Abstract
Streptozotocin (STZ) is known to induce renal tumors in rodents, but their similarity to human tumors remains poorly defined. We characterized and comparatively validated a mouse model of STZ-induced renal tumorigenesis by administering a single intraperitoneal dose of STZ (250 mg/kg) to female CBA/NSlc mice and maintaining them for 182 days. Renal tumors developed in 25 of 28 surviving mice (89%), with mean and median numbers of 3.4 and 2.5 tumors per animal, respectively. Histopathologically, the tumors were diagnosed as adenomas or adenocarcinomas and exhibited clear, eosinophilic, or mixed cytoplasm. Immunohistochemical analysis of four representative adenocarcinomas revealed positivity for CK AE1/AE3, CK7, PAX8, CD10, CD82, E-cadherin, CD117, and S100A1, and negativity for CK20 and vimentin. These morphological and immunohistochemical features resembled those of human chromophobe renal cell carcinoma (chRCC). Furthermore, the tumors expressed collecting duct markers (uromodulin, CD15, MUC1, and GLUT-1) but lacked proximal convoluted tubule markers (AQP1 and megalin), suggesting a collecting duct origin. Taken together, STZ-induced mouse renal tumors closely resemble human chRCC, providing a reproducible model for investigating the biology and potential therapeutic approaches for this tumor type.
Keywords
Introduction
Various experimental animal models have been developed to study human renal tumors, including chemical tumorigenesis, tumor cell line allografts (syngeneic), tumor cell line xenografts, and genetically engineered models. Chemical tumorigenesis models, in which chemical agents are administered to animals to induce renal tumorigenesis, lack tissue specificity. Nevertheless, they offer considerable advantages, such as no requirement for genetic modification or immune system suppression, while enabling the observation of the sequential tumorigenesis process. 19 Therefore, chemical tumorigenesis models are valuable research tools. Several chemical agents, including streptozotocin (STZ), 2-acetylaminofluorene, and dimethylnitrosamine, have been reported to induce renal tumors in such models. 19 However, despite these developments, a well-characterized renal chemical tumorigenesis model in mice remains limited. Validating mouse models of renal chemical tumorigenesis has been inadequately explored.
The most well-known and relatively well-investigated mouse model of renal chemical tumorigenesis involves the use of STZ, an organic compound produced by the bacterium Streptomyces achromogenes. STZ is known to induce tumorigenesis in the kidneys and other organs and destroy pancreatic β-cells in mammals. 19 In medical research, STZ is primarily used to establish animal models of diabetes, while clinically, it is employed as a therapeutic agent for pancreatic and gastrointestinal neuroendocrine tumors (commercial name: Zanosar®). 8 In 1967, Arison and Feudale first reported that a single administration of STZ can induce renal tumors in rats. 2 Following this discovery, Rakieten et al, 18 Horton et al, 11 and Kazumi et al 12 further developed an STZ-induced renal tumor model in rats by maintaining large cohorts. In 1981, Mandel et al 14 observed that STZ could also induce renal tumors in mice during diabetes research investigating the effects of pancreatic islet transplantation, marking the beginning of studies on STZ-induced renal tumorigenesis in mice. In 1985, Hard conducted experiments with a large number of mice and demonstrated that intravenous administration of STZ (250 mg/kg) to 6-week-old CBA/H/T6J male and female mice increased the incidence of renal tumors in both sexes. 10 Hard’s model was used as a prototype for subsequent mouse models of STZ-induced renal tumorigenesis. Building on Hard’s work, Miyao et al 15 divided CBA/H/T6J female mice into several groups, euthanized them after different rearing periods, and reported a high incidence of renal tumorigenesis when mice were maintained for more than five months after STZ administration. Since then, although mouse models of STZ-induced renal tumorigenesis have been reported sporadically, no study has comprehensively examined the similarities between STZ-induced mouse renal tumors and human renal cancers.6,9
To characterize and comparatively validate a robust and useful mouse model of STZ-induced renal tumorigenesis, it is essential to confirm that a high incidence of renal tumors can be consistently achieved by maintaining at least several dozen mice for an appropriate duration. In the present study, we demonstrated that maintaining a sufficient number of CBA/NSlc female mice for 182 days (26 weeks) following STZ administration reliably generated renal tumors. Moreover, immunohistochemistry (IHC) and colloidal iron staining revealed that STZ-induced mouse renal tumors exhibited characteristics similar to those of human chromophobe renal cell carcinoma (chRCC). Here, we described the experimental protocols used and the histopathological features of STZ-induced mouse renal tumors in detail.
Materials and Methods
Animals and Chemicals
Based on the report by Hard 10 that female mice exhibited better survival than males approximately one year after STZ administration, we used female mice in this study. Thirty-five female CBA/NSlc mice (4 weeks old) were purchased from Japan SLC. Upon arrival, the mice were randomly assigned to one of two groups: 30 mice were designated as the experimental group, while the remaining five mice served as the control group. STZ was purchased from the FUJIFILM Wako Pure Chemical Corporation (Osaka, Japan). After a two-week acclimation period, at 6 weeks of age, mice in the experimental group were administered a single intraperitoneal injection of STZ (250 mg/kg body weight); the control group mice received a single intraperitoneal injection of saline (0.2 mL/20 g body weight). STZ and saline were administered via intraperitoneal injection according to the method described by Miyao et al, 15 with the STZ dosage also based on their protocol. Five mice were housed per cage under controlled conditions, including a 12-hour light/dark cycle (lights on from 8:00 a.m. to 8:00 p.m.) and room temperature of 22°C, and ad libitum access to food (MF diet; Oriental Yeast Co., Ltd., Tokyo, Japan) and water. Twenty-six weeks post-injection, the mice were euthanized by carbon dioxide inhalation and necropsied for tissue collection. All mice were maintained at the Osaka Medical and Pharmaceutical University in accordance with the institutional guidelines for animal experimentation. All experimental procedures were approved by the Institutional Animal Care and Use Committee of Osaka Medical and Pharmaceutical University.
Histology
Kidneys were sectioned sagittally to produce two separate slices per kidney, as described by Hard. 10 All harvested tissues, including the kidneys, pancreas, lungs, and liver, were fixed in 10% neutral-buffered formalin, dehydrated, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E) using standard protocols. Histopathological examination of STZ-induced renal tumors was performed using light microscopy, focusing on mitotic activity and nucleolar atypia, according to the method described by Miyao et al 15 . Furthermore, we counted the number and maximum diameter of all renal tumors identified in each renal H&E-stained specimen from the experimental group.
Human Tissue Samples
Patients diagnosed with clear cell renal cell carcinoma (ccRCC; n = 5), papillary renal cell carcinoma (pRCC; n = 5), chRCC (n = 7), or renal oncocytoma (n = 4) at Osaka Medical and Pharmaceutical University Hospital (between 2006 and 2020) with complete clinicopathological data were selected. Formalin-fixed paraffin-embedded (FFPE) specimens from these patients were sectioned and subjected to H&E staining using standard procedures. This study was approved by the Research Ethics Committee of Osaka Medical and Pharmaceutical University.
Immunohistochemical, Periodic Acid-Schiff, and Colloidal Iron Staining Assessment
Immunohistochemical staining was performed for CK AE1/AE3, CK7, CK20, vimentin, CD10, CD82, PAX8, E-cadherin, CD117, S100A1, AQP1, megalin, uromodulin, AQP3, CD15, MUC1, and GLUT-1. The dilutions, antigen retrieval conditions, and manufacturer details are presented in Table 1. FFPE specimens of mice and human kidneys were sectioned, mounted on CREST-coated slide glasses (Matsunami Glass Ind., Ltd., Osaka, Japan), and then dried overnight at 45°C. Sections were deparaffinized in xylene, rehydrated through graded ethanol to distilled water, and subjected to heat-induced antigen retrieval using HEAT PRO II (Nichirei Biosciences Inc., Tokyo, Japan) for 20 min or treated with protease for 5 minutes. Immunostaining was performed using a HISTOSTAINER (Nichirei), with incubation with primary antibodies for 30 minutes, followed by incubation with the corresponding secondary antibodies for 30 minutes. This automated system used 3,3'-diaminobenzidine (DAB) as the chromogen.
List of antibodies used in this study.
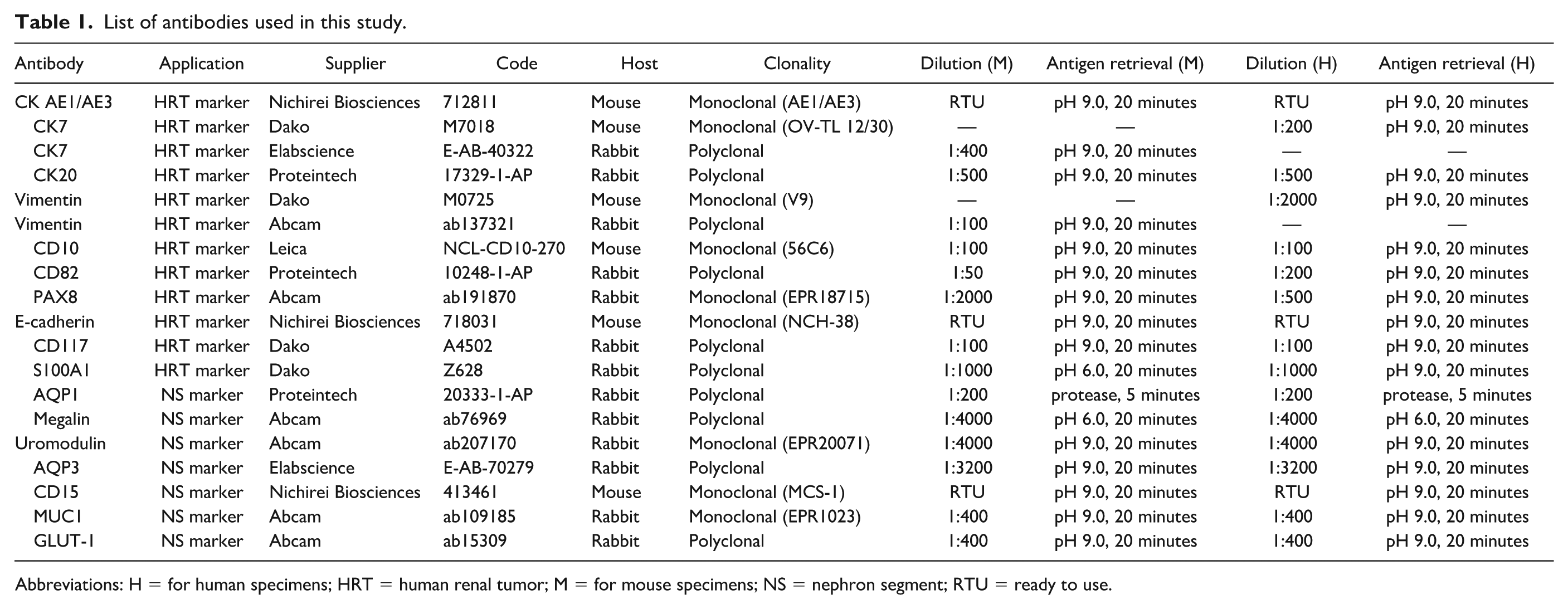
Abbreviations: H = for human specimens; HRT = human renal tumor; M = for mouse specimens; NS = nephron segment; RTU = ready to use.
To perform IHC/PAS double staining, kidney specimens from the control group mice and tumor-free areas of human kidneys diagnosed with ccRCC were selected. Periodic acid-Schiff (PAS) staining was performed after IHC staining. Hale’s colloidal iron stain, which is traditionally used to differentiate chRCC from other renal tumors, was also applied to STZ-induced mouse renal tumors.
Assessment
Immunohistochemical staining of mouse or human renal tumors was scored for extent and intensity as described by Bing et al. 3 The extent was divided into 0, focal (< 25%, score 1), intermediate (25%-50%, score 2), and diffuse (> 50%, score 3). The intensity was scored as 0, weak (score 1), moderate (score 2), and strong (score 3). The final score, defined as “IHC score,” was the sum of the extent and intensity scores divided by 2.
Statistical Analyses
Data analyses were performed using JMP® Pro 17 (JMP Statistical Discovery LLC). Data are presented as mean ± standard error of the mean (SEM). Considering the small sample size (n = 4-7) and non-normal distribution of the data, statistical analyses were performed using the Kruskal-Wallis and Steel-Dwass tests. Differences were considered statistically significant at P < .05. Based on the IHC scores described above, a power analysis was performed for the sample sizes of the five groups compared in this study (STZ-induced mouse renal tumor, ccRCC, pRCC, chRCC, and renal oncocytoma). For CD82, which displayed marked differences in staining among the five groups, the mean IHC scores were 2.5 for STZ-induced mouse renal tumors, 0 for ccRCC, 0 for pRCC, 2.8 for chRCC, and 0 for renal oncocytoma. Using these mean values and a sample size of n = 5 in JMP for power analysis, the statistical power was calculated to be 96.30%, indicating that the actual sample size (n = 4-7) used in this study would be appropriate. This post hoc estimate should be interpreted as an approximation of detectable effect sizes rather than definitive evidence of sufficient power for all markers.
Results
Survival Rate of Mice in the Experimental Group
Twenty-eight of 30 mice in the experimental group (93%) survived until the end of the 182-day observation period. Two mice died of natural causes or were euthanized owing to apparent debilitation and substantial weight loss (> 20% body weight loss within several days) approximately 30 days after STZ administration. As necropsies were not performed on these two mice, the causes of their rapid weight loss and spontaneous death remain unclear. No additional deaths occurred thereafter (Figure 1). Body weights of all mice were measured weekly for 182 days following STZ administration. At day 182, the mean body weight was 20.7 g in the experimental group (n = 28) and 22.9 g in the control group (n = 5). While the body weights of mice in the control group generally remained within a range of 20 to 25 g, those in the experimental group were more variable, ranging from 14 to 27 g. Although quantitative measurements were not performed, mice in the experimental group tended to exhibit increased urine output compared with those in the control group, suggesting that STZ administration caused polyuria, which might reflect diabetes in the treated animals.
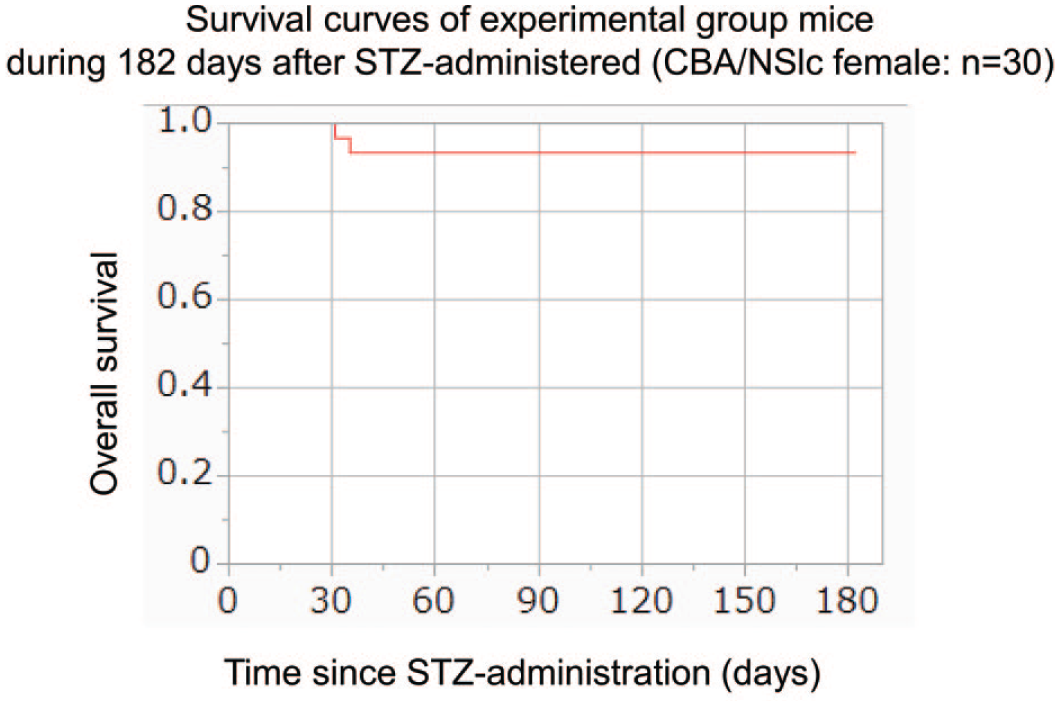
Survival curves of mice in the experimental group during 182 days after STZ administration (n = 30).
Incidence, Number, and Diameter of STZ-Induced Tumors in Mice
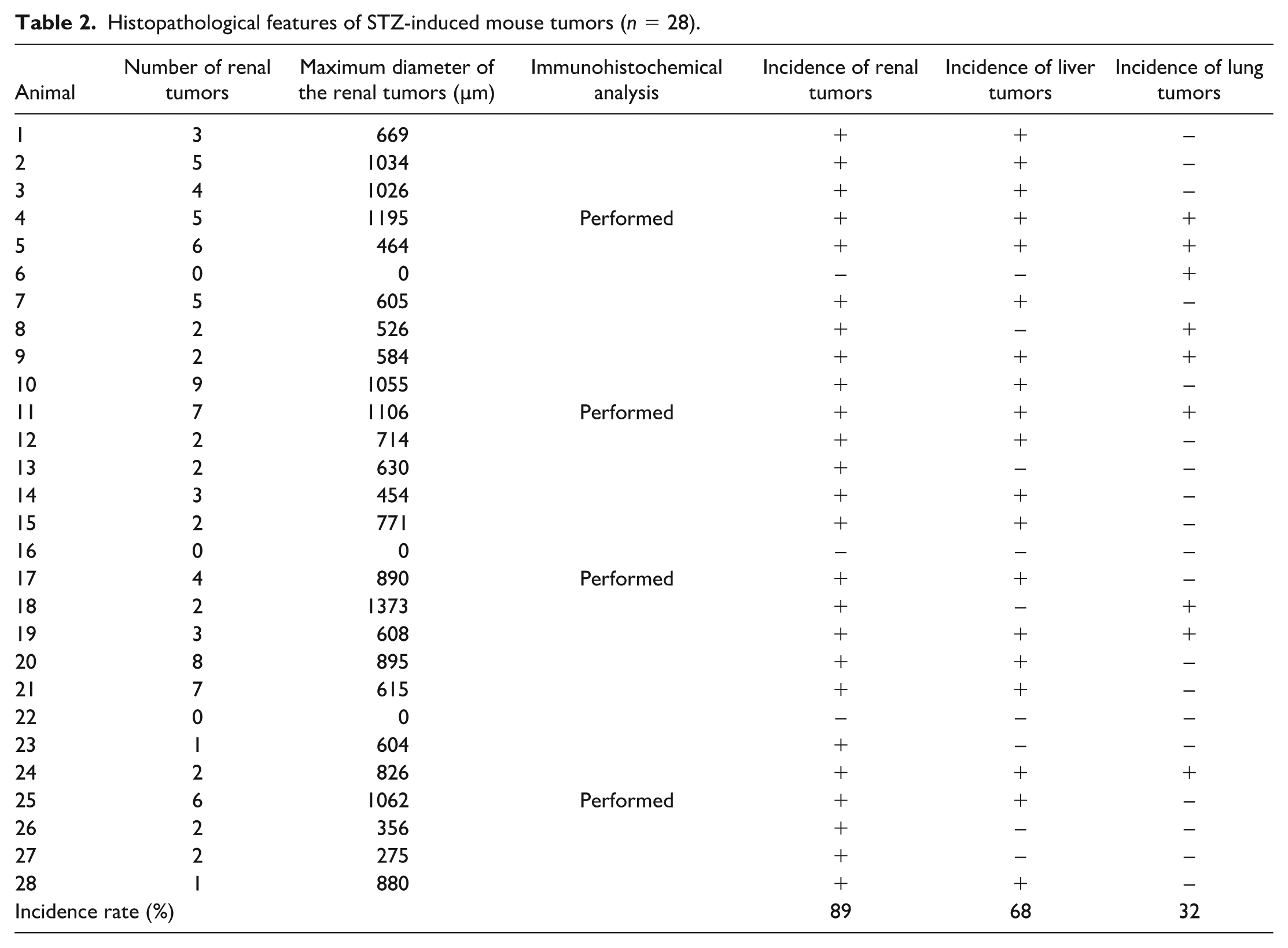
Of the 28 mice that survived for 182 days after STZ administration, 25 developed STZ-induced renal tumors, corresponding to a tumor incidence rate of 89% (25 of 28). The mean and median numbers of renal tumors per mouse were 3.4 and 2.5, respectively. The mean and median maximum tumor diameters were 686 and 650 µm, respectively. Some mice developed liver and lung tumors, as well as cutaneous myomas. Although liver and lung tumors were histopathologically diagnosed as adenocarcinomas, they did not resemble STZ-induced mouse renal tumors. The liver tumors in mice were multiple, well-demarcated lesions within the hepatic parenchyma, composed of proliferating tubular structures with moderate nuclear atypia. No tumor tissue resembling bile ducts was observed. Although the lesions showed adenocarcinoma-like histopathological features, they were not histopathologically similar to typical human hepatocellular carcinomas, and the specific histologic type could not be determined. The lung tumors in mice were histopathologically similar to invasive pulmonary adenocarcinoma (papillary or solid patterns) in humans. It remains unclear whether they represented primary carcinomas or metastases of the renal tumors. Myomas were observed in only one mouse in the experimental group; however, no detailed histopathological examinations were performed. The incidence, number, and diameter of renal tumors, as well as the incidence of liver and lung tumors, are summarized in Table 2.
Histopathological features of STZ-induced mouse tumors (n = 28).
Histopathological Features of STZ-Induced Renal Tumors in Mice
STZ-induced renal tumors in mice were diagnosed as adenomas or adenocarcinomas based on criteria described by Miyao et al. 15 Most tumors exhibited distinct ductal differentiation and nuclear atypia with prominent nucleoli; however, mitotic figures were rarely observed. Two predominant histological subtypes were identified: one with clear cytoplasm and the other with eosinophilic cytoplasm. Representative histopathological features of STZ-induced mouse renal tumors (adenocarcinoma and adenoma) are shown in Figures 2 to 4, while representative histopathological features of the human renal tumors examined in this study (ccRCC, pRCC, chRCC, and renal oncocytoma) are shown in Figure 5. Among human renal tumors, ccRCC typically shows clear cytoplasm, whereas renal oncocytomas usually exhibit eosinophilic cytoplasm. In contrast, chRCCs possess both of these features. 5 These findings suggest that the histopathological characteristics of STZ-induced renal tumors in mice resemble those of human chRCCs.
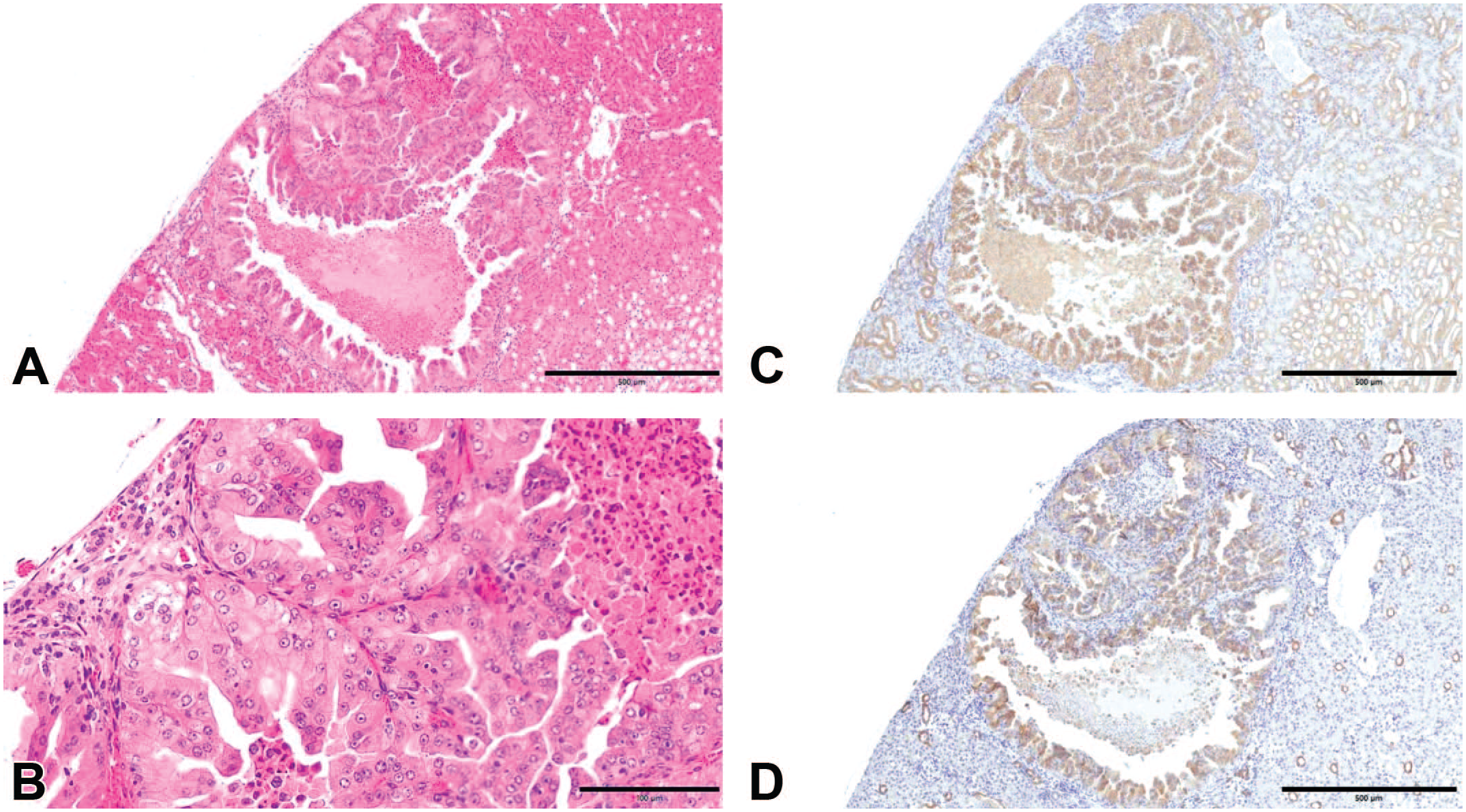
Histopathological and immunohistochemical features. (A) A renal tumor recognized in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 5X). (B) A renal tumor in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 20X). The tumor was histopathologically diagnosed as adenocarcinoma with eosinophilic cytoplasm. (C) E-cadherin is positive in mouse normal distal tubules, normal collecting ducts, and the renal tumor (original objective 5X). (D) Cytokeratin 7 is positive in mouse normal collecting ducts and the renal tumor (original objective 5X).
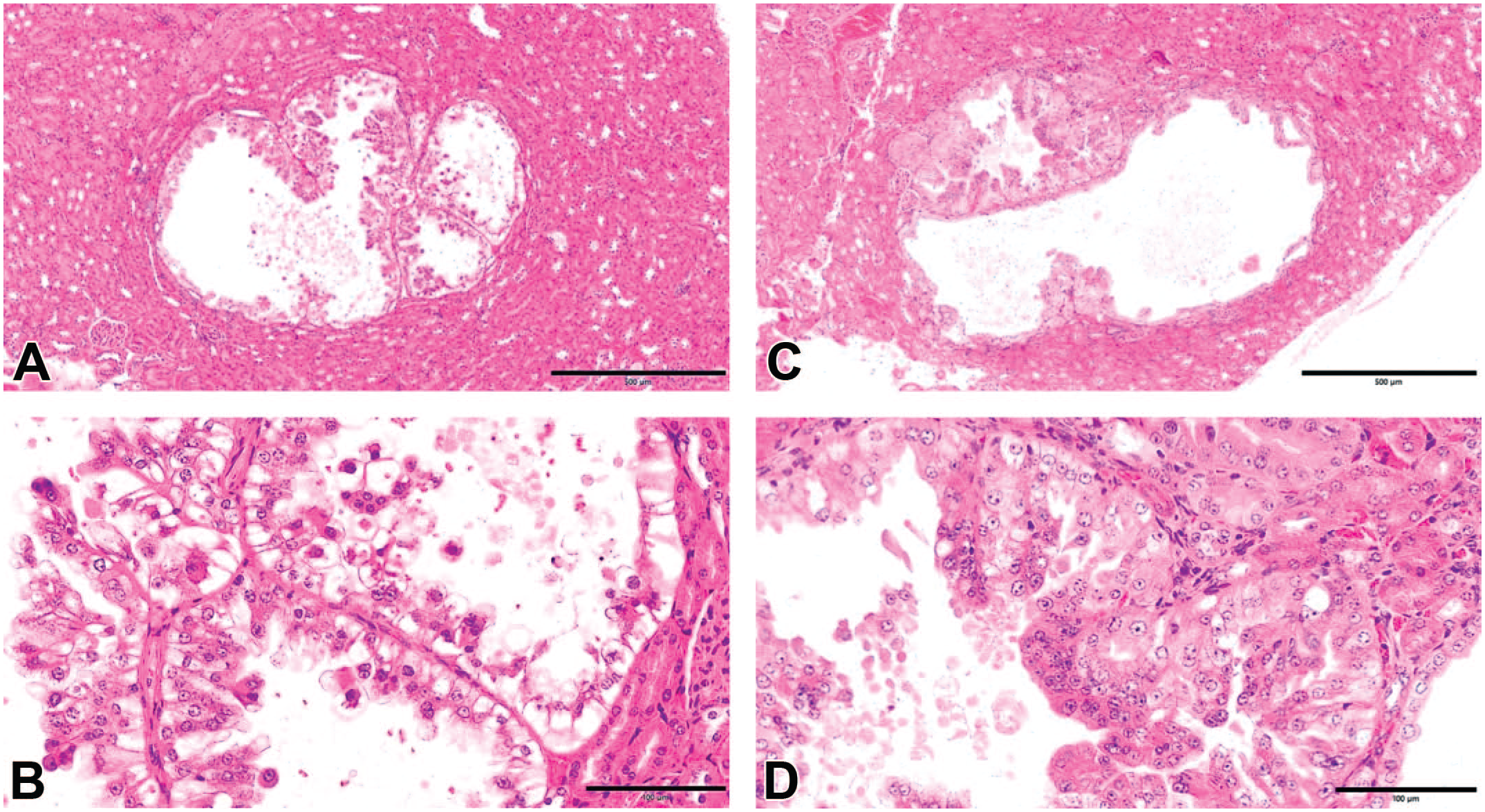
Histopathological features. (A) A renal tumor recognized in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 5X). (B) A renal tumor in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 20X). The tumor was histopathologically diagnosed as adenocarcinoma with clear cytoplasm. (C) A renal tumor recognized in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 5X). (D) A renal tumor in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 20X). The tumor was histopathologically diagnosed as adenocarcinoma with eosinophilic-clear mixed cytoplasm.
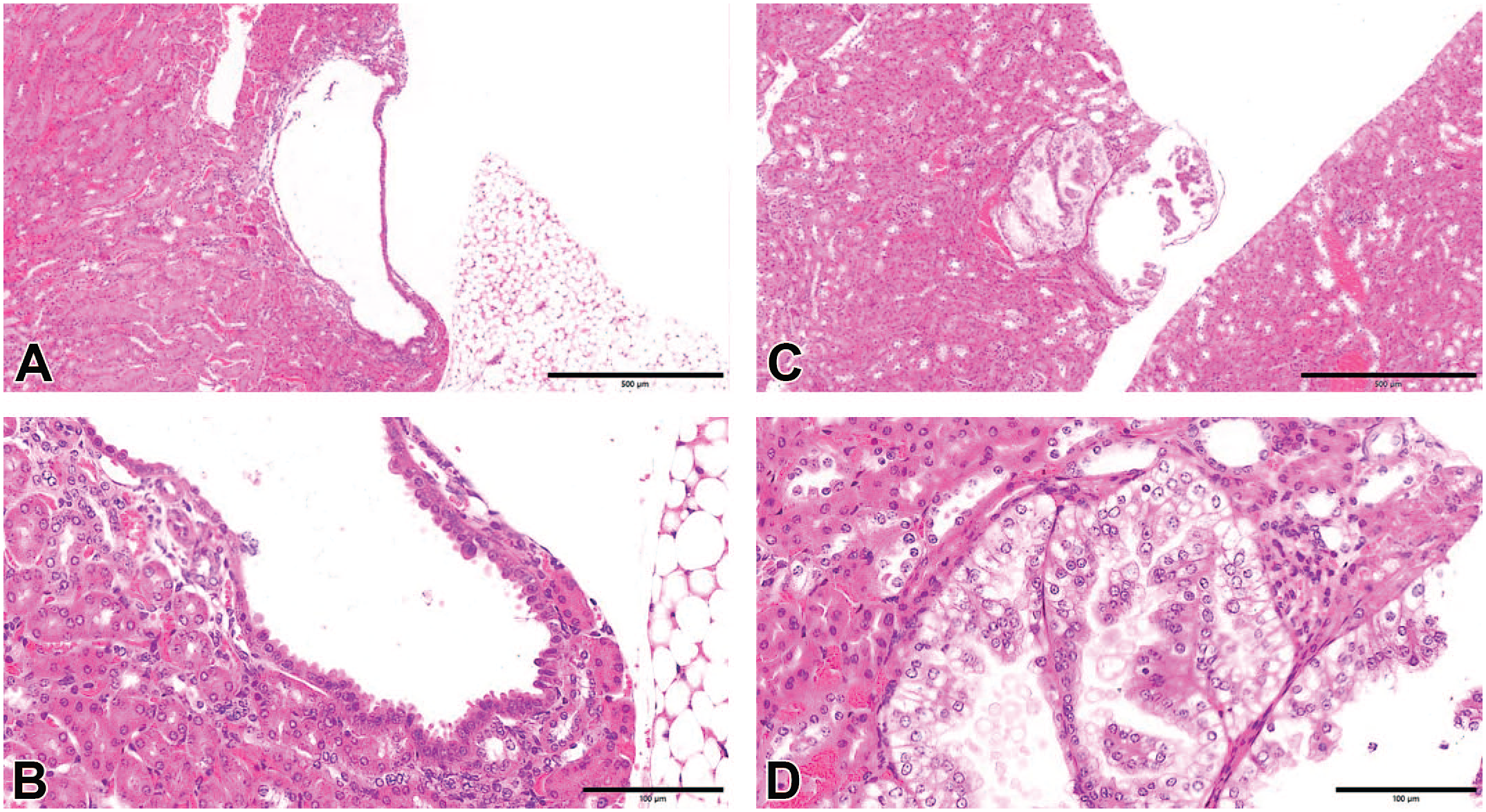
Histopathological features. (A) A renal tumor recognized in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 5X). (B) A renal tumor in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 20X). The tumor was histopathologically diagnosed as adenoma with eosinophilic cytoplasm. (C) A renal tumor recognized in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 5X). (D) A renal tumor in a mouse that survived 182 days after STZ administration (hematoxylin and eosin, original objective 20X). The tumor was histopathologically diagnosed as adenoma with clear cytoplasm.
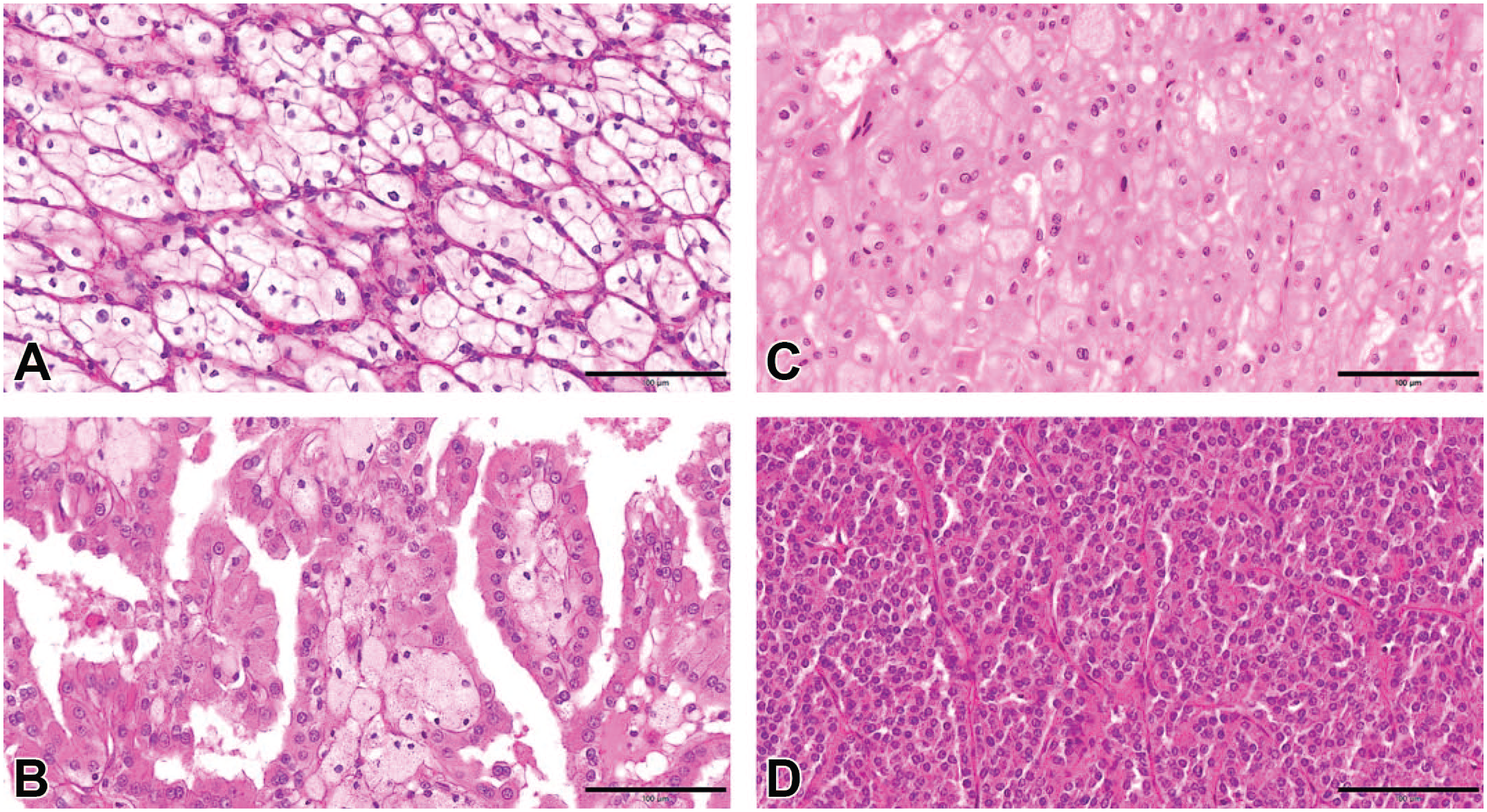
Histopathological features. (A) Representative image of human ccRCC (hematoxylin and eosin, original objective 20X). (B) Representative image of human pRCC (hematoxylin and eosin, original objective 20X). (C) Representative image of human chRCC (hematoxylin and eosin, original objective 20X). (D) Representative image of human renal oncocytoma (hematoxylin and eosin, original objective 20X).
Immunohistochemical Features of STZ-Induced Renal Tumors in Mice: Comparison With Human Renal Tumor Markers
For immunohistochemical analysis and colloidal iron staining of STZ-induced mouse renal tumors, renal tissues from four of the 25 mice that developed renal tumors were used. These four mice were selected because the largest renal tumor in each specimen was histopathologically diagnosed as adenocarcinoma and was of sufficient size. In addition, each specimen contained at least four renal tumors, and the occurrence of tumors in the liver or lungs was also considered. We compared the immunohistochemical profiles of STZ-induced mouse renal tumors with those of various human renal tumor types using antibodies commonly employed in the differential diagnosis of human renal neoplasms. The IHC scores and P values of these markers for STZ-induced mouse and human renal tumors are summarized in Table 4.
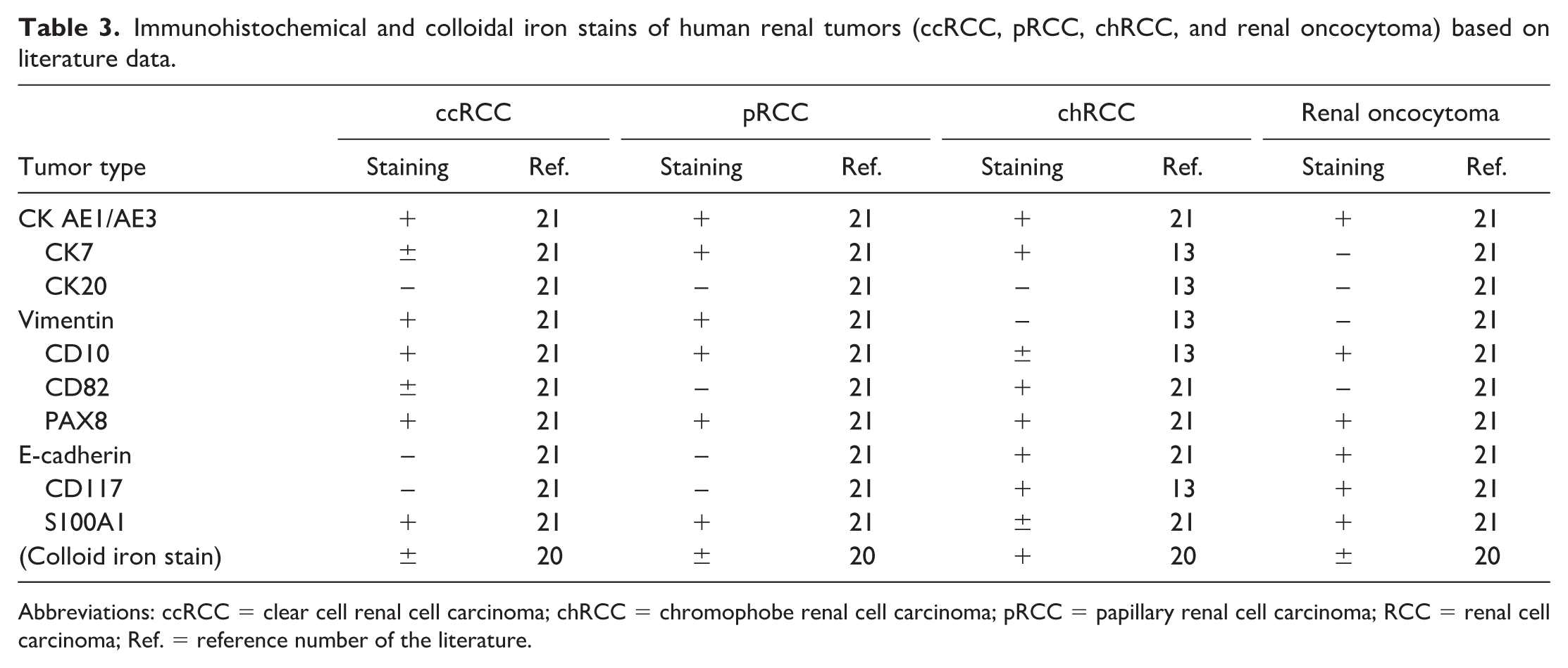
STZ-induced renal tumors in mice were positive for CK AE1/AE3, PAX8, CK7, CD10, CD82, E-cadherin, CD117, and S100A1 and negative for CK20, vimentin, and Hale’s colloidal iron stain (Figure 2 and Table 4). In contrast, the typical immunoprofiles of human renal tumors13,20,21 are as follows (Table 3):
ccRCC. Positive for CK AE1/AE3, vimentin, CD10, PAX8, and S100A1, and negative for CK20, E-cadherin, and CD117.
pRCC. Positive for CK AE1/AE3, CK7, vimentin, CD10, PAX8, and S100A1, and negative for CK20, CD82, E-cadherin, and CD117.
chRCC. Positive for CK AE1/AE3, CK7, CD82, PAX8, E-cadherin, CD117, and Hale’s colloidal iron staining; negative for CK20 and vimentin.
Renal oncocytoma. Positive for CK AE1/AE3, CD10, PAX8, E-cadherin, CD117, and S100A1 and negative for CK7, CK20, vimentin, and CD82.
These known immunoprofiles largely aligned with the findings of our study (Table 4). Collectively, the immunohistochemical features of STZ-induced renal tumors in mice were similar to those of human chRCC and renal oncocytoma.
Immunohistochemical and colloidal iron stains of human renal tumors (ccRCC, pRCC, chRCC, and renal oncocytoma) based on literature data.
Abbreviations: ccRCC = clear cell renal cell carcinoma; chRCC = chromophobe renal cell carcinoma; pRCC = papillary renal cell carcinoma; RCC = renal cell carcinoma; Ref. = reference number of the literature.
Immunohistochemical stains and IHC scores of STZ-induced mice renal tumors and human renal tumors (ccRCC, pRCC, chRCC, and renal oncocytoma) using human renal tumor markers.

Kruskal-Wallis followed by Steel-Dwass multiple comparisons. Reported pairwise P values are Steel-Dwass-adjusted. P < .05 considered significant.
Abbreviations: ccRCC = clear cell renal cell carcinoma; chRCC = chromophobe renal cell carcinoma; PR = positive rate (including weakly positive); pRCC = papillary renal cell carcinoma; STZ mice = renal tumors recognized in CBA/NSlc female mice survived 182 days after STZ administration.
Immunohistochemical Features of STZ-Induced Renal Tumors in Mice: Analysis Using Nephron Segment Markers
Each nephron segment in the kidney exhibits a distinct immunoprofile. The major types of renal epithelial tumors are considered to originate from specific nephron segments and retain their corresponding marker expression. For example, CD10, as a nephron segment marker, is expressed in the proximal convoluted tubules (PCTs) and is commonly expressed in ccRCC and pRCC, whereas S100A1 and kidney-specific cadherin are markers of distal nephron segments that are expressed in chRCC and renal oncocytoma. 21 Tumors originating from collecting ducts may express markers localized in the collecting ducts. However, the expression patterns may vary between mice and humans, even when the same antibodies are used.
To address this, we performed double staining with IHC and PAS to determine the marker localization in both mouse and human renal cortices. PAS staining was positive in the brush borders of the PCT, but negative in the distal convoluted tubules and collecting ducts, 4 allowing accurate identification of nephron segments. The localization of human renal tumor markers within the mouse and human renal cortices is shown in Table 5. STZ-induced mouse renal tumors were generally positive for markers localized in the collecting ducts of the mouse renal cortex (Tables 4 and 5).
Localization of human renal tumor markers in mouse or human renal cortex.
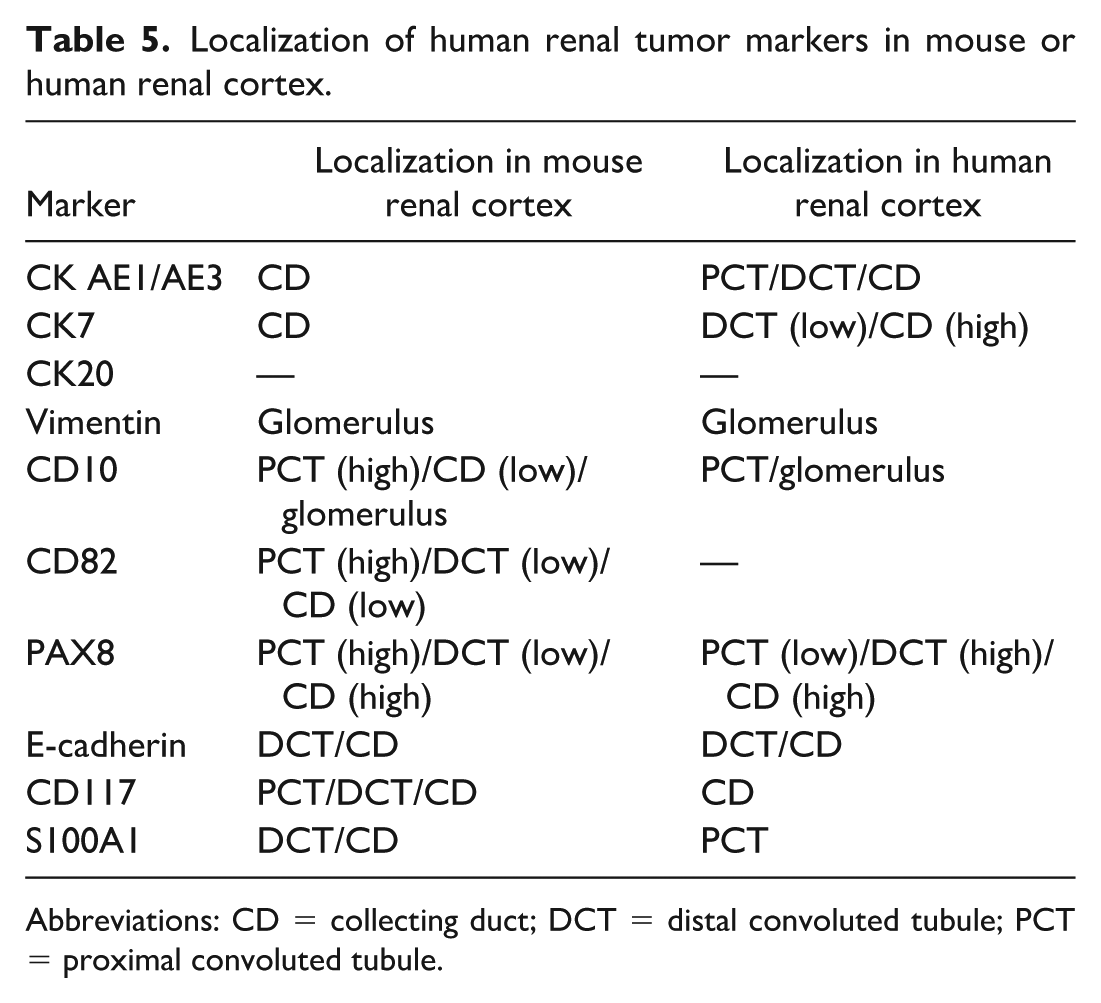
Abbreviations: CD = collecting duct; DCT = distal convoluted tubule; PCT = proximal convoluted tubule.
Furthermore, we evaluated the expression of AQP1, megalin, uromodulin, AQP3, CD15, MUC1, and GLUT-1 as nephron segment markers. The IHC scores and P values of these nephron segment markers for mouse and human renal tumors are presented in Table 6, and their localization in the renal cortex is shown in Table 7. STZ-induced renal tumors in mice were positive for uromodulin, CD15, MUC1, and GLUT-1 and negative for AQP1, megalin, and AQP3. Notably, STZ-induced renal tumors, as well as chRCC and renal oncocytoma, were typically positive for markers localized in the collecting ducts and negative for those localized in the PCT (Figure 6). Conversely, ccRCC and pRCC were positive for PCT markers and negative for collecting duct markers (Tables 6 and 7). These results suggest that STZ-induced renal tumors in mice originate from the collecting duct system, similar to human chRCC and renal oncocytomas. 21
Immunohistochemical stains and IHC scores of STZ-induced mice renal tumors and human renal tumors (ccRCC, pRCC, chRCC, and renal oncocytoma) using renal cortex markers.
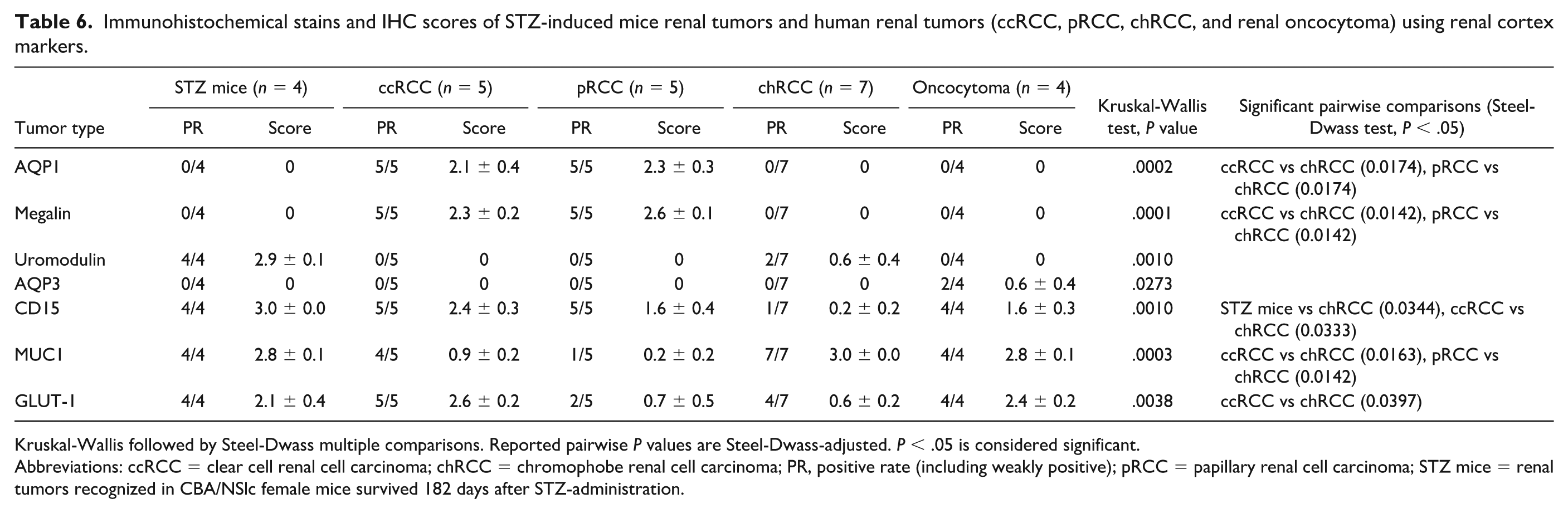
Kruskal-Wallis followed by Steel-Dwass multiple comparisons. Reported pairwise P values are Steel-Dwass-adjusted. P < .05 is considered significant.
Abbreviations: ccRCC = clear cell renal cell carcinoma; chRCC = chromophobe renal cell carcinoma; PR, positive rate (including weakly positive); pRCC = papillary renal cell carcinoma; STZ mice = renal tumors recognized in CBA/NSlc female mice survived 182 days after STZ-administration.
Localization of renal cortex markers in mouse or human renal cortex.
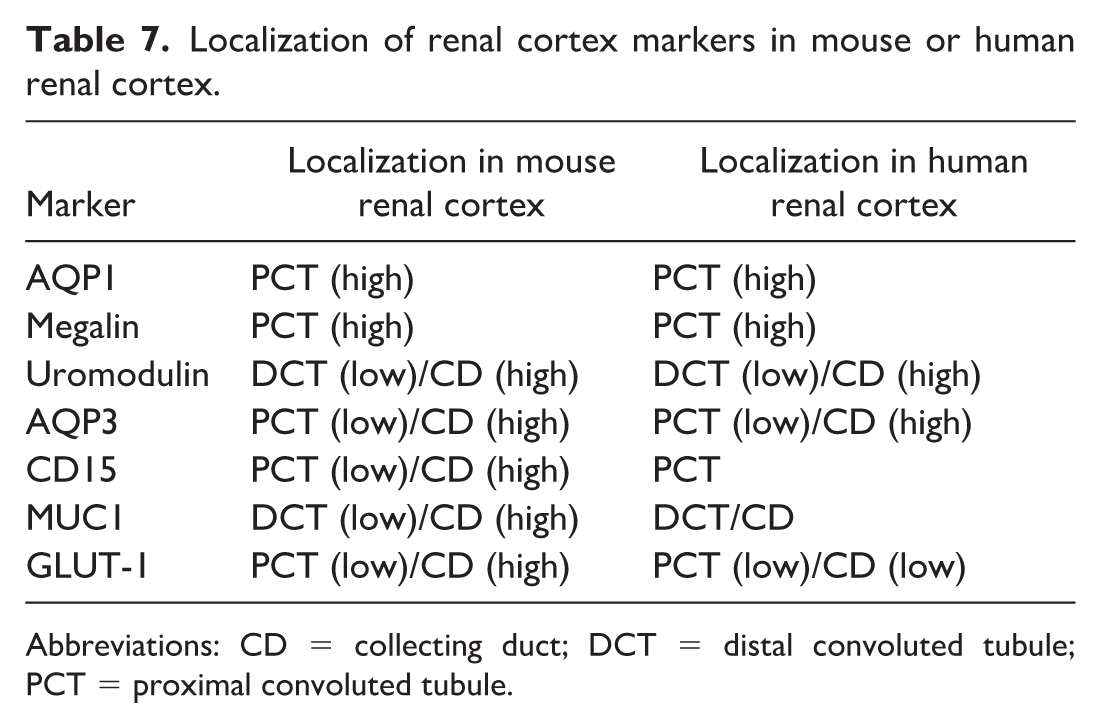
Abbreviations: CD = collecting duct; DCT = distal convoluted tubule; PCT = proximal convoluted tubule.
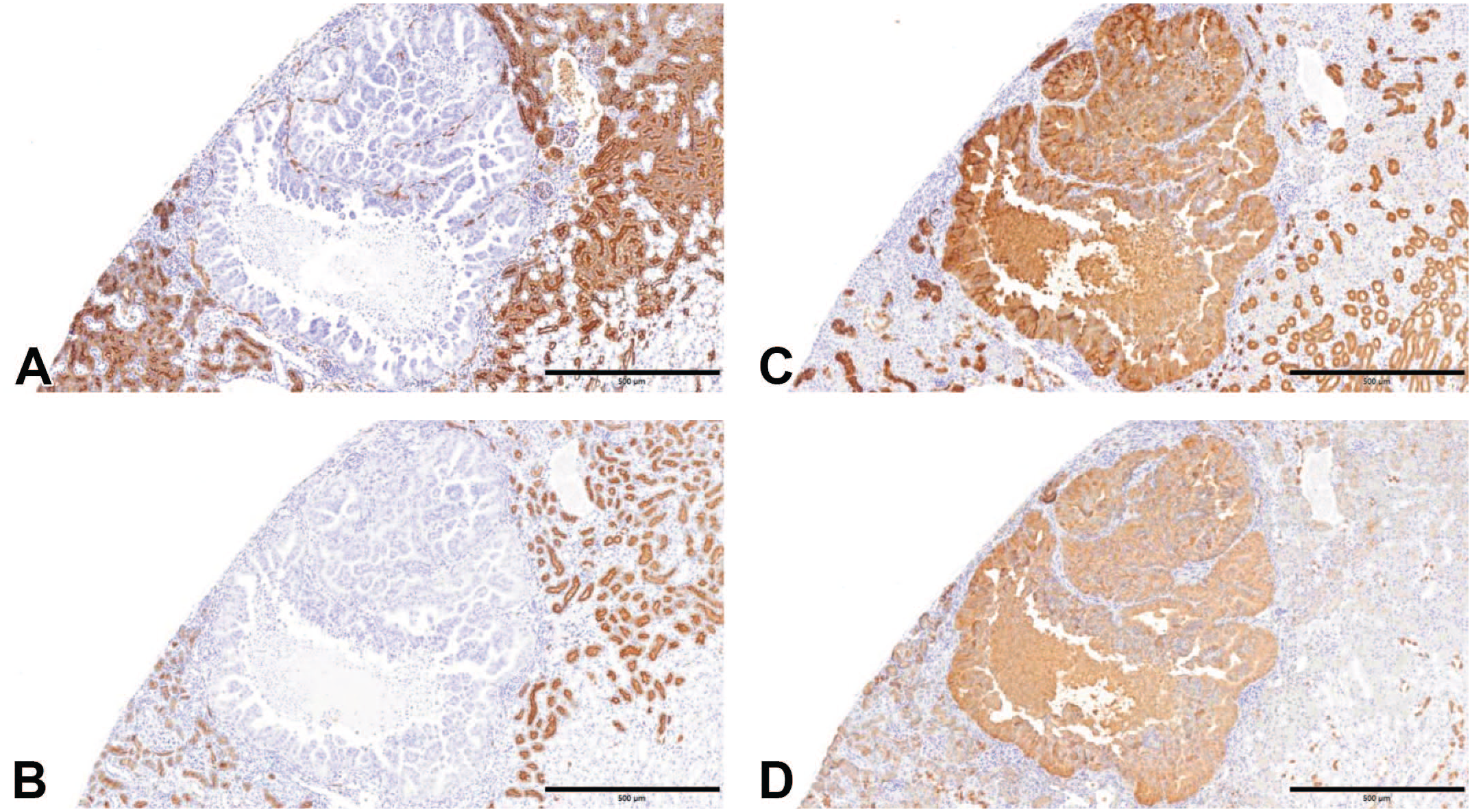
Immunohistochemical features. (A) A renal tumor in a mouse that survived 182 days after STZ administration. AQP1 is positive in mouse normal proximal tubules but negative in the renal tumor (original objective 5X). (B) Megalin is positive in mouse normal proximal tubules but negative in the renal tumor (original objective 5X). (C) Uromodulin is positive in mouse normal distal tubules (low), normal collecting ducts (high), and the renal tumor (original objective 5X). (D) CD15 is positive in mouse normal proximal tubules (low), normal collecting ducts (high), and the renal tumor (original objective 5X).
Discussion
In this study, we evaluated the usefulness of our experimental mouse model of renal tumorigenesis by comparing it with previously reported models. Among the 28 mice euthanized 182 days after STZ administration, the mean and median numbers of renal tumors were 3.4 and 2.5, respectively, and the incidence of renal tumors was 89%, thereby indicating both a sufficient tumor burden and high incidence. The mean and median maximum tumor diameters were 686 and 650 µm, respectively. In Hard’s model, the maximum tumor diameters ranged from 500 to 2000 µm after a maximum rearing period of 80 weeks, 10 while in Miyao’s model, tumors measured approximately 200 µm at 5 months and 450 µm at 9 months post-STZ administration. 15 Therefore, the tumor sizes achieved in our model were considered sufficiently large compared with those reported in earlier studies. Importantly, only 2 of 30 mice died before 182 days after STZ administration, highlighting the stability and reliability of our experimental system. Taken together, these findings indicate that maintaining 6-week-old CBA/NSlc female mice for 182 days after STZ administration reliably validates a reproducible and robust mouse model of STZ-induced renal tumorigenesis.
Two distinct histopathological types of STZ-induced mouse renal tumors were identified: tumors with pale cytoplasm and those with eosinophilic cytoplasm. No notable immunohistochemical differences were observed between these two types of tumors, suggesting a common cellular origin. Compared with human renal tumors, mouse tumors with a pale cytoplasm resemble either ccRCC or the classical subtype of chRCC, whereas eosinophilic tumors resemble the eosinophilic subtype of chRCC or renal oncocytoma. 5 Histopathological assessment alone is insufficient for differentiating human renal tumors; thus, immunohistochemical assessment is crucial, especially for less common tumors, such as chRCC and renal oncocytoma. Although chRCC is malignant and renal oncocytoma is benign, both tumors are believed to originate from intercalated cells of the renal collecting duct. 1 They share immunohistochemical features: both are positive for CD117 and E-cadherin and negative for vimentin, in contrast to ccRCC and pRCC, which are considered to originate from PCT. 21 STZ-induced mouse renal tumors exhibited immunohistochemical profiles consistent with those of chRCC and renal oncocytomas. Specifically, the tumors were positive for CK7 and CD82 (consistent with chRCC), CD10 and S100A1 (consistent with renal oncocytoma), and CK AE1/AE3, PAX8, E-cadherin, and CD117, and were negative for CK20 and vimentin.13,16,21 Although no human tumor precisely matches the immunohistochemical profile of STZ-induced mouse renal tumors, these similarities strongly suggest that these tumors most closely resemble human chRCCs.
Immunohistochemical staining for nephron segment markers revealed that STZ-induced mouse renal tumors tended to express markers that were highly localized in the collecting ducts of the mouse renal cortex, similar to human chRCCs and renal oncocytomas. Although AQP3, a principal cell marker of the human collecting duct, 7 was absent in our model, the localization of nephron markers differed slightly between humans and mice. Nevertheless, the tendency of STZ-induced mouse renal tumors to express collecting duct markers supports their similarity to human chRCCs. In addition, the lack of expression of AQP1 and megalin, PCT markers, further emphasizes their distinction from PCT-derived tumors, such as ccRCC or pRCC. STZ exhibits cytotoxicity in PCT cells via p53-pathway activation in acute settings. 17 In contrast, STZ-induced mouse renal tumors in our experiment expressed distal nephron markers and lacked PCT markers. This apparent discrepancy suggests that tumorigenesis in our model may reflect STZ’s genotoxicity (direct DNA damage) rather than cytotoxic injury to specific nephron cells. Alternatively, injury-repair dynamics could create a permissive microenvironment for transformation of distal nephron cells. Time-course injury mapping and genomic profiling would be valuable to clarify the pathogenesis of STZ-induced mouse renal tumorigenesis. Negative colloidal iron staining, traditionally used in diagnosing chRCC, should be interpreted cautiously; although colloidal iron positivity has been associated with chRCC, its diagnostic specificity is not necessarily high, 20 and data regarding colloidal iron staining in mouse renal tumors remain limited. Therefore, although our results do not fully contradict the similarity to chRCC, they highlight important considerations for comparative studies of mouse and human renal tumors.
Previous studies on STZ-induced mouse renal tumors have performed ultrastructural comparisons between mouse and human renal tumors, reporting that ultrastructures resemble those of human chRCC. 6 However, ultrastructural analysis was not performed in the present study. In addition, no genetic comparisons have been reported between STZ-induced mouse renal tumors and human renal tumors. Ultrastructural and genetic analyses of mouse renal tumors obtained in the present experimental model may further clarify the similarity between STZ-induced mouse renal tumors and human chRCC. Rat models of chemical tumorigenesis have also been investigated, and experiments similar to those in the present study using rats may facilitate the development of a novel STZ-induced renal tumorigenesis model. In addition to chemical tumorigenesis models, various other mouse models of renal tumorigenesis, such as genetically engineered models, have been established. 19 Comparing the renal tumors obtained from these models with STZ-induced mouse renal tumors may further aid the development of experimental models. Although chRCC is less common than ccRCC in humans, 21 the STZ-induced mouse renal tumorigenesis model described here could serve as a valuable platform for understanding and advancing research on the biology and treatment of chRCC.
Conclusion
In conclusion, maintaining 6-week-old female CBA/NSlc mice for 182 days after STZ administration successfully validated a reliable mouse model of STZ-induced renal tumorigenesis. To the best of our knowledge, this is a robust mouse model of renal chemical tumorigenesis. Although we did not perform an ultrastructural or genetic comparison between STZ-induced mouse renal tumors and human renal tumors in this study, the renal tumors generated in this model exhibited morphological and immunohistochemical features resembling those of human chRCC, suggesting their potential utility in future studies on human renal tumors, particularly chRCC.
Footnotes
Acknowledgements
Abbreviations
STZ, streptozotocin; IHC, immunohistochemistry; chRCC, chromophobe renal cell carcinoma; ccRCC, clear cell renal cell carcinoma; pRCC, papillary renal cell carcinoma; H&E, hematoxylin and eosin; PCT, proximal convoluted tubule.
Author Contributions
Authors contributed to conceptualization (YF, HM, TT, and YH), methodology (YF, HM, TT, and YH), software (YF), data curation (YF), formal analysis (YF), validation (YF and YH), investigation (YF and YH), visualization (YF and YH), project administration (YF and YH), resources (YF and YH), writing—original draft (YF), writing—review and editing (YF, HM, TT, and YH), supervision (YH), and funding acquisition (YH).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosure of Generative AI or AI-Assisted Technologies Used in the Writing of the Manuscript
ChatGPT and DeepL Translate were used to enhance the readability of the manuscript.
Ethics Approval
This study was conducted in accordance with the Basic Guidelines for the Conduct of Animal Experiments at Research Institutes (Ministry of Education, Culture, Sports, Science, and Technology, Japan).
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
