Abstract
Water pollution by organic dyes continues to pose a serious health and environmental threat to the ecosystem. Although adsorption using biopolymer-based hydrogels has proven to be an ideal technique for the treatment of these dye contaminants from aqueous solutions, these hydrogels suffer from lack of mechanical stability and recovery as compared to synthetic polymers. Herein, we review the low-cost synthesis of hydrogel incorporated with inorganic components mainly focusing on strategies to improve the mechanical stability and separation of the hydrogel in removing methylene blue (MB) dye from aqueous solution. The literature shows that hydrogel nanocomposites are a class of materials that have flourished significant consideration, especially concerning water treatment. In adsorption technology, hydrogel nanocomposites act as absorbents, prominent to enhance their removal efficiency towards contaminants. This review highlights the preparation and use of hydrogel nanocomposites as efficient adsorbents. In-depth discussions on adsorption and diverse synthetic routes of hydrogels have been devoted to applications of these nanocomposites and are compared in this contribution to the removal efficiency of MB dye from wastewater.
Introduction
Water is very important for the survival of living things on earth. 1 Despite the need for this resource, water pollution continues to be a problem in most countries including South Africa, where the mainstream water supplies are underground and surface water. 2 Water pollution may be defined as any water that is unsafe for drinking by humans and animals. 2 There are two classes of water contaminants, namely, point sources and non-point sources in which they are defined as a source of pollution at a fixed location (mines, industries, power stations, water treatment station, etc.) and pollution from moving sources (cars, buses and trains), respectively.3,4 In point source, water pollutants may be classified as either inorganic (fertilizers and toxic metals), organic (dyes) or microbial (viruses and bacteria). 5 For example, dyes are organic complexes mostly used by textile industries to give colour to fabrics and contribute largely to pollution. 6 Other applications may include use in medical, pharmaceutical, paper, rubber, plastics, leather, food and cosmetics industries. 7 Dyes contain aromatic rings in their structure and can be made up of either chromophores or auxochromes.7,8 Chromophores are responsible for the production of colour (OH, NH2, NHR, NR2, Cl, COOH and R =alkyl group), and auxochromes (NO2, NO and N = N) improve chromophores, make molecules soluble in water and improve their affinity to bind materials. 8 The discharging of dye effluents into either surface and/or groundwater sources leads to contamination which ultimately results in various health and environmental problems.8,9 Consumption of contaminated water by humans can lead to vomiting, mutation, cancer, breathing problems, diarrhoea, eyes burn, nausea shock, cyanosis, jaundice and tissue necrosis.7,8–10 The environmental issues include the death of aquatic organisms, leading to the development of foul smell,11,12 hence the need to eliminate dyes from waste effluents before discharging them into rivers and other water streams.
Methods of removing dyes from water.

The most used commercial adsorbent is activated carbon (AC) (powder or granular).6,25 The application of AC as an adsorbent is possible due to its high adsorption capacity, high removal efficiency, large surface area and high porosity.
29
It effectively removes various types of organic micropollutants as well as their secondary oxidation products and inorganic compounds.
29
The main disadvantage of this adsorbent, however, is the high cost and its regeneration problems.
6
To reduce the cost of using commercial AC, agricultural by-products including shells and seeds of fruits have been used for production of AC.30,31 For example, a study by Geçgel et al. (2012) generated AC from Pea Shells (
Hydrogels are a class of three-dimensional (3D) soft polymeric materials, which can imbibe a substantial amount of liquids.
41
In this direction, their innate ability to absorb and hold a high volume of liquid qualifies them as ideal materials for the adsorption of aquatic pollutants.
41
Hydrogels attracted a great deal of attention in various fields (gas sensing,
42
drug delivery
43
and cosmetics
44
) owing to their non-toxicity, low cost, permeability and biocompatibility.
10
However, the applicability of hydrogels has been overshadowed due to their weak mechanical properties and partial adsorption capacity.
10
The yearning for enhancing inherent shortcomings exhibited by hydrogels has instigated tremendous research interest in the development of hydrogel nanocomposite, which is a robust process to improve the disadvantages without losing the advantage of hydrogels. In the past decades, the use of hydrogel nanocomposites for the removal of MB dyes in aqueous environment has increased considerably. In the past 10 years, a developing trend of publications in hydrogel nanocomposites for removal of MB dyes is shown in Figure 1. The literature survey statistics show an exponential increase in the number of publications per year considering hydrogel nanocomposites for the removal of MB dye. The total number of publications per year on hydrogel nanocomposites for removal of dyes during the period 2011–2021 (using science direct database). *Data collected in May 2021.
This review summarizes the recent advances and developments of hydrogel nanocomposites as adsorbents for wastewater treatment. This is realized by doing a detailed review of the reactions and mechanisms of adsorption including an overview of the adsorption of MB dye. Then, we introduce hydrogel nanocomposite as an adsorbent for MB removal with great emphasis on the structure and synthetic routes.
Adsorption
Background
The attachment or adherence of molecules on the surface of solid material is known as adsorption. The adsorbent material contains active adsorptive sites on its surface to which molecules (gas or liquid) from the bulk solution (adsorbate solution) bind to.45,46 Adsorption of molecules may occur either physically or chemically. 47 The mass transfer process may occur in the form of migration, diffusion or convection, in which liquid or gaseous molecules are transferred to a solid phase.48,49 Interactions such as Lewis acid-base, van der Waals and Columbic are characteristic of physisorption50,51 whereas chemisorption is distinguished through the development of new adsorbate–adsorbent bonds. 52 The adsorptive sites can have the same or different energy, depending on the nature of the material. 53 Owing to its principle, adsorption has gained interest for application in hydrogen storage, sensing, drug delivery, gas capture and water treatment.54–57
The recent interest in applying adsorption for the treatment of dyes from wastewater was attracted by its low operation costs, easy design, efficiency and fast removal of dye.58,59 Before applying an adsorbent material on an industrial scale (column adsorption), it is first optimized in batch mode experiments; then if it possesses a high removal efficiency, it can be employed for column adsorption studies.60,61 Factors that affect the adsorption process in wastewater treatment include the following55,56,60: 1. The adsorbent and adsorbate charges, 2. the solution pH, 3. temperature, 4. dye concentration, 5. adsorbent dose and 6. the surface area of the solid material.
When an adsorption process reaches equilibrium, it means the active sites of the adsorbent are saturated with adsorbate molecules and no further adsorption can take place. 61
Adsorption parameters
Adsorption isotherms
The isotherms of adsorption are significant for describing the adsorbate–adsorbent interactions. 62 Additionally, they provide information about the mechanisms of adsorption, adsorption capacity and surface properties. This study employs the Langmuir, Freundlich and Temkin isotherm models. Below are their brief descriptions including their linear and non-linear equations.
Langmuir isotherm model
Irving Langmuir
63
established the Langmuir isotherm in 1916. The model theory assumes that the active sites on the solid surface have the same energy, leading to homogeneous adsorption of adsorbate molecules (monolayer coverage).
64
Graphically, adsorption equilibrium is observed by a plateau, which is a point where all active sites are fully occupied and no further adsorption can occur.65,66 The model is expressed as follows, non-linear (equation (1)) and linear (equation (2)) forms

The 
If
Freundlich isotherm
The model assumes multilayer coverage of the adsorbent. The model is expressed by the following equations in a non-linear (equation (4)) and linear (equation (5)) form

Temkin isotherm
The Temkin isotherm theory ignores the concentration values and assumes that the decrease in heat of adsorption concerning the temperature is linear instead of decreasing logarithmically as suggested in the Freundlich equation. The non-linear (equation (6)) and linear (equation (7)) forms of the Temkin model are expressed as follows below
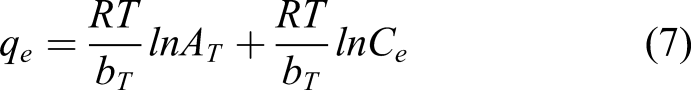
Kinetic isotherms
Adsorption is a process that involves the transfer of mass of a solute from the liquid phase to the surface of the adsorbent.
69
The mechanism of adsorption can be researched using either Lagergren’s pseudo-first-order equation or pseudo-second-order equation. The amount of contaminant adsorbed with time can be analysed provided an appropriate model is used.
70
To identify which model is appropriate, the correlation coefficient (
The non-linear (equation (8)) and linear (equation (9)) equations for the pseudo-first-order kinetic model are
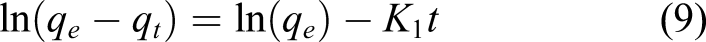
The non-linear and linear equations for the pseudo-second-order kinetic model are

Thermodynamics
During the adsorption process, thermodynamic parameters such as enthalpy (


Adsorbent materials for MB
Although the adsorption technique is effective for dye removal, the type of adsorbent used limits its efficiency. 75 Various materials have been used for MB dye removal. For example, AC is reported to be the most frequently used nano-adsorbent for removing a variety of inorganic and organic contaminants owing to its high surface area, porous structure, thermal stability and amphoteric nature.76,77 However, AC has some drawbacks such as intraparticle resistance in industrial application, high production costs and regeneration costs.77–79 Recent research studies have attempted to find ways to improve the regeneration of AC and produce it at a lower cost. 77 On the other hand, alternative adsorbents such as fly ash, graphene, clay, carbon nanotubes and polysaccharides were reported for removal of MB.80–87 Among these adsorbents, Mittal et al. 80 reported that using polysaccharides-based adsorbents is a promising strategy for MB dye removal from aqueous solutions. This is due to their enhanced adsorption capacity amongst other attractive properties.
Polysaccharides
Polysaccharides can be defined as highly hydrophilic and non-toxic natural polymers made up of multiple small units of saccharides that are connected through glycosidic bonds.83–85 These biopolymers are found and used in plants or animals for structural support and energy storage. 86 Examples of polysaccharides include carboxymethyl cellulose (CMC), alginate, carrageenan, chitosan, guar gum, starch and locust bean gum.85–87 These natural polymers offer the advantages of high absorption capacity, biodegradability and cheap synthesis. 88 However, due to low solubility and other physical drawbacks, synthetic polymers are incorporated into the biopolymeric backbone to improve their properties. 89 Polysaccharides-based materials have recently attracted use for adsorption of dyes and toxic metals because of their non-toxic nature, biodegradability, easy availability and low-cost synthesis. 90 Amongst other polysaccharides, CMC and alginate offer more attractive properties for application as dye adsorbents.
Carboxymethyl cellulose
Cellulose is the main component of most plants. 6 Carboxymethyl cellulose is a highly reactive, hydrophilic, water-dissolving derivative of cellulose with many carboxyl groups on its surface. 82 Commercially it can be obtained as sodium CMC with different degrees of saturation (DS), which may range from 0.6–0.95 depending on how it was prepared, and the solvent used.91,92 Carboxymethyl cellulose is mostly used in paper, packaging, textile, food, cosmetics and pharmaceuticals.85,93 Due to its sensitivity to pH, non-toxic nature, hydrophilicity, low cost and ability to form gels, CMC has been applied on its own or in a composite form as an adsorbent for removing inorganic and organic contaminants.92,94
Carboxymethyl cellulose (Figure 2) is a cellulose derivative with carboxymethyl groups at carbon 2, 3 and 6.
95
In a study conducted by Kono,
96
the resonances of CMC were determined using carbon-13 nuclear magnetic resonance (13C NMR) as shown in Figure 3. In the spectrum of CMC, the carbonyl carbon (C1, C4), overlap of C2, C3, C5, the carboxymethyl groups, and C6 were assigned to broad 13C resonances at 178, 104, 83, 75 and 63 ppm.
96
The existence of carboxylic groups (COO−) on the polymer chains of CMC allows for interactions with multivalent metal cations (Al3+ and Fe3+) which results in the formation of stabilized ionotropic hydrogels. Interactions with metal ions through the hydroxyl groups of the CMC may improve the water insolubility and stability of the polymer aggregates.95,96 Structure of sodium carboxymethyl cellulose.
63
13C NMR of CMC.
75
CMC: carboxymethyl cellulose.

The reaction of cellulose with monochloroacetic acid (MCA) or its sodium salt in the presence of an organic solvent under basic conditions produces NaCMC. 97 There are various materials from which CMC can be derived; for example, Petri 98 prepared CMC from pineapple peels, and in another study, Begum et al. 99 prepared CMC from sugarcane bagasse. The synthesis of CMC occurs in three steps: (1) alkalization, (2) carboxymethylation and (3) neutralization as outlined in the steps below.
Cell-OH + NaOH
Cell-OH.NaOH + ClCH2COONa
NaOH+ Cl−CH2COONa During the synthesis process, carboxymethyl groups replace hydroxyl groups at positions 2, 3 and 6 of cellulose. In the synthesis of CMC, it is important to consider the degree of substitution, which will govern the porosity, and adsorption capacity of CMC.
99
Degree of Substitution (DS) refers to the number of hydroxyl groups replaced by carboxymethyl groups. Each β-glucopyranose unit of cellulose has three hydroxyl groups; therefore, hypothetically DS value will range between 0 and 3.0.
100
Various factors may affect the DS of CMC such as the solvent, temperature and the NaOH concentration.
101
For example, most researchers report that the use of isopropanol as the solvent yields CMC with very high DS.
101
Huang et al.
102
synthesized CMC from the pulp of six different plants; the conditions that yielded the best CMC were 20% NaOH concentration, the temperature of 60°C and 3–5 g of MCA. The group reported that CMC yield increased with increasing MCA content. The degree of saturation influences the application of CMC.
102
The hydrophilicity of CMC increases with an increase in DS. When DS > 0.4 (higher), CMC becomes soluble in water, has better viscosity, enhanced cationic exchangeability and improved covalent crosslinking through radiation. Whereas when DS < 0.4 (lower), CMC is insoluble, and it swells in solution.
102
For example, a study reported the degradation of a polymer with irradiation of 10% CMC (0.7 DS), whereas irradiation of CMC of 1.32 DS resulted in outstanding crosslinking and high gel-fraction.
103
For adsorption, the properties of CMC can be modified by crosslinking to form a gel, which will improve the solubility and hydrophilicity of CMC.
Alginate
Alginate is a sentimental term used in the dietetic, cosmetic, pharmaceutical and biotechnological industries. 104 The alginate can be found in salts of magnesium (Mg), potassium (K), calcium (Ca) and sodium (Na). 104 E.C.C Stanford who was a pharmacist 105 discovered alginates in 1929. SA is found in brown algae, marine algae and produced by some bacteria, hence abundant in nature.106,107 Applications of sodium alginate include drug delivery, thicker, gel-forming agent, binder during the production of tablets, use as a stabilizer and mixing agent.105–108 The recent use of polysaccharides as adsorbents for dyes and other pollutants is mostly due to the attractive properties that polysaccharides possess, such as high hydrophilic structures, their non-toxic nature, biodegradability and cheap synthesis.109–111
Sodium alginate (SA, Figure 4) is made up of poly- Structure of sodium alginate.
62

Hydrogels
Background
Hydrogels are hydrophilic polymer chains that are cross-linked to form gel structures that swell in aqueous solution and trap fluids for a long period without dissolving.83,84 Hydrogels may contain carboxylic, amine, imide, hydroxyl and sulfonyl groups in their 3D structure that are responsible for the hydrophilicity and swelling capacity.
118
Depending on the nature of the hydrogel, it can be classified based on various properties. Classification of hydrogels (Figure 5) can be based on whether they are synthetic (involves the use of synthetic monomers), natural (involves using biopolymers) or a combination of synthetic and natural monomers resulting in a hybrid hydrogel.
119
The polymeric composite classification can be based on the method used to synthesize the hydrogel
119
: a. Homopolymeric hydrogels: they are hydrogels consisting of the same type of monomer. b. Copolymeric hydrogels: these hydrogels comprise two or more different kinds of monomers such that the network would have at least one hydrophilic component on the polymer network chain. c. Multipolymer interpenetrating polymeric hydrogel (IPN): the hydrogel network consists of two components (natural and/or a synthetic polymer) that are independently cross-linked. Classification of hydrogels.

It was also demonstrated that hydrogels can be categorized based on whether they are
120
a. crystalline, b. amorphous or c. semi-crystalline: showing properties of both crystalline and amorphous phases.
The other classification is based on whether the crosslinking of hydrogels occurs via chemical or physical means. Briefly, a. physical crosslinking in hydrogels may be through (1) formation of a hydrogen bond, (2) hydrophobic interactions between chains, in which the dissipation energy of the bond prevents breakage and improves the hydrogel strength, and (3) crystallization where gels are subjected to freeze and thaw process in PVA/PVP solution 10 h at 15°C and then placed at room temperature for 2 h. Lastly (4) between oppositely charged groups.
84
b. Chemical crosslinking can be achieved by (1) using aldehydes such as acetaldehyde, glutaraldehyde (GA) and formaldehyde and (2) radiation using an electron beam, gamma rays or ultraviolet rays at room temperature in which a free radical is formed, monomers are added to the chain for growth and the gel forms at the critical gelling point. Lastly (3) free-radical polymerization using a cross-linker, for example, MBA. In this method, an initiator such as potassium persulfate (KPS) generates free radicals, the radicals react with other monomers and then MBA is added to the polymer chain to form the hydrogel.84,121
In addition, the hydrogels may be classified based on the charge on their cross-linked polymer network.122,123 The charge may be a. ionic (cationic/anionic), b. non-ionic (neutral), c. amphoteric (comprising both basic and acidic groups) and d. zwitterionic (contains anionic and cationic components on each repeating structural unit). The net charge of the gel is zero.
Physical and chemical stimuli classifications.

Application of hydrogels
Hydrogels have gained much attention for use in various applications owing to their outstanding qualities; these include use in the making of disposable diapers, absorbent pads, use in hydrogen storage, sensing, CO2 capture, biomedical field (drug and cell delivery systems), immobilization of enzymes, wastewater treatment and agriculture for trapping water.11,48,49 The ideal properties of a hydrogel depend on its specific application. For application in water treatment, the hydrogel must have the following properties below
98
: 1. High absorption capacity; 2. low residue monomer and soluble content; 3. cost-effective; 4. reusable; 5. high biodegradability and not produce harmful by-products; 6. must retain neutral pH after swelling in water and 7. easy recoverability.
The swelling capacity of hydrogels is indicative of their absorptive capacity which may be affected by factors such as the intermolecular spaces in the 3D network, the existence of hydrophilic groups on the hydrogel polymer backbone and the pore size of the hydrogel surface. 10 Hence, parameters such as the amount of initiator, cross-linker, polymer, monomer and solvent volume are optimized during the preparation of the hydrogel to obtain optimum conditions for preparing an ideal hydrogel. 122
Preparation methods
As stated previously, biopolymers suffer poor mechanical, thermal stability and they dissolve in water. 89 Most researchers have reported that crosslinking, grafting and modification with inorganic constituents such as metal oxides and clay improves solubility and stability problems associated with biopolymers.3,121 The type of method used to prepare the hydrogel, therefore, affects its structural make-up or physical properties. Amongst several physical and chemical techniques used for hydrogel synthesis, the most widely used methods are discussed below.
Grafting
Grafting is the modification of the polymer backbone using synthetic polymers such as acrylamide, acrylic acid, methacrylamide and vinyl alcohol as support materials. 123 During grafting, an initiator creates a free radical site, and then a monomer unit is added to the generated free radical site. 124 Grafting can occur via either a chemical or radiation. Chemical grafting involves using chemical reagents such as ammonium persulfate, KPS or other chemical initiators. 125 Radiation grafting involves initiating free radicals using UV visible or microwave radiation. 126
It was shown in the literature that using a microwave radiation method for synthesizing hydrogels produces sterilized hydrogels.
126
Naturally, polysaccharides suffer from poor chemical and physical stability.
89
Grafting has been reported to solve these issues including enhancing the performance of hydrogels through the introduction of new functionalities from grafted monomers.
127
For example, in a study conducted by Tally and Atassi,
86
it could be observed from the thermogravimetric analysis (TGA) curves of (a) SA and (b) SA-g-P (AA-co-AM)/PVP semi-IPN SAP (Figure 6) that grafting with synthetic polymers enhanced the thermal stability of hydrogels by more than 20% weight loss at above 200°C. Thermogravimetric analysis of (a) SA and (b) SA-g-P (AA-co-AM)/PVP semi-IPN SAP.
62
SA: sodium alginate; PVP: polyvinylpyrrolidone; SAP: semi-IPN.
A study of the modification of CMC by grafting with poly (3-sulfopropyl methacrylate), P (SPMA), in the presence of KPS initiator for MB removal was carried out.
93
The main vibrational peaks at C =O (1718 cm−1), S =O (1085 cm−1) and S-O (626 cm−1) were observed in Figure 7(a) of the Fourier transform infrared (FTIR) of the CMC-g-P (SPMA) hydrogel.
93
The scanning electron microscopy (SEM) image showed the well-defined pores and 3D interconnections of CMC-g-P (SPMA) hydrogel, for which the hydrogel could allow easy penetration of dye molecules in solution through its pores as observed in Figure 7(b).
93
The adsorption of MB by the hydrogel was characterized by an absorption peak at 664 nm corresponding to the characteristic absorption band of MB (Figure 7(c)). From the X-ray diffraction (XRD) (Figure 7(d)), the partially crystalline structure of CMC was characterized by sharp peaks at 2θ = 9.4 and 20.1° resulting from the intermolecular and intramolecular hydrogen bonding between carboxylic groups and hydroxyl groups of the biopolymer. Upon grafting on the CMC-g-P (SPMA), it could be observed that the peaks were broader, indicating the hydrogen bonds were destructed, allowing grafting to take place.
93
FTIR (a), SEM (b), UV-vis (c) and XRD (d) of CMC-g-P (SPMA).
68
FTIR: Fourier transform infrared; SEM: scanning electron microscopy; P (SPMA): poly (3-sulfopropyl methacrylate); CMS: carboxymethyl cellulose; XRD: X-ray diffraction.
Crosslinking
The process of crosslinking can occur through physical or chemical interactions. Physical interactions are irreversible and include electrostatic interactions, hydrogen bonding and the van der Waals forces.
128
Chemical crosslinking involves forming irreversible covalent bonds in the hydrogel polymer chain usually through the reaction of complementary groups.
129
Chemically cross-linked hydrogels are mostly used in medical applications. For example, Liu et al.
130
synthesized chitosan-based hydrogels from PEG for application in drug delivery. In another study by Kumar et al.,
131
cross-linked hydrogels were produced from GA as a covalent cross-linker (Scheme 1). Crosslinking using glutaraldehyde to produce hydrogel.
109

The most commonly used chemical cross-linkers include ethylene glycol dimethyl-acrylate, N, N-methylene-bis-acrylamide (MBA), tetra-ethylene glycol dimethyl-acrylate (EGDMA) or tri-propyleneglycol diacrylate (TPGDA).
132
However, these cross-linkers are usually toxic and result in non-biodegradable hydrogels.
133
In addition, because of the lack of internal structural homogeneity and an effective mechanism for energy dissipation, chemically cross-linked hydrogels are very brittle.
134
This prompted many researchers to consider introducing nanocomposites, ionic interactions, hydrogen bonding and hybrid hydrogel systems to improve the hydrogel properties.135–137 A hybrid system consists of physical interactions that will aid in energy dissipation and chemical interaction, which will mainly improve structural properties.
138
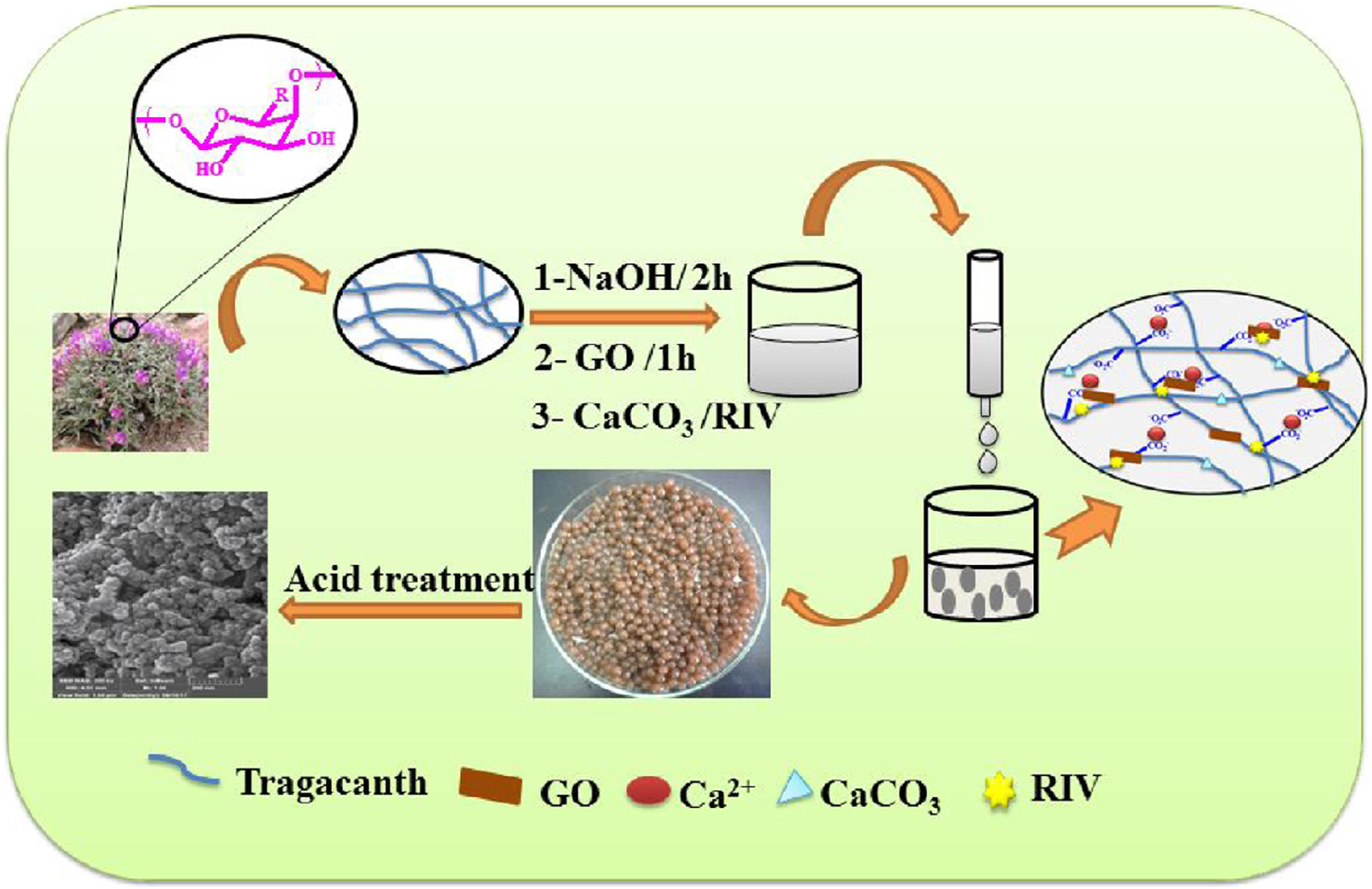
Figure 8 illustrates the preparation of hydrogel beads through ionic interactions by Rahmani et al.
139
Briefly, a mixture containing gum tragacanth (GT), graphene oxide (GO) and calcium carbonate (CaCO3) was added dropwise into a concentrated solution of Ca2+ which interacted with -OH and COO− groups on the GT and GO to form hydrogel beads. Ionic crosslinking offers the advantage of less toxicity.
133
Scheme showing the preparation of gum tragacanth–based hydrogel beads.
117
In a study by Eftekhari-sis et al.,
140
hybrid hydrogels constructed from poly (N-isopropyl acrylamide-co-itaconic acid) and non-toxic octa-vinyl polyhedral oligomeric silsesquioxane (OV-POSS) cross-linker were prepared. Their obtained SEM (Figure 9), XRD and TGA (Figure 10) results are discussed. The rough non-homogeneous surface was observed on the SEM image (a) of poly (NIPAM-co-IA). Upon hybridization with 8% POSS (b), 12% POSS (c) and 12% POSS (d), the morphology changed to a honey-comb like structure with homogeneous pores of varying pore sizes as POSS content was increased. The study reported that at 12% POSS the honey-comb pattern was disrupted, indicating that the degree of uniformity in the hydrogel hybrid can be controlled by changing the crosslinking content.
140
SEM images of poly (NIPAM-co-IA) hydrogel (a) and hybrid poly (NIPAM-co-IA)/OV-POSS (8%, 10% and 12% POSS) (b–d).
118
SEM: scanning electron microscopy; OV-POSS: octa-vinyl polyhedral oligomeric silsesquioxane. X-ray diffraction and thermogravimetric analysis.
118


For example, the XRD patterns in Figure 10(a) showed a successful hybridization and a uniform dispersion of the cross-linker as confirmed by the disappearance of characteristic crystalline diffraction peaks of OV-POSS at 2θ = 9.8, 23.1 and 23.9°. 140 The TGA thermogram (Figure 10(b)) showed that the crosslinking with POSS in the hybrid hydrogels increased the thermal stability, wherein the weight loss at temperatures 340–500°C was less than weight loss obtained for poly (NIPAM-co-IA) at the same temperature. 140 From the obtained literature, it could be observed that the chemical and physical properties of the hydrogels were improved, including creating porous structures that enable penetration of adsorbate molecules.
Free-radical polymerization
Free-radical polymerization is a technique in which polymerization is achieved by generating free radicals to which chain growth occurs in succession.
141
This method involves a combination of grafting and crosslinking. The decomposition of the initiator may occur using temperature, light or photon, which then forms a free radical.
142
Decomposition by photon offers advantages such as low costs; it allows better spatial and time-based control of the reaction process and does not use chemical solvent.
143
Scheme 2 shows a schematic representation of an overview of free-radical polymerization for synthesizing hydrogels. Firstly, there is the generation of free radicals by an initiator (initiation). Secondly, the free radicals react with the monomer to generate vacant active sites (propagation) and lastly, the formation of a polymer network through crosslinking (termination).
144
An overview of thermal free-radical polymerization and crosslinking.
122

Free-radical polymerization is the most frequently used procedure in the polymer industry. 1 The advantages of using free-radical polymerization are that it is easy to carry out, convenient, it is not easily affected by impurities and it is suitable for designing and preparing polymers that can be used for a variety of applications. Free-radical polymerization allows well-characterized reaction kinetics and achievement of in situ properties. 145
Modification with inorganic materials
Depending on the intended application of the hydrogels, their physical properties can be further enhanced through the incorporation of inorganic materials. For example, for use in removing contaminants from aqueous solutions, the hydrogel must be mechanically and thermally stable for application especially for removing effluents from industries that utilize water for cooling reactions,146,147 wherein the contaminants may be introduced at that point. In addition, the hydrogels must be easy to recover. Many recent studies have incorporated nanofillers such as clay-based materials, carbon-based materials and metal oxides into the hydrogel matrix during the polymerization process to improve stability and recoverability. The blended product is then called a hydrogel nanocomposite.
Carbon-based
Carbon-based hydrogel nanocomposites are hydrogels synthesized by incorporating materials such as graphene oxide (GO), biochar, AC and carbon nanotubes.148,149 GO is a carbon material prepared by oxidizing graphene through chemical or thermal reduction processes. It contains highly hydrophilic groups such as hydroxyl, carboxylic and epoxy groups that are essential for adsorbing dyes and toxic metals. 150 GO may interact with contaminants through pi-to-pi interactions, electrostatic interactions or hydrogen bonding. 151 Owing to its outstanding mechanical, electrical and thermal properties, GO has attracted its use in the biomedical, energy and environmental field. 152
Carbon nanotubes are simply graphene sheets rolled up in cylinders of 1 nm in diameter.
153
As a result of their porous structure, large surface area, high tensile strength (0.15 TPa) and elastic modulus (0.91 TPa), CNTs have attracted interests in use for adsorption of pollutants such as dyes, dichlorobenzene, ethylbenzene and some heavy metals.154,155 CNTs can be categorized into two forms, namely, single-walled carbon nanotubes (SWCNTs), which are made up of single layers of graphene sheets, and multi-walled carbon nanotubes (MWCNTs), which are made up of multiple layers of concentric cylinders.153,156 For example, the incorporation of CNTs in gels improves the mechanical properties; however, the rate of degradation decreases. This is because carbon-based materials have high hydrothermal stability, which makes them resistant to harsh environments.
154
Scheme 3 shows various interactions or applications that carbon-based materials can take part in such as surface complexation, physical adsorption through its surfaces and electrostatic interactions.
157
Amongst recent studies that have used carbon-based materials to modify properties of the hydrogel, the incorporation of MWCNTs into XG/PAA hydrogel improved the surface hydrophilicity and specific area of xanthan gum.
10
Makhado et al.
119
conducted studies on xanthan gum polymer cross-linked with polyacrylic acid and incorporated with reduced graphene oxide (rGO); the successful synthesis was confirmed by FTIR. Various carbon-based nanocomposite hydrogels and their adsorption properties for removing contaminants from aqueous solutions are listed in Table 3. Showing possible interactions of carbon-based materials.
135
Carbon-based adsorbent hydrogels and their adsorption properties in removing contaminants from aqueous solution.

Clay-based hydrogels
Clay-based adsorbent hydrogels and their adsorption properties in removing contaminants from aqueous solution.


‘Properties of cellulose−clay nanocomposite hydrogels. (a) Time dependence of storage modulus (G′) and loss modulus (G″) for different gelation systems at 60°C. The data are shifted along the
Metal oxide–based hydrogels
Metal oxide–based nanoparticles have been reported to have a high density and restricted size, which are responsible for their very interesting and unique chemical and physical properties.170,171 Examples of metal oxides include titanium dioxide (TiO2), iron oxide (Fe3O4), magnesium oxide (MgO) and aluminium oxide (Al2O3). Owing to their non-toxic nature, high surface area, high chemical stability and economical friendliness, Fe3O4 nanoparticles are widely used for the removal of toxic metals and organic pollutants from water.170,171,172,173 Ion-oxide and zinc oxide nanoparticles are some of the most frequently used metal oxides in treating dyes from aqueous solutions. 174
Magnetic (Fe3O4) nanocomposite hydrogels
Magnetite (Fe3O4) also known as black iron oxide amongst other transition metals has the strongest magnetism and is stable at ambient temperatures.
175
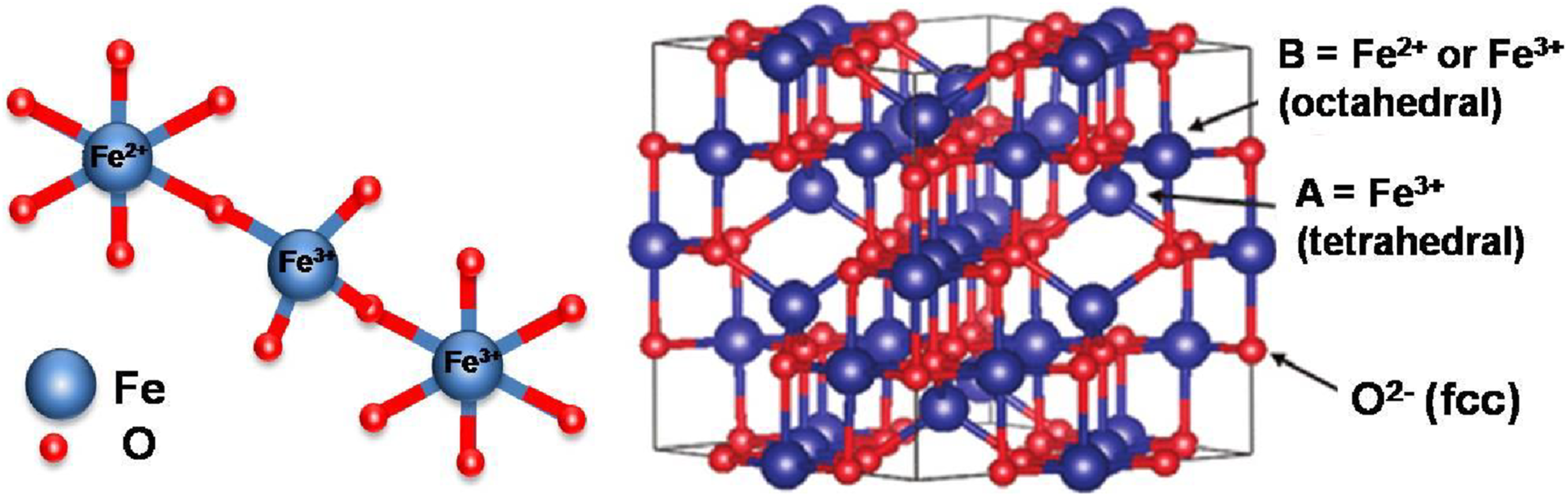
Magnetite is prepared from the co-precipitation of iron oxide salts that result in an inverse spinel crystal structure consisting of half of the Fe3+ in tetrahedral coordination and the other half Fe2+ ions in octahedral coordination (Figure 12).176,177 The co-precipitation method is the most useful and suitable technique in preparing magnetite at both lab-scale and industrial scale.
178
The inverse spinel structure of magnetite Fe3O4.
155
Metal oxide–based adsorbents for organic dyes removal.

Zinc oxide–based hydrogels
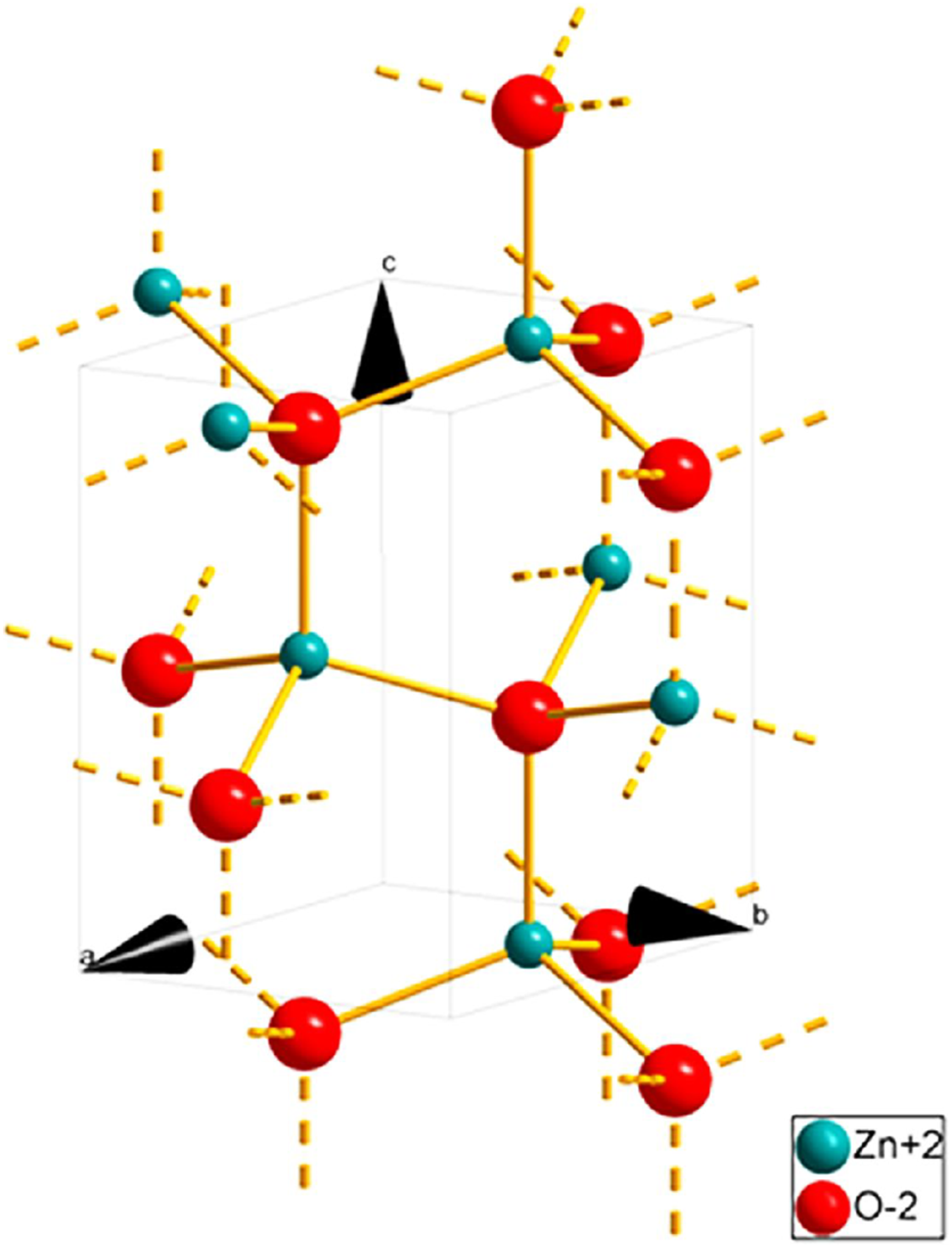
Zinc oxide (ZnO) can be found in nature as a zincite mineral; however, the majority of it is prepared synthetically. Its crystal structure can be found in a hexagonal wurtzite form or cubic zinc blende form (Figure 13). The form that is most stable and commonly found at ambient temperature is the wurtzite structure.
188
ZnO is commonly used for treating skin-related problems such as nappy rash, dandruff and incorporation in ointments used in wound dressing.
188
Other applications of ZnO include use in catalysis, batteries, sensors and adsorption of contaminants.189–192 Khan et al.
193
reported their application as adsorbent material in a study, where a guar gum adsorbent hydrogel incorporated with ZnO nanoparticles was used for removing chromium (VI) from water. The group reported that incorporating ZnO nanoparticles improved the recovery of the adsorbent from the aqueous solution after the removal of Cr (VI). In another study, CMC hydrogel was modified with ZnO for antimicrobial activity, which was influenced by their inexpensiveness and lacks colour.
194
Therefore, looking at these properties, the ZnO would be ideal for preparing hydrogels for adsorbing dyes. Crystal structure of zinc oxide.
162
Titanium dioxide–based hydrogels
Titanium dioxide is a naturally occurring oxide of titanium. It exists in three crystalline phases, namely, brookite, anatase and rutile. 195 The rutile phase is thermodynamically stable compared to the anatase and brookite phases, which are metastable. 195 The advantages of TiO2 are that it is non-toxic, chemically stable, highly reactive, biocompatible, cost-effective, contains electrochemical properties and is safe to produce.195,196 In addition, TiO2 possesses antimicrobial and UV-protection properties. 196 Applications of titanium dioxide include the use of a photocatalyst for water-dye degradation, in food and pharmaceuticals as a white colourant, in sensors, ceramic industries, inorganic membranes and biological implants.195,196 For example in a study by Kangwansupamonkon et al., 197 TiO2/poly [acrylamide-co-(acrylic acid)] hydrogel was reported to have 91.6% removal for MB using 5% TiO2 as an inorganic initiator. Another study by Ari et al. 195 used P (AMPS)-TiO2 composite hydrogel for photocatalytic degradation of MB and reported high catalytic performance and high thermal stability due to the incorporated TiO2 nanoparticles. The group also reported over 80% removal capacity for MB after repeated cycles of more than five, hence improved reusability and regeneration. Another study by Han et al. 198 also reported excellent performance in terms of regeneration, self-cleaning and recovery of the prepared hydroxyethyl-titanium dioxide-CMC (HEC-TiO2-CMC) hydrogel cage for the removal of MB. 198 Adsorption of MB onto polyacrylamide-grafted gum ghatti (PAAm-g-Gg)/TiO2 hydrogel nanocomposite prepared by Mittal and Ray was reported to be greatly dependent on the adsorbent dose, the ionic strength, pH and temperature of the solution. 199 The pair reported a removal capacity of 1305.5 mg g−1 dominated by pseudo-second-order rate model and Langmuir adsorption isotherm. 199 The incorporation of TiO2 NPs onto sodium alginate cross-linked polyacrylic acid (SA-cl-poly (AA) hydrogel) was reported to have improved removal capacity from 80% using hydrogel with no TiO2 to 94.1% and 99.4% for nanocomposites containing 0.05 and 0.2 g of TiO2 NPs, respectively. 200 XRD patterns from this study showed the disappearance of some TiO2 peaks after MB absorption indicating a reduction in the crystallinity of the nanocomposite that is said to be caused by the trapping of dye molecules within the micropores and macropores of the absorbent. 200
Advantages and disadvantages of several hydrogel nanocomposites for the removal of methylene blue from aqueous solution.

Conclusions
This review discusses recent studies applied for removing MB from aqueous solution using biopolymer-based hydrogel nanocomposites. Owing to various advantages such as low cost and easy design, the adsorption method is recognized as the most promising treatment technique for the removal of MB dye. This work discussed the principle behind the adsorption method, common adsorbent materials used and its advantages. We also briefly explained the hydrogels and methods to improve their properties. Three isothermal models: Langmuir, Freundlich and Temkin models have been thoroughly explained with their respective principles. From this review, it was observed that the preparation of hydrogels can be affected by various factors and its removal capacity is affected by pH, temperature and dye concentration. This contribution emphasized the importance of incorporating metal oxide nanoparticles in hydrogels for the removal of dyes and also their advantages and disadvantages towards the gel structure and properties. Concerning the research up to date, it has been observed that the realization of hydrogels for practical purposes needs more research, especially the preparation of hydrogel nanocomposites with the ability to remove organic contaminants such as dyes. It was seen that both magnetic nanoparticles and ZnO nanoparticles have contributed greatly to improving stability and enabling easy separation of adsorbent materials after application. Although the actual wastewaters are complex to be treated because they possess various ions and pollutants, it is more pivotal to conduct analysis on wastewater for adsorption studies using the actual industrial water samples. This is mainly because the adsorbent performance may be noticeably different from the one carried out in batch mode in the laboratory.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors immensely acknowledge the financial support from the National Research Foundation (NRF) (Grant Nos. 116679, 117727, and 118113), Sasol Foundation and University of Limpopo, South Africa.
