Abstract
Background:
Perinatal depression (PD) has important implications for maternal and infant well-being but largely goes undetected. There is a need to develop low-intensity psychosocial interventions applicable to obstetric health care facilities.
Objective:
To assess the effectiveness of a brief psychological intervention for mild to moderate PD delivered by a nurse as compared to treatment-as-usual (TAU).
Methods:
This study is a randomized, open-label, parallel-group, multicentric trial being conducted in four sites of India. A total of 816 pregnant women with mild to moderately severe depression (Patient Health Questionaire-9 score of 5–19) are being assessed for the effectiveness of the intervention. Participants are randomly allocated to two groups of trial intervention (psychological intervention given by nurse) and TAU. The primary outcome is to compare the proportion of women reporting improvement across both groups. Participants are serially followed-up in each trimester and at 6, 10, 14 weeks, and 6 months postpartum. Secondary outcomes include pregnancy outcomes, feeding practices, physical growth, and immunization status of the infants.
Conclusion:
It is a prospective pregnancy birth cohort with a robust design and long-term follow-up. This is one of the largest studies utilizing non-specialist health workers for the screening and management of PD. This study also holds promise to cast light on the course and outcome of depression during pregnancy in different parts of India. It is envisaged to help in developing a sound screening and referral-based protocol for obstetric settings.
This is the study protocol of a multicentric study whose primary objective is to assess whether a brief psychological intervention for mild to moderately severe PD delivered by nurses be effective when compared to treatment-as-usual. The secondary objective is to identify the sociodemographic, obstetric, and pregnancy outcome predictors associated with mild to moderately severe depression.Key Messages:
Depression is the most common cause of disability worldwide, with over 300 million people living with depression, an increase of more than 18% between 2005 and 2015. 1 Pregnancy and transition to parenthood are associated with significant biological, psychological, and social changes. Of this, the perinatal period that commences at the time of conception, continues throughout the pregnancy and extends till one year after delivery is considered as a highly vulnerable phase. 2 One in five women are reported to have depression during this period, known as, perinatal depression (PD). 3 PD has emerged as a cause of global concern. 4 The onset of PD usually begins in the antenatal period.5-7 If unaddressed during the antenatal period, this could result in postnatal depression.8-10 PD not only increases maternal morbidity but also has deleterious effects on fetal outcome. 11
Early identification and prompt management is the key to reduce morbidity associated with PD. To achieve this, the American Congress of Obstetricians and Gynecologists (ACOG) recommends that all obstetric care providers carry out a comprehensive assessment of the pregnant woman’s mood and emotional well-being, including screening for depression and anxiety using a validated instrument, during antenatal and postnatal visits. This needs to be linked with appropriate referral care. 12 Further, the World Health Assembly (WHA) recommended the incorporation of maternal mental health as an integral component of maternal and child health care. 13
In recent years, there has been an increased focus on the development of feasible and effective psychological interventions for PD.14-16 An ongoing study in Italy aims to develop and assess the efficacy of a psychological intervention for PD. 14 The United States Preventive Task Force reported that psychological interventions are effective in reducing PD through a systematic review. 17 A cluster randomized controlled trial (RCT) conducted among Indian rural women reported that a participatory intervention with a women’s group reduced maternal depression by 57%. 18 Another RCT carried out in South India found that a community-based depression intervention program implemented through the existing health system was effective in managing women with depression in primary care. 19 There are a few other studies that have attempted to assess the integration of perinatal mental health initiatives within existing maternal health settings.20,21
However, despite the existing evidence and recommendations, there is a high treatment gap of 76%–85% for mental health disorders in developing countries. 22 In India alone, the treatment gap for any mental health disorder is as high as 83%. Reasons cited for such a huge gap include the dearth of mental health professionals (0.2 psychiatrists per 10,000 population against the ideal of 1 per 10,000) and inequitable distribution of available trained health professionals. 23
To address this gap, task-shifting with the involvement of non-specialist health workers (NSHWs) becomes imperative.24,25 This group may include general physicians, nursing professionals as well as personnel like the accredited social health activists (ASHAs) and the auxiliary nurse midwives (ANMs). Further, the World Health Organization (WHO) stressed the need for developing a low-intensity psychological intervention. This refers to an intervention being delivered by a less intense level of specialist human resources, that is, utilizing NSHWs for health care delivery. 26
There have been no systematic assessments of the effectiveness of these low intensity psychosocial interventions delivered by NSHWs across various levels of health care in India. It is important to assess the effectiveness of such interventions in different health care settings and challenges of implementation. In this context, the present study, funded by the Indian Council of Medical Research (ICMR) Task Force on Capacity Building for National Mental Health Programme (NMHP), is being conducted to assess whether a brief psychological intervention delivered by the nurse is effective in the treatment of depression in pregnancy as compared to treatment-as-usual (TAU), which includes assessment and management for PD by psychiatrists using a common protocol. 27 It is hypothesized that this intervention will not be inferior to TAU.
Material and Methods
Study Design
This study is an RCT that compares a brief psychological intervention for PD with TAU.
Study Duration
The study is ongoing at the time of this report and is being carried out for three years. The study timeline is depicted in Table 1.
Study Setting
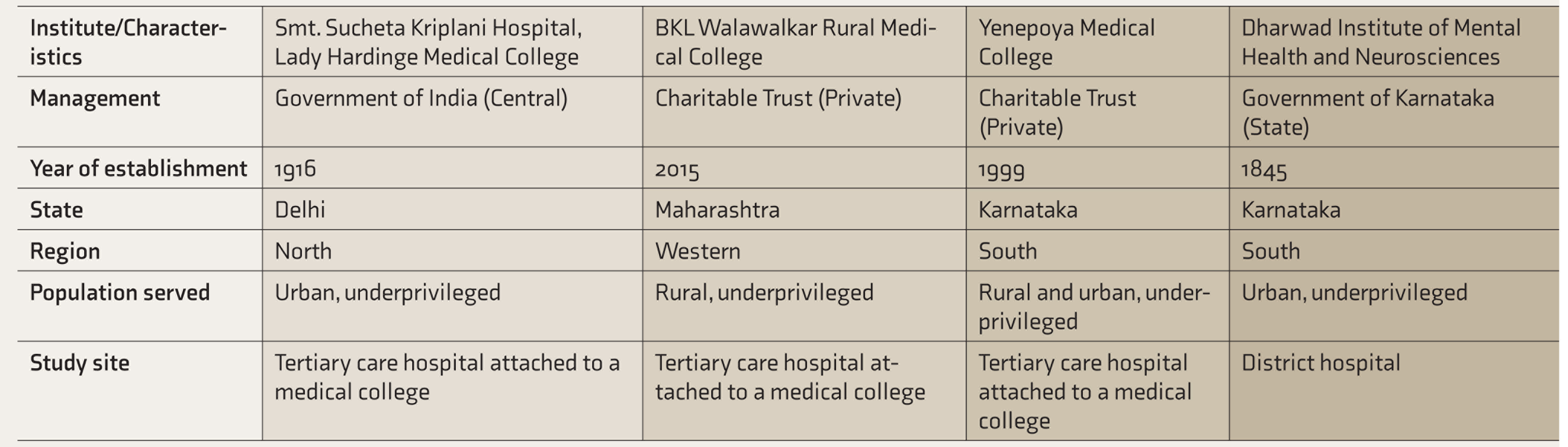
This study is being conducted in four health care facilities across India: (1) Smt. Sucheta Kriplani Hospital, attached to Lady Hardinge Medical College, New Delhi in north India, (2) BKL Walawalkar Rural Medical College, Sawarde, Maharashtra state, western India, and two sites in the southern state of Karnataka, (3) Yenepoya Medical College in Mangaluru, and (4) District Hospital, Dharwad. These sites were selected considering the geographical representation and levels of health care provided. The first three sites mentioned above are tertiary care teaching hospitals attached to Medical Colleges, while the fourth site is a secondary care district hospital. The characteristics of the four study sites are described in Table 2.
Sample Size
To be able to detect a 20% difference in the depression scores between the intervention and control group with 80% power using a two-sided test with an alpha (α) value of 0.05, a total of 170 pregnant women with mild depression are required. After considering an attrition of 30% (design effect of 0.5), the sample size is calculated to be 204. 28 Thus, the sample size is calculated to be 204 for each site, so 816 is the total sample size for all the four sites. Thus, 102 women each in both the experimental and control groups are being recruited in every study site.
Complete enumeration is being done to screen all pregnant women visiting the antenatal OPD. Thereafter, pregnant women with a PHQ-9 score of 5–19 are being invited to participate in the trial, until the desired sample size of 204 pregnant women is reached.
Participants
Participants must satisfy all of the following inclusion criteria to be considered for this study: (1) pregnant women with a gestational age of ≤20 weeks (first trimester/early second trimester) attending the OPD of the four study sites for antenatal care, (2) those with a score of 5–19 on Patient Health Questionnaire-9 (PHQ-9) (mild to moderately severe depression), and (3) intending to continue antenatal care, undergo delivery at the study site and post-delivery, willingness to visit the study site for postnatal care and immunization of the infant.
Timeline of the Study
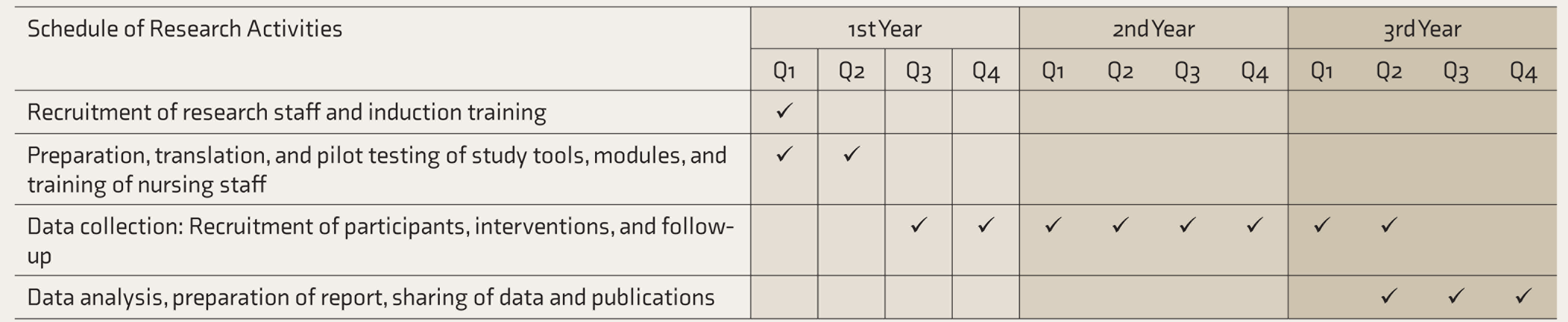
Q: quarters = three months.
Characteristics of the Four Study Sites
The exclusion criteria are as follows: (1) unmarried women, (2) history suggestive of assisted conception, (3) history suggestive of intellectual disability, (4) history suggestive of already being on psychiatric treatment for any mental illness, and (5) those expressing self-harm ideation or having a score of ≥20 on PHQ-9 (severe depression).
Brief Psychological Intervention (BIND-P Intervention)
An assessment of mental health needs and help-seeking for PD was conducted. We also gained insights from health care providers, particularly physicians and nurses, about their experiences of providing care for PD.13,29 We brainstormed and prepared the first draft of the intervention after an extensive literature review on various aspects of PD like systematic reviews on clinical and psychosocial presentations, epidemiological studies, existing interventions and models, potential challenges in implementations and strategies. We sought inputs from research experts in a capacity building workshop and fine-tuned the intervention. 30 The content was then validated by obstetricians, pediatricians, psychiatrists, and public health experts across India. Thereafter, we arrived at a consensus and finalized the intervention. The intervention was developed in the English language, and then translated to three languages (Marathi, Hindi, and Kannada).
The BIND-P intervention is primarily conceptualized as a health care worker based psychological intervention comprising of three brief sessions of 15–20 minutes duration sessions delivered once in two weeks. The first session includes psychoeducation with a focus on depression and it's implications in pregnancy and measures to alleviate the same. During this session, initially, the nurse introduces herself and builds a rapport with the participant. Thereafter, the nurse provides more details about mental health, depression, and the core therapeutic component for the three sessions. Two weeks later, the second session that includes relaxation and breathing exercises and visual imagery techniques is being delivered. The second session begins with a quick recap of the contents of the first session, after which, the nurse demonstrates certain brief and simple progressive muscle relaxation exercises. This is followed by demonstration of simple breathing techniques and visual guided imagery. The third session carried out two weeks after the second session. This session focuses on health promotion activities like nutrition, exercise, social support, sleep hygiene, and training for thinking healthy. The self-explanatory diet, exercise, and sleep charts will be provided to the participants for self-monitoring. All the three sessions conclude with a summary of the contents delivered.
The BIND-P intervention is brief, pragmatic, generalizable, and culturally grounded. It includes integral components of the WHO Thinking Healthy Program like psychoeducation, sleep, nutrition, and exercises. 31 However, the BIND-P intervention has been developed considering factors like the diverse Indian health care setting, ease of implementation, long-term sustainability, and improvement of the referral system for specialist care. On the contrary, the WHO Thinking Healthy Program is an evidence-based,non-specialist-delivered depression intervention.
Treatment as Usual
The control group were assessed and treated by a psychiatrist independent of the research team. A TAU protocol for this arm was developed after reviewing the recent guidelines published in 12 countries and the Indian Psychiatric Society (IPS) guidelines for the management of depression in pregnancy.32,33 Further, we also reviewed the guidelines provided by Kazdin et al. while drafting the TAU protocol. 27 The TAU protocol was kept uniform across all four sites. However, the psychiatrists may use the drugs or therapy based on their experiences, as and when required.
Study Tools
The following validated tools will be used: (1) a semi-structured pro forma to capture the study variables, (2) Modified BG Prasad Scale, 2016 and Modified Kuppuswamy Scale for socioeconomic status assessment,34,35 (3) Patient Health Questionnaire-2 (PHQ-2) for depression screening and Patient Health Questionnaire-9 (PHQ-9) for rating the severity of depression in both groups,36,37 and (4) diagnostic and statistical manual 5th edition (DSM 5) for diagnosis of depression by a psychiatrist in the TAU group. 38
Recruitment of Staff and Training
In each site, two Junior Research Fellows (JRFs), who are qualified psychologists or psychiatric social workers, and one staff nurse or two ANMs were recruited. The JRFs and the staff nurse/ANMs underwent induction training by the PI at each site for participant enrollment and data collection. The PI and the other members of the study team, which included experts from psychiatry, obstetrics and gynecology, and community medicine trained the staff nurses/ANMs at each site, through a three-day workshop. To ensure uniformity of contents of the training program across the four sites, a training manual was developed by the study team in English and translated to Hindi, Marathi, and Kannada. All four sites adhered to the training manual while conducting the training program for nurses. The training included didactic lectures, case-based learning, demonstrations, and assignments. Emphasis was given to hands-on training. The topics covered were rapport building, communication skills, screening for PD, BIND-P intervention, referral, and follow-up assessment. Monthly reorientation sessions are also being conducted. 39 Additionally, we have also trained registered staff nurses working in antenatal or postnatal OPD of all the four sites to deliver the intervention. Reorientation sessions to these staff nurses are also being held every quarter.
Data Collection Procedure
Participant Recruitment
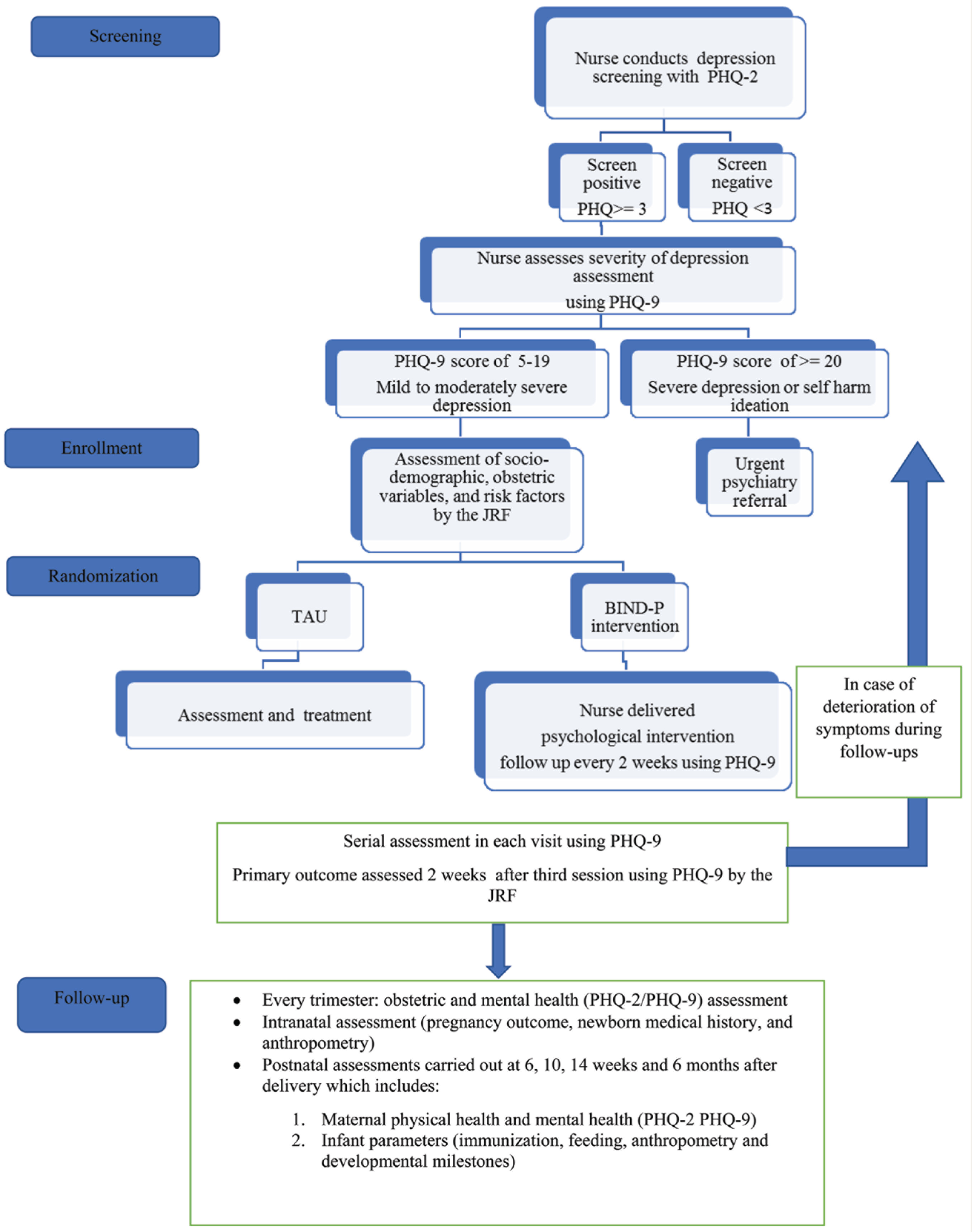
The internationally accepted and validated PHQ is used as the screening tool for PD. 40 After the JRFs obtain consent for screening, the staff nurse/ANMs screen the pregnant women with PHQ-2. It consists of two items that enquire about the frequency of the symptoms of depressed mood and anhedonia, scoring each as 0 (not at all) to 3 (nearly every day). The purpose of PHQ-2 is for initial screening as a “first step” approach. 41 A score of ≥3 is considered as the cut-off for risk of PD and such women are then further evaluated with PHQ-9 by the nurse/ANMs. It has nine questions with a score from 0 to 3 for each question (maximum score of 27). A threshold score of 5–9 is regarded as mild depression, a score of 10–15 is regarded as moderate depression, 15–19 signifies moderately severe depression, and 20 or higher severe major depression. 42 Those with a PHQ-9 score of 5–19, that is, mild to moderately severe PD, which includes (1) minor depression (PHQ-9 scores ranging from 5 to 9), (2) dysthymia and major depression-mild (PHQ-9 scores ranging from 10 to 14), and (3) major depression, moderate (PHQ-9 scores ranging from 15 to 19) are invited to participate in the trial with another written informed consent obtained by the JRFs. Recruited participants are further interviewed with a semistructured and pretested pro forma by the JRFs. The pro forma gathered socio-demographic variables, marital history, key obstetric variables like gravidity, complications in the present, and past pregnancies, and modes of past deliveries.
Randomization
Eligible and consenting participants are allocated to the intervention or the control groups using a computer-generated random number sequence by the members of the research team/JRFs. Block randomization using random permuted blocks of different sizes, where the size of the next block is randomly chosen from the available block sizes is followed. To ensure that all participants have an equal chance of being in the intervention or control group, we selected a 1:1 allocation ratio. 43 As the research staff were actively involved in the screening and recruitment process, it was not possible to mask the information about the allocation of the trial participants into the intervention and the control groups.
Delivery of BIND-P Intervention, TAU, and Outcome Assessment
Both the intervention and the control groups receive routine antenatal care. The intervention group has been receiving the BIND-P intervention delivered by the trained staff nurses/ANMs. The intervention composes of three sessions delivered fortnightly at an interval of two weeks. The first session is being delivered at the time of recruitment, followed by the second session, which is delivered two weeks after the first session. The third session is delivered two weeks after the second session. Participants allocated to the TAU group were referred for assessment and treatment of PD by psychiatrists independent of the research team. The primary outcome assessment for both the arms is being carried out using PHQ-9 at two weeks after the third and final session of BIND-P intervention/TAU is delivered.
Follow-up
All the trial participants are being followed up in the antenatal OPD of the study sites once in every trimester during their routine antenatal visits. Later, the participants are being follow-up at 6, 10, 14 weeks, and 6 months post-delivery, during the infant’s scheduled immunization visits. Timely telephonic reminders are given to ensure that the participants adhere to the follow-up schedule and to minimize attrition. Any participant missing three consecutive follow-up visits is considered as a non-respondent.
Each of these follow-up visits include an interview with PHQ-9. Additionally, semi-structured follow-up pro formas tailored for each trimester of pregnancy and post-delivery are being used to collect key information related to maternal and infant outcomes, as and when applicable. At any stage during the follow-up visits, a participant having signs or symptoms of major depression-severe with a PHQ-9 score of ≥20 is referred for psychiatric evaluation.
Outcome Measures
Data on socio-demographic variables, obstetric profile, and PHQ-9 scores were taken at the baseline at the time of recruitment and at different intervals of the peripartum period (each trimester, at the time of delivery, 6, 10, 14 weeks, and 6 months post-delivery).
The primary outcome refers to the proportion of participants in the intervention group reporting improvement in depression as defined by a 20% reduction in PHQ-9 scores following three sessions of the intervention when compared to the TAU group.
The secondary outcomes include the following:
Association between socio -demographic variables and depressive symptoms, which includes maternal age, education, family income, occupation, and socioeconomic status, which will be captured at recruitment. Association between key obstetric variables and depressive symptoms, which includes past obstetric history, past pregnancy details, medical comorbidities, and complications in the current pregnancy, which will be captured once in every trimester. Maternal-related outcomes like mode of delivery and outcome (live birth/ stillbirth/ neonatal death), which will be assessed at birth. Infant-related outcomes measured at birth, 6, 10, 14 weeks, and 6 months of age, which includes birth weight, breastfeeding practices, weight at 6 months of age, immunization practices, and developmental milestones, which includes the gross motor, fine motor, language, and social/cognitive, which are a part of the maternal and child health card, Government of India. These assessments are being carried out by the JRFs. The data collection procedure is diagrammatically depicted in Figure 1.
Statistical Analysis
Data are entered in Microsoft Excel in addition to entry at District Health Information System-2 (DHIS-2) developed and maintained by the Data Management Unit (DMU), ICMR. Statistical analysis will be performed using Statistical Package for the Social Sciences (SPSS) Inc., Chicago, USA, Version 17.0. Continuous variables will be expressed as means/medians ±SD/quartiles and categorical variables as percentages. Percentages and proportions will be used to summarize categorical variables. The dependent variables include categories of depression scores, while variables like sociodemographic factors, obstetric risk factors, and pregnancy outcomes will be considered as covariates. The bivariate analysis will be conducted by applying the chi-square test (Fisher’s exact test when the expected value is <5) for categorical variables. A P value of <0.05 will be considered as the criterion for statistical significance. Multiple logistic regression analysis will be done on the outcome variables found to be statistically significant in the bivariate analysis. Adjusted odds ratios (AOR) with 95% confidence intervals (95% CI) will be computed for adjustment of the confounding variables and to explain the net bearing effect of the different independent variables. Missing data will be dealt with using several methods: completer analysis, last observation carried forward, and multiple imputations to do sensitivity analyses.
Ethical Considerations
The study is approved by the Institutional Ethics Committee of all four study sites. Permission from the authorities like Principal/Director of the institutes, Medical Superintendent of the attached hospitals have been obtained. Detailed information about the nature, objectives of the study, the risks and benefits if any, the contents of the brief intervention, number of follow-up visits are provided to the study participants. Written informed consent is obtained at two stages, once for the eligibility survey and subsequently, before recruiting the participants into the trial. To ensure the privacy of the participants, data collection is carried out in a separate space or room in the OPD attached to the study site. Counseling is delivered on a one-on-one basis with only the research staff being present at the site. The participants are compensated for travel and indirect costs. The anonymity of the study participants is ensured. Strict confidentiality of the information collected is maintained. The participants are given the absolute right to withdraw from the study at any stage without specifying the reason.
Expected Harms and Management
The expected foreseeable unwanted events in this trial could be stigma, psychological trauma, worsening of existing symptoms, or the emergence of new symptoms and lack of improvement. To address the issue of stigma due to the detection of depression among the participants, the data collection will be carried out in a separate space within the OPD ensuring privacy. To address the issues of the severity of symptoms or lack of response, the participants will be followed up periodically and anyone with the PHQ-9 scores ≥20 or with self-harm ideation will be referred to a psychiatrist urgently.
All four study sites have the required infrastructure, expertise, and facilities for handling any adverse event. In case of any adverse event/s related or unrelated to an investigational intervention, the PI will notify the Institutional Ethics Committee and the sponsor. In case of any injury occurring to a participant as a result of her/his participation in the study, the PI will facilitate free medical treatment at the hospital.
Data Monitoring
A National Coordinating Unit (NCU) has been set up by the ICMR, which will facilitate the project in terms of scientific inputs, administrative oversight (meeting deadlines, meeting scientific targets, quality control). Each site interacts with members of the NCU for a progress review of the project through conference calls once a month. Standard Operating Procedures (SOPs) have been formulated by the NCU and have been shared with the PIs of individual sites. The DMU at the ICMR has developed a software called the DHIS-2, in which the data collected at each site are entered by the research staff daily. Data collected on their respective sites are checked for errors by the PI weekly so that corrections can be made in a timely fashion. All registers and records maintained by the research staff are reviewed by the PI weekly. At least 25% of the interviews carried out by the research staff are cross-checked randomly on-site by the PI for completeness and adherence to the study protocol. Supportive supervision is being provided to the research staff by the PI. Additionally, random site visits are carried out by the PI and other members of the study team, once a week on any working day.
Data Collection Procedure Which is Being Followed in This Trial
Discussion
The existing research on the effectiveness of a brief psychological intervention for depression in pregnancy is confined to a few studies conducted abroad and studies with follow-up are scant. To our knowledge, this is one of the largest multicentric studies that test the effectiveness of a psychological intervention for PD delivered by nurses. It also includes follow-up of antenatal women right from conception to mother—child dyad till 6 months postpartum with serial assessments. This is likely to provide valuable insights about maternal depression, it’s course during the perinatal period, it’s impacts on pregnancy and infant outcomes in low middle-income countries.
Furthermore, our study uses PHQ for screening as well as assessment of the severity of PD. It has been translated and is widely available in various Indian languages. It has been validated for screening and severity assessment of PD.44,45 Other scales like Beck’s Depression Inventory (BDI), Edinburgh Postnatal Depression Scale (EPDS), Hospital Anxiety and Depression Scale (HADS), and Hamilton Depression Rating Scale (HAM-D) were considered during the development of the protocol. However, translated and validated versions of these scales having robust psychometric properties in different Indian languages were not available at the time of protocol development. 46 Therefore, the use of a study tool that has robust psychometric properties like PHQ adds merit to our study.
Our study is not devoid of limitations. The four study sites were not randomly selected across India. There is no representation from central and eastern India that would have made the findings of the study more generalizable. However, this study is an outcome measure of a capacity building workshop on implementation research in mental health that was organized by the ICMR. Four of the authors of this publication (PR, RR, PK, MM), who are faculty members in the four study sites were trainees at this workshop. They demonstrated an interest in developing and testing the effectiveness of a psychological intervention for PD. Due to this like-mindedness, the four authors decided to collaborate for this multicentric trial. Therefore, the four study sites (one each in northern and western India and two in the south Indian state of Karnataka) were selected.
Nevertheless, our study has policy implications and would facilitate the incorporation of mandatory screening of pregnant women for depression into routine antenatal care. This may be instrumental in developing an effective program for universal screening of common mental disorders in antenatal clinics at par with screening for physical illnesses. This could also facilitate the development of an effective referral system and empower NSHWs in providing care for mild illnesses, thereby reducing the treatment gap. In the long run, studies of such nature would help in early detection of depression and prevent the associated unfavorable outcomes.
Footnotes
Acknowledgements
This work is part of the BIND-P project (CTRI/2018/07/014836) under the Capacity Building group of NCD-ICMR. The work was supported by the ICMR under Capacity Building Projects for NMHP. We thank Dr Soumya Swaminathan, then Secretary, Department of Health Research (DHR), Dr Balram Bhargav, current Secretary DHR, Prof. V. L. Nimgaonkar, Dr Ravinder Singh, and Dr Harpreet Singh. We thank the faculty of “Cross-Fertilized Research Training for New Investigators in India and Egypt” (D43 TW009114, HMSC File No. Indo-Foreign/35/M/2012-NCD-1, funded by Fogarty International Centre, NIH). We are also thankful to the National Coordinating Unit (NCU) of ICMR for NMHP Projects for their constant support and guidance. We thank the DMU of ICMR for designing the database. The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Health (NIH) or ICMR. NIH and ICMR had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Declaration of Conflict of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Opinions, discussions, views, and recommendations expressed in this article are solely those of the authors and do not necessarily represent that of the organizations they are affiliated with.
Funding
This protocol paper describes the methodology of a research project funded under ‘Capacity Building Task Force for Mental Health Research in India’ funded by the Indian Council of Medical Research vide file number 5/4-4/151/M/2017/NCD-1. International mentors were funded by the training program 'Cross Fertilized Research Training for New Investigators in India and Egypt' funded by FIC, NIH (No. D43 TW009114).
Trial Registration
The trial is registered prospectively before the recruitment of the study participants commenced in the Clinical Trial Registry maintained by the Government of India (CTRI/2018/07/014836).
This detail can be found at the International Clinical Trial Registry Platform maintained by the World Health Organization.
