Abstract
Background:
Suicidal thoughts and behaviors (STBs) are a major concern in India, but there is limited research on effective interventions. Cognitive Therapy for Suicidal Patients (CT-SP) has shown promise, but its implementation in India needs further investigation.
Novelty:
This study adopts a transdiagnostic approach to suicidality, recognizing the strong link between various mental health conditions and STBs. The study aims to use an evidence-based intervention for acute suicidal risk and understand its outcomes in the Indian context using systematic implementation. The blended delivery format of the intervention (both in-person and online) further enhances its relevance for diverse clinical settings.
Objectives:
The study aims to assess the effectiveness of a brief CT-SP intervention in reducing the severity of suicidal ideation and behaviors. Secondary objectives include evaluating changes in emotional regulation, cognitive flexibility, hopelessness, depression, anxiety, problem-solving skills, frequency of dysfunctional cognitions, and overall functioning.
Methods:
The study will use an open-label, single-group design with baseline, post-intervention, and follow-up assessments at one, three, and six months. Patients with recent suicidal ideation or behaviors will be recruited across diagnoses. The intervention consists of a ten-session CT-SP protocol delivered in a blended format over five weeks.
Expected Outcome:
The study would contribute to the empirical understanding of suicide risk management in clinical settings and inform the development of culturally relevant interventions specific to the Indian context.
Keywords
This study protocol outlines the design of one of the few research projects in India to explore the effectiveness of Cognitive Therapy for Suicidal Patients (CT-SP) in reducing suicidal ideation and behaviors among individuals seeking psychiatric care. This study aims to improve knowledge of suicide risk management by addressing the cognitive and emotional aspects of suicidality from a transdiagnostic perspective using an evidence-based intervention and multiple outcome measures.Key Messages:
Protocol Registration
Clinical Trials Registry of India, April 2022 (CTRI/2022/04/042090).
Introduction
Background
Suicide is a global public health concern, with approximately 800,000 deaths occurring annually, and India bears a substantial burden with a mortality rate of 17.9 per 100,000 population, one of the highest in Southeast Asia.1,2 Suicidal thoughts and behaviors (STBs) range from fleeting ideation to tragic completed suicides. The World Mental Health (WMH) Survey Initiative found the lifetime prevalence of suicidal ideation, plans, and attempts to be 9.2%, 3.1%, and 2.7%, respectively, across 17 countries.3,4 Though reducing suicide is an important task, the National Mental Health Survey 2016 reported a treatment gap of over 80% in India. 5
Mental health conditions are inseparably linked to STBs, with 60%–90% of those who died by suicide having a psychiatric diagnosis.3,6 Specific phobia, substance use, and conduct disorders are predictors of planned suicide attempts. 7 Compared to the general population, higher risk was associated with alcohol-related conditions, schizophrenia, mood, anxiety, eating, and personality disorders, especially borderline personality disorder (BPD). 8 Considering these findings, STBs can be meaningfully conceptualized as a transdiagnostic entity.
Literature Review
Existing theories of suicide provide frameworks to identify dynamic factors contributing to STBs and propose potential targets for intervention. The Cognitive-Behavioral Model of Suicidal Mode describes that in addition to dispositional vulnerabilities, cognitive, affective, behavioral, and physiological characteristics such as impulsivity and maladaptive cognitive styles increase suicide risk. 9 Evidence-based psychological interventions have shown optimistic results, such as reducing suicide risk and enhancing coping and problem-solving skills.10,11 One of the most promising approaches, the cognitive-behavioral theory of suicidal mode coupled with the cognitive conceptualization of suicidal acts, enhances coping with stressors by using adaptive patterns of thoughts and behaviors.12,13 The principles of this intervention have been effectively adapted to be delivered to people in out-patient care, in-patient psychiatric care, and veterans.14–16While these findings highlight Cognitive Behavior Therapy (CBT) as an effective intervention, studies on the management of STBs remain scarce in the Indian context. 17
Novelty
This study conceptualizes suicide as a transdiagnostic factor and aims to address the distress and dysfunction experienced by individuals across different diagnoses that lead to STB. It seeks to explore the application of evidence-based brief CBT intervention in a blended offline and online format to improve the accessibility of treatment. The study aims to mitigate the severity of STBs and understand the cognitive, affective, and emotional outcomes. Exploring the utility of such interventions using a systematic approach in India is necessary to ensure that the existing treatment gap for suicide-related concerns can be addressed effectively in a culturally sensitive manner.
Objectives
The primary objective is to study the effectiveness of CT-SP in reducing the severity of suicidal ideation and behaviors in individuals with suicidal risk attending psychiatric services. The secondary objectives are to examine the effectiveness of the intervention in (a) reducing the level of hopelessness, depression, anxiety, and negative automatic thoughts, (b) improving emotion regulation, problem-solving, cognitive control, and flexibility in individuals, and (c) improving personal and socio-occupational functioning.
Methodology
Study Design/Study Participants/Setting
This study will employ an open-label, single-group design with baseline, post-intervention, and follow-up assessments up to 6 months post-intervention. Individuals with significant suicidal ideation or suicidal behaviors indicated on the Columbia Suicide Severity Rating Scale (C-SSRS) by either a score of three or more on the Suicide Ideation Severity subscale in the last month or the presence of any suicidal behaviors on the Suicidal Behavior subscale scale over the previous three months, aged between 18 and 60 years and providing consent to participate in the research study will be recruited. 18 Presence of actual, aborted, or interrupted attempts and preparatory acts will be considered significant suicidal behaviors and included in this study. The presence of non-suicidal self-injurious behaviors alone will not be considered as meeting the inclusion criteria but will be recorded. Individuals with a diagnosis of schizophrenia spectrum disorders and those with bipolar affective disorder, severe depression with psychosis or obsessive-compulsive disorder whose symptom severity prevents engagement in therapy based on clinical evaluation, any cliniczal history suggestive of intellectual disability or autism spectrum disorders and those who underwent brain stimulation interventions such as electroconvulsive therapy (ECT) in the last 15 days will be excluded. The research will be conducted within the in-patient and out-patient services, Behavioral Medicine Unit, and NIMHANS Centre for Well-being of the National Institute of Mental Health and Neurosciences (NIMHANS), Bangalore, India. These participants would be recruited using the convenience sampling method. A brief cognitive therapy intervention that addresses the cognitive and behavioral processes that play a role in suicidal ideations and behaviors has been chosen for this study. Cognitive Therapy for Suicidal Patients (CT-SP) is a cognitive-behavioral treatment for adults with suicidal ideation and behaviors. 19 It is a manualized and evidence-based ten-session treatment protocol based on a cognitive-behavioral model of STBs. The intervention includes three phases focused on risk assessment and safety planning, cognitive case conceptualization, application of cognitive and behavioral strategies, and relapse prevention protocols (Table 1). The first phase of this skill-based cognitive-behavioral intervention addresses high-risk conditions such as recent attempts or active ideation by focusing on crisis management and ensuring safety. It employs crisis management planning, restriction counseling, identifying reasons for living, and making a hope kit and coping cards. Crisis management with safety planning has been validated as a stand-alone intervention in clients with acute suicidal risk. 20 Cognitive intervention for suicidality will begin in this phase with the cognitive conceptualization of a recent suicidal crisis, identification of suicide-specific cognitions, and treatment goals. These are addressed in the second phase. Skill consolidation for future suicidal crises will be done in the third phase of relapse prevention. The sessions will be conducted on a biweekly basis and are likely to be completed in five weeks. The sessions will be carried out in a blended format, combining a minimum of four in-person sessions in the beginning, with the following sessions having an option of being held online based on the client’s preferences. After completion of the intervention sessions, the outcome measures will be applied, and follow-up assessments will be carried out at one, three, and six months.
Three Phases and the Various Tasks of Intervention Distributed Across Ten Sessions of the Planned Intervention Period. 19

Sample Size Estimation
The sample size estimation was done based on an assumed moderate effect size. A sample size of 34 was initially calculated using Cohen’s d value of 0.5, an alpha level of 0.05, and a power level of 0.80. However, to account for the various outcome variables, missing data, and potential attrition, a sample size of 50 participants was arrived at.
Study Tools
A clinical information sheet will collect baseline data on demographics, medical and psychiatric diagnoses, comorbid conditions (including substance use), onset and duration of illness, current and past treatments, past suicidality (including attempts), family history of mental health issues, premorbid personality, developmental history, adverse childhood experiences and triggers for the onset of current STBs.
Screening Measure
The C-SSRS will be used for suicide risk screening, assessing suicidal ideation, behavior, and self-injurious behavior without suicidal intent. This scale is a clinician-administered structured interview.
Diagnostic Measures
Diagnostic Assessment of Mental Health and Related Disorders (DIAMOND) is a semi-structured interview to assess participants’ current psychiatric conditions, including mood, anxiety, personality, and substance use disorders. 21 Diagnostic classifications are based on the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) criteria. The International Personality Disorder Examination (IPDE) is a comprehensive assessment tool used to evaluate personality disorders based on the ICD. These will be administered by the researcher at the point of recruitment to record the presence of any of these disorders owing to the transdiagnostic approach of the current intervention.
Primary Outcome Measures
The C-SSRS will be used as the primary outcome measure. It uses standard definitions of suicidal ideation as defined by the Centers for Disease Control and Prevention. 22 The scale contains three subscales: the severity of suicidal ideation, the intensity of suicidal ideation, and suicidal behaviors.
Secondary Outcome Measures
The STHS is a 23-item, self-report measure to assess hopelessness in two dimensions. 23 Hamilton Depression Rating Scale (HAM-D) is a clinician-rated measure of the severity of depressive symptoms. 24 Patient Health Questionnaire 9 (PHQ-9) is a self-reported measure of the client’s perception of the severity of depressive symptoms. 25 The severity of anxiety symptoms will be assessed using the Hamilton Anxiety Rating Scale (HAM-A), a clinician-rated tool. 26 The clients’ emotional functioning in terms of awareness, clarity, non-acceptance of emotions, difficulties engaging in goal-directed behaviors, impulsiveness, and lack of strategies to manage emotions will be assessed using the Difficulties in Emotion Regulation Scale (DERS), an 18-item self-report assessment. 27
The ATQ is a 30-item self-report instrument that will be used to assess the frequency of automatic negative self-statements. 28 The Cognitive Control and Flexibility Questionnaire (CCFQ) is an 18-item self-report measure that assesses an individual’s perceived cognitive control over emotion and their appraisal and coping flexibility. 29 The Personal Problem-Solving Inventory (PPSI) is a 32-item self-report measure of three problem-solving process domains: problem-solving confidence, approach-avoidance style, and personal control. 30
The WHODAS 2.0 is a 36-item version, a self-administered measure of the functional limitations experienced by the individual across six domains in the last 30 days. 31
In addition to these measures, the perceived levels of distress, the intensity of suicidal ideation, and intention will be continuously assessed using visual analog scales (VAS) to track changes and ensure safety with appropriate referrals. They will be administered at the beginning of every session during the intervention phase.
Implementation Plan
Based on a pilot study with two participants, the relevant outcome measures, the number and frequency of intervention sessions, and follow-ups were finalized for this study. Individuals reporting suicidal ideation or a recent suicide attempt will be screened using the C-SSRS from in-patient and out-patient services of the hospital. The study participants will be screened and recruited by the first author (SS). Those who meet the inclusion criteria and consent to participate will be recruited, and the diagnostic and secondary outcome measures will be administered. After completion of the baseline assessments, the intervention will be initiated. During this period, the VAS will be administered at the beginning of every session to track distress and suicidality. Participants who complete 80% of the total number of sessions would be considered completers. Dropouts will be defined as participants who undergo less than 80% of the planned intervention. On completion of the intervention, the primary and secondary outcome measures will be administered. The study participants will continue to receive concurrent pharmacological treatments as per the advice of the clinical treating team. Any changes in the medication that occur during the study period will be recorded. Follow-up assessments will be carried out at one, three, and six months after the completion of the intervention. Attrition and retention rates will be recorded during the study and reported in future publications of the study findings. The schedule of enrollment, intervention, and assessments are presented in Figure 1. The data collected at the different time points will be maintained confidentially as deidentified data by SS and stored for 5 years after the completion of the study. During the course of study participation, an increase in distress or symptoms noted in the VAS or reported by the participant will be communicated to the treating team, and necessary referrals will be made. To address the potential occurrences of suicidal crises during the study period, all participants and emergency contact persons will be provided with contacts of suicide helplines and crisis response plans for immediate crisis intervention. The therapist will provide limited assistance between sessions, in addition to making timely referrals for in-patient psychiatric management and involving the emergency contact person to ensure safety. Despite these precautions, any adverse events occurring during the study period will be recorded and reported as per standard reporting guidelines.
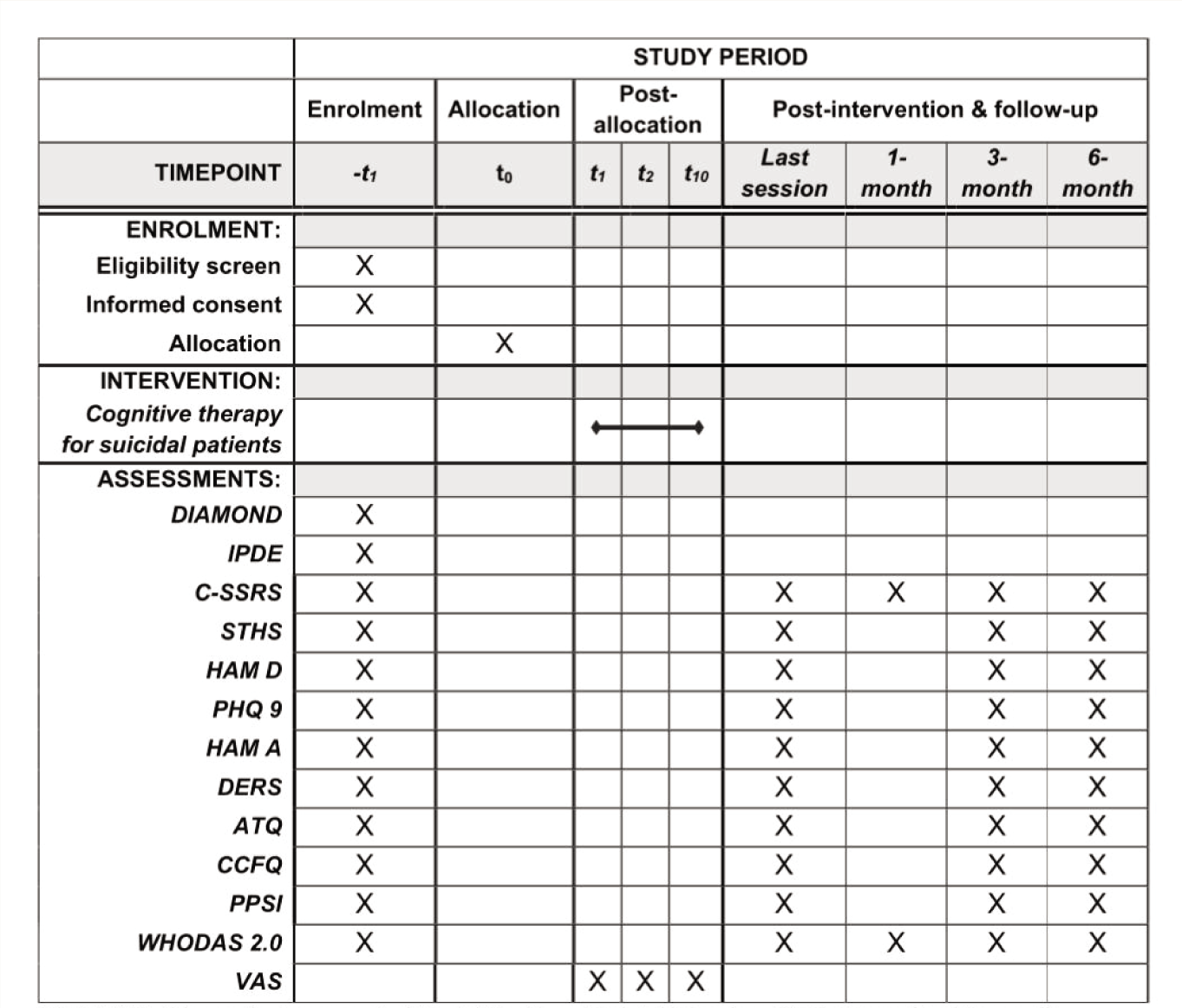
DIAMOND: Diagnostic Assessment of Mental Health and Related Disorders; IPDE: International Personality Disorder Examination; C-SSRS: Columbia Suicide Severity Rating Scale; STHS: State-Trait Hopelessness Scale; HAM D: Hamilton Depression Rating Scale; PHQ 9: Patient Health Questionnaire 9; HAM A: Hamilton Anxiety Rating Scale; DERS: Difficulties in Emotion Regulation Scale; ATQ: Automatic Thoughts Questionnaire; CCFQ: Cognitive Control and Flexibility Questionnaire; PPSI: Personal Problem-Solving Inventory; WHODAS 2.0: World Health Organization Disability Assessment Schedule 2.0; VAS: Visual Analogue Scales.
Therapist training, treatment adherence, and fidelity: The primary therapist who carries out the intervention will be the first author (SS) under the supervision of the second and third authors (MM and VSKR). The primary therapist is a licensed clinical psychologist in India with post-graduate training in clinical psychology. The therapist has undergone training in CBT, which is considered sufficient to deliver cognitive therapy interventions. Treatment adherence and fidelity will be maintained through case discussions with the supervisors (The second author is a trained CBT therapist with over 25 years of experience in delivering and supervising CBT across psychiatric disorders and the third author provides inputs on screening, recruitment, and delivery of interventions in this population) and other experts in the field. The expert rating of a random selection of the sessions on the Cognitive Therapy Rating Scale 32 will also assess it.
Data Collection and Statistical Analysis Plan
The data obtained using the tools will be entered in Microsoft Excel and subjected to statistical analysis using R software. Descriptive statistics such as mean, standard deviation, frequency, and proportion will be used to express the data. Appropriate tests of association will be carried out to study the association between the demographic variables and the outcome variables considered in this study. Repeated Measures ANOVA will be used to test the main hypothesis about change in suicidal ideation across different time points. Pearson’s correlation coefficient or Spearman’s Rho will be used to measure the relationship between variables. A P value of < .05 will be considered as statistical significance. The changes observed in the VAS assessing suicidality and distress will be examined using the Friedman test. Further, we plan to carry out subgroup analyses based on whether the clients have sought psychotherapeutic treatment after completion of the intervention, using appropriate tests of association.
Timelines
The study protocol was developed as part of a five-year doctoral program spanning from January 2021 to December 2025. The preparatory phase included a review of the literature, the development of the research proposal and subsequent modifications, and registration with a PhD. Committee and Institutional Ethics Committee, and registration in CTRI were carried out between Jan 2021 and April 2022. Participant enrollment in the research was initiated in May 2022 and is ongoing. The data collection phase is planned for completion by December 2024. Following this, analysis and publication of findings will be done. The flow of the study procedure is detailed in Figure 2.
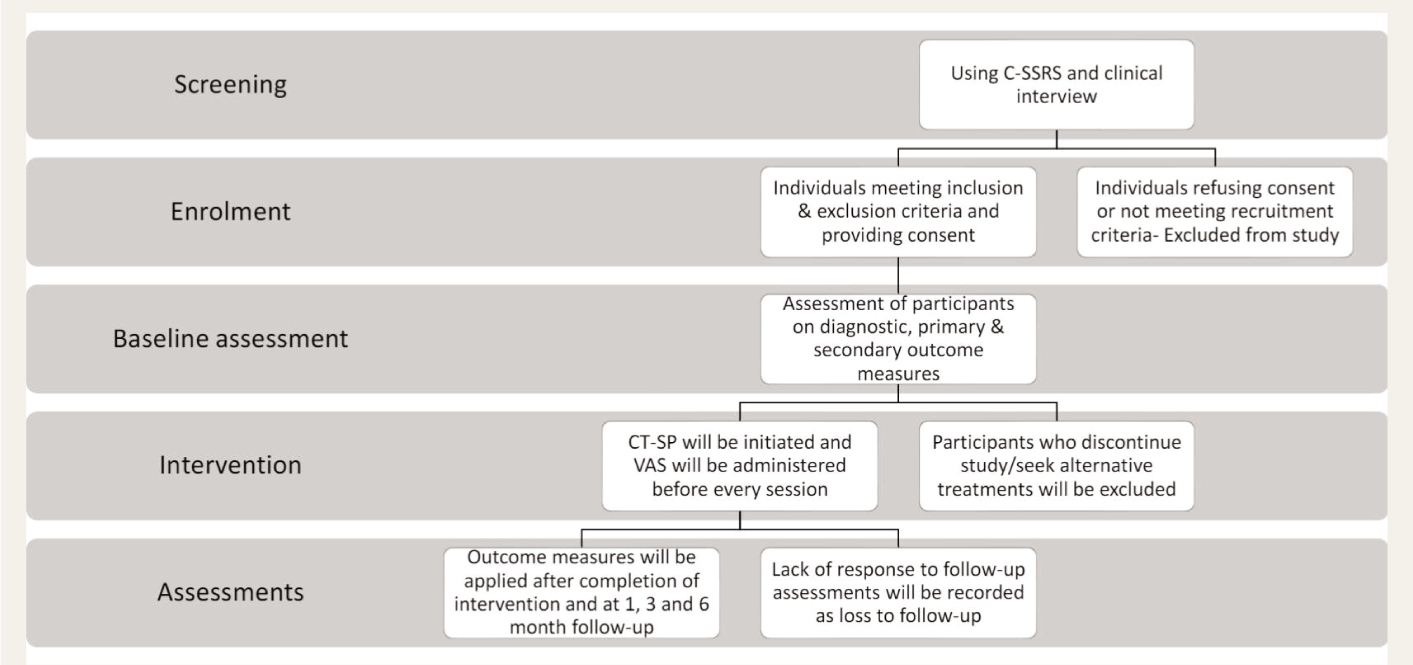
C-SSRS: Columbia suicide severity rating scale; CT-SP: Cognitive therapy for suicidal patients; VAS: Visual analogue scale.
Discussion
Considering the high suicide rates in India, the present study addresses the management of suicidality using an intervention (CT-SP) with a transdiagnostic approach. Such interventions that directly target suicidal cognitions and behaviors have been shown to facilitate better patient outcomes. 33 The psychological factors associated with long-term suicide risk, assessed by the secondary outcome measures, facilitate a holistic understanding that includes improvement in problem-solving, cognitive flexibility, and hopelessness.34–36 Safe and ethical application of the intervention in working with high-risk populations is ensured by monitoring the VAS scores through the intervention period. Despite the existing literature on the effectiveness of CBT for suicidality, its applicability may not be directly transferable to a culturally and economically diverse society like India.37–39 This study is among the first few research initiatives in India that aim to identify the utility of an evidence-based, structured, efficient suicide prevention intervention.
Expected Limitations and Strengths
The study’s strengths are the transdiagnostic view of suicidality, evidence-based intervention, comprehensive outcome measures, and a proactive approach to ensure participant safety. This highlights the relevance of the protocol for diverse clinical populations. It aims to provide insights into adapting a validated intervention to the local context and expand the existing literature on suicide risk management in India. The blended delivery format of the study, combining in-person and online sessions, will help improve the accessibility of potentially life-saving care beyond geographic barriers. Structured intervention can help therapists develop specific skills and competencies according to treatment guidelines. The brief nature of the intervention can reduce healthcare costs by providing timely, effective care, possibly preventing expensive in-patient treatments. This will be beneficial in improving the standard of care for suicidality in our country by serving as a foundation for future research. Limitations of the study include the lack of a control group, the delivery of intervention by a single therapist, the length of time required for assessments, and a relatively small sample size. This research design limits the possibility of attributing causality to the intervention and confirming its effectiveness.
Expected Outcomes
This study addresses a major public health concern by investigating the use of CT-SP as a brief, evidence-based intervention to reduce the severity of suicidal ideation and behaviors in psychiatric treatment-seekers in India. By taking a transdiagnostic approach and using a wide range of outcome measures, the study seeks to shed light on the effectiveness of this intervention in a context where research on suicidality is limited. Despite its limitations, this study contributes to existing knowledge on managing suicide risk in a country with high suicide rates. The results have the potential to inform future interventions and policy decisions aimed at reducing the burden of suicide.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Acknowledgements
The authors acknowledge the suggestions given by the members of the registration committee and the faculty of the Department of Clinical Psychology, National Institute of Mental Health and Neuro Sciences, Bangalore, India, in the development of the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration Regarding the Use of Generative AI
Grammarly and its generative AI were used while editing this manuscript. 40 We assume full responsibility for the entire content of the manuscript, including the parts generated by the AI tool.
Ethical Approval
The study protocol was approved by the ethics committee of the National Institute of Mental Health and Neuro Sciences (NIMHANS), Bangalore, Approval Number: No. NIMH/DO/BEH. Sc. Div./2021–22, dated: 29 January 2022, and registered with the Clinical Trials Registry of India in April 2022 (CTRI/2022/04/042090).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: University Grant Commission, India; National Eligibility Test- Junior Research Fellowship (NET-JRF).
Patient Consent
Written informed consent was obtained from each participant prior to enrollment in the study.
Reporting Guideline
The SPIRIT guideline is uploaded as Supplementary online material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
