Abstract
Perfluorohexane adsorption on porous activated carbon was studied via Monte Carlo methods. In order to estimate the microporous adsorption capacity at relative pressures (P/P0) 0.1, 0.3, and 0.6, microporous structures were generated by simply packing corannulene and oxygenated corannulene fragments together into a simulation box and the benchmark data were validated by calculating CO2 and Ar adsorption isotherms prior to estimating the perfluorohexane adsorption capacity of the candidate material BAM-P109. Since ∼50% of the total pore volume of this material is mesoporous, the mesoporous adsorption capacity was also estimated using a slit-pore model with varying pore widths at P/P0 = 0.3 and 0.6. Prior to the adsorption simulations, the saturation pressure of perfluorohexane at 273 K was estimated as 8.13 kPa through a series of Monte Carlo simulations, which is in good agreement with the experimental data of 8.55 kPa.
Introduction
The 8th Industrial Fluid Properties Simulation Collective (IFPSC) considered the perfluorohexane (PFH) adsorption onto activated carbon (AC) as a suit to the 7th challenge addressing PFH adsorption on BCR-704 faujasite (FAU) type zeolite (http://fluidproperties.org/simulation-challenge). The candidate material, BAM-P109, was characterized and the benchmark data were published in the literature (Ross et al., 2016). We used Monte Carlo (MC) simulations to address the challenge and were awarded the first prize.
Perfluorocarbons (PFCs) are linear molecules with the chemical formula C x F y . In PFCs, all hydrogen atoms in their hydrocarbon counterparts are replaced with fluorine atoms. PFCs are one of the most powerful greenhouse gases. Even their concentration in the atmosphere is lower when compared with the other potent greenhouse gases, such as methane (CH4) and carbon dioxide (CO2), and they can remain for longer times because of their chemical stability, especially due to the very stable C–F bonds. PFH, in particular, is known as a good solvent for oxygen dissolving and is widely used because of its chemically inert and non-toxic properties in medicine (Dias et al., 2003).
ACs, on the other hand, are one of the most promising materials to separate and store gases (Esteves et al., 2008; Shafeeyan et al., 2010; Wilson and Danner, 1983) due to their intrinsic properties, such as high surface area, low cost of processing, thermal and mechanical stabilities. They have been using widely for removing heavy metals and contaminants from aqueous solutions (Yin et al., 2007).
In the literature, there are limited number of molecular simulation studies on PFH adsorption in porous materials and no remarkable studies in ACs, with the exception of the study of Müller (2005). The adsorption of simplest perfluoroalkanes (PFA), up to C3 has been investigated on zeolites (Mellot and Cheetham, 1999; Mellot Draznieks et al., 2000). Mellot and Cheetham (1999) studied CF4 and various chlorofluorocarbons and hydrofluorocarbons in siliceous Y and NaY zeolites, whereas Müller (2005) and Mellot Draznieks et al. (2000) focused on the adsorption of the three simplest PFAs on the AC.
Mosher et al. (2013) studied CH4 adsorption on microporous and mesoporous carbons. Carbon systems were modeled as slit-pore models, where the carbon sheets were positioned parallel to each other with a gap between two adjacent carbon sheets to enable the gas molecules to adsorb. They revealed the distinction between adsorption on micropores and mesopores.
In this study, we investigated the PFH adsorption behavior of the candidate material BAM-P109 AC. We first modeled the carbon fragments and packed them in a simulation cell based on the experimental chemical composition determined via X-ray photoelectron spectroscopy (XPS) reported in the challenge website. The calculated X-ray diffraction (XRD) pattern was compared with the experimentally reported pattern (Ross et al., 2016). Once the structure characterization was completed, the adsorption of the gases, CO2 and Ar, was performed via MC simulation to validate our model and compared with the benchmark data (Ross et al., 2016). The phase properties of PFH were already reproduced in our previous study addressing the 7th IFPSC (Demir and Ahunbay, 2014). Using the same methodology, the vapor pressure of PFH at 273 K was estimated to make sure of the validity of the force field used for both adsorbent and adsorbate. After all, PFH adsorption simulations were performed either on micropores or on mesopores. This study is the first attempt to predict PFH adsorption on the AC model via molecular simulation methods.
Simulation details
The pore size distribution (PSD) data indicated that ∼50% of the total pore volume of BAM-P109 is mesoporous (Ross et al., 2016). Therefore, two different pore models were constructed to estimate the PFH adsorption capacity of the material: a microporous model that is based on the corannulene (CR) type carbon fragments; and a mesoporous model that is based on a collection of slit pores.
Microporous pore model
Two different fragments were used to construct the AC model, as depicted in Figure 1. Both are CR and one of them is functionalized by one hydroxyl group (CR_OH) to render the model containing oxygen. The XPS results revealed different atomic compositions of the candidate material, BAM-P109 (http://fluidproperties.org/simulation-challenge). The carbon composition is lower in the individual pellets. Moreover, a trace amount of Na has been detected in the pellets, which may be due to the use of binders to the ACs more packed. On the other hand, the crushed samples have higher carbon composition and no Na ions. Therefore, the crushed AC is considered as the basis for the atomic compositions. The values based on the mass are then converted to the number based values. As a result, the C/O ratio is calculated as 47 in number based. Therefore, 135 CR and 100 CR_OH fragments were packed in a cubic simulation box by using Amorphous Cell module of Materials Studio 5.0®. Ten different configurations having the least energy were generated at a density range 0.5–1.0 g/cm3. In the reported experimental report (Panne and Thünemann, 2010), the surface area measurements were performed by using nitrogen gas (N2) at 77 K. Therefore, we used the same probe radius reported in the experimental report (Panne and Thünemann, 2010), which was equal to 2.271 Å. Since the Brunauer, Emmett and Teller (BET) surface area of the AC BAM-P109 is known, equal to 1396 m2/g, the configurations generated at 0.7 g/cm3 (which corresponded to a cubic simulation box with sides of 52.3 Å) had surface areas closer to the experimental one. Among 10 configurations generated at 0.7 g/cm3, the configuration having the closest surface area to the experimental one, which is equal to 1387 m2/g, was selected as the simulation box, as well as two more simulation boxes which were selected to compare the adsorption results. The surface area values of the selected three simulation cells are reported in Table 1.
Two fragments (a) CR and (b) CR_OH. The curvature of each fragment is also shown on the right. Surface area values of generated simulation cells.

LJ parameters and atomic partial charges for the AC.

LJ: Lennard-Jones model; AC: activated carbon.
As reported by Di Biase and Sarkisov (2013), the curvature of carbon fragments has key importance in the adsorption. They also performed adsorption simulation by using flat carbon fragments (coronene type fragments based on the six-membered cyclic aromatics) and their results revealed that the adsorbate–adsorbent interaction near curved surfaces are stronger compared with the flat surfaces, which may be attributed to the hybridization of carbon atoms between sp2 and sp3.
The choice of the oxygenated group is based on the simulation studies performed by Di Biase and Sarkisov (2013). They reported that the important point is the C/O ratio rather than the type of the oxygenated groups. Based on this, we created the oxygenated carbon fragments by simply adding a hydroxyl group to one of the end C atoms.
The number of carbon fragments packed in a simulation box was selected, so that a smaller box containing lower number of carbon fragments may lead to the statistically unhealthy results during adsorption simulations. Therefore, the number of carbon fragments is also consistent with other simulation studies in the literature (Di Biase and Sarkisov, 2013).
Mesoporous pore model
The mesopores were modeled as a collection of independent, non-interconnected, graphitic slit pores (Mosher et al., 2013). The pore surface was represented using a six-layer perfect graphite sheet. The simulation box containing a given slit pore has dimensions of Lx = 3.4080 × Ly = 2.9520 nm in the x- and y-directions, respectively, and with periodic boundary conditions. The distance between the two slabs in the z-direction was adjusted to model at three different slit pores widths: w = 7.3, 9.7, and 13.3 nm. Figure 2 shows the simulation box for the adsorption of PFH in mesopores. Both surfaces, oxygenated and then saturated with hydrogen, were in contact with PFH molecules. The position of oxygen atoms was selected randomly. In the literature (Di Biase and Sarkisov, 2013), it is reported that the important point in the adsorption process on activated carbon is not the position of oxygen atoms on the surface, but the C/O ratio. This ratio was the same that we used for the adsorption of PFH molecules in the micropores. The number of graphite sheets was chosen so that the PFH molecules adsorbed on one surface would not interact with PFH molecules adsorbed on the other surface. The distance between two C atoms in the two adjacent graphite sheets is 3.685 Å, whereas the bond length between two C atoms is 1.420 Å.
Mesoporous model for PFH adsorption.
In order not to break the two-dimensional structure of the graphite sheets, the geometry optimization was performed to adjust the positions of O and H atoms connected to the surfaces, where all C atoms were kept rigid.
When slit pores are used, the mesoporous adsorption capacity (qmeso) can be estimated based on the pore width distribution. If the pore width distribution is discretized with an associated probability (pi) for the ith pore width (wi), the adsorption capacity can be estimated from the following relationship:

Benchmark data validation
The XRD pattern for the candidate material has been reported in the literature (Ross et al., 2016). In an XRD experiment, a sample is bombarded by a beam of electrons which, in turn, ionize electrons from the target atom. This leads to the emission of X-rays from the target atom. The well-defined narrow peaks are the outcomes of the XRD for crystalline structures, whereas broader peaks are characterized in amorphous structures. Figure 3 depicts the benchmark (Ross et al., 2016) and calculated XRD patterns for the candidate material, BAM-P109. The comparison shows that there is slight discrepancy between the experimental and calculated XRD patterns; nevertheless the model may be assumed to reproduce the amorphous structure of the material reasonably.
Experimental (Ross et al., 2016) and calculated XRD patterns for BAM-P109 AC.
To validate the benchmark data (Ross et al., 2016), some preliminary adsorption simulations were carried out using Materials Studio Sorption Module®. Grand Canonical Monte Carlo (GCMC) simulations were performed to predict the adsorption isotherms, which relied on the parity of the chemical potential between gas phase and adsorbed phase. Constant chemical potential was imposed indirectly through the fugacity of the sorbate molecules, which was assumed to be equal to vapor pressures of sorbates.
LJ parameters and partial charges of Ar and CO2.

Note: LJ: Lennard-Jones.
First, CO2 adsorption isotherms were calculated for three cells having the same density but different surface area values, which is reported in Table 1. Cell 1 which has the closest BET surface area to the experimental one gave the best fitted CO2 adsorption isotherm to the experimental one, as shown in Figure 4. Even though the isotherms obtained from the other two cells, cells 2 and 3, were slightly different. The adsorption isotherm obtained for cell 3 is shifted up when compared with the others, which proved the largest surface area among these three cells. For further simulations, only cell 1 was used.
Experimental (Ross et al., 2016) and predicted CO2 adsorption isotherms at 273.15 K.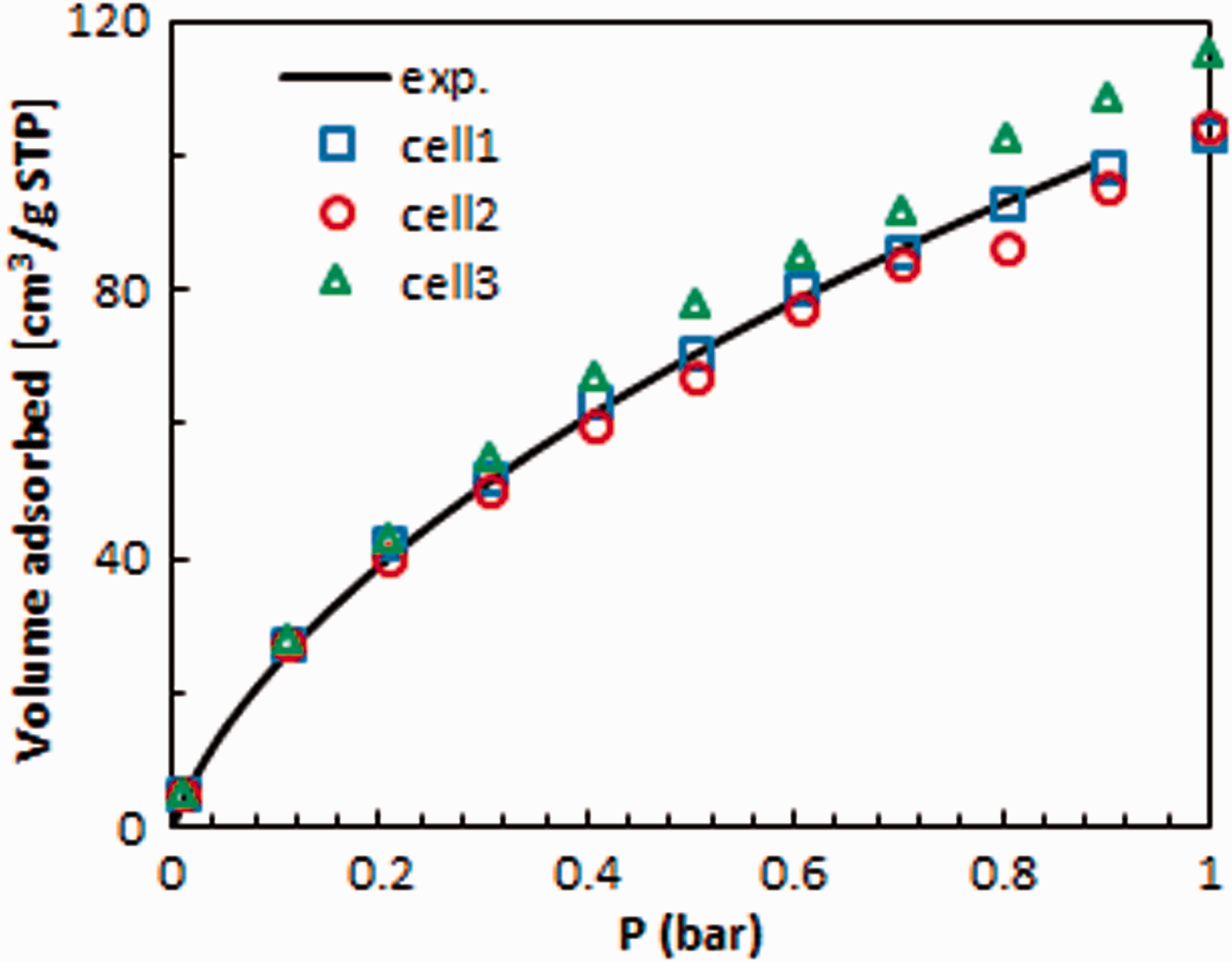
Figure 5 shows the comparison of calculated and measured Ar adsorption isotherms. Our calculations slightly overpredicted the Ar adsorption, which may be attributed to several cases. First, the carbon and Ar models that we adopted may have led to this overestimation. Second, we presumed a defect-free and uncontaminated AC; however, these effects were normally inherent to the adsorbent.
Experimental (Ross et al., 2016) and predicted Ar adsorption isotherms at 87 K.
Bonded interaction parameters for the perfluorohexane molecule.

LJ parameters and atomic partial charges for the perfluorohexane molecule and the activated carbon.

LJ: Lennard-Jones.
PFH vapor pressure calculations
The calculated vapor pressure of PFH at 273 K was 7.2 kPa using our previous data, whereas the experimental result reported in the literature (Ross et al., 2016) is 8.55 kPa. The difference, which is 15.8% with respect to the experimental value, necessitated to perform further simulations to obtain closer vapor pressure to the experimental one. Therefore, we performed two additional NPT simulations at 277 K and 266 K in addition to our previously reported results (Demir and Ahunbay, 2014), below the normal boiling point of PFH, and the results are depicted in Figure 6.
Saturated vapor pressures of Perfluorohexane at different temperatures. Simulation results are compared with the experimental data obtained from National Institute of Standards and Technology (NIST) (http://webbook.nist.gov/cgi/cbook.cgi?Name=perfluorohexane&Units=SI).
The vapor pressure of PFH at 273 K was found by fitting the Antoine Equation to the calculated vapor pressure data.

Results
Microporous PFH adsorption on BAM-P109
PFH adsorption on the BAM-P109 at 273 K was investigated at three different relative pressures, 0.1, 0.3, and 0.6, which corresponded to 0.813, 2.439, and 4.878 kPa, respectively. The simulations were performed by using Fixed Pressure–Gibbs ensemble Monte Carlo (FP-GEMC) method (McGrother and Gubbins, 1999). In FP-GEMC, the total number of atoms, N, the temperature, T, and the pressure in the gas reservoir were kept constant. Two simulations boxes were set: one was for gas reservoir from where the gas molecules come to adsorb and the other one was for the AC. Initially, 250 PFH molecules were loaded into the gas reservoir box and the volume of the gas reservoirs was adjusted according to the pressure set. In addition, the volume of the AC was kept unchanged during the adsorption simulations. MC simulations were run for 5 × 106 steps where the last 1 × 106 steps were used for production. Figure 7 shows the adsorbed PFH amounts in micropores at the three relative pressures. The adsorption capacities in micropores (qmicro) at the relative pressures of 0.1, 0.3, and 0.6 are shown in Table 6.
Perfluorohexane adsorption isotherm in micropores at 273 K. Microporous Perfluorohexane adsorption capacity of BAM-P109 in different units. PFH: perfluorohexane.
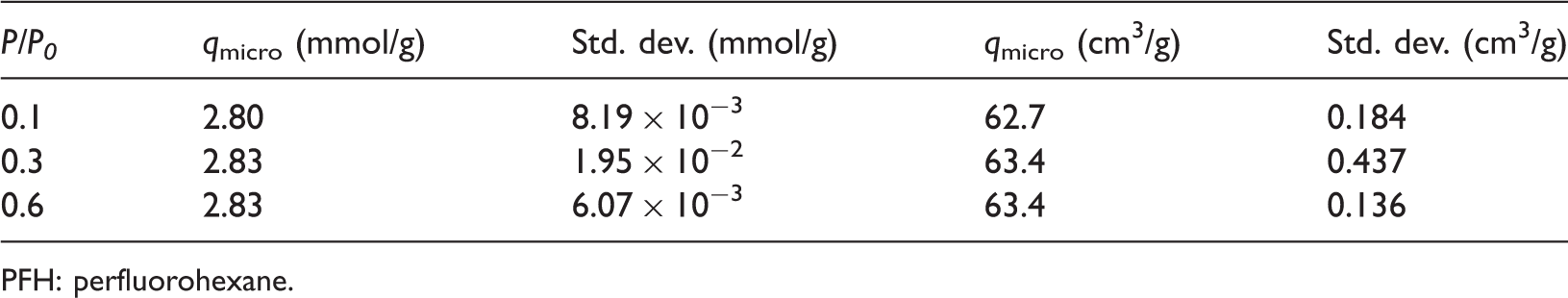
Figure 8 shows the radial distribution functions (RDFs) between adsorbed PFH molecules and C atoms of the BAM-P109 as well as PFH and O atoms situated at the surface of the AC fragments.
RDFs between (a) PFH and C atoms, and (b) PFH and O atoms of the AC.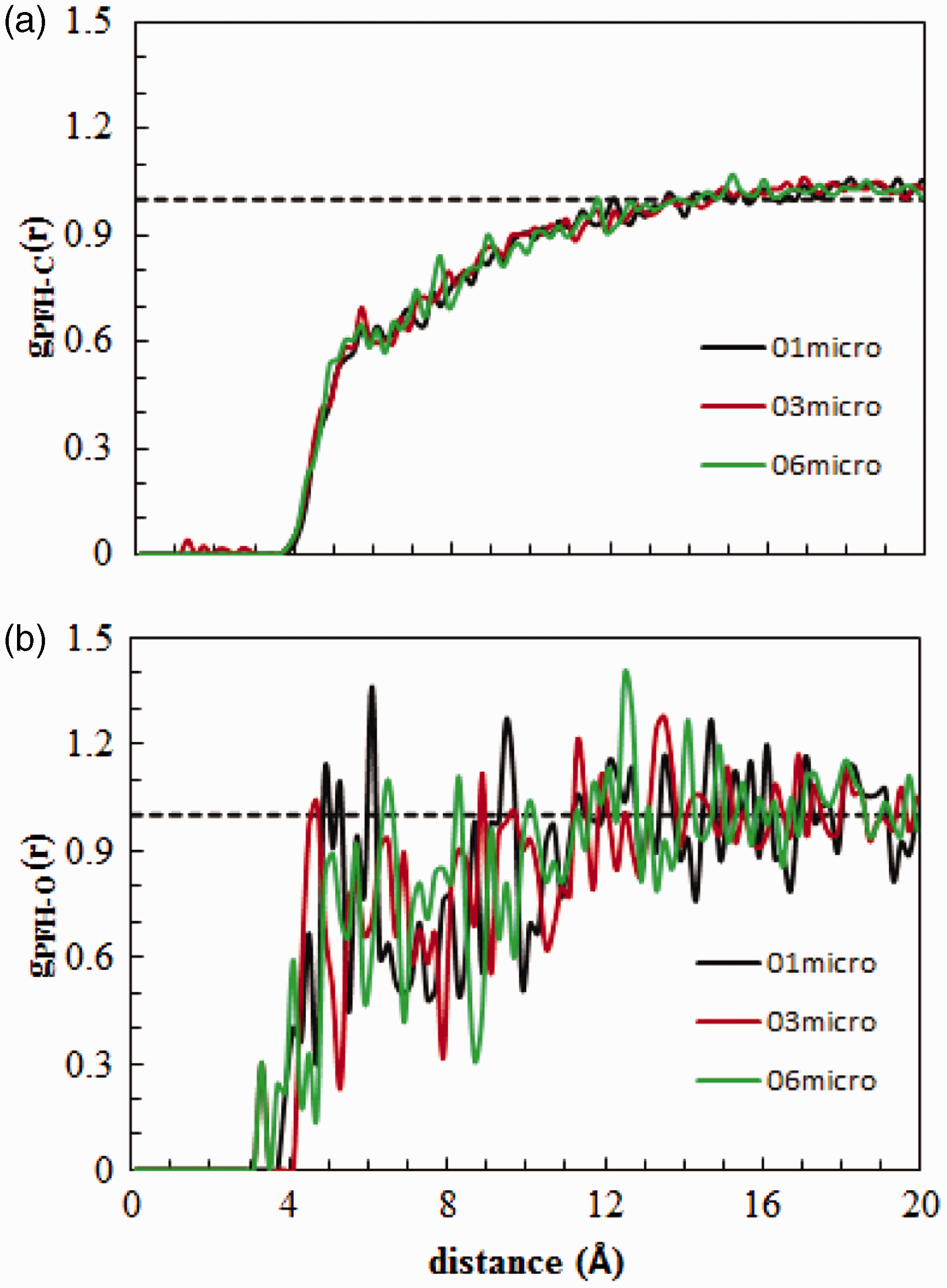
Mesoporous PFH adsorption on BAM-P109
Mesoporous adsorption simulations that were performed at P/P0 = 0.3 and 0.6 are given in this section. MC simulations steps were between for 3 × 106 (for w = 7.3 and 9.7) and 9.5 × 106 (for w = 13.3) steps. Figure 9 shows the PFH concentration profile in the mesopore model simulation box for w = 7.3, 9.7, and 13.3 nm at P/P0 = 0.3 and 0.6. The area between the dashed lines accounted for the graphene sheets. The results show that only surface adsorption takes place at these relative pressures. Hence, the number of molecules can be assumed to be independent of the pore width within the mesoporous range. These runs were sufficient to ensure that no pore filling took place at these pore widths. On the basis of these results, we used an average amount adsorbed to estimate mesoporous adsorption capacity. Thus, the amount adsorbed at the surface (ni) was assumed to be constant at average values (and standard deviations) of 39.1(1.54) and 58.4(2.39) molecules at P/P0 = 0.3 and 0.6, respectively. Next, the PFH density was calculated for pores with different widths varying in regular intervals from 3 to 30 nm. Finally, the mesoporous adsorption capacities at P/P0 = 0.3 and 0.6 were estimated according to equation (1).
Perfluorohexane concentration distribution in three mesopores of w(i) = 13.3, 9.3, and 7.3 nm for relative pressures 0.3 and 0.6.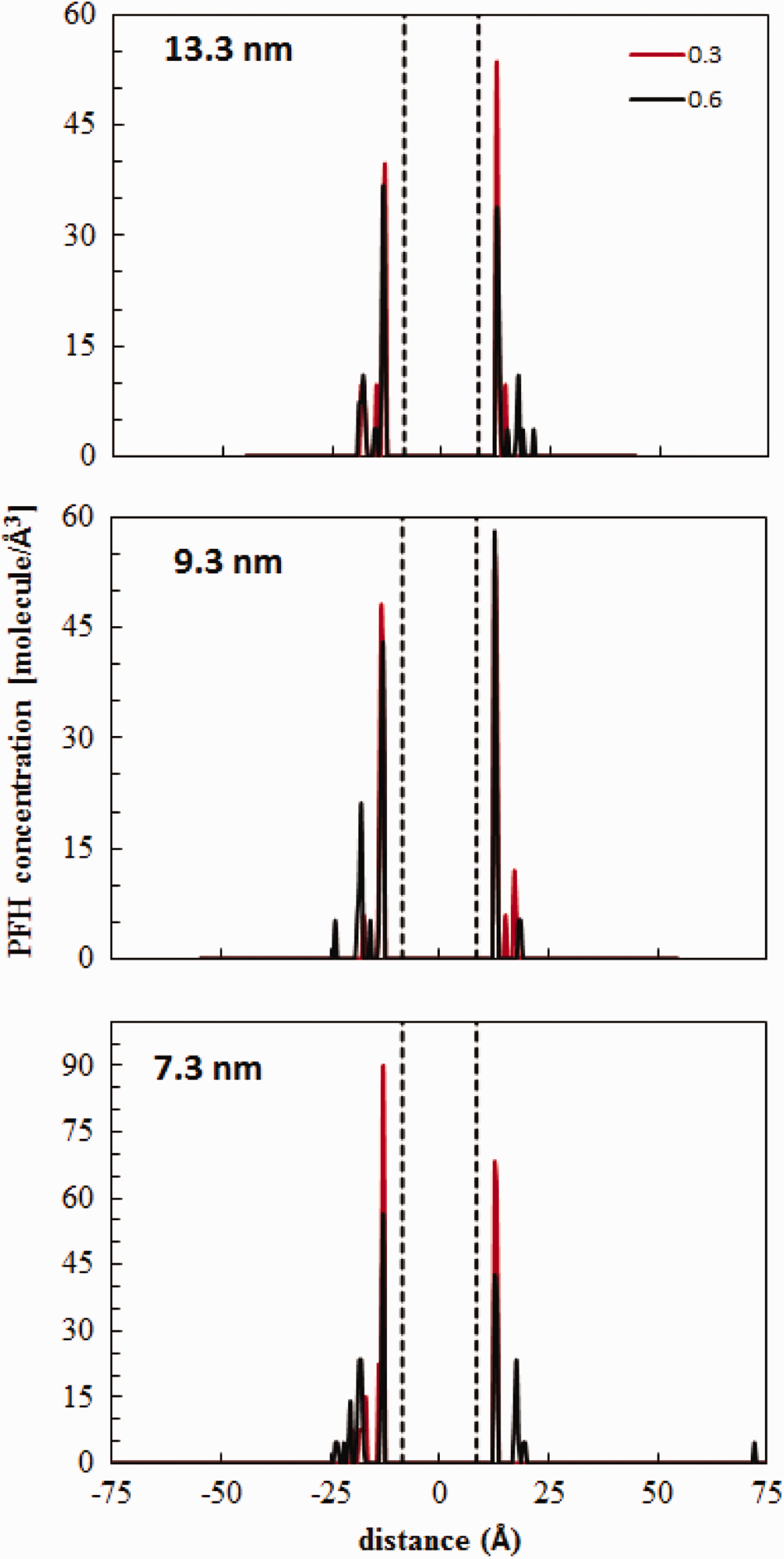
Total PFH adsorption on BAM-P109

PFH adsorption capacity of BAM-P109 at three different relative pressures.

PFH: perfluorohexane.

Total and microporous perfluorohexane adsorption capacity of BAM-P109 at 273 K in comparison with the experimental data (Ross et al., 2016).
Conclusions
We used a two-step procedure to predict the PFH adsorption capacity of the amorphous AC BAM-P109. First, we constructed an amorphous carbon model to account for the microporosity of the material. Next, we used a slit-pore model to account for the mesoporous adsorption capacity of the carbon. The overall adsorption capacity of the material was then estimated by adding together the adsorption capacities calculated for both models. Although the accuracy of the simulations may be improved further by increasing the number of MC steps, they are not estimated to differ significantly from the current ones.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
