Abstract
In this article, a full factorial experimental design 24 was used to evaluate four factors (contact time, temperature, initial concentration of anthocyanins, and adsorbent mass) and their interactive effects on adsorption of anthocyanins from elderberry and chokeberry extracts on bentonite. The factors were investigated at two levels (−1 and +1). The regression models considering the main effects and interactions were suggested. The variance analysis and the statistic data showed adequate models. The contact time has the most positive effect and the initial concentration of anthocyanins has the most negative effect that influences the removal of anthocyanins from berry extracts on bentonite.
Introduction
During the last years, an increasing interest to anthocyanins, as natural colorants in food industry (Delgado-Vargas and Paredes-Lopez, 2003; Francis and Markakis, 1989) and anti-oxidants in medicine (Castaneda-Ovando et al., 2009; Konczak and Zhang, 2004) is observed. These compounds have positive effects on different diseases, including cardiovascular, ophthalmic, inflammatory, and neurological ones, as well as cancer and diabetes (Ghosh and Konishi, 2007).
Anthocyanins are found in many vegetables, fruits, and berries (Castaneda-Ovando et al., 2009; Delgado-Vargas and Paredes-Lopez, 2003). It should be noted that dark red or dark blue berries have the highest content of anthocyanins and may become cheap sources of anthocyanins on a large scale (Denev et al., 2010). Compared with the other methods, on the one hand, adsorption is effective for removal and purification of anthocyanins from extracts. On the other hand, it is also relatively simple in design and operation (Zhao et al., 2015).
Macroporous resins (Beye et al., 2013; Buran et al., 2014; Chen et al., 2016; Zhao et al., 2015) and reversed-phase adsorbents (Chulkov et al., 2012) have successfully been used for the removal of anthocyanins. These synthetic adsorbents are characterized by a high surface area, big pore size, good physical and thermal stability, however they are expensive.
Natural clay minerals are cheap adsorbents and can be an alternative for the synthetic adsorbents (Lopes et al., 2011; Yoshiumi et al., 2009). Unfortunately, there is no information in the literature about the studies which investigated changes of important adsorption factors during removal of anthocyanins on clay minerals. Meanwhile influence of the factors and interactions among these factors are very important in case of industrial production of anthocyanins by means of adsorption. Conventional methods of studying the effect of different adsorption factors require numerous experiments to determine the optimal levels (Baral et al., 2009; Ponnusami et al., 2007).
In this study, the full 24 factorial design was performed to evaluate the importance of contact time, temperature, initial anthocyanin concentration, and adsorbent mass on removal efficiency of anthocyanins from elderberry and chokeberry extracts on bentonite as well as to suggest regression models for prediction of adsorption removal of anthocyanins.
Materials and methods
Plant materials and chemicals
Samples of ripe elderberries and chokeberries were harvested in Vinnitsa region (Ukraine) in 2015. The fresh berries were immediately frozen and kept in a freezer at −20℃ before being used in the experiments.
Chemical reagents of analytical grade (hydrochloric acid, glacial acetic acid, sodium acetate (trihydrate), potassium chloride) were purchased from Cherkassy State Chemical Plant (Ukraine).
Preparation of berry extracts
The thawed berries were smashed using a blender. The berries were macerated with 0.1 M HCl (1:2 = m:V) for 24 h in the darkness at 20℃. Then the berry extracts were separated from the berries by filtration through a filter paper and stored in a fridge at 4℃.
Adsorbent preparation
The adsorbent was bentonite from Dashukovsky deposit in Ukraine (TU U14.2-00223941-006:2010) that refers to alkaline earth silicate sheet and has the following chemical composition (%): SiO2, 49.6; Al2O3, 13.5; Fe2O3, 7.2 (Rakitskaya et al., 2012).
Before adsorption studies bentonite was pounded in a porcelain mortar, washed with distilled water at 20℃ to remove impurities, and then dried at 95℃ for 24 h to remove moisture.
Analysis of anthocyanins
Total monomeric anthocyanin concentration was determined by pH-differential method (Lee et al., 2005), using two buffer systems: potassium chloride buffer (pH 1.0 (0.025 M)) and sodium acetate buffer (pH 4.5 (0.4 M)). The absorbance of each extract was measured at 515 and 700 nm using UV-VIS spectrophotometer (SF-56, Spectral, S.-Petersburg, Russian).
Total anthocyanins (mg/L) were calculated as cyanidin-3-glucoside according to the following equation:
Adsorption studies
The adsorption of anthocyanins from berry extracts on bentonite was performed by batch experiments. In the batch adsorption studies, anthocyanin extracts (pH = 1.5) with bentonite were shaken at 150 rpm/min. Mixtures of anthocyanin extracts with bentonite were filtered off after the adsorption and concentrations of anthocyanins were determined by pH-differential method.
The experimental anthocyanin removal efficiency was defined using the equation
Experimental design and statistical treatments
Experimental design method is an important tool in engineering science for improving the performance of the process (Baral et al., 2009). In addition, a factorial experiment is the only source making possible to assess quantitatively the impact and interaction of different factors. In this study, contact time (min), temperature (℃), initial concentration of anthocyanins (mg/L), and adsorbent mass (g/L) were chosen as independent variables. The anthocyanin removal efficiency was chosen as a dependent response variable. The low and high levels for the factors were selected according to our preliminary experiments. The order in which the experiments were made was randomized to avoid systematic errors.
Selected values of the variables investigated in the factorial experiment.
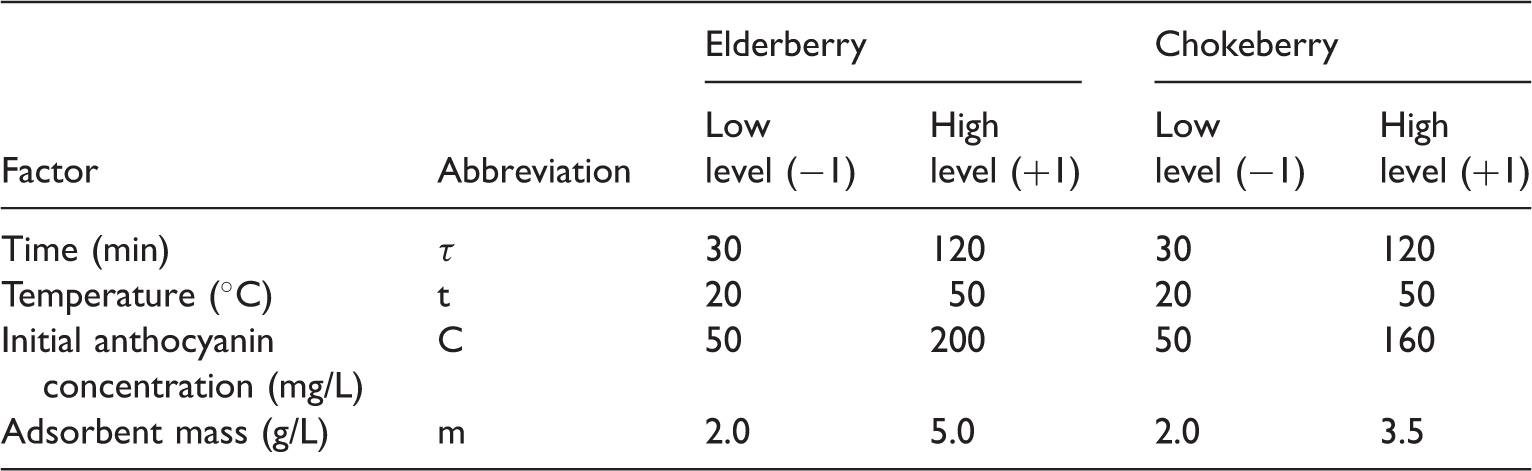
The total number of 16 experiments was performed and each experiment was repeated thrice. Experiments were chosen randomly to prevent partiality of researchers. The results of the experimental design were analyzed using MINITAB 16 statistical software to evaluate the effects as well as the statistical parameters, the statistical plots (Pareto, main effects, and interaction plots). The interactions between independent factors were determined with the analysis of variance (ANOVA) and the main effects of anthocyanin adsorption were identified based on the P-value with >95% of confidence level.
Results and discussion
Full factorial experiments can help to provide a mathematical model and to predict results, as well as to evaluate influence of the main effects and their interactions on process. The main steps of the factorial experimental design technique are planning and performing of the statistically worked out experiments, examination of the main factors and their interactions, estimation of the coefficients in a mathematical model, checking the adequacy of the model.
Removal efficiency of anthocyanins from berry extracts on bentonite.

Statistical parameters for 24 designs.

ANOVA
Analysis of variance.

DF: degrees of freedom; SS: sum of squares; MS: mean square.
Current literature includes adsorption studies using full factorial design where three- and four-way interactions were significant (Baral et al., 2009; Çoruh and Gürkan, 2014; Moura et al., 2004). It should be understood that experimental designs are not a priori tool for basic research, because they never allow explanation of the studied physical-chemical phenomenon (Khammour et al., 2016). However, the contribution of all significant interactions is extremely important when regression models are used for the prediction of the process efficiency.
Pareto chart
As it is shown in Figure 1, the vertical line with 2.04 value in the Pareto chart indicates the minimum statistically significant effect magnitude. It was calculated for the 95% confidence level and 32 freedom degrees. The values which exceed a reference line are significant ones. According to Figure 1, the main factors (C, τ, t, and m) and their two-way interactions τ × m, C × m only for elderberry extracts, two-way interaction τ × t, three-way interaction τ × C × m, and four-way interaction τ × t × C × m for both berry extracts extend the limits of the reference line and are significant at the level of 0.05. All other their interactions have smaller effects and are statistically insignificant.
Pareto charts of standardized effects on the removal efficiency of anthocyanins from extracts: (a) elderberry and (b) chokeberry.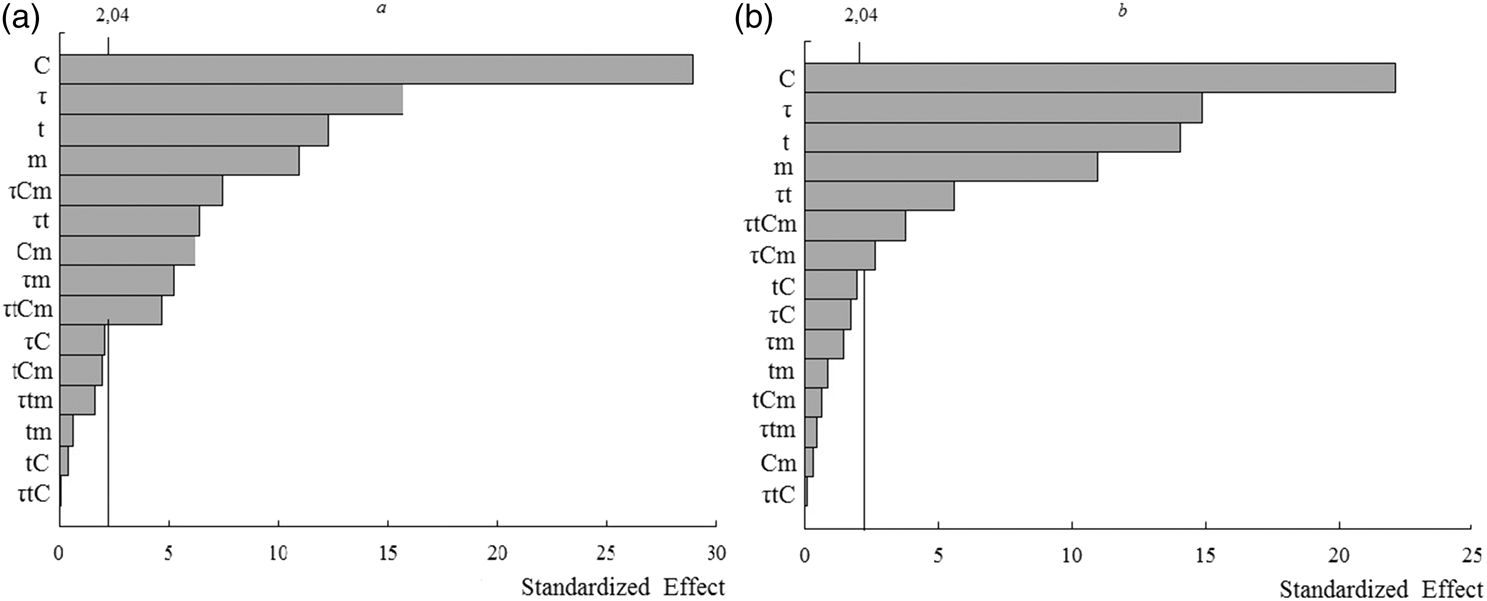
Main effects
Main effects of each factor on removal efficiency of anthocyanins from berry extracts are shown in Figure 2. It can be seen that contact time has a strong positive effect upon responses. The adsorbent mass has less positive influence upon removal efficiency of anthocyanins than contact time. The contact time and the adsorbent mass increase make larger the anthocyanin removal efficiency. The initial anthocyanin concentration has a very strong negative effect upon responses. The temperature has a less negative influence upon removal efficiency than the initial anthocyanin concentration. The increase of the initial anthocyanin concentration and temperature decreases the anthocyanin removal efficiency. Maximum removal efficiency of anthocyanins from elderberry and chokeberry extracts takes place at low initial concentration and temperature, high contact time, and adsorbent mass.
The plot of the main effects for the removal efficiency of anthocyanins from elderberry (a) and chokeberry (b) extracts on bentonite.
Unfortunately, little information was published about the main effects of control factors on removal efficiency of anthocyanins from berry extracts. According to Zheng et al. (2015), adsorption time had the strongest effect on adsorption of blueberry anthocyanins on AB-8 macroporous adsorbent, followed by pH value, and adsorption temperature was the least.
Interaction effects
It is necessary to take into account the fact that the effects of each factor depend on the levels of the other factors. The two-way interactions among different control factors for the removal efficiency of anthocyanins are shown in Figure 3. The interaction between factors is effective when the change in the response from low to high levels of a factor is dependent on the level of the second factor, viz. when the lines of the interaction effects do not run parallel (Mathialagan and Viraraghavan, 2005).
Interaction effects for the removal efficiency of anthocyanins from elderberry (a) and chokeberry (b) extracts on bentonite.
As it is shown in Figure 3, the lines of interaction effects such as adsorbent mass and contact time, adsorbent mass and temperature, adsorbent mass and initial anthocyanin concentration in case of anthocyanin removal from elderberry extracts are far from being parallel and the interaction between these factors is effective. That is, the high level effect of the adsorbent mass on the removal of anthocyanins from elderberry extracts will be higher if contact time is maintained at a high level, initial anthocyanin concentration and temperature are kept at a low level.
As for the anthocyanin removal from chokeberry extracts regularities are similar to the ones listed above but all lines of interaction effects are parallel and the interaction factors are less effective.
Modeling of anthocyanin removal on bentonite
Based on the findings given above, the quantitative models for anthocyanin removal can be represented by the following first-order polynomial equations:


Analysis of variance for removal efficiency of anthocyanins.
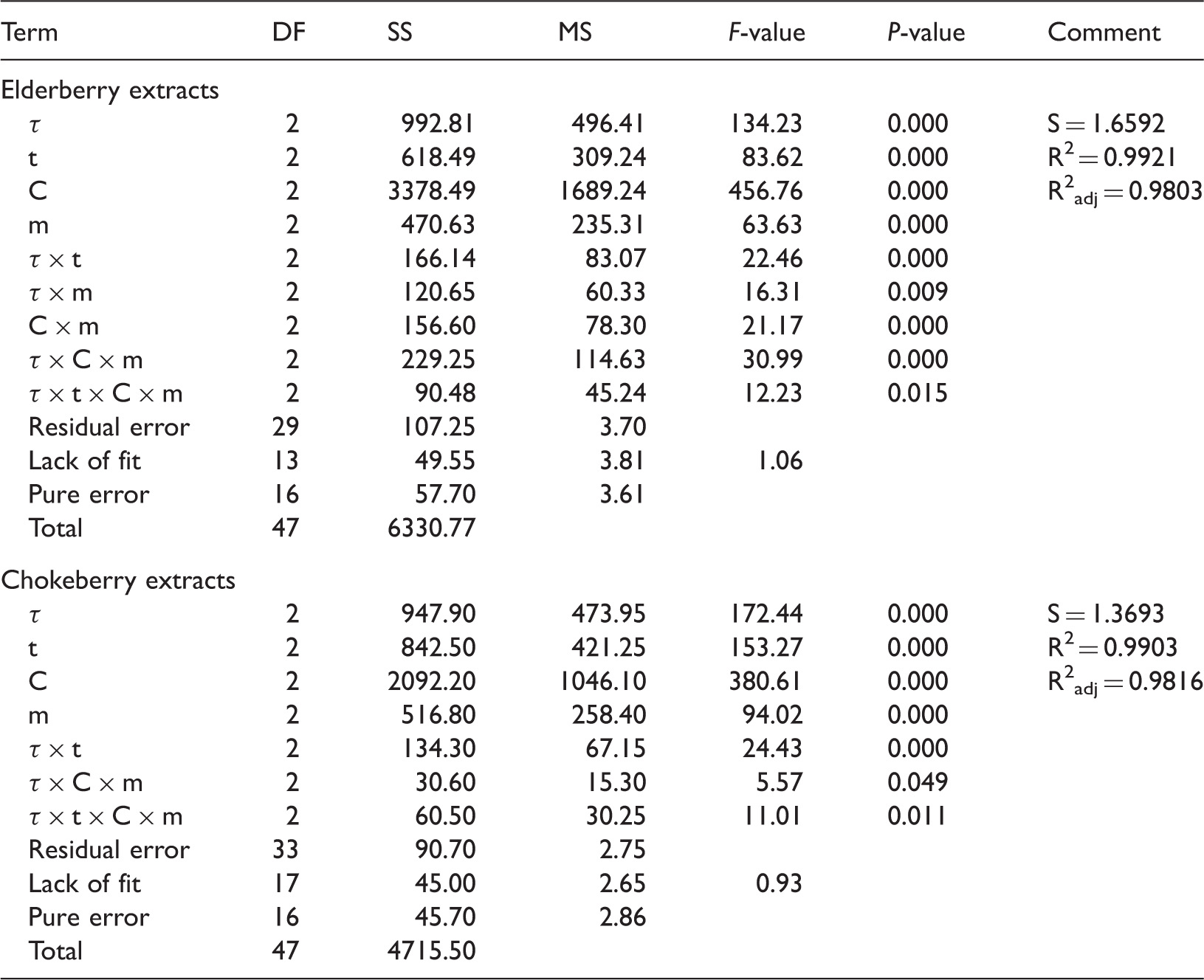
DF: degrees of freedom; SS: sum of squares; MS: mean square.
It can be seen (Table 2) that for equations (3) and (4), the values of the determination coefficient R2 were 0.9921 and 0.9903, respectively, while the values of adjusted determination coefficient R2adj were 0.9803 and 0.9816, respectively. It indicates about a high degree of correlation between the experimental and predicted values.
Using of equations (3) and (4), it is possible to predict the removal of anthothyanins from berry extracts on bentonite. Table 2 shows the removal efficiency of anthothyanins across the experimental and predicted values. These data indicate that there is a good fit between the observed and the predicted values.
Conclusions
In the present study, bentonite was selected as a cheaper and readily available adsorbent for the removal of anthocyanins from aqueous berry extracts. The influence of contact time, temperature, initial concentration of anthocyanins, and adsorbent mass on removal efficiency of anthocyanins was designed by using 24 full factorial design. A full first-order polynomial equation was used to analyze the experimental results and to predict the percentage removal of anthocyanins. The most significant factors on removal efficiency of anthocyanins can be written with increasing importance in the following way: initial anthocyanin concentration > contact time > temperature > adsorbent mass. Two, three, and four ways interactions between them were also significant. The ANOVA showed high coefficients of determination (0.9921 and 0.9816) of equations, thus confirming adequacy of adjustment of the regression models with the experimental data. Maximum removal efficiency of anthocyanins from elderberry and chokeberry extracts occurred at low initial concentration and temperature, high contact time and adsorbent mass. Due to the obtained results, bentonite could be recommended as an effective and low-cost adsorbent for the removal of anthocyanins from aqueous berry extracts.
Footnotes
Acknowledgements
This article was first presented at the 15th Ukrainian-Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


