Abstract
Two coordination polymers of erbium(III) and neodymium(III) ions with 1,3,5-benzenetricarboxylic acid were synthesized under the solvothermal conditions from the dimethylformamide solution. They were characterized by the attenuated total reflectance Fourier transform spectroscopy (ATR-FTIR), thermogravimetry and differential scanning calorimetry (TG-DSC), thermogravimetric analysis coupled with Fourier transform infrared spectroscopy (TG-FTIR) and X-ray diffractions methods. The single-crystal X-ray analysis confirmed formation of three-dimensional framework of Er(III) 1,3,5-benzenetricarboxylate with the channels occupied by dimethylformamide and water molecules. Porosity of crystalline complexes was investigated by the nitrogen sorption experiments. The Nd and Er compounds exhibit real porosity with the BET surface area of 259 and 225 m2/g, respectively. The Horvath–Kawazoe analysis of pore-size distributions for the obtained complexes points out to their microporous character.
Keywords
Introduction
Metal–organic frameworks (MOFs) and coordination polymers (CPs) have been particularly highlighted as an emerging class of new porous materials due to their potential applications in diverse fields like gas storage, molecular separation, catalysis, optics and drug delivery (Furukawa et al., 2013). Porous MOFs possess some advantages which are beneficial for the gas/solvent sorption application, such as high surface areas, uniform and tunable pore size (Chen and Ma, 2012). Among different types of carboxylate-based linkers employed for the synthesis of MOFs, 1,3,5-benzenetricarboxylic acid (H3btc) belongs to the most explored ligands used for the construction of porous CPs (Basolite®C-300 BASF analogue of HKUST-1 (IRMOF-6)) (Chui et al., 1999). Terbium 1,3,5-benzenetricarboxylate (MOF-76) is one of the examples of microporous lanthanide MOFs which demonstrates permanent microporosity and molecular sieving behaviours, as revealed from the sorption isotherms of different gas/solvent molecules (N2, Ar, CH2Cl2, C6H6, C6H12) (Rosi et al., 2008). Also porous rare-earth MOF based on the yttrium ions (Y-btc) and 1,3,5-benzenetricarboxylic acid was prepared under the solvothermal conditions (Luo et al., 2005). It possesses tetragonal pores with a size of approximately 6 Å and exhibits highly selective sorption behaviour of hydrogen over nitrogen. The series of isostructural microporous Ln-MOFs enantiomers, Ln(btc)(H2O)(DMF)1.1 (Ln = Y, Tb, Dy, Er and Yb) features free windows of 6–7 Å with accessible exposed metal sites, moderate surface areas and high thermal stability (over 450℃) (Jiang et al., 2010). Another interesting example of btc-based Ln-MOF is Dy(btc)(H2O)·DMF (Guo et al., 2006). The removal of the guest and terminal coordinated molecules in the Dy-MOF rendered the permanent porosity and accessible Lewis acid sites which afforded interesting performances properties regarding hydrogen adsorption, sensing and catalysis.
The lanthanide MOFs with benzene polycarboxylic acids have been investigated by our group for many years. The whole series of lanthanide complexes with rigid (Łyszczek, 2007; Łyszczek and Lipke, 2013; Łyszczek et al., 2011) or more flexible benzene polycarboxylic acids (Łyszczek and Mazur, 2012, Łyszczek et al., 2016a, 2015) were synthesized and investigated. As the continuation of our research on lanthanide MOFs, neodymium(III) and erbium(III) complexes with 1,3,5-benzenetricarboxylic acid were prepared by the solvothermal method using dimethylformamide (DMF) as a solvent (Łyszczek et al., 2016b). Previously, the series of nonporous lanthanide 1,3,5-benzenetricarboxylates obtained under the hydrothermal conditions was studied (Rzączyńska et al., 2005). The aim of this work was to investigate some physicochemical properties, crystal structures, thermal and sorption properties of Nd(III) and Er(III) CPs as representatives of light and heavy lanthanides.
Experimental
Synthesis
The mixtures of stoichiometric amounts of lanthanide chlorides (1 mmol), 1,3,5-benzenetricarboxylic acid (1 mmol) and 20 ml of DMF were sealed in 150 ml stainless steel reactors with a Teflon liner and heated at 140℃ for 72 h. Afterwards the reaction systems were gradually cooled to room temperature. The obtained compounds were filtered off and washed with DMF. The erbium complex [Er(btc)(DMF)2]·0.5DMF·0.5H2O was obtained in the form of single crystals suitable for X-ray diffraction measurements whereas the neodymium complex Nd(btc)(DMF)1.5 was obtained in the form of polycrystalline powder.
Characterization
The contents of C, N and H in the obtained compounds were determined by the elemental analysis with a Perkin-Elmer CHN 2400 elemental analyser. The elemental analysis for C13.5H13.5N1.5 O7.5Nd: Calcd: C, 35.18%; H, 2.93%; N, 4.55%. Found: C, 35.92%; H, 3.05%; N, 4.21%. The elemental analysis for C16.5H21.5N2.5O9Er: Calcd: C, 34.99%; H, 3.80%; N, 6.19%. Found: C, 34.98%; H, 3.67%; N, 5.88%.
The ATR-FTIR spectra were recorded over the range 4000–600 cm−1 on the Nicolet 6700 FTIR spectrophotometer equipped with a universal ATR attachment with a ZnSe crystal. The TG-DSC analyses in air were made on the Setsys 16/18 analyser (Setaram). The samples (6–8 mg) were heated in the range of 30–800℃ in ceramic crucibles at a heating rate of 10℃/min. The TG-FTIR coupled measurements were performed using a Q5000 (TA) apparatus coupled with the Nicolet 6700 spectrophotometer.
Single-crystal diffraction data were collected at 100 K on an Oxford Diffraction Xcalibur CCD diffractometer with the graphite-monochromated Mo Kα radiation (λ = 0.71073 Å). The programs CrysAlis CCD and CrysAlis Red (CrysAlis PRO, 2013) were used for data collection, cell refinement and data reduction. A multi-scan absorption correction was applied. The structures were solved by direct methods using SHELXS-97 and refined by the full-matrix least squares on F2 using SHELXL-97 (Sheldrick, 2015). The water molecule (O1w) and the lattice DMF molecule are disordered over the crystallographic two-fold axis with the carbonyl O3s atom been located on the axis. The positions of their atoms were refined with the site occupancy factor of 0.5. All non-H atoms were refined with anisotropic displacement parameters. The water H-atoms were found in difference Fourier maps and not refined. All remaining ones were placed in geometrically calculated positions and treated as riding atoms with Uiso = 1.2 − 1.5Ueq(C).
The powder patterns were recorded on an Empyrean diffractometer PANalytical.
Nitrogen adsorption and desorption isotherms were measured using an ASAP 2010 apparatus (Micromeritics) at −196℃.
Results and discussion
Structural and spectroscopic characterization
Microporous CPs based on neodymium(III) and erbium(III) ions and 1,3,5-benzenetricarboxylic acid of the formulae Nd(btc)(DMF)1.5 and [Er(btc)(DMF)2]·0.5DMF·0.5H2O (where btc = C9H3O63−) were prepared by the solvothermal route using DMF as a solvent. The powder X-ray diffraction patterns confirmed that both complexes are crystalline. The single-crystal X-ray diffraction measurements were performed only for the erbium complex. Erbium 1,3,5-benzenetricarboxylate [Er(btc)(DMF)2]·0.5DMF·0.5H2O crystallizes in the monoclinic system, space group C2/c and unit cell parameters a =18,5397(7) Å, b = 11,3386(4) Å, c = 19,6493(8) Å, β = 106,175(4)°, V = 3967.1(3) Å3. The molecular structure of the investigated complex is the same as previously reported structures of lanthanide 1,3,5-benzenetricarboxylates Ln(BTC)(DMF)2 H2O (Li et al., 2007). Nevertheless, the composition of outer sphere of complex is different according to distinct conditions of synthesis. The examined [Er(btc)(DMF)2]·0.5DMF·0.5H2O complex is a three-dimensional coordination polymer. Each Er atom is coordinated by eight oxygen atoms derived from four different btc3− moieties and two DMF molecules. Two COO groups exhibit bidentate-chelating character while the remaining one appears as the bidentate-bridging group. All erbium(III) ions are surrounded by two chelating and two bridging carboxylate groups from different ligands (Figure 1(a)). In this three-dimensional coordination network, channels occupied by DMF and water molecules (located around the two-fold axis) can be distinguished while in the previously described lanthanide complexes only water molecules were observed in channels.
(a) TG curves of thermal decomposition of Nd and Er 1,3,5-benzenetricarboxylates in air. The inset shows the part of crystal structure [Er(btc)(DMF)2]·0.5DMF·0.5H2O. (b) Nitrogen adsorption/desorption isotherms measured at −196℃. TG: thermogravimetric.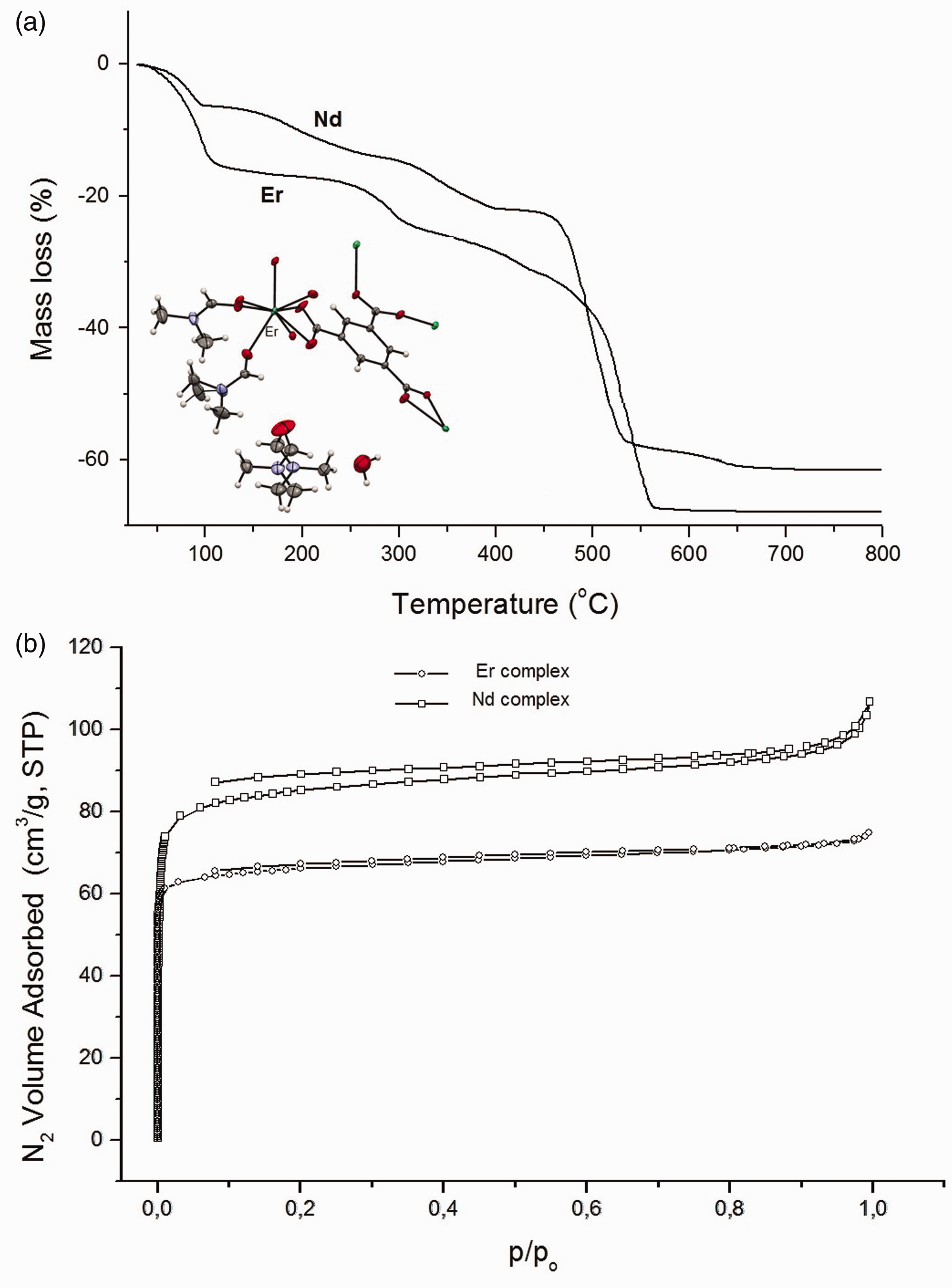
The ATR-FTIR spectra of free acid and investigated complexes confirmed deprotonation of all carboxylic groups COOH after lanthanide ions coordination. Characteristic bands for free acid at 1720, 1280, 1249 and 932 cm−1 corresponding to the stretching vibrations of ν(C=O) and ν(C–OH) groups as well as deformation vibrations from γ(OH) group are not observed in the spectra of the complexes (Rzączyńska et al., 2005). On the other hand, both spectra exhibit the bands from asymmetric stretching vibrations of carboxylate groups which appear at 1607 cm−1 and symmetric stretching vibrations at the slightly different wavenumbers: 1374 cm−1 (Nd) and 1366 cm−1 (Er). Moreover, the spectra show two bands characteristic of DMF molecules. The bands at 3066 and 2928 cm−1 from the stretching vibrations of CH3 groups as well as bands at 1667 and 1643 cm−1 were assigned to the stretching vibrations of free and coordinated carbonyl groups from the DMF (Łyszczek and Lipke, 2013).
Thermal analysis
Thermal stability as well as pathways of thermal decomposition of both complexes was investigated by means of the TG-DSC (air) and TG-FTIR methods (nitrogen) (Figure 1(a)).
During heating of Nd(btc)(DMF)1.5 complex, solvent molecules are released at first. Mass loss of 7% corresponding to removal of 0.5 molecule of DMF (calc. 7.92%) occurs in the range 30–110℃. Further gradual weight loss of 16% observed in the range 120–400℃ is assigned to the release of one DMF molecule (calc. 15.84%). Endothermic effects connected with removal of DMF molecules are observed at 89, 188 and 330℃. Over 420℃, the decomposition process of desolvated framework as well as burning of organic ligand takes place. Formation of final solid product of heating (Nd2O3) is observed at 700℃ (calc. total mass loss 63.50%, found 62%).
The desolvation process of the [Er(btc)(DMF)2]·0.5DMF·0.5H2O complex takes place in three hardly distinguishable steps. The first weight loss (17%) found in the temperature range of 30–110℃ is associated with release of solvent molecules from the channels (0.5 molecule of DMF and 0.5 molecule of water) and about half molecule of bonded DMF (calc. 15.82%). Then the remaining 1.5 molecules of DMF (found 19%, calc. 19.04%) are released in two hardly distinguishable steps in the temperature ranges 110–300 and 300–430℃. Removal of solvent molecules is accompanied by the endothermic effects observed on the DSC curve at 76, 100 and 290℃. Further heating causes the decomposition of the desolvated form of the complex along with burning of organic ligand. The final solid product of decomposition (Er2O3) is formed at about 560℃ (calc. total weight loss 66.74%, found 68%).
The presence of solvent molecules in the structures of complexes was also confirmed by the analysis of the FTIR spectra of gaseous products of thermal decomposition of investigated complexes. Heating of the erbium complex leads to the release of water molecules up to about 100℃. The FTIR spectra show characteristic bands in the regions: 4000–3500 and 1800–1300 cm−1 due to stretching and deformations vibrations of water molecules. Further heating of the complexes leads to the departure of DMF molecules. They give a characteristic gaseous FTIR spectrum with the bands: at 2941 and 2845 cm−1 from the ν(CH3) groups, the band at 1719 cm−1 from ν(CO) and the band at 1384 cm−1 from the bending vibrations of CH groups. Additionally, the bands at 1271 and 1082 cm−1 derived from the rocking vibrations of CH3 from the (CH3)2N moiety are observed. The two-stage process of solvent molecules removal can confirm the presence of differently bonded DMF molecules in the complex structures. At a higher temperature, the decomposition process of the complexes leads to the release of carbon oxides. Carbon monoxide molecules give a doublet of bands at 2177, 2113 cm−1. The bands observed at 2359, 2343 and 690 cm−1 are derived from the stretching and deformation vibrations of carbon dioxide molecules. At a higher temperature (600℃) release of some hydrocarbons can be assumed based on the presence of stretching vibrations C–H in the range 3000–2700 cm−1.
Sorption properties
In order to verify porous nature of investigated lanthanide 1,3,5-benzenetricarboxylates, the nitrogen adsorption–desorption isotherms were measured at −196℃. The Nd and Er compounds exhibit real porosity with the BET surface area of 259 and 225 m2/g, respectively. The total pore volumes for the Nd and Er complexes are 0.1651 and 0.1156 cm3/g, respectively. The pore-size distributions for the complexes were calculated from the nitrogen isotherms by applying the Horvath–Kawazoe (H–K) analysis. The median pore width for the Nd and Er complexes is 7.46 and 4.86 Å, respectively.
The obtained nitrogen isotherms shown in Figure 1(b) are of type I according to the IUPAC classification with the pore-filling step at p/po ≈ 0.1 that is indicative of the presence of micropores in the structures. In the range p/po (0.1–1.0) the slight increase of the adsorbed amount of nitrogen was observed. This fact can be related to the adsorption on the external surface on nanocrystallites. Both isotherms exhibit a hysteresis between the adsorption and desorption curves down to the lowest attainable pressure. The desorption process of nitrogen is reversible to about 0.1 p/po pressure and removal of nitrogen at low pressure proceeds with some difficulties. The most reasonable explanation of this observation is irreversible uptake of molecules in pores.
Conclusions
Two lanthanide MOFs based on the 1,3,5-benzenetricarboxylate linker were solvothermally synthesized and characterized. The lanthanide ions in the complexes are coordinated by carboxylate oxygen atoms of btc3− ligand and DMF molecules. The obtained complexes are thermally stable up to about 400℃. The nitrogen sorption measurements confirmed their microporous character. The single-crystal X-ray analysis of the erbium complex showed that it has a form of three-dimensional CP with the channels occupied by DMF and water molecules. Based on the obtained results it can be concluded that also the Nd complex has the three-dimensional structure with solvent molecules filling the free voids in the crystal.
Footnotes
Authors’ note
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
