Abstract
The inappropriate use of pain medication has led to the appearance of salicylic acid molecules (aspirin’s metabolite excretion) in surface water, which causes problems for the environment. The adsorption process using activated carbon is one of the processes that is used for the removal of organic compounds present in the aqueous phase; however, the percentage of removal depends on the physicochemical properties of the adsorbent and the adsorbate such as: pore size, surface area, surface chemistry, molecular size and solubility. In this work, we have studied the adsorbent–adsorbate interactions through the determination of the immersion enthalpy of the activated carbon in salicylic acid aqueous solutions; due to the solute and activated carbon surface have functional groups capable of ionized with the pH, the adsorption study was carried out at pH 2, 7, and 11. The activated carbons selected for the study were a granular activated carbon and a reduced activated carbon to 1173 K; as the immersion liquid were used salicylic acid aqueous solutions with concentrations between 0.072 mmol/ l and 0.72 mmol/ l, the solutions at pH 2 and 11 were prepared with HCl and NaOH solutions, respectively. It was determined that the immersion enthalpy is between −7.39 J /g and −22.5 J/g for the reduced activated carbon and between −7.63 J/g and −16.73 J/g for the granular activated carbon.
Introduction
Pain is defined as an unpleasant sensory and emotional experience associated with real or potential tissue damage. Burch (2011) indicates that one in five people suffer from chronic pain today; in the USA, the estimated number of patients who suffer from chronic pain is 116 million people, with the number growing constantly.
One of the strategies to treat pain is to use painkillers that are non-steroidal analgesics (NSAIDs), which have anti-inflammatory and antipyretic properties, and these painkillers are sold worldwide (Divins Triviño, 2001).
Although NSAIDs are 42 active principles, acetaminophen, ibuprofen, and salicylic acid are the drugs most commonly used by the population because they are over-the-counter medications; however, consumption and disposal inappropriate practices of expired or partially used medications generates the appearance of these drugs in wastewater, surface water and drinking water.
The contamination of water sources with pharmaceutical compounds has generated environmental problems associated with toxic effects of bioactive molecules (Saif Ur Rehman et al., 2015). The continuous exposure to traces of active principles not only affects animals; despite the fact that salicylic acid is synthesized by endogenous plants and plays an important role as a defense against pathogens, overexposure to this compound generates the synthesis of hydrogen peroxide, a free radical that alters the redox balance of plants when it is in high concentrations (Borsani et al., 2001).
Due to the environmental consequences associated with the presence of pharmaceutical compounds in water sources, it is necessary to remove these molecules in water treatment plants; however, techniques such as coagulation, flocculation, advanced oxidation are insufficient taking into account the physico-chemical properties of drugs (Martínez- Bueno et al., 2012).
Activated carbon is a porous solid with high surface area and chemical properties that allow to remove drugs with different characteristics. Jung et al. show that activated carbon adsorbs greater than 90% of drugs such as steroids, antidepressants, anti-inflammatory, lipid regulators and psychiatric control medications; however, the process efficiency will depend on the solid and the physico-chemical properties of the drug; some variables to be taken into account in the adsorbent are the surface area value, pore size and total acidity and basicity; in the case of drugs, the molecular size, solubility, and pKa are the properties that influence the adsorption process (Jung et al., 2015).
The adsorption process using activated carbon comprises two stages: the passage of molecules through the porous network until the molecules reach the pores, takes place the adsorption, and the interaction between the surface chemical groups present on the activated carbon and the functional groups present in the pharmaceutical compound, such interactions can be electrostatic or specific chemical, the interactions magnitude present between the adsorbate and adsorbent is a system characteristic and it allows describing from the energetic view point the adsorption process (Ocampo-Perez et al., 2011).
In this work, we have compared the salicylic acid (excretion metabolite of acetylsalicylic acid) adsorption on two activated carbons with different surface chemistries; it shows the functional groups present in the adsorbent and the importance of the adsorbate during the adsorption process. We varied the pH values in order to propose the possible adsorption mechanisms from the determination of adsorption isotherms, and the thermodynamic functions as immersion and interaction enthalpy, Gibbs energy, and entropy.
Methodology
Activated carbons
Two activated carbons are used as adsorbents: a commercial activated carbon, carbochem brand GS50, which is immersed in concentrated hydrochloric acid, washed with distilled water until the pH became constant and dried at 100℃; this sample is called as granular activated carbon (GAC).
GAC is subjected to heat treatment in a thermolyne furnace at 900℃ for 2 h with a heating ramp of 2℃/ min in nitrogen atmosphere; in this work, this sample will be called as the reduced activated carbon (GACr).
The physical properties of the activated carbons were determined by nitrogen adsorption at −196 ℃ in an apparatus called autosorb 3B (Quantachrome), from the isotherm obtained and the application of the BET model was calculated the apparent surface area, to determine the micropore volume was used Dubinin Radushkevish model; for this study, we did not take into account the pore size distribution, assuming that the adsorbate is a small molecule and has no diffusion problems throughout the activated carbon structure.
The chemical properties of the activated carbons were determined by Boehm’s (1989) titration that allows quantifying acidic and basic functional groups present on the surface of the activated carbon by reverse titration.
Five hundred milligrams of activated carbon GAC and GACr were weighed in hian analytical balance (Ohaus) with an accuracy of 10 mg; then these are suspended in 50 ml either of NaOH 0.1 M, Na2CO3, or NaHCO3, stored for 5 days at 20℃ with constant agitation. Subsequently, the potentiometric tritation of 10 mL of each one sample with HCl were made with hydrochloric acid. The equipment used for the qualification is a potentiometer, CG 840B Shott.
The above procedure is repeated using liquid hydrochloric acid as the immersion medium for quantifying the total basic groups on the activated carbon surface. After five days of storage, an aliquot of acid is titrated with sodium hydroxide.
Due to the presence of ionizable groups on the activated carbon surface, it is necessary to determine the electrical charge of the solid when there is a variation of the pH; for the activated carbons, the pHs were determined at the point of zero charge (pHpzc) through the mass titration which consists in weighed amounts of activated carbon between 4 and 4.5 g in containers with 10 ml of 0.1 M NaCl, are stored for 48 h with agitation and constant temperature, subsequently measured the pH value (Kodama and Sekiguchi, 2006).
Determination of the immersion enthalpy
The immersion enthalpy of the activated carbons GAC and GACr in aqueous solutions of salicylic acid between 0.072 mmol/ l and 0.72 mmol/l in neutral solution, acid and base, as well as in water, hydrochloric acid and sodium hydroxide to 20℃ was determined. The solutions were prepared with distilled and deionized water, and salicylic acid reagent grade Alfa with purity of 99%.
Hydrochloric acid and sodium hydroxide solutions were prepared from analytical grade reagents from Merck.
Immersion calorimetry took place in a heat conduction microcalorimeter with a cell in stainless steel with a capacity of 15 ml in which 10 ml of the immersion liquid was placed. Weigh 100 mg of each activated carbon in a glass ampoule with a peak fragile that is placed in the calorimetric cell, begins to capture the electric potential for approximately 60 min to get a stable baseline, the immersion of the sample, the increase of potential product of the wet of the solid, is expected until it comes back again to the base line and the electrical calibration.
The enthalpy of the immersion corresponds to the energy transferred when the activated carbon is in contact with a liquid immersion; this parameter is used to characterize the solid–liquid system and is related to the affinity of the solid by the immersion liquid. By applying the Hess law, it is possible to calculate the energy that is exchanged when the adsorbate interacts with the activated carbon surface previously knowing the enthalpy of immersion with the solvent, which provides information complementary to the thermodynamics of adsorption isotherms.
Determination of the adsorbed quantity of salicylic acid on the activated carbons
The salicylic acid-adsorbed molecules during the time of immersion calorimetry were determined by UV–vis spectroscopy using Genesys 0.1; the initial concentration was determined before the start of the immersion calorimetry, and once this assay determined the final concentration, filtering the solution and reading directly in a quartz cell with capacity of 3 ml, the amount adsorbed was determined on the basis of equation (1)
The temperature reading corresponds to 20℃, temperature during which the calorimetry assays were carried out.
For salicylic acid solutions at pH 2, the maximum wavelength (λmax) corresponds to 326 nm, 296 nm at pH 7 and pH 11 298 nm.
Discussion and results
Physicochemical properties of salicylic acid.

Figure 1 shows the species distribution diagram of salicylic acid; at pH values less than 2 were diprotic species in a percentage higher than 90%, from pH 4 to pH 13 the predominant species is monoprotic, and above these pH values the completely dissociated species are located close to 70%.
Salicylic acid species distribution diagram.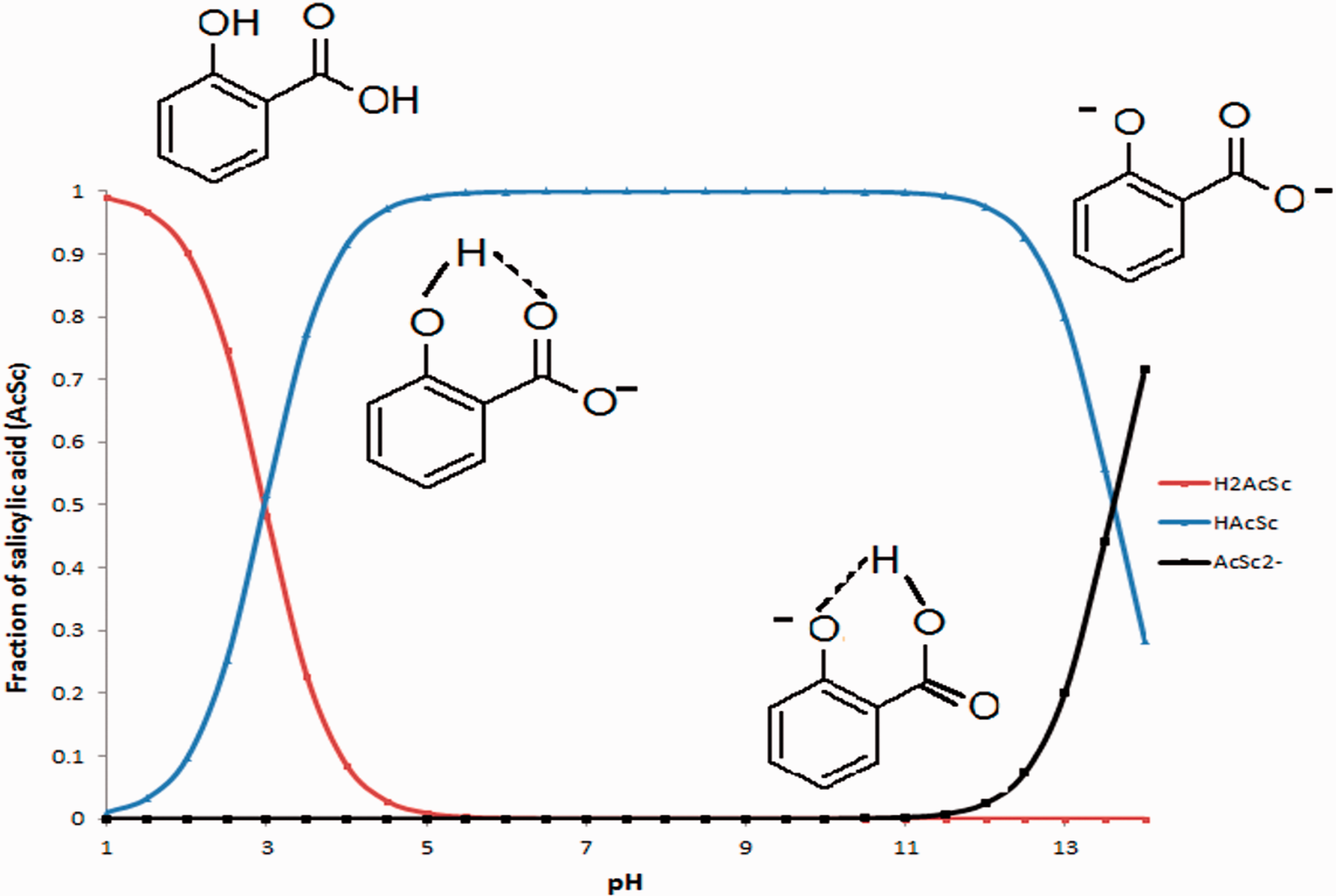
The slight solubility of this compound in water is associated with the formation of intramolecular hydrogen bonds that prevent the formation of interactions with water; this can influence the behavior of adsorption, taking into account the Lundelius rule, which relates to a lower value of solubility with an increase in adsorption (Sfouq Aleanizy et al., 2015).
Granular activated carbon (GAC) and reduced activated carbon (GACr) – Physicochemical properties.
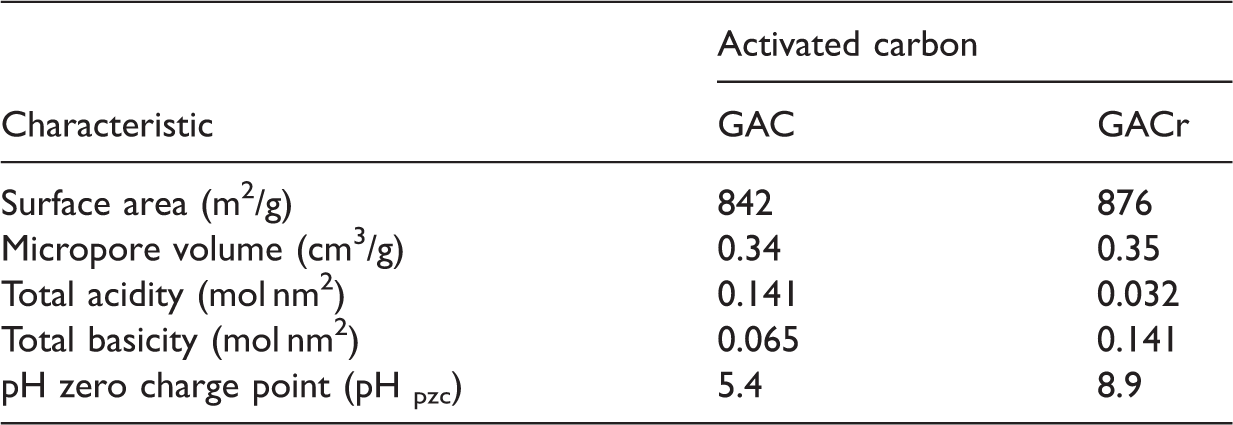
The variation in the surface functional groups present on the activated carbons creates changes in the acid–base properties including the pH at the point of zero charge. The determined value for GACr was 8.9 and for the GAC of 5.4, so at pH 2 both carbons have positive charge, at pH 7 GACr maintains the positive charge while GAC has a negative charge; at pH values greater than 9 the surface charge of solids is negative. In their work, Soylak et al. (2000) show how the treatment of activated carbon with acids and bases changes the properties of adsorption of metals such as gold, silver, and palladium in aqueous solution and how this solid is a sorbent suitable for concentrated solutions of transition metals and ensure its recovery.
Immersion enthalpies of the granular activated carbon in salicylic acid solutions at pH 2, 7, and 11 .

Immersion enthalpies of the reduced activated carbon in salicylic acid solutions at pH 2.7 and 11.
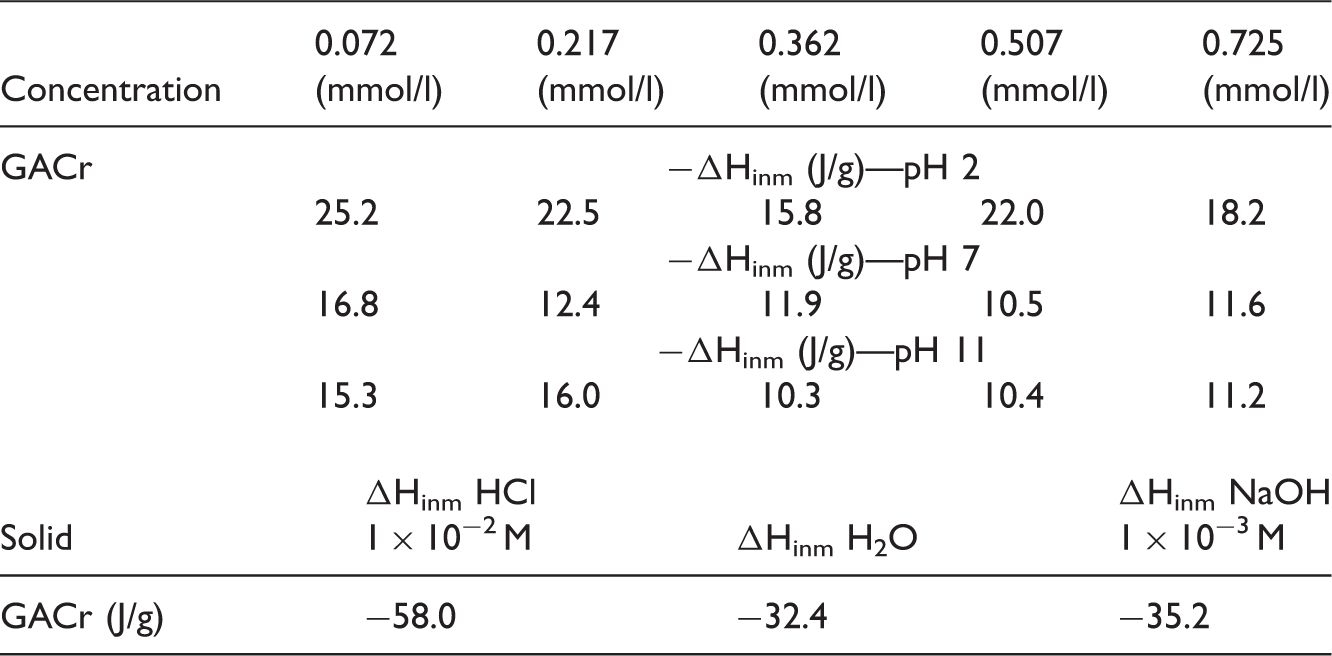
The immersion enthalpy allows to determine the transferred energy when the activated carbon is in contact with an immersion fluid (Rouquerol et al., 2008); in the case of solvents, GAC has a higher value of enthalpy in water and bases compared with GACr; this is due to the acid–base interactions and hydrogen bonds that occur between the acid groups present on the adsorbent surface and the hydroxyl groups present in the medium or the water molecules as appropriate. In an acid medium the highest value of enthalpy corresponds to the immersion of the sample GACr, which presents the basicity values higher than the GAC and therefore increases the acid-base interactions with the hydroniums presents in the medium.
The immersion enthalpies of the activated carbons in salicylic acid solutions ranged in concentrations between 0.072 mmol/ l and 0.72 mmol/l at pH 2, 7, and 11; the results show that a higher concentration is related to an increase in the immersion enthalpy value. This behavior can be attributed to the displacement of solvent of the carbonaceous surface after interaction with the salicylic acid.
The enthalpy values determined for the immersion of the activated carbon GACr in the salicylic acid solutions at pH 2 and 7 presented lower values to compared with GAC. This is due to the presence of basic functional groups on the surface of the adsorbent that interact with the salicylic acid and the hydronium ions; at pH 11, GAC also presents the greatest value of immersion enthalpy due to repulsions that occur between this activated carbon that has negatively charged product of acidic functional groups deprotonation (present in greater proportion than in the activated carbon GACr) and the hydroxyl groups of the solvent.
The interaction enthalpy is calculated by Hess law and allows obtaining the change in energy due to the interaction of adsorbent–adsorbate without taking into account the interactions of activated carbon with the solvent and despising other interactions as the solute–solvent assuming that given the low solubility of the adsorbate in water the affinity is lower.
Figure 2 shows the interaction enthalpy of the GAC and salicylic acid at pH 2, 7, and 11; it shows that for all pHs, the enthalpies are endothermic indicating that the interactions formed with the adsorbent surface require energy from the environment to be able to form. At pH 11, the energy requirement is greater because salicylic acid has a negative charge equal to the charge on the adsorbent surface. This behavior low to generates that a low concentrations the enthalpy increase due to the force of repulsion that occur in the system, however, subsequently the enthalpy remains constant indicating that generates few energy exchange which demonstrates low affinity between the adsorbate and adsorbent.
Enthalpies of interaction of granular activated carbon in salicylic acid solutions at pH 2, 7, and 11.
At pH 2, the enthalpy change due to the interaction of the salicylic acid with activated carbon CAG shows an increase proportional to the change of concentration of the adsorbate. This is due to the fact that the ions present in the medium as hydroniums ions interact with the surface groups present on the activated carbon reducing the capacity of the interaction with salicylic acid; at this pH, the adsorbate has charge-neutral for which the hydrogen bonds formation with activated carbon is possible; however, the increase of solute molecules is not enough to compete with the hydroniums ions that can protonate the adsorbent surface groups, thus preventing the formation of this type of interactions.
At pH 7 activated carbon GAC has a negative charge and salicylic acid also due to the carboxyl group deprotonation, at low concentrations the change in enthalpy increases due to the repulsion forces present in the system, however to increase the concentration of salicylic acid may be the formation of hydrogen bonds between the adsorbate’s phenol group and oxygenated groups on the activated carbon (GAC), however, it must be taken into account that due to the ortho position in which the substituents are located the formation of these interactions may be of an intramolecular character. Figure 2 shows the interaction enthalpies between 0.36 mmol/ l and 0.5 mmol/ l remain constant and at higher concentrations decrease, this behavior can be related with the saturation of the functional groups on the activated carbon after the adsorption process.
Figure 3 shows the salicylic acid adsorption on the GAC at pH 2, 7, and 11. After the immersion calorimetry, the data exhibit the same behavior of the interaction enthalpies; it is shown that at pH 7 the process is favored and therefore reached the highest adsorption capacity. Table 5 shows the adsorption capacities determined by the higher adjustment models that are Langmuir and Freundlich. At pH 11 the lower adsorption capacity is reached due to the low affinity between the adsorbate and the activated carbon; however, at low concentrations, other interactions different to ionic interactions become relevant as is the case of π interactions that allow the salicylic acid adsorption in an amount greater than those that are achieved at the same concentration at pH 2.
Adsorption isotherms of the granular activated carbon (GAC) in salicylic acid solutions at pH 2, 7, and 11. Parameters of the Langmuir and Freundlich models applied to adsorption isotherm for salicylic acid on granular activated carbon at pH 2, 7, and 11. The r2 of the fitted models were 0.85 in the three pHs.

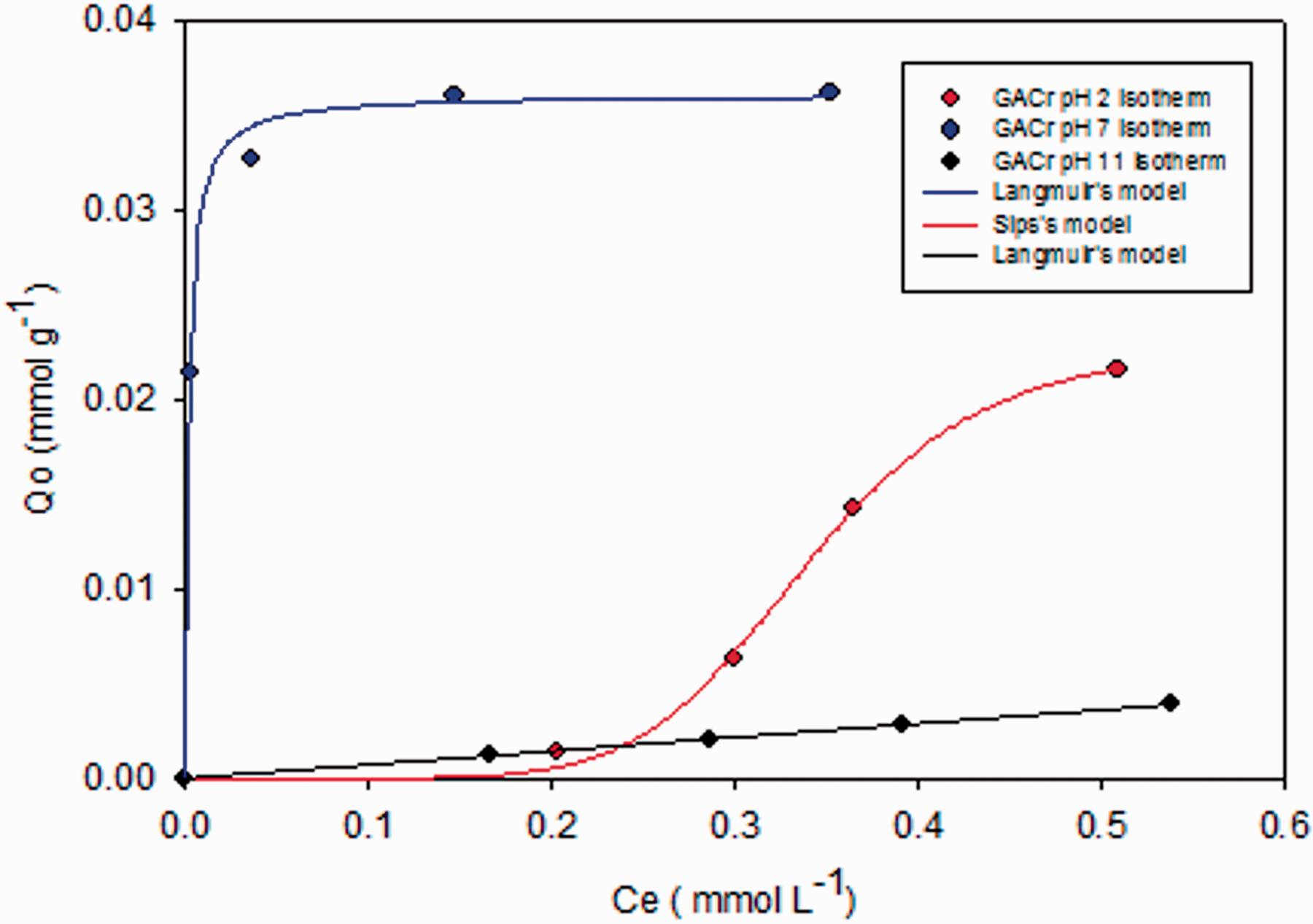
Parameters of the Langmuir and Sips models applied to adsorption isotherm for salicylic acid on reduced activated carbon at pH 2, 7, and 11.

The r2 of the fitted models were 0.90 in the three pHs.
Figure 4 shows the interaction enthalpies of reduced activated carbon, GACr, in the salicylic acid solutions at pH 2, 7, and 11. The results show the same tendency as the data obtained with the GAC; the positive values are related to the formation of new adsorbent–adsorbate interactions as well as the displacement of solvent molecules that are attached to the surface of the activated carbon. At pH 11, the change in enthalpy is constant due to the repulsion that occurs between the surface of the sorbent (with negative charge from pH 8.9) and salicylic acid which is deprotonated. The enthalpy values in this pH are above those determined to GAC. This is due to the thermal reduction process in which oxygenated groups such as carboxylic acids, aldehydes, ketones, and esters on the GAC surface are reduced to CO2, CO, and phenol (Chingombe et al., 2005); the latter in basic medium is deprotonated generating a negative charge on the surface. Figure 5 shows that the adsorption isotherm is determined after the immersion calorimetry; the model of greater adjustment corresponds to Langmuir model, which indicates that the energy in the adsorption sites on activated carbon GACr is constant and may be related to the high polarity on the surface due to phenol groups, it can be observed that the effects repulsive generate a strong effect on the adsorption process since the monolayer capacity decreases respect to the process carried out at pH 2 and 7.
Interaction enthalpies of reduced activated carbon in salicylic acid solutions at pH 2, 7, and 11. Adsorption isotherms of reduced activated carbon (CAGr) in salicylic acid solutions at pH 2, 7, and 11.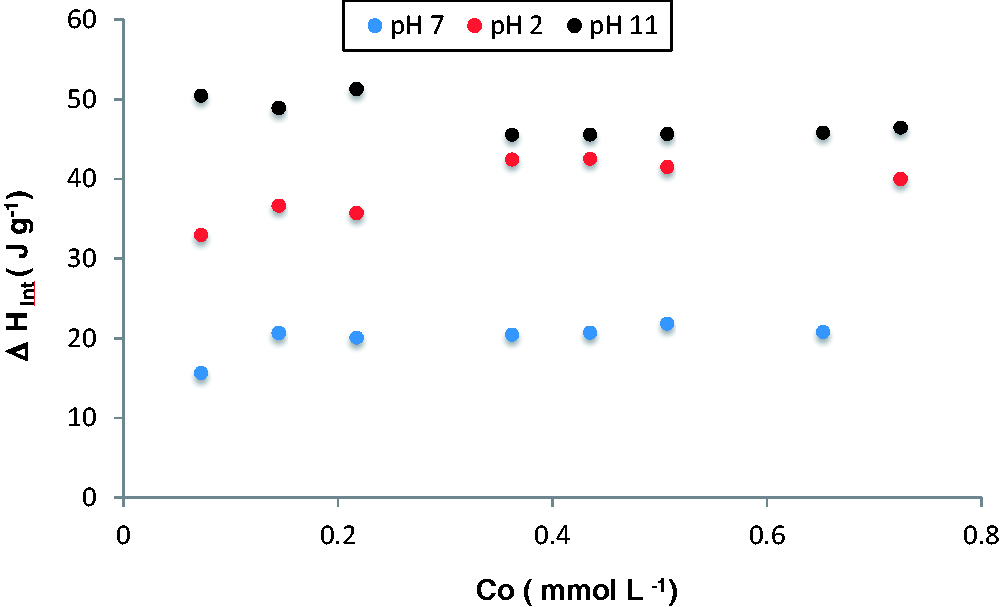
Comparing the interaction enthalpies between the two activated carbons of this study, it can be seen that the formation of a greater number of adsorbent–adsorbate interactions during the adsorption process carried out in the activated carbon reduced GACr to pH 7. This behavior is related to the formation of acid–base interactions between the basic groups present in the activated carbon and the carboxylic acid present in the molecules of adsorbate. In addition, during the thermal reduction process the carboxylic acids present in the GAC are transformed in Phenol groups, which can form hydrogen bond interactions with functional groups present in the salicylic acid.
The presence of weak acids and bases on the activated carbons surface and in the adsorbate generates that the adsorption process depends on the pH. Due to the acidic nature of the adsorbate at high pH values the molecule is negatively charged generating repulsion forces with the surface of the adsorbent. This effect increases in activated carbons with high amount of acidic oxygenated groups given that negatively ionized are also increasing the field of repulsion.
At acid pH values the granular and reduced activated carbon adsorb H+ ions, so that the values of enthalpy change are similar, however, the adsorption of salicylic acid is favorable at low concentrations in the GAC due to the formation of specific interactions with the adsorbate, in the case of reduced activated carbon the basic groups on the surface increasing the adsorbent–solvent interactions.
At pH 2, the enthalpy change increases with the adsorbate concentration and above concentrations of 0.36 mmol/ l the enthalpy remains constant. This indicates that at low concentrations salicylic acid molecules require more energy to move the H+ ions attached to the surface; part of the energy that is needed to carry out the process is required by the decrease in the aromatic ring reactivity of the salicylic acid because it is replaced with a carboxylic acid that attracts the electrons of the resonance ring and prevents the stabilization of the carbocation intermediate in the aromatic substitution reactions. This effect is compensated at high concentrations according to Le Chatelier’s principle; there is a displacement of the solvent molecules attached on the surface and adhered to the adsorbate.
Figure 5 shows the adsorption isotherm at pH 2, which was adjusted to the Sips model, which combines the models described by Langmuir and Freundlich and allows to predict the adsorption on heterogeneous systems in concentration ranges higher than that was possible with the Freundlich model; at low concentrations the system is heterogeneous because the H+ ions compete with salicylic acid, however to increase the concentration of adsorbate generate the displacement of the solvent on the activated carbon surface and the system becomes more homogeneous until saturation of the functional groups capable of interacting with the adsorbate and therefore is presented the monolayer’s formation as described by the Langmuir model.
In addition to the adsorption process on GAC, the salicylic acid adsorption on GACr is favored at pH 7; the enthalpy values are positive for the reasons outlined above; however, the energy requirement is lower due to ionic interactions between the activated carbon surface which has positive charge and the adsorbate with negative charge. Given that the ionic interactions are stronger than the hydrogen bond interactions it reaches a greater adsorptive capacity compared with activated carbon GAC. Figure 5 shows that the adsorption isotherm was adjusted to the Langmuir model that presented the highest adjustment, but this model is used to describe processes that are carried out in homogeneous surfaces, a feature that does not have the activated carbon. To the adjustment to the adsorption models is related to the system conditions, as at pH 7 there are mainly electrostatic interactions between the adsorbent and the adsorbent it can be assumed that the surface charge is homogeneous and therefore the adsorbate only interacts with a group in the adsorbent possessing the opposite charge and it is assumed that interactions between the adjacent groups are negligible because the adsorbate has a negative charge that prevents interaction between equal molecules.
The Gibbs energy change of the adsorption process is associated with the spontaneity of the process as well as to changes in the chemical potential of the system when there are variations in conditions such as concentration and the pH (Borisover, 2016), Figure 6 shows the ΔG for the adsorption of salicylic acid on GAC; negative values indicate that the process is spontaneous in the pH range and concentration that were covered this work; however, there are differences in the Gibbs energy change with the change in the pH; the increase in the Gibbs energy proportional to the increase in the concentration of adsorbate in the solution to pH 11 is attributed to the forces of repulsion between the adsorbate and adsorbent that possess the same charge. Due to an increase in salicylic acid molecules with negative charge is greater repulsion and increase of the energy needed to carry out the adsorption of salicylic acid. Gundogdu et al. (2012) in their work of adsorption of phenol on a low-cost activated carbon produced from tea industry waste carried out a study on the thermodynamics, kinetics, and equilibrium of the process and arrived at the conclusion that the pH that favors the process is 7, as shown for the study of the adsorption of salicylic acid in this work.
Gibbs energy change for the adsorption process of salicylic acid on granular activated carbon at acidic, neutral, and basic pH.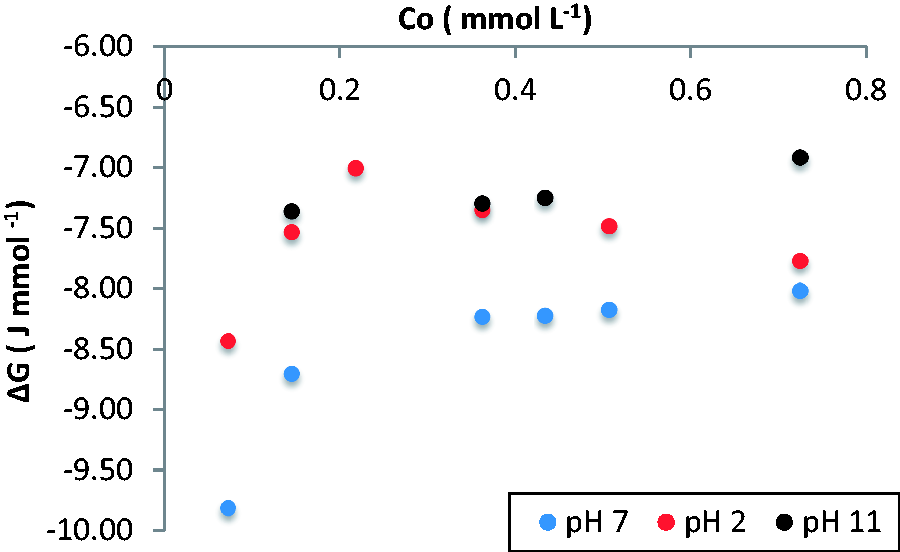
The Gibbs energy change is associated with the adsorption equilibrium constant as shown in equation (2) (Giraldo and Moreno-Piraján, 2008)

The term
The chemical potential is equal to the molar Gibbs energy as shown in equation (3)

At pH 2, it is shown that the Gibbs energy increases values above 0.36 mmol/ l and in higher concentrations decreases, as shown in Figure 3 during the adsorption of salicylic acid in the activated carbon GAC at low concentrations the adsorption capacity is low due to competition that occurs between the adsorbate and the H+ ions for the adsorption sites on the activated carbon surface, this generates that the equilibrium constant have low values and therefore the change in the Gibbs energy is greater, to the extent that more salicylic acid molecules are adsorbed and displace H+ ions from the adsorbent surface the values of the equilibrium constant increase and therefore the Gibbs energy decrease.
At pH 7 the Gibbs energy increase at low concentrations and increases from 0.36 mmol/ l remains constant, Figure 3 shows that the Gibbs energy change presents a similar behavior to the adsorption process at low concentrations, the Gibbs energy change increases due to the force of repulsion adsorbate–adsorbent that decrease the equilibrium constant; however, the formation of specific interactions between the phenol’s hydrogen from the adsorbate and oxygenated groups on the adsorbent increases the adsorption capacity until the adsorbent active sites is saturated whereby the adsorbent quantity remains constant as does the Gibbs energy.
In Figure 7, it is observed that at pH 11 the Gibbs energy change for the salicylic acid adsorption on reduced activated carbon, GACr, remains constant due to the low adsorption capacity obtained during the process associated with the low adsorbate–adsorbent interactions.
Change in the Gibbs energy for the process of adsorption of salicylic acid in activated carbon reduced to pH acidic, neutral, and basic.
A decrease in the Gibbs energy change to increase the concentration is observed at pH 2. This is related to the low adsorption capacity of the salicylic acid at low concentrations due to adsorption competitive the hydronium ions that are found in greater proportion in the medium; as well as the presence of these ions and the deactivation of the aromatic ring generates the destabilization of the carbocation intermediate reaction which implies that at low concentrations required more energy to carried out the process as shown in Figure 8; above concentrations 0.5 mmol/ l takes less energy due to a change in the balance due to the increase of adsorbate molecules that favors interactions adsorbate–adsorbent and solvent–adsorbent.
Formation of the carbocation intermediate in the salicylic acid adsorption on activated carbons at pH 2.
The lowest value of Gibbs energy change was reached at pH 7 due to the environmental conditions favoring the adsorption process of the salicylic acid in the reduced activated carbon GACr. Figure 5 shows the formation of a monolayer from concentrations of 0.1 mmol/ l. This indicates that once they have saturated the active sites, the adsorption process ends; however, adsorbate molecules with negative charge in the medium generating repulsion forces that increase the Gibbs energy change proportional to the concentration.
Aburub et al. indicate that the adsorption process of drugs on activated carbon is directed by enthalpic or entropic ways; in this work, we have evaluated the importance of the adsorbent–adsorbate interactions during the process of adsorption; this factor that is related to the enthalpy of the process shows that specific interactions such as ionic or hydrogen bonding predominate in carbons that possess chemical surfaces with abundant oxygenated functional groups; however, when the characteristics of the adsorbate or adsorbent prevent this type of interactions it becomes relevant that the nonspecific interactions are related to the entropic processes in the adsorption. These interactions involve mainly interactions of π electrons that are along the carbonaceous adsorbent (Aburub and Wuster, 2006).
Figures 9 and 10 show the entropy change for the adsorption process of salicylic acid on the activated carbons GAC and GACr at pHs 2, 7, and 11 The values of entropy change are greater in the GAC despite having a greater number of acid groups on its surface. This behavior related to the conditions that disfavor the process how is the change in pH; which has relevance due to the presence of weak acids in the adsorbate and adsorbent.
Entropy change for the adsorption process of salicylic acid on granular activated carbon at acidic, neutral, and basic pH. Entropy change for the adsorption process of salicylic acid on reduced activated carbon at acidic, neutral, and basic pH.
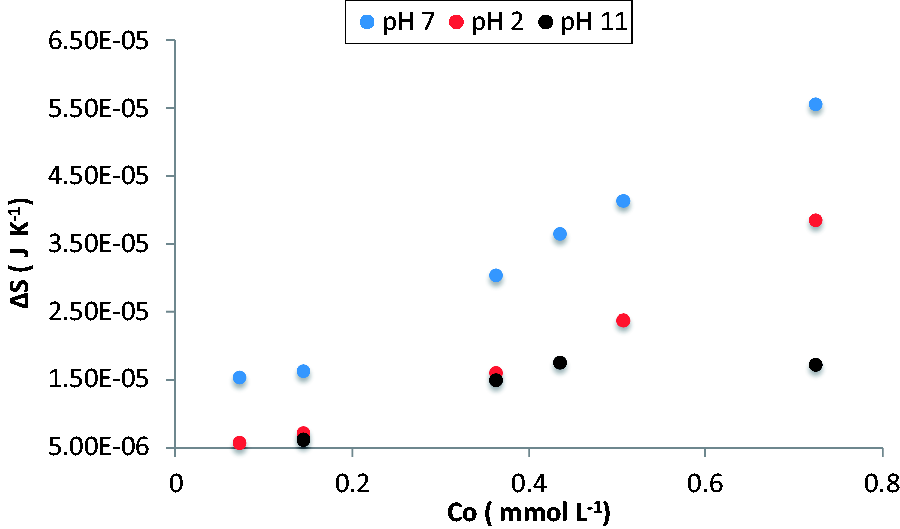
For the adsorption process carried out at pH 11 in the activated carbons GAC and GACr it is evident that the entropy change increases due to the fact that the only possible interactions to form are non-polar interactions between the aromatic regions in the adsorbent and the adsorbate due to the force of repulsion that is predominate in the system.
At pH 2, the entropy change is proportional to the change in the concentration, although Figures 2 and 4 show that the enthalpic factor is relevant in the adsorption process at this pH. The presence of H+ ions in the medium creates competition for the active groups, to low concentrations of adsorbate decrease the adsorption capacities due to the fact that the surface chemistry of activated carbons is to retain molecules of solvent, however the hydrogen bonds formation between the phenol and salicylic acid surface groups available to interact, as the adsorbate concentration increases, the affinity between the activated carbon surface and salicylic acid increases, which decreases the adsorbate–solvent and adsorbent–solvent interactions, this generates the apolar regions desolvation on the activated carbon and in salicylic acid increasing the π–π interactions and in this way the entropy change.
At pH 7 the entropy change differs in the two activated carbons, in the GAC, the repulsion forces are present due to the adsorbate and adsorbent have the same charge, the repulsion between the salicylic acid molecules generates that is organized in such a way that the charges do not stay in contact direct, this favors the formation of interactions between the phenol and surface groups that favor the adsorption and allow the formation of the monolayer until the saturation of the groups that may interact, this organization of the system increase the entropy change.
In the case of activated carbon GACr is observed an increase in entropy that indicates that not only the ionic interactions are relevant during the process of adsorption, parallel processes to the adsorbent–adsorbate interactions can increase the entropy change, similar to the process carried out at pH 2 water molecules is required to be displaced from the surface to be able to affect adsorbent–adsorbate interactions, this water displacement not only affects the surface but also to the adsorbate that are required to be desolvated leaving the molecule to interact, it must be taken into account that due to the charges present in the on the activated carbon and salicylic acid the polarity increases and therefore more water molecules will be attached which implies a greater entropy change to the extent that more adsorbate molecules are in the system and generate the displacement of the same.
Conclusions
The extreme pH conditions disfavor the adsorption process due to the surface chemistry on the activated carbons and the physicochemical properties of salicylic acid . At pH 2 is presented in both activated carbons positive surface charge due to the functional groups protonation thus preventing the formation of interactions as hydrogen bond with the adsorbate, as well as effects of resonance in the salicylic acid prevent the stabilization of the carbocation intermediate reaction by increasing the energy required to carried out the process, however interactions by π electrons can be generated in the highest concentration of adsorbate, at pH 11 the conditions are unfavorable due to presented repulsions between salicylic acid and the activated carbons, the energy required is greater for the activated carbon (GAC), because it has a greater amount of negative charges due to contain a greater amount of acid functional groups.
The immersion and interaction enthalpies, the Gibbs energy change and the entropy change indicate that the process is favored in the reduced activated carbon, GACr, in this activated carbon are reached the maximum adsorption, capacity 0.036 mmol/ l; the values of the thermodynamic functions ΔHint 20.76 J /g, ΔG −9.67 J/ mmol and a ΔS 5 × 10−5 J /K shown that at pH 7 the process requires energy to carried out the adsorbent–adsorbate interactions, however, is spontaneous and it reaches the highest entropy change due to the system order that is associated with the reduction of the freedom degrees in the salicylic acid which is found in solution and then retained on the activated carbon surface.
Footnotes
Acknowledgements
The authors thank the Framework Agreement between the Andes University and the National University of Colombia and the act of agreement established between the Chemistry Departments of the two universities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

