Abstract
Background:
Empathy plays a fundamental role in the context of psychotherapy. Mental health professionals (MHP) are required to express empathy on a daily basis. “Perspective taking” (cognitive empathy) and “emotional contagion” (affective empathy) are elements of empathy that are both innate and acquired. This study aimed to explore the underlying neural correlates of empathy using functional magnetic resonance imaging (fMRI).
Method:
A total of six healthy subjects from MHP and other professionals (OP) were recruited in a single-assessment study design. Subjects were screened using the Mini-International Neuropsychiatric Interview and the Standard Progressive Matrices. Behavioral measures such as cognitive and affective empathy, interpersonal reactivity, and emotional and social quotient were assessed. Perspective taking was examined with the fMRI face recognition task and the reading mind through eyes task. Emotional contagion was examined by the negative, positive, and pain emotions task. The fMRI was conducted in a 3T Siemens Magnetom Skyra scanner, using a block design paradigm.
Results:
Activation was noted in the following areas: cingulate and thalamus for positive and negative emotions, precuneus for negative emotion and pain, inferior parietal lobe for reading mind task and negative emotion, declive for reading mind and pain, and precuneus and frontal gyrus for reading mind task and facial recognition.
Conclusions:
There was no significant difference between MHP and OP groups on the behavioral measures. However, there were variations in cerebral and cerebellar activation in the functional imaging parameters.
Keywords
A comparison of mental health and other professionals show a variation in the functional networks of empathy. The differences in cerebral and cerebellar activation in MHPs may be attributed to the impact of training and practice on the dynamics of the brain organization.Key Message:
Acentury ago, Titchener introduced the word “empathy” based on the German concept of “Einfühlung” of Theodor Lipps. Empathy can be conceptualized as an attitude, a competency, or a behavior, which include authenticity, respect, communication skill, trust building, etc. 1 Empathy has multiple components, namely the capacity to recognize emotions in oneself and others via cues, such as facial expressions, speech, and behavior. The cognitive component of empathy, identified as theory of mind, involves the ability to understand the perspective of another person while maintaining the distinction between self and others. The affective element of empathy includes the capacity to share emotional states with others or the ability to experience similar emotions as others. 2 The individual variations in empathic ability are determined by genetic, biological, environmental, and socialization processes. 3 In a critical review of the empathy neuroscience literature, Zaki and Ochsner 4 identified three main facets of empathy: affect sharing, which is the tendency to share the emotions of others; “mentalizing,” which is the capacity to reason and draw inferences of others’ mental states; and prosocial concern or prosocial motivation, which is the desire to improve the well-being of others. They highlighted that frontal, temporal, and parietal regions are related to affect sharing and mentalizing.
As per the current opinion in affective neuroscience, empathy comprises dual systems: one that involves affect sharing or emotional empathy. It refers to the emotional reaction evoked automatically by observing another person’s state; it is an “affective–perceptual” form of empathy. 5 According to Preston and de Waal’s perception–action hypothesis (2002), perception of a behavior in another automatically activates one’s own representations for the behavior. The output from this “shared” representation automatically proceeds to motor areas of the brain, where responses are prepared and executed. This state-matching reaction is associated with the simulation theories, and motor empathy, imitation, and emotional contagion are linked to the mirror neuron system. The second system involves “cognitive–evaluative” empathy, which is a more deliberate and controlled system, where the empathic responses are determined by the extent of attention one allocates to the feelings of the target, together with the explicit conceptualization and appraisal of the emotions.6–10
Empathy is a cognitive, imaginative process, requiring effort and voluntary setting-aside of one’s own perspective, and it involves the ability to envision and comprehend the situation of another person. 11 Hence, empathy is influenced by contextual factors and appraisal processes, and it comprises a definite self-other differentiation. 12 Elliot et al. 13 indicated that empathy is a moderately strong predictor of therapy outcome. In 59 independent samples and 3599 clients, they found that client and observer perceptions of the therapists’ empathy predicted better outcomes than the therapists’ perceptions of empathic accuracy. Another study found that the clients’ and observers’ perceptions of the therapists’ understanding of the client’s internal experiences were related to outcome. These results highlight the importance of therapists making an effort to understand their clients by acknowledging their needs, so that the client may perceive empathy during therapy. 14
Being a core component of effective psychotherapy, empathy will benefit from further research. The ability to experience empathy is a fundamental skill. By studying the neural variability associated with empathy, one can further understand the differences in trait empathy, empathic abilities, or habitual tendencies. Our objectives were to evaluate the neural correlates of empathy using fMRI (Glass Brain View) and establish whether there is a difference between the neural correlates of mental health professionals (MHP) and other professionals (OP).
Materials and Methods
Six healthy subjects were recruited for the study after obtaining written informed consent. They formed two groups (MHP and OP). The MHP group consisted of clinical psychologists. The OP group comprised of engineers. Those with a history of medical, surgical, neurological, neurosurgical, or psychiatric illnesses were excluded. Those with electronic, metallic, or mechanical implants, such as stents, pacemakers, intra-uterine devices, cochlear implants, neurostimulators, or contact lenses, were also excluded. The age range of the participants was 25–28 years. The MHP group consisted of two females and one male, whereas the OP group consist of two males and one female. A single behavioral assessment and fMRI design was adopted. All subjects were screened using the Mini-International Neuropsychiatric Interview 15 and the Standard Progressive Matrices (SPM). 16 The behavioral questionnaires used for the study were Interpersonal Reactivity Index (IRI), 17 Questionnaire of Cognitive and Affective Empathy (QCAE), 18 Emotional Quotient (EQ), 19 Social Stories Questionnaire, 20 and Faux Pas Recognition Test. 21 All scales were administered individually. The subjects had to undergo an fMRI.
The Institute Ethics Committee approved the study.
Experimental Procedure
An empathic paradigm (using E-prime 1.1) was designed for the study. Empathy was examined using the following: 1. Face recognition task. 2. Reading mind through eyes task. 3. Negative emotions task. 4. Positive emotions task. 5. Pain task fMRI paradigm.
Pleasant images for the paradigms were selected from the International Affective Picture System (IAPS) 22 and the selected images ranged on a scale of 10, with 10 being most pleasant and 1 being most unpleasant. The researcher selected pictures based on both valence and arousal; those with values in the higher and lower ranges were chosen. The pleasant images were in the range of 6–8.2, with mean(±SD) valence being 7.25 (±0.60). Arousal was maintained in the range of 2.9–5.79 (mean 4.2±0.70). The range for the unpleasant images was 1.56–3.15 (mean 2.29±0.43), and arousal values were from 4 to 6.79 (5.54±0.71). Similarly, images depicting physical pain were chosen for the pain task. The block design paradigm was used to record blood-oxygen-level dependent signals with maximum efficiency. All the paradigms had four rest and active blocks and ten dynamics per block, with a total of 80 dynamics. For the face recognition and reading mind through eyes tasks, the subjects had to identify the appropriate emotions and use the respective buttons to choose the correct option out of two. The order of the presentation was predetermined, as listed above. Four paradigms were presented with intermediate MPRAGE (for the structural brain), and three more tasks were given. Subjects were asked to press buttons for two paradigms where the faces and eyes were shown. For the positive, negative, and pain images, the subjects were asked to empathize with the emotion shown in the picture. In the first paradigm, images were shown for the active block, followed by a rest. The rest block of all the IAPS paradigms displayed a crosshair. However, for the face and eyes task, clip art face and eyes were used as a rest task. The overall duration of the task was approximately 40 min, including the imaging time of the structural brain of each subject (Figure 1). Statistical analysis was carried out for the behavioral scales. Statistical parametric mapping using MATLAB was utilized for analyzing the imaging data. 23
Representative Sample of the Block Design Paradigm

fMRI Scanning
MRI scanning was conducted in a 3 Tesla Siemens Magnetom Skyra scanner. An anatomical scan was acquired with a T1 MPRAGE sequence. The Field of View (FoV) was 240 mm, slice thickness was 0.9 mm, the number of slices per slab was 176, and voxel size was 0.9×0.9×0.9 mm3. fMRI was acquired with an Echo Planner Imaging (EPI) sequence. The FOV was 192 mm, slice thickness was 4 mm, the number of slices obtained was 36, and voxel size was 3×3×4 mm3. The matrix was 64×64, Repetition Time (TR) was 4 s, and TE was 0.03 s. fMRI analysis was done using Statistical Parametric Mapping 8. The data from the first five dynamics were discarded. Pre-processing consisted of realignment, normalization, and smoothing. Realignment removed movement artifacts in fMRI time series. The translation and rotation did not exceed ±3 mm for any subject. The data were resliced after movement correction, with an estimation quality of 1 and separation of 4. Second-degree B-spline interpolation with registration to the first image was used. Normalization was done to the EPI.nii.1 template. Bounding box parameters were x of –85 to 85, y of –120 to 90, and z of –80 to 96. Second-degree B-spline interpolation was repeated. The source image was the mean image after realignment. The images to write included both mean and realignment images. The smoothing at full width at half maximum was 8 mm. Masking and wrapping were not used in any step. The first-level analysis was done with the general linear model with family-wise error. In order to carry out statistical analysis, the smoothed images were entered and analyzed using standard SPM. Second-level analysis was estimated, and a contrast was defined to get the results of the analysis.
Results
On the SPM, all the subjects fell into the above-average intelligence category. Means and standard deviations were calculated and nonparametric analyses were done for the behavioral measures (Table 1). Perspective taking (PT), fantasy (FS), empathic concern (EC), and personal distress (PD) were assessed on IRI. The mean PD (P = 0.72), mean FS (P = 0.72), mean EC (P = 0.88), and mean PT (P = 0.42) of the two groups did not show any statistically significant difference (t-Test). On QCAE, the groups had comparable means for cognitive empathy (P = 0.09) and affective empathy (P = 0.59). There was no statistical difference between the groups for perspective taking, online simulation (cognitive empathy), emotion contagion, or proximal and peripheral responsivity (affective empathy). The EQ scale assessed sensitivity (P = 0.14), maturity (P = 0.26), competence (P = 0.27), and total EQ (P = 0.5); there was no significant statistical difference between the groups. Both groups had a high social quotient as assessed by social story questions. The Faux Pas test demonstrated an adequate theory of mind in both groups.
Mean and SD on Interpersonal Reactivity Index, Questionnaire of Cognitive and Affective Empathy, and Emotional Quotient
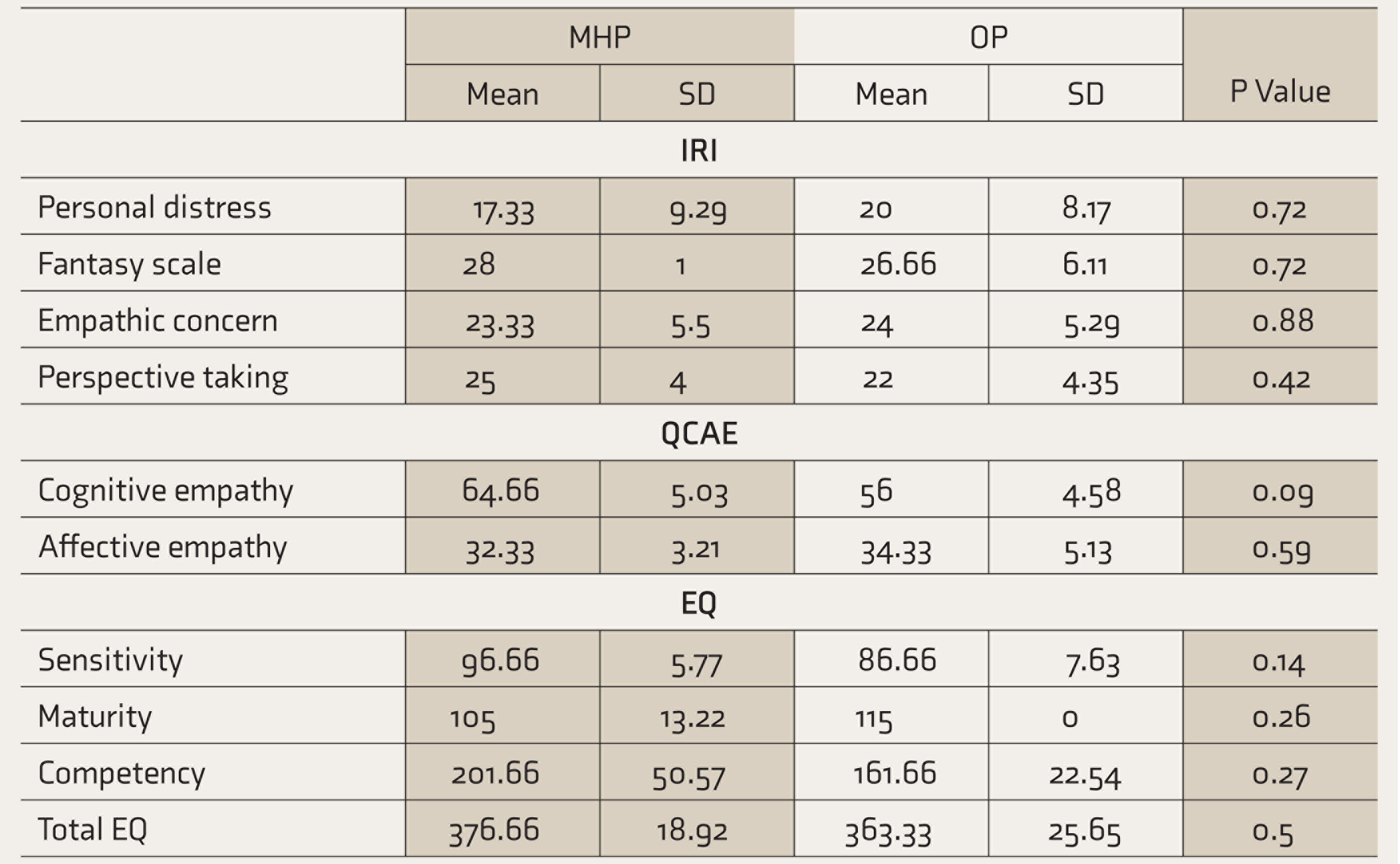
IRI: Interpersonal Reactivity Index, QCAE: Questionnaire of Cognitive and Affective Empathy, EQ: emotional quotient, MHP: mental health professionals, OP: other professionals.
Glass Brain View of Empathy
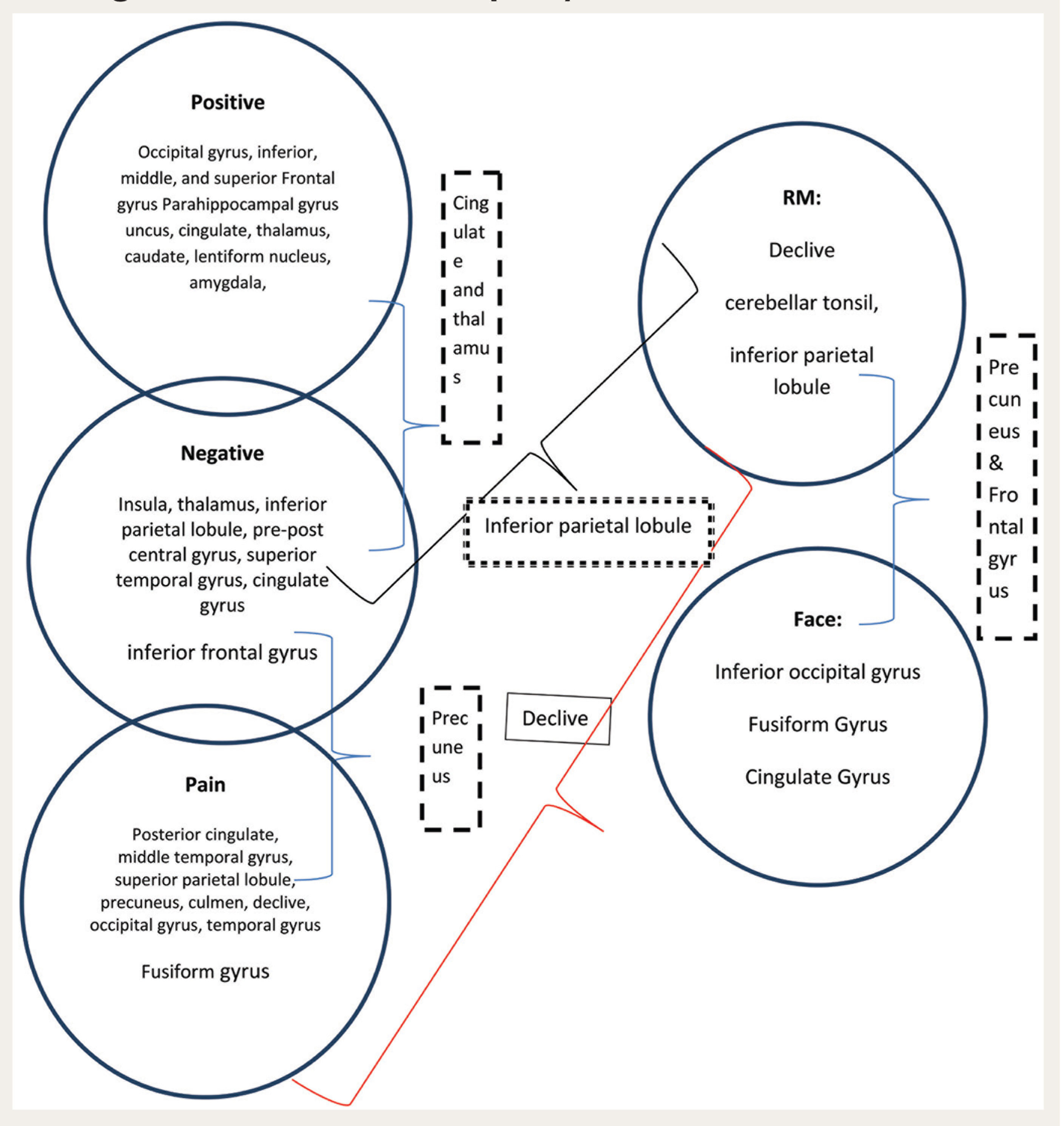
Cognitive empathy or perspective taking was assessed using the recognition and identification of facial expression and the reading mind through eyes tasks. The facial expression task activated bilateral occipital gyrus, fusiform gyrus along with culmen, and bilateral precentral and medial frontal gyrus—Brodmann’s areas (BA) 18, 19, 31, 4, and 20. When compared to the OP group, the MHP group had greater activity in the frontal lobe, precentral gyrus, and middle frontal gyrus (BA; 4, 6, and 9). Bilateral inferior parietal lobe was also activated (BA 7 and 40). The anterior and posterior lobes of cerebellum, such as declive and culmen, were found to be active for the reading mind through eyes task for the MHP group when compared to the OP group (Figures 1 and S1).
Affective Empathy
Emotional contagion was assessed through negative emotions task, positive emotions task, and pain task. Activations of inferior frontal gyrus (BA 37), precentral gyrus (BA 44), supramarginal gyrus (BA 40), and superior temporal gyrus (BA 13) were greater in the MHP group. Subcortical, anterior, and posterior cerebellar lobe (pyramis, declive, and culmen of the vermis), thalamus, insula, and putamen (lentiform nucleus) were activated in the negative emotions task. Positive emotions task activated bilateral occipital gyrus (BA 17, 18, and 19), limbic lobe—uncus, anterior and posterior cingulate, parahippocampal gyrus and amygdala (BA; 36, 20, 32, 28, and 30), frontal lobe—inferior, medial, and inferior gyrus (BA 6, 8, 9, and 47), bilateral temporal lobe—superior, medial, and fusiform gyrus (BA 38, 21, and 37), and sublobar areas—thalamus, caudate, and posterior declive. Pain stimuli activated the superior parietal lobule (BA 7), fusiform gyrus (BA 20), medial temporal gyrus (39), and the anterior and posterior lobes of cerebellum (declive and culmen) in the MHP group when compared to OPs (Figures S2, S3, and S4).
Discussion
Empathy is a concept that is multidimensional in nature. The objective of this study was to determine the neural correlates of empathy in Mental MHP and OP. The subjects were administered with behavioral measures and fMRI (Glass Brain View). There was no significant difference between both groups on the measures of interpersonal reactivity, cognitive and affective empathy, or EQ. In the fMRI paradigm, the first task of cognitive empathy involved the facial recognition and mind reading task, in which the subjects were asked to watch the screen and identify the emotions expressed through faces/eyes. The underlying neural network for cognitive empathy consists of the ventromedial prefrontal cortex, temporoparietal junction, and the medial temporal lobe.
In our study, when compared to the OP group, the MHP group had greater activations in some regions. Facial expression task activated bilateral occipital and frontal lobes. The nature of the activation suggests that in facial recognition, the discrimination of emotions appears to be at a perceptual level. There was evidence of activation of the cerebellum and bilateral frontal and parietal lobes for the reading mind task. Research has shown that the cerebellum does have a role in cognitive operations. 24 The frontoparietal loops are well known for processing information related to many cognitive domains, behavior, and emotion, including empathy and theory of mind. The results indicate the possibility of a cognitive loop over an emotional loop in the processing of information. Reading mind tasks shared the component of inferior parietal lobule and declive with negative emotions, suggesting that empathy is a shared representative of both affect and cognition. 25
The tasks that required empathizing with the stimulus (positive/negative/pain) showed the following: frontotemporal lobe, along with posterior and sublobar structures, was activated for the negative emotion task, whereas the positive emotions task activated the bilateral frontoparietal and temporo-occipital lobes. The positive emotions also elicited amygdala activation. The amygdala is not only associated with the intensity detection of stimuli, in addition, it is responsive to novelty and prediction error. Moreover, it has the ability to suspend on-going mental activity, and re-direct attention to stimuli, thus promoting appropriate motivational actions. The amygdala is associated with both unpleasant and pleasant emotions, and responds to fear as well as reward-related stimuli.26–28 The MHP were sensitive to both stimuli and affect.
The negative emotions suggested the involvement of the insula. In one study, the anterior insula was found to be associated with interpretation of stimuli in threat appraisal. Lesions to the insula can cause deficits in the perception of others pain. Increased activity in the anterior insula and frontal cortex results in increased empathy for pain. The insula is known to play a part in both affective–perceptual and cognitive–evaluative forms of empathy. 29 In the negative task, the insula could have played the role of detecting threat appraisal and in empathizing with pain.
The pain task activated the parietal lobule, temporal gyrus, and anterior and posterior cerebellar lobes. The pain stimuli did not elicit activation in the frontal lobe. The mid-anterior cingulate cortex is linked to the affective and motivational aspects of first-hand pain. It may also decrease the motivational significance of others’ pain. However, the narrative of emotional pain differs. When there is emotional suffering (loss/illness), it yields activity in cortical regions, bilateral and temporoparietal junctions, precuneus, medial prefrontal cortex, and amygdala, representing both cognitive states and mentalization.30–32 The MHP may have used more of the emotional pain modality network versus the physical pain matrix. The temporoparietal junction influences emotional and cognitive empathy. According to theorists, the temporoparietal networks are a part of the social cognition network, which are responsible for the ability to empathize and mentalize, and they are independent and directly impact behavioral responses. For example, empathizing can interfere with mentalizing in response to a strong negative emotion.33–34 Similarly, in this study, positive, negative, and pain stimuli have evoked activation in cortical and subcortical structures. Though these systems are independent of one another, it appears that empathic responses still consist of both components to a certain degree. There were common areas activated for both perspective taking and emotional contagion. Positive and negative emotions have elicited cingulate and thalamus activation. Negative emotions and pain elicited precuneus. Reading mind and negative emotions have the inferior parietal lobule in common. Reading mind and pain have the declive in common.
In view of the small sample size, one must exercise caution while generalizing the results. However, from an empathy building perspective, the results indicate no significant difference in behavioral measures of the MHP group with the OP group. The possibility of type II error cannot be ruled out due to small sample size. On the other hand, findings from the functional networks differ. On the fMRI, there was a significant difference between the groups for cerebral and cerebellar activations, which could imply that empathy maybe a developmental trait, which gets enhanced with training and practice. Empathy is a multifaceted skill, which enables individuals to comprehend the affective states of others and culminates in pro- social and altruistic behavioral responses. Empathy is determined by the cognitive, emotional, behavioral, and moral capacities. The model of empathy involves a bottom-up processing with respect to affect sharing and top-down processing concerning motivation, intentions, and self-regulation that influence the extent of an empathic experience.35–36 The neurodevelopmental perspective of empathy suggests that affective empathy develops earlier than cognitive empathy. Affective responsiveness tends to be automatic and employs mimicry and somato- sensorimotor resonance between self and others. In the cognitive component, emotional understanding in relation to oneself and representations of the other are relied on. This study reflects the interface of nature and nurture, in the neurodevelopmental framework of socio-emotional functions (Figure 2).
Neural Activation for MHP Compared to OPs Within and Between the Cognitive and Affective Empathy Tasks
The limitations include small sample and convenient sampling method, consisting of only clinical psychologists and engineers as part of the mental health and other group. The statistical analysis carried out is not robust; the region of interest analysis of the imaging data would have strengthened the study. In addition, static pictures were used for eliciting cognitive and affective empathy, and the tasks were chosen from western data. Empathizing with a static picture, without eliciting any feedback during the imaging, is a limitation. However, it has to be done to avoid movement during the scanning. The study focuses on components of empathy (perspective taking and emotional contagion) that play a crucial role in the psychotherapy process and outcome. By looking at the empathy of MHP and OP through the Glass Brain View, we found that there are differences in the underlying neural processes of empathy.
Conclusions
The aim was to study the neural correlates of the cognitive and affective components of empathy in MHP and OP, using fMRI. The ability to demonstrate empathy toward our fellow species is an ability unique to the repertoire of human social behavior. The results indicate the possibility of a cognitive loop over the emotional loop in perspective taking. For emotional contagion, the analysis indicates that positive emotions predominately utilized cortical and subcortical areas, whereas negative emotions and pain shared networks with cognitive empathy of the reading mind task. The reading mind task seems to incorporate elements of processing both negative emotions and pain, whereas positive emotions have activated unique patterns. On comparing the MHP and OP groups, there was no significant statistical difference between the groups on behavioral data, whereas on fMRI, there was a statistical difference between the groups for cerebral and cerebellar activations of brain areas. However, in view of the small sample, findings must be generalized with caution.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
