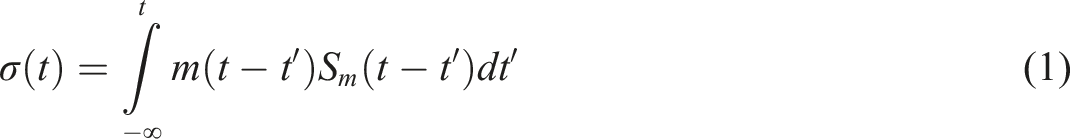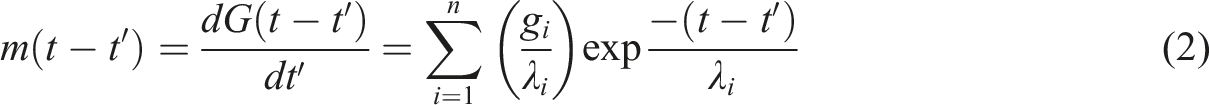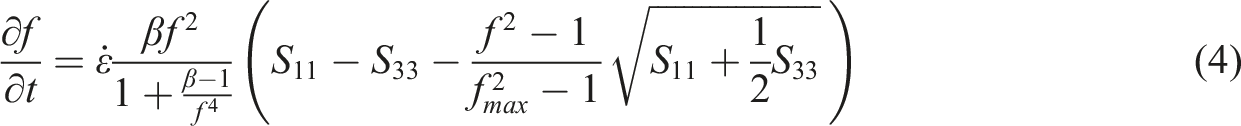Abstract
In this study, we investigated the effect of recycling process on the molecular structure, viscoelasticity and foaming behavior of low density polyethylene (LDPE). A series of LDPE samples with different recycling process was prepared by multiple extrusion using a twin-screw extruder. The molecular weight distribution (MWD) was characterized by gel permeation chromatography (GPC). Wider MWD indicated the generation of higher molecular weight products. Small-amplitude oscillation rheology showed reduced loss factors, indicating that the chain entanglement was more difficult to relax. Moreover, nonlinear viscoelasticity was investigated using elongational rheology and molecular stress function (MSF) model. The results showed a steeper strain hardening exhibited in recycled LDPE. The correlated parameter β in the MSF model indicated that the recycling did not significantly change the branches regularity in LDPE, while the increasing
Introduction
Low density polyethylene (LDPE) is one of the most produced polymers in the world, which total output of the global market reached 6.8 × 107 tons in 2019. 1 It is mainly applied in the field of plastics packaging, such as packing bags, films, cushioning foams and other disposable products.2–4 Compared to polymers in building blocks and mechanical devices, packaging materials have an average life time of 6 months, 5 which makes them subject to only limited chemical variation at end-of-life. These wastes have a high potential for secondary utilization, but there is still a large proportion not involved in effective recycling. 6
The environmental pollution will be reduced7,8 and the consumption of oil resources will be saved if a suitable use can be found for the waste LDPE. Considering that the polymer foams are more profitable with the market demand having been expanding in recent years9,10 and the stable manufacturing of LDPE foams has been well developed in industry, using recycled LDPE to replace virgin materials in foams sector is a potentially viable option. For now, most studies have reported some particular treatment options (such as filling, 11 capacitating, 12 etc.) for wasted LDPE to meet the specific use requirements. Only a few studies investigated the chemical foaming.13,14 However, few literatures have reported the physical foaming of recycled LDPE. Furthermore, there were considerable differences in waste sources in previous studies, which made the experimental results difficult to summarize and promote to other solutions. The contribution of recycling needs to be further evaluated to apply recycled LDPE more universally in manufacturing foams.
Although virgin LDPE have been frequently used in foaming,15,16 the influence of molecular structure changes from recycling still needs to be reevaluated. During recycling,17–19 free radicals are generated when exposed to air at high temperature. The molecular topology is changed due to random free-radical collisions. Products can be divided into two categories based on the perspective of molecular weight variations: (1) free-radical reactions will be terminated after chain scission to obtain low molecular weight (LMW) components; (2) the combination of free radicals will cause grafting reaction 20 to form high molecular weight (HMW) components. The competitive relationship between these two reactions determines the final molecular weight distribution (MWD) and molecular topology. Thus, it is necessary to reevaluate the rheological properties and foamability after recycling.
Rheological assessment has become a common tool in the study of polymers foamability.21–24 The viscoelasticity was used to assess the foaming process. Long branched chain (LCB) structures25–28 are beneficial in foaming as providing more entanglement sites and can inhibit overgrowth of bubbles to regulate the cellular structure.29,30 LDPE is an excellently expandable material,31,32 which is also related to the existence of LCB in its molecules. The recyclability of LDPE on foaming can be highly expected. Therefore, to identify if the recycled LDPE is still suitable for foaming, the changes in molecular structure, rheological behavior and foamability need to be clarified.
In this study, recycling behavior was simulated using multiple extrusions in a twin-screw extruder and co-blended with additives to investigate the influence of recycling on LDPE foamability. First, LDPE samples with different degradation time were prepared and the molecular weight changes were measured using gel permeation chromatography (GPC). The crystallization behaviors were analyzed by differential scanning calorimetry (DSC). Second, linear and nonlinear rheology was measured to evaluate the contribution of molecular structure changes towards chain entanglement. Thirdly, foam samples were prepared using batch foaming so that the influence of chain entanglement behavior on viscoelasticity could be analyzed from the microcellular structure. At last, the essential function of chain entanglement in evaluating the foamability of recycled LDPE was clarified.
Experiments and simulations
Materials and sample preparation
LDPE (Purell 2420F, MFR = 0.75 g/10 min at 190°C) was purchased from LyondellBasell Inc., Netherlands. CO2 (purity: 99.995 wt.%) and N2 (purity: 99.995 wt.%) were purchased from Air Liquide Co.. LDPE samples with extrusion times of 0, 2, 4, 6, 8, 10 were prepared using a twin-screw extruder (Nanjing Giant SJ-20, L/D = 30), named as E-0 (virgin material), E-2, E-4, E-6, E-8 and E-10, with the screw zones temperatures: 160°C, 190°C, 220°C, 220°C and 220°C. The extrusion head temperature was 215°C, main screw speed 160 rpm, and feeder screw speed 16 rpm.
The recycled LDPE in industry is inevitably mixed with other materials, thus the additives existing in the system also need to be taken into account. In this experiment, PP (K8003, MFR = 2.2 g/10 min at 190°C, Sinopec Yangzi Petrochemical Co., China) and activated light CaCO3 (1250 mesh, Suzhou Shenba Fine Chemical Co., Ltd, China) were used as impurities in the recycled LDPE. Under the same conditions, LDPE and PP were separately extruded 7 times to prepare mixed samples. LDPE and PP were mixed according to the ratio of 95/5, 90/10, 80/20 and then to extrude for the 8th time, named: LD95/P5, LD90/P10, LD80/P20, as LDPE/PP samples extruded 8 times. LDPE, PP and CaCO3 were mixed according to the ratio of 92/5/3, 95/5/5, 85/5/10 and then to extrude for the 8th time, named: LD95/P5, LD90/P10, LD80/P20, as LDPE/PP/CaCO3 samples extruded 8 times.
Rheological measurements and model correlation
Small-amplitude oscillation measurements were performed using a rotational rheometer (Haake Mars III, Thermo Fisher Scientific Co., Ltd) with a parallel plate geometry (35 mm in diameter, 2 mm in thickness). The linear viscoelasticity was measured at 180°C using the frequency range of 0.1 to 100 rad/s and the stress of 20 Pa. The geometric samples were prepared by compression molding at 160°C and 6 tons pressure.
Uniaxial elongational rheology measurements were performed using the same rheometer equipped with the SER universal test platform and sheet samples (20 mm × 10 mm × 1 mm) to obtain the nonlinear viscoelasticity at 180°C. Each sample was respectively subjected to four strain rates (0.1 s−1, 0.3 s−1, 1 s−1, 3 s−1). Measurements are all performed in N2 atmosphere to prevent oxidation.
The MSF model was used to calculate the nonlinear viscosity, given by a history integral of this form:
The strain is calculated according to the following equation:
Molecular weight measurement
The molecular weight and molecular weight distribution (MWD) of all samples were measured using a gel permeation chromatograph (PL-GPC220, Agilent Technologics Inc., USA). The experimental temperature was 150°C and 1,2,4-trichlorobenzene was used as the eluent. The flow rate was 1 mL/min, with 0.025 wt.% BHT (2,6-di-tert-butyl-4-methylphenol, also known as butylated hydroxytoluene) added as an antioxidant. Linear polystyrene standards were used to calibrate the curve.
Thermophysical properties
A differential scanning calorimeter (DSC25, TA Instruments, USA) was used. About 10 mg of the samples were weighed and warmed up from room temperature to 180°C, to eliminate the thermal history of samples. The temperature was then cooled down to 40°C at a rate of 10°C/min, and the cooling curve was recorded and held for 5 min to ensure the temperature is stable. Then heat up to 180°C at a rate of 10°C/min, hold for 10 min and record the heating curve. Crystallization temperature (Tc), melting temperature (Tm), crystallization enthalpy (∆Hc) and melting enthalpy (∆Hm) were recorded. The crystallinity (χc) of samples was calculated as follows:
The
Batch foaming at melt state
The foaming of LDPE was carried out in an intermittent foaming system using the rapid pressure relief method. The polymer sample (10 mm × 10 mm × 2 mm) was first placed in a high-pressure foaming kettle and purged three times with CO2, then the system temperature was stabilized at the foaming temperature, and the high-pressure CO2 and N2 were pumped into the kettle. 5 MPa CO2 + 10 MPa N2 are used for a total saturation pressure of 15 MPa to diffuse CO2 and N2 gas into the melt, saturated for 60 min, rapidly drop to atmospheric pressure at a pressure drop rate of 100–150 MPa/s, and then rapidly cool down to room temperature.
Foam morphology characterization
The expansion ratio of LDPE foam (Rv) is defined as the ratio of the bulk density of the LDPE solid sample to the bulk density of the LDPE foam sample, determined by the following equation:
The density of LDPE solid samples and foam samples was determined by the drainage method according to ASTM D792-00. ρf was calculated from precision balance measurements with equation as follows:
The foam samples were quickly quenched after being immersed in liquid nitrogen for 10 min, and the fracture surfaces were coated with Pt spray to enhance conductivity. A scanning electron microscope (SEM, Nova, NanoSEM450, FEI, U.S.) was used to observe and photograph the fracture surfaces. Image Pro Plus (Media Cybernetics, Bethesda, MD) was used for cell statistics, calculated as follows:
Results and discussion
Molecular weight of recycled LDPE
The molecular weight of LDPE was characterized by using gel permeation chromatography (GPC). The results of GPC are shown in Figure 1. The change of MWD illustrates the effect of simulated recycling on the structure of LDPE. The MWD curves of E-2 basically overlap with E-0. Accordingly, the molecular structure is not greatly affected by less than twice recycling. Compared with virgin material, the peak molecular weight (Mp) of multiple recycled LDPE decreased from 1.09 × 105 g/mol to 8.40 × 104 g/mol. The number average molecular weight (Mn) generally decreased, indicating that the total number of molecules per unit mass increased. The degradation as a whole always shows molecular chain scissions. However, it can be found that since E-4, there is not only a decrease of Mn, but also the curve is higher than E-0 on the right side of about 8 × 105 g/mol, which shows an obvious HMW tail. (a) Molar weight distribution of LDPE samples; (b) Average molecular weights of LDPE samples with different extrusion times.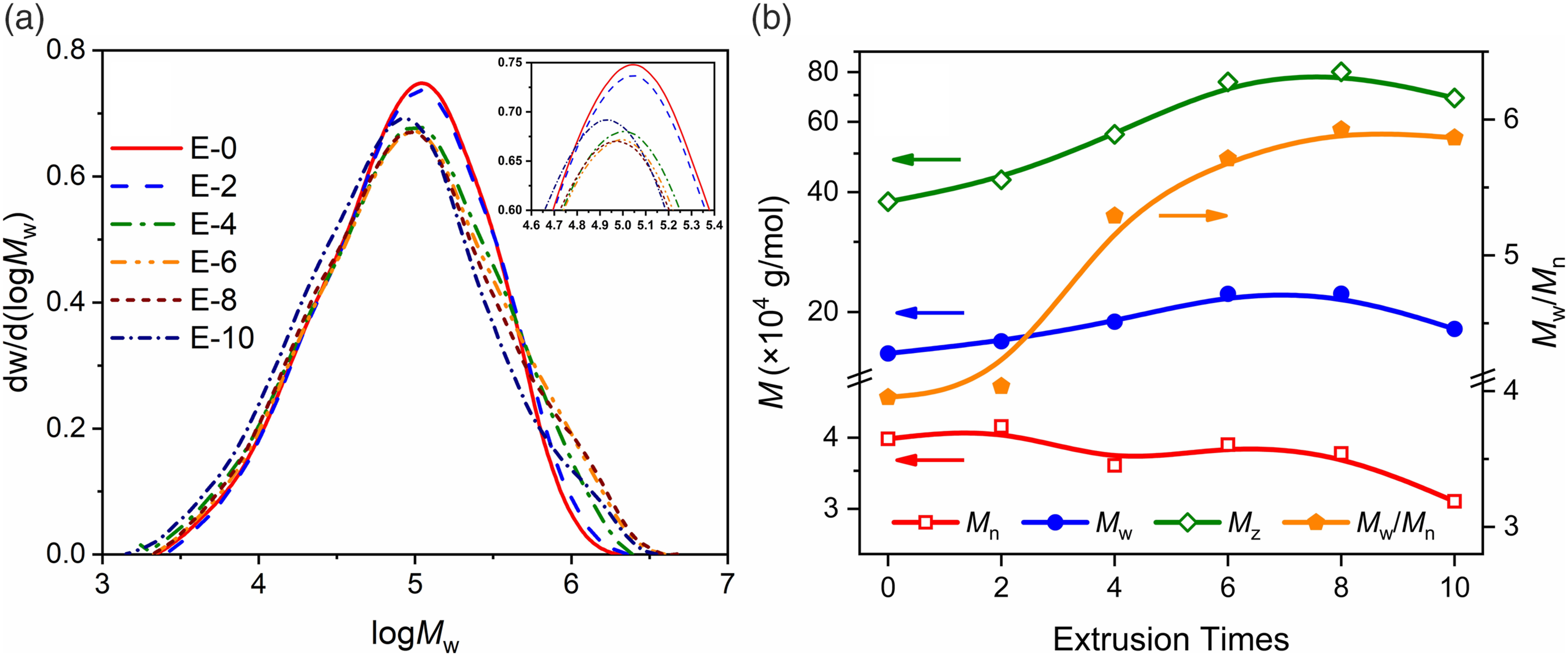
The calculation of the heavy average molecular weight (Mw) and the z-average molecular weight (Mz) is influenced by the molecular molar mass, with higher molecular weight components having higher statistical weights. Thus, Mw and Mz are more sensitive to higher molecular weight components. And both showed significant improvement, peaking in E-8, where Mw increases from 1.57 × 105 g/mol to 2.22 × 105 g/mol and Mz increases from 3.78 × 105 g/mol to 8.01 × 105 g/mol. In the GPC curves, it can be observed that the increased HMW tails in E-6 and E-8 compared to E-0 curves are also reduced in E-10.
The dispersion Mw/Mn increases with the recycling number from E-0 to E-8. Two types of products exist simultaneously in the free radical reaction: the LMW component and the HMW component. The MWD broadens owing to the growth of both components. The result will undoubtedly affect the motility and crystallization behavior of molecules in the melt state. Therefore, an indirect effect on the foaming process is also expected. This is due to the randomness of free-radical reactions.19,20 In a more similar actual recycling, that is, strong shear in a twin-screw extruder, simultaneous molecular chain scissions and random connections are still observed. As the recycling times increases, the obtained HMW fraction will likewise be irreversible oxidation and molar mass reduction. Predictably, there is an optimal time interval for the increase of Mw and Mz by recycling.
Thermophysical properties of recycled LDPE
The DSC calculation results in Figure 2 are listed in Table 1. DSC experiments showed that the crystallization enthalpy of multi-extruded E-8 was increased and the crystallization temperature was slightly shifted upward compared to E-0. It indicates that the tendency of crystallization is stronger for multi-recycled LDPE. (a) Cooling curves; (b) heating curves of samples in DSC. Results of DSC calculation. aPeak of low temperature. bPeak of high temperature.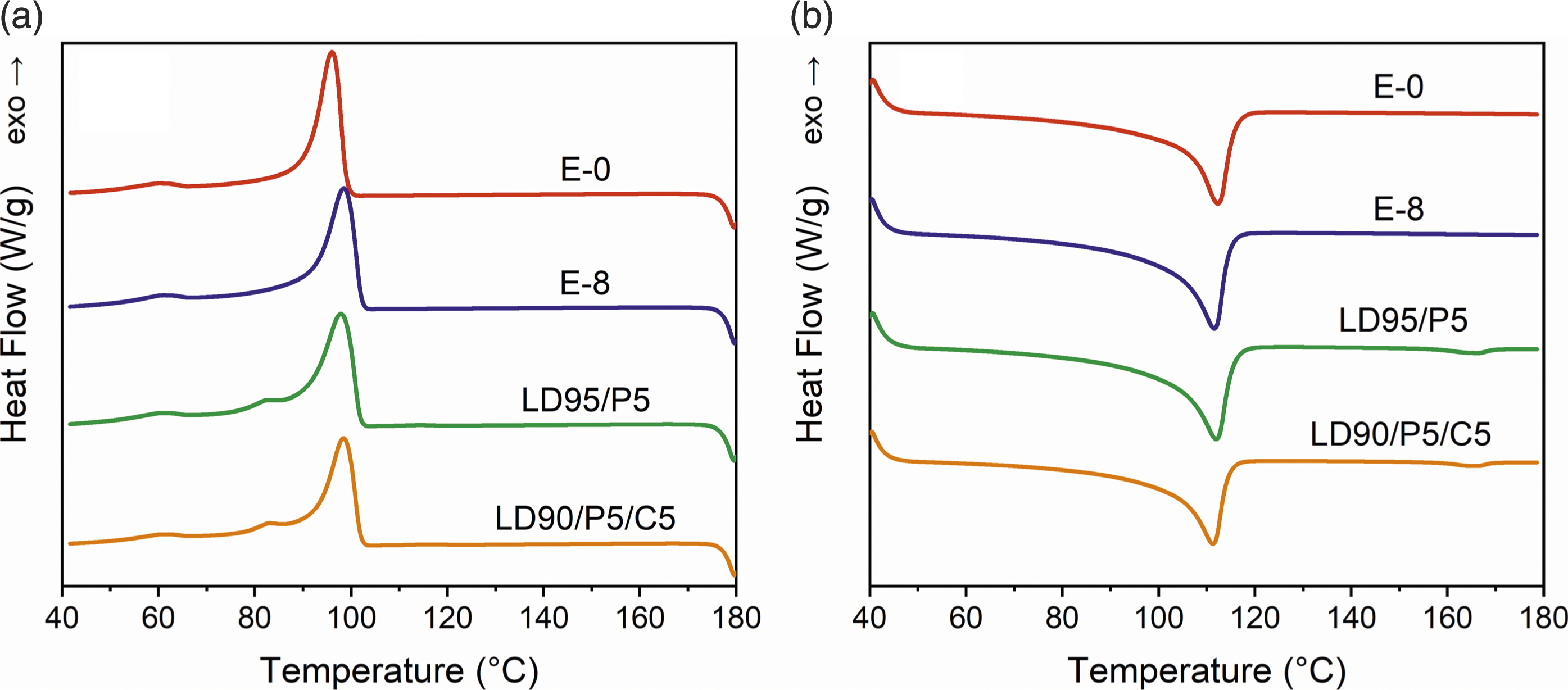

After PP added, no new crystallization peak was observed near the crystallization temperature of PP. It shows that the mixed system had no obvious phase separation and then separate to crystallize. However, a newly emerging peak was observed near 80°C. It is speculated that due to its low concentration and uniform dispersion in the system, PP was difficult to nucleate at its crystallization temperature. Thus PP would be induced to crystallize only when LDPE crystals provided sufficient nucleation sites. 38 In the secondary heating curves of LD95/P5 and LD90/P5/C5, a minor melt peak was observed near 165°C which also indicates that the residual PP crystals melted at this temperature.
Furthermore, the addition of active CaCO3 had no significant effect on the crystallization temperature, as well as no significant change in the peak at 82°C. However, the crystallization enthalpy was further reduced. The crystallization temperature only increased by about 0.6°C, indicating that the induction of heterogeneous crystallization nucleation was also not obvious. The effect was mainly manifested by hindering the orderly collapsation of chain segments, which reduced the crystallinity under the same cooling rate.
Linear and nonlinear rheology of recycled LDPE
In this work, the viscoelasticity of recycled samples was evaluated by using small-amplitude oscillation and uniaxial elongational rheology. Figure 3 show complex viscosity (|η*|) and loss factor (tanδ) of the melts at 180°C. Easily noticed, the viscoelasticity patterns are similar for virgin and multiple extruded samples. The trends of storage modulus G′ and loss modulus G″ are consistent, with not obvious change of |η*|. LDPE is a typical pseudoplastic fluid, so each sample exhibits significant shear thinning behavior. |η*| and shear thinning behavior did not change obviously. It indicates that the heating, oxidation, and strong shear behavior in the experimental range did not lead to serious degradation. Recycled LDPE still has a considerable utilization potential. (a) Complex viscosity |η*|; (b) loss factor tanδ of multi-extrusion samples.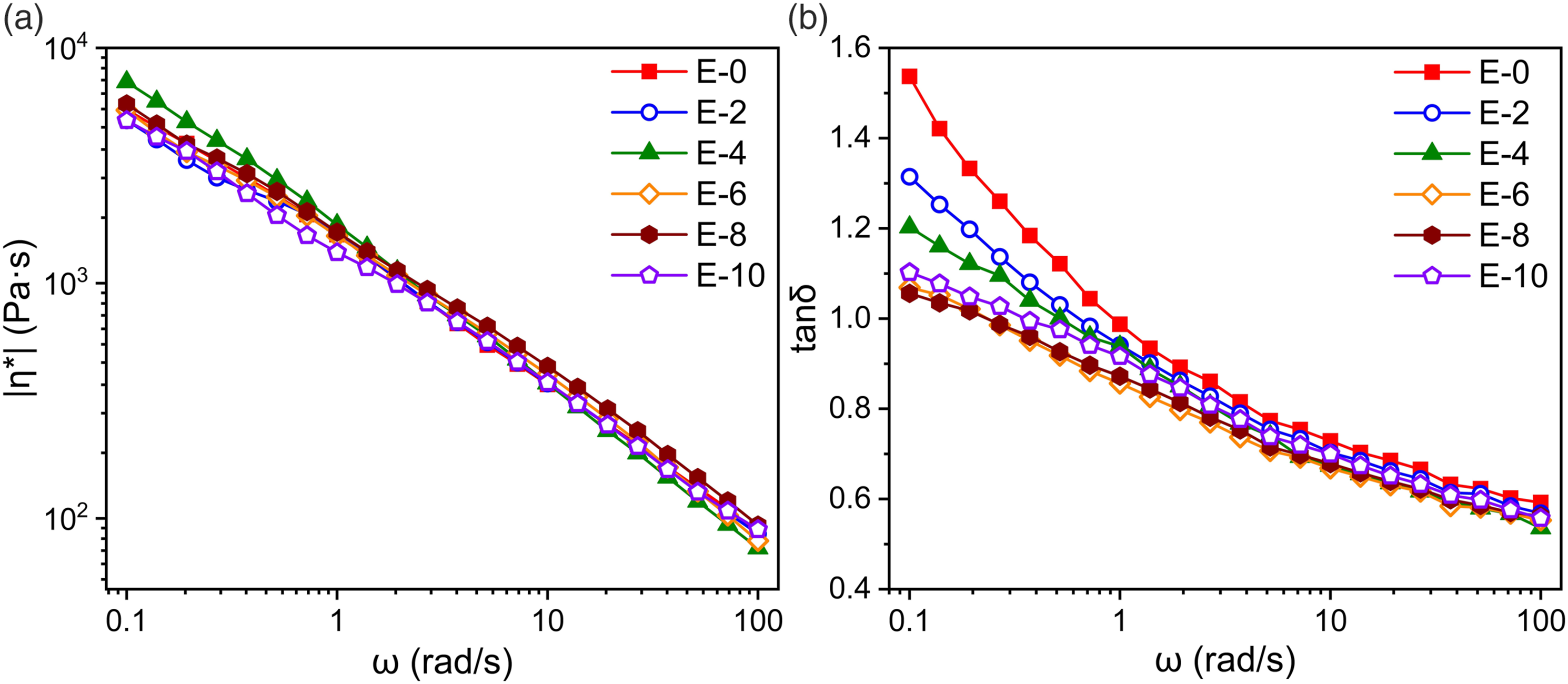
The samples basically maintained essentially chemically stable in this experiment. This is related to the fact that LDPE, as a polyolefin, lacks functional groups that can be easily oxidized and decomposed. Meanwhile, the tanδ gradually drops with increasing recycling times. More notable in low-frequency range, tanδ reduced from 1.5 to 1.0 at 0.01592 Hz. This indicates that the newly formed components with more complex structures have longer relaxation time. The flow properties of the material change from viscous dominant to elastic dominant. This is supported by the relaxation time spectra of multi-extruded samples, which are detailed as Tables S1–S6 of Supporting Information and further to used in the fitting of MSF model.
The nonlinear viscoelasticity of these samples was characterized by uniaxial elongational rheology experiments. As shown in Figure 4, E-0 already exhibits noticeable strain hardening. Strain hardening was further enhanced after several extrusion cycles. Hugo Rolón-Garrido et al.
39
observed similar phenomena in samples after heat-oxygen treatment, and our study further verified that the enhancement of strain hardening remained under conditions closer to the actual. LDPE after oxidation treatment obtained some products with complex structures. This additional fraction contributes significantly to nonlinear rheology, which offsets the tendency of overall molecular weight shift to LMW. Elongational viscosity 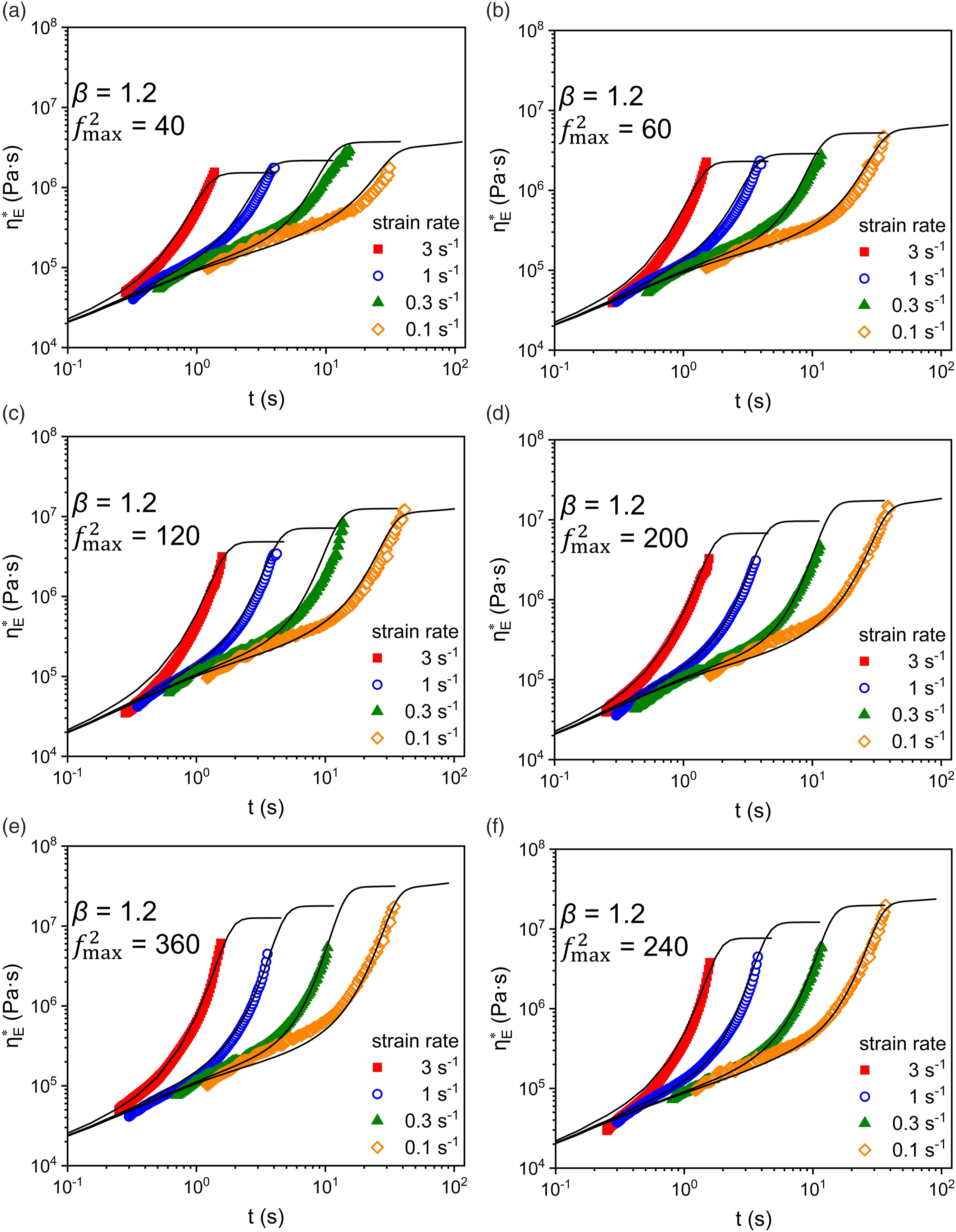
According to the results from MSF model fitting,
Based on the variation pattern of
Rheological analysis was also performed for the blended systems. As shown in Figure 5, after 8 times recycling, the viscosity of PP is significantly lower than that of LDPE, while the shear thinning phenomenon is weaker. On the one hand, this is related to the structure of the virgin material PP itself. On the other hand, since the degradation of PP is dominated by chain-scission. The chain scission during degradation is severe and it is difficult to produce HMW components. Therefore, it is manifested in rheology as a simple viscosity reduction. (a) Complex viscosity (|η*|); (b) loss factor tanδ of LDPE/PP samples with 8 times extrusion.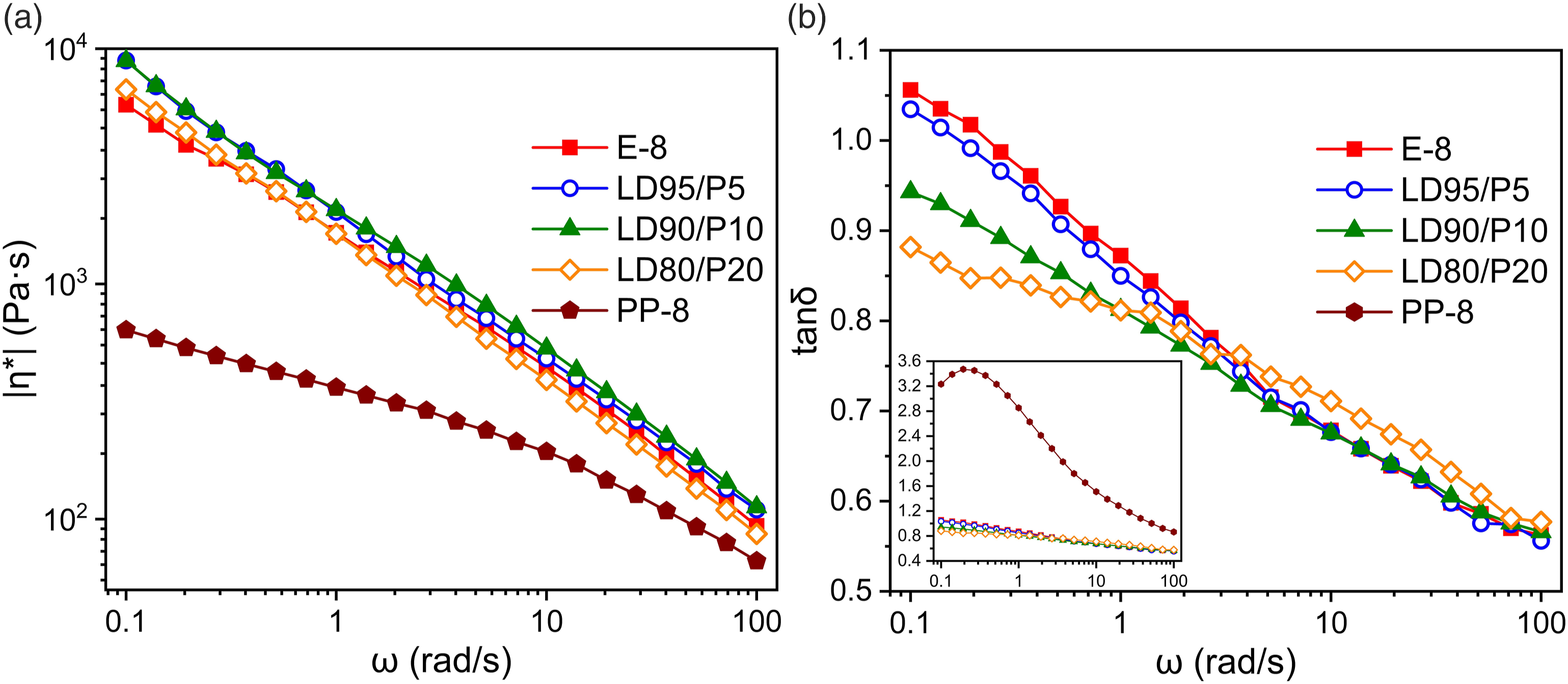
After LDPE/PP blended extrusion, the |η*| appeared slightly elevated. It indicates that the system containing less than 10% of PP can still maintain excellent compatibility. The blended system is molecularly dispersed to have the effect of polymer alloy. Consequently, blending with lower viscosity PP, LDPE instead shows increase in |η*| and decrease in tanδ. Compared to LD90/P10, LD80/P20 showed decreased |η*|, but tanδ showed opposite patterns in high and low frequency parts. The reason for this phenomenon may be that at 20% PP addition, LDPE and PP already exhibit severe phase separation forming the PP phase with lower viscosity. Thus LD80/P20 showed decrease in |η*|. Meanwhile, because its lower viscosity, PP phase has flows more easily under small shear at high frequency. Therefore, LD80/P20 has higher tanδ than LD90/P10. In low frequency part, the presence of PP phase forms phase interface, which prevents the relaxation of LDPE chain segment, making the elasticity more prominent. Therefore, tanδ is slightly lower.
As shown in Figure 6 5% PP content was added 1250 mesh CaCO3 for simulating the inorganic filler actually contained. The linear rheological properties of LDPE/PP system were unchanged with the addition of CaCO3 up to 10%. Small additions of CaCO3 had no obvious influences on linear viscoelasticity. The relaxation time spectra of blended samples demonstrate this as detailed in Tables S7–S13 in Supporting Information. (a) Complex viscosity (|η*|); (b) loss factor (tanδ) of LDPE/PP/CaCO3 samples with 8 times extrusion.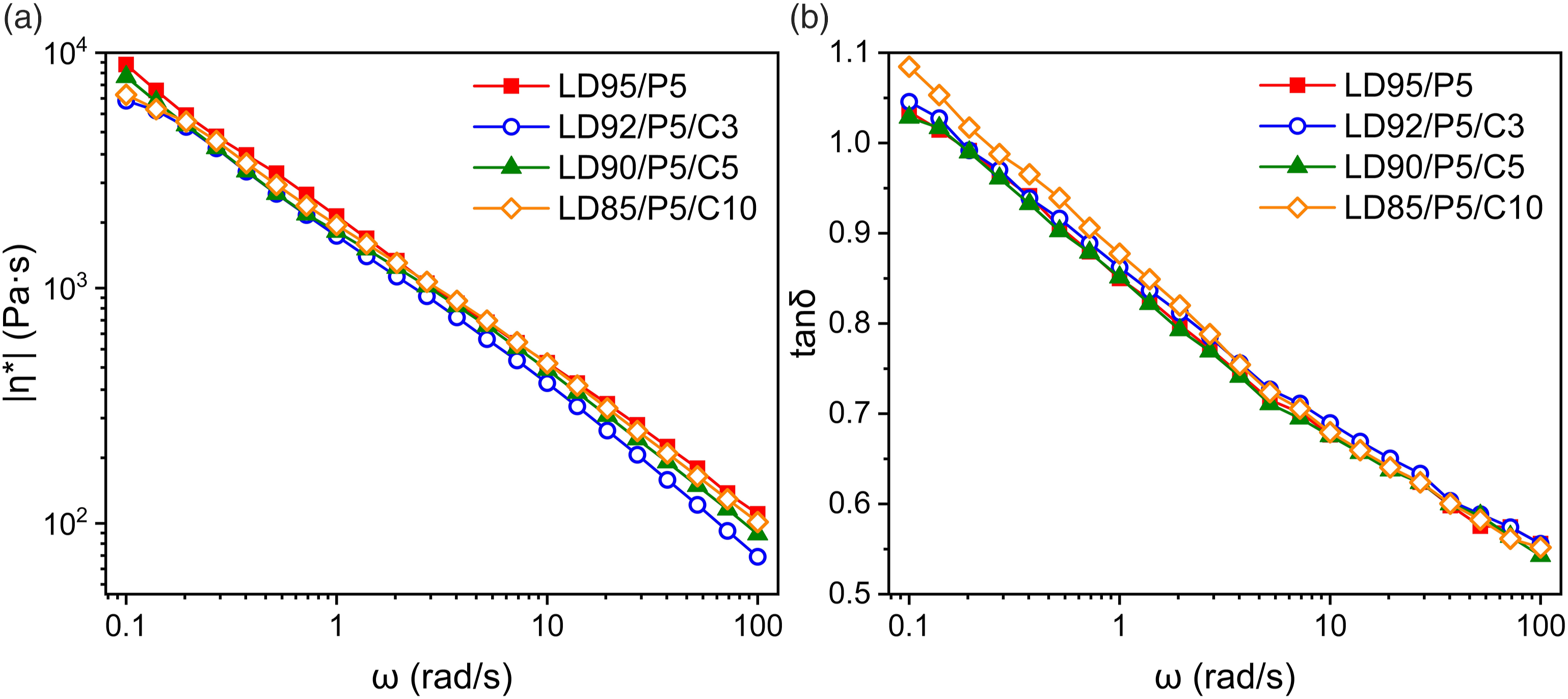
As shown in Figure 7, the addition of PP and active CaCO3 did not change strongly on the strain viscosity curve, indicating a continuous LDPE phase in the system at a large addition of PP and active CaCO3. PP additions within the continuous phase, or even the formation of phase interfaces have little significant impact on entanglement sites in LDPE phase. Both Elongational viscosity 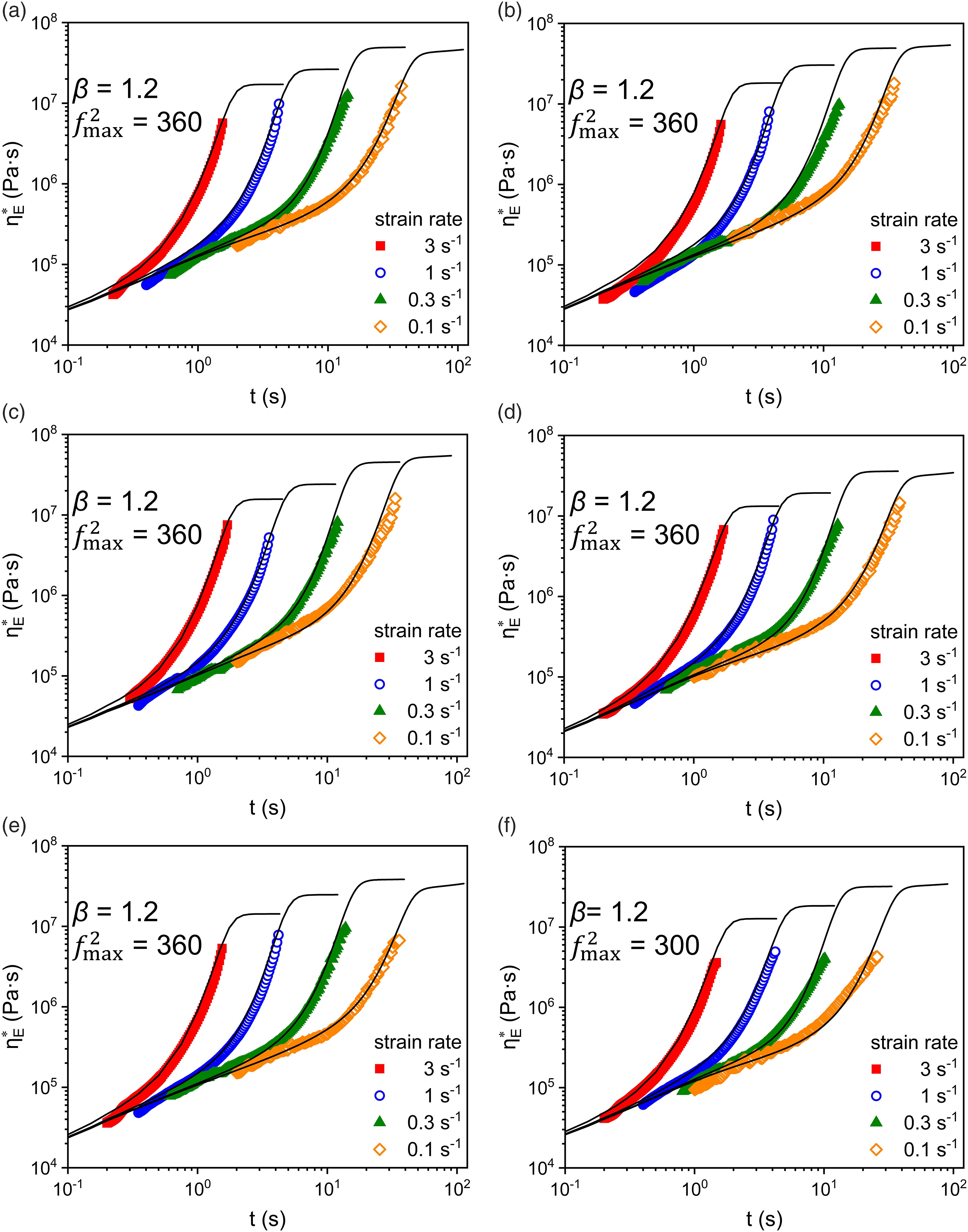
Foaming behavior of recycled LDPE
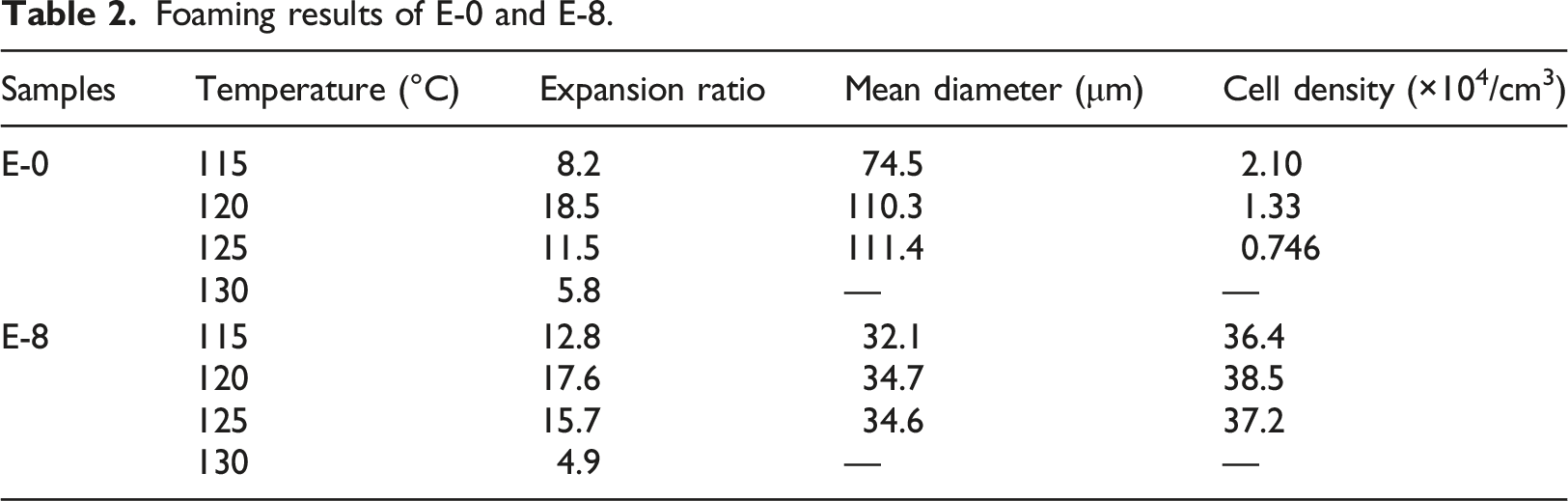
Foaming results of E-0 and E-8.

SEM images of all samples prepared at 115–125°C and LD90/P10 prepared at 120–130°C.
Crystallization and rheology work together to maintain the appropriate resistance during foaming and optimize the cell structure. After recycling, the change of molecular topology led to more pronounced elongation hardening phenomenon and also improved crystallinity. During the bubble growth period with high deformation rate, the maintenance of cells structure relies mainly on the viscosity improvement caused by strain hardening. On the one hand, it serves to inhibit the rupture of cell walls, which reduces the gas escape rate. On the other hand, the stress near the cell wall during the growth process is higher, which can reduce the threshold nucleation radius to induce cell nucleation. 40 Also the improved crystallinity will further enhance the melt strength during the cooling and molding of bubbles. In addition, the bubble wall of larger cell diameter has higher deformation degree suffering more obvious stretching effect, so the growth resistance is greater. The strain hardening phenomenon also plays a role in reducing and homogenizing the cell diameter. Arguably, for indicators of industrial interest, such as production stability and mechanical properties, recycled LDPE undergoing thermal-oxidative and mechanical degradation instead has some advantages compared with virgin materials.
Besides, the existence of impurities complicated the foaming results. As shown in Figure 9, the LDPE/PP system appeared obvious changes in expansion ratio and cell structure. Comparing LD95/P5 with E-8, the cell diameter of the sample at 120°C is enlarged, meanwhile the expansion ratio is almost constant. However, the addition of PP clearly improves the expansion ratio at 125°C. It indicates that minor PP addition is helpful to maintain the cell structure under high temperature. The upper limit of foaming temperature was therefore increased. Proper increased crystallinity helps to set the bubbles and then to rise the upper limit of foaming temperature. Expansion ratio of (a) LDPE/PP; (b) LDPE/PP/CaCO3; mean diameter of (c) LDPE/PP; (d) LDPE/PP/CaCO3; cell density of (e) LDPE/PP; (f) LDPE/PP/CaCO3.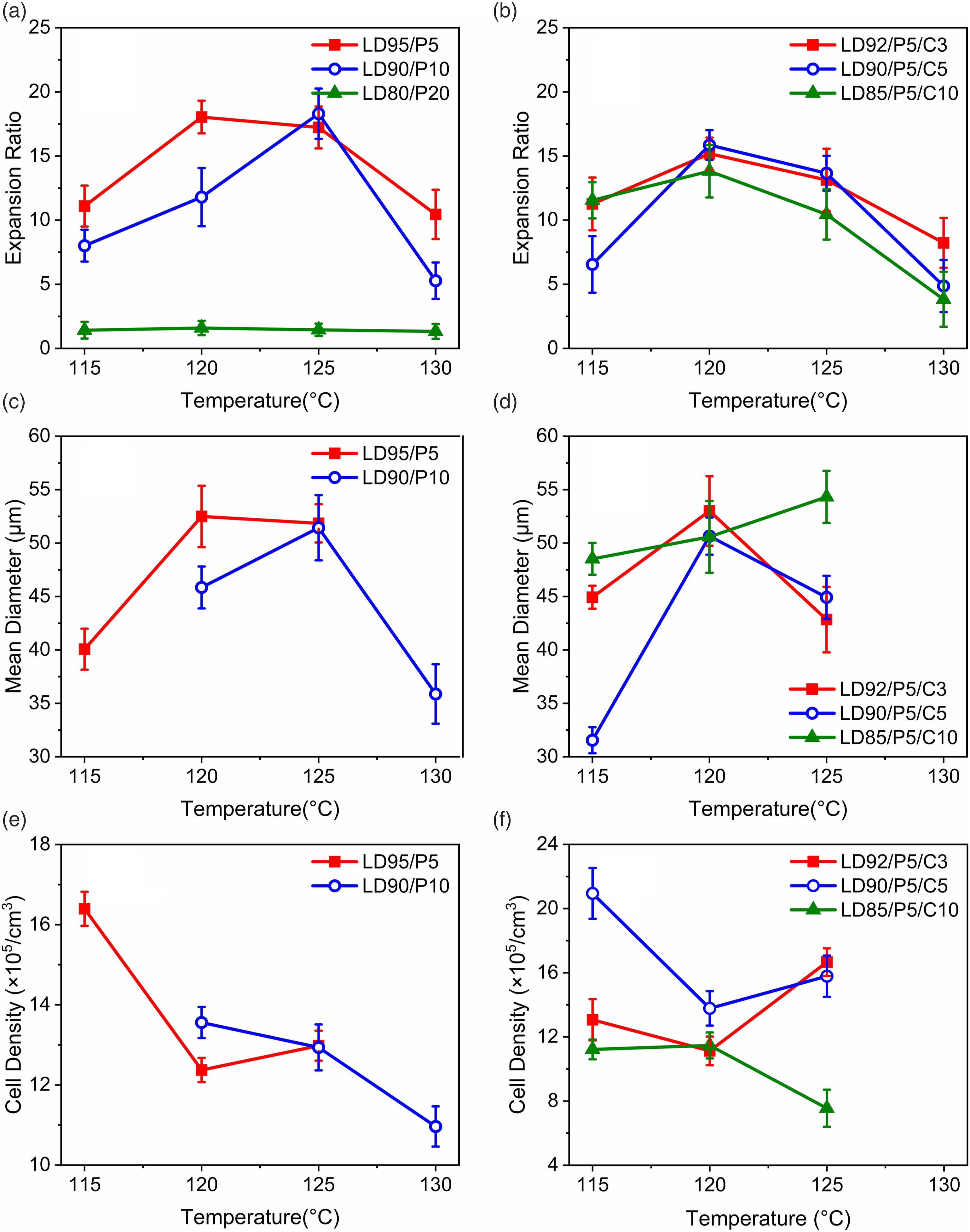
The expansion ratio curve of LD90/P10 indicates that the addition of PP also raises the lower limit of foaming temperature. Since the Tg of PP is higher than that of LDPE and they remain well compatibility at this content. While raising the crystallinity, the addition of PP will also improve the overall Tg. Accordingly, in LD90/P10, the foaming temperature shows a low temperature performance at 120°C and the optimum expansion ratio temperature moves up to 125°C. The interval is narrower compared to LD95/P5, which is negative for continuous production.
With the PP content further increased to LD80/P20, samples did not change substantially in the rheological tests, but varied enormously in the foaming experiments. The expansion ratio of LD80/P20 did not reach 2 times at all temperatures. This result has no application value for high expansion ratio materials like LDPE. LDPE can ensure some compatibility at low PP content, but as the PP content is comparable, the system without additional fillers is inevitably difficult to be gradually compatible, the system without additional fillers is inevitably difficult to be compatible. The high PP concentration leads to agglomeration and there is already obvious phase separation in the system. When the bubble is growing, the blowing agent will leak severely at the phase interface so that the driving force is insufficient to expand the melt. For the recovery of such thermodynamically incompatible systems and application in foaming industry, limiting the proportion of incompatible components, or the addition of fillers is optional, otherwise serious loss of performance is unavoidable.
The foaming results revealed that containing a small amount of active CaCO3 had only a little changes on expansion ratio and cellular structure. The excessive addition of CaCO3 led to the aggregation and enlarged diameter of bubbles. The expansion ratio loss was considerable, and increased open cells were noted. It suggested that the introduction of activated CaCO3 had stronger effect on destabilizing the cell wall than nucleation in foaming. Presumably, the interface energy barrier was not high because of the excellent compatibility of active CaCO3, thus the nucleation effectiveness was not obvious. On the one hand, the excessive filler would further enhance the melt strength of matrix, increasing the growth resistance and reducing the multiplicity. On the other hand, CaCO3 is selected with 1250 mesh, which the diameter of many particles is in the similar magnitude with the thickness of cell walls, which can be observed in Figure 8. The stress near CaCO3 particles in the melt will be different from other parts, which may destroy the stress uniformity of the cell wall, then affect the stability. Therefore, the percentage of waste polymer containing inorganic fillers needs to be strictly controlled in recycling, in order to ensure that the total amount of fillers does not seriously affect the quality of foam products.
Conclusion
In this study, LDPE was repeatedly extruded in a twin-screw extruder to investigate the effect of recycling on its rheological properties and foamability from the viewpoint of molecular structure change. First, in GPC, Mn reduced slightly from 1.09 × 105 g/mol to 8.40 × 104 g/mol, but Mw increased from 1.57 × 105 g/mol to 2.22 × 105 g/mol, indicating that higher molecular weight fractions were formed during recycling. Second, in viscoelastic rheology, tanδ at 0.01592 Hz decreases from 1.5 to 1.0, which shows a transition from viscous to elastic state. Besides, for the parameters calculated using MSF model in nonlinear viscoelastic rheology, β remained constant at 1.2, but
Supplemental Material
Supplemental Material - Effect of thermal-oxidative and mechanical degradation of recycled LDPE on foaming
Supplemental Material for Effect of thermal-oxidative and mechanical degradation of recycled LDPE on foaming by Huanyu Zou, Jiawei Lu, Pengfei Zhou and Tao Liu in Journal of Cellular Polymers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (2016YFB0302200).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.