Abstract
Bromophenol blue (BPB) is a toxic, non-biodegradable, and mutagenic dye, which poses threats to the ecosystem and human health. Various nanocomposites have been used for the removal of BPB dye. However, their removal efficiency is low, and recyclability is still a challenge. In this report, efficient and recyclable silica-chitosan-guar gum (Si-CH-GG) nanocomposites were used as an adsorbent for the removal of BPB dye from aqueous media. Si-CH-GG was developed by the dispersion of silica nanoparticles (Si NPs) into the chitosan-guar gum (CH-GG) matrix. The nanocomposites (NCs) were characterized by techniques such as FT-IR, SEM-EDX, TGA, and XRD. The resulting NCs showed improved thermal properties compared to their components. The adsorption studies for BPB dye were investigated for both CH-GG and Si-CH-GG adsorbents to examine the influence of Si NPs on the BPB removal efficiency. Under the optimum conditions [pH (6), initial concentration (20 ppm), dosage (20 mg), and time (40 min)], the maximum removal percentage (%) for BPB dye was 96% for both CH-GG and Si-CH-GG. However, the maximum adsorption capacities (qmax) were 4.56 and 7.25 mg/g for CH-GG and Si-CH-GG, respectively. It was established that the adsorption followed the Langmuir isotherm and pseudo-second-order kinetics in all cases. Furthermore, CH-GG and Si-CH-GG can be easily regenerated with HCl (0.1 M) and recycled five times, resulting in 100% removal of BPB dye. Both CH-GG and Si-CH-GG proved to be promising candidates for the efficient and highly sustainable removal of BPB dye.
Introduction
Various industries release a vast amount of coloured effluents into the environment, comprising more than 100,000 types of commercial dyes with over seven million tons produced annually.1,2 Among these dyes, bromophenol blue dye remains prominent due to its high solubility and stability in water. It can migrate via aquifers, resulting in surface water pollution. 3 Bromophenol blue (BPB) is a triphenylmethane derivative, and its structurally related compounds such as fluoresceins and xanthenes are widely used in various industries such as textiles, papers, additives, plastics, and laboratory indicators. 4 BPB blue is known to cause mutagenic and heterogenic effects in living organisms due to its strong inhibition of oxygen penetration into water media and reduction in photosynthesis. 5 Due to their synthetic origin and complex chemical structure, dyes are durable to light, oxidation, and biodegradable processes; hence, they can exist in the environment for a very long time.1,6 Therefore, the removal or reduction of dye concentrations in water is crucial to achieving sustainability.
Numerous techniques, such as ion exchange, coagulation, the photocatalytic degradation process, and membrane filtration, have been employed for the removal of BPB dye from wastewater. These methods are reported to be efficient, fast, simple in operation, and eco-friendly.7,8 However, these methods have some shortcomings, such as being expensive, causing membrane fouling, being case-sensitive, and being associated with sludge production.9,10 In the last decade, the adsorption technique has been developed and deployed as a more effective technique for the removal of dyes from aqueous media. This is due to its simplicity, cost-effectiveness, renewability, flexibility, excellent selectivity, ability to recover and reuse adsorbents, wide application, and high efficiency.11,12,13,14 Several adsorbents like zeolites, activated carbons, and graphene have been used for the removal of dyes from water. For example, Ghaedi et al. reported the removal of BPB using activated carbon. 15 In another study, Fathy et al. studied the adsorption of BPB dye using graphene oxide. 16 Graphene oxide was also used by Ortun and coworkers for the removal of BPB dye. 17 While these nanocomposites demonstrated significant removal in the process, they suffer from separation inconvenience and adsorption-desorption kinetics. 18
Recently, the use of nanocomposites consisting of nanoparticles and various biopolymers has gained attention as a viable adsorbent for the removal of BPB. NPs have been extensively used as fillers in the fabrication of polymeric nanocomposites due to their excellent properties such as thermal stability, high surface area, physical rigidity, tunable porosity, and chemical inertness. 19 Various authors have investigated the adsorption of bromophenol blue using different kinds of nanocomposites. Mohammadzadeh et al. synthesized Fe2O3-ZnO-ZnFe2O4/carbon nanocomposites for the adsorption of bromophenol blue dye with an ultrasound-assisted method. The synthesized nanocomposites obtained a maximum adsorption capacity of 90.91 mg/g. 20 A study on the adsorption of bromophenol blue dye was done by Liu and coworkers. They used magnetic Fe3O4MIL-88A nanocomposites, resulting in a maximum adsorption capacity of 167.2 mg/g. 21 In another study, Rastgordani and others were able to achieve a maximum adsorption capacity of 129.6 mg/g using polyaniline@SiO2 nanocomposites. 4
The use of chitosan and guar gum polymers for the adsorption of bromophenol blue dye from the aqueous solution or wastewater has been reported individually or with other polymers, nanoparticles, etc. However, there’s limited research on the use of chitosan-guar gum blended with silica for the removal of dye from water.
Chitosan-based nanocomposites have gained attention in water treatment due to their outstanding qualities, including chemical stability, surface area, mechanical strength, and structural characteristics.22,23
In this work, we report the development of Si-CH-GG nanocomposites by dispersing silica nanoparticles into the chitosan-guar gum matrix. We explored the removal of BPB using CH-GG and Si-CH-GG to understand the effect of Si NPs on the properties of CH-GG as well as the adsorption efficiency of the BPB molecules. The adsorption of BPB dye was investigated using a batch method as a function of pH, contact time, initial concentration, and adsorbent dosage. Various isotherms and kinetic models were applied to study the adsorption behaviour of the BPB onto the adsorbents. Moreover, the regenerative and recyclable capabilities of the adsorbents were assessed.
Experimental
Materials and reagents
Chitosan, guar gum, tetraethyl orthosilicate (TEOS), bromophenol blue, methyl orange, methylene blue, acetic acid, ethanol, hydrochloric acid, and sodium hydroxide were purchased from Sigma-Aldrich. All chemicals were used without further purification. Deionized water was used for all solution preparation.
Synthesis of Si NPs
The Si NPs were synthesized by the sol-gel method. This was achieved by mixing tetraethyl orthosilicate (TEOS), ethanol, and distilled water (4:4:1). The mixture was stirred for 6 h at room temperature, and then left to dry overnight in an oven at 60°C.
Preparation of CH-GG
To obtain chitosan solution, chitosan powder was dissolved in 2% (v/v) acetic acid solution under constant stirring at room temperature to form a solution. The guar gum solution was achieved by dissolving guar gum powder in distilled water at room temperature. The two solutions were physically blended at a weight ratio of 75:25 and then freeze-dried overnight to obtain the CH-GG blend.
Preparation of Si-GG-CH
The Si-GG-CH nanocomposites were prepared by dispersing 2% of the as-synthesized Si NPs into the CH-GG solution with constant stirring for 2 h. The solution was then sonicated in an ultrasonic bath for 3022min at room temperature to allow a good dispersion of NPs. After sonication, the solution was left to dry overnight in a freeze drier.
Instrumentation
Fourier-Transform Infrared Spectroscopy (FT-IR) analysis was performed using a Perkin-Elmer PE 1600 FTIR spectrophotometer to study the chemical interaction between the silica NPs and the CH-GG blend. The morphology and chemical composition of the Si-CH-GG nanocomposites were determined by scanning electron microscopy (SEM) fitted with energy dispersive spectroscopy (EDS), using a JSM-IT 300 instrument. X-ray diffraction (XRD) patterns were obtained using the Bruker D8 and Rigaku Ultima IV X-ray diffractometers. The thermal degradation temperature was determined by gravimetric analysis (TGA) SII TG/DTA6200. The experiment was performed in a nitrogen atmosphere with a heating rate of 10°C/min in the temperature range from room temperature to 900°C. The spectra were recorded in the 400–4500 cm-1 region. The surface area, pore volume, and pore diameter of the Si-CH-GG nanocomposites were evaluated using Brunauer-Emmett-Teller (BET) QuantaChrome Autosorb IQ3. The absorbance of the dye solution was measured at its λmax value (591 nm) using a UV-vis spectrophotometer (UV/VIS Spectrometer Lambda 650).
Batch adsorption studies
A stock solution of bromophenol blue (BPB) dye was prepared by dissolving a known amount of BPB in distilled water. The pH of the dye solution was adjusted using 0.1 M of HCl and NaOH to obtain the desired pH. A known amount of CH-GG and Si-CH-GG was added into 15 mL of BPB dye solution, then agitated in a mechanical shaker (150 r/min) under room temperature for a given time. Afterward, the adsorbent was filtered, and the absorbance of the dye solution was measured at its λmax value (591 nm) using a UV-vis spectrophotometer (UV/VIS Spectrometer Lambda 650). The absorbance was then used to calculate the adsorption capacity (qe) (mg/g) and the adsorption percentage efficiency (%R) of the CH-GG blend using the following equations:
Adsorption of BPB ions in a mixture of dyes
A solution consisting of bromophenol blue (BPB), methyl orange (MO), and methylene blue (MB) ions was used for the selectivity of the CH-GG and Si-CH-GG on BPB ions. The solution was shaken using a mechanical shaker at room temperature for 60 min. At the end of the experiment, the adsorbent was filtered, and the absorbance of BPB, MO, and MB ions was analyzed by using a UV-vis spectrophotometer.
Reusability of the CH-GG and Si-CH-GG
The adsorbed BPB ions on CH-GG and Si-CH-GG were washed several times with deionized water to remove any unadsorbed BPB ions, and then 0.1 M of HCl was used as a desorbing agent. The regenerated wet CH-GG and Si-CH-GG were then used for the next adsorption cycle. To test the reusability of the adsorbents, the adsorption-desorption cycles were repeated five times.
Results and discussion
Morphology, structural, and thermal characterizations
The SEM images in Figure 1(a) show the morphologies of silica NPs, CH-GG, and Si-CH-GG. Particles with an uneven shape were observed in the Si NPs, and the CH-GG showed a porous and rough surface. In the morphology of the Si-CH-GG, it can be seen that the Si NPs were dispersed on the surface of the CH-GG as white spots. Moreover, the introduction of the NPs resulted in a different structure, with the pores found on the CH-GG completely closed up. This showed the interaction between the Si NPs and the CH-GG.
24
(a) SEM images of Si NPs, CS-GG, and Si-CH-GG; (b) EDS spectrum with (c) elemental mapping of Si-CH-GG.
In Figure 1(b), the EDS spectrum and mapping confirmed the presence of Si NPs in the CH-GG matrix, together with O, N, C, Cl, and Na elements.
Figure 2(a) indicates the FTIR spectra of Si NPs, CH-GG, and Si-CH-GG, respectively. The Si NPs spectrum shows a broad band of OH stretching vibration attributed to the absorbed water and silica silanol groups. In addition, peaks observed at 789.71 cm−1, 940.79 cm−1, and 1036.77 cm−1 correspond to the Si-O-Si symmetric and asymmetric vibrations. For the CH-GG spectrum, it displays a strong absorption band of OH water molecules around 3339.81 cm−1, while the absorption bands at 1645.41, 1555.20, and 1408.24 cm−1 are assigned to the stretching vibrations of C = O (amide I), N-H (amide II), and C-N (amide III), correspondingly.
25
The spectrum of Si-CH-GG showed a slight change in characteristic bands when compared to those of CH-GG. The difference in the obtained spectra proves that Si NPs were effectively dispersed in the CH-GG matrix. The OH, amide I, amide II, and amide III absorption peaks in Si-CH-GG showed a shift in the lower frequency as well as an increase in intensity, and an overlap between the silica Si-O-Si group and the C-OH stretching band in CH-GG was observed. From the above information, it can be concluded that there was an interaction between the Si NPs and the CH-GG matrix through hydrogen bonding (Figure 3).
26
FTIR (a), XRD (b) spectra of silica NPs, CH-GG and Si-CH-GG, and (c) TGA spectra of CH-GG and Si-CH-GG. Schematic representation of the fabrication of Si-CH-GG.

The XRD patterns of Si NPs, CH-GG, and Si-CH-GG are presented in Figure 2(b). All three materials exhibit broad diffraction peaks, indicating an amorphous structure. The Si NPs showed a diffraction peak at 2θ of 22.71° and the CH-GG at 2θ at 21.17°.25,27 The dispersion of Si NPs into the CH-GG matrix resulted in a shift of diffraction peaks from 2θ = 21.17° to 22.54°. The slight shift in the diffraction peak was due to the interaction between the functional groups of silica and the CH-GG polymer blend, resulting in the formation of Si-CH-GG nanocomposite. A decrease in intensity was also observed after the dispersion of silica, indicating an increase in the amorphicity of the material. The findings also revealed that no additional peaks were found, pointing to a homogenous mixture of the Si NPs and CH-GG polymer blend.
The thermal stability of CH-GG and Si-CH-GG was assessed using TGA (Figure 2(c)). The CH-GG TGA curve showed two weight losses of 18% and 52.2% at the temperature range of 29.9°C–260°C and 355°C–540°C, respectively. The first weight loss was due to water vaporization, and the second weight loss was due to the decomposition of the main CH-GG matrix. The Si-CH-GG showed three weight losses. The first weight loss of 18% was due to the loss of moisture at the temperature range of 29.9°C–260°C. The last two weight losses of 45.5% (252°C–603°C) and 27.8% (656°C–819°C) were attributed to the thermal degradation and complete combustion of the material. From the above information, an increase in thermal stability was observed when Si NPs were incorporated into the CH-GG matrix. This might be due to the fact that silica is known to have exceptional barrier properties that inhibit the permeation of several atmospheric gases [20].
Porosity analysis
The surface properties of CH-GG and Si-CH-GG.

According to IUPAC, both CH-GG and Si-CH-GG have a mesoporous structure with pores ranging in size from 2.0 nm to 50 nm, as evidenced by the results of the mean pore diameter measurement of 17.072 and 15.286 nm, respectively.28,29
The N2 adsorption-desorption isotherms and BJH pore width distribution for both CH-GG and Si-CH-GG are shown in Figure 4. The results showed that the CH-GG and Si-CH-GG N2 adsorption-desorption isotherm may be classified as Type-IV, indicating that a sizeable component of their structure is mesoporous.30,31 This is consistent with the findings of the pore size distribution of CH-GG and Si-CH-GG. In general, there are a lot of active sites in the synthesized CH-GG and Si-CH-GG that might work well to interact with the BPB dye molecules. The N2 adsorption-desorption isotherms and pore size distribution of (a) CH-GG and (b) Si-CH-GG.
Adsorption of BPB ions
Effect of pH
The pH of the solution plays an important role in the removal of BPB dye. It influences the adsorbent surface charge and the degree of ionization of the materials present in the solution [2]. The effect of pH on the adsorption of BPB dye onto CH-GG and Si-CH-GG was investigated over a pH range of 2-12. As shown in Figure 5, BPB dye solution appears as a yellow colour at pH 2-4, but, it gets ionized to a blue colour at pH 6-12. At pH 2-4 (yellow), BPB dye is identified as having a double-protonated form. However, it’s deprotonated at pH 6-4 (blue).
32
For both CH-GG and Si-CH-GG adsorbents, the highest removal efficiency was observed at pH 6. This might be attributed to the electrostatic attraction between the deprotonated dye molecules (blue solution) and the positive surface active sites of the adsorbents. However, an increase in pH beyond 6 led to a decrease in the adsorption of the molecule due to BPB dye precipitation caused by a high concentration of hydroxide ions (OH-). Furthermore, the electrostatic repulsion between the negatively charged dye and the negatively charged adsorbents results in poor removal of BPB dye at higher pH. It was observed that the adsorption of BPB dye onto the Si-CH-GG (21% at pH 10) drastically decreased as compared to the CH-GG (89%). This might be due to the presence of silica NPs, which contained siloxane groups (Si-O-Si). The latter are negatively charged at higher pH, thus increasing the number of active adsorption sites on the Si-CH-GG. For the next adsorption batch studies, pH 6 was selected for both adsorbents. Effect of pH on the adsorption of BPB onto (a) CH-GG and Si-CH-GG and (b) structure of BPB species according to a colour solution.
Effect of initial BPB ion concentration
The initial concentration impacts the removal of BPB dye. It provides the driving force to overcome mass transfer resistance between the dye ions and the solid phase. 21 In investigating the effect of the initial concentration on the removal of BPB ions by CH-GG and Si-CH-GG, the initial concentration was varied from 10 to 50 mg/L. In Figure 5(a), the results show that the removal efficiency of BPB dye increased with an increase in the initial concentration from 10 to 20 mg/L for both adsorbents. A further increase in initial concentration led to a decrease in BPB dye removal efficiency for CH-GG, while it became constant at 40 mg/L for Si-CH-GG. The increase might be due to the increase in the driving force of the concentration gradient, which encourages enhanced dye molecule diffusion into open interior pores and/or active adsorption sites on Si-CH-GG’s surface.14,33,34 For both adsorbents, a 20 mg/L initial concentration was selected for all following adsorption experiments.
Effect of adsorbent dosage
The effect of CH-GG and Si-CH-GG dosages on the removal of BPB dye varying from 10 to 50 mg was investigated, as shown in Figure 5(b). The results showed that the adsorption efficiency increased as both adsorbents’ dosages increased from 10 to 20 mg. The increase might be due to the increase in the number of vacant adsorption sites, 35 which then get saturated with a further increase in dosage, resulting in a slight decrease in adsorption efficiency. It can be observed from the results that in Si-CH-GG, silica NPs provide more active sites, thus showing high removal efficiency of BPB dye as compared to CH-GG. Therefore, it can be concluded that Si-CH-GG showed more potential for removing the BPB ion than CH-GG. The optimum dosage was found to be 20 mg.
Effect of time
Adsorption of BPB dye on CH-GG and Si-CH-GG was studied as a function of contact time ranging from 10 to 60 min. It can be seen in Figure 5(c) that the adsorption efficiency for both CH-GG and Si-CH-GG increased with an increase in contact time and reached equilibrium at 50 min. The rapid increase in the initial contact time might be attributed to the availability of a large number of vacant sites on the surface of the CH-GG and Si-CH-GG. 36
However, the Si-CH-GG showed a slow adsorption rate at the beginning of the adsorption process as compared to the CH-GG. This shows that the introduction of silica NPs onto the CH-GG delays the interaction of the BPB dye molecules with the adsorbent, hence the low adsorption rate at the start of the adsorption process. This might be attributed to the decrease of the pores, which led to a small surface area Figure 6. (a) Effect of initial concentration; (b) effect of dosage; and (c) effect of time on the adsorption of BPB onto CH-GG and Si-CH-GG.
Adsorption isotherms
To understand the adsorption mechanism of BPB dye on the CH-GG and Si-CH-GG, the equilibrium data of BPB dye was analyzed using Langmuir and Freundlich isotherm models. The Langmuir isotherm describes monolayer adsorption on a homogeneous adsorbent surface with no interactions between the adsorbed molecules, whereas the Freundlich isotherm model proposes monolayer adsorption on a heterogeneous adsorbent surface where interactions between adsorbed molecules are involved.12,37
Langmuir isotherm model
Langmuir model is studied using the following equation
16
:
Figure. 7(a) shows a plot of qe versus Ce with a correlation coefficient of R2 = 0.9346 and 0.9333 for CH-GG and Si-CH-GG, respectively. The R2 showed that the adsorption of BPB dye on the CH-GG and Si-CH-GG followed the Langmuir model, indicating monolayer adsorption. CH-GG and Si-CH-GG showed the maximum adsorption capacities for BPB dye of 5.56 and 7.25 mg/g, respectively. Again, this shows that incorporating Si NPs into the matrix of CH-GG caused an increase in the adsorption capacity. (a) Langmuir isotherms, (b) Freundlich isotherms, and (c) pseudo-first order and pseudo-second order for the adsorption of BPB ions on CH-GG and Si-CH-GG.
Freundlich isotherm model
Freundlich model is studied using the following equation
38
:
Isotherm constants for the adsorption of BPB ions from an aqueous solution.

Kinetics model analysis
To understand the mechanism of adsorption kinetics of BPB dye, two kinetic models were considered, i.e., pseudo-first-order and pseudo-second-order kinetic models.
The pseudo-first-order equation (1):
The pseudo-second-order equation is expressed as follows:
Kinetic parameters for the adsorption of BPB ions.

Mixture of dyes
The mixed dyes experiments were carried out to study the adsorption selectivity of CH-GG and Si-CH-GG. The experiments were carried out under the optimum conditions obtained above. The results in Figure 8(a) and (b) show that both materials have excellent selectivity towards the BPB dye in the mixture of other dyes. BPB and MO dyes can be adsorbed onto the CH-GG and Si-CH-GG, while no adsorption performance was observed on the MB dye. Both BPB and MO are anionic dyes, and MB is a cation. MB is a highly positively charged dye, resulting in electrostatic repulsion with the adsorbents at pH 6. Consequently, the adsorbents showed selectivity toward the anionic dyes. It can be concluded that the adsorption of dyes followed the order of BPB (58%), MO (39%), MB, and (no adsorption performance) for both CH-GG and Si-CH-GG under experimental conditions: pH = 6, initial concentration = 20 ppm, adsorbent concentration = 10 mg, and time = 40 min. Table 4 shows the structure, wavelength, and adsorption efficiency of the mixed dyes. Removal efficiencies of BPB, MO, and MB mixed dyes onto (a) CH-GG, (b) Si-CH-GG, and (c) FTIR spectra of Si-CH-GG and BPB-loaded Si-CH-GG. Mixed dye structure, wavelength, and adsorption efficiency.

Adsorption mechanism
FTIR analysis was used to examine the adsorption mechanism of the BPB ions onto the Si-CH-GG (Figures 8(c) and 9). By relating the spectra of Si-CH-GG before and after BPB adsorption, it can be established that: (1) The initial intermolecular hydrogen-bonded OHstr and N-Hstr (primary) broad band between 3600 and 3100 cm-1 became narrower with higher intensity. (2) The initial C-Hstr signal disappeared after adsorption. (3) The C = Ostr from the dye structure was apparent on the adsorbent after adsorption. (4) A slight decrease in Si-O-Si peak intensity was observed after adsorption. (5) There was an increase in amide I intensity, while amide III showed a decrease in intensity, and lastly, the disappearance of the amide II peak was noticed. The proposed schematic adsorption mechanism of BPB onto Si-CH-GG.

This shows that the functional groups involved in the removal of BPB dye were amino, amide, hydroxyl, and silicon-oxygen groups.
Adsorption-desorption (reusability) studies
The reusability of the material is essential for economic purposes. HCl (0.1 M) was used as an eluent to separate the adsorbed BPB ions from the adsorbents. The use of HCl as a desorbing agent at pH 1.50 completely desorbed the dye from the surface of both CH-GG and Si-CH-GG. This was due to electrostatic repulsion between the protonated ions (H+) and the positive surface of the materials. Thus, it was established that CH-GG and Si-CH-GG could be effectively regenerated with HCl after the adsorption of BPB ions.
The removal efficiency of the BPB increased from 96% (1st cycle) to 100% (2nd-5th cycle), as shown in Figure 10. Following each adsorption cycle, the BPB dye was desorbed using HCl. There was a shift from blue (pH 6) to yellow (pH 3) when the BPB dye solution came into contact with the adsorbents in cycles two through five. This might have produced a better interaction between the BPB ions and the available adsorption sites, resulting in a removal efficiency of 100% (Table 5). The removal efficiency of the BPB ions remained at 100% after 5 adsorption-desorption cycles. This proves that both materials are highly stable, recyclable, and cost-effective. UV-vis absorption spectra and number of cycles of (a) CH-GG and (b) Si-CH-GG reusability for the adsorption of BPB dye. Reusability of the adsorbents for the adsorption of BPB.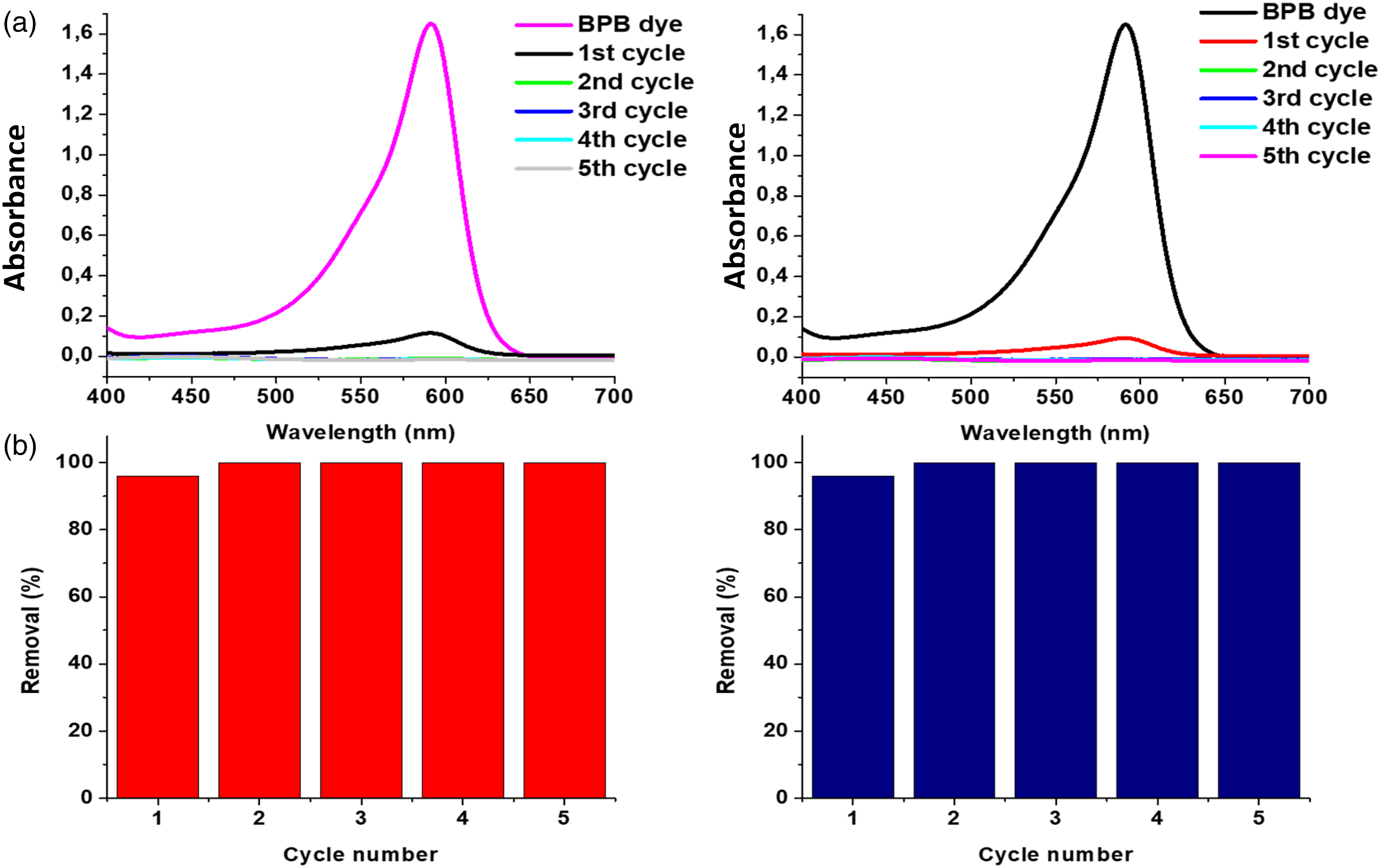

The maximum adsorption capacity value of BPB ions on Si-CH-GG compared with that of other adsorbents.

aUD = Undisclosed.
Conclusions
Si-CH-GG nanocomposite, an effective and recyclable adsorbent for BPB dye in an aqueous solution, was successfully synthesized. The fabricated nanocomposite was characterized by FT-IR, SEM-EDX, TGA, and XRD. The results showed that Si NPs were well dispersed within the CH-GG matrix, leading to an improved maximum adsorption capacity from 4.56 mg/g (CH-GG) to 7.25 mg/g (Si-CH-GG) (about a 59% increase). The adsorption process followed the Langmuir adsorption isotherm (qmax = 7.25 mg/g, R2 = 0.9333) and best-fit pseudo-second-order kinetics (qe = 4.30 mg/g, R2 = 0.9924). The optimum pH was 6.0. The as-synthesized nanocomposite demonstrated excellent stability and reusability (up to 5 cycles without any change in the removal efficiency from cycles 2-5) and thus exhibited potential for removing BPB dye from water.
Footnotes
Acknowledgments
The authors acknowledge the Department of Chemistry, College of Science, Engineering, and Technology of the University of South Africa.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



