Abstract
Currently for engineered nanomaterials (ENMs), there are no alternatives to bioaccumulation testing in fish as prescribed by OECD TG 305. New approaches are being sought, in order to reduce the numbers of animals used and address the ethical concerns associated with the use of vertebrates in such testing. This study aimed to demonstrate the value of an alternative method — the in chemico digestibility assay — as a tool to assess the bioaccumulation potential of ENMs. Titanium dioxide ENMs, or their equivalent bulk form, were incorporated onto a commercial fish diet and the fish digestion process was simulated in vitro at pH 2 (simulated stomach phase) and pH 7.8 (simulated intestinal phase). Only the 1000 mg/kg dry weight feed treatment showed measurable Ti release in the stomach phase, with the results being similar for the TiO2 ENM and the bulk form. More Ti from TiO2 was released from the feed during the intestinal phase of digestion compared to the stomach phase, but there were no statistically significant differences between either the ENM or bulk form. There was higher incidental Ti release from untreated control feed in the intestinal phase compared to the stomach phase. On a percentage basis, the stomach generally showed a statistically significant digestibility of around 3% of the total Ti in the feed, compared to around 7% in the intestine. The in chemico digestibility assay shows potential for use in the determination of the bioaccessible fraction of ENMs, and could be a useful screening tool to identify ENMs of concern for bioaccumulation.
Introduction
New substances, including engineered nanomaterials (ENMs), are assessed for their environmental safety by using standardised regulatory test methods. The key triggers of concern in environmental risk assessment are persistence in the environment, bioaccumulation potential and toxicity to wildlife (so-called ‘PBT’), with the measurement of bioaccumulation often involving the use of fish. For novel materials such as ENMs, a wealth of data currently exists on their persistence1,2 and toxicity,3,4 but much less is known about their ability to bioaccumulate.3,5,6 One of the most widely used methods for determining the bioaccumulation potential of chemicals is the Organisation for Economic Co-operation and Development Test Guideline (OECD TG) 305, which typically requires 150 fish per test, with exposures lasting up to 28 days.7,8
In the EU alone, 136 new ENMs were recently listed, in addition to the 341 unique materials and particles already listed for use in the EU. 9 The European Union Observatory for Nanomaterials (EUON) Database lists 336 substances (https://euon.echa.europa.eu/search-for-nanomaterials; accessed January 2024). Under the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) regulations, any substance with an annual production of more than one tonne requires a hazard classification, and ENMs produced in quantities of 100 tonnes/year or more may require a bioaccumulation test. This burden of work will inevitably increase the use of fish for in vivo testing according to OECD TG 305, as well as similar testing to comply with other regulations elsewhere in the world. Clearly, a testing strategy that considers the replacement, reduction and refinement (i.e. Three Rs) of animal use when testing ENMs is advantageous.
Recently, a tiered testing strategy has been suggested for screening ENMs prior to in vivo bioaccumulation testing,5,10,11 and this approach is also being considered by the Working Party on Manufactured Nanomaterials at the OECD. 12 The overall aim of the scheme is to replace or reduce the use of TG 305 (which uses live fish), whilst still ensuring chemical safety for ENMs. There are four proposed tiers, with the final tier being the in vivo testing on fish as a last resort. The first tier uses environmental chemistry triggers specific to ENMs, such as particle settling and dissolution rates, to highlight ENMs of concern. The second tier involves alternative bioaccumulation tests using invertebrates (e.g. earthworms, 11 freshwater amphipods 13 ), the application of in silico models to predict the bioaccumulation potential of ENMs, and/or existing data from bioaccumulation studies on invertebrates or fish cell lines. The third tier has two aspects: the first step is an in chemico ‘digestibility assay’ that simulates the digestion of ENM-supplemented diets in the gut lumen of fish in order to calculate the bioaccessible fraction; the second step is an ex vivo measurement of uptake into the gut by using a gut sac method. 14 Only materials of the highest concern would then go on to Tier 4. The fourth tier is an in vivo test (e.g. OECD TG 305), involving the use of fish. 10
The idea of using in chemico digestibility assays for predicting bioaccessible fractions, and therefore aspects of the safety of substances, is well-established. For example, the assays have been used to predict the hazard to humans from the incidental ingestion of metal-contaminated soils (see Oomen et al. 15 and Reis et al. 16 ). Some of the methods have been standardised (e.g. the ‘BARGE’ method 17 ), and used for the testing of some ENMs. 18 In the case of ENMs, with appropriate modification of the method for particulate materials, it has been possible to determine the bioaccessible fraction of ENM-contaminated feeds in the simulated gut lumen of fish (e.g. copper oxide (CuO) ENMs 19 and Ag-containing ENMs 20 ). This approach simulates the gastrointestinal lumen conditions of a carnivorous fish, such as rainbow trout, through the use of two ‘compartments’: one to mimic the stomach (pH 2) and the other to mimic the intestine (pH 7.8). The rainbow trout is also used routinely for the in vivo fish bioaccumulation test, i.e. OECD TG 305. The in chemico digestibility assay for ENMs has shown significant correlation with the in vivo data on the bioaccumulation of metals from ENMs in fish, 5 highlighting its utility as a screening tool and perhaps as an alternative to in vivo fish testing.
However, for wider acceptance of such methods in a regulatory context, data on different materials are still required. So far, most studies have been on ENMs that release dissolved metals by dissolution, or are synthesised from known toxic substances. Data are also needed on apparently ‘inert’ materials expected to be of low hazard, and especially those that have low solubility within the gut, such as titanium dioxide (TiO2). The aim of this study was to perform the in chemico digestibility assay on a relatively inert ENM, TiO2, and compare its behaviour to that of the TiO2 bulk form. In this context, the ‘bulk’ form has a particle size at the micron scale (above 0.1 μm) and the ENM is within the nano scale (i.e. 1–100 nm). In addition, the previous reports on the use of the digestibility assay have used a single concentration of ENM in the diet to inform on the in vivo bioaccumulation potential (e.g. Clark et al. 20 ). However, bioaccumulation potential may be concentration-dependent for metallic materials. Here, testing with the digestibility assay was extended by using Ti materials over a range of concentrations (10, 100 and 1000 mg/kg dry weight (dw)) to establish whether a threshold of the bioaccessible fraction can be calculated and used to help inform regulatory decision making regarding the need for further in vivo testing.
Materials and methods
Diet formulation and nanomaterial characterisation
The TiO2 ENM used in the present study was supplied by the NanoSolutions EU project (https://nanosolutionsfp7.com/), and previously reported and characterised in Vassallo et al. 21 Briefly, the TiO2 nanoparticles (NPs) were uncoated (Lot YF1310291; PlasmaChem GmbH, Berlin, Germany) with an average diameter of 10–20 nm, a surface area of 98 ± 10 m2/g, and a mean aggregate size in 0.90% NaCl of 36.00 ± 1.15 nm. The bulk form of TiO2 had a nominal size of 150 nm ± 42 nm (Acros Organics 277370010, Lot A0224336) with 98.0–100.5% purity, and a mean aggregate size in 0.90% NaCl of 301.00 ± 27.61 nm. 21 The bulk form of the material consisted of a 75% anatase and 25% rutile crystal mix, to match the nanomaterial form.
The two forms of TiO2 material were supplemented into diets, according to the method of Clark et al. 20 Briefly, a commercial fish diet (Skretting Elite Trout Pellets, size 2.5 mm), containing (dry weight) 42–45% protein and 21% oil, was used. To make 100 g of each experimental diet, a 10 ml suspension of the relevant TiO2 was added to 90 g of the commercial feed. The TiO2 suspensions contained either 1, 10 or 100 mg of TiO2 NPs or bulk form TiO2 powder, suspended in ultrapure water. The suspensions were sonicated (FB15048 ultrasonic bath, 35 W; Thermo Fisher) for at least 1 hour prior to adding to the pellets. Once added, the suspension was allowed to soak into the pellets and then they were mixed carefully, but thoroughly, for 5 minutes using a commercial food mixer (Kenwood KM810/KM816, 2004). The pellets remained intact during this step. The TiO2 materials were then immediately sealed onto the pellet surface through the addition of a 10% w/v gelatine topcoat. The gelatine solution was prepared by adding 1 g of porcine gelatine (> 98% purity, Sigma-Aldrich) to 10 ml of ultrapure water, and dissolved by heating in a water bath set to 50°C. The solution was allowed to cool before being added to the pellets. The coated pellets were then dried overnight in a 45°C incubator. The measured Ti concentrations in the prepared pellets, which should be 60% less than the nominal dose of TiO2 compound because of the stoichiometry of the metal oxides and their hydroscopic nature, were determined by metal analysis (see below).
In chemico digestibility assay
Seven diets were prepared: one control (no added TiO2); three each containing 10, 100, 1000 mg/kg dw of TiO2, as ENMs or as the bulk form. The in chemico digestibility of each of the diets was assessed. To understand the behaviour of the materials in the gastrointestinal tract of carnivorous fish, two compartments were simulated in 0.9% NaCl: the stomach (in which the NaCl was adjusted to pH 2 with dilute nitric acid) and the intestine (in which the NaCl was adjusted to pH 7.8 with 2 M NaOH). Twenty millilitres of the respective saline solutions were dispensed into a series of 50 ml Falcon tubes, along with ∼1 g of each diet (n = 3 tubes/treatment), then rotated at 30 rpm for 4 hours (Stuart Tube Roller STR6) at room temperature. At 1, 2 and 4 hours, a 0.5 ml sample was taken from each tube, ensuring that no undigested pellets were collected in the solution. These 0.5 ml samples were each transferred into separate 15 ml centrifuge tubes, to be further processed for metal analysis.
Total Ti analysis
The diets were checked prior to the experiments, in order to determine the total Ti metal concentrations in each. A 250 mg sample (n = 5) of each diet was transferred into a Falcon tube with 5 ml of concentrated nitric acid, and placed in a water bath at 90°C for 4 hours to fully digest. Following digestion, the samples were allowed to cool, diluted to 50 ml with ultrapure water, and analysed for total Ti by inductively coupled plasma optical emission spectroscopy (ICP-OES) (iCAP 7400 ICP-OES, Thermo Fisher Scientific). The 0.5 ml saline samples collected during the digestibility assay were diluted to 2 ml with 1.5 ml of a solution containing 2% v/v Triton X-100 and 5% v/v nitric acid. 22 These samples were kept in the fridge overnight, before Ti determination by ICP-OES. All the samples were compared to matrix-matched standards, with standard checks run every 10–15 samples. The wavelength for Ti analysis was 334.931 nm. The limit of detection for the Ti metal analysis was calculated to be 0.13 mg/kg dw, based on the lowest standard. Measured total Ti in the feed was expressed as mg/kg dw.
Total Ti concentrations in oven-dried TiO2 powders.
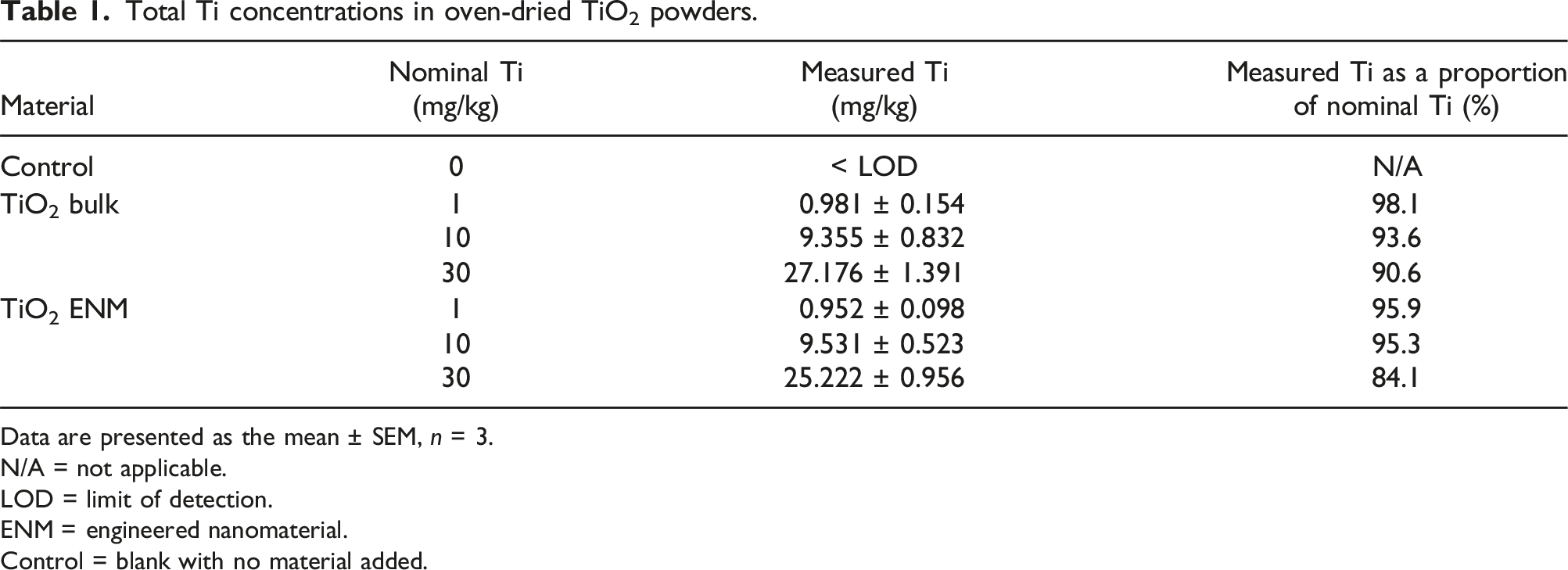
Data are presented as the mean ± SEM, n = 3.
N/A = not applicable.
LOD = limit of detection.
ENM = engineered nanomaterial.
Control = blank with no material added.
Measured Ti concentrations in feed treated with oven-dried and non-dried TiO2 powders.
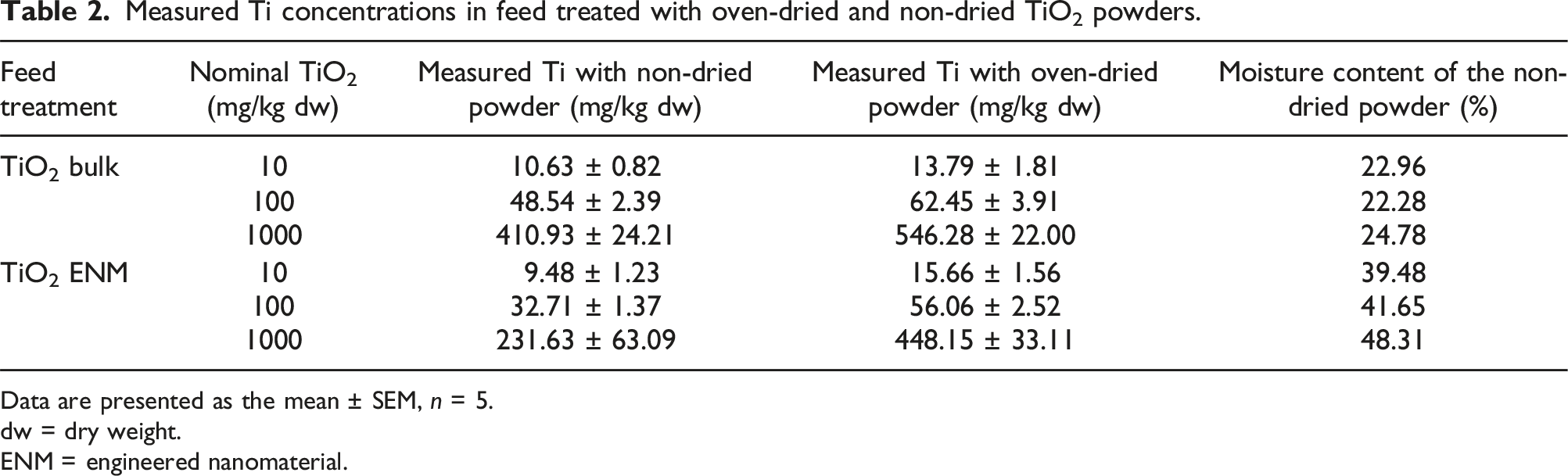
Data are presented as the mean ± SEM, n = 5.
dw = dry weight.
ENM = engineered nanomaterial.
Expected and measured total Ti concentrations in the treated diets.
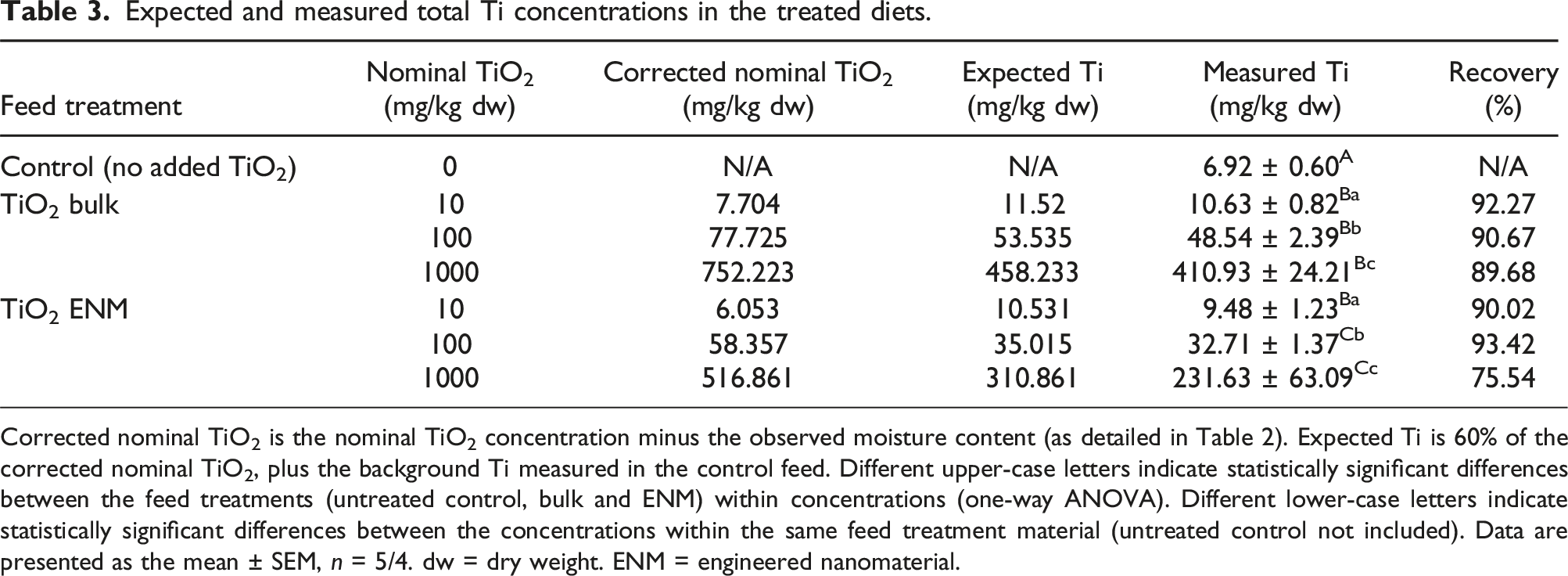
Corrected nominal TiO2 is the nominal TiO2 concentration minus the observed moisture content (as detailed in Table 2). Expected Ti is 60% of the corrected nominal TiO2, plus the background Ti measured in the control feed. Different upper-case letters indicate statistically significant differences between the feed treatments (untreated control, bulk and ENM) within concentrations (one-way ANOVA). Different lower-case letters indicate statistically significant differences between the concentrations within the same feed treatment material (untreated control not included). Data are presented as the mean ± SEM, n = 5/4. dw = dry weight. ENM = engineered nanomaterial.
Statistics and calculations
The bioaccessible fraction of Ti in the feed was calculated from the measured amount of Ti released into the digestion solution (simulated gut lumen) after 4 hours, relative to the total Ti concentration determined in the feed. This assumed bioaccessible fraction was expressed as a percentage, calculated as the total mass of Ti released into the digestion solution divided by the determined mass of Ti in 1 g of feed, multiplied by 100%. Statistical analysis of all data was performed with SigmaPlot version 14.5. After descriptive statistics, the data were tested for equal variance by using the Brown–Forsythe test and normality was assessed via the Shapiro–Wilk normality test. Non-parametric data were log10-transformed if necessary. Statistical tests were then performed, either via a one-way ANOVA test for the measured feed concentrations and the differences between treatments across pH solutions, or a two-way ANOVA test for differences between time and treatment. Where data could not be transformed, the Kruskal–Wallis test was used. The default 5% rejection level was used for the statistics. The p values reported are for the Holm–Sidak test, or Tukey test for non-parametric data where relevant.
Results
Measured total Ti concentration in the diets
The control diet, as expected for animal feed, contained some trace amounts of Ti, at a level of around 7 mg/kg dw (Table 3). All of the TiO2-treated diets showed significantly elevated Ti concentrations, as compared to the control (untreated) diet, confirming that Ti absorption had occurred during the preparation of the diets (Table 3). Serial increases in measured Ti were observed in the different supplemented diets, with ∼10-fold differences in the measured Ti concentrations. When nominal TiO2 concentrations were corrected for moisture content (see Tables 1 and 2), the percentage recovery of measured Ti from the experimental diets relative to the expected total Ti, was within an acceptable range.
Behaviour of the TiO2-treated feed in the simulated stomach phase at pH 2
Total Ti released from the TiO2-treated feed in the simulated stomach phase at pH 2.
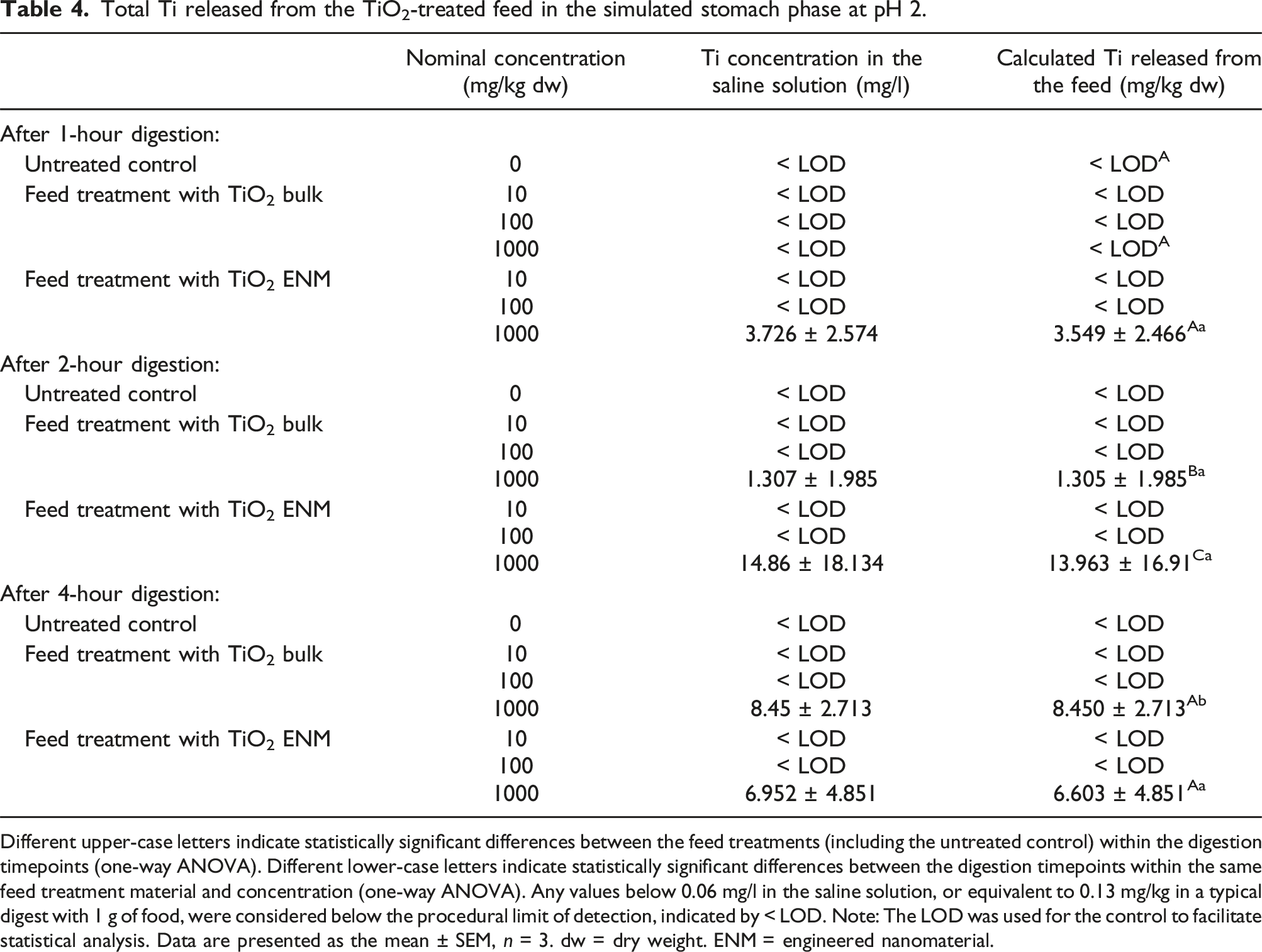
Different upper-case letters indicate statistically significant differences between the feed treatments (including the untreated control) within the digestion timepoints (one-way ANOVA). Different lower-case letters indicate statistically significant differences between the digestion timepoints within the same feed treatment material and concentration (one-way ANOVA). Any values below 0.06 mg/l in the saline solution, or equivalent to 0.13 mg/kg in a typical digest with 1 g of food, were considered below the procedural limit of detection, indicated by < LOD. Note: The LOD was used for the control to facilitate statistical analysis. Data are presented as the mean ± SEM, n = 3. dw = dry weight. ENM = engineered nanomaterial.
Behaviour of the TiO2-treated feed in the simulated intestinal phase at pH 7.8
Total Ti released from the TiO2-treated feed in the simulated intestinal phase at pH 7.8.
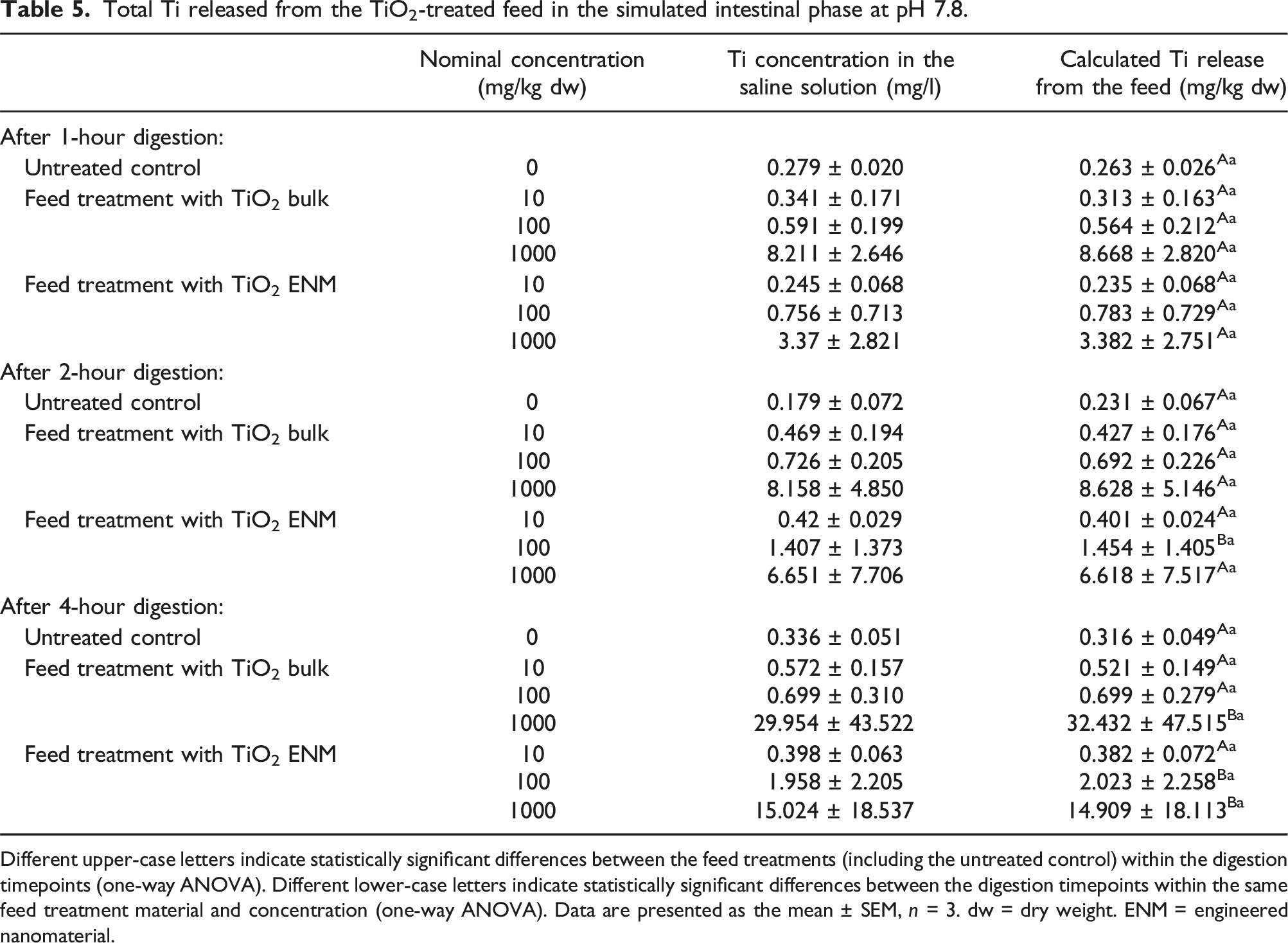
Different upper-case letters indicate statistically significant differences between the feed treatments (including the untreated control) within the digestion timepoints (one-way ANOVA). Different lower-case letters indicate statistically significant differences between the digestion timepoints within the same feed treatment material and concentration (one-way ANOVA). Data are presented as the mean ± SEM, n = 3. dw = dry weight. ENM = engineered nanomaterial.
Proportion of total Ti released from the diet after a 4-hour simulated digestion
Percentage of Ti released from the TiO2-treated feed after a 4-hour digestion in each simulated gut compartment.

The proportion of Ti released after a 4-hour digestion in each simulated gut compartment was calculated as a percentage of the measured total Ti in the treated diet formulations (see Table 3). Different upper-case letters indicate statistically significant differences between the feed treatment material and the untreated control (one-way ANOVA). Different lower-case letters indicate statistically significant differences between the concentrations within the feed treatment materials (one-way ANOVA). Any values below 0.13 mg/kg were below the procedural limit of detection, indicated by < LOD. Data are presented as the mean ± SEM, n = 3. dw = dry weight. ENM = engineered nanomaterial.
Discussion
Utility of the in chemico digestibility assay
This study is one of the first to perform the in chemico digestibility assay on TiO2 ENMs and its bulk counterpart, across a range of test concentrations in animal feed. Digestion in the simulated stomach phase and simulated intestinal phase both caused Ti release from the feed, with the highest concentrations released from the 1000 mg/kg dw diet after 4 hours (Tables 4 and 5). When this Ti release was expressed as a percentage of the total Ti measured in the feed, only the stomach phase showed an increase relative to the untreated control, with the total Ti from the intestinal phase being similar to the control background leaching (Table 6). This difference is attributed partly to a lack of detectable Ti release from the feed in the control of the simulated stomach phase. Notably, there were few treatment differences in the measurements, indicating a similar bioaccessible fraction of both the bulk and nano forms of TiO2; with both forms reaching a maximum of around 7% in the simulated intestinal phase.
The digestibility assay has been previously applied successfully to CuO ENMs, showing some dissolution of soluble metal, 19 and to Ag ENMs made of a known toxic metal (i.e. silver). 20 However, less is known about very sparingly soluble metal-containing ENMs such as TiO2. 23 One concern with respect to using the digestibility assay to inform regulatory decisions on the bioaccumulation potential of ENMs is that the assay may readily detect metal from materials that show dissolution, but not the particles themselves, or apparent dissolved metal from ‘insoluble’ particles. This would lead to the underestimation of the metal release and therefore bioaccessible fractions, i.e. it would represent a false negative in terms of regulatory decision making. For simplicity and utility as a standardised method, the approach described here is based on the determination of the total metal concentration released into the saline, without the need for more complicated single particle inductively-coupled plasma mass spectrometry (spICP-MS; see Laycock et al. 24 ) to measure particulate versus dissolved fractions. For TiO2, Shaw et al. 22 developed a total metal method for routine ICP-OES that relied on digesting any organic material (i.e. food or tissue), to leave a dispersion of TiO2 in a liquid phase that, with some mixing, could be used for total Ti determination. This approach was also applied here to the fish feed and the digestibility assay.
The digestibility assay is intended as a rapid method and decision tool in a tiered approach to bioaccumulation testing, which may help waive the need for in vivo testing on fish. 10 The method is quick (with a 4-hour incubation time) and simple, so that any competent technician could conduct the protocol in a regulatory setting. However, for practicality, it would be desirable to simplify the method further to a single TiO2 concentration. The TiO2 concentrations used in this study were deliberately within the same order of magnitude as those used for in vivo TiO2 bioaccumulation studies, 25 and for other ENMs.19,20,26 The single-concentration approach has been advocated for the bioaccumulation testing of chemicals in vivo, 8 and could also be applied to alternative methods. However, consensus on the concentration-dependence of the bioaccumulation of ENMs is not yet established — for example, it is not known whether the bioaccessible or bioavailable fractions change with the exposure dose. Here, concentrations below 1000 mg/kg dw of TiO2 in the feed were not detected by the assay at pH 2 (simulated stomach, Table 4), but the assay at pH 7.8 (simulated intestine) was much more effective, as the Ti released into the saline during the incubation reached a detectable level in the same incubation period (Table 5). For the intestinal phase, the released Ti was readily detectable and there was little evidence of concentration dependence for the feed treatments (Table 5), suggesting that any one concentration could have been selected for the assay. A concentration of around 10–100 mg/kg dw of TiO2 might be regarded as more physiologically relevant, so one of the lower TiO2 concentrations within the tested range is recommended. However, metal detection above background is also important for animal feed, and this is problematic for ubiquitous metals in the Earth’s crust (such as Ti, Fe or Al25,27,28), which inevitably are found in the protein sources and other ingredients used in feed manufacture. In the present study, the feed contained about 7 mg/kg dw of Ti (Table 3). In such cases, to ensure detection, a test concentration of at least ten-fold higher than the background might be recommended.
Prediction of bioaccessible fractions compared to in vivo accumulation
A recent meta-analysis by Handy et al. 5 showed a statistically significant relationship between the total metal concentration in the liver of trout from in vivo dietary exposures (i.e. the OECD TG 305 approach) and the total metal released during the in chemico digestibility assay for a range of coated ENMs, including those with Ag or Cu in their core. In the present study, around 3% of the TiO2 ENM and around 2% of the bulk form was released from the fish feed at the highest treatment concentration (1000 mg/kg dw) in the simulated stomach phase, and around 7% for both materials in the intestinal phase (Table 6), indicating a low dietary bioaccessibility of TiO2. This was also shown in vivo by Ramsden et al. 25 Together, these data argue in favour of the in chemico digestibility assay being considered as a suitable prediction tool in a tiered approach to bioaccumulation testing.
The data presented here for TiO2 is also broadly consistent with that obtained for other metallic ENMs, where only a few percent of the total metal was released from the feed using the same methodology. For instance, in the stomach phase, silver nanoparticles (Ag NPs) at 100 mg/kg dw showed around a 4% release from the feed after 4 hours of digestion, which was a similar level to that of the dissolved metal salt counterpart (silver nitrate). However, there was significantly less Ag release (0.5%) from the feed in the case of insoluble silver sulphide nanoparticles (Ag2S NPs) under the same conditions. 10 Moreover, feed containing CuO ENMs, when digested under the same stomach phase conditions, released approximately one third of the CuO material (250 mg/kg dw) from a feed Cu concentration of 750 mg/kg dw (see Boyle et al. 19 ). This suggests that the more soluble ENMs were more readily released from the feed, as one might expect — but nonetheless, the data for CuO still correlated with the in vivo results. 5
An initial prediction equation, for extrapolation of the in chemico assay results to the in vivo scenario, is available for a range of metallic ENMs of different sizes and with different surface coatings. 5 This could be used to help inform a decision to either waive or include the in vivo fish test in the overall evaluation of metallic ENMs. In a regulatory context, more data from other materials, including carbon-based ENMs such as carbon nanotubes or graphene oxides and organic ENMs such as dendrimers, and preferably all with different surface chemistries, would help to build a more powerful prediction equation. This would provide a weight of evidence for the predictive power of the in chemico digestibility assay and help to expedite its regulatory acceptance.
Benefits to animal welfare of a tiered approach to bioaccumulation testing
The determination of bioaccumulation potential is one aspect of ENM safety where alternative methods to in vivo testing on vertebrates have not yet been implemented. The approach presented here could help inform regulatory decision making to waive or include testing according to OECD TG 305, or other similar in vivo testing, for a given ENM. 10 For instance, OECD TG 305 and other similar in vivo fish tests often employ dose range-finding experiments prior to the main study. This is done to ensure that the exposure concentrations are not lethal, but still allow bioaccumulation to be confidently detected in the tissue. Instead, the in chemico digestibility assay could be used to identify likely bioaccessible fractions, and if the material showed concentration-dependence, inform on the choice of dosing for any subsequent in vivo study.
As part of the justification to waive the need for data from OECD TG 305 entirely for an ENM, a ‘negative result’ from the in chemico digestibility assay would be needed (i.e. a low or negligible bioaccessible fraction). A threshold level in the assay would need to be agreed for regulatory decision-making. For example, a value of 5% release from the feed (statistically 1:20 chance), or 10% release (less stringent, but more protective of false negatives) could be used to ‘trigger’ further in vivo testing. For example, for Ag NPs, around 3–4% of the dose is bioaccessible 10 and causes concentrations of total Ag in the organs of trout to be above the background level in vivo, 20 albeit without toxicity. Thus, a lower threshold might also be considered on a substance-specific basis. A decision-tree and possible thresholds for the bioaccumulation testing of ENMs are currently being discussed at the OECD.
Conclusions
The in chemico digestibility assay has been demonstrated for TiO2 materials here, adding to the evidence of its successful use in the similar analysis of other metallic ENMs, and showing that the assay does have utility to indicate the potential for bioaccumulation of metal oxides classed as sparingly soluble. The assay takes only a few hours to conduct and is relatively robust for the purposes of helping decision-making as to whether or not further in vivo studies on bioaccumulation potential are needed for metallic ENMs. In this respect, we recommend adopting the assay to help reduce in vivo bioaccumulation testing on fish. Further research is underway to collect data on carbon-based ENMs and other particulates, such as plastics, to extend the applicability of the assay.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Summer Studentship award from Replacing Animal Research (formerly known as FRAME (Fund for the Replacement of Animals in Medical Experiments)) to CMcH. RDH and NJC were supported by the NanoHarmony project, grant agreement No. 885931 under the EU Horizon 2020 research and innovation programme.
