Abstract
This work explores the kinetics, equilibrium and thermodynamics of peroxidase adsorption onto spherical guar gum–alginate matrices. The effect of contact time, solution pH, initial protein concentration and temperature was studied in batch experiments. The results show that peroxidase adsorption increased with rising contact time and initial enzyme concentration, and was higher at pH 4.0. The kinetic processes can be predicted by both the pseudo-first-order rate kinetics and the pseudo-second-order rate kinetics. Equilibrium adsorption data were analyzed with different isotherm models. The experimental data fitted to the Freundlich model in agreement with the low energy activation, demonstrating the presence of a high physical and unspecific interaction between the enzyme and the matrix.
Introduction
Adsorption is defined as the ability of some materials (biomass, biological material, polymers, inorganic solids, etc.) to bind solutes present in aqueous solution. Adsorption is a complex physicochemical process, which includes many steps such as diffusion of the solute through the boundary layer, intra-particle diffusion and adsorption of the solute on the sorbent surface. There are countless studies about using different biomass wastes from vegetal or microbiological origin, as substitutes to the expensive and more conventional use of activated carbon, in the adsorption mechanism to remove heavy metal and different toxic components from liquid wastes (Dotto et al., 2012; Pardo et al., 2003; Rangabhashiyam et al., 2014). At present adsorption has emerged as an eco-friendly alternative technology to remove several molecules from aqueous solutions (Zhang et al., 2013). This technology has numerous advantages, such as, its simplicity, being easy to do, using environmentally friendly components and having non-expensive preparation methods of the adsorbent (Gadd, 2009). Much attention has been given to the obtainment of non-soluble matrices formed by polysaccharides and natural polyelectrolytes such as alginate, chitosan, carrageenan, etc. (Gotoh et al., 2004; Kulkarni et al., 2000; Roy et al., 2005; Somers et al., 1993; Spelzini et al., 2011; Woitovich Valetti and Picó, 2016).
Sodium alginate (Alg) is a particularly attractive biopolymer that has a wide range of applications, particularly, in the biomedicine, industrial, food and pharmaceutical fields, because of its capacity to hold water, form gels and form stable emulsions. Alg is a water-soluble linear, polysaccharide composed of 1,4-linked β-D-mannuronicand α-L-glucuronic acid residues, which are found in varying composition and sequence (Lee and Mooney, 2012). This polymer has the advantage of being of natural origin, having a friendly behaviour when it is discarded in the environment and being low-priced. The gelation of Alg can be carried out under an extremely mild environment using non-toxic reactants. The most important property of Alg is its ability to form gels by reaction with divalent cations. Alg beads can be prepared by extruding a solution of Alg as droplets into a divalent cations solution such as Ca2+ or Ba2+ (Paques et al., 2014). There are a great number of papers where Alg is transformed into a non-soluble matrix adding Ca2+ to the medium (Daly and Knorr, 1988; Spelzini et al., 2011). However, the working pH range of the matrix is limited and when Ca2+ is lost by the matrix, the Alg
Guar gum (GG) is a non-ionic polysaccharide found abundantly in nature and has many properties desirable for drug delivery applications. However, due to its high swelling characteristics in aqueous solution, the use of GG as delivery carriers is limited. GG is a water soluble polysaccharide derived from the seeds of
Alg–GG beads are not very stable, but the cross-linked Alg–GG matrix showed stability over a wide pH range and also in the presence of phosphate or Ca2+ chelator (Roy et al., 2005).
The goal of this work was to analyse the molecular mechanism of adsorption of a model protein as peroxidase onto a matrix formed by Alg and GG cross-linked with epichlorohydrin to provide stability. To achieve this goal, we have determined the kinetics mechanism and isotherms of adsorption together with thermodynamic variables.
Materials and methods
Chemical
Alginic acid sodium salt (Alg), GG, epichlorohydrin and peroxidase from
Preparation of Alg–GG beads
One hundred twenty-five milligrams of GG were dissolved in 25 mL of distilled water. Alg–GG beads with distinct Alg to GG percentage weight ratios were prepared (0.6:0.5, 1.0:0.5, and 1.5:0.5). Alg was dissolved in the GG solution prepared as above, and left stirring until complete dissolution. Beads were formed by dropping this solution through a syringe into a 50 mL of 0.1 M CaCl2 solution according to the method previously reported (Roy et al., 2005). The beads were maintained in stirring overnight and finally incubated in 6 mM CaCl2 solution at 5℃.
Preparation of non-soluble Alg–GG beads
Cross-linked Alg–GG particles were prepared by adapting the procedure of Roy et al. (2005). Alg–GG beads (5 g) were transferred to absolute ethanol containing 0.1 M CaCl2 (25 mL) and incubated for 30 min at 45℃. To this 1.5 mL or 3 mL of epichlorohydrin was added in small aliquots and were kept for 10 min stirring constantly. To this 4.2 mL and 8.4 mL, respectively, of 5 N NaOH were added and kept overnight at 30℃. Finally, acetic acid was added to the system until the pH became neutral. The cross-linked Alg–GG beads thus formed were washed with 30 mL of a 3:1 mixture (v/v) of absolute ethanol and water, followed by 20 mL of 99% ethanol. The matrix was finally re-suspended in distilled water and equilibrated with the work buffer before use.
To check the intersection of matrices we combine a certain amount of 0.1 M EDTA, which binds strongly to Ca2+. EDTA forms complex with the Ca2+ present in the sample, and if the cross-linking was not successful the cation loss leads to the dissolution of the beads.
Measurement of enzymatic activity of POD
POD activity was spectrophotometrically monitored by following the oxidation of pyrogallol to purpurogallin. The reaction mixture is: 300 µL of pyrogallol 21.32% (w/v), 200 µL of H2O2 8 Vol, 2.40 mL 100 mM phosphate buffer pH 6.0 and sufficient enzyme to give a considerable change in the 420 nm absorbance between 0 and 90 seconds. The slope of the initial linear portion of the absorbance vs. time curve is proportional to the POD activity. Measurements were taken every 0.1 s and the solution remained under continuous agitation during the measurements. The activity of POD was calculated by the following equation:

Determination of total protein concentration
It was carried out using the Bicinchoninic assay. A fresh standard working reagent (SWR) was prepared mixing 98 Vol of reagent A (Bicinchoninic acid solution purchased from Sigma Aldrich) and 2 Vol of reagent B (CuSO4 solution 4% w/v). A volume of 10 µL of protein solution (maximum concentration of 1 mg/mL) was added to 1 mL of SWR. The tubes were incubated at 37℃ for 30 min. After leaving them to cool down at room temperature, the absorbance was measured at 562 nm using a cell with a 1 cm path length. The calibration curve was performed using dilutions of a standard solution of BSA 1 mg/mL.
Determination of adsorption conditions
The batch experiments for the adsorption kinetics of commercial POD onto cross-linked Alg–GG beads were carried out measuring the enzymatic activity of free POD in the supernatant over time. The mixtures were prepared with constant activity of POD in 25 mM sodium acetate buffer at different pH (4.0, 5.0 and 5.5) and stirring constantly at 20 rpm until the adsorption equilibrium was reached.
Adsorption kinetics
To analyze the adsorption kinetic mechanism the adsorbed POD amounts vs. time curves were fitted with two models, namely pseudo-first- and pseudo-second-order. The kinetic adsorption was assayed at two temperatures and at three different initial concentrations of enzyme. The mixtures were prepared with constant activity of POD in 25 mM sodium acetate buffer, pH 4.0 and stirring constantly at 20 rpm until the adsorption equilibrium was reached.
Adsorption isotherms
Adsorption isotherm of POD was determined by equilibrating different quantities of POD with 100 mg cross-linked Alg–GG beads at pH 4.0 and two temperatures: 25℃ and 6℃. The mixtures were stirred until the adsorption equilibrium was reached and free protein in the supernatant was determined. The amount of POD adsorbed at equilibrium time by unit of mass adsorbent (m) was determined by the following equation:
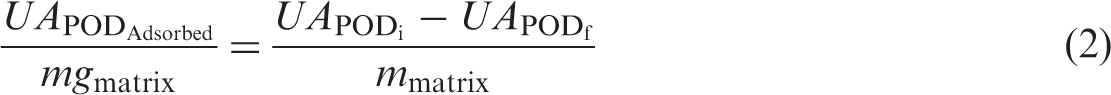
Thermodynamic evaluation
The thermodynamic state functions (free energy, enthalpy and entropy) are important indicators when estimating the mechanism of adsorption process. The distribution coefficient (
The standard Gibbs energy (Δ


Data analysis
Nonlinear regression analysis was applied to estimate the isotherm and kinetic model parameters. Nonlinear regression was performed using trial and error method with the help of Sigma Plot v11 software. In trial error procedure, isotherm and kinetic parameters were estimated by maximizing the coefficient of determination (
Least sum of squares:

The 
Results and discussion
Determination of best adsorption conditions
Adsorption of POD expressed as percent (%) on the initial POD activity in function of pH, using the matrix system 1, 2 and 3. a

The concentration of GG was 0.5% (w/v), temperature 21℃ and medium 25 mM sodium acetate buffer. Total amount of Alg–GG matrix 100 mg.
These results can be explained taking into account the electrical charge of POD and Alg. Generally, commercial Alg have a pKa of 3.2; therefore posses negative charge at pH higher than 3.2. POD isoenzymes posses an isoelectrical pH of 5.5. At pH lower than 5.5 the positively charged groups of POD interact with the negatively charged groups present in Alg resulting in adsorption. The best pH for adsorption is 4.0 since in this value the opposite charges present in the enzyme and polymer are higher. On the other hand, it was expected that a higher concentration of Alg in the matrix increases its ability to interact electrostatically with the enzyme due to the presence of more charges for interact.
Kinetic studies
The kinetic of adsorption was assayed at two temperatures and at three different initial amounts of POD (Figure 1). It can be seen that the adsorbed protein increases with the contact time until it reaches a plateau. The point where the plateau begins is the equilibrium time required to achieve the maximum adsorption in these conditions. A relatively rapid uptake and establishment of equilibrium in a short time indicate the efficacy of that adsorbent for its use in protein absorption from a biomass solution. Available adsorption studies in literature reveal that the uptake of adsorbate species is fast at the initial stages of the contact period, and thereafter, it becomes slower near the equilibrium (Chiou et al., 2004; Woitovich Valetti and Picó, 2016). Between these two stages of the uptake, the rate of adsorption is found to be nearly constant. This is obvious from the fact that a large number of vacant surface sites are available for adsorption during the initial stage, and after a lapse of time, the remaining vacant surface sites are difficult to be occupied due to repulsive forces between the solute molecules on the solid and bulk phases (Luo et al., 2013).
Kinetics of POD adsorption on the Alg–GG matrix S2. Medium: 25 mM sodium acetate buffer, pH 4.0. Total amount of Alg–GG matrix 100 mg. Total amount of POD 0.01564UA. Temperature 21℃. (Results from S3 system are similar – data not shown.)
To analyze the adsorption kinetic mechanism, the experimental data at different temperatures and concentrations were fitted with two models, namely pseudo-first-order and pseudo-second-order, respectively as shown in equations (9) and (10).


Kinetic parameters of adsorption of POD onto Alg–GG beads. a

Medium: 25 mM sodium acetate buffer, pH 4.0. The initial total amount of POD is constant in all the systems. Temperature 21℃ and 6℃.
Intra-particle diffusion mechanism
The solute transfer is usually characterized by either external mass transfer (boundary layer diffusion), intra-particle diffusion or both. The sorption dynamics can be described by the following three consecutive steps which are as follows (Vadivelan and Kumar, 2005):
‐ Transport of the solute from bulk solution through liquid film to the adsorbent surface; ‐ Solute diffusion into the pore of adsorbent except for a small quantity of sorption on the external surface; parallel to this is the intra-particle transport mechanism of the surface diffusion; ‐ Sorption of solute on the interior surfaces of the pores and capillary spaces of the adsorbent.
The third step is assumed to be rapid and considered to be negligible. The overall rate of sorption will be controlled by the slowest step, which would be either film diffusion or pore diffusion. The most commonly used technique for identifying the mechanism involved in the sorption processes by fitting the experimental data in an intra-particle diffusion plot. Previous studies by various researchers (Daneshvar et al., 2012; Vadivelan and Kumar, 2005) showed that the plot of Intra-particle diffusion plot for POD onto Alg–GG matrices S2 (at different initial total concentration of POD), the data have been taken from Figure 1. All the other conditions are the same as Figure 1. The total POD amounts were: A: 0.0134UA, B: 0.0156UA and C: 0.0405UA. (Results from S3 system are similar – data not shown.)

The relationship between the pseudo-second-order rate constant 
Isotherm modelling
Equilibrium adsorption isotherms are known to be very important when it comes to understanding the mechanisms of the adsorption and describing how adsorbates interact with adsorbents. We performed the isotherm absorption at pH 4.0 because this pH value was found to be optimal to perform the adsorption process, so, the temperature effect on the adsorption was analysed. By visual inspection of the adsorption isotherm experimental data (Figure 3), a poor adsorption of enzyme at low protein concentration can be seen, but at increased POD total activity, the bound fraction of enzyme was significantly enhanced. S2 showed high adsorption capacity than S3 (data not shown).
Adsorption isotherm of POD onto non-soluble Alg–GG beads S2 and S3. Medium: 25 mM sodium acetate buffer, pH 4.0. Temperature: 25℃.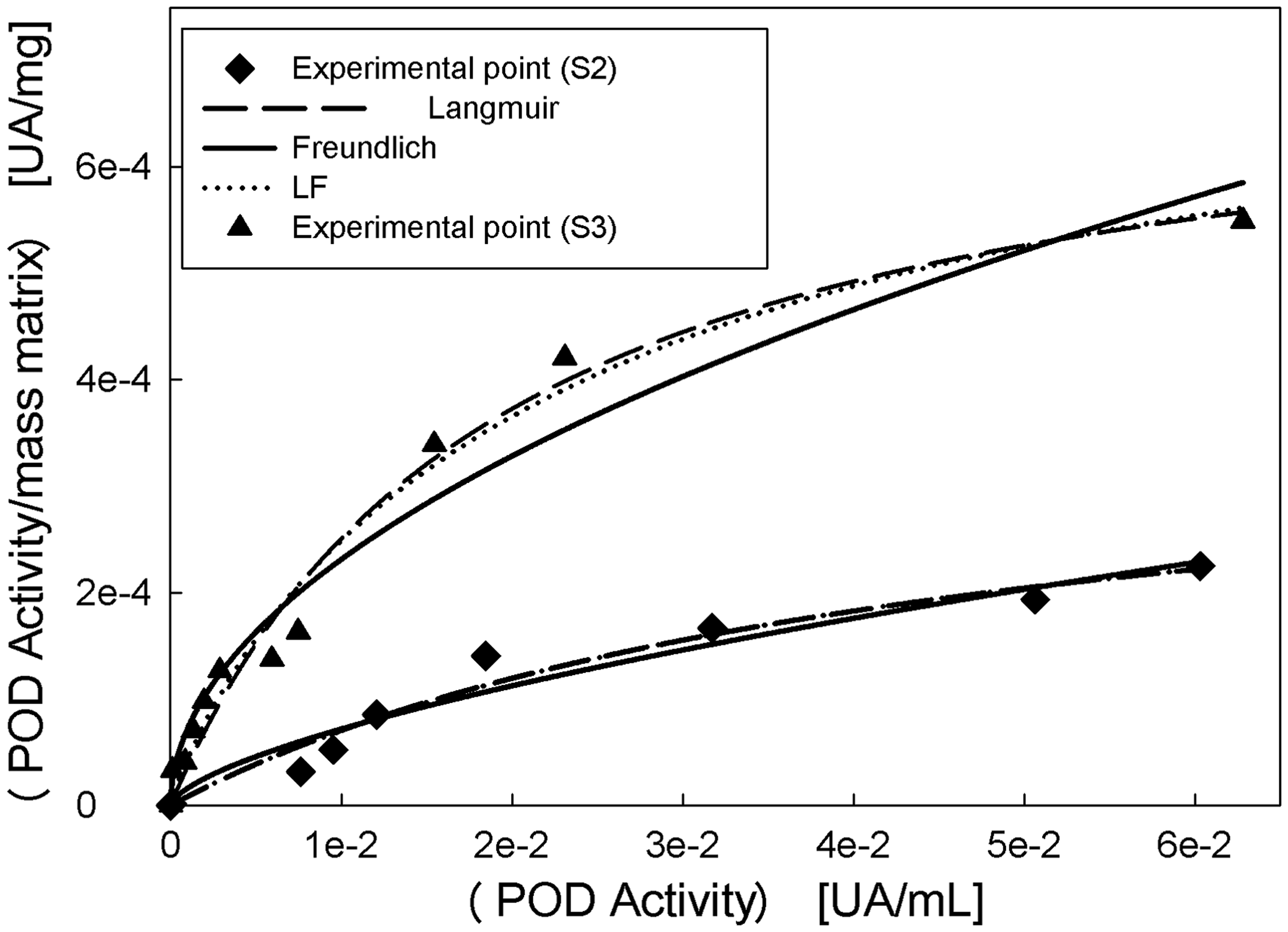
To optimize the design of an adsorption system for the removal of adsorbate, it is important to establish the most appropriate correlation for the equilibrium data. Various isotherm equations have been used to describe the isotherm curve. In order to estimate the validity of isotherm models with experimental data, two-parameter equations were used namely, Freundlich and Langmuir which are given by equations (13) and (14):



Isotherm constants for POD adsorption onto Alg–GG matrix.

The statistical analysis of the error function shows that the equation that best fitted the data for both system is the Freundlich model (see Table 3), this occurs at the two temperatures assayed.
Thermodynamics studies
Thermodynamic constants for POD adsorption ontoAlg–GG beads

The adsorption process was temperature dependent as shown in Figure 3 and, it was found that increasing the temperature induced a significant decrease in the adsorption process in agreement with the negative Δ
Net chemical adsorption is associated to Δ
The temperature increase induced a decrease in the negative entropy value suggesting an increase in the disorder of the system, this finding should be assigned to the loss of water molecules which interact with the OH polysaccharides groups by temperature effect.
At ambient temperature S2 showed a greater capacity to adsorb the enzyme than S3, probably due to the larger separation between the polysaccharide chain caused by S2 low alginate content. At low temperature the inverse effect was observed; the increase in the alginate induces an increase in the adsorption of protein. These results suggest that the temperature can induce a modification in the distance between the polysaccharide chains in a significant manner, facilitating the penetration of the enzyme molecule to the interior of the matrix.
Adsorbed POD activity measured
With the goal to verify the effective bound of the enzyme to the matrix, activity measures were done using the matrix with adsorbed enzyme. The catalytic activity of the immobilized enzyme remains constant over time. Besides from the mass balance of the system the measured value correspond to the results obtained using the depletion method (data not shown).
Conclusions
The possibility of using Alg–GG matrices cross-linked by epichlorohydrin for the adsorption of a vegetal peroxidase was examined in this work. The capacity of adsorption showed to be optimal at pH 4.0 for matrices with 1.0% and 1.5% of alginate (w/v) and 0.5% GG (w/v). At this pH the carboxyl groups of alginate have negative electrical groups, which interact with the positively groups of POD, when the pH was increased, the adsorption process decreased, being null at pH 6.0. The kinetics of the sorption process showed fitted to a first- and second-order-process, however, the analysis of the data suggests that two process are involved in the adsorption mechanism: a diffusion of the protein from the solution to the surface of the matrix and secondly the intra particle diffusion. Thermodynamic analysis of the data showed low values of Δ
Footnotes
Acknowledgements
Ana Silva thanks the EC for her fellowship in IPROBYQ-Rosario, Argentine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: FonCyT, Projects PICT 2013–271 – Argentina Innovator 2020 and Biotechnologies to Valorize the regional food Biodiversity in Latin America – Marie Curie Actions – IRSES Project number 611493 – European Community.
