at the lower right corner: a display of the magnetic separation of PRGO-Fe3O4 from water under an external magnetic field). PRGO: partially reduced graphene oxide.
The pHzpc determines the direction of the electrophoretic mobility where the net total particle charge comes to zero. Figure 9 shows pHpzc of PRGO-Fe3O4 is approximately 5, thus will be negatively charged when pH exceeds 5.
Dependence of zeta potential of PRGO-Fe3O4 hydrogel on pH. PRGO: partially reduced graphene oxide.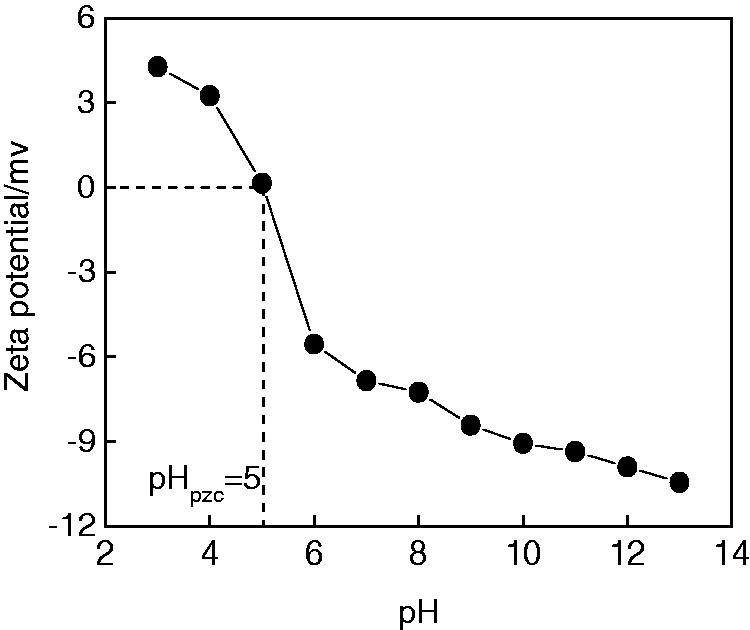
Adsorption properties
Effect of pH
To investigate the effect of pH on adsorption performance, adsorption experiments were carried out under different pH values ranging from 3 to 13, shown in Figure 10. When pH < 5, Pb2+ is the dominant species of Pb(II) (shown by the inset), and PRGO-Fe3O4 is positively charged (Figure 9), the electrostatic repulsion between the two hinders adsorption, resulting in a poor adsorption performance. Given that 5 < pH < 6, PRGO-Fe3O4 is negatively charged (Figure 9), then Pb2+ is the dominant species of Pb(II), the electrostatic attraction between the two facilitates adsorption, resulting in a dramatic enhancement in adsorption. Given that 6 < pH < 10, the main species of Pb(II) turns from Pb2+ to both Pb2+ and Pb(OH)+, making Pb(II) decreased in positive charge, weakening the electrostatic force between PRGO-Fe3O4 and Pb(II), resulting in adsorption decrease. Given that 10 < pH < 11 and pH > 11, the dominant species are Pb(OH)2 and Effect of pH on Pb(II) adsorption.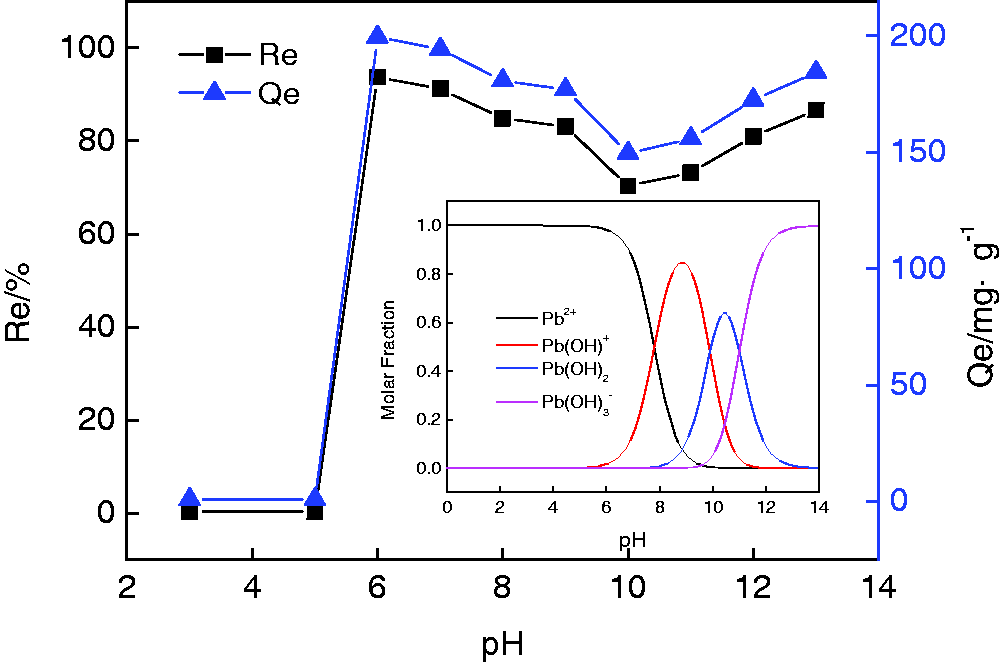
Effect of contact time
Figure 11 shows a fast adsorption process, with the adsorption equilibrium being reached in 10 min, manifesting an extraordinarily fast kinetics. This may be attributed to the thin graphene sheets in PRGO-Fe3O4 (as seen in Figure 3), which facilitates the intra-particle diffusion of Pb(II) due to short diffusion path.
Effect of contact time on Pb(II) adsorption.
Effect of adsorbent dosage
Figure 12 shows, with the increase of the adsorbent dosage, the adsorption percent increased, while the adsorption quantity decreased, respectively. The number of active sites available for adsorption increase as the adsorbent dosage increase, providing Pb(II) more probabilities to be adsorbed, consequently leading to the increase of the adsorption percent. However, the adsorption quantity decreases as the adsorbent dosage increase, because the initial amount of Pb(II) in the solution is fixed before the adsorption. The adsorption quantity and percent reached 777.28 mgċg−1 and 80%, respectively, under the dosage of 100 mgċL−1. Clearly, a high adsorption quantity is favored by low dosage, implying a strong adsorptive ability of PRGO-Fe3O4.
Effect of adsorbent dosage on Pb(II) adsorption.
Effect of foreign substances
In general, organic substances and electrolytic ions exist simultaneously in wastewater. In our work, humic acid and NaCl were added into the Pb(II) solution to simulate the wastewater environment. Figure 13 shows that adsorption percent and quantity decrease drastically as the concentration of humic acid increase. Humic acid adsorbs metal ions via chelating with its abundance of oxygen containing groups. Given that pH > 5 (adsorption is conducted under pH = 6), there are more free humic acid molecules in the solution (Li et al., 2012), forming stable complexes with Pb(II), remarkably worsening the adsorption performance. However, Figure 14 shows that the adsorption percent and quantity decrease only by a small extent due to the increase of NaCl concentration. The electrolytes impact the adsorption performance by changing the thicknesses of the electric double layers as well as the value of zeta potential. The electrolytic ions lie in a same plane with outer sphere complex during adsorption, thus outer sphere complex is more sensitive to the variation of ionic strength compared to that of inner sphere complex (Li et al., 2012). In the current work, only a minor influence on the adsorption performance by NaCl was observed, indicating that adsorption can be attributed to the formation of inner sphere complexes on the surface of PRGO-Fe3O4.
Effect of humic acid concentration on Pb(II) adsorption. Effect of NaCl concentration on Pb(II) adsorption.

Effect of temperature
Figure 15 shows, when the temperature increased from 20℃ to 35℃, the adsorption percent and quantity decreased from 95.93% and 201.13 mgċg−1 to 82.52% and 173.01 mgċg−1, respectively, implying that the adsorption of Pb(II) onto PRGO-Fe3O4 may be exothermic in nature. Consequently, a lower temperature facilitates adsorption.
Effect of temperature on Pb(II) adsorption.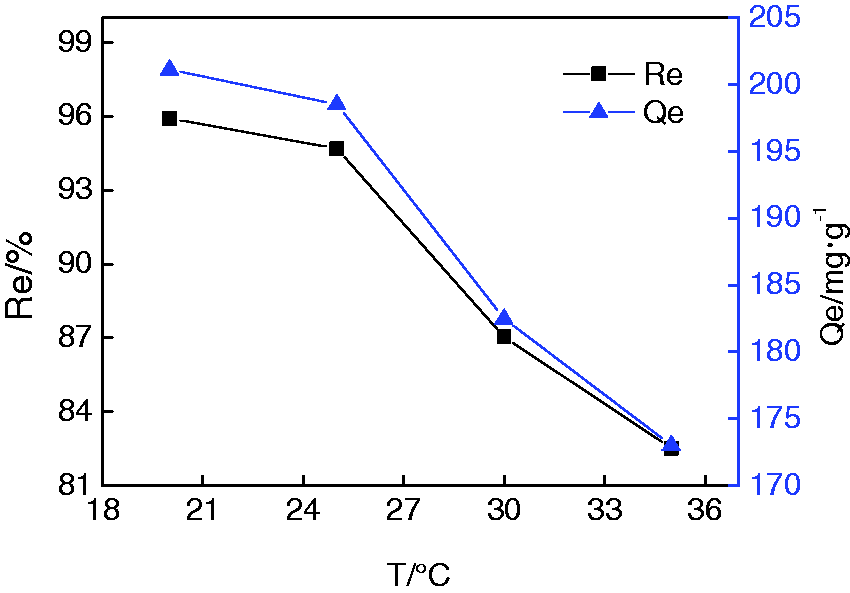
Adsorption mechanism
Adsorption models were used to explore the adsorption mechanism, thermodynamics, and kinetics, as shown by Table S1 and Figure S1, S2, and S3 in the Supporting Information. By comparing the R2 (linear correlation coefficient) values, the agreement of adsorption isotherm models fitted with Pb(II) adsorption in the below orders
The Temkin isotherm model assumes that the adsorption heat of all the molecules in the layer decreases linearly with coverage due to chemical interactions between adsorbent and adsorbate, and that the adsorption is characterized by a uniform distribution of the binding energies, up to a maximum binding energy (Guo et al., 2014). Based on the assumptions of the Temkin model, we can draw a conclusion that monolayer chemisorption is the main adsorption mechanism in our work. This may be attributed to the plenty of evenly distributed oxygen containing groups as the active sites for adsorption. Figure S2 shows, ΔH = −91.39 kJ mol−1, ΔG = −9.55 kJ mol−1, ΔS = −279.31 J K−1mol−1(293 K), confirming that the adsorption is exothermic, entropy decreasing, and spontaneous process. The absolute value of ΔH is higher than 20 kJ mol−1, implying a strong chemical interaction between adsorbent and adsorbate, a proof of chemisorption, which supports the conclusion drawn from the Temkin model. The absolute value of ΔG decreased with the rise of temperature, indicating a weaker adsorption under a higher temperature, as shown by the inset. During the adsorption process, the water molecules in the hydration sheath of the PRGO-Fe3O4 be transferred into the solution, leading to entropy increase, while Pb(II) be adsorbed onto PRGO-Fe3O4 from the solution, leading to entropy decrease. ΔS < 0 indicates that the degree of freedom decreases during the adsorption process. By comparing the R2 (linear correlation coefficient) values, the agreement of adsorption kinetic models fitted with Pb(II) adsorption in the below orders
The pseudo-first-order model gives the lowest linear correlation coefficient among all the four kinetic models, with the fit result of equilibrium adsorption quantity being 62.28 mgċg−1, which is far lower than the experimental result of 190.17 mgċg−1, indicating that pseudo-first-order model is not appropriate to describe the adsorption process. In contrast, pseudo-second-order model gives a linearly dependent coefficient of 0.9974 and a fit result of equilibrium adsorption quantity 190.84 mgċg−1, which is very close to that of the experimental result, meaning that pseudo-second-order model is appropriate for describing the adsorption kinetics, with chemisorption the rate controlling step, which is consistent with the conclusions drawn from Temkin model and the value of ΔH. The linearly dependent coefficient of the intra-particle diffusion model and liquid film diffusion model is 0.9885 and 0.9999, respectively. However, the extended fit line of the latter go through the original point, while the former not, as shown in Figure S3, implying that the adsorption process is controlled by both chemisorption and liquid film diffusion, whereas intra-particle diffusion is not a major rate controlling step. This may be ascribed to the ease of the Pb(II) diffusion inside PRGO-Fe3O4, which is facilitated by the thin graphene sheets in its structure. A conspicuous influence of temperature on the adsorption as shown by Figure 15 also supports the chemisorption nature.
Performance evaluation
Comparison of adsorption performance toward Pb(II) by PRGO-Fe3O4 with other adsorbents.

Conclusion
In this work, partially reduced graphene oxide-Fe3O4 (PRGO-Fe3O4) was successfully prepared, and characterized by XRD, HRTEM, XPS, FTIR, RS, N2 adsorption–desorption, VSM, and zeta potential analyses. PRGO-Fe3O4 exhibit excellent adsorption property toward aqueous Pb(II), as well as efficient separation from water: Adsorption equilibrium was reached in 10 min with the adsorption percent and quantity of 95.77% and 373.14 mgċg−1, respectively, under a condition of pH = 6, adsorbent dosage 250 mgċL−1, and Pb(II) initial concentration 97.68 mgċL−1, with the subsequent magnetic separation process taking only 10 s. As supported by Temkin isothermal model, the adsorption of Pb(II) onto PRGO-Fe3O4 is chemisorption, which produces inner sphere complexes. As confirmed by the thermodynamic and kinetic analyses, the adsorption is an exothermic, entropy decreasing, and spontaneous process, the rate of which is controlled by both chemisorption and liquid film diffusion. The facile synthetic route and good adsorption performance may endow PRGO-Fe3O4 with a promising application prospects.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



