Abstract
Background:
Suboptimal management of depression in type 2 diabetes mellitus (T2DM) often translates into poor glycemic control, medical complications, and impaired quality of life. Feasibility and effectiveness of collaborative care models of depression in diabetes in low- and middle-income countries (LMICs) remain unexplored. DIAbetes Mellitus ANd Depression (DIAMAND) study, a multicentric single-blind randomized controlled trial (SBRCT) comparing effectiveness of fluoxetine and mindfulness in primary care settings, addresses this gap in scientific literature.
Methods:
This trial conducted in diverse geographic settings of New Delhi, Bengaluru, and Bhubaneswar will comprise module-based training of primary care providers (PCPs) for screening, diagnosing, and managing depression in diabetes in phase I. Phase II will involve four-arm parallel group RCT on 350 participants with T2DM with comorbid depressive episode randomly allocated to receive fluoxetine, mindfulness therapy, fluoxetine plus mindfulness therapy, or treatment as usual at primary care settings. Interventions would include fluoxetine (up to 60 mg/day) and/or sessions of mindfulness for 16 weeks. Primary outcomes on standardized rating scales include depression scores (Hamilton Depression Rating Scale), treatment adherence (Adherence to Refill and Medication Scale), self-care (Diabetes Self-Management Questionnaire), diabetes-related distress (Diabetes Distress Scale), and glycemic control. Secondary outcomes include quality of life (World Health Organization Quality of Life Brief version [WHO-QOL BREF]) and mindfulness (Five Facets Mindfulness Questionnaire).
Discussion:
This RCT will investigate the effectiveness of module-based training of PCPs and feasibility of collaborative care model for managing depression in T2DM in primary care settings in LMICs and effectiveness of fluoxetine and/or mindfulness in improving diverse outcomes of T2DM with major depression.
Depression is a common comorbidity in diabetes with large community-based studies reporting prevalence as double (20.03%) than the general population (10.8%).1,2 Diabetes and depression share a bidirectional relationship with common risk factors and shared etiopathogenesis, and adversely impact outcomes of each other through neurohormonal and behavioral pathways.3–5 Comorbid depression often results in poor treatment adherence and impaired glycemic control, increased incidence of microvascular and macrovascular complications, and poor quality of life (QoL). 5
Currently, 80% people with diabetes live in low- and middle-income countries (LMICs) of which only half receive some form of clinical care which is often suboptimal. 6 By 2030 a 69% increase in prevalence of diabetes is estimated for LMICs, i.e. three times higher than for high-income nations. 7
In India, community level diabetes care is provided through National Program for Prevention and Control of Diabetes, Cancer and Stroke (NPPCDCS) whereas community mental healthcare is provided through National Mental Health Program (NMHP). Poor organizational and system level integration difficulties alongside financial, infrastructure, and manpower deficits further compound the problem of mental healthcare delivery in diabetes clinics. 8 Task sharing, task shifting, and integration of mental healthcare have been proposed as mechanisms of scaling up care for patients with diabetes. 9
Collaborative Care for Depression in Diabetes
There are few evidence-based guidelines and successful models for combined psychological and pharmacological management of depression in diabetes. Service delivery for depression in patients with diabetes in collaborative care settings requires integration of specialist mental healthcare with existing primary and secondary level healthcare or stepped up care models to effectively treat depression and improve glycemic outcomes.9–16
Effectiveness of Antidepressant Medications
Improvement of depression usually translates into better treatment compliance and glycemic control by improving physical activity, dietary regulation, and blood glucose monitoring and adherence to medications mediated through behavioral pathways. 5 Specific serotonin reuptake inhibitors (SSRIs) like fluoxetine are known to have the most favorable efficacy vs side-effect profile in treatment of depression in comorbid medical conditions.17–19 In an RCT conducted by Lustman et al., fluoxetine significantly reduced depression as well as improved glycemic control as compared to placebo in patients with diabetes. 18
Role of Combined Pharmacological and Psychological Treatments
The Cochrane review on pharmacological and psychological interventions in diabetes and depression reported moderate effect sizes for improvement of depression, glycemic control, treatment adherence, QoL, and intervention cost at short and medium term. However, the trials had methodological issues of not investigating most outcomes sufficiently and lacking follow-up data, thereby limiting the evidence on the sustainability of treatment effects. 20
Mindfulness-based practices (MBPs) are characterized by focus in present moment, awareness of breath, bodily sensations, emotions and thoughts, inculcation of positive emotional experiences like joy, gratitude, regulation of emotion, attention, and behavior by performing both formal and informal structured mindfulness exercises. 21 MBPs are unique in their approach towards metacognition with focus and acceptance of experiences of emotion, thoughts, and bodily sensations rather than challenging and altering thoughts that are central to cognitive approaches. 22
MBPs have proved effective in the management of anxiety, depression, stress, chronic pain, and improving QoL, reducing diabetes distress, improving medication adherence, diabetes-related self-efficacy, and even medical outcomes of glycemic control.22–25 MBP therapy has also been found to be as effective as cognitive behavior therapy in reducing diabetes, distress, and depression, and improving well-being in depressed diabetic patients. 26 However, mindfulness-based therapies are also associated with some risks. 27
It is important to demonstrate feasibility and sustainability of mindfulness intervention for patients with depression and diabetes in primary care settings for possible future incorporation as low cost nonpharmacological interventions in NPPCDCS and NMHP in the future.
Materials and Methods
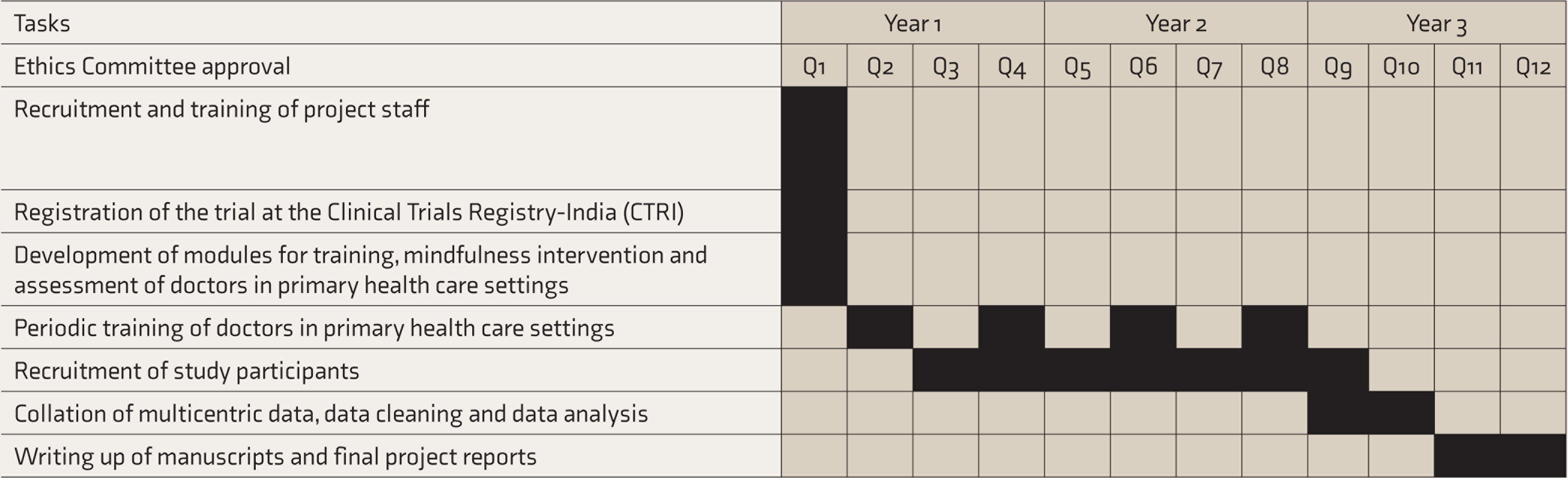
The proposed timeline for study is given in Figure 1.
Study Design and Ethical Approval
The study design of DIAbetes Mellitus ANd Depression (DIAMAND) study is a multicenter, 16-week parallel group randomized single-blind trial in patients undergoing treatment for type 2 diabetes mellitus (T2DM) and diagnosed with depressive episode as per ICD-10 (International Classification of Diseases Tenth Revision) Diagnostic Criteria for Research, and not on any antidepressants for any indication at baseline. The study participants will be allocated in 1:1:1:1 ratio into fluoxetine, mindfulness, combination (fluoxetine and mindfulness), or treatment as usual (TAU). The choice of fluoxetine was based on known efficacy and safety profile as well as easy availability (available free of cost at most primary healthcare centers or government psychiatric facilities in India as an essential drug). 27
The TAU group will not receive any antidepressants but will also receive standard diabetes care. In an effectiveness study, it is not always possible to ensure that patients may not start additional treatments on their own. In order to maintain internal validity, an inventory of all concomitant medications taken by the patient will be obtained at each visit. If a patient has newly started treatment with a medication that is excluded by the protocol, ongoing continuation in the study for that patient will be evaluated on a case-by-case basis. In case of medications such as antidepressants, the patient may be terminated from the study.
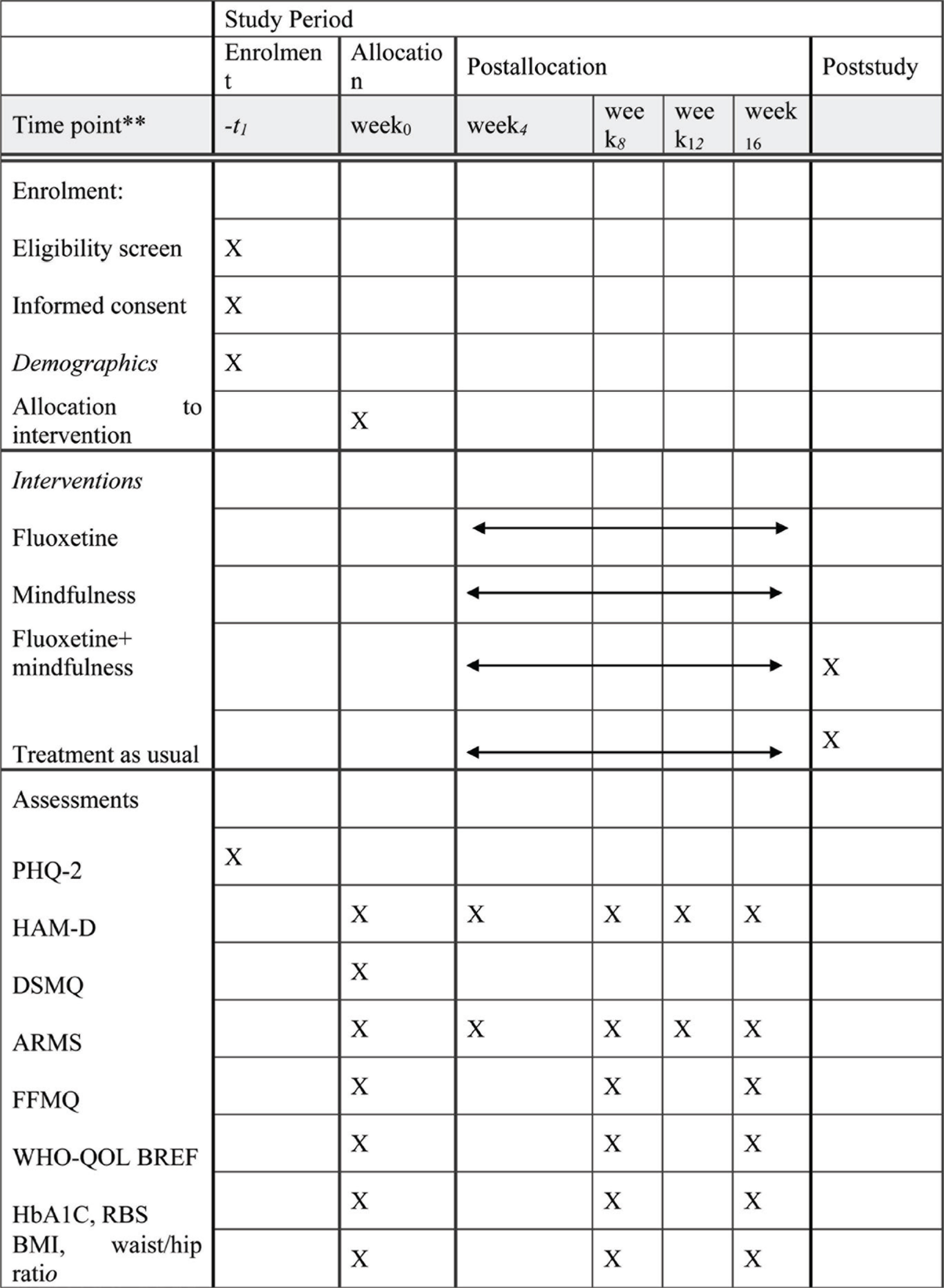
ARMS: Adherence to Refills and Medication Questionnaire, DSMQ: Diabetes Self-Management Questionnaire, HbA1C: glycosylated hemoglobin, FFMQ: Five Facets Mindfulness Questionnaire, HAM-D: Hamilton Depression Rating Scale, RBS: Random Blood Sugar, PHQ-2: Patient Health Questionnaire, WHO-QOL BREF: World Health Organization Quality of Life Brief version. **Exact time-points mentioned in this row.
This trial will be carried out in the form of a collaborative care provision for patients with diabetes attending primary health centers. Mental healthcare treatment that includes pharmacological treatment of depression will be provided through trained primary care physician under supervision, support, and guidance of specialist psychiatrists. Psychological treatment would be provided through MPhil Clinical Psychology/MPhil Psychiatric Social Work qualified Junior Research Fellows (JRFs) trained in mindfulness techniques by mindfulness experts at the National Institute of Mental Health and Neurosciences, Bengaluru and at the Atal Bihari Vajpayee Institute of Medical Sciences (ABVIMS) and Dr Ram Manohar Lohia (RML) Hospital, New Delhi as well as online training on mindfulness-based stress reduction (https://palousemindfulness.com/). The study has received ethical approval from Institutional Ethics Committee of all the three participating centers and registered in the Clinical Trials registry of India (vide ID: CTRI/2019/01/017239 dated 24.01.19).
Study Settings
The study will be conducted at three sites in north (Delhi), east (Bhubaneswar), and south (Bengaluru) zones of India. In Delhi, Central Government Health Scheme (CGHS) Wellness Centre, Timarpur near Delhi University will be the primary site. CGHS Wellness Centres are run by Government of India for providing comprehensive medical care to central government employees and pensioners. In addition, participants attending primary care settings of Delhi Government and Delhi Jal Board near CGHS Wellness Centre, Timarpur, Delhi will also be recruited at this site (site supervised by Chandra M).
In Bhubaneswar, Non-Communicable Disease Clinic of All India Institute of Medical Sciences (AIIMS), a model flagship center under program of Non-Communicable Disease Control and Prevention Program for the state of Odisha, will be the study site. The services are oriented towards maintaining adequate control and prevention of complications of diabetes mellitus and hypertension (supervised by Patra S).
In Bengaluru site, the participants will be recruited from the Community Health Centre in Kengeri satellite town and Anekal General Hospital in Anekal town which are run by the Department of Health and Family welfare of the Government of Karnataka (sites will be supervised and managed by Raveendranathan D and Johnson Pradeep R).
In phase 1, all PCPs at study sites will receive a standard module-based training for diagnosis and management of diabetes mellitus along with a pre and post case vignette and questionnaire-based assessment for knowledge, attitude, and clinical skills of primary healthcare personnel.
To incorporate subjects with diabetes who do not attend primary care for diabetes or any other health-related needs and self-refer to tertiary care settings, module-based training programs will also be organized for physicians at ABVIMS and Dr RML Hospital, AIIMS Bhubaneswar, and St John’s Medical College. This will lead to a much larger pool of physicians trained to diagnose and manage depression in patients with diabetes and these centers may also function as additional recruitment sites.
Inclusion and Exclusion Criteria
These criteria reflect the real-world clinical characteristics of patients with T2DM who seek care are primary health centers. The inclusion criteria are: (1) T2DM diagnosed as per American Diabetes Association guidelines 27 , (2) both genders, (3) age 20–60 years, (4) on standard of care for management of diabetes for past 3 months, 28 (5) mild to moderate depressive episode diagnosed as per ICD-10 Diagnostic Criteria for Research (DCR), 29 and severity of depression as assessed by Hamilton Depression Rating Scale (HAM-D) 30 score between 8 and 24.
The exclusion criteria are: (1) antidepressant use for any indication in last 3 months; (2) major complications of diabetes mellitus such as retinopathy, nephropathy, etc.; (3) poorly controlled hypertension; (4) major psychiatric disorders (schizophrenia, bipolar affective disorder); (5) substance use disorders except nicotine; (6) suicidal ideation, suicidal gestures, psychotic symptoms, or agitation; (7) neurological/neurosurgical disorders, cerebrovascular disorders, seizure disorder, and head injury; (8) disorders of cognitive impairment like dementia, mental retardation; (9) recent myocardial infarction within past 6 months; and (10) history of allergy to any antidepressants.
Sample Size Estimation
Sample size (N) was calculated by keeping the primary outcome of mean depression scores on HAM-D 29 and response to fluoxetine at 66.7% (p1) compared to 37% for placebo (p2) based on the study by Cipriani et al., 27 type I error at 5%, power at 90%, using two-sided Tukey’s critical Z value for multiple comparisons for type I error of 5% and six pairwise comparisons (fluoxetine and TAU; mindfulness and TAU; fluoxetine and mindfulness, combination and TAU, combination and fluoxetine, combination and mindfulness).
The sample size is determined to be 76 for each arm, i.e. 304 for four arms and enhanced to 350 considering anticipated attrition of 15%. All sites will recruit 350 participants each with a total sample size of 1050.
Enrolment and Randomization
The enrolment, randomization, and follow-up protocol of the study is given in Figure 2. Informed written consent would be taken from participants in their vernacular language in a proforma approved by the local ethics committee. Screen positive patients having Patient Health Questionnaire (PHQ)-2 scores of ≥3 29 would be further assessed using ICD-10 DCR. 30 All consenting patients diagnosed with major depressive episode as per ICD-10 by primary care physician and confirmed by trained mental health professional and scoring between 8 and 24 on HAM-D would be included in the study.
Timeline of DIAbetes Mellitus ANd Depression (DIAMAND) Project
Recruited participants would then be randomized in 1:1:1:1 ratio into fluoxetine, mindfulness, combination, and TAU groups using a computer-based randomization program using permuted block randomization method with blocks of eight allocations to ensure equal number of participants in each treatment group. The allocation sequence would be concealed by using sequentially numbered opaque sealed envelopes. Independent evaluators blinded to treatment assignment will rate participants. Blinding will be removed at the recommendation of the site principal investigator (PI) in case of any medical emergency in order to know the medications received by the patients.
Study Medication
Fluoxetine will be initiated at 20 mg/day in participants in fluoxetine and combination arms by the primary care physician and titrated up to a maximum of 60 mg/day for optimal management. Adherence to medication will be monitored by self-report and pill count. Concurrent use of antipsychotics or mood stabilizers will not be allowed to the participants in the study period. Only benzodiazepines will be allowed on as-needed basis for the study duration. However, medications for any other medical indication are permitted in the study period in keeping with the TAU protocol.
Mindfulness Therapy
Various RCTs have been reported using mindfulness-based interventions for depression in primary care.31–34 Mindfulness-based cognitive therapy has been found effective in managing depression in adolescents as well as elderly Indian population.35,36 Mindfulness-based stress reduction, mindfulness-based cognitive therapy, and brief mindfulness interventions have been used for depression in primary care. We shall be using brief mindfulness-based intervention to the study participants randomized to mindfulness and combination arms as a scripted module of on mindfulness interventions designed for patients with diabetes and depression based on evidence-based mindfulness interventions provided in primary care settings.25,26 The sessions will include introduction and understanding of the concept of mindfulness, its applicability to patients with diabetes and depression, and general principles of practice of mindfulness. 34 The module will include mindful breathing, body scan, gratitude, and compassion exercise. In addition to formal mindfulness techniques, informal mindfulness procedures like practicing mindfulness in daily activities like cooking, cleaning, showering, etc. will be discussed. The participants will be taught the techniques of slowly disengaging from personal thoughts, emotions, and bodily symptoms, gain a sense of personal balance, and reach a state of self-compassion. 34
Script for mindfulness module will also be audio recorded at the Delhi center in English and Hindi which will then be translated into Kannada and Odiya. Bilingual experts at individual sites would be involved in the translation of mindfulness intervention module. The final version would then be prepared by consensus of the PIs of the three sites. An audio recording of the mindfulness intervention will be shared with participants of mindfulness and combination arms in the language of their fluency to aid daily practice of mindfulness in home settings. Five mindfulness sessions lasting 40–45 minutes in duration would be provided at baseline, 4, 8, 12, and 16 weeks monthly intervals. Booster sessions will be provided at each follow-up. Mindfulness therapy intervention would be delivered by mental health professionals trained in mindfulness. The JRFs would have access to the written module as well as audio recording of mindfulness intervention for reference and self- monitoring. The mindfulness sessions administered on-site by project staff will be recorded and randomly checked for fidelity on a periodic basis by site PI. Adherence to mindfulness practice shall be ascertained and reinforced by weekly telephonic enquiry by the research staff. In addition, all study participants in mindfulness (n = 76) and combination arms (n = 76) will be shared a monitoring format to document daily practice of mindfulness which will be evaluated on monthly follow-ups.
Follow-Up Plan and Outcome Measures
All participants will be assessed at baseline, 2, 4, 8, 12, and 16 weeks. We are using SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials Template) flowchart which is as per the SPIRIT guidelines, a standard reporting guideline for intervention protocols (Figure 1). The study flow and participant recruitment are shown in Figures 2 and 3. For the primary outcome, participants will be rated using the HAM-D. 37 Other outcomes like Adherence to Refill and Medication Scale (validated for treatment adherence even in low literacy populations [α = 0.828]), 39 Diabetes Self-Management Questionnaire (has four subscales Glucose Management, Dietary Control, Physical Activity, Health-Care Use and a Sum Scale assessing self-care behaviors predicting glycemic control [convergent correlation with HbA1C: –0.40]), 40 and Diabetes Distress scale (assesses four distress-related domains: emotional burden, physician-related distress, regimen-related distress, and diabetes-related interpersonal distress with high reliability (alpha > 0.87) and validity), 41 QoL measured with WHO-QOL BREF (standardized scale validated in diverse cultural settings), 42 mindfulness measured with Five Facets Mindfulness Questionnaire (comprising of five independently developed mindfulness questionnaires), 43 and changes in glycosylated hemoglobin (HbA1C) levels as marker of glycemic control would be considered secondary. In addition, body mass index, waist hip ratio, and fasting blood glucose levels will be recorded for subsequent analysis. The project staff will be trained for administration and rating of all scales by site PI prior to commencement of study. Inter-rater reliability exercises will be carried out weekly with site PI as the gold standard rater to ensure robustness of collected data. The protocol for collection of anthropometric data, scale-based assessments, and laboratory investigation is given in Figure 4.
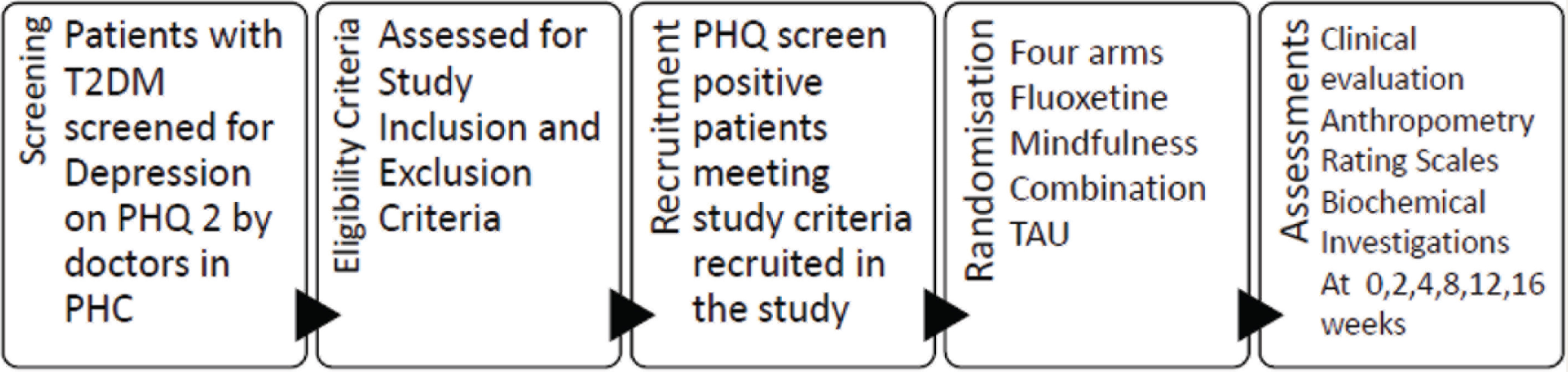
PHC: primary health center, PHQ-2: Patient Health Questionnaire: 2 items, T2DM: type 2 diabetes mellitus; TAU, treatment as usual.
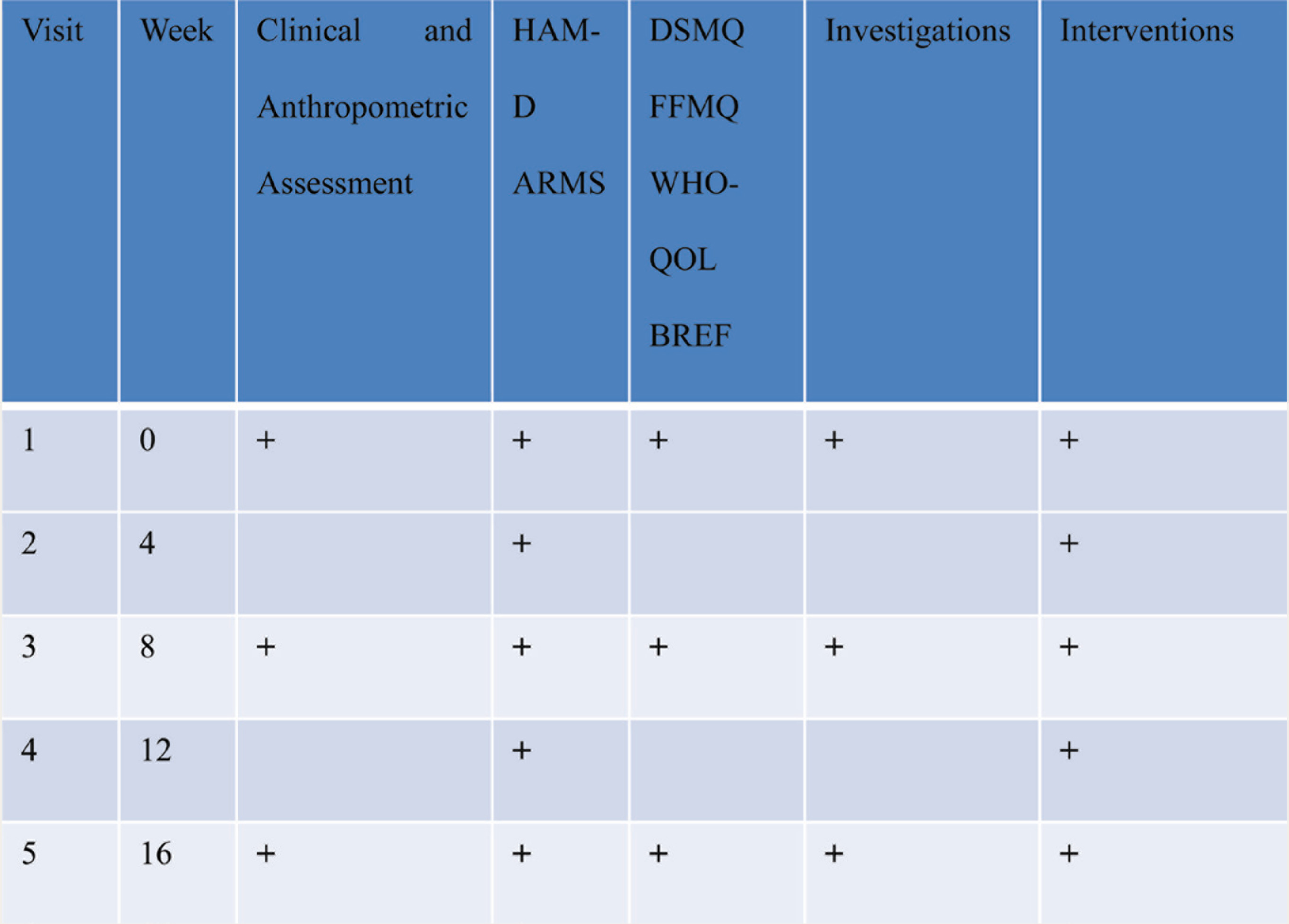
ARMS: Adherence to Refills and Medication Questionnaire; DSMQ: Diabetes Self-Management Questionnaire, HbA1C: glycosylated hemoglobin, FFMQ: Five Facets Mindfulness Questionnaire, HAM-D: Hamilton Depression Rating Scale, WHO-QOL BREF: World Health Organization Quality of Life Brief version.
Unblinding Protocol and Safety Measures
Identification of death wishes, suicide ideas, suicide plans, suicide attempts, psychosis, or mania would be done clinically while evaluation of depression at baseline and follow-up. Monitoring for adverse effects of both fluoxetine (like dermatological, respiratory, gastrointestinal side effects, manic switch) and mindfulness (somatic pain, disorientation, addiction to meditation, suicidal ideation, and destructive behavior) will be done during follow-up. 42 Similarly, any serious or life-threatening event like myocardial infarction, diagnosis of cancer, head injury etc. occurring during the course of the trial will also be documented. All such participants will be unblinded and excluded from the trial, and appropriate clinical care would be provided including emergency referral to a higher center.
A trial monitoring committee has been constituted at each site to monitor the sites for any adverse events. A six-monthly report would be sent to them for their decision on continuation or termination of the trial.
Statistical Analysis Plan
All patients who complete randomization will be included in the intent-to-treat analysis regardless of compliance with study visits or treatment. The efficiency of randomization will be examined to ascertain whether important sociodemographic and clinical confounders are evenly distributed at baseline in the different arms of the study.
A mixed model regression analysis with fixed and random effects will be carried out to compare changes in primary and secondary outcomes in treatment conditions, namely fluoxetine, mindfulness, combination of both versus TAU, while also investigating for confounders like age, gender, and duration of illness for diabetes. Effect sizes between treatment arms will be calculated to assess the magnitude of differences within groups. All the analyses will be done using IBM SPSS version 26.0 or STATA version 13.0.
The differences in outcomes across the three sites will also be investigated using site as an interaction term during analysis of pooled dataset of all three sites.
Discussion
The evidence base for treatment of depression in diabetes is limited. 27 German Diabetes Association recommends simultaneous improvement of psychological as well as medical outcomes as standard of care for depressed diabetics, i.e. remission of depression and achievement of glycemic control. 45
The results of DIAMAND study may provide insights into the process of improvement of psychological and glycemic outcomes and hence may add to the scientific literature on the role of adequate antidepressant treatment in the management of diabetes. The results may also provide the much-needed evidence for collaborative care for depression in diabetes in the Indian context. Integration of mental healthcare with existing health services delivery system through mutual cooperation, proactive follow-up of patients, monitoring of patient outcomes, and support for primary care physician would be unique in our model. In addition, the results of DIAMAND study would be informative in terms of evaluation of feasibility as well as efficacy of both simplified psychological therapy and pharmacological intervention in routine primary care clinical settings.
The study protocol has incorporated provision of formal clinical assessment using standardized nosological system for a definite diagnosis of depression by PCPs who have not been adequately trained in psychiatry. This will help demonstrate the success of task-shifting and task-sharing approaches advocated by World Health Organization for reducing treatment gap in mental disorders in LMICs. 47 Use of validated instruments for screening, diagnosis, and assessment of depression, QoL, adherence to medications, and self-management is another strength of our study.
Our study will investigate both psychological and glycemic targets as treatment outcomes. Hence, careful evaluation of depression, QoL, and self-care would be helpful in assessing psychological response, whereas adherence to medications and glycemic control will indicate the impact of antidepressant treatment on diabetes control and management. Our results would add to the existing evidence base for effect on glycemic control upon medium-term use of SSRIs which is lacking to date. 10
There are a few limitations in the study design. The single-blind design may introduce systematic bias based on the participant’s perception of the nature of intervention. Participants may have preconceived notions about medication (more effective/less desirable) and mindfulness intervention (culturally more acceptable/less effective) which can contribute to a placebo or nocebo effect. However, as the study is a framework for the intervention for depressed patients with T2DM seeking healthcare services at primary care level in LMICs, a pragmatic effectiveness and feasibility approach, which can be translated into clinical practice, was chosen.
There is a possibility of treatment contamination if any study participant does not follow the protocol for the assigned treatment either by nonadherence or partial adherence (not receiving the recommended intervention because of patient preference or treatment intolerance) or treatment crossover (receiving the intervention intended for the other group in a trial, e.g. if participants of different arms interact and on their own take intervention not assigned to them). A Contamination Adjusted Intention to Treat (CA ITT) analysis will be a better statistical strategy in such a case but requires a bigger sample size. 45 As each site is achieving minimum sample size individually, the pooled data set of all three sites will provide sufficient numbers to conduct CA ITT analysis as well, if required.
The experience from collaborative care treatment in three different medical setups from varied geographical regions from northern, eastern, and southern parts of India would help in understanding the challenges and difficulties in providing collaborative care for depression in primary healthcare settings in different cultural contexts with diverse constraints in healthcare resources and logistics. This experience would help in formulating and planning future research in identifying active treatment ingredients for unique sociocultural needs of India. This treatment model would help in formulating integration and provisioning of mental health care within the existing framework of NMHP and NPCDCS.
Trial Status
Phase I of the trial involving training of primary healthcare physicians has been completed, and phase II involving recruitment and randomization of participants has been initiated at all sites.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This protocol paper describes the methodology of a research project funded under ‘Capacity Building Task Force for Mental Health Research in India’ funded by the Indian Council of Medical Research vide file number 5/4-4/151/M/2017/NCD-1. International mentors were funded by the training program 'Cross Fertilized Research Training for New Investigators in India and Egypt' funded by FIC, NIH (No. D43 TW009114).
