Abstract
Background:
Among persons with schizophrenia (PwS), tobacco use is higher in comparison to the general population, contributing to greater morbidity and mortality. Pharmacological interventions combined with psychosocial interventions are effective in tobacco cessation. While the effectiveness of extant psychosocial interventions—when used in isolation—seems limited, developing better combinations of interventions could help treatment providers deliver tobacco cessation services to PwS at different stages of motivation to quit. We aimed to develop a personalized tobacco cessation intervention package (PTCIP) for PwS.
Methods:
The stage-based intervention package was developed through a systematic review of interventions for tobacco cessation, the authors’ clinical experience, and expert validation. The components of the intervention package, developed for PwS visiting the outpatient psychiatric department, were retained, removed, or added based on the content validity ratio (CVR).
Results:
The final components included brief advice, principles of motivational interviewing, psychoeducation, decisional balance matrix, positive reinforcement, offering various treatment options, personalized feedback using a smoking-risk calculator, and prediction of cardiovascular risk using the WHO package of essential noncommunicable disease (PEN). The delivery of the intervention package was refined based on pilot testing in eight participants.
Conclusion:
The tailored package was designed to be delivered by a mental health professional as a single comprehensive 40 min to 45 min face-to-face session, integrated with routine follow-up visits, followed by two telephonic conversations in the second and third week of the initial session. The package needs to be tested in a randomized controlled trial for its effectiveness.
Psycho social interventions are an important treatment option for tobacco cessation in persons with schizophrenia. Individualized interventions based on the stages of motivation to quit or cut-down tobacco use is necessary. Low-cost personalized feedback is an important ingredient of individualized intervention. The developed intervention needs to be tested in randomized controlled trials before it can be routinely used in the psychiatric out-patient setting.Key Messages:
Psychosocial interventions are an important treatment option for tobacco cessation in persons with schizophrenia; individualized interventions based on the stages of motivation to quit or cut-down tobacco-use is necessary; low-cost personalized feedback is an important ingredient of individualized intervention; the developed intervention needs to be tested in randomized controlled trials before it can be routinely used in the psychiatric outpatient setting.
Tobacco use is significantly higher amongst people with mental health conditions than the general population and is even higher in patients with schizophrenia (PwS), with prevalence estimated to be 58% to 88%.1, 2 People with severe mental illness die 10 to 20 years earlier than the general population, 3 with smoking being the major contributor. 4 Despite the desire to quit tobacco, PwS may continue to use it for perceived mental health benefits.5, 6 However, chronic tobacco use, in any form, worsens cognitive functions7–11 and performance-based functional capacities. 12
Interventions for tobacco cessation have focused on quit attempts. The long-term benefits of quitting include improved depression, anxiety, and stress; improved positive mood and quality of life 13 ; reduced readmissions; 14 and decreased need for higher doses of antipsychotics. 15 Evidence suggests that PwS can give up smoking with the help of pharmacological interventions such as varenicline, bupropion, and nicotine replacement therapy (NRT) either alone or in combination,16–19 or along with behavioral interventions such as cognitive-behavioral therapy (CBT), motivational interviewing (MI), and behavioral counseling.18, 20–22 A recent systematic review reported that at six months, the rate of smoking cessation among PwS using psychosocial interventions (11%–18%) was comparable to pharmacotherapy (12%–19%). 23
Psychosocial strategies have been considered to be important ingredients in addiction treatment. 24 Interventions such as stimulus control techniques, social skills training, relapse prevention training, psychoeducation, brief interventions such as 5As, and motivational techniques have been effective in settings that cater to persons with mental illness and tobacco use. 24 However, these intense psychosocial interventions are underused in the treatment of nicotine dependence among PwS 24 because of the belief that they are likely to be less successful at quitting and are not offered consistently across different levels of mental health services. 21 Moreover, the primacy attached to the symptoms of schizophrenia in psychiatric settings besides the use of tobacco as a reward or in token economies and its perceived role in social facilitation can result in tobacco use being condoned in mental health settings.
The “stages of change” or DiClemente’s transtheoretical model (TTM) by Prochaska et al. 25 include precontemplation, contemplation, preparation, action, and maintenance. 26 Cessation interventions tailored to the person’s stage of readiness to quit would help PwS move forward through the stages of motivation toward action and maintenance. 27 Given that stage-based specific interventions are effective in tobacco cessation,28–31 combinations of various psychosocial interventions are needed to overcome the challenges of quitting tobacco 32 among PwS.
This study aimed to develop a nonpharmacological personalized tobacco cessation intervention package for PwS attending the psychiatry outpatient department (OPD) of a tertiary care mental health setting for follow-up visits.
Materials and Methods
The study was approved by the Institute Ethics Committee. The personalized tobacco cessation intervention package (PTCIP) was developed between October 2018 and January 2019. The intervention package was developed as a part of a Ph.D. research and developed for use and testing in a randomized control trial (RCT; yet to be published) among persons (18–65 years) of any gender with a schizophrenia spectrum disorder. A systematic literature review was carried out using the following databases—PubMed, Cochrane, EBSCO, PsycINFO, ProQuest, and Google Scholar—for existing pharmacological, nonpharmacological, and combination interventions for tobacco reduction/cessation in patients with severe mental illness, schizophrenia in particular. 33 The PTCIP was based on the systematic review, and the components were initially selected or added based on the authors’ clinical experience (mean years ± SD = 16.5 ± 6.32) of working with patients with mental illness and substance use disorders, as the evidence was available only from high-income countries. Components were selected keeping in mind the local clinical realities in terms of treatment resources, the volume of patients, time available for face-to-face consultation for treatment during their routine psychiatric follow-up, and access to mobile phones. Components for providing personalized feedback were added considering that carbon monoxide monitoring was unavailable for this study and that urine cotinine analysis could be made available for a subset of patients only. The package developed was presented to 14 mental health experts (two psychiatrists, four addiction psychiatrists, two psychiatric social workers, two clinical psychologists, two psychiatric nurses, one epidemiologist, and one mental health educator) having at least five years of experience working with patients with severe mental illnesses and substance use disorders. Based on the expert validation and feedback and estimation of the Lawshe’s content validity ratio (CVR), the components were retained, removed, or modified (Table S1).
The experts were required to assess the components in the intervention package as “very essential,” “essential,” or “not essential.” The final intervention package was developed (Figure 1) to provide 40 min to 45 min of face-to-face intervention followed by two telephonic conversations in the second and third weeks from the initial intervention.
Process in the Development of the Final Intervention Package
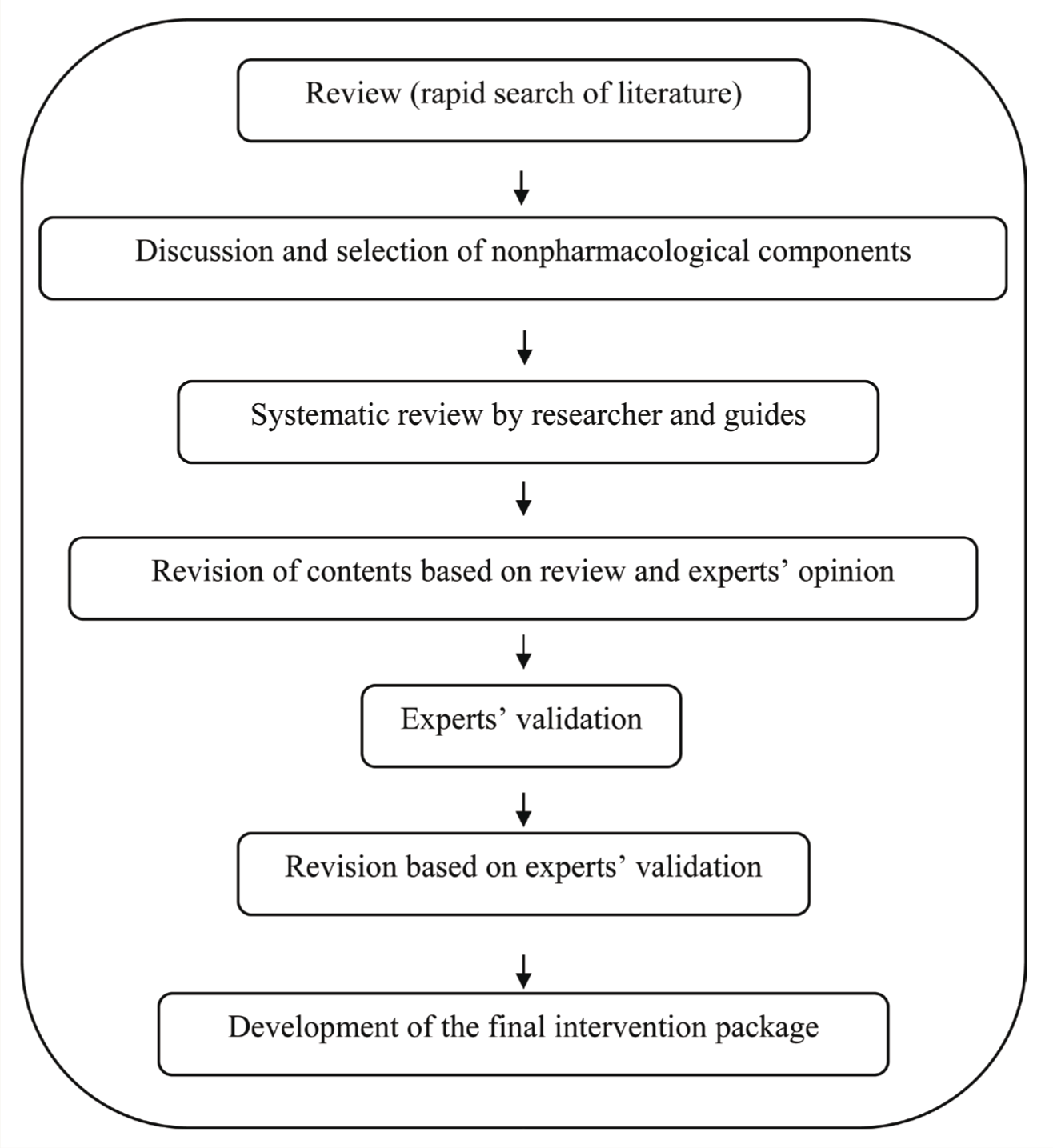
After obtaining written consent, the intervention package was pilot tested on eight patients diagnosed with schizophrenia spectrum disorders. The patients were asked about their intention to change tobacco use behavior and categorized based on the TTM model to deliver the interventions. The patients were randomly recruited from the regular out-patient follow-up patients using computer-generated random numbers. Pilot testing was done to assess the time required to deliver the face-to-face session of the intervention package, the ability of the participants to understand the intervention, and other practical aspects in a busy OPD, such as availability of space and privacy, internet facility to use smoking-risk calculator, and arranging regular psychiatric follow-up.
Results
Development of a Personalized Tobacco Cessation Intervention Package (PTCIP)
Systematic Review Summary
We have reported the findings of the systematic review elsewhere. 33 In summary, six RCTs (two nonpharmacological interventions, four combinations of nonpharmacological and pharmacological interventions compared with routine care) that measured reduction and cessation of tobacco use after a minimum follow-up period of six months were included in the systematic review. The review found that combinations of pharmacological and nonpharmacological treatments facilitated abstinence and reduced cigarette smoking more than nonpharmacological interventions used in isolation. The nonpharmacological interventions varied widely, and specific nonpharmacological management such as home visits and MI were more effective than others. 33
Nonpharmacological Components Included in the Intervention Package
Table S1 summarizes the selected nonpharmacological interventions from the review of literature, components removed with reasons after experts’ validation based on their clinical experience, practical issues and suitability of the settings, and the objective measures added to the package for immediate personalized feedback on tobacco users. The CVR for the components validated was 0.8 (each component ranged from 0.6 to 1.0) using the following formula: content validity ratio (CVR) = (Ne - N/2)/(N/2), Ne being the number of experts indicating an item as “essential” and N being the total number of experts. The final intervention package was tailored to the individual’s need during the face-to-face intervention.
Intervention Package Modification According to the Stage of Change
Tobacco cessation counseling based on the stage of change has previously been reported to be effective in helping patients to quit. 34 The complete intervention package tailored according to the stages of change has been provided in Table S2.
The final components in the PTCIP included:
Components of brief advice and 5As. Principles and techniques of MI such as empathy, developing discrepancy, acknowledging feelings, supporting self-efficacy, and optimism. General educational information on tobacco use and its harm by elicit-provide-elicit method (that included oral health issues, cancer in various organs, exacerbation of lung diseases, cardiovascular diseases, etc.), the importance of physical health and mental health, craving management, and relapse prevention. Psychoeducation about mental illness, antipsychotic medications and the need for increased dose of medication because of tobacco use, and decisional balance matrix. Positive reinforcement such as verbal reinforcement in “change talk,” “cut down or quitting” tobacco, and treatment adherence. Information that treatment was available at the tobacco cessation clinic of the institute and that pharmacological management options (bupropion, varenicline, NRT) were available. Personalized feedback based on comorbid-medical illness, laboratory values, quantifying and communicating the percentage of cardiovascular risk using the WHO package of essential non-communicable disease (PEN) visual chart, and “number of days lost” in life span because of smoking, using free online Smoking Risk Calculator on a computer or mobile phone. WHO Package of Essential Non-Communicable Disease (PEN): The World Health Organization/International Society of Hypertension (WHO/ISH) risk prediction chart is a tool that enables integrated risk assessment and risk prediction in non-Western populations. The risk prediction charts enable the total risk stratification approach for the management of cardiovascular disease (CVD) to be introduced in WHO regions where cohort data and resources are not readily available for the development of population-specific risk prediction charts. The risk of CVD in the next ten years is predicted using color coding, based on the percentage of risk calculated. Different charts are available for different regions.
35
Unique charts are available for people with and without serum total cholesterol value and with and without diabetes. The package is available in the public domain for free use. The information required to use the chart includes age, gender, systolic blood pressure, presence or absence of Type 2 diabetes mellitus, and availability or unavailability of total serum cholesterol level. The visual chart was not modified for the study. However, because the colors used in the WHO/ISH risk chart (green, yellow, orange, and red) to interpret the percentage of CVD risk were like the traffic light signal, we primed the participants by using a traffic signal color chart to assess the understanding of green, yellow, and red color signals, before using the WHO PEN chart.
Smoking risk calculator: The calculator requires information such as age; gender; ethnicity; approximate day, month, and year of initiating smoking; any quit attempt, and the number of cigarettes per day to calculate the number of “days lost” in the life span because of smoking. 36 Permission from “Med India” content team was obtained through e-mail to use the free online calculator.
Brief telephonic follow-up regarding advice to quit, challenges related to abstinence/reduction attempts, verbal reinforcement for cutting down or stopping tobacco use, and discussion about the reasons for the inability to cut down.
Practical Challenges
Eight males in different stages of motivation (precontemplation n = 2, contemplation n = 3, preparation n = 1, action n = 1, and maintenance n = 1) [mean age = 38.4 ± 6.26 years, mean duration of illness = 11.2 ± 6.65 years, mean duration of tobacco use = 13.7 ± 6.67 years, mean Fagerstrom test for nicotine dependence (FTND) score = 6.0 ± 1.33, mean tobacco craving questionnaire (TCQ) score = 61.7 ± 5.91, mean Clinical Global Impression–Severity (CGI-S) score = 1.6 ± 0.52, forms of tobacco use (smoker: smokeless users: dual users) = 4:2:2, mean number of cigarette/bidi smoked = 8.7 ± 1.63, mean number of smokeless sachet chewed = 1.75 ± 0.50] diagnosed with schizophrenia spectrum disorder with FTND scores of ≥3 and Global Clinical Improvement score <4 were recruited for the pilot. They were on treatment with varying doses of medications such as risperidone, chlorpromazine, olanzapine, and fluphenazine, based on individual needs (mean chlorpromazine equivalent doses = 531.2 ± 342.20). The crowd in the OPD limited the privacy of the patients, and completion of a regular follow-up visit would take 2 h to 4 h. Patients’ responses such as “not interested,” “not needed,” “not so important” for intervention, and disinterest from caregivers were observed. To address this, a separate room was made available to deliver the interventions, in discussion with the administration. The pilot experience necessitated the coupling of the delivery of the intervention with regular follow-up, keeping in view the time spent by the patients in the OPD. This would help develop a rapport during the initial follow-up and provide a single point of care for physical and mental health. While the package was designed in English, the package can be delivered by mental health professionals in local languages because of the simple, clear language used in the module. The contents of the telephone conversation were finalized after discussion with the senior authors after the pilot experience. The other practical challenge was the telephone being answered by the caregivers, who were then informed about the purpose of the telephonic conversation, which required additional time for the researcher.
Discussion
In this study, we have reported the development of a personalized, stage-based intervention package for tobacco cessation for PwS that involves a single face-to-face session and two telephonic follow-up sessions. As each stage of change is characterized by short- and long-term intentions and behaviors, 34 stage-based interventions were developed—for patients in the precontemplation stage, creating awareness about the benefits of quitting would be more appropriate; those in contemplation would require several cessation strategies along with interventions to address barriers and resistance to quit; action-oriented strategies would help patients in the preparation stage; and relapse prevention would be apt for patients in action or maintenance stages. 34 The stage-based interventions used in this study have been reported to be beneficial in tobacco users without mental illness 37–39 and formed the basis for use in this group. Issues distinct to PwS compared to general population tobacco users would include heavier use of tobacco, use of tobacco for self-medication, and the clinical features of schizophrenia such as negative symptoms and cognitive deficits, thereby necessitating a relatively intense face-to-face session coupled with routine follow-up and persistent efforts from the service provider, exemplified by strategies such as the use of teach-back, frequent summarization, and the telephonic follow-ups.
The package has been developed based on an extensive literature review, 33 expert validation, and considering the practical challenges in a busy OPD. It included unique components of personalized feedback for settings with limited resources and has the potential to be used in regular follow-up visits by a psychiatric nurse or any other mental health professional.
Besides their effectiveness in relapse prevention among the general population, short individual sessions and telephone-based support have been reported to be as effective as more intense, face-to-face, and long-term treatments for tobacco use 21 in persons with mental illness. 40 Telephonic counseling can further supplement the interventions. 41
Psychoeducation is essential to make the patients understand the need for tobacco cessation. According to the American Psychiatric Association (APA) treatment guidelines, psychoeducational interventions should be part of the standard therapy for PwS. 42 The Substance Abuse and Mental Health Services Administration has identified psychoeducation as an evidence-based practice that should be implemented in psychiatric settings. 43 Educational components proposed for tobacco cessation have been observed to be an integral element in preventing heavy use and encouraging tobacco cessation.44, 45 Elicit-provide-elicit method of education—used in this intervention—helps avoid the information that the patients already know, acknowledge their lived understanding and expertise, and seek permission respectfully to share additional information. 46
Brief interventions were included as components of the intervention package as they effectively cut down tobacco use and lead to cessation among people with schizophrenia or other psychosis 47 and the general population. Brief advice is believed to be the single most cost-effective way to help those who smoke, 48 and the other components such as MI and psychoeducation 49 used in the package have also been reported to be effective in the general population.48, 50, 51
The important guiding principles of MI 52 were included in the intervention package to help resolve patients’ ambivalence, thereby enhancing their motivation to change toward reducing/quitting tobacco use. Adaptation of MI and personalized feedback as an approach has been reported to increase quit attempts, strengthen the commitment to positive change behavior, and improve the therapeutic alliance with the therapist in persons with schizophrenia and other severe mental illnesses.20, 53, 54
During the pilot testing, two of the eight participants expressed a need for pharmacotherapy and requested tobacco cessation services from the addiction unit of the institute. They were referred to the tobacco cessation clinic. Patients were made aware of the different treatment options for tobacco cessation to allow them to avail all locally available treatment options, including pharmacotherapy, which probably might raise interest in patients motivated to seek treatment for tobacco cessation. Addiction specialists might be more comfortable in prescribing pharmacotherapy for tobacco cessation as compared to psychiatrists who have not received similar training and exposure and who handle a high volume of patients in psychiatry OPD with “diagnostic overshadowing”—treating the psychopathology associated with mental illness may assume priority over intervening to reduce the tobacco use. However, addiction specialists are not available in most centers. Therefore, for more PwS to access effective treatment options for their tobacco use behavior, 55 general adult psychiatrists will be required to increase their responsibility in modifying their management strategies to seriously include treatment for tobacco use 56 while providing treatment for the mental conditions.
Biochemical verification with breath carbon monoxide or cotinine levels for personalized feedback is not always available in routine clinical settings. The biomarkers are not a necessity to enter change talk. The novel components of the smoking-risk calculator and the WHO PEN package included in the intervention package would help provide immediate feedback. Personalized feedback could reinforce PwS to quit tobacco 5 and, as an add-on to standard treatment for tobacco cessation, has been reported to be effective in the general population also. 57 WHO PEN package and smoking risk calculators can be used amongst those who smoke and not amongst users of smokeless tobacco. In our setting, around 40% of patients with mental illness use more than one form of tobacco. 58 Therefore, the objective measures could be used for dual users. Visual charts that utilized the traffic signal colors, the use of a pen/pencil to draw the readiness ruler, the use of a decisional matrix to improve the motivation to quit tobacco, and the use of a computer or mobile phone to perform online calculations were novel to this package.
The PTCIP has been developed following the CONSORT 2010 updated guidelines, for sufficient details to be included in interventions, 59 and criteria for developing public health interventions (provided as online-only supplementary file).60, 61
Implications
The PTCIP requires to be tested in an RCT, comparing it with brief advice/treatment as usual and evaluated for its cost-effectiveness. Nonpharmacological interventions for tobacco cessation are less likely to be associated with adverse effects. If found effective across levels of care, the intervention could be recommended for use in routine clinical practice during follow-ups. Across the levels of care, the human resource for intervention delivery—for example, psychiatrist or psychiatric nurse—needs to be carefully chosen based on availability. The intervention can be integrated into the District Mental Health Program if found cost-effective.
Strengths and Limitations
Coproduction of interventions with all stakeholders involved will maximize the effectiveness of the interventions, leading to greater acceptability, practicality, evaluability, and uptake by the stakeholders and policymakers. 62 While the brevity of the content in the intervention package, a single face-to-face session with only two telephonic follow-ups, and tobacco cessation intervention integrated into a routine follow-up were advantages of the study, the interventions could have been further enhanced by the involvement of the stakeholders—patients and caregivers as experiential experts—in codesign or through Focus Group Discussions/Key Informant Interviews.
The intervention is likely to be challenging in PwS in the presence of cognitive deficits, formal thought disorder, disorganization, or negative symptoms. It may not be suitable for the actively symptomatic (CGI-severity score ≥4). The active engagement of caregivers in intervention delivery, at least in a subset of patients, may be necessary in Indian cultural settings. Issues that may arise in intervention fidelity across providers, inter-provider differences in delivery, or a qualitative assessment of specific components suitable for specific patient subtypes were not examined. The trials included in the systematic review were conducted in high-income and upper-middle-income countries. Thus, the suitability of these interventions for low- and middle-income countries (LAMICs) needs to be explored.
Nevertheless, given the higher prevalence of tobacco use among PwS, 51 and with higher nicotine dependence in them, 50 there is a need to develop such interventions to improve both physical and mental health. People in the preparation and action stages of quitting would require types of interventions different from those in the earlier stages of precontemplation or contemplation. 63 Stage-based tobacco cessation models 26 might be more effective and efficient than “one size fits all” interventions. 31
Conclusion
The intervention package was developed according to the individual’s stage of motivation to quit tobacco. The components in the package were based on a systematic review of literature and experts’ validation and were in keeping with the criteria for developing public health interventions. The pilot experience was used to refine the components of the package so that the intervention could be integrated seamlessly into the routine follow-up OPD care at a tertiary psychiatric hospital. The effectiveness of the developed psychosocial intervention package needs to be evaluated through RCTs.
Supplemental Material
Supplemental material for this article is available online.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Acknowledgements
We thank Dr Pratima Murthy, the Director, NIMHANS, and Dr Jagadisha Thirthalli, the Head of Psychiatric Rehabilitation Services, for their constant support in the development of the intervention package.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest concerning this article’s research, authorship, and publication.
Funding
The authors received no financial support for the research, authorship, and publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
