Abstract

In the past two decades, the realm of nicotine consumption underwent a radical change with the introduction of the basic design of electronic nicotine delivery devices (ENDS) or e-cigarettes. While on one hand the ENDS were proposed as a substitute or replacement to the existing nicotine-containing products that contained smoking tobacco forms, on the other hand, these offered another avenue to consume nicotine. Since their introduction, the ENDS have captured attention of consumers, researchers and policymakers alike. The discussion on use of ENDS has gathered greater momentum over the past few years with manufacturers investing extensively in their products on the one hand, and growing voices challenging the proposed benefits and highlighting the harmful consequences, on the other. An intense and controversial debate regarding the safety, efficacy, and long-term effects of ENDS has ensued. 1 At the core of this debate lie the benefits offered by the ENDS and the cost (undesirable consequences) of these benefits.
In this article, we explore the implications for India with respect to the ENDS in context of the experience that has emerged over the past years. We aim to consider the implications of the possible introduction of ENDS as a smoking cessation medicinal product as well as an alternative to the smoking products that are already available in the country.
This article is aimed at offering insights into the controversies and limitations in context of use of ENDS. We have tried to highlight the key themes that should be taken into consideration while deliberating on policy for ENDS from a scientific perspective.
We have accessed the relevant published academic literature as well as documents that have chronicled the experience and impact of use of ENDS in other jurisdictions. We have focused on three jurisdictions with diverse regulatory control of ENDS. Later we have presented the Indian scenario and discussed our views on how the experience from the other jurisdictions offers learning that is relevant to Indian context.
What Is the Current Status of Legislation in India?
India enacted The Prohibition of Electronic Cigarettes Act 2019 that prohibits and penalises the production, manufacturing, import, export, transport, sale, distribution, storage and advertisement of these products.
2
The Indian Council of Medical Research (ICMR) also published a white article in the month of May 2019 that concluded that
Based on the currently available scientific data from multiple streams of research, the Council recommends complete prohibition on ENDS or e-cigarettes in India in the greater interest of protecting public health, in accordance with the precautionary principle preventing public harm from a noxious agent.
3
While the Prohibition of Electronic Cigarettes Act 2019 does not preclude use of the products as a medicinal products if registered under the Drugs and Cosmetics Act of 1940, the Drugs Technical Advisory Board in its 83rd meeting held in June 2019 recommended that
The manufacture, sale (including online sale) and distribution of Electronic Nicotine Delivery System (ENDS) including E-cigarette, Heat-Not burn devices, Vape, e-Sheesha, e-Nicotine, Flavoured Hookah and the like products shall be prohibited under Section 26A of the Drugs and Cosmetics Act, 1940 as well as their import (including personal purpose) shall also be prohibited under Section 10A of the said Act.
4
Consequently, currently the ENDS are not allowed to be made available either as a consumer product or as a medicinal product in the country.
What Are the Regulatory Frameworks for ENDS Globally?
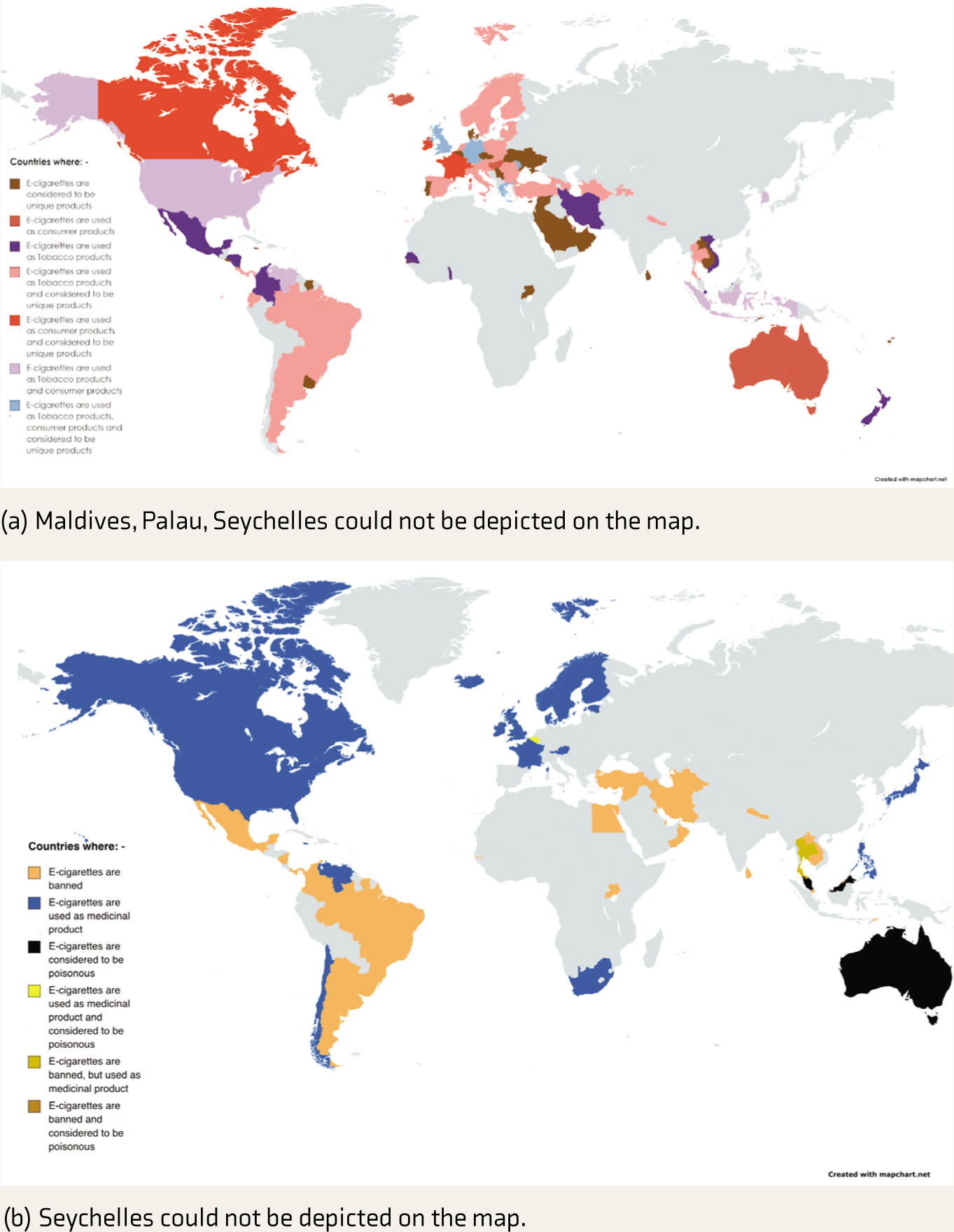
The landscape surrounding ENDS has undergone a significant transformation as a growing number of countries have enacted bans and implemented stringent regulations, and other countries easing the restrictions and even promoting its use. Of all the formulations of the psychoactive substances available globally this is the most diversely regulated. The Institute of Global Tobacco Control reported that by July 2022, 109 countries or jurisdictions had either regulated or banned ENDS. 5 Smith and Hilton (2022) reported that 30 countries had prohibited the ENDS, 20 had registered these as medicinal products, and four had identified these as poisonous or hazardous substances. 6 Also, 54 countries had identified these as tobacco products, 15 as consumer products and 68 as unique products. The regulatory control of the ENDS across different jurisdictions has been presented in Figure 1.'
(a and b) Regulatory Framework for ENDS Across Different Jurisdictions (Based on Data Presented in Smith and Hilton, 2022).
Do ENDS Cause Physical Harms?
The ENDS have been subject to research aimed at assessing their adverse health effects. While some of the proposed adverse effects remain unsubstantiated in research studies, the others have been well established. Table 1 offers an overview of the level of evidence for different types of adverse effects of ENDS on health.''
Level of Evidence for Different Adverse Effects due to ENDS Based on Banks et al. 2023 25 .

EVALI: e-cigarette or vaping use-associated lung injury.
The current evidence suggests that the ENDS are not completely safe. Also, it can be concluded that the adverse effects due to smoking are more diverse, more common and are responsible for a greater health burden at this point in time, although there are no head-to-head comparisons between the adverse effects due to ENDS and smoking.
Are ENDS Effective for Smoking Cessation?
While the systematic reviews agree that ENDS are as effective as Nicotine Replacement Therapy (NRT) for smoking cessation, there is a difference in interpretation and conclusions about the studies that have identified ENDS to be more effective than NRT. A review published in the year 2023 included five randomised controlled trials (RCTs) and concluded that among individuals attempting to quit smoking nicotine e-cigarettes were more efficacious than conventional NRT. 7 On the contrary, a previous review that was published in the year 2021 and had included 10 RCTs concluded that there was no clear evidence of a difference in effect between nicotine-containing e-cigarettes and NRT on incidences of smoking cessation. 8 It appears that the conclusions drawn across these reviews are shaped, to a large extent, by the inclusion criteria they used for identification of the eligible studies based on which the inferences are drawn.
Another significant issue that is relevant to this discussion stems from a living Cochrane review that assesses evidence for effectiveness of electronic cigarettes for smoking cessation. 9 Many of the authors on this review have disclosed that they had undertaken educational sessions sponsored by manufacturers of smoking cessation medications; received honoraria, travel support, support in kind from manufacturers of smoking cessation medications; provided consultancy to manufacturers of smoking cessation medications; were investigators on studies by ENDS manufacturer; were investigators on clinical trial on a project that supplied ENDS free of cost; served on the advisory board of manufacturer of smoking cessation medicine. In fact, Cochrane acknowledged that the 2014 version of this review was ‘not compliant with the Cochrane commercial sponsorship policy, as updated in 2014’. The authors with these conflicts of interest continued to have authorship on the subsequent versions of the review.
This variation in the understanding of effectiveness of ENDS as effective smoking cessation intervention or a safe(r) substitute to smoking also extend to the national policies. While countries such as the UK have accepted their effectiveness, others such as Australia have held a position that these are not effective while not being safe as well. 10
What Has been the Experience from Countries Where ENDS Are Available?
The United Kingdom (UK) has one of the most permissive regulatory frameworks on e-cigarettes. E-cigarettes have been available in the UK for many years. These are regulated under the Tobacco and Related Products Regulations. The government has identified e-cigarettes as the key strategy to bring down the smoking prevalence in the UK. It reflected, most recently, in the national ‘swap to stop’ scheme where the aim is to encourage one million smokers to swap smoking for vapes to help cut tobacco smoking rates. 11
The data collected by the Action on Smoking and Health has demonstrated that there has been an increase in the percentage of population who currently used e-cigarettes in Great Britain from 1.7% in 2012 to 9.1% in 2023. 12 Also, among the current adult e-cigarette users, the percentage of ex-smokers has increased from 28% (2013) to 56% (2023). The percentage of dual users (those who used e-cigarettes and smoked) has seen a decline from 67% (2013) to 37% (2023). In addition, the percentage of never-smokers who are currently using e-cigarettes has remained consistently lower than 10% over this period. All these observations are in the intended direction whereby a larger proportion of the adults who smoke are taking up e-cigarettes as desired by the government. At the same time, most of the adults who did not smoke also do not tend to take up c-cigarettes either.
However, there are some other observations that either are not encouraging or are even a cause of concern. While there has been a decline in the percentage of current smokers who have never tried e-cigarettes from the high of 62% in 2013, the number is still substantial in 2023 (27%). Also, a significant percentage of adults who are current smokers tried using e-cigarettes but discontinued these in favour of smoking (43% in 2023).
The data from the Smoking Toolkit Study suggests that there has been an increase in the percentage of adults in England who used higher-strength e-cigarettes (2% or more) over the years since 2016. 13 This despite the government restricting the strength of nicotine in e-cigarettes to 2% in 2016. In addition, more than 2.5 million illicit e-cigarettes (that did not conform to the regulatory framework) were collected in the UK between 2020 and June 2023. 11 These observations suggest that the regulatory framework is not sufficient and efficient to ensure that the unintended products do not enter the market. This becomes more significant given the fact that the access to e-cigarettes among adults is easy as these are sold at retail shops where any adult can purchase them.
The limited effectiveness of the regulatory framework on e-cigarettes is also reflected in the data on their use among the underaged. While the sale of e-cigarettes to those under 18 years of age is prohibited in the UK, a substantial percentage of those aged 11–18 years reported their use. 14 The ever use of e-cigarettes has reached 15% (11–15 years old); 34% (16–17 years old and 38% (among 18 years old). Also, ‘just to give it a try’, ‘other people use them, so I join in’, ‘I like the flavours’ are the commonly cited reasons for use of e-cigarettes among non-smokers as well as never-smokers. Use of e-cigarettes as an aid while trying to quit smoking (endorsed by 1.3%) and as an alternative to smoking (endorsed by 0.6%) is rather rare. Among the current users of e-cigarettes aged 11–17 years, around one-third tried e-cigarettes before trying smoking. The percentage of those underage who ever smoked and the percentage of those who ever used e-cigarettes but never smoked is getting closer. Another NHS-funded study found that use of e-cigarettes was the most strongly associated factor with current smoking status among those aged 11–15 years. 15 The strength of association was higher than other factors such as taking drugs, having friends who smoke, family does not discourage smoking, having smokers at home, taking alcohol and sex.
Australia has taken a contrarian approach to e-cigarettes. The policy and regulatory approach to electronic cigarettes identify these as gateway to a lifetime of nicotine addiction among children and young adults; increase uptake of smoking among young; having harmful consequences to the users and the bystanders; and nicotine being a poison. 16 The e-cigarettes have been identified as detrimental to the public health gains made in terms of reduction of the smoking prevalence in the country over the past decades. E-cigarettes are not recommended as first-line treatment for smoking cessation in the country. The e-cigarettes are available only on prescription from a medical practitioner in the country. 17
A recent report submitted to the Department of Health and Aged Care documented that the six-month prevalence of current use of e-cigarettes among those aged 14 years or more has increased over the years from 2018 onwards. 18 The most recent rate was reported at 7.7%. The increase has been the highest among those aged 18–24 years (25.1% in 2022); 14–17 years (15.8% in 2022); and 15–34 years old (14.0% in 2022). The increase has been comparatively lower among older age groups (4.0% for 25–49 years and 1.4% for those aged 50 or more for 2022). These increases in rates suggest that despite the regulatory restrictions the e-cigarettes are accessible even to those aged less than 18 years.
In the United States of America (USA) e-cigarettes have been the most used nicotine-containing product among the youth since 2014, with the current rate of use of e-cigarettes being 14.1% and that of all tobacco products being 16.5% among high school students. 19 Nicotine, however, is not the only vaping product being used by the students. The trend in vaping nicotine among 8th, 10th, and 12th graders is closely followed by the trends in vaping marijuana. 20 The lifetime prevalence of vaping nicotine and vaping marijuana among 12th graders in 2022 was 38.8% and 27.5%, respectively.
In addition, no applications have been received for registration of any of the ENDS as a medicinal product in the UK and Australia. The Medicines and Healthcare Products Regulatory Agency (MHRA) in the UK had granted a licence to one of the companies for a nicotine inhaler. However, the company quit plans to market it as it wanted to focus on consumer products instead. 11 So, despite the government in the UK deciding that all NCPs shall be regulated by MHRA as medicines to make these products safer and more effective to reduce the harms of smoking in the year and then updating the guidance in 2021, currently no ENDS is registered as a medicinal product. Similarly, while the ENDS can be registered as a therapeutic good under the Therapeutic Goods Association, no applications were received for the same. In the USA the manufacturers have argued that their products are targeted at smokers and not at those seeking to quit and hence these should not be regulated by the Food and Drugs Administration (FDA).
Some inferences can be drawn from these observations from three jurisdictions with different approaches to e-cigarettes. These have been summarised in the Box 1).
Inferences Drawn from Observations Form the UK, Australia and the USA in Context of e-cigarette use.
• E-cigarettes use does not seem to be a concern among never smokers
• Dual use happens among a substantial proportion of users
• E-cigarettes do not work for some (close to half of) smokers
• Some (around one third) smokers not interested in using e-cigarettes
• Smokers are able to quit using current mechanism of sale that do not involve the health care professionals with the end users and sellers managing between them (in a large proportion of cases)
• Electronic cigarettes are the first exposure to nicotine in a sizable proportion of population
• Most use e-cigarettes out of curiosity and conforming to presumed normative behaviour
• Use of electronic cigarettes strongly associated with smoking
• The legislative/regulatory framework is not fully effective against underage use of e-cigarettes (like smoking)
• Proportion of ever electronic cigarette users (and never smokers) getting closer to ever smokers
• Regulatory status does not necessarily translate into availability and use
• Nicotine is not the only vaping product available
The Indian Context
While we have presented the observations and experiences from other countries, it is important to state that the Indian context is different in many ways. Consequently, it shall be inappropriate to extrapolate these to the Indian setting directly.
The situation in India differs from the other countries in many ways. First, smoking forms of tobacco are not the most common form of tobacco used in India. According to the Global Adult Tobacco Survey (GATS 2,) 2016–2017, the number of only smokeless tobacco users (including khaini, gutkha, betel quid with tobacco, mishri, gul, gudakhu) is almost twice that of smokers among men and twelve times higher among women. 21 Second, among smokers, biri users outnumber the cigarette users. Biri is a relatively less expensive tobacco product as compared to cigarettes. Also, the money spent on the last tobacco purchase is smaller at INR 12.50 for biris and INR 29.56 for cigarettes. This is relevant as in certain Western countries like the UK the cost of using e-cigarettes in the long run is lesser as compared to smoking. Bringing the cost of e-electronic cigarettes lower than that of biri would be challenging and unlikely if not subsidised. Third, currently, the use of NRT for smoking cessation is low. Among those who tried to stop smoking in the past 12 months, only 4.1% used pharmacotherapy. Fourth, the awareness of e-cigarettes among those aged 15 years or more is low. Only 3.03% of respondents reported being aware of the e-cigarettes in the GATS 2. Similarly, the use of e-cigarettes was low at 0.66% among those aware of these. The awareness about e-cigarettes among those aged 13–15 years, however, is higher at 26.9% as reported in the Global Youth Tobacco Survey. 22 The use is still low at 2.8%.
In addition, there has been a downward trend in the rates of use of tobacco products both for smoking as well as smokeless forms—an observation similar to many developed countries. However, even now the burden due to tobacco use in the country is substantial. Another important fact in this context is that smoking products continue to remain available despite of these being responsible to high mortality and morbidity; e-cigarettes being banned in the country; gutkha being banned in many states; underage smoking being a significant concern despite of the regulatory framework in place; a lesser proportion of Indians interested in smoking as previous years; effective interventions being available for smoking cessation; tobacco products contributing to less than 3% of the gross tax revenue to the central government 23 and years of advocacy efforts. This fact is a reality check on how challenging it is to withdraw a consumer product despite a strong argument that can be put in place.
Implications of Introduction of ENDS on Various Stakeholders
The ENDS can be introduced in two ways—as a consumer product or as a medicinal product. In case the ENDS are introduced in the country, given the current level of understanding around its use (based on evidence from other countries) and the contextual factors specific to India, there shall be various potential implications for different stakeholders.
In case the ENDS are introduced as a medicinal product, the smokers (mainly cigarette users) shall have access to an effective (at least as effective as NRT) and ‘safer’ (as compared to cigarettes) treatment option. However, the advantage offered by ENDS over the existing pharmacological options (NRT) shall remain debatable given the varied interpretation of the evidence of its effectiveness. A large proportion of smokers in the country who use biri will find it difficult to access these due to cost (unless these are subsidised). Another challenge shall be to get the manufacturers interested in introducing such products as they may be more interested in introducing these as consumer products rather than medicinal products.
In case the ENDS are introduced as a consumer product, the smokers (mainly cigarette users) shall have access to a ‘safer’ (as compared to cigarettes) alternative to smoking; the biri smokers are unlikely to afford these (unless the prices are subsidised).
In either of these scenarios, the underage non-smokers can be expected to have access to these given the extent of effectiveness of the existing regulatory framework as suggested by the data on underage use of tobacco products. Given the low baseline level of current use and an increase in visibility of the ENDS, the illicit market is expected to grow. This shall be driven by the supply of those products that do not conform to the established standards (e.g., higher nicotine strength products and products available at lower than the regulated market prices). Also, the role of the healthcare professionals in either of these scenarios remains to be defined. They can make suggestions and offer advice to those interested in looking for alternatives to smoking or are interested in quitting. However, to support wider and easy availability of ENDS as either a medicinal product or a consumer product these shall be made available the way the existing NRT are available (that can be accessed OTC) or on local pan shops and kiosks where the cigarettes are currently sold. Any other approach may be deemed restrictive.
In case the ENDS are not introduced as a medicinal product or a consumer product the underage non-smokers shall continue to have access to smoking tobacco products that are one of the largest known contributors to mortality and morbidity, albeit their use is on the decline; and there shall remain a possibility of growth of the illicit market as the demand grows. In addition, the current smokers shall continue to have the option of the currently under-prescribed NRT.
Impact of the Perspective
The way the messaging around the continuum between smoking and no tobacco/nicotine is conceptualised has important public health implications. The discussion on the role of ENDS is shaped largely by the perspective on the tobacco products and harms related to them. In one of the paradigms used to explain this, there are two extremes—one being smoking that is pitched to be ‘not safe’ and the other being no tobacco/nicotine use that is the ‘safest’ option. In between lie the e-cigarettes (ENDS) that are labelled as ‘safer’ than smoking.
The other paradigm also has smoking and no tobacco/nicotine use at the two extremes. However, in this paradigm the smoking is labelled as ‘unsafe’ and no tobacco/nicotine as ‘safe’. The e-cigarettes (ENDS) shall still be somewhere in between these two extremes, but these shall be labelled as ‘less unsafe’ in this second paradigm (Figure 2).
Two Paradigms to Conceptualise and Communicate the Safety Profile of Nicotine Products.
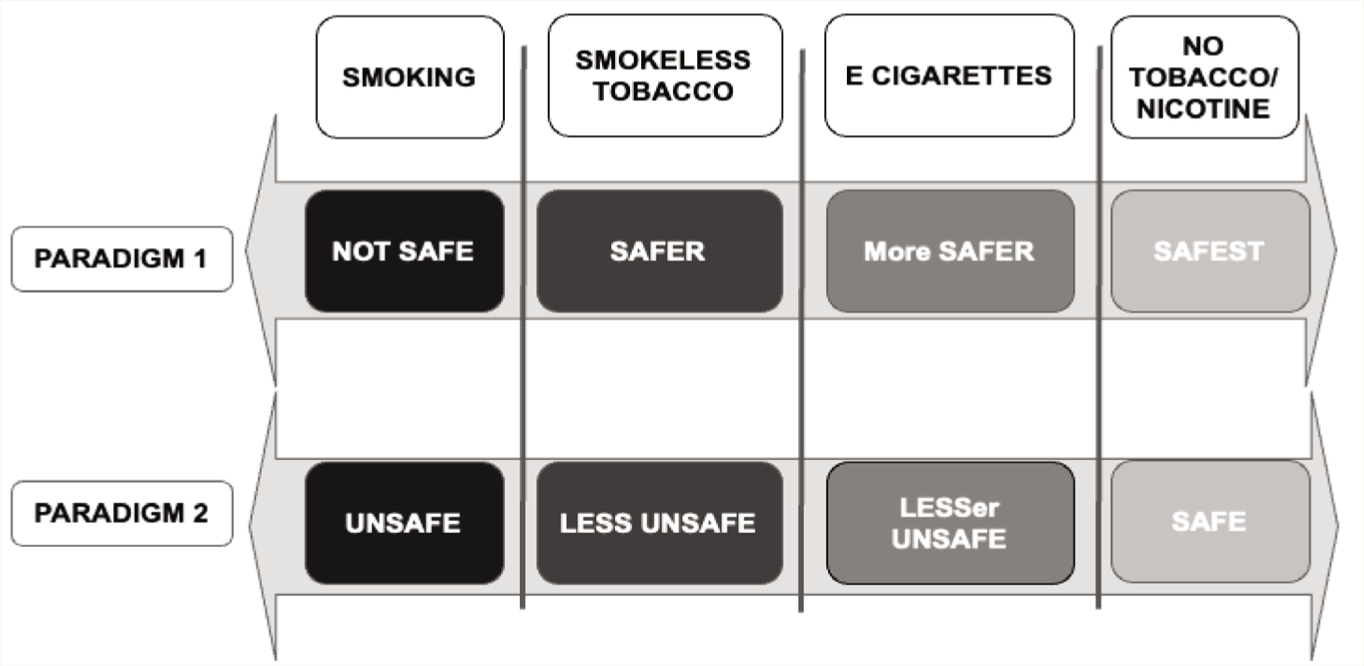
These two conceptualisations and their context can be better understood if we incorporate the smokeless tobacco products into these paradigms. Smokeless tobacco products are associated with less types as well as less magnitude of harm to the users. Given this knowledge, if we try to fit in the smokeless tobacco products in the first of the two paradigms discussed above, these shall be placed somewhere between smoking and e-cigarettes and then these shall be labelled as ‘safer’ and e-cigarettes shall be labelled as ‘more safer’. However, in the second paradigm, these shall be labelled as ‘less unsafe’ and the e-cigarettes shall be labelled as ‘lesser unsafe’.
The concept of smokeless tobacco forms being ‘safer’ than smoking is not a hypothetical one. In fact, the use of a smokeless tobacco product (snus) has been proposed as a harm reduction strategy against harms due to smoking. 24 The data from Sweden suggests that the prevalence of smoking is lowest in the country among all the member states of the European Union. However, the concept of promoting smokeless tobacco use as a ‘safer’ alternative to smoking remains debatable and would be hard to fathom in Indian context.
Implications of Exercising the Options
India has two obvious options to choose from with regards to the ENDS-introduce these now or not to introduce these now, that is, maintain the status quo. Both these have their own set of implications that shall impact a large section of the population.
If ENDS are introduced, these are likely to benefit a proportion of current (cigarette) smokers (who may otherwise access other existing interventions). However, there are concerns in the form of interpreting the existing evidence on their effectiveness; increased access to the ENDS among those underage and a possible growth of the illicit market (given the low current levels and increased interest fuelled by increased visibility). In this scenario, one waits for more data on long-term safety of ENDS and in case there are ‘concerning’ findings then a decision on withdrawing these might have to be taken—a decision that is likely to come with great difficulty as suggested by the experience with cigarettes.
On the other hand, if ENDS are not introduced now and the status quo is maintained, there is likely to be a reduced chance of future generations initiating their use, although a proportion of them shall use other tobacco products the way they do now, with a possibility in reduction of prevalence in the future given the reducing trend. Also, at least a proportion of them (which is low currently) shall still have access to the ENDS through the illicit channels. While we wait for more definite evidence on their superiority as smoking cessation intervention and safety, we shall be withholding access to an intervention and/or a product that is ‘safer’ or ‘less harmful’ than smoking to a proportion of current smokers (mainly cigarette users) who otherwise can access to other tobacco cessation interventions.
It is the pros and cons of both these approaches that pose the ethical dilemma in the context of ENDS. We need to remain open to carry this discussion forward as more data is generated, with an aim to move closer to an approach that does not deprive those likely to benefit from scientific discovery, while at the same time keeping the unintended consequences to the lowest possible levels. Also, we must ensure that the discussion on ENDS is not taken over by the non-scientific, opinionated discourse, that shall do more harm than good for sure.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Declaration Regarding the Use of Generative AI
None used.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
