Abstract
Background:
Vilazodone, a novel selective serotonin reuptake inhibitor and 5-HT1A partial agonist, was approved in 2011 for treatment for major depression. We aimed to compare the efficacy and safety of vilazodone versus escitalopram in patients with major depression at 4 weeks.
Methods:
Participants (
Results:
No significant difference was noted between groups on depression scores at study endpoint (
Conclusion:
Clinical ratings of major depression did not differ significantly between vilazodone and escitalopram groups at the end of 4 weeks. Our findings are limited by lack of statistical power to detect smaller differences between groups, should they exist.
Flexibly dosed vilazodone (20–40 mg/day) was not superior to escitalopram (10–20 mg/day) for the primary outcome of endpoint depression scores at the end of 4 weeks of treatment. Diarrhea was more commonly reported in the vilazodone group. Our findings should be interpreted with caution, given that the study was underpowered to detect a medium or small effect size.Key Messages:
A ntidepressants are now the mainstay in the treatment of depression. The goal of treatment of depression is to induce response and remission. Selective serotonin reuptake inhibitors (SSRI) are often the preferred first-line antidepressants, owing to a better adverse effect profile and low risk of toxicity in overdose compared to older agents. Nevertheless, treatment-emergent sexual dysfunction with SSRIs is often a concern and the most common cause of discontinuation of these agents; figures quoted in studies are 24%–73%.1, 2
Comparative assessment of various antidepressants shows that escitalopram is often the first choice antidepressant, judged mainly by combined efficacy and tolerability. 3 However, a major drawback of escitalopram is treatment-emergent weight gain and sexual dysfunction, which predisposes to treatment discontinuation in a significant number of patients. 4 Hence, there is a need for an antidepressant that should combine the advantages of a rapid onset of action and a more favorable adverse effect profile, including lower sexual dysfunction, weight gain, and other adverse effects.
Vilazodone, a serotonin partial agonist reuptake inhibitor, was approved by the Food and Drug Administration in January 2011 for the treatment of depression. Its dual action of serotonin reuptake inhibition and partial agonism on serotonergic 5HT1A receptor is postulated to enhance its antidepressant effect and tolerability. 5 Emerging evidence points to a better side-effect profile and earlier onset of therapeutic action for vilazodone, compared to SSRIs. 6
Given this background, head-to-head comparison studies of newer agents against the ongoing standard of care are necessary to establish their efficacy and tolerability. To date, only three studies have performed a head-to-head comparison of escitalopram and vilazodone, and the results were inconsistent.7–9 The study by Bathla et al. was an open-label randomized, controlled, head-to-head comparison of vilazodone with a maximum dose of 40 mg/day versus escitalopram maximum dose of 20 mg/day over 12 weeks, showing better efficacy, lesser weight gain, and lesser sexual dysfunction with vilazodone. 6 In a randomized, prospective, parallel-group, open-label clinical study by Kadam et al., newly diagnosed patients with major depressive disorder (MDD) were randomized to receive vilazodone 20 mg daily, escitalopram 20 mg daily, or amitriptyline 75 mg daily for 12 weeks. Vilazodone was more efficacious and well-tolerated compared to escitalopram and amitriptyline. 7 Sexual side-effects and weight gain were not the focus of this study. The third one was a randomized open-label comparison of escitalopram versus vilazodone over 6 weeks. Both drugs were effective in decreasing depression and anxiety scores. In comparison, escitalopram was found to be better than vilazodone. 8 This study also has not mentioned sexual issues or weight gain.
Hence, we conducted this study to compare treatment efficacy, in terms of clinical depression symptom ratings, between vilazodone and escitalopram at 4 weeks study endpoint (primary objective) and also compare treatment-emergent sexual dysfunction and weight gain at the study endpoint (secondary objective). Additionally, we also planned to compare 2-week depression symptom ratings between groups (secondary objective); this was done to examine the proposed early onset of action with vilazodone.
Materials and Methods
Setting and Sample
The study was carried out at the psychiatry outpatient clinic of a tertiary care cum teaching hospital in Kerala, South India, from August 2020 to November 2020. All clinical diagnoses were made by one consultant psychiatrist. The eligibility criteria were as follows:
Inclusion Criteria
Adult outpatients aged 18–65 years, with a diagnosis of MDD without psychotic features based on Diagnostic and Statistical Manual of Mental Disorders—5th edition (DSM-5) criteria 10 and scoring ≥ 15 on the 17-item Hamilton Depression Rating Scale (HDRS), were included 11 if they have been drug-free for 8 weeks.
Exclusion Criteria
Patients with other comorbid psychiatric disorders, including substance use disorders
Patients with concurrent major physical illnesses such as malignancies or renal/cardiac failure
Patients who were acutely suicidal, pregnant, or lactating mothers
Trial Design and Workflow
Eligible subjects were randomly assigned, using a computer-generated random number sequence, to receive either vilazodone (
Assessments
Clinical assessments were done at baseline and at the 2-week and 4-week follow-up time points. The following measures were employed.
Hamilton Depression Rating Scale (HDRS)-17 (primary outcome) 11 : This clinician-administered tool is widely used to rate depression symptom severity and also as a guide to assess clinical recovery. Items are scored on a 3-point or 5-point Likert scale, with total scores ranging from 0–52. The following cutoffs have been proposed for stratifying clinical severity: mild depression (8–16), moderate depression (17–23), and severe depression (≥24). 12
Montgomery–Asberg Depression Rating Scale (MADRS) (secondary outcome): 13 This is a popular 10-item, observer-rated measure to assess the intensity of depressive symptoms. Items are rated on a 7-point Likert scale ranging from 0 (absent) to 6 (severe), total scores may range from 0–60. The MADRS predominantly focuses on the core symptoms of depression (sadness/lassitude/cognitive symptoms such as pessimistic thoughts), in contrast to HDRS, which emphasizes the physical symptoms of depression. Hence, it was used as an additional measure of depression.
Clinical Global Impression—Severity (CGI-S) scale (secondary outcome) 14 : It is part of a 3-item observer-rated measure that seeks to assess the severity of illness. Ratings are made relative to past clinical experience with other subjects having the same diagnosis. A seven-point Likert scale ranging from 1 = “normal” to 7 = “among the most extremely ill patients” is used for the ratings.
Rater-blinded assessment of depression was done using HDRS, MADRS, and CGI-S at baseline, 2nd, and 4th weeks. This was done by an independent qualified psychiatrist with prior experience in administering the measures and not involved in any other aspects of the trial. Adverse events were elicited by directly querying the patients at all the study visits. A specific scale was not used to assess sexual side-effects; it was recorded verbatim. Additionally, we recorded the weight of study participants at every visit.
Sample Size Calculation
We estimated that a sample size of 48 (24 per group) would have an 80% power to detect a difference of 2.5 units on the HDRS (Standard Deviation [SD] 1 = 3.5 and SD 2 = 2.7)
15
between groups, with a precision level of 5%, using OpenEpi software version 3.01 (Copyright (c) 2003, 2008 Andrew G. Dean and Kevin M. Sullivan, Atlanta, GA, USA;
Ethics Approval
Written informed consent was obtained from all study participants. Prior approval was obtained from the Institutional Ethics Committee, and the study was registered with the Clinical Trial Registry of India (CTRI/2020/07/026520).
Statistical Analysis
Descriptive sample data were expressed using mean (with standard deviation [SD]) or median (with interquartile range [IQR]) for continuous variables, while frequencies with percentages were used for categorical variables. We used Student’s
The primary analysis was performed on the Intention-to-treat (ITT) sample, which we defined as every patient randomized regardless of subsequent protocol deviations. In addition, we performed two analyses—a prespecified analysis on the 2-week scores—to determine if the groups separated early on the clinical outcomes and an exploratory analysis on the completer sample. The last observation carried forward method was used to impute missing values. Log transformation was attempted for outcome measures that violated normality assumptions. For those variables that conformed to normality following transformation, a one-way between-subjects analysis of covariance was conducted to examine the impact of group assignment on endpoint outcome measures while simultaneously controlling for baseline scores; otherwise, the Quade test, a nonparametric alternative, was done with nontransformed scores itself, to avoid loss of information. The main ITT analysis was for the prespecified primary outcome (endpoint HDRS scores); additional analyses were for prespecified secondary outcomes (MADRS and CGI-S). Effect sizes were computed as standardized mean difference (Cohen’s
To check the independent effects of variables that were significantly different between groups at baseline, we performed multivariable linear regression analyses with all these variables, in addition to the grouping variable, as the independent variables and endpoint HDRS (primary outcome), MADRS, or CGI-S (secondary outcomes) scores as the dependent variable, in three sequential analyses. The total number of independent variables was capped at five in each of these analyses to avoid model overfitting; these were group assignment, sex, baseline HDRS, baseline MADRS, and baseline CGI-S scores. Similar multivariable analyses were performed to check the potential influence of endpoint clonazepam dose on outcome variables; again, the covariates in the model were grouping variable and those that were significantly different between groups at baseline, subject to a maximum of five covariates in the model.
Results
Sample Description (Table 1)
Comparison of Demographic and Clinical Variables at Baseline.
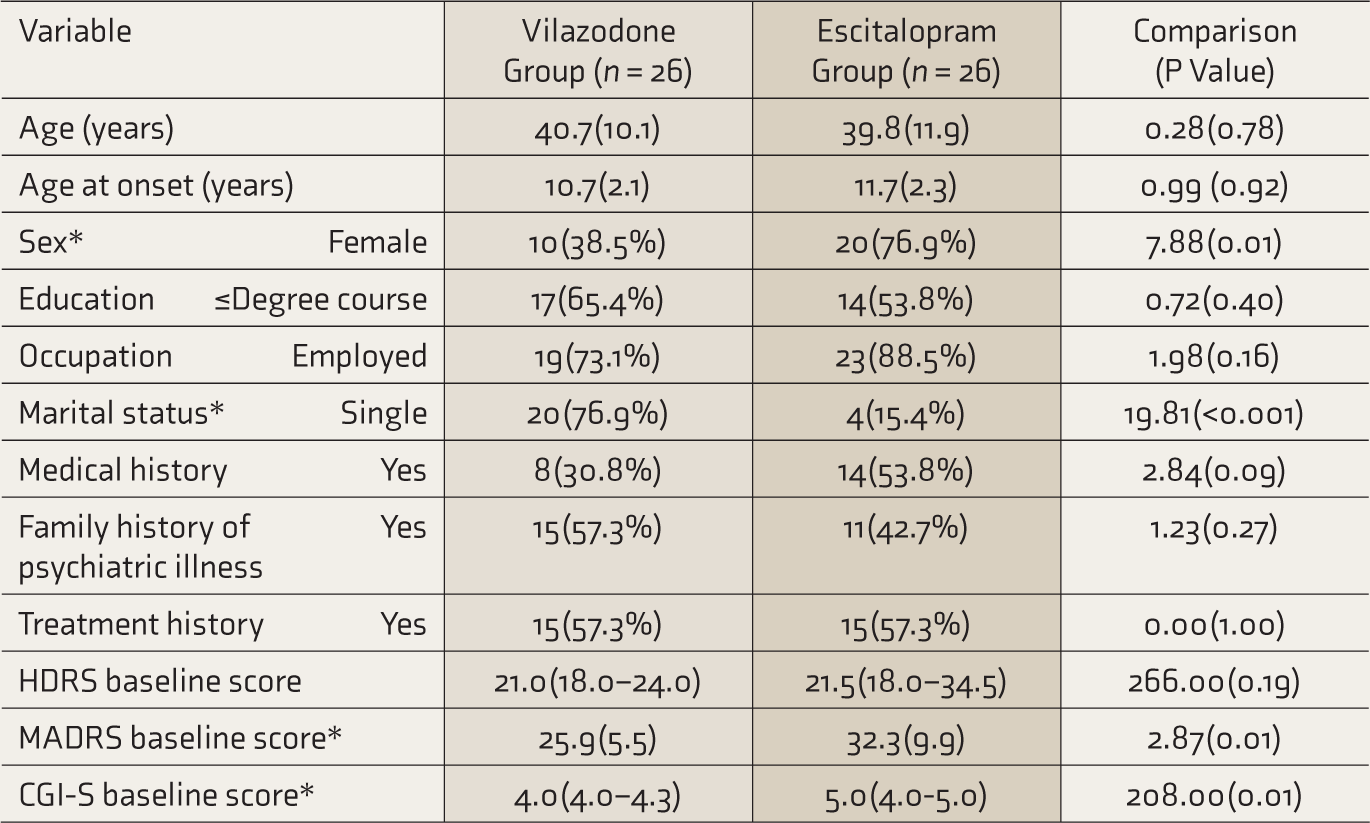
CGI-S: clinical severity of illness, HDRS: Hamilton Depression Rating Scale, MADRS: Montgomery–Asberg Depression Rating Scale. Values expressed as mean (standard deviation), median (interquartile range), or frequency (percentage). Comparisons done using independent t-test, Mann–Whitney U test, or chi-square test. * Significant at P < 0.05.
Participants’ mean age (±SD) was 40.3 (±10.9) years; the age range was 22–64 years. Most participants were aged ≤ 45 years (
Sample Disposition (Figure 1)
Consolidated standards of reporting trials (CONSORT) flow diagram.
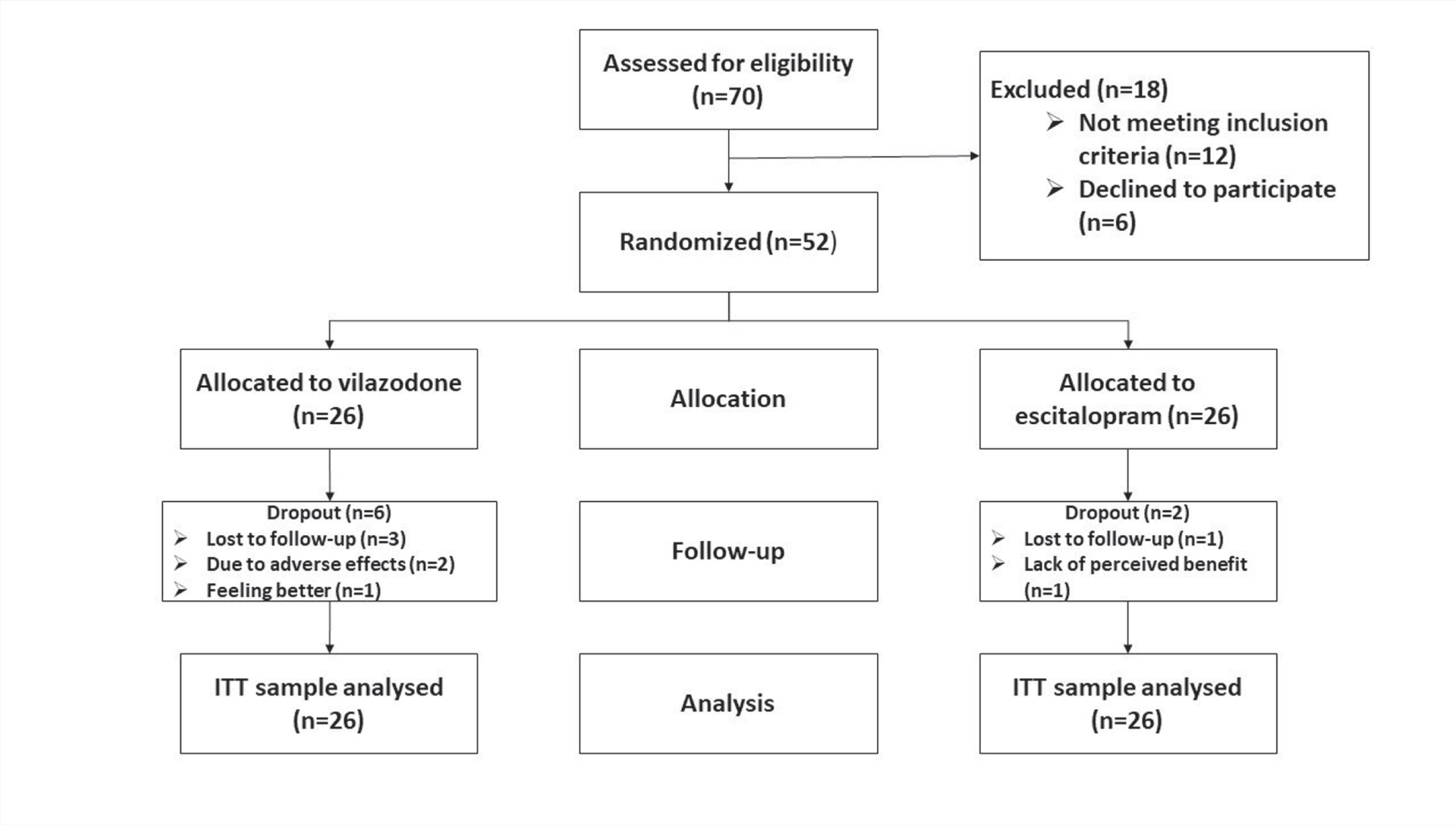
Of the 52 included subjects, 44 completed the study (4 weeks). Of the 8 dropouts, 6 and 2 were from vilazodone and escitalopram groups, respectively; there was no statistically significant difference in proportion of dropouts between groups (χ
2
= 5.36, P = 0.25). Further, all those who dropped out provided data till 2 weeks. Reasons for dropouts (
Primary Outcome: Endpoint HDRS Scores (Table 2)
The groups did not significantly differ on the primary outcome in the ITT analysis (
The prespecified analysis at 2 weeks showed no significant between-group differences (
Secondary Outcome: Endpoint MADRS Scores (Table 2)
Significant difference emerged between groups in the ITT (
Clinical Outcome Measures across the Study.
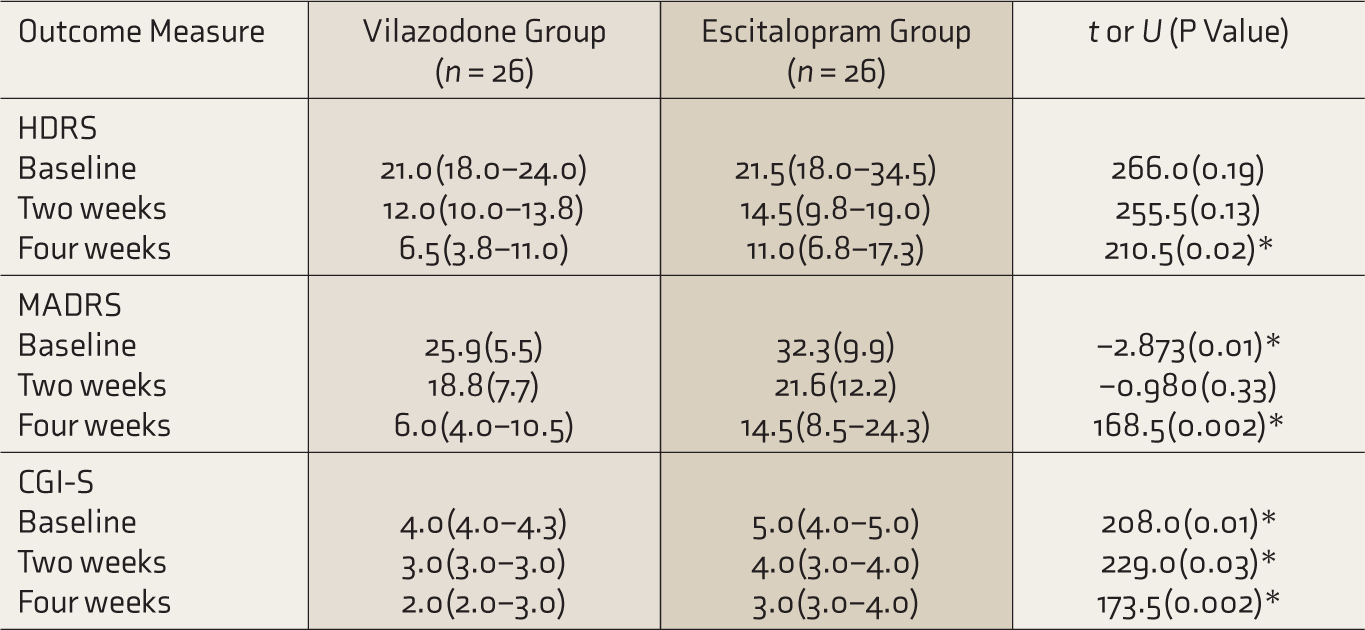
CGI-S: clinical severity of illness, HDRS: Hamilton Depression Rating Scale, MADRS: Montgomery–Asberg Depression Rating Scale. Values for the two groups are mean (standard deviation). Comparisons done using independent
Secondary Outcome: Endpoint Clinical Severity of Illness Scores (Table 2)
The vilazodone group had significantly lower endpoint clinical severity of illness scores in both the ITT (
Adverse Drug Reactions
Instances of diarrhea were significantly more in the vilazodone group (
At baseline, 17(32.7%) participants reported sexual problems. Five additional patients (all in the escitalopram group) reported sexual adverse effects during the trial; this difference in proportion was not statistically significant.
Regression Analyses
We performed the first regression analysis with the primary outcome (endpoint HDRS scores) as the dependent variable. Independent variables in the model were group assignment status and important ones that differed between groups at baseline: these included sex, baseline HDRS, baseline MADRS, and baseline CGI-S scores. Except for the grouping variable, we found that all the others had nonsignificant regression coefficients (Table 3); this implies that none of the baseline variables that distinguished the groups were significantly associated with the primary outcome when controlling for the effects of other variables in the model. When endpoint MADRS and CGI-S scores were regressed on the same set of independent variables, it was noted that the grouping variable continued to remain significant even after controlling for the other covariates in the model (Table 3).
Regression Analyses for Primary and Secondary Outcomes.
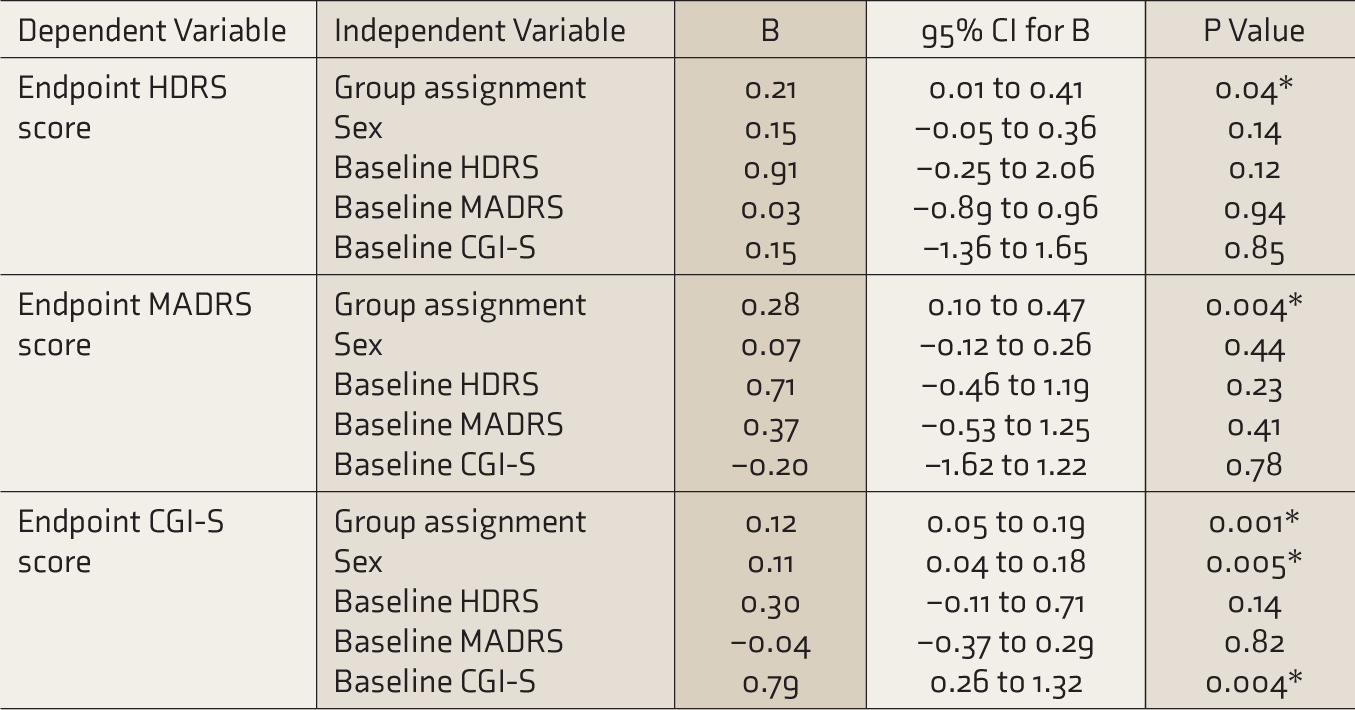
B: unstandardized coefficient, CGI-S: clinical severity of illness, CI: confidence intervals, HDRS: Hamilton Depression Rating Scale, MADRS: Montgomery–Asberg Depression Rating Scale. * Significant at P < 0.05.
In the subsequent multivariable analysis carried out with endpoint HDRS scores as the dependent variable and sex, marital status, baseline HDRS, baseline MADRS, and endpoint dose of clonazepam as independent variables, the regression coefficient for the dose of clonazepam was not significant, suggesting that clonazepam dose was not significantly associated with the primary outcome. Similar results were noted when endpoint MADRS and CGI-S scores were regressed on the same set of independent variables.
Discussion
The major finding of the study was that there were no significant differences in endpoint depression scores between subjects in the vilazodone arm and the escitalopram arm. However, among those who completed the study, those in the vilazodone arm experienced lower endpoint depression scores as well as clinical severity of illness. Previous head-to-head comparisons of vilazodone versus escitalopram have shown inconsistent results7–9; while some authors found no difference in efficacy for both agents, 7 others observed superiority for escitalopram 9 or vilazodone. 8 In an indirect comparison of vilazodone with several antidepressants, including escitalopram, all the studied antidepressants studied demonstrated similar efficacy 17 ; similar results were noted by investigators who compared vilazodone and escitalopram using a noninferiority design. 18
We did not find evidence for the early-onset hypothesis action of vilazodone. By directly stimulating the 5HT1A autoreceptors in the central nervous system in addition to augmenting synaptic serotonin (5HT) through Serotonin Reuptake Transporter inhibition, vilazodone, theoretically, may have a more rapid onset or more pronounced therapeutic action compared to conventional antidepressants.19, 20 So far, there is no human evidence of rapid onset or more robust antidepressant action of vilazodone compared to SSRI or Serotonin Norepinephrine Reuptake Inhibitors. 21 In the present study, we noted no differences between the two drugs on the primary outcome at 2 weeks. However, given that this secondary analysis was underpowered and given that the groups separated on both the secondary outcomes at the 2-week timepoint, our findings may represent a signal worth investigating further with a suitably powered study.
Sexual dysfunction, which was not significantly different between groups in our study, is one of the main concerns in managing depressive illness. Depression per se is associated with sexual dysfunction, and this may reduce the quality of life. 22 Simultaneously, sexual dysfunction could also be induced by antidepressants, which may predispose to poor treatment adherence. 23 In an earlier trial comparing vilazodone and citalopram, no statistically significant differences emerged in sexual functioning between groups though trends pointed to a greater improvement in sexual functioning in the vilazodone arm. 24 On the other hand, in a study from India, sexual functioning was noted to be better in those treated with vilazodone compared to escitalopram. 7 These differences are relevant because we compared vilazodone with a first-choice antidepressant drug with a well-known side effect of sexual dysfunction. However, compared to placebo, vilazodone may have a greater risk of sexual side effects such as decreased libido and orgasmic dysfunction. 25 There is a caveat here: SSRI-related sexual adverse effects are more commonly reported among males. 26 However, our sample was predominantly female, and this skewing was more evident in the escitalopram group. This may have impacted our observations, in addition to the lack of statistical power to detect differences in adverse effects. More research, focusing on adverse effects as a primary objective, is needed to clarify the relative risk of sexual dysfunction with vilazodone, compared to conventional antidepressants.
Weight gain is another important risk factor for suboptimal adherence to psychotropic agents in major mental illnesses. 27 In the present trial, no significant differences in weight gain were noted between groups. There are several reasons for weight gain among patients with depressive illness; it could be a symptom of depression itself or a side effect of the medication used to treat depression. 28 Weight gain with antidepressants is associated with negative long-term outcomes, including the risk of cardiovascular diseases. 29 Clinical evidence suggests that vilazodone is not associated with clinically significant weight changes. 16 However, in most of these studies, the comparator was a placebo. Consistent with our findings, a previous study noted no weight gain with vilazodone compared to escitalopram in depression. 7
Adverse reactions most associated with vilazodone are diarrhea, nausea, vomiting, and insomnia. Other less common adverse effects include dizziness, fatigue, dry mouth, decreased libido, and palpitations. 24 More than a third of our patients in the vilazodone group reported diarrhea, while just under one-third reported sexual side effects. No serious adverse events were noted in either treatment group.
Our study has limitations. The sample was underpowered to detect a medium or small effect size and the follow-up duration was short. We did not use a noninferiority design because of prior trials showing superiority of vilazodone over escitalopram. Further, we have not used any specific scale for assessing important adverse effects such as sexual functioning or medication adherence. Our study duration of 4 weeks is on the shorter end of the adequate trial period recommended to assess antidepressant efficacy.30, 31 However, this duration may match real-world practice where clinicians are unlikely to wait beyond 4 weeks to decide whether to persist with or discontinue the antidepressant, based on response. Finally, the baseline scores of primary and secondary outcome measures were significantly different between groups, which may indicate issues with the randomization process. A greater numerical difference in MADRS baseline scores, compared to HDRS scores, between groups may have been due to the difference in content and coverage of symptoms between the two.32, 33 However, none of these variables significantly influenced the primary outcome. In view of these limitations, our findings should be interpreted cautiously.
Methodological strengths of the study included its randomized design, assessment of efficacy using standard measures, and blinding of outcome raters thereby minimizing the chances of detection bias. Coprescription of medications that can potentially contribute to sexual dysfunction was avoided. Our study adds to the limited body of evidence providing data for head-to-head comparisons between vilazodone and escitalopram.
Conclusions
We found no differences between the vilazodone and escitalopram groups on the primary outcome of endpoint depression scores at the end of 4 weeks. However, among those who spontaneously complete the trial and take vilazodone as advised for 4 weeks, it is possible that vilazodone may outperform escitalopram on endpoint depression scores. We found no statistically significant differences between groups on the 2-week depression scores or proportion with ADRs. However, this may have been a type-II error as our analysis was limited by a lack of statistical power to detect smaller differences between groups, should they exist. Future trials comparing vilazodone and escitalopram should be adequately powered to detect smaller differences between groups and be carried out for a longer duration with a more rigorous assessment of the adverse effect burden.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study has not been funded. Being an investigator-initiated study, patients purchased medicines on their own, which was mentioned in the informed consent form.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
