Abstract
A physical mixture of
Introduction
Pollution of the water sector by toxic heavy metals like chromium generated from industries is a serious environmental problem. Because of their high solubility in water, heavy metals are taken up by plants or animals and hence easily entering the food chain. In addition, heavy metals are non-biodegradable, thus may last longer in the environment causing detrimental health effects such as diseases and disorder on humans and
The removal of Cr(VI) from aqueous solutions by adsorbents is a complex process that could involve complexation, reduction, ion exchange, precipitation and adsorption. Because Cr(VI) is a strong oxidant, it is prone to undergo reduction to Cr(III) under strongly acidic conditions and/or in the presence of electron donors (Yang and Chen, 2008). Adsorbents used for Cr(VI) removal in literature include natural polymers (Jung et al., 2013), plant-based materials (Altun and Pehlivan, 2012), synthetic polymers (Bayramoglu and Yakup Arica, 2011), activated carbons (ACs) (Demirbas et al., 2004; Jung et al., 2013; Pakade et al., 2016a) or inorganic materials (Dubey et al., 2016). Among these adsorbents, ACs produced from locally available low-cost agricultural wastes or by-products (Anirudhan and Sreekumari, 2011) have received extensive consideration owing to their large surface area, high adsorption capacity and well-defined microporous structure. Typical agricultural wastes/by-products that have been explored for the preparation of ACs for the removal of Cr(VI) include, but not limited to, sawdust (Kapur and Mondal, 2013), walnut shell (Altun and Pehlivan, 2012),
On the other hand, clay minerals, owing to their high surface areas, layered structure, low cost, non-toxic, easy accessibility and chemical stability (Ballay et al., 2014; Sarkar et al., 2010), have also received broad applications on the removal of pollutant from aqueous solutions particularly the removal of heavy metal ions (Bradl, 2004), dyes (Arbeloa et al., 2002) and other organics (El-Nahhal and Safi, 2004). Chiefly, it has been shown that clays can be cost-effective adsorbents for the removal of heavy metals from acid mine drainage samples (Rios et al., 2008). Numerous studies have reported the transformation of Cr(VI) to Cr(III) during adsorption (Li et al., 2017; Lv et al., 2016; Pakade et al., 2016a; Stoica-Guzun et al., 2016; Tang et al., 2014; Yang and Chen, 2008), but no attempt was made to remove the formed Cr(III). Therefore, the current study attempts to use cationic clay (attapulgite clay which is a magnesium aluminium silicate rich clay) for the removal of the formed Cr(III) while AC will be responsible for the removal of Cr(VI) and reduction. The physical mixture of clay and AC is a simple and low-cost method as it involved no synthesis. Combinations of clay with other adsorbents like nanotubes (Ballay et al., 2014), AC (Chen et al., 2011) and polymers (Kotal and Bhowmick, 2015) have been investigated for heavy metal removal from aqueous solutions. The attapulgite clay@carbon composite material prepared by hydrothermal carbonisation process exhibited high adsorption capacity for Cr(VI) removal of 177.74 mg g−1 (Chen et al., 2011). In this work, it was reported that the removal mechanism was a combined effect of complexation, redox reaction and ion exchange processes (Chen et al., 2011).
Materials and methods
Chemicals and instrumentation
All the chemicals and reagents used were of high-quality grade and were used without further purification. K2Cr2O7, NaOH and HCl were sourced from Merck Chemical Co. (Johannesburg, South Africa). 1,5’-Diphenylcarbazide was purchased from Sigma-Aldrich (Johannesburg, South Africa).
All solutions were prepared with ultrapure water obtained from the Siemens LaboStar equipment (Warrendale, Pennsylvania, USA). An ADWA AD111 OPR pH meter from Adwa Instruments (Szeged, Hungary) was used for measuring the pH of the solutions. A multipoint MS-53 M stirrer model Jeio Tech (Seoul, Korea) was utilised for performing batch adsorption experiments. Total chromium was measured on an atomic absorption spectrophotometer AA7000 from Shimadzu (Kyoto, Japan) whilst UV-VIS (T80+) supplied by PG Instruments was used for the determination of Cr(VI). Centrifugation was performed on a CL10 ThermoScientific centrifuge from Labotec (Johannesburg, South Africa). Adsorption and kinetic isotherms were plotted using Origin 7.0 software.
Adsorbent and adsorbate
Commercial
Adsorption studies
Parameters controlling adsorption of Cr(VI) ions by clay and AC mixture were evaluated in batch experiments with two repetitions. The influence of solution pH (pH 1–9), initial Cr(VI) concentration (10–250 mg L−1), adsorbent dosage (0.02–0.30 g) and stirring time (15–240 minutes) was investigated. Solid material was separated from the solution after adsorption by first centrifugation and then filtered through a Whatman #4 filter paper. Then the concentration of Cr(VI) and total chromium was determined using UV-VIS spectroscopy and atomic absorption spectrophotometer, respectively. For the determination of Cr(VI), a calorimetric method entailing the use 1,5’-diphenylcarbazide complexing agent in acidic medium was followed and the absorbance was measured at 540 nm wavelength (Greenberg et al., 1992).
The adsorption capacity (

Ionic strength
Effect of salts on the removal of chromium by the AC–clay combination was studied in the presence of various concentrations of different electrolyte solutions. Binary solutions containing Cr(VI) ions and each of the following electrolytes NaNO3, NaCl, KNO3, KCl and NH4Cl were prepared. The concentration of the competitor anion was varied from 0.5%, 1.0%, 3.0% and 5.0% while the concentration of Cr(VI) was kept constant at 100 mg L−1.
Results and discussion
Characterisation of materials
Morphological analyses were carried out on an FEI Quanta 200 SEM (FEI, Hillsboro, OR, USA). The SEM images for the adsorbent before and after Cr(VI) adsorption are displayed in Figure 1. A dispersion of clay powder in the pores of MAC can be observed in Figure 1(a). After adsorption, MAC pores are much clearer and visible. This could imply that segregation of MAC and clay adsorbents took place after adsorption. Both adsorbents displayed heterogeneous surface properties which is suitable for adsorption. Elsewhere (Hashemian et al., 2013), it was also observed that the surface of the adsorbent changed significantly after adsorption.
SEM images for adsorbents before (a) and after (b) chromium(VI) adsorption.
Optimisation of MAC–clay mixture and adsorption by pure materials
AC–clay physical mixture was optimised by mixing MAC and attapulgite clay in the following mass ratios: 3:1; 1:1; 1:3, representing 0.3 g:0.1 g; 0.1 g:0.1 g; 0.1 g:0.3 g, respectively, and performing the adsorption of Cr(VI) at uniform conditions, that is, at 100 mg L−1 initial Cr(VI) concentration, initial pH 1, solution volume 30 mL and 2 hours of contact time. Figure 2(a) shows the effect of MAC–clay ratio on the adsorption of chromium. It can be observed that the removal efficiency of chromium increased as the amount of MAC was increased. All chosen ratios showed adsorption efficiency greater than 99%. Even though 3:1 ratio seemed to be optimum, 1:1 was used in succeeding experiments simply because there was only 0.1% difference between 3:1 and 1:1. Therefore, to save material (MAC), 1:1 was the best option. Adsorption of Cr(III) and Cr(VI) by pure materials (clay or MAC) showed high %removal of Cr(III) by clay (82%) and very low %removal of Cr(VI) by clay (7%) (Figure 2(b)). In pure MAC, the trend was reversed, that is, 42% removal of Cr(VI) and 5% removal for Cr(III). However, these results revealed that hybrid material had higher %removal than pure materials. This study was performed using saturated solution of chromium solution at pH 3.
Optimisation of MAC:clay ratio for adsorption of total chromium (a) and removal of Cr(III) and Cr(VI) by pure samples (b).
Adsorption studies
Effect of pH
It has been shown that the removal of chromium (VI) by adsorbents could be through adsorption and/or reduction (Li et al., 2017; Lv et al., 2016) with pH playing a significant role. The influence of pH on the adsorption of chromium by the MAC–clay is depicted in Figure 3. The percentage removal (% Effect of solution initial pH (a) and concentrations of total chromium and chromium(VI) after adsorption (b) (conditions: amount of adsorbent 0.1 g; solution volume 25 mL; contact time 2 hours).


Equilibrium concentration for Cr(VI) and total chromium was plotted against the initial pH of the solution (Figure 3(b)). As can be seen, at pH below 2 the amount of Cr(VI) at equilibrium was almost zero, but the amount of total chromium in solution was about 40 mg L−1 which signified possible reduction. However, as the pH was increased from pH 3 to pH 9, the amount of Cr(VI) in solution increased. This translated to less percentage adsorption of Cr(VI) by the sorbent as most of it was remaining in solution. Total chromium also increased indicating that less adsorption was achieved. At pH 3, the equilibrium concentration of Cr(VI) and total chromium was similar. This implied that the reduced Cr(III) was adsorbed by the material. In addition, the reduction of Cr(VI) to Cr(III) causes depletion of protons in solution leading to increase in solution pH essential for Cr(III) uptake by clay. Hence, pH 3 was used for subsequent studies because adsorption and reduction were efficient.
Effect of concentration and adsorbent dosage
Figure 4(a) shows the percentage removal (% The percentage removal and adsorption capacity on the effect of initial Cr(VI) concentration and total Cr removal (a) and effect of adsorbent mass (b). ((a) conditions: amount of adsorbent 0.1 g; solution volume 25 mL; pH 3; contact time 2 hours. (b) conditions: pH 3; solution volume 25 mL; concentration 100 mg L−1; contact time 2 hours).
To further confirm the synergistic effect of MAC and clay in the removal of Cr(VI) and Cr(III), SEM-EDS images of the exhausted materials revealed the presence of chromium in both materials (Figure 5). The EDS showed a presence of Au and Pb in both materials because the samples were coated with Au and Pb to create a conductive layer of the metal on the sample to prevent charging. The carbon sample showed the presence of C while Al, Si, Mg and Fe were observed in the clay sample, and these formed the backbone of the investigated adsorbents.
SEM-EDS images for MAC (a) and attapulgite clay (b) after adsorption.
Effect of time
Figure 6(a) shows the equilibrium concentration of chromium versus contact time. It can be observed that after 15 minutes, the amount of Cr(VI) left in solution was almost zero while the amount of Cr(III) left was 32 mg L−1. This observation illustrated that the Cr(VI) that got attached to the carbon was first reduced to Cr(III) and released back into solution. However, as the time was prolonged the equilibrium concentration of total chromium decreased to the same level as Cr(VI) from 60 to 240 minutes. The decrease of Cr(III) in solution with increasing time proved that adsorption of Cr(III) through cationic exchange resin was a slow process. Optimal contact time was found to be 60 minutes, but 120 minutes were used in subsequent experiment so to allow the reactions to reach completion. Figure 6(b) also illustrates that adsorption and transformation of Cr(VI) is a rapid process while adsorption of Cr(III) by the attapulgite clay is a slow process. This is demonstrated by the amount of Cr(III) left in solution after 30 minutes.
The effect of contact time on Cr(VI) adsorption (a) and equilibrium Cr concentrations (b) (Conditions: amount of adsorbent 0.1 g (0.05 g: 0.05 g); solution volume 25 mL; pH 3; Cr(VI) concentration 100 mg L−1).
Effect of coexisting anions and ionic strength
Figure 7 illustrates the influence of ionic strength on the adsorption capacities of Cr(VI) and Cr(III) by MAC–clay mixture. It can be observed that the adsorption capacity of Cr(VI) and Cr(III) decreased as the ionic strength of solution increased. This was consistent with Chen et al.’s (2012) and Gladysz-Plaska et al.’s (2012) observations that increasing the ionic strength of the solution and decreasing the pH resulted in decreased Cr(III) adsorption capacity. When looking at the KCl:KNO3 and NaCl:NaNO3 pairs, it could be observed that higher removal of Cr(VI) was achieved in the presence of Cl− in KCl:KNO3 pairs, but a contrasting observation was observed in the NaCl:NaNO3 pairs as higher removal was obtained in NaNO3. Gladysz-Plaska et al. (2012) also observed high percentage adsorption of Cr(VI) in the presence of NaNO3 than in NaCl at lower concentration dosages, but the order was reversed at higher concentration of electrolyte. The removal of Cr(III) by MAC–clay mixture decreased in the following order KCl>NaCl>KNO3>NaNO3>NH4Cl. Therefore, it can be concluded that the cation exchange was greatest in the presence of K+, then Na+ and lastly The effect of ionic strength and co-ions on Cr(VI) (a) and Cr(III) (b) adsorption capacity by MAC-attapulgite mixture (Conditions: amount of adsorbent 0.1 g; solution volume 25 mL; pH 3; Cr(VI) concentration 100 mg L−1; contact time 2 h).
Figure 8 shows the effect of different electrolytes on the reduction of Cr(VI) by MAC and subsequent adsorption of Cr(III) by attapulgite clay. Figure 8(a), (d) and (e) shows that there was Cr(III) present in solution after adsorption implying that substantial amount of Cr(VI) was reduced to Cr(III) and not adsorbed on the sorbents. The amount of Cr(III) present ranged from 45–57 mg L−1, 25–28 mg L−1 and 20–28 mg L−1 for solutions containing NaNO3, KCl and NH4Cl, respectively. Therefore, removal of Cr(III) was mostly affected in the following order NaNO3>KCl>NH4Cl. There was very little Cr(III) generated in the presence of KNO3 and NaCl (Figure 8(b) and (e)). These results show that it was not only about the cation that interfered with the removal of Cr(III) but also the balancing counter ion played a crucial role. Cr(VI) removal was most efficient in the presence of KCl followed by NaNO3 then KNO3, NH4Cl and NaCl had similar impacts. The amount of Cr(VI) present in solution after adsorption decreased in the order KNO3>NaCl≈NH4Cl>NaNO3>KCl. These results demonstrated that simultaneous removal of Cr(VI) and Cr(III) in the presence of electrolytes was a complex process. In addition, counter ions Cl− and The effect of ionic strength and co-ions on Cr(VI) (a) and Cr(III) (b) adsorption capacity by MAC-attapulgite mixture (Conditions: amount of adsorbent 0.1 g; solution volume 25 mL; pH 3; Cr(VI) concentration 100 mg L−1; contact time 2 h).
Adsorption isotherms
Freundlich and Langmuir isotherms were used to model the adsorption capacity data. Information about the maximum adsorption capacities is important in order to infer about exhaustion of sorbents particularly for industrial application. Both models were used in their linear forms to fit the effect of concentration data. A linear plot of 
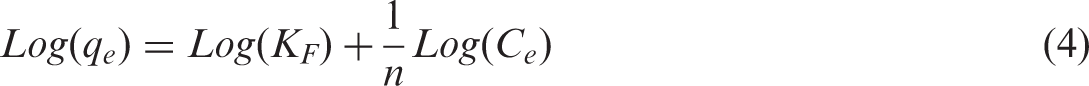
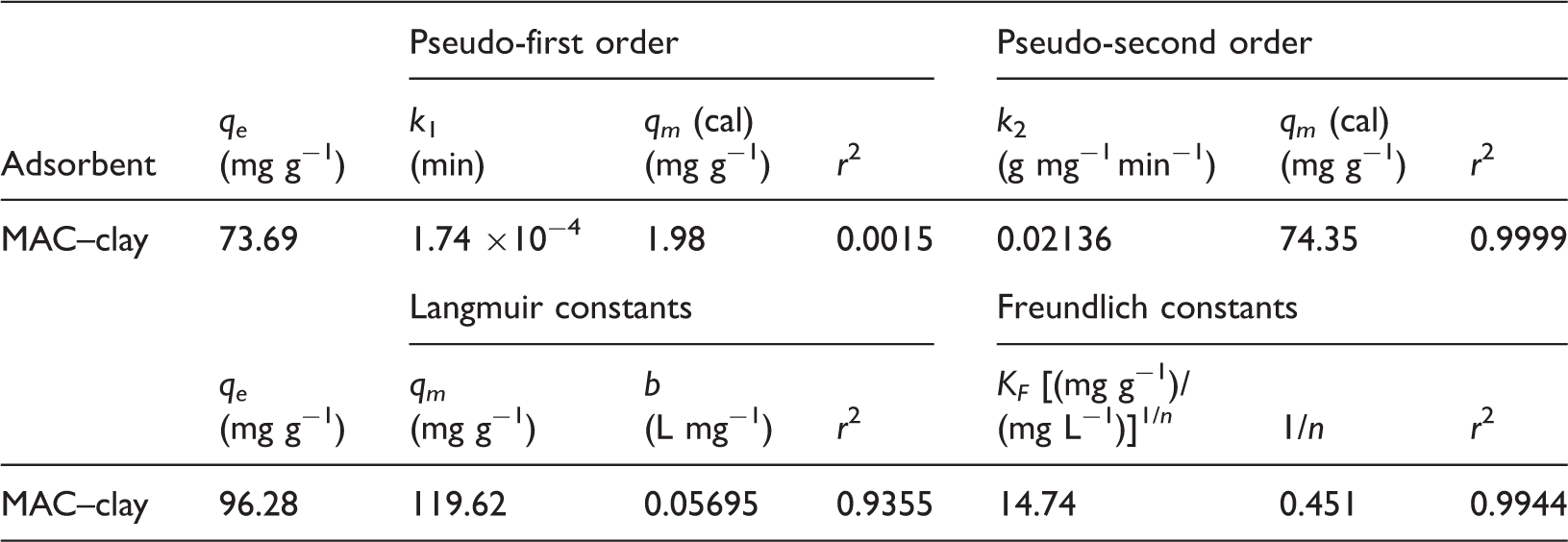
Langmuir and Freundlich isotherm constants, and pseudo-first-order and pseudo-second-order constants for the adsorption of Cr(VI) on MAC–clay adsorbents.
Adsorption kinetics
The influence of stirring time on adsorption of chromium on the sorbent mixture was investigated by varying stirring time from 15 to 240 minutes while other variables were kept constant. The data obtained was fitted in the pseudo-first-order (PFO) and PSO rate models to assess the processes governing sorption of adsorbate and mass transfer rate. Linearized forms of the PFO and PSO models are supplied in equations (5) and (6), respectively
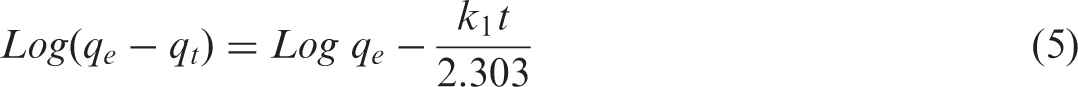

Table 1 presents the correlation data for PFO and PSO kinetic models for the adsorption of Cr(VI) on MAC–clay. The
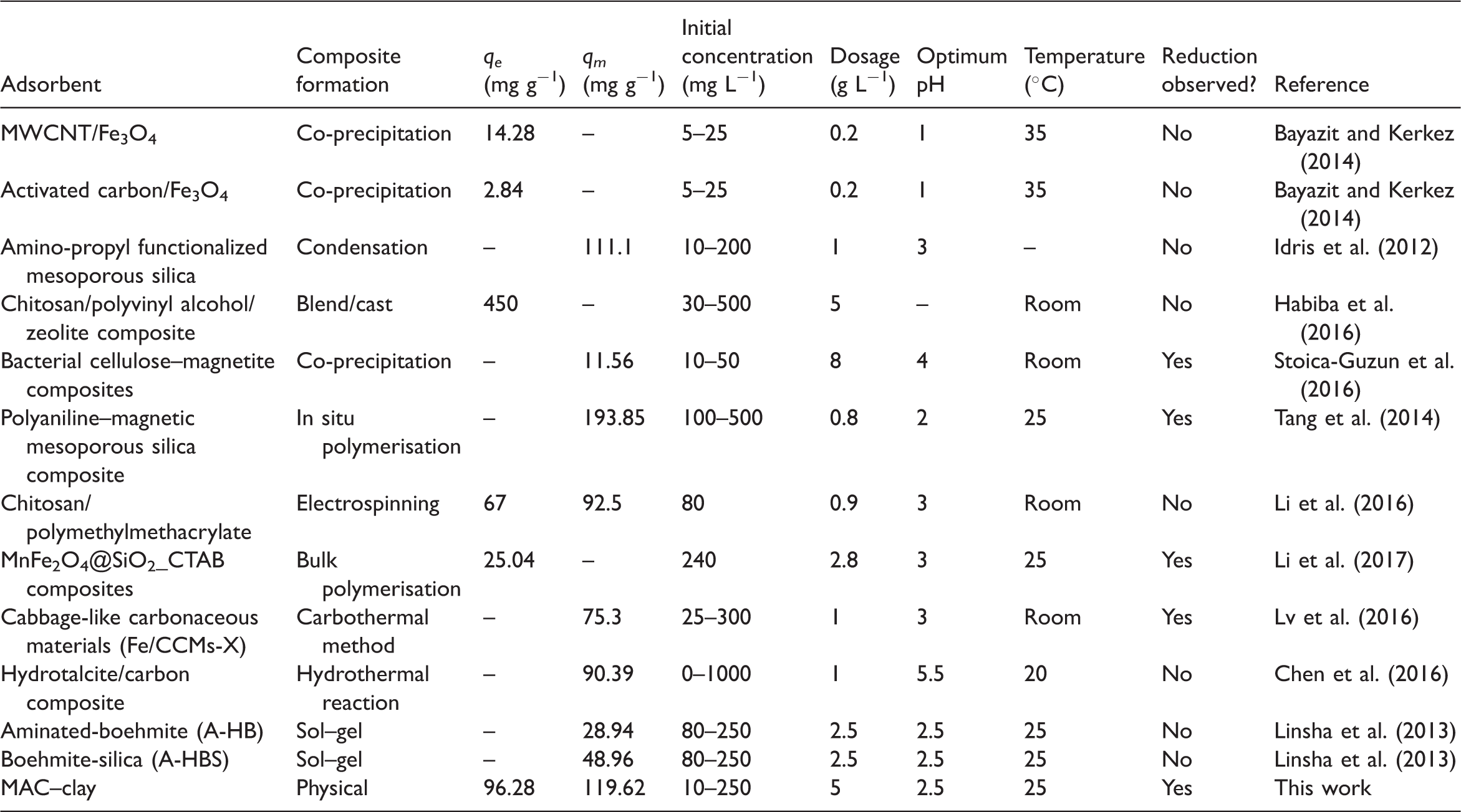
Comparison of adsorption capacity with composite materials
Comparative studies of adsorption capacities of Cr(VI) onto adsorbents.
Conclusion
These results demonstrate that a combination of two sorbents, MAC and attapulgite clay may be used to combat the scourge of Cr(VI) contamination in wastewater systems. Cr(VI) is a tricky pollutant that easily transforms to Cr(III) when its removal is attempted using adsorbents bearing heteroatoms. This is because the heteroatoms impart electrons that facilitate the reduction of Cr(VI) into Cr(III) leading to incomplete removal of chromium. In this case, Cr(VI) was adsorbed and some reduced by MAC while the produced Cr(III) was taken up by attapulgite clay through ion exchange mechanism fashioning synergistic effects for simultaneous removal of Cr(VI) and Cr(III). SEM-EDS revealed the presence of chromium in both pure MAC and clay samples, an indication that Cr was adsorbed by both materials. Parameters affecting adsorption were evaluated and optimum values were pH 3, contact time (1 hour), adsorbent mass (0.2 g) and concentration of 50 mg L−1. The effect of ionic strength was also investigated and it was found that the amount of Cr(VI) after adsorption decreased according to the following order KNO3>NaCl≈NH4Cl>NaNO3>KCl, demonstrating that total and simultaneous removal of Cr(VI) and Cr(III) in the presence of electrolytes is a complex process. It was clear that the counter cation also played a role in the adsorption of Cr(VI). Hence, this presents a challenge in treating contaminated wastewater with adsorbents as the presence of electrolyte will inhibit the removal of chromium. Furthermore, it was shown that the data fitted best in Freundlich isotherm and PSO rate model, leading to a conclusion that a multilayer-chemisorption process was involved for the removal of chromium and this was no surprise due to the diversity of binding sites present in MAC and clay.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial assistance from the National Research Foundation (TTK13061018779 and TTK160510164648) and the Vaal University of Technology is greatly appreciated.
