Abstract
Purpose of the review:
Delirium occurs in up to 31% of general hospital patients in the medical, surgical, or critical care wards. Agitation is common during hyperactive delirium, which poses a risk to themselves, other patients, or staff. Evidence for using oral or parenteral haloperidol from randomized controlled trials and published systematic reviews shows divergent findings, ranging from good response to no difference compared to placebo, despite its popularity in routine clinical use. We decided to perform an umbrella review of systematic reviews on the efficacy and safety of haloperidol in delirium.
Collection and Analysis of Data:
This protocol was registered with PROSPERO CRD42024502020. We searched PubMed, Ovid MEDLINE, and PsychINFO for systematic reviews with meta-analyses published in English only over the previous 20 years. These were searched under title, abstract, and Medical Subject Headings (MeSH) terms as applicable. We used Assessing the Methodological Quality of Systematic Reviews 2 to assess the quality of the articles. Ten systematic reviews were finally selected for review that focused on either preventing or treating delirium. The summary of results did not find that haloperidol is effective in preventing or treating delirium, though all included reviews found it a safe drug to use. Heterogeneity from
Conclusions:
The authors conclude that there is limited evidence for haloperidol in preventing or treating delirium. Future studies must not aim to assess the resolution of delirium; instead, they must modify outcome measures, to mainly behavioral and psychiatric symptoms such as agitation, hallucinations, and persecutory delusions in delirium.
Key Messages:
Delirium occurs commonly across healthcare setups, with its prevalence rates at admission up to 31% of critical care, general medicine, surgical, and orthopedic wards. It is associated with increased mortality, length of stay, institutionalization, and care needs. 1 These in-patient units include emergency, preoperative, and postoperative recovery units. Agitation is commonly associated with a hyperactive type of delirium where patients pose a risk to themselves or others, including medical staff. 2 Patients with intraoperative hyperactive delirium may pose a risk of becoming uncooperative and may break barriers of asepsis, leading to poorer outcomes of procedures. Many older adults, after major surgeries such as hip surgery or cardiac surgery, develop delirium. In postoperative recovery units, this is an additional burden for the treating team, family members, and hospital management, which is preventable or controllable if anticipated. Sometimes, it takes time to investigate the cause of delirium. During this period in the hospital, when the behavior becomes unmanageable, it becomes necessary for the trained ward staff to administer either physical or chemical restraints to control the behavior. Perioperative delirium with anesthetic agents, particularly in older people, is well documented, in addition to a higher risk of critical medical complications. 3 Ketamine or opioids, particularly in partially sedated elderly patients with suboptimal doses of intravenous anaesthetic agents, benzodiazepines, and volatile anesthetic agents, often lead to postoperative agitative delirium. Tranquilization is one of the options to manage agitation in delirium in medical wards, as there is an immediate need to manage the behavior, which poses challenges to healthcare staff members. 4 Haloperidol, either oral or parenteral, is routinely administered to reduce agitation when verbal de-escalation techniques fail. 5
Haloperidol is a typical antipsychotic that seems ideal for tranquilization by intravenous or intramuscular route in delirium due to its rapid onset of action and not known to cause severe sedation, which is important. At the same time, other medical treatments for the cause of delirium are given a priority. 6 There is a need for evidence-based prescription of pharmacological agents, including haloperidol, in potentially life-threatening physical illnesses. Haloperidol is frequently prescribed for delirium. 7 It is also used perioperatively and in critically ill elderly patients at risk of developing delirium as a prophylactic drug.8–10 Safety concerns about haloperidol include the risk of extrapyramidal side effects (EPSEs) and prolongation of the corrected QT (QTc) interval in the ECG. 6 Compared to other antipsychotics, the anticholinergic effect, which is known to worsen delirium, is relatively lesser with haloperidol. 11
In the early trials of haloperidol in delirium, haloperidol was considered as a treatment for delirium. 12 Trials comparing haloperidol with other atypical antipsychotics, namely olanzapine and risperidone, began to emerge in the new millennium.13,14 Initial trial results indicated no significant difference between haloperidol and these atypical antipsychotics regarding response and safety. Delirium severity, measured by the delirium index score or score on the Memorial Delirium Assessment scale, was the primary outcome measure. 15 Trials were also done on at-risk patients, such as in older adult patients before undergoing larger surgical procedures such as hip surgery and cardiac surgery, and a naturalistic policy-based service evaluation study showed a reduced incidence of delirium significantly. 16 Some specifically looked at the survival post-delirium using haloperidol. 17
Agitation and aggression in hyperactive delirium have recently been a management focus in patients with delirium. 18 Agitation with aggressive behavior in delirium needs multidisciplinary management. Liaison psychiatrists working with lead physicians develop a care plan that includes non-pharmacological environmental measures to reduce the impact of other contributing factors. The intervention must not interfere with or worsen the medical or perioperative complications. The symptoms are generally different in the hypoactive type of delirium.
The evidence based on the response and the adverse effect profile of haloperidol from the randomized controlled trials (RCTs) is contradictory.
10
The literature review suggested several systematic reviews with meta-analyses on haloperidol in delirium. However, many published systematic reviews, which compared haloperidol against placebo or atypical antipsychotic drugs reported varied responses varied such as (a) significant response and safer as treatment or prophylaxis, (b) promising, but no robust, generalizable evidence, (c) no difference with placebo on delirium-free days, mortality, incidence, length of stay (d) no response.19–22 There are variations in the systematic review studies’ objectives and reported
Methods
Protocol, Register, and Guidelines
This umbrella review of systematic reviews followed and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement 23 and particularly followed the Preferred Reporting Items for Overviews of Reviews (PRIOR) checklist. 24 The study protocol was registered in PROSPERO CRD42024502020. In this study, we included prevention as a part of management.
Eligibility Criteria
Population
The study population included general hospital inpatients in the clinical trial.
Intervention
The intervention studied was haloperidol administered orally or parenterally at the dose as per the trial protocol.
Comparator
The comparator drug for this review is a placebo. The reviews that included studies must have had RCTs comparing haloperidol with placebo.
Outcomes
The specific outcomes were based on the preliminary search and the literature review of systematic reviews and RCTs. The objective is to find out the efficacy of haloperidol in the prevention or treatment of delirium. The primary outcomes in this study were (a) prevention of delirium by studying the incidence of delirium and (b) management of delirium by resolution of behavioral symptoms related to delirium. Secondary outcomes: safety profile includes adverse effects: (a) neurological = EPSEs; (b) cardiac = QTc interval prolongation on ECG, arrhythmias, and sedation; and (c) length of hospital stay.
Setting
The reviews must have included the clinical trials conducted on participants admitted to general hospitals in medical, surgical, or intensive care unit (ICU) settings.
Inclusion Criteria
Systematic reviews with meta-analysis of RCTs involving haloperidol compared with placebo in medical hospital inpatients, including ICU/critical care units; medical, surgical, and orthopedic wards in hospital/healthcare units with inpatient facility; articles published in the English language between the years 2004 and 2023; haloperidol is used either for prevention or management of delirium; and the systematic reviews must have followed PRISMA guidelines.
Exclusion Criteria
Unpublished systematic reviews (e.g., conference abstracts and review protocols), narrative reviews, systematic reviews without meta-analysis, and the systematic reviews that studied non-RCTs were excluded. No supplementary primary RCTs were considered. Systematic reviews of the RCTs and umbrella reviews of the systematic reviews of the RCTs are considered superior in the hierarchy of evidence base; it was decided to exclude the reviews on non-RCTs.
Search Methods
Electronic searches: The following databases were searched: PubMed, Ovid MEDLINE, and PsycINFO. The search included systematic reviews published in English between 2004 and 2023.
Search Strategy
The search terms included delirium (ti, ab & MeSH) and haloperidol (ti, ab & MeSH), and systematic review or meta-analysis, using filters, 20-year period, published in English, was done on March 11, 2024 (Supplementary Material 1). After the search, the files were exported to Covidence software, which removed duplicates and made 59 articles available for screening.
Study Selection
Two authors (VHa and VHe) independently screened the articles initially based on title and abstracts. A third author (MK) resolved the discrepancies between the authors; all of these were done on the Covidence software. In the second stage, the same two authors independently examined full texts using the eligibility criteria (inclusion and exclusion criteria), followed by the same third author addressing discrepancies. The excluded systematic reviews with reasons are presented in the supplementary material 2.
Data Extraction
The data collected from the eligible systematic reviews were as follows: the author and the year of publication, study aims, number of studies, number of studies included in the meta-analysis, outcomes, results, authors’ conclusions, limitations, and comments. The data from the forest plot were gathered to verify the number of participants and pooled effect size, and the information on the event rate and control rate was utilized to calculate the numbers needed to treat.
Quality Assessment of the Included Systematic Reviews
The Assessing the Methodological Quality of Systematic Reviews 2 (AMSTAR 2) tool was used to appraise the methodological quality of the included systematic reviews. 25 This tool evaluates the methodological aspects using 16 items. For each question, if all parts are presented in the systematic reviews completely, they are marked as “Yes”; where it is reported but incomplete, they are marked as “Partial Yes”; when the information is not available, they are marked as “No.” Two authors (ST and ASH) independently completed a quality assessment on Microsoft Excel, and the discrepancies were resolved by a third author (VHa).
Analyses
As the individual studies in this systematic review were systematic reviews with meta-analysis, pooling the results from the meta-analyses was not done. However, a qualitative type of analysis was done. The risk ratios available in the meta-analyses calculated the number needed to treat (NNT) from the pooled results. This was done where risk ratios were provided. Data on heterogeneity in the individual systematic reviews were also extracted from the meta-analyses. Sensitivity analysis was also performed with the exclusion of low-quality studies.
Results
Selection of Systematic Review Studies
One hundred thirty-three studies were identified, of which 37 were assessed for eligibility. Based on the predetermined eligibility criteria, 10 studies were suitable for this systematic review. The PRISMA flowchart (Figure 1) outlines the process of selecting articles. After the title and abstract screening between the two independent investigators, the inter-rater reliability data found that the random agreement score of 0.51 had a Cohen’s
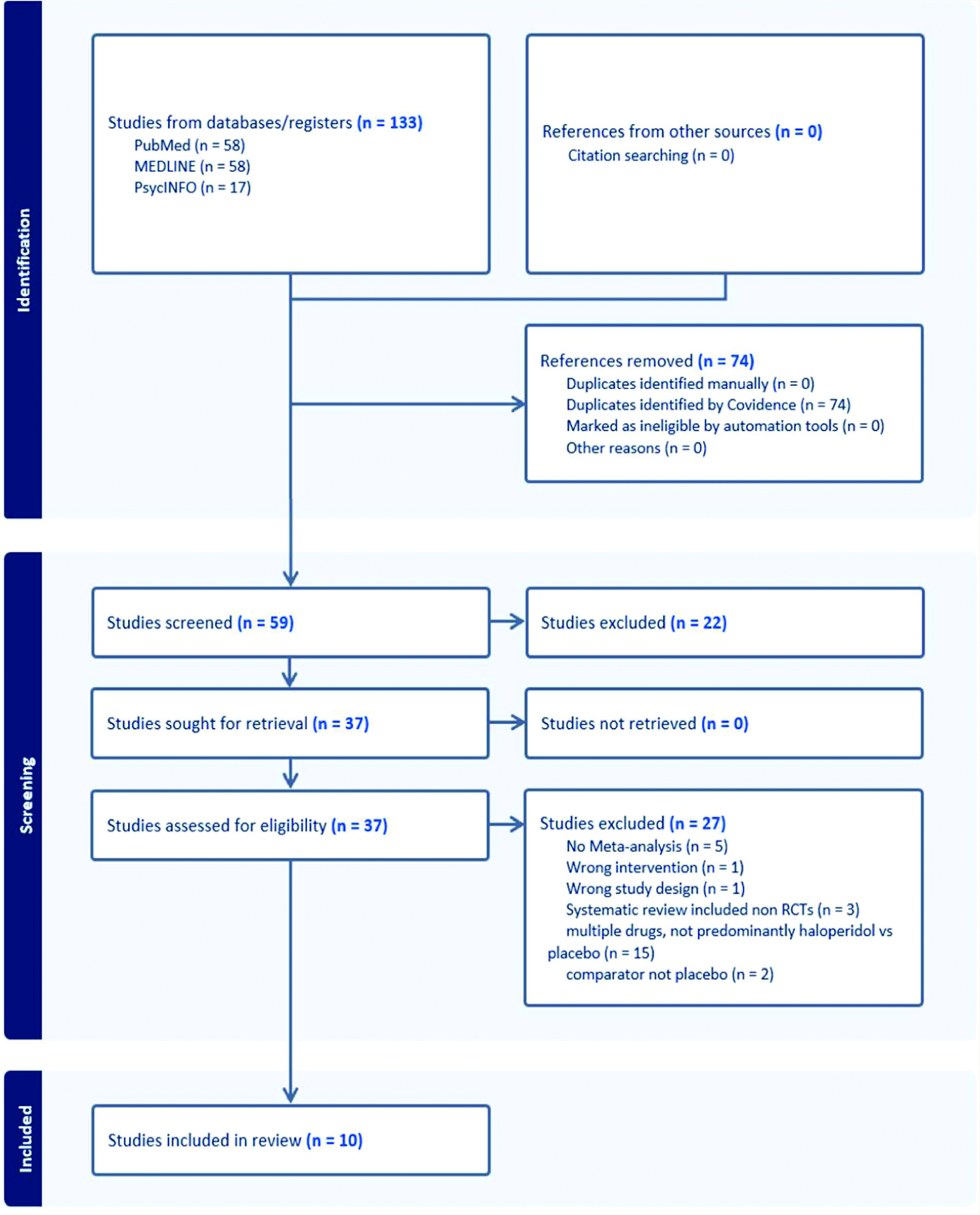
A total of 27 systematic reviews focused on multiple drugs, with no predominant focus on haloperidol; some studies were on atypical antipsychotics, and a few did not have meta-analysis and were excluded from the final review (see Supplementary Material 2).
This final list included systematic reviews that exclusively studied haloperidol against placebo and other primary systematic review articles where the data related to haloperidol against placebo were extracted and presented. The characteristics of the included systematic reviews are presented in Table 1. The majority of the studies had the incidence of delirium as their primary outcome measure.
Of the 10 systematic reviews in the review, 8 included systematic reviews aimed at studying haloperidol in the prevention of delirium in ICUs and during the perioperative period.26–36 Two reviews aimed to study haloperidol as both prophylaxis and treatment of delirium.31,34 One study by Siddiqui et al. (2007) 26 was updated in 2016 and published in the Cochrane Database of Systematic Reviews. 27
Population and Setting
Only one review (Igwe
Other Outcomes
Except for the Zayed et al.’s review, 31 where the primary outcome was mortality, the rest of the included reviews had outcomes related to the incidence of delirium. None of the reviews studied the symptoms of delirium specifically. It cannot be assumed that the resolution or reduced incidence of delirium is related to reduced symptoms—length of delirium. Although two reviews27,32 concluded that there was a reduction in the length of hospital stay or shorter duration of delirium, and another review 33 suggesting reduced incidence of postoperative delirium, five reviews28,29,31,34,36 found contradicting evidence that there was no difference in these secondary outcomes (see Table 1).
Safety Profile
All reviews concluded that haloperidol was safe and tolerable, which is an interesting finding. The reviews included RCTs that studied neurological adverse effects, particularly the EPSEs, and cardiac adverse effects that mainly included QTc prolongation on ECG and hypotension, bradycardia, and arrhythmias. The reviews concluded no significant rise in incidence of EPSE and QTc prolongation. Three reviews29–31 had mortality as one of the secondary outcomes and were not found to be significant in the haloperidol group compared to the placebo group (see Table 1).
Characteristics of Included Systematic Reviews (N = 10).
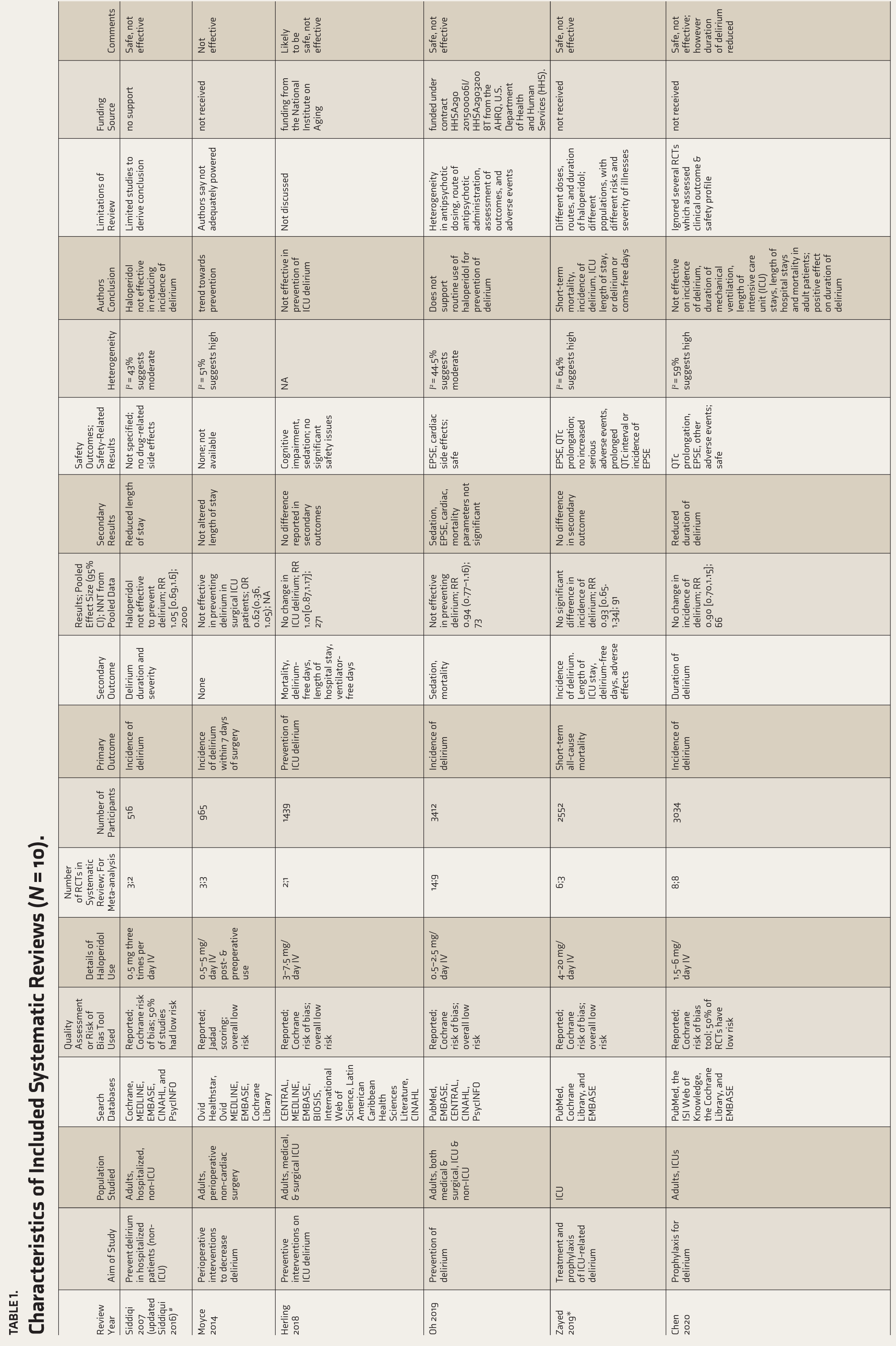
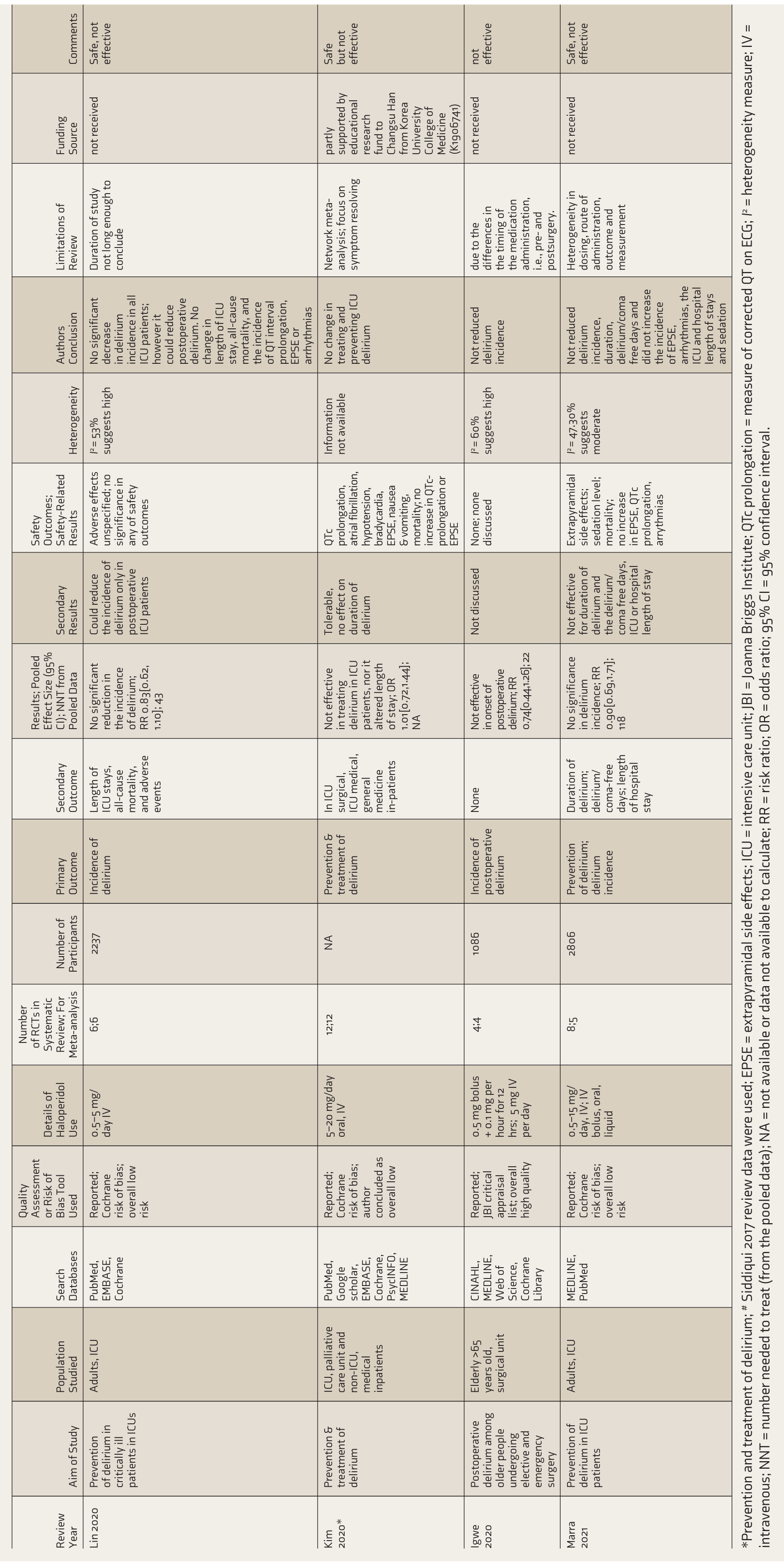
*Prevention and treatment of delirium; # Siddiqui 2017 review data were used; EPSE = extrapyramidal side effects; ICU = intensive care unit; JBI = Joanna Briggs Institute; QTc prolongation = measure of corrected QT on ECG;
Overlap of the Primary Studies
The RCT studies, namely Kalisvaart et al. 8 Wang et al. 9 and van den Boogaard et al. 37 appeared in at least three meta-analyses. One RCT study by van den Boogaard et al. 37 had the highest weight from the review and visual inspection of all meta-analyses, suggesting it appeared relatively more robust.
Quality Assessment
Most of the systematic reviews have not adopted comprehensive search strategy as per the AMSTAR 2. The reason for studying only RCTs was not discussed in all systematic reviews except one. Many systematic reviews also did not mention details of the funding of the individual RCTs of the individual reviews. The quality assessment results for the 10 included studies are presented in Table 2.
Publication bias and subgroup analyses were not assessed or conducted as this is a systematic review of systematic reviews. After the minor deviation from eligibility criteria as per the protocol, only 10 systematic reviews were yielded. For the purpose of sensitivity analysis, when the lowest-quality article, Moyce et al. 28 is excluded from this review, the qualitative results do not seem to be any different.
Analysis
Most of the meta-analyses pooled the risk ratio from the RCTs. A few reviews presented the odds ratio (OR). The NNT was calculated from the data available from the forest plots for some of them (see Table 1). Though NNT is a type of effect size, we noted a considerable variation in the value from 22 to 2000. Due to variations in Patient, Intervention, Comparison, and Outcome (PICOS) and heterogeneity, we cannot pool the data from the different meta-analyses as studies. The included reviews had moderate to high heterogeneity based on the
Quality of Included Systematic Reviews Using AMSTAR 2 (N = 10).
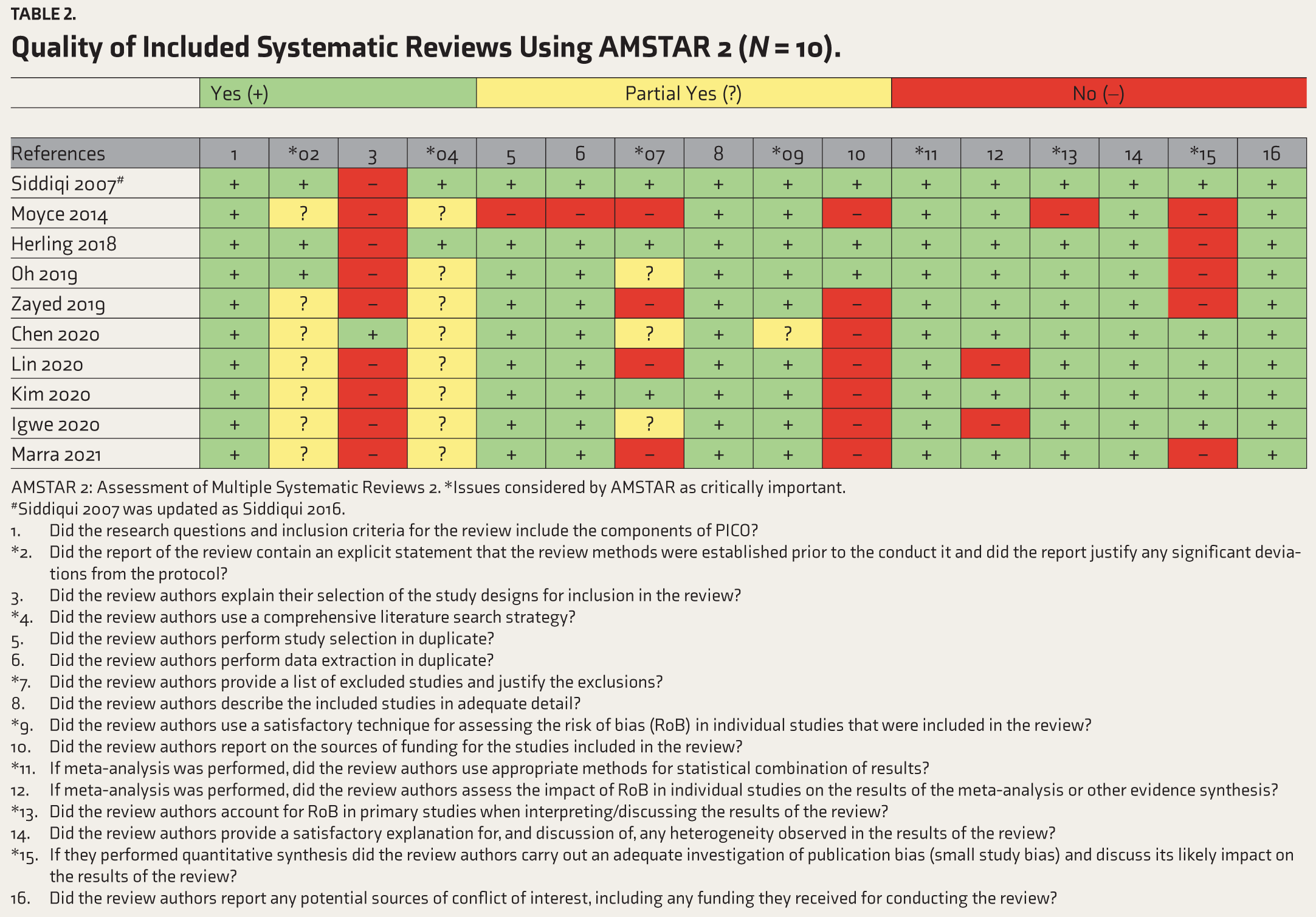
AMSTAR 2: Assessment of Multiple Systematic Reviews 2. *Issues considered by AMSTAR as critically important.
#Siddiqui 2007 was updated as Siddiqui 2016.
Did the research questions and inclusion criteria for the review include the components of PICO? *Did the report of the review contain an explicit statement that the review methods were established prior to the conduct it and did the report justify any significant deviations from the protocol? Did the review authors explain their selection of the study designs for inclusion in the review? *Did the review authors use a comprehensive literature search strategy? Did the review authors perform study selection in duplicate? Did the review authors perform data extraction in duplicate? *Did the review authors provide a list of excluded studies and justify the exclusions? Did the review authors describe the included studies in adequate detail? *Did the review authors use a satisfactory technique for assessing the risk of bias (RoB) in individual studies that were included in the review? Did the review authors report on the sources of funding for the studies included in the review? *If meta-analysis was performed, did the review authors use appropriate methods for statistical combination of results? If meta-analysis was performed, did the review authors assess the impact of RoB in individual studies on the results of the meta-analysis or other evidence synthesis? *Did the review authors account for RoB in primary studies when interpreting/discussing the results of the review? Did the review authors provide a satisfactory explanation for, and discussion of, any heterogeneity observed in the results of the review? *If they performed quantitative synthesis did the review authors carry out an adequate investigation of publication bias (small study bias) and discuss its likely impact on the results of the review? Did the review authors report any potential sources of conflict of interest, including any funding they received for conducting the review?
Discussion
This seems to be the first umbrella review of systematic reviews on haloperidol for treating delirium. Umbrella reviews are conducted when multiple systematic reviews present conflicting findings. Simply updating a systematic review would not have addressed the central issue identified—a significant drawback of the studies conducted. The most important finding of this study is that haloperidol is ineffective in these primary outcome measures of prevention or treatment of delirium when administered intravenously in perioperative care or ICUs or when orally administered. The adverse neurological and cardiac effects were insignificant, suggesting that the drug is safe for these indications. The outcome measures varied from evaluating the incidence of delirium or the number of days to delirium resolution in keeping with the study objective. None of the included studies discussed the types of delirium.
Haloperidol is a popular agent, a regularly used drug to reduce acute agitation and aggression, which is related to the hyperactive type of delirium. 7 It is also known to reduce hallucinations, delusions, and agitation acutely and without causing sedation or cardiac abnormalities. Doses vary with increasing age and frailty. Contrary to this understanding of the use of haloperidol, all the systematic reviews focused on haloperidol for the prevention or treatment of delirium. This systematic review demonstrated that haloperidol did not reduce the length of stay in the hospital, duration of delirium, incidence of delirium, number of delirium-free days, or mortality. The summarized results suggest that it was not efficacious in preventing or treating delirium. Most importantly, none of the systematic reviews that were studied had the particular objective of studying the reduction of the level of agitation or behavioral problems in delirium, and therefore, the required results were not available, which this review had set out to study.
The negative result could be explained by the fact that the systematic reviews predominantly had either prevention or treatment of delirium as the primary outcome measure. However, it is known that haloperidol is used mainly for behavioral and psychiatric symptoms in the context of delirium and not for the complete remission of delirium. By its nature, the course of delirium is variable, based on the underlying cause, severity, and treatment of the underlying cause. Despite the completion of appropriate treatment of delirium, there can be a time lag before delirium completely subsides. Factors that influence the duration of delirium such as age, comorbidities, sensory deprivation, environment, stress, and pain. Treating the underlying cause will gradually lead to the remission of delirium. The findings from this study and understanding of the current literature indicate that haloperidol may be used in the symptomatic management of delirium but not in the remission or prevention of delirium. Given that it appears to be safe, it can still be tried to reduce the behavioral and psychiatric symptoms of delirium.
The recent hypothesis in the etiopathogenesis of delirium suggested that precipitating factors include neuronal aging, neuronal inflammation, oxidative stress, neuroendocrine dysfunction, and circadian dysregulation. 38 Delirium is more common in older adults, where there is neuronal loss, neurotransmitter dysregulation, reduction in blood flow, and diminishing compensatory mechanisms. 39 In the context of this hypothesis, haloperidol may have no scope to reduce or modify the underlying etiopathogenesis. Therefore, it has no role in the remission or prevention of delirium. The outcome in individual studies should have been tranquilising effect of haloperidol in delirium rather than remission of delirium itself, or effect of haloperidol as adjunct in managing behaviour in delirium.
There is a lacuna in the evidence base for managing delirium in general. More research is needed in this field. There are limitations in conducting such studies in delirium as this condition runs a fluctuating course. It also needs more opportunities in terms of resources in this area. The primary treating team is either medical, surgical, or critical care unit, that need to take the lead in conducting studies in collaboration with other disciplines, including psychiatrists. The primary systematic review studies in our review have included delirium as one group and have not been classified into hypoactive, hyperactive, or mixed and examined. Also, the delirium is grouped as one, though the cause of delirium could have been different. This makes the delirium group heterogeneous. The rationale for using haloperidol for prevention is not described. It is clear from the review that this agent has no role in prophylaxis. Also, haloperidol works on dopamine receptors, but so far, the role of dopamine has not been implicated in hypotheses of delirium. Delirium is known to have multifactorial causes. It requires comprehensive, holistic treatment, including environmental modification and care. Haloperidol may be considered as an adjunct drug specifically to reduce the agitation in delirium, which the systematic reviews fail to focus on.
There was one review where the authors concluded a trend towards preventing delirium with haloperidol, Moyce et al. 28 which aimed to determine the efficacy of perioperative interventions in decreasing the incidence of postoperative delirium. This review was on surgical patients and the prevention of delirium. The outcome measure was the incidence of delirium within 7 days of surgery. The dose was from 0.5 mg to 5 mg/day intravenous (IV) post- and preoperative use. The review included three RCTs in a systematic review and meta-analysis. The pooled OR was 0.62 (0.36, 1.05) and thus was ineffective in preventing delirium. We found significant inconsistency and moderate heterogeneity, and variations in the visual inspection of the forest plot were seen. The results from the meta-analysis also showed imprecision, as there were variations in the width of the confidence intervals.
This umbrella review is based on the systematic reviews done on the RCTs. There are apparent variations in the populations, interventions (in terms of route of administration), and outcomes between the included reviews. The results from meta-analyses also vary with heterogeneity scores. Thus, the methodological variations have an impact on the results. Despite that, all reviews found haloperidol safe, and this result was consistently obtained. Concerning efficacy, there was no significant reduction in the incidence or resolution of delirium. The researchers are focusing on the resolution of delirium or reduction of incidence of delirium, that is, prevention or treatment of delirium when they are meant to be focusing on the reduction of the specific symptoms such as behavioral, namely agitation, aggressiveness, and irritability, or psychiatric symptoms, namely hallucinations or delusions in the presence of naturally fluctuating course of a delirium.
Strengths and Limitations
The strength of this study is that it had a standard approach with a preregistered protocol and followed PRISMA guidelines. The limitation of this study is that the majority of systematic reviews were heterogeneous in terms of population, routes, and dose of administration. Most systematic reviews have not mentioned the funding source for individual RCTs. Another limitation of reviewing the reviews is that there is no scope for studying their efficacy or dosage issues in specific older adult population groups, where delirium is more common.
Clinical Implications and Future Directions
Haloperidol is still being used in medical settings to treat agitation. Most systematic reviews aimed at preventing or treating delirium did not find a favorable response. One may have to conclude that there is no evidence base for using haloperidol in the management of delirium. Psychopharmacology of haloperidol suggests that it is the drug of choice as a tranquilizing agent meant for use in reducing agitation or aggression behaviors. It does not improve cognitive functions in delirium either. There is no need for any further trials on haloperidol in the prevention or treatment of delirium. Treating the underlying cause is the mainstay of the treatment in delirium, and it is not from haloperidol or any other antipsychotics. The outcome measures need to be sensible when designing further systematic reviews on treatment. However, that needs to point toward specific behaviors, mainly psychiatric, and additionally may examine the functional symptoms in hyperactive delirium and not as treatment. Unfortunately, the researchers have focused on preventing delirium with haloperidol. This study recommends that future studies focus on the use of haloperidol in particular symptom profiles or reduction in the number of incidents or reduction in the need for physical restraints in addition to behavioral symptoms such as agitation, aggression, paranoia, hallucination, as well as their intensity could be the primary outcome measure. Studies must modify outcome measures to the behavioral and psychiatric symptoms such as agitation, hallucinations, or persecutory delusions in delirium (see Table 3).
Recommendations for Future Studies on Haloperidol or Any Other Antipsychotics in Delirium.

ABS = Agitated Behavior Scale; BI = Barthel Index; BPRS = Brief Psychiatric Rating Scale; CGI = clinical global impression; NPI = neuropsychiatric inventory; RCTs = randomized controlled trials; SF-36 = short-form health survey; ZBI = Zarit Burden Inventory.
Deviation from Protocol
We decided to exclude systematic reviews that did not have PROSPERO registration or without any mention of protocol made available before the study began. At the eligibility criteria stage, the authors felt higher quality articles might be missed. Therefore, this exclusion criterion was modified to include systematic reviews with at least a mention of having followed PRISMA guidelines. Our assumption of the higher quality of such articles was validated after the AMSTAR 2 assessment. For uniformity reasons, we used the term umbrella review of systematic reviews at the time of manuscript preparation.
Conclusion
Haloperidol does not seem to reduce the incidence of delirium when used pre- or postoperatively as a prophylactic agent and does not induce delirium resolution. These were based on the outcome measures used in individual systematic review articles. None of the systematic review studies in the past two decades attempted to examine specific symptom reduction in delirium and haloperidol as an adjunct in the overall management of delirium. The authors had the assumption that haloperidol could prevent or treat delirium, which is not the case. The heterogeneity of delirium must be understood. The outcome measure needs to be appropriate. Possible alternative options, such as atypical antipsychotics or dexmedetomidine, may need to be explored and studied. There is a need to study the efficacy of haloperidol on behavioral or psychiatric symptoms in delirium as a safe tranquilizing agent rather than studying delirium resolution.
Supplemental Material
Supplemental material for this article available online.
Supplementary Material
Supplemental material for this article available online.
Footnotes
Data Availability
This article contains supplementary material. The PROSPERO protocol is in the public domain.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration Regarding the Use of Generative AI
No part of this article was written or generated by a generative AI tool. The authors take full responsibility for the accuracy, integrity, and originality of the published article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Reporting Guidelines
PRISMA 2020 Guidelines. The protocol was registered with PROSPERO prior to undertaking the search.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
