Abstract
Introduction: Sarcomatoid differentiation has been reported in approximately 8% of chromophobe renal cell carcinoma (RCC) and is associated with a worse prognosis. We aim to describe the clinicopathologic and molecular findings of chromophobe RCC with sarcomatoid differentiation. Methods: Surgical pathology database was searched to identify chromophobe RCC with sarcomatoid differentiation from January 2015 to December 2021. Results: Five patients were diagnosed with chromophobe RCC with sarcomatoid differentiation. The median age at the time of diagnosis was 57 years (range 51-61 years). Three patients died after median follow-up of 12.1 months (range 1.6-18.2 months). The median tumor size was 10.7 cm (range 5.6-13.6 cm). The median percentage of sarcomatoid component was 60% (range 10-90%), and the median percentage of necrosis was 30% (range 10-50%). One tumor demonstrated osteoid formation. PAX8, keratin 7, KIT (CD117), and Hale colloidal iron were positive in the epithelial component, whereas the sarcomatoid component was positive for vimentin, CD10, and high Ki67 proliferative index. Molecular testing was performed in three specimens: all were TP53 mutated and microsatellite stable. One aggressive tumor had RB1 frameshift mutation and copy number gains for TERT and CUL4A. Conclusion: Chromophobe RCC with sarcomatoid differentiation is a rare entity with aggressive behavior. Percentage of sarcomatoid component, necrosis, and the occurrence of metastasis is associated with worse prognosis. Molecular profiling reveals frequent TP53 mutation. While TERT promoter mutation has no prognostic implication, FLCN inactivation may be associated with a less aggressive course. The clinical significance of RB1 loss is unclear.
Introduction
Sarcomatoid renal cell carcinoma (RCC), although not a true subtype of RCC, represents dedifferentiation of any type of RCC. It is characterized by the presence of high-grade sarcoma-like spindle cell proliferation admixed with a conventional RCC component. Sarcomatoid RCC is clinically aggressive with dismal prognosis. Current therapies for sarcomatoid RCC is limited and not highly effective, regardless of the original tumor histology.
Chromophobe RCC is the third most common subtype of RCC, representing 5-10% of all kidney tumors. 1 Compared to clear cell RCC, chromophobe RCC is associated with favorable outcomes, lower risk of metastasis, and a 10-year survival rate of 80-90%. 2 However, sarcomatoid differentiation is estimated to occur in approximately 8% of chromophobe RCC and is associated with high metastatic risk and worse prognosis. 3 Here, we present the clinicopathologic and molecular findings of five patients presenting with chromophobe RCC with sarcomatoid differentiation.
Methods
Nephrectomies with a diagnosis of chromophobe RCC with sarcomatoid differentiation were retrieved from a search of the surgical pathology database between January 2015 and December 2021. Patient's demographics, clinical presentation, pathologic parameters, treatment, and follow-up were recorded. Available slides, including hematoxylin and eosin (H&E), special stain Hale colloidal iron (Artisan, Argilent®), and the following immunohistochemistry (IHC) were reviewed: PAX8 (Roche Ventana®, MRQ-50, pre-dilute), keratin 7 (Roche Ventana®, SP52, pre-dilute), KIT (CD117; Dako®, c-kit, polyclonal rabbit, 1/200 dilution), vimentin (Roche Ventana®,V9, pre-dilute), CD10 (Leica®, 56C6, pre-dilute), and Ki67 (Roche Ventana ®, 30–9, pre-dilute).
Molecular analyses were performed in three tumors: #3: TEMPUS xT® (648 genes, hybrid capture next generation sequencing, detection of single nucleotide variants, indels and translocations); #4 and #5: Foundation ONE CDx® (324 genes, hybrid capture next generation sequencing, detection of substitutions, indels, copy number alterations, and select gene rearrangements).
Results
Clinicopathologic findings for each patient are summarized in
Clinicopathologic Features of the Five Patients.
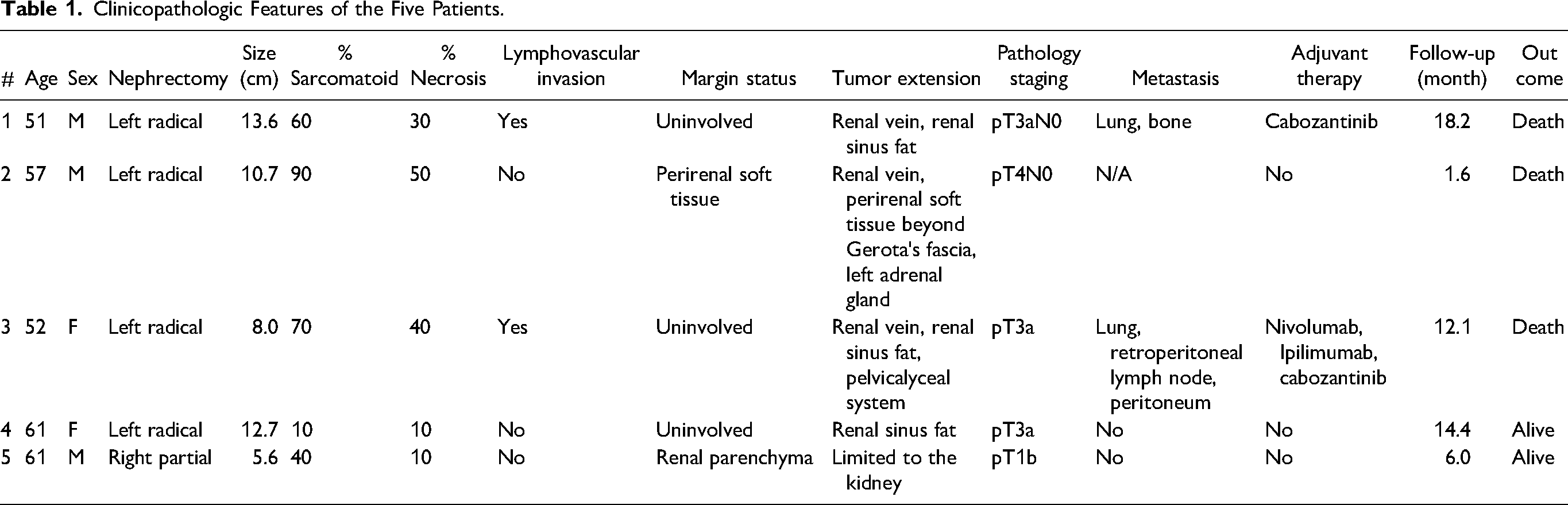
Radical nephrectomy was performed in four patients (80%), whereas one patient underwent a partial nephrectomy (#5). Four tumors (80%) occurred in the left kidney. The median tumor size was 10.7 cm, ranging from 5.6 to 13.6 cm. Only one patient (#5) had a pathologic staging less than pT3 (pT1b). Conversely, tumor extension into perinephric tissue beyond Gerota's fascia and direct invasion of the adrenal gland was identified in one specimen (#2), and that tumor was staged as pT4 (Figure 1a). Lymphovascular invasion, defined as tumor cells invading vessels other than renal vein and its segmental branches, was identified in two specimens (40%). Renal vein invasion was identified in three specimens (60%), renal sinus fat invasion was identified in three specimens (60%), and extension into the pelvicalyceal system was identified in one specimen (#3). Margins were involved by tumor in two specimens (#2: perirenal soft tissue; #5: renal parenchyma). The median sarcomatoid component was 60%, ranging from 10% to 90% (Figure 1b). Necrosis was identified in all specimens, with a median percentage of 30% (range 10-50%) (Figure 1c). One specimen (#4) had heterologous component with osteoid formation (Figure 1d).
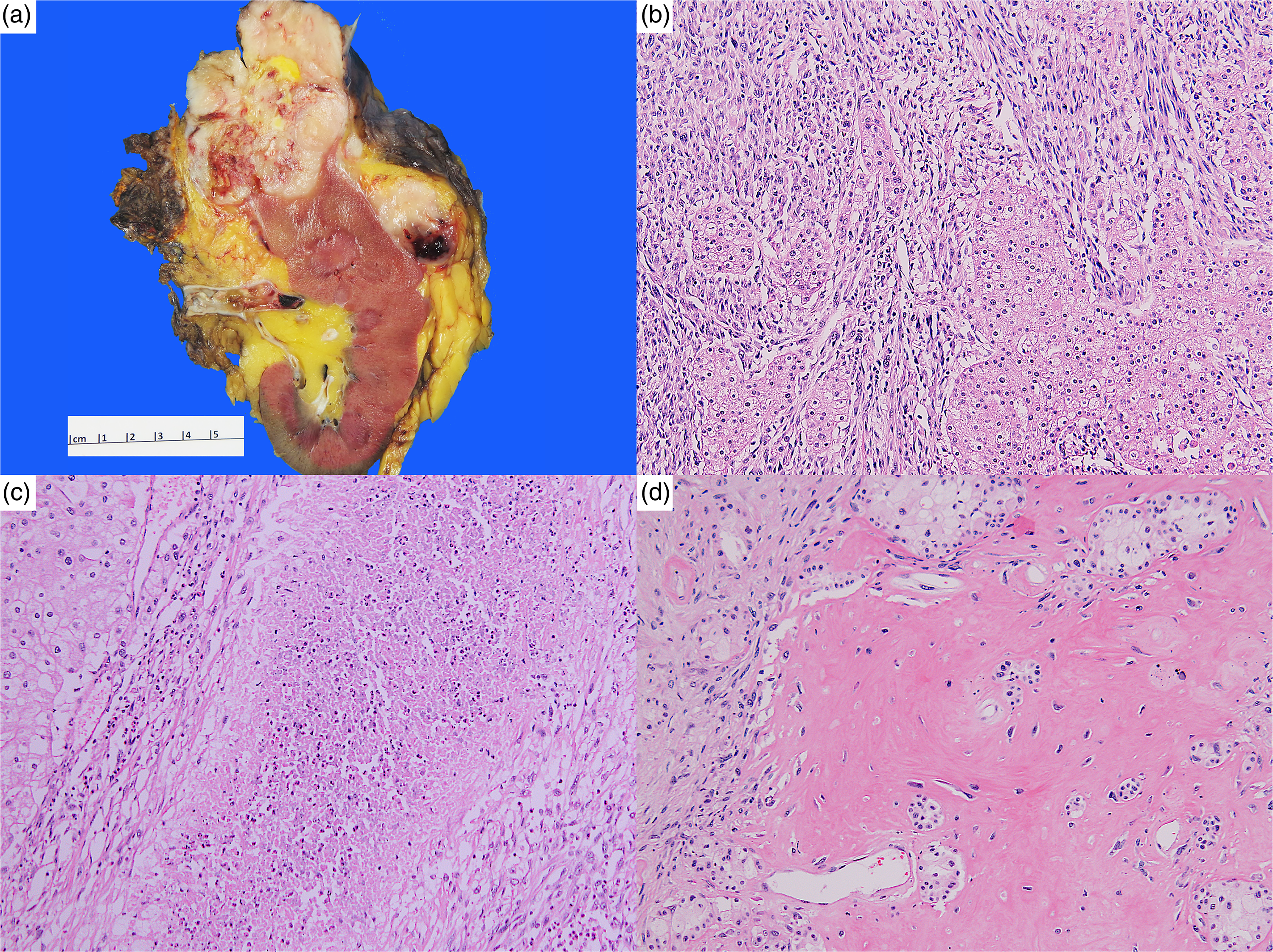
(a) Specimen #2, macroscopic appearance, including direct adrenal gland invasion and extension into renal vein; (b) Specimen #2, epithelial component and adjacent sarcomatoid differentiation; (c) Specimen #2, sarcomatoid differentiation with coagulative necrosis; (d) Specimen #4, osteoid formation.
After a median follow-up of 12.1 months (range 1.6-18.2 months), two patients were alive (40%). Patient #2, who presented with the highest percentage of sarcomatoid component (90%) and necrosis (50%), had a very aggressive disease course. He was not eligible for adjuvant therapy and died after 1.6 months from the time of diagnosis. Metastatic disease was identified in two patients (40%), with lung being the most affected organ. Both patients with metastatic disease received adjuvant therapy: one patient (#1) received cabozantinib and the other (#3) received nivolumab, ipilimumab, and cabozantinib. However, both died after 18.2 and 12.1 months of follow-up, respectively.
IHC and special stains were performed in four specimens (80%). PAX8, keratin 7, KIT (CD117), and Hale colloidal iron were consistently positive in the epithelial component. Areas of sarcomatoid differentiation showed reduced PAX8 and keratin expression, as well as positivity for vimentin and CD10. Additionally, Ki67 proliferative index was markedly higher in the sarcomatoid areas. Figure 2 illustrates the morphology, IHC, and special stain performed on specimen #5.
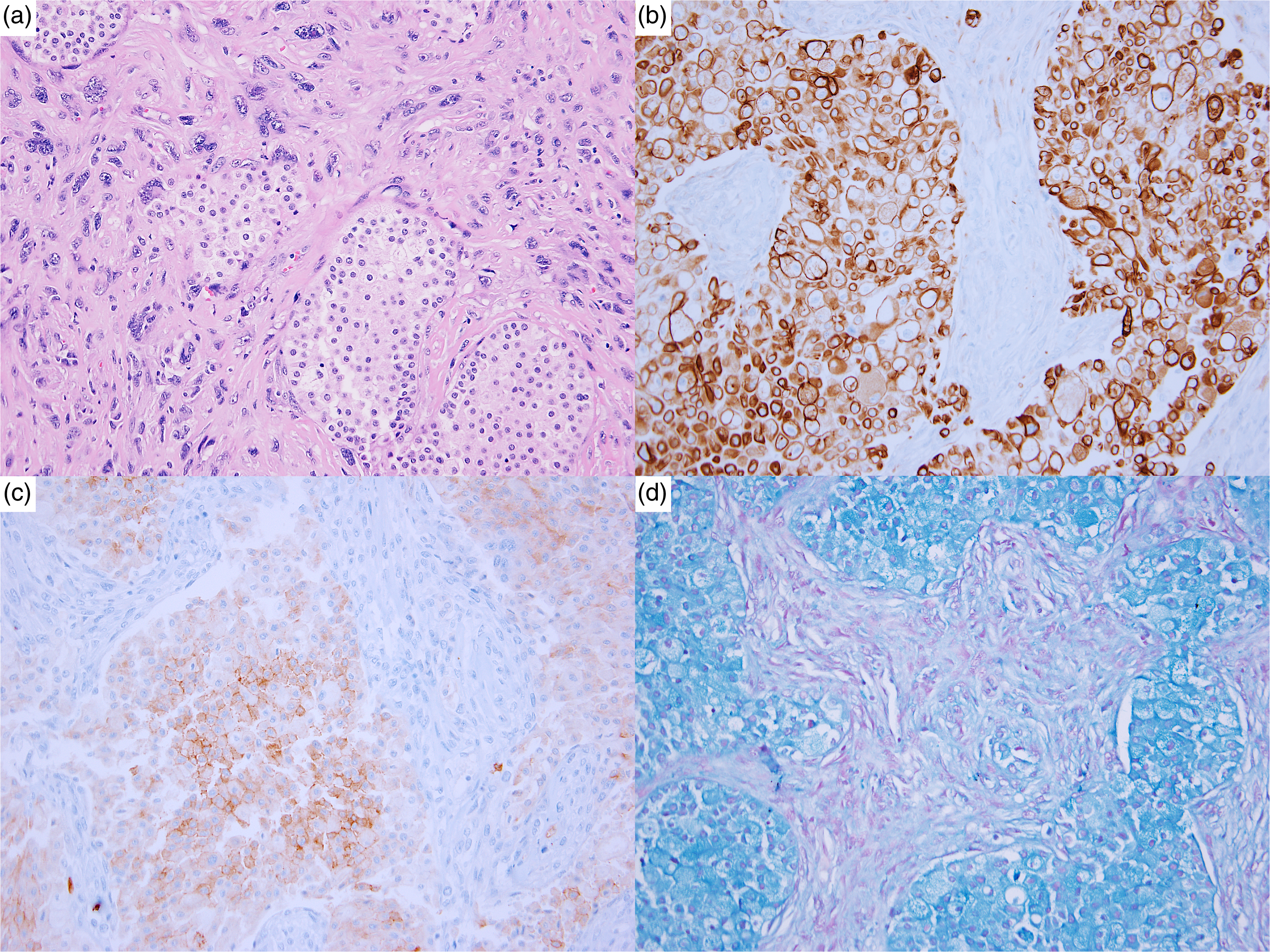
Specimen #5: (a) Epithelial components intermixed with areas of sarcomatoid differentiation; (b) Keratin 7, diffusely positive in epithelial component and patchy positive in sarcomatoid component; (c) KIT (CD117), positive in epithelial component and negative in sarcomatoid component; (d) Hale colloidal iron, positive in epithelial component and negative in sarcomatoid component.
Comprehensive next-generation sequencing was performed in three specimens (#3, #4, and #5): all were TP53 mutated and microsatellite stable. Patient #3 (pT3a), subsequently diagnosed with metastasis to retroperitoneal lymph nodes, peritoneum, and lungs, had RB1 frameshift mutation and copy number gains for TERT and CUL4A. This patient died 12.1 months after diagnosis. Patient #4 (pT3a) had no other relevant molecular finding, and she was alive after 12.1 months. Patient #5 (pT1b) had FLCN inactivating mutation and TERT promoter mutation. He was alive after 6 months of follow-up and did not present with any features suggestive of Birt-Hogg-Dubé syndrome, such as cutaneous hamartomas, multiple kidney neoplasms, lung cysts, or spontaneous pneumothorax.
Discussion
Sarcomatoid transformation has been described in multiple renal malignancies, and it is believed to represent a final dedifferentiation pathway for kidney epithelial neoplasms. 4 The presence of sarcomatoid component in chromophobe RCC is characterized by a variable amount of anaplastic spindle cell population. Immunoreactivity of the sarcomatoid component differs from the epithelial component with decreased expression for keratins and positivity for vimentin. 5 Sarcomatoid differentiation occurs in approximately 8% of chromophobe RCC and is an important histologic prognostic factor being associated with high risk of metastatic disease. 3
Among sarcomatoid chromophobe RCC, the percentage of geographic necrosis and presence of lymphovascular invasion are key features of a highly aggressive malignancy. 6 In our series, one patient (#2) presented with multiple poor prognostic factors (90% sarcomatoid differentiation, 50% necrosis, renal vein invasion, extension beyond Gerota's fascia, and direct tumor invasion into the adrenal gland) and died 1.6 months after the diagnosis. Heterologous components, especially osseous formation, have also been described in association with sarcomatoid chromophobe RCC, but their clinical significance is uncertain due to the rarity of this phenomenon. 6 Only one specimen (#4) had heterologous component (osteoid formation). This tumor had only 10% of sarcomatoid differentiation and 10% of necrosis. The patient's disease course was mild, and no metastatic disease was identified. No adjuvant therapy was required, and the patient is currently alive with no signs of recurrence 15.1 months following radical nephrectomy. Interestingly, our case series showed a left-sided preference (80%). The largest case series of sarcomatoid chromophobe RCC to date (14 patients, Lauer et al. 4 ) reports 64% of left-side tumors. Taking together both series, 13 of 19 (68%) of tumors were located on the left kidney. The significance of this finding is unknown.
Multiple previous reports have associated chromophobe RCC with losses of chromosomes 1, 2, 6, 10, 13, 17, 21, and Y chromosome. 7 In 2007, Brunelli et al 8 reported the cytogenetic findings of six chromophobe RCC with sarcomatoid differentiation. They reported a distinct genetic profile compared to classic chromophobe RCC, including gains of chromosomes 1, 2, 6, 10, and 17. Recently, comprehensive genomic analyses of chromophobe RCC showed a low somatic mutation rate, with TP53 and PTEN being the most frequently mutated genes, especially in those with metastatic disease. 7
In our cohort, next generation sequencing was performed in three specimens (#3, #4, and #5). Tumor #3 (pT3a, with metastasis to retroperitoneal lymph nodes, peritoneum, and lungs) was microsatellite stable but demonstrated TP53 missense mutation, RB1 frameshift mutation, and copy number gains for both TERT and CUL4A. This patient died 12.1 months after diagnosis. Tumor #4 (pT3a) was microsatellite stable and presented with only TP53 mutation. The patient was alive after 12.1 months. Tumor #5 (pT1b) was microsatellite stable but had TP53 mutation, FLCN inactivating mutation, and TERT promoter mutation. This patient was alive after 6 months of follow-up.
As previously mentioned, TP53, along with PTEN, are the most frequent mutations found in chromophobe RCC, regardless of the presence of sarcomatoid differentiation. The prevalence of TP53 mutation ranges from 32% to 64%. 9 TP53 mutation was seen in all specimens in which molecular studies were performed. TERT copy number gain was seen in patient #3, who had an aggressive clinical course. On the other hand, TERT promoter mutation was found in patient #5 and the patient had a mild course, treated with partial nephrectomy. Even though TERT promoter mutation is correlated with poor prognosis in clear cell RCC, 10 it does not appear to have the same impact when found in chromophobe RCC. TERT promoter mutation has been detected in 6-12% of chromophobe RCC without prognostic significance. 9 RB1 loss has been described as a useful marker to distinguish chromophobe RCC from renal oncocytoma. 11 Its clinical significance in patient #3 is unknown. Finally, patient #5 also presented with FLCN inactivation. This is classically associated with oncocytic tumors in Birt-Hogg-Dubé syndrome, 12 in which the renal tumors are considered to be less aggressive. However, the current patient did not have any other features suggestive of Birt-Hogg-Dubé syndrome, including cutaneous hamartomas, multiple renal tumors, pulmonary cysts, or spontaneous pneumothorax. The good prognosis, in this case, may be attributed to the incidental discovery of the tumor and diagnosis at an early stage of the disease.
The presence of sarcomatoid component may influence the selection of appropriate immunotherapy with checkpoint inhibitors. 13 It has been demonstrated that treating sarcomatoid RCC with immuno-oncology agents (nivolumab + ipilimumab) confers significant improvement in long-term survival as compared to sumatinib. 14 In our cohort, one patient (#3) was treated with this approach but did not survive. In the near future, molecular profiling of sarcomatoid RCC may direct personalized therapy. 15
Conclusion
Chromophobe RCC with sarcomatoid differentiation is a rare entity with aggressive behavior. Percentage of sarcomatoid component and necrosis appears to be associated with prognosis, as well as the occurrence of metastasis. Molecular profiling reveals frequent TP53 mutation. While TERT promoter mutation has no prognostic implication, FLCN inactivation may be associated with a less aggressive course, but more definitive conclusions are limited by the small sample size. The clinical significance of RB1 loss seen in an aggressive tumor is unclear.
Footnotes
Author's Contributions
A Gama: project development, data collection and analysis, manuscript writing and editing.
H Xu: project development, data collection, manuscript editing.
XJ Yang: project development, manuscript editing.
B Choy: project development, data collection and analysis, manuscript writing and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
