Abstract
The reduction of NO2 in air at ambient temperatures with activated carbons can be increased by the infiltration of metal oxide nanoparticles into the sorbents. The NO2 is first adsorbed to the activated carbon and subsequently catalytically reduced to physiologically neutral substances by the metal oxides. The catalytic reduction at ambient temperatures is rather slow. In a former study concerning the application in cabin air filters, it was shown that the modification of activated carbon with 5 wt% CuO/ZnO leads to reduced breakthrough of NO2 and that the adsorbent was able to regenerate between repeated NO2 adsorption cycles. Here we show that the efficiency of the sorbent can be more than doubled by increasing the metal oxide infiltration to 20 wt% whereas a further increase in loading yields no additional improvement, due to a partial transformation of the oxidic compounds.
Introduction
The purpose of adsorptive cabin air filters is to remove noxious gases from the air entering the passenger cabin. NO2 is a critical gaseous pollutant and in contrast to other hazardous compounds, such as benzene, its concentration in ambient air has not been reduced in the last decade (Bruckmann et al., 2014). Thus, our aim was to improve NO2 retention in cabin air filters by modifying the activated carbon via deposition of nanoscale metal oxides. The operation parameters for cabin air filtration are characterized by ambient conditions, thin layers of activated carbon, and frequent interruptions of uptime due to nonoperation times between driving cycles (Bräunling, 2000). The conditions with respect to temperature, adsorptive concentration, oxygen concentration, and relative humidity differ considerably to those of the catalytic reduction of NOx in flue gas treatment which is investigated intensively (e.g. Huang et al., 2008; Muniz et al., 1999; Park and Kim, 2005; Palomares et al., 2012). In the last decade, investigations on the adsorption of NO2 at ambient temperatures (e.g. Bandosz et al., 2013; Belhachemi et al. 2014; Gao et al., 2011) and with modified activated carbons (e.g. Bashkova and Bandosz, 2009; Kante et al., 2009; Levasseur et al., 2011; Nowicki et al., 2015; Pietrzak and Bandosz, 2007) increased. However, the focus of the investigations was on exhaust gas cleaning with correspondingly higher concentrations of NO2 than in cabin air filtration.
The process-related concept for this study was that NO2 is adsorbed on the activated carbon during the driving cycle and catalytically transformed into physiologically neutral substances (N2, O2, CO2) by the metal oxide nanoparticles in the micropores of the activated carbon. The time required for the catalytic reduction, being a slow process at ambient temperatures, is provided by the nonoperating times of the motor vehicle. In this way at least partial regeneration of the adsorbent takes place.
In the preliminary study it was shown (see Sager et al., 2013) that the concept could be realized successfully by the infiltration of 5 wt% metal oxide nanoparticles into activated carbon using incipient wetness impregnation. Under the conditions of cabin air filtration the capacity of the modified sorbent could be maintained for repeated loading cycles. Various metal oxides (e.g. Fe2O3, CuO, Co3O4) and combinations of metal oxides with copper oxide were tested. A mixture of stoichiometric amounts of ZnO and CuO was found to be most efficient for reducing NO2 and subsequent investigations were performed with this mixture. The basic selection criteria for the metal oxides had been that they are redox active, readily available, reasonably priced, and easy to handle.
After the first study had shown that cabin air filter performance can be improved by using activated carbon infiltrated with 5 wt% of metal oxide nanoparticles (Sager et al., 2013), we now investigated to which extent the efficiency of activated carbon modified with CuO/ZnO could be optimized by increasing the amount of infiltrated metal oxide catalysts. This amount is limited by the manufacturing method, the pore volume, and the accessibility of the pores.
Experimental
Preparation and characterization of the modified activated carbon
Activated carbon, denoted as Kugelkohle R 1407, was provided by CarboTech AC GmbH. In order to obtain the activated carbon loaded with CuO/ZnO, the incipient wetness method was used. During incipient wetness impregnation, the total volume of the salt solution is adsorbed by the pores of the activated carbon (the respective volume is determined prior to the impregnation). After impregnation no separate liquid phase exists any more. During the successive heating step, water evaporates and the nitrates decompose to yield the oxides. The chemical ratios of the metal cations (here Cu : Zn = 1) and therefore also the chemical compositions of the oxides are identical to those in the impregnation solution. The bulk chemical compositions of the resulting oxide-loaded carbons are thus entirely determined by the preparation method. In a typical preparation for a carbon loaded with 5 wt% oxides with a Cu-to-Zn ratio of 1, 6.34 ml aqueous 5.32 M Cu(NO3)2 solution, 8.51 ml aqueous 4.06 M Zn(NO3)2 solution, and 54.55 ml deionized water were mixed to achieve the impregnation solution. The solution was slowly added to 104.51 g activated carbon under intensive stirring. Overheating of the sample due to release of heat of adsorption during uptake of the salt solution into the pores was prevented by using an ice bath for cooling. The solution was completely absorbed by the activated carbon and the dry powder was intensively stirred for additional 5 min. The obtained powder was dried for 16 h at 90℃ and then heated to 250℃ with a heating rate of 4℃ min−1 and kept at that temperature for 30 min. For higher loadings, the volumes of salt solutions were adjusted accordingly. To obtain a loading of 30% CuO/ZnO in the activated carbon, 8.86 ml aqueous 5.32 M Cu(NO3)2 solution was admixed to 11.35 ml aqueous 4.06 M Zn(NO3)2 solution. Since the amount of impregnation solution was higher than the maximum pore volume of the activated carbon, the impregnation was performed in two steps. Half of the aqueous solution was added in a first step, followed by drying for 16 h at 90℃. In a second impregnation step, the remaining solution was added and drying was repeated for 16 h at 90℃. Finally, the sample was heated to 250℃ and kept at that temperature for 30 min. For a loading of 40% CuO/ZnO, the impregnation was performed in three successive loading steps. For simplicity, all samples are denoted as carbon loaded with CuO/ZnO even though some of them also contain Cu2O as will be shown below.
The sorption properties of the samples were investigated by nitrogen adsorption at -196℃ using Micromeritics ASAP 2010 sorption analyzers. Prior to the adsorption measurements, the samples had been activated by heating at 200℃ under vacuum for 16 h. The apparent specific surface areas were calculated from the sorption isotherms via the Brunauer Emmett Teller method using data points in the relative pressure range of 0.05–0.2. Total pore volumes were calculated from nitrogen volumes adsorbed at a relative pressure close to 0.995. Pore size distributions were derived with the non-local density functional theory (NLDFT) method, using the equilibrium model for nitrogen adsorption in carbon slit pores at –196℃. Micropore volumes were calculated from these pore size distributions as the volume adsorbed in pores smaller than 2 nm. Data evaluation was performed with the Quantachrome Autosorb software package.
Thermogravimetric (TG) experiments combined with differential scanning analyses (DSC) were performed with a Netzsch 449 F3 Jupiter TG/DSC analyzer. Samples were heated either in an air or in an argon flow (40 ml min−1) to 500℃. The composition of exhaust gas was analyzed with a Netzsch QMS 403 D Aëolos quadrupole mass spectrometer attached directly to the TG/DSC analyzer.
XPS spectra were measured with a Kratos spectrometer equipped with a hemispherical analyzer. A monochromatized Al X-ray source (E= 1486.6 eV) was operated at 15 kV and 15 mA. Pass energy was set to 160 eV for the survey scans. Hybrid mode was used as lens mode. The base pressure in the analysis chamber was 4×10−9 Torr (5.3×10−7 Pa). The binding energy scale was corrected for surface charging by use of the C 1s peak of carbon as reference at 284.5 eV. In order to expose the oxide within the activated carbon, the carbon samples were ground prior to analyses. Oxides within the carbon beads thus get exposed to the surface of the crushed particles and can be analyzed with XPS.
X-ray diffraction patterns were measured on a Stoe STADI P powder diffractometer in reflection mode (Bragg–Brentano geometry) using a secondary graphite monochromator and a scintillation detector. The samples were measured on reflection-free quartz sample holders. Rietveld refinements were performed with the TOPAS software suite and line profile analyses via whole powder pattern modeling with the PM2K software package (Leoni et al., 2006). This method allows evaluation of microstructure parameters, such as crystalline domain size, domain size distribution, stacking fault probability, and twinning probability, from diffraction profiles (Scardi, 2008). Here, only average domain sizes, Dave, and the standard deviations,
Experimental procedure of the breakthrough tests
The sorbents were tested by repeated breakthrough experiments in a fixed-bed flow reactor (inner diameter 0.05 m) which is illustrated in Figure 1. The adsorptive NO2 was directly supplied to air (23℃ and 50% relative humidity) as carrier gas from a reservoir using a mass flow controller so that the volumetric inlet concentration of NO2 was 4 ppm. The equipment of the NO2 supply was heated to ensure the required vapor pressure and avoid condensation. The fixed bed consisted of 2 g of the adsorbent and the flow rate was 0.2 m/s. At the inlet and outlet of the fixed-bed reactor, the concentrations of NO2 and NO were recorded quasi continuously with two nitrogen oxide analyzers (type AC 31M from Ansyco, Karlsruhe, Germany). The measuring range of the analyzers using chemiluminescence is up to 10 ppmV, the lower detection limit is <1 ppb, and the precision of the measurement is ±1% of the upper range value. At input concentrations of 4 ppmV the measurement uncertainty causes a lack of significance of breakthrough values below 2.5%. Each breakthrough experiment (cycle) was conducted for 90 min. Between the first three cycles, the adsorbents were left to rest periods of 3 and 15 h. During the resting times, the respective sorbents were stored in small sample containers. The experimental procedure is described in more detail in Sager et al. (2013).
Sketch of the experimental set-up for the breakthrough experiments.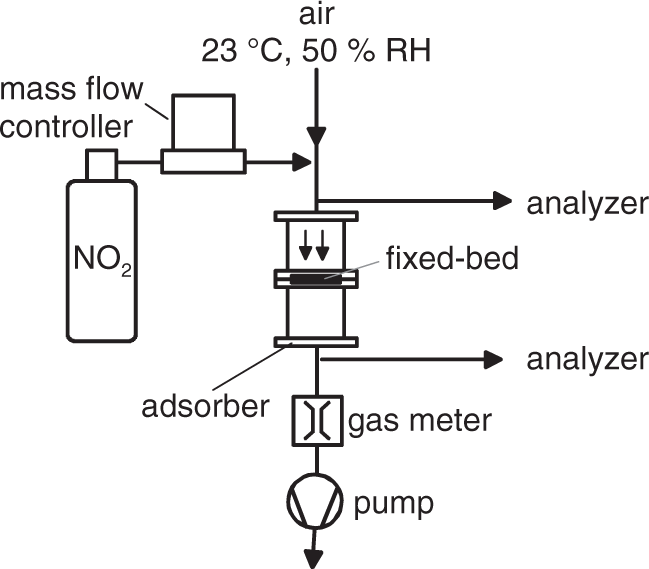
Results and discussion
NO2 breakthrough tests
As described, the test of individual adsorbent samples included measurement of three breakthrough curves with two intermediate rest periods. In addition to the breakthrough curves of NO2, a significant fraction of NO was measured downstream of the sorbent bed. The phenomenon of NO2 reduction to NO by activated carbon is commonly observed and not caused by the metal oxide catalysts. With the released oxygen, the carbon is oxidized to CO or CO2 and/or oxo-functionalized groups are generated on the inner surface of the activated carbon (Heschel and Ahnert, 1998; Unseld, 1969). Details on the time course and the influence of the adsorptive concentration on the generation of NO were described by Sager and Schmidt (Sager and Schmidt, 2009). The fraction of NO is considered in the evaluation by the breakthrough curves of the sum parameter NOx. Thus, the results of one experiment cycle for an individual adsorbent sample are three breakthrough curves of NO2 and three of NOx.
For example, in Figure 2 the breakthrough curves of NOx and NO2 of one sample of activated carbon impregnated with 30 wt% of CuO/ZnO are shown. Both, the breakthrough of NOx and of NO2 increase during the subsequent adsorption cycles. In the first 2 min of each cycle, the breakthrough curves are distorted by effects related to the experimental procedure. The breakthrough of NOx reaches 21% at the end of the third cycle. The breakthrough curves of NO2 are below 2% and thus, as described above, within the measurement uncertainty. The breakthrough curve of NO2 during the first cycle is too close to the x-axis to be perceptible.
Breakthrough of NOx and NO2 through activated carbon impregnated with 30 wt% of CuO/ZnO (23℃, 50% RH, c1 NO2 = 4 ppmV, c1 NO = 0).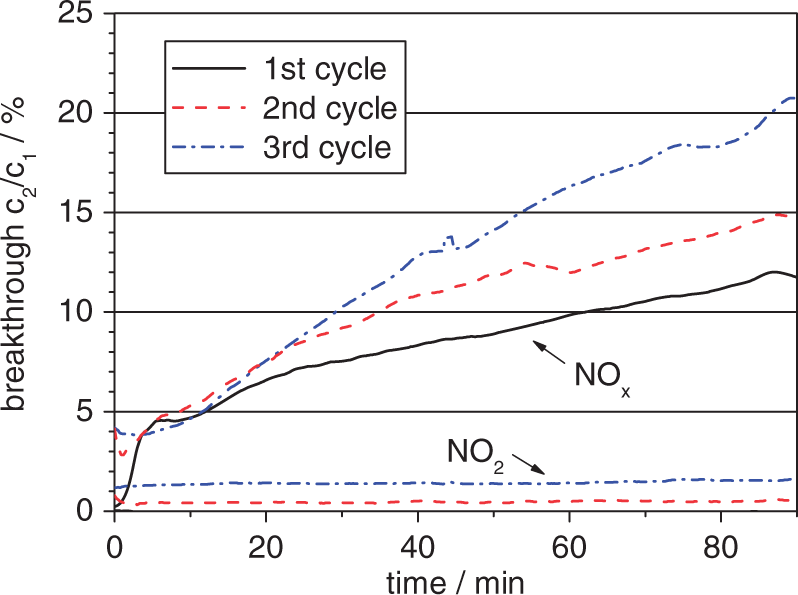
The breakthrough curves of NO2 of the differently strong infiltrated adsorbents are nearly all within the measurement uncertainty as shown in Figure 2 for the infiltration degree of 30 wt%. By the modification of the activated carbon, the reduction of NO2 is considerably enhanced. For comparison, the breakthrough curves of NO2 through one sample of nonmodified adsorbent are shown in Figure 3. From the second adsorption cycle on, the NO2 breakthrough is beyond measurement uncertainty and increases during subsequent cycles to exceed 11% at the end of the third cycle.
Breakthrough of NO2 through nonmodified activated carbon (23℃, 50% RH, c1 NO2 = 4 ppmV, c1 NO = 0).
For comparison of the efficiency of modified and nonmodified adsorbents with respect to the separation of NO2 and NOx, the respective NOx breakthrough after 90 min at the end of specific adsorption cycles is plotted against the degree of infiltration in Figure 4. Furthermore, the dependence on the adsorption cycle is illustrated. The values of the breakthrough at 90 min through the activated carbon infiltrated with different oxide loadings are plotted in Figure 4. The three different symbols mark breakthrough values measured after each of the three cycles (see Figure 2).
Breakthrough of NOx after 90 min at each adsorption cycle through activated carbon with different degree of infiltration.
Figure 4 shows that the infiltration of CuO/ZnO catalysts into the activated carbon leads to reduced NOx breakthrough. This improvement of the NOx separation is increased up to a degree of infiltration of 20 wt%. Beyond this degree of infiltration the breakthrough remains more or less the same. Figure 4 also points out that the deterioration of the breakthrough during the successive adsorption cycles is lower for the modified adsorbents. Thus, the assumption is supported that at least a partial regeneration of the modified adsorbent is achieved by the metal oxide catalysts.
Characterization of adsorbent materials
Apparent specific surface areas, and pore volumes of pure activated carbon and of carbon loaded with 10–40 wt% CuO/ZnO. The percentages of the micropore volumes from the total pore volumes are given in parentheses in the last column.

BET: Brunauer Emmett Teller.
X-ray diffraction patterns of the different CuO/ZnO-loaded samples are shown in Figure 5. For low loadings, only CuO is visible as crystalline phase. In these samples ZnO is present either as amorphous phase or its crystallites are too small to result in distinct XRD reflections. For oxide loadings of 30 and 40 wt% also crystalline ZnO is observed in the carbon along with CuO. Also a second copper oxide phase, i.e. Cu2O, is found in these samples. The remaining broad reflections are caused by the carbon matrix (2θ ≈ 26.6° and 43.5°). Quantitative Rietveld refinements reveal that the crystalline oxidic compounds in the carbon matrix consist of 73.7% CuO, 12.8% Cu2O, and 13.5% ZnO for the sample loaded with 30 wt% oxide and 29.9% CuO, 40.7% Cu2O, and 29.4% ZnO for the sample loaded with 40 wt% oxides (see Figure S4, ESI). The content of Cu(I) oxide increases with amount of oxide loading. The amount of crystalline ZnO observed is substantially below the total concentration of ZnO in these samples. Thus, samples in which crystalline ZnO is observed contain also significant amounts of ZnO that is invisible to X-ray diffraction, i.e. it is either amorphous or crystallites are too small to cause diffraction patterns.
XRD patterns of activated carbon loaded with 5, 10, 20, 30, and 40 wt% CuO/ZnO (from bottom to top).
The diffraction data have been analyzed with PM2K in order to calculate particle size distributions of the crystalline oxidic compounds. Fits of peak profiles to the experimental data are shown in Figure 6. The flat difference curves show the very good match of the fits to the experimental data. Lognormal particle size distributions for CuO, ZnO, and Cu2O have been calculated from the parameters used for the modeling of the experimental data and are presented in Figure 7 and the results are summarized in Table 2.
WPPM fitting of diffraction data of the activated carbon samples loaded with different amounts of CuO/ZnO. The black line corresponds to the measured data, the red line to the modeled peak profiles, and the gray line at the bottom to the difference curve. WPPM: whole powder pattern modeling. Domain size distributions of the different crystalline phases in the activated carbons. Average domain sizes, Dave, and standard deviations, s.d., of lognormal domain size distributions as calculated with PM2K. Data from Tseng et al. (2015).


The mean crystalline domain sizes of the individual crystalline phases correspond well to the small mesopore diameters of 2–3 nm that are present in the activated carbon. During the decomposition of the nitrate precursors, NO2 and O2 are generated that can combust a small fraction of carbon within the pores of the activated carbon and thus slightly widen the pore diameters. In this way, also slightly larger particles can get accommodated within the mesopores. However, the mean crystalline domain sizes of all observed crystallites fall well into the range of pores that are expected for the activated carbon. At higher oxide loading, Cu2O is formed in significant amounts, which is likely the result of reduction of CuO by CO that is generated during the partial combustion of the activated carbon during the nitrate decomposition. The results of the breakthrough experiments show that the presence of Cu(I) oxide has no beneficial effect on the breakthrough of NO2 or on the regeneration ability of the activated carbon.
Grinding of the carbon beads exposes the interior of the spheres. XPS investigations on ground samples reveal that the Cu:Zn ratio on the newly generated surfaces is significantly above unity (see Table S2, ESI). For loadings with 5, 10, 20, and 30 wt% CuO/ZnO, Cu:Zn ratios of 2.8, 2.9, 1.9, and 2.2 were observed. Thus, a major fraction of Zn seems to be invisible to XPS. In carbon the escape depth of photoelectrons is rather small, and therefore, Zn that is buried deeper in the carbon is not detected. As known from the sorption experiments, micropores in the activated carbons get successively blocked with increasing loading with the mixed oxides. Zn species in micropores within the carbon matrix would get less exposed to the surface during grinding of the carbon spheres and thus be less visible to XPS. It can therefore be concluded that a major fraction of the Zn species is deposited in the micropores of the activated carbon.
After using the oxide-loaded activated carbon samples for NO2 removal during up to six breakthrough experiments with rest periods of up to three months, the Cu:Zn ratio of ground samples changes significantly. For low loading with only 5 wt% CuO/ZnO, the ratio becomes 1, i.e. the ratio of metals used for the preparation of the material. For higher loadings, the amount of Zn is also increased but not to unity (Cu:Zn = 1.6 for 30 wt% CuO/ZnO). It thus seems that Zn species migrate from the micropores to the larger mesopores and get deposited there. This process is likely induced by water that is accumulated within the micropores during the long NO2 removal tests in humid air (50% humidity). XRD investigations show that the content of Cu2O is significantly reduced after the tests in samples with higher loading, at the same time a new phase (Zn,Cu)5(OH)6(CO3)2 (aurichalcite) is formed within the mesopores (Figure S5, ESI). Hydroxycarbonates of Zn are formed easily in humid air in the presence of CO2, a process that is likely to proceed within the carbon pores. The presence of Cu in close proximity causes the formation of the mixed metal hydroxycarbonate aurichalcite. TG/DSC experiments confirm the formation of such a phase (Figure S6, ESI). After the NO2 removal tests, the residual mass after carbon combustion in air is significantly less than expected for the oxide loaded samples, i.e. the total weight of the samples increased. The weight increase corresponds well to the formation of aurichalcite since water and carbon dioxide get fixed in the crystal structure. Upon heating under argon, about 6.8 wt% of mass is lost in the temperature range where decomposition of aurichalcite is expected to proceed. At the same time, analysis of the composition of the exhaust gas by mass spectrometry shows the release of water and CO2 in that temperature range (Figure S7, ESI). Thus, aurichalcite is formed from Cu(II) and Zn(II) during the long-term NO2 removal tests. In none of the samples nitrogen could be detected by XPS indicating that no metal nitrates were formed in significant amounts during the NO2 removal cycles.
Conclusions and outlook
Increasing the loading of CuO/ZnO in the activated carbon up to about 20 wt% causes a significant enhancement of the NO2/NOx removal capability of the adsorbent material. Higher loadings do not cause substantial improvement of the breakthrough behavior of the adsorbent. Even though the NO2/NOx breakthrough after 90 min is more or less identical for samples containing 20, 30, and 40 wt% of the mixed oxides, the composition of the oxidic compounds differs substantially in these materials. This implies that the presence of an additional Cu2O phase at higher oxide loadings does not affect the properties of the adsorbent and its regeneration capability in a beneficial manner. At lower loadings, only CuO is observed as crystalline phase in the adsorbent materials. Higher oxide loadings result in the formation of crystalline ZnO next to CuO and Cu2O. However, a major fraction of Zn is likely deposited as amorphous oxide/hydroxide within micropores of the activated carbon. Upon exposure to humid air during the NO2 breakthrough experiments, zinc species migrate into mesopores. With time, aurichalcite, a mixed zinc and copper hydroxycarbonate, is then formed in these pores. All crystalline particles observed have domain sizes of 2.5–3.5 nm which are accommodated in the mesopores of the activated carbon. An optimum loading with the mixed oxide seems to be achieved at about 20 wt% CuO/ZnO. At this composition, the adsorbent material shows high retention of NO2 and NOx whereas higher oxide concentrations do not alter the performance of the adsorbent significantly.
The exact role of the individual species within the metal oxide-loaded activated carbon will be investigated in further studies. However, direct assessment of catalytic reactions will be impossible for reactions taking place over a very long time period at room temperature and with NO2 concentrations on ppm level. Thus, future investigations will focus on tests at somewhat higher temperatures and higher NO2 concentrations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the German Federal Ministry of Economics and Technology for financial support within the agenda for the promotion of industrial cooperative research and development (IGF) based on a decision of the German Bundestag. The access was opened by the IUTA e. V., Duisburg, and organized by the AiF (IGF-Project No. 17516N).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
