Abstract
In this paper, new styrene microspheres with pendant methylenethiol or sulphonic groups are presented. The copolymer was obtained by the emulsion–suspension polymerization of aromatic tetrafunctional monomer 1,1′-bis[4-(2-hydroxy-3-acryloiloxypropoxy)phenyl] cyclohexane (C.DA) with styrene. This process was performed at constant mole ratio of C.DA to styrene (1:1 w/w). In order to introduce sulphur groups on the surface of the microspheres, the multistage modification was carried out. After modification very attractive methylenethiol (–CH2SH) and sulphonic (–SO3H) groups were introduced into the structure of microspheres. Elemental analysis and the spectroscopic method (attenuated total reflectance-Fourier transform infrared spectroscopy) were used to confirm the modification reaction. Thermal stabilities and degradation behaviours of the obtained copolymers were characterized by using thermogravimetry/differential scanning calorimetry/quadrupole mass spectrometry analyses.
Introduction
Heavy metals are used in many industrial areas particularly in power and transport industries. Their occurrence in the natural environment is an undesirable phenomenon. The most dangerous heavy metal ions for living organisms are: Cr(II), Hg(II), Cd(II), Pb(II), As(II), etc. Purification of the environment from toxic metals is very important problem. Ion exchangers with sulphur groups on the surface are used for the removal of toxic metals. These groups exhibit great affinity for heavy metal ions, particularly for mercury ions (Dujardin et al., 2000; Podkościelna and Kołodyńska, 2013; Podkościelna et al., 2016).
Ion exchange is a process in which mobile ions of one substance are replaced by similarly charged ions of another substance that are electrostatically bound to the functional groups present in a solid matrix. This process is reversible and stoichiometric. Ion exchangers such as high molecular weight acids or bases, as a result of ion exchange, are converted to their corresponding salts. Depending on the type of the functional group, ion exchangers are called anionic if they exchange negative ions (e.g. CrO4−2, SO4−2, NO3−, Cl−) and cationic if they exchange positive ions (Fe+3, Pb+2, Cd+2, Ca+2, NH4+). Ion exchangers are widely used in laboratories and in the industry. They are used, for example, for softening and demineralization of water for power industry, separation of ions of the valuable metals in metallurgy, catalyzing a chemical processes, the removal of heavy metal ions from aqueous solutions, isolation of antibiotics and the separation of aminoacids in the pharmaceutical industry. Ion exchange is one of the basic chromatographic analytical techniques (ion exchange chromatography) (Wheaton and Anderson, 1958).
In this article synthesis of sulphur-containing microspheres are presented. Copolymerization was performed using 1,1′-bis[4-(2-hydroxy-3-acryloiloxypropoxy)phenyl] cyclohexane (C.DA) (Podkościelny and Bartnicki, 2000), styrene, α,α′-Azoiso-bis-butyronitrile (AIBN) (initiator) and pore forming diluents (toluene and decan-1-ol). In the first stage of modification chloromethyl derivative was obtained with paraformaldehyde in the presence of hydrochloric acid at about 100℃. Then chloromethyl groups reacted with thiourea resulting in the formation of isothiouranium salt, which was hydrolysed with the NaOH solution and acidified with hydrochloric acid. In the final stage, the microspheres containing methylenethiol groups on its surface (–CH2SH) were synthesized. The sulphonic groups (–SO3H) were obtained by the reaction between C.DA and sulphuric acid for 3 h at temperature range of 50–100℃. The course of the modification was controlled by elemental analysis and the spectroscopic methods. This work was first presented at the 15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Experimental
Reagents for copolymerization
Styrene, bis(2-ethylhexyl)sulphosuccinate sodium salt (DAC,BP) and decan-1-ol were from Sigma-Aldrich (Buchs, Switzerland). C.DA was obtained from Department of Polymer Chemistry (UMCS Lublin, Poland). AIBN was obtained from Merck (Darmstadt, Germany). Toluene was from POCh (Gliwice, Poland).
Copolymerization
One hundred fifty millilitres of redistilled water and 1.5 g of bis(2-ethylhexyl)sulphosuccinate sodium salt were stirred for 0.5 h at 80℃ in a three-necked flask fitted with a stirrer, a water condenser and a thermometer. Then, 10 g of C.DA and 2.03 g of styrene were dissolved in a mixture of solvents (10 ml toluene/10 ml decan-1-ol). To the resulting solution 1% AIBN (initiator) was added and then transferred to a flask containing an aqueous phase. Copolymerization was performed for 18 h at 80℃. The obtained copolymers were filtered off, washed with distilled hot water, dried and extracted in a Soxhlet apparatus (firstly with acetone, next with methanol). Chemical structures of monomers used for synthesis of microspheres are shown in Figure 1.
Chemical structures of monomers used for copolymerization: (a) 1,1′-bis[4-(2-hydroxy-3-acryloiloxpropoxy)phenyl]-cyclohexane (C.DA) and (b) styrene (St).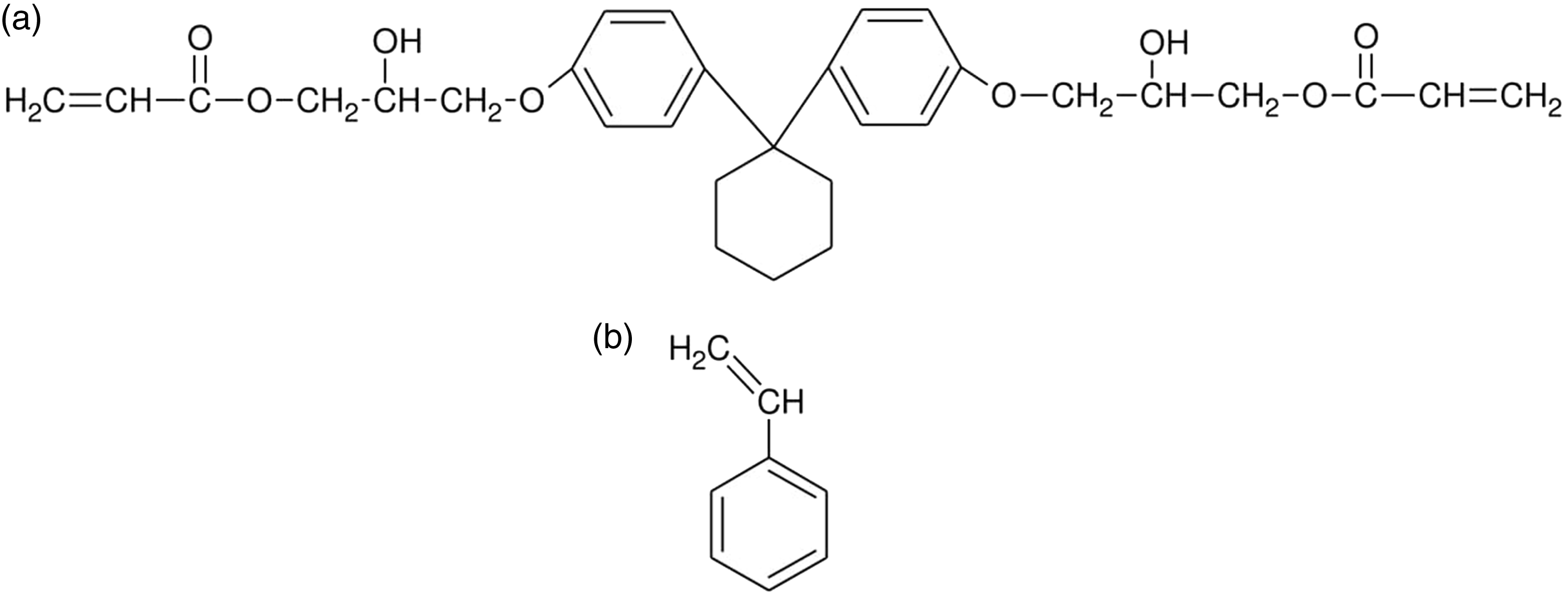
Modifications
Modification I with sulphuric acid
In a 500 ml round-bottomed three-necked flask, equipped with a mechanical stirrer, thermometer and condenser, 50 g of C.DA-St microspheres were placed and then 300 ml of sulphuric acid was added dropwise slowly (for about 1.5 h at 50℃). Then the content of the flask was heated to 100–120℃, this temperature was kept constant for 3 h and after switching off the heating, stirring was continued for 15 h. The obtained modified microspheres with –SO3H groups on the surface were cooled. The sulphuric acid was washed by decantation, and then filtered on a Buchner funnel by washing with distilled water.
Modification II
Step 1: To a 250 ml three-necked flask equipped with mechanical stirrer, thermometer and condenser, 10 g of polymer microspheres C.DA-St, 15 g of paraformaldehyde and 100 ml of concentrated hydrochloric acid were added. The mixture was heated using a heating mantle for 22 h, maintaining the temperature at about 100℃. The obtained modified microspheres with –CH2Cl groups on the surface were cooled, washed with distilled water, filtered off and extracted in a Soxhlet apparatus with boiling acetone and toluene. Next, the product was dried in air.
Step 2: In a 250 ml round-bottomed three-necked flask, equipped with mechanical stirrer, thermometer and condenser, 10 g of chloromethylated microspheres, 15 g of thiourea and 100 ml of 1,4-dioxane were placed. The mixture was heated for 9 h at a temperature of about 100℃. After cooling, the contents of the flask were filtered and washed with hot water to remove excess thiourea. The modified microspheres were placed in a Soxhlet apparatus and washed precisely with methanol by continuous extraction.
Step 3: In a 250 ml round-bottomed three-necked flask, equipped with a mechanical stirrer, thermometer and condenser, 25 g of NaOH dissolved in 75 ml of H2O and then 20 g of microspheres containing thiourea groups on its surface were added. The mixture was heated at 80℃ for 2 h. After cooling the flask, modified microspheres were filtered and washed with distilled water. Then the filtered product was placed in a glass beaker (250 ml) and concentrated hydrochloric acid (40 ml HCl + 50 ml H2O) was added and this mixture was stirred at periodic intervals. After about 0.5 h microspheres were filtered, washed with distilled water and dried. Finally, microspheres with –CH2SH groups on their surface were obtained. Chemical structures of the microspheres obtained by modifications I and II are presented in Figure 2.
Microspheres with –SO3H and –CH2SH groups on their surface.
Characterization
Elemental analysis of the modified microspheres was carried out using the Perkin-Elmer CHNS 2400 apparatus and elemental analyser Vario EL III Elementar (S).
Porous structure of copolymers was investigated by nitrogen adsorption at 77 K using the adsorption analyzer ASAP 2405 (Micrometrics Inc., USA).
Attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR) spectra were obtained using a Bruker FTIR spectrophotometer TENSOR 27 in the Department of Polymer Chemistry UMCS. The spectra were acquired in the spectral region of 400–4000 cm−1.
Thermal analysis of the modified samples was carried out on a STA 449 Jupiter F1, Netzsch (Selb, Germany) under the following operational conditions: heating rate 10℃ min−1, dynamic atmosphere of helium (50 ml min−1) in the temperature range of 30–600℃, sample mass of about 5 mg and sensor thermocouple type S thermogravimetry–differential scanning calorimetry (TG–DSC). The identification of gas composition coming out during decomposition (degradation) was detected and analyzed by quadrupole mass spectrometer QMS 403 C Aëolos (Germany) coupling on-line to STA instrument.
Scanning electon microscopic (SEM) photos of microspheres were obtained by means of an optical microscope Morphologie G3 Malvern (UK).
Results and discussion
The results of elemental analysis of modified microspheres.

The ATR-FTIR analysis of copolymers.

Swellability coefficient results.

Thermal stabilities of all samples.


Thermogravimetric/differential scanning calorimetric/quadrupole mass spectrometric (TG/DTG/DSC) curves.

Mass spectrometric (MS) curves.
In Figure 5 the photos of the microspheres are presented. After modification some partial destruction of the surface of C.DA-St-SO3H is visible.
Scanning electon microscopic (SEM) photos of microspheres.
Conclusion
Polymer microspheres are obtained as a result of the copolymerization reaction of C.DA with styrene. Modifications of the copolymer results in the formation microspheres with –CH2SH and –SO3H groups on their surface; By elemental analysis the percentage elemental composition of the obtained copolymers was determined and it confirmed the correct course of the modification reaction to form a 7.55% (for the –SO3H) and 2.30% sulphur (for –CH2SH), respectively. ATR spectra analysis confirmed the presence of characteristic signals derived from the groups that are included in structure of these compounds in the C.DA-St-CH2SH and C.DA-St-SO3H copolymers. In the next stage the obtained modified microspheres will be tested as polymeric ion exchangers. Based on TG/DTG/DSC/QMS studies it was concluded that the sample C.DA-St have the highest thermal resistance.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: People Programme (Marie Curie Actions) of the European Union's Seventh Framework Programme FP7/2007-2013/ under REA grant agreement n° PIRSES-GA-2013-612484.
