Abstract
Knee osteoarthritis (OA) poses a significant health care burden globally, necessitating innovative therapeutic approaches. CCoat, a novel poly(2-[methacryloyloxy]ethyl phosphorylcholine) (pMPC)ylated liposome device, protects the cartilage surface of the joint from mechanical wear through an entropy-favored process. Two preclinical studies were performed to explore the safety of CCoat following repeated intra-articular (IA) injections into the knee joint (i.e., femorotibial joint) in Sprague-Dawley rats. The studies involved 2 or 3 IA injections, at an interval of 2 or 3 weeks, and an observation period of 1 or 13 weeks after the last injection. Assessments included clinical, histopathological, and immunofluorescent evaluations. In study 1, no mortality or abnormal clinical signs occurred. At 1 week post last injection, histopathology revealed minimal vacuolated macrophages beneath the synovial membrane, predominantly M2-like, indicating a nonadverse response. Immunofluorescent staining supported M2-like macrophage predominance. Study 2 confirmed these findings with no systemic effects over 13 weeks. Statistical analyses indicated no significant differences in body weight, clinical pathology, or organ weights compared with controls. Results affirming the safety of pMPCylated liposomes following repeated IA injections in rat. This novel lubricant coating approach shows promise in OA therapy, with this safety assessment supporting its potential clinical application.
Keywords
Introduction
Knee osteoarthritis (OA) is a widespread, long-term condition characterized by the gradual development of degenerative damage in the articular cartilage or secondary hyperostosis (e.g., femoral condyle osteophyte, ossification of the patellar tendon, enthesopathy of the patellar ligament anterior to the patella and at the tibial tuberosity and calcification of the posterior knee capsule).9,12,17,26,46 Symptoms of knee OA encompass joint pain, swelling, stiffness, and limitations in joint movement. Knee OA impacts the three compartments of the knee joint—medial, lateral, and patellofemoral. Typically, this condition progresses gradually over a period of 10 to 15 years, significantly disrupting daily activities. With advancing age, bilateral knee OA becomes increasingly prevalent, leading to the majority of affected individuals eventually exhibiting radiographic evidence of the disease in both knees.6,19,27 In 2020, the global prevalence of knee OA was estimated at 16% among individuals aged 15 years and older, rising to 22.9% among those aged 40 years and above. Notably, the prevalence of knee OA was higher among women, at 21.7%, compared with 11.9% in men. 9
The diagnosis of OA is primarily based on clinical history and physical examination findings, with radiographic confirmation often obtained through X-rays. OA imposes a significant burden on health care systems, encompassing both direct and indirect costs. Direct costs include surgical interventions such as total joint replacements, as well as nonsurgical management options like pain medications, physical therapy, routine physician visits, and intra-articular (IA) injections. Indirect costs arise from lost workplace productivity, early retirement, and disability. Furthermore, OA severely affects an individual’s quality of life. The direct costs associated with OA vary depending on the joint involved, disease severity, treatment options, the health care system of the country, and the overall health status of the individual. Factors such as aging populations and increasing obesity rates are associated with higher health care costs related to OA.21,22,31 The progression of knee OA is influenced by several risk factors, including advancing age, female sex, obesity, systemic and local joint inflammation, genetics, diet (e.g., low level of vitamins D, C, and K), as well as injury and abnormal loading of the joints.2,3,9,32 Extensive literature supports the critical role of inflammatory mediators in initiating and perpetuating the OA process. These mediators originate both locally from joint cells and systemically from other tissues, such as adipose tissue. Once released, they enter the bloodstream and reach the joint through the subchondral bone vasculature, contributing to the progression of OA. 4 The concept of “low-grade” joint inflammation and its regulatory mechanisms is gaining acceptance as critical not only to joint pain and disability in OA but also to joint trauma and the biomechanical damage of joint tissues. Persistent synovitis and subchondral bone damage are considered major contributors to joint destruction, especially in knee OA. The association of meniscal damage with OA progression underscores the meniscus’s biomechanical and potential inflammatory roles in affected joints. Furthermore, the synovium is a significant player in the “low-grade” inflammation process in OA, with evidence supporting interaction between articular cartilage, subchondral bone, and synovium. Synovial cells, particularly type A macrophage-like synoviocytes, are likely the primary sources of pro-inflammatory mediators within the joint. 29
In 2013, following a meeting of the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO), a comprehensive review of biomarker research for knee OA was published. The review concluded that none of the investigated biomarkers had demonstrated sufficient evidence to guide clinical trials or to be used in clinical settings. Building on this, a systematic review was conducted in 2021, which searched MEDLINE and Embase databases for studies published from August 2013 to May 2018. This review aimed to compile a comprehensive list of candidate molecular biomarkers for knee OA post the 2013 ESCEO findings. It included a total of 80 studies and identified 89 statistically significant molecular biomarkers. Notably, the C-telopeptide of type II collagen (CTXII), a byproduct of articular cartilage breakdown, was highlighted across multiple studies for its ability to predict the progression of knee OA in both urine and serum. 8
The crucial aspect in treating OA involves preventing and managing the progression of the disease, restoring knee joint function, and alleviating pain. 46 Management options for knee OA can be categorized into nonpharmacological, pharmacological, procedural, and surgical approaches. Nonpharmacological treatments that provide short-term pain relief include physical therapy—such as muscle strengthening and range-of-motion exercises—biomechanical interventions, weight loss for overweight or obese patients, and therapeutic ultrasound. Pharmacological therapy typically involves analgesics and anti-inflammatory agents. The procedural category comprises IA injections of substances like corticosteroids, hyaluronic acid (HA), and platelet-rich plasma (PRP), which primarily aim to provide symptomatic short-term relief and improvements in pain and function. For severe grades of knee OA, joint replacement surgery is considered the gold standard, although it is associated with a higher rate of complications and an increased risk of requiring revision surgery. 40 Among these, articular injection of compounds stands out as a particularly significant approach. Intra-articular therapies offer several physiological and practical advantages over systemic medications, particularly in terms of safety. These advantages are particularly notable in patients with specific comorbidities such as cardiovascular or bleeding disorders. Benefits of IA treatments include enhanced bioavailability, potential placebo effects, and in some cases, a novel mechanism of action that more directly targets the pathophysiology of OA. Furthermore, IA injection is a minimally invasive procedure that can be efficiently performed in an outpatient setting, requiring only a brief recovery period. Joints commonly affected by OA are well-suited to this treatment approach. 44
One approach to OA therapy involves addressing cartilage mechanics through the injection of liposome treatment as a lubricant coating. 16 Surface-attached layers of phosphatidylcholine (PC) liposomes may reduce the friction coefficient μ (=force-to-slide/load) between the sliding surfaces. This phenomenon is attributed to hydration lubrication, an emerging paradigm for lubrication in aqueous and biological environments. 23 The concept of utilizing liposomes as effective lubricant coating has been explored in series of tribology tests aimed to measure the friction force, lubrication and load bearing capacity using mica surfaces coated with a layer of PC liposomes. The studies demonstrated that extremely low friction can be achieved between boundary layers of PC liposomes. The extreme lubricity of the liposome layers is attributed to the hydration lubrication mechanism acting via the strongly attached, fluid hydration layers surrounding the exposed phosphocholine groups at the outer liposome surfaces.13,25,39,41,42 Hence, there is a genuine need for innovative IA therapy that facilitates prolonged liposome retention in joints. 23 This approach aims to coat the damaged cartilage surface resulting in restoring cartilage natural lubrication, enhanced functionality and a reduced wear-induced pain.
A promising novel approach to stabilize PC liposomes and preserve their ultralow friction properties involves the incorporation of a lipid/poly-phosphocholine conjugate, as highlighted in previous studies.23,24,25,33 Poly(2-[methacryloyloxy]ethyl phosphorylcholine) (pMPC) stands out due to its ease of polymerization, biocompatibility, and antifouling (i.e., resistance to nonspecific adsorption of proteins and/or adhesion of cells) properties derived from its highly hydrated nature, forming a thick hydration shell around the phosphocholine groups. The use of pMPC has garnered increased attention and recent findings indicate that poly-phosphocholinated liposomes significantly enhance retention time following IA injection in mouse joints. An in vivo study assessing IA retention time was conducted in mice following a single injection of various formulations: DiR (1,1′-dioctadecyl-3,3,3′,3′-tetramethylindotricarbocyanine iodide) fluorescently labeled pMPCylated liposomes, HA, and PEGylated liposomes. 23 Fluorescence monitoring was performed at predetermined intervals over 10 days using a near-infrared camera with a wavelength of approximately 755 nm. The results demonstrated that pMPCylated liposomes exhibited a retention half-life (T1/2) of approximately 85 hours, significantly surpassing the retention half-lives of HA (less than 1 hour) and similarly sized PEGylated liposomes (less than 20 hours). 23
To ensure that new biolubricants do not cause adverse events, particularly local inflammatory reactions that could lead to chondrocyte death, it is essential to conduct well-planned preclinical studies in animals. Recent findings from our research indicate that a single IA injection of pMPCylated liposomes in rats and sheep did not induce local reactivity or systemic toxicity after 1 and 6 weeks. 33 The objective of the present preclinical studies in rats was to evaluate the local and systemic effects of pMPCylated liposomes more comprehensively through repeated IA injections, thereby better simulating their intended human use. Additionally, we extended the duration of observation to 13 weeks and conducted a more thorough histopathological assessment, employing immunofluorescent staining in order to provide more details on the type of inflammatory cell infiltrates in the synovium and joint capsule following the IA injection.
Materials and Methods
Animal Husbandry and Maintenance
A total of 24 young adult healthy Hsd:Sprague-Dawley (SD) rats, comprising 12 males and 12 females, were included in study 1, and 60 rats (30 males and 30 females) were included in study 2. The animals were at the age of 8 weeks at the start of treatment and were obtained, housed, and treated at Envigo CRS LTD (Israel). Animals were housed in polypropylene cages, fitted with solid bottoms and filled with certified commercial wood shavings as bedding material, 2 to 3 rats per cage separated by sex. The animals were given access to a commercial rodent diet ad libitum and provided with free access to drinking water. The environment was maintained at a temperature of 20-24°C, with a relative humidity of 30-70%, and operated on a twelve-hour light/dark cycle regulated by an automatic timer. Approval for the studies was obtained from the National Council for Animal Experimentation (study 1: IL-2208-178-3; study 2: IL-2305-305-3).
Test Item
The Test Item, CCoat, comprised pMPCylated liposomes at concentrations of 15 mM and 30 mM was manufactured by Liposphere Ltd. (Givat-Shmuel, Israel). Both concentrations were prepared in a similar fashion, by dispersing the lipid layer in PBS to obtain the required concentrations of 15 mM and 30 mM. The process was controlled by measuring the particle size. Each final liposome solution was filled into the ready-to-use sterile vials. Several vials of CCoat from either concentration were provided in liquid form and stored in the refrigerator at 2-8°C until dosing. Commercial 0.9% NaCl injection, physiological saline (Baxter, United Kingdom) served as the Control Item.
Experimental Design
Study 1.
The study comprised three Test Item groups and one Saline Control group, each consisting of n=6 rats (3 males and 3 females) per group. Two Test Item groups received 2 or 3 repeated IA injections of CCoat at the high-dose level (30 mM) into both knee joints, with a 2-week interval between successive injections, in order to verify that there are no local or systemic adverse effects when CCoat is accumulated in the knee joint. An additional Test Item group received 3 repeated IA injections of CCoat at the low-dose level (15 mM), into both knee joints, also at a 2-week interval between successive injections. The control group underwent 3 repeated injections of Physiological Saline with a 2-week interval between successive injections as well. Animals were sacrificed 1 week after the last injection session (Table 1). No claim of compliance with Good Laboratory Practice (GLP) was made for this study.
Experimental design of study 1.
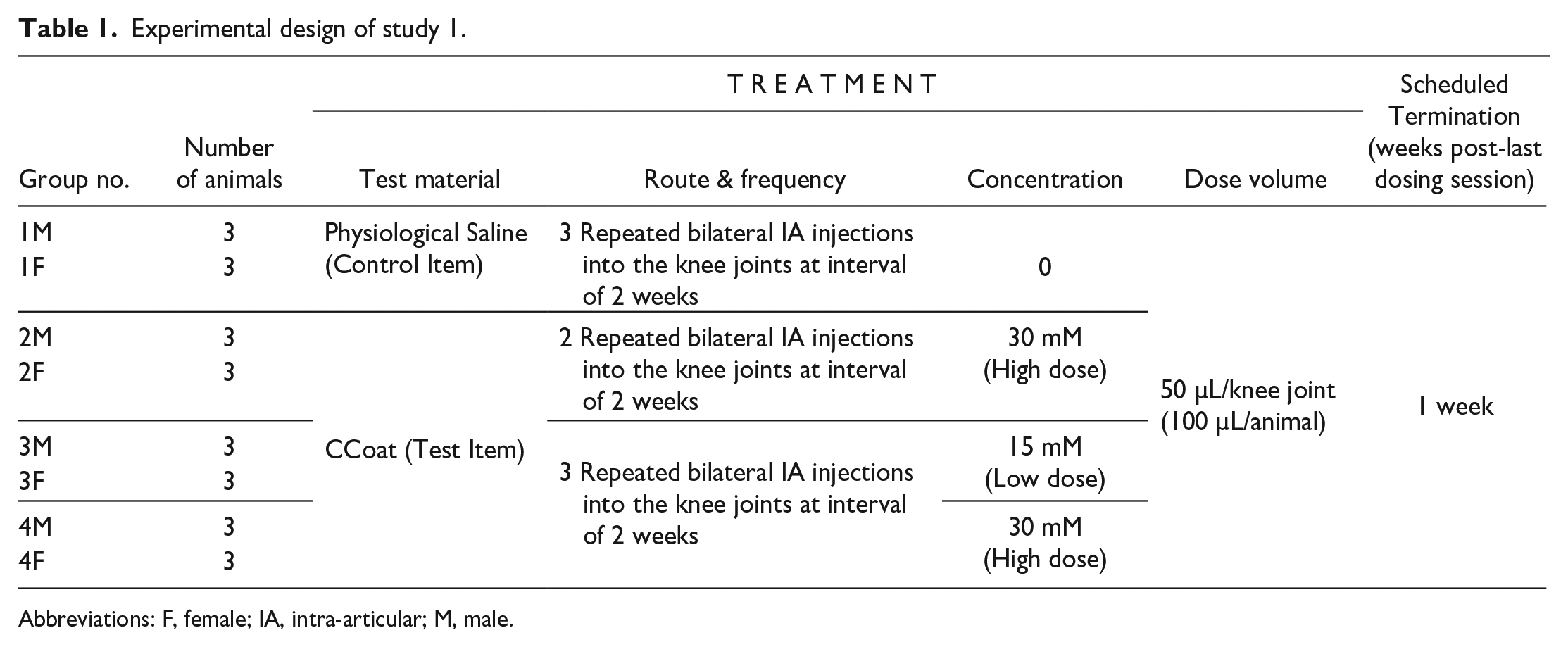
Abbreviations: F, female; IA, intra-articular; M, male.
Study 2.
This study was performed after completion of study 1 using a different batch of CCoat. The study involved two groups: the Test Item group (30 mM of CCoat) and the Saline Control group, both receiving two repeated IA injections at a 3-week interval. Each group consists of n=30 animals, comprising 15 males and 15 females. The animals were observed for up to 13 weeks after the second injection. Five male and five female rats from each treatment group were sacrificed 1 week following the last dosing session, and the remaining ten male and ten female rats in each group were sacrificed 12 weeks later (Table 2). The study was conducted in compliance with GLP principles and according to the ISO 10993-11 and ISO 10993-6 Standards.14,15
Experimental design of study 2.
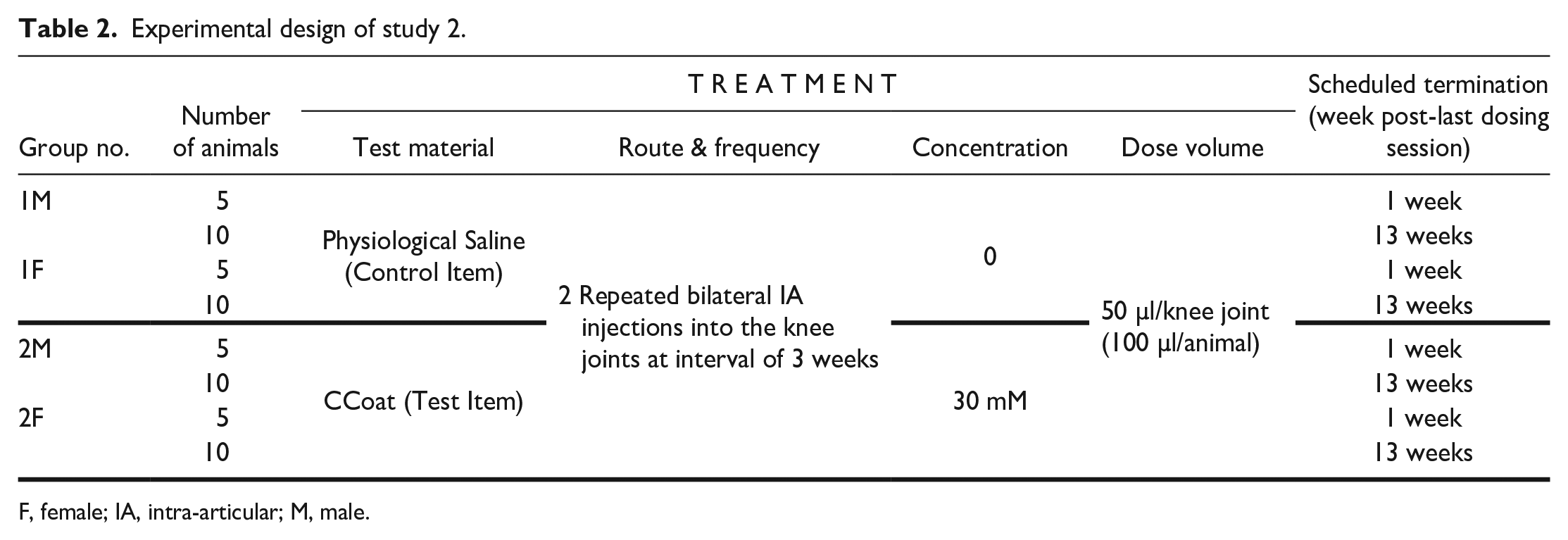
F, female; IA, intra-articular; M, male.
Experimental Procedure
In both studies, IA injections were carried out under general anesthesia by Isoflurane inhalation. The IA injection was performed to both knee joints per animal using a 1-ml syringe connected to a 27G needle after ensuring adequate anesthesia. A new set of syringe and needle was designated for each knee joint. To pinpoint the precise injection site, each hind leg was secured at an angle of approximately 45 degrees. The needle was then inserted through the patellar ligament directly into the joint space of the knee. The maximal feasible volume of 50 microliters was injected to each joint.
Observations and Examinations
The following observations and examinations were performed in both studies.
During the in-life phase, all animals underwent thorough clinical examinations, which also included assessments of local reactions and gait scoring. These examinations were conducted on each day of dosing (once animals had recovered from anesthesia), 1 day after each dosing session, and once weekly thereafter throughout the observation period. Gait scoring was performed using a semi-quantitative grading system with five grades (0-4), based on the gait analysis method for rodents. 20 Furthermore, measurements of body weight and food consumption were conducted once weekly. Ophthalmoscopy examinations were performed on the animals participating in study 2 prior to the first injection and during the last week prior to sacrifice.
At the end of observation period and prior to sacrifice, blood samples were collected for clinical pathology examination by retro-orbital sinus bleeding.
Following euthanasia, all animals in both studies underwent a complete necropsy and gross pathological examination. In study 1, tissues and organs collection was limited to the following organs: Both knee joints (injection site), popliteal lymph nodes, liver, spleen, and thymus. All organs were fixed using 10% neutral buffered formalin (approximately 4% formaldehyde solution) for a minimum fixation period of 48 hours. In study 2, a full guideline-based list of tissues and organs 15 (i.e., Supplementary Table 1) were collected from all animals and fixed using either 10% neutral buffered formalin (approximately 4% formaldehyde solution) or Davidson’s Solution (only eyes together with the optic nerve and harderian glands), allowing for a minimum fixation period of 48 hours.
Histopathology examinations were performed on all collected tissues and organs from all animals in study 1. In study 2, all collected tissues and organs from 10 animals (5 males and 5 females) per group per termination time point were examined (a total of 40 animals). The bilateral knee joints (i.e., injection sites; 2/animal) were placed in a decalcification solution (Formical-2000) for three days. On the second day, the solution was changed. After completion of the decalcification process, the bones were rinsed, sectioned in the coronal plane, and sampled at a level consistent with the methods outlined by Bolon et al. 5 The appearance of each trimmed joint showed the distal femur and proximal tibia separated by the joint space as well as the physis (growth plate) for both bones. 5 The trimmed joints were subsequently processed, embedded in paraffin, and sectioned before being stained with H&E.
Histopathological examinations of the knee joints were conducted based on parameters and grading scales derived from the ISO 10993-6 Guideline. 14 The evaluation encompassed cell type and response, tissue response, and total reactivity score. Additional histopathological changes in the knee joints were assessed and scored utilizing parameters and grading scales previously described 33 (Supplementary Tables 2-4), adapted from Bolon et al, 5 Schafer et al, 36 and Silva et al. 38 Any additional histopathological changes in organs/tissues were described and scored using a semi-quantitative generic grading system with five grades (0-4) 36 as follows: Grade 0—No change, Grade 1—Minimal, Grade 2—Mild, Grade 3—Moderate, and Grade 4—Severe.
Immunofluorescence Evaluation
Immunofluorescent staining (IF) evaluations for identification of synovial macrophages type were conducted at StageBio (Frederick, MD, USA) on a total of 8 fixed SD rat knee joints, corresponding to one male and one female from each group from study 1 (Table 3). After optimization runs with appropriate rat colon, rat lymph node, and human tonsil tissues, the knee joint sections for identification of were subjected to staining using a triple-label immunofluorescent (IF) assay (Table 4) as follows: (1) CD68 and CD163 Cocktail as pan-macrophage detector, (2) P-Stat1 as M1 macrophage marker and (3) C-Maf as M2 macrophage marker. Since C-Maf and P-Stat1 are not specific to macrophages, the interpretation that the stained cells are M1 or M2 macrophages were performed in conjunction with CD68/CD163 cocktail Immunofluorescence labeling. Matching isotypes included Rabbit IgG (Thermo Fisher Scientific, 31235) and Mouse IgG (Thermo Fisher Scientific, 31903). The staining procedure was carried out using the Leica Bond Rx staining platform. Resulting slides were digitally scanned under fluorescence using the Zeiss Axioscan and were assessed for local response. The intensity of specific reactivity was graded on a scale of 0-5, 0=negative, 1=Weak (Blush), 2=Mild, 3=Moderate, 4=Strong, 5=Intense. The frequency of specific reactivity was graded based on the percentage of a particular cell type or tissue element (N=Negative, no reactivity; VR=Very rare, estimated as less than 10%; R=Rare, estimated as 10-25%; O=Occasional, estimated as 26-50%; F=Frequent, estimated as 51-75%; VF=Very frequent, estimated as 76-100%). Positive cells for CD68 and CD163 cocktail were indicated by red cytoplasm, C-Maf signal was indicated by pink/purple nucleus and Pstat1 by green nuclear signal. Additionally, DAPI staining was used as a general method for identification of cells in the tissue sections and was indicated by the blue nucleus.
Samples evaluated by immunohistochemistry.

Antibodies used for the immunofluorescence study.

Statistical Analysis
Calculations and statistical evaluations were conducted using R (Version 3.2.1) or Microsoft Excel (Version 2403) MultiComp. Rnw (validated R-Script for statistical evaluation between multiple groups and/or multiple parameters between 2 groups) was used. Prior to application of the appropriate statistical method a normality test was performed considered Gaussian distribution (e.g. Shapiro-Wilk normality test; P < .01). For the MultiComp.Rnw, when the normality test passed for all groups, analysis was performed using an equal-variance Bartlett test; P < .01. If the Bartlett test passed, 1 way ANOVA with Dunnett’s posttest was performed. If the Bartlett test did not pass, analysis was performed using nonparametric method, Kruskal-Wallis test with Mann-Whitney U test. When the normality test did not pass for all groups, analysis was performed using nonparametric method, Kruskal-Wallis test with Mann-Whitney U test.
Results
Study 1
Mortality, clinical signs, local reactions, and body weights
No mortality was observed in any of the animals throughout the entire study period. Abnormal clinical signs (e.g., changes in activity or mobility, abnormal posture, piloerection, shallow and/or rapid respirations, etc.) were not detected, and all animals exhibited normal gait (score 0). All groups demonstrated comparable body weight gain by the end of the study period. No statistically significant differences were noted.
Clinical pathology
There were no Test Item related changes in any of the clinical pathology parameters compared with control values.
Organ weight and organ weight to body weight ratio
Organ weights included both absolute values and relative to terminal body weight values. There were no Test Item related changes in organ weights and organ weights to body weight ratio compared with control values.
Macroscopic observations
No gross pathological findings were observed in any of the animals at the time of their scheduled necropsy.
Histopathological findings
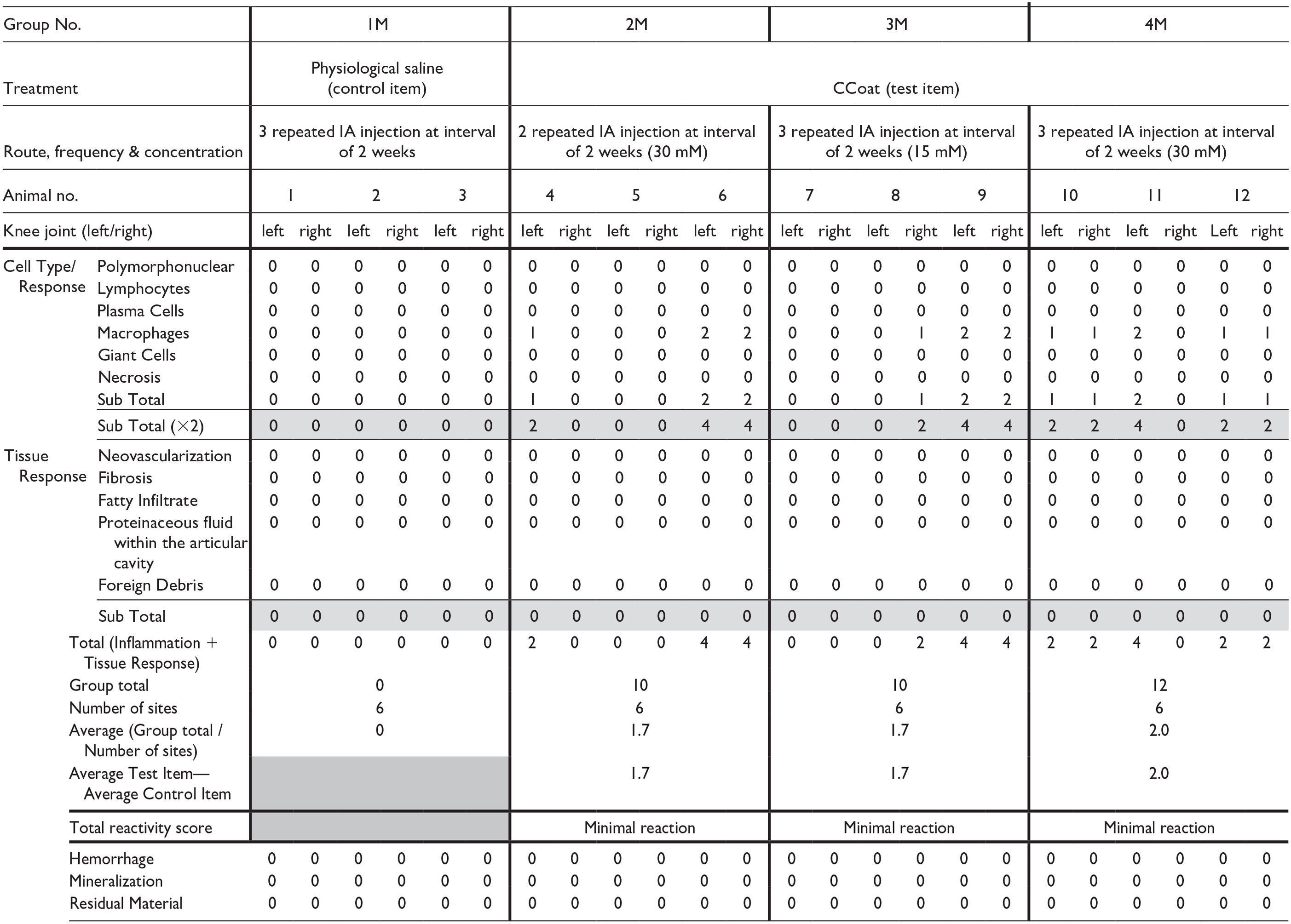
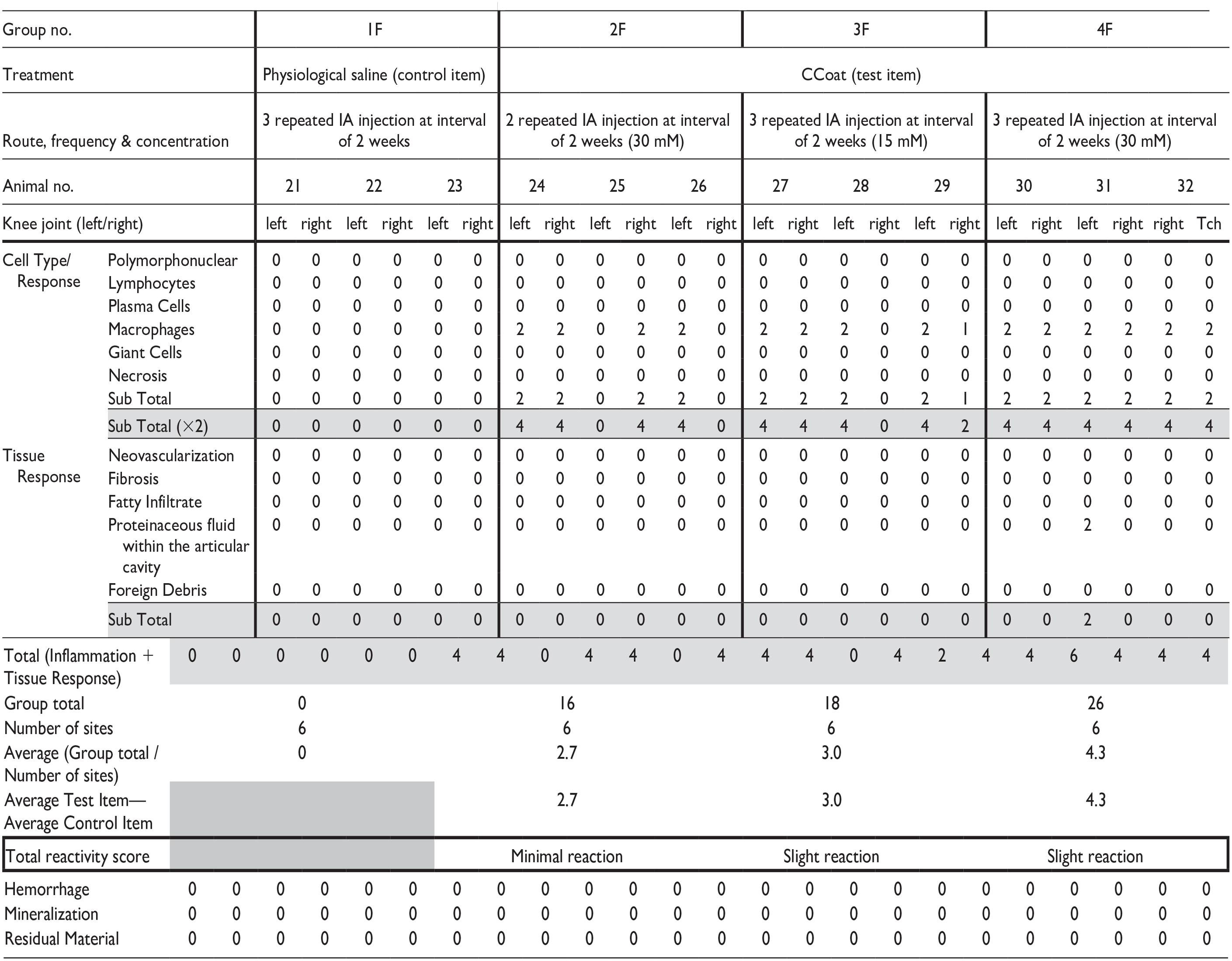
Test Item related changes were dose-related and observed in the injected knee joints of all Test Item treated groups in both sexes. These changes primarily involved the presence of macrophages consisting of minimal to mild accumulation of monomorphic and monotypic macrophages with abundant vacuolated cytoplasm beneath the synovial membrane, with the absence of any inflammatory cells (i.e., polymorphonuclear cells or lymphocytes) (Figure 1). In a singular instance from Group 4F, a mild presence of pinkish, acellular proteinaceous fluid was noted within the articular cavity, without any additional damage to the articular structure. The total reactivity was scored up to minimal in all-male treated groups (i.e., Groups 2, 3 & 4), and slight in female Group 2 and Groups 3 and 4 (Tables 5 and 6). No additional changes were noted in the injected knee joints of all Test Item treated groups in both sexes.
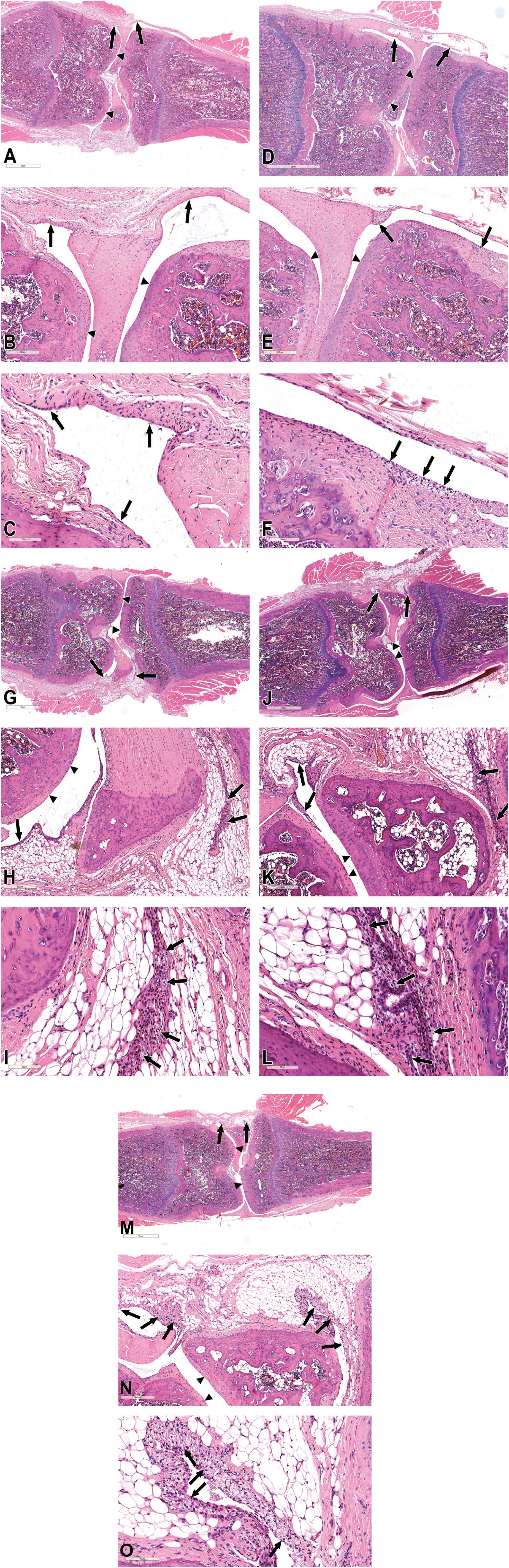
Histopathological findings of the knee joint in study 1. (A-C) Histopathological section from a control group rat (Group 1M). No abnormalities are detected. Synovial membrane—arrows; articular cartilage—arrowheads. A. low magnification, B. medium magnification, C. Higher magnification. (D-F) Histopathological section from a rat treated with 2 repeated IA injections of 30 mM (high dose) CCoat (Group 2M). Minimal accumulation of vacuolated macrophages beneath the synovial membrane is noted (arrows point to the structures of interest), without any inflammatory reaction. Articular cartilage—arrowheads. D. low magnification, E. medium magnification, F. Higher magnification. (G-I) Histopathological section from a rat treated with 3 repeated IA injections of 15 mM (low dose) CCoat (Group 3M). Minimal accumulation of vacuolated macrophages beneath the synovial membrane is noted (arrows), without any inflammatory reaction. Articular cartilage—arrowheads. G. low magnification, H. medium magnification, I. Higher magnification. (J-L) Histopathological section from a rat treated with 3 repeated IA injections of 30 mM (high dose) CCoat (Group 4M). Minimal accumulation of vacuolated macrophages beneath the synovial membrane is noted (arrows), without any inflammatory reaction. Articular cartilage—arrowheads. J. low magnification, K. medium magnification, L. Higher magnification. (M-O) Histopathological section from a rat treated with 3 repeated IA injections of 30 mM (high dose) CCoat (Group 4F). Mild accumulation of vacuolated macrophages beneath the synovial membrane is noted (arrows), without any inflammatory reaction. Articular cartilage—arrowheads. M. low magnification, N. medium magnification, O. Higher magnification.
Individual and Group Reaction Scoring according to the ISO 10993-6 Standard—males (study 1).
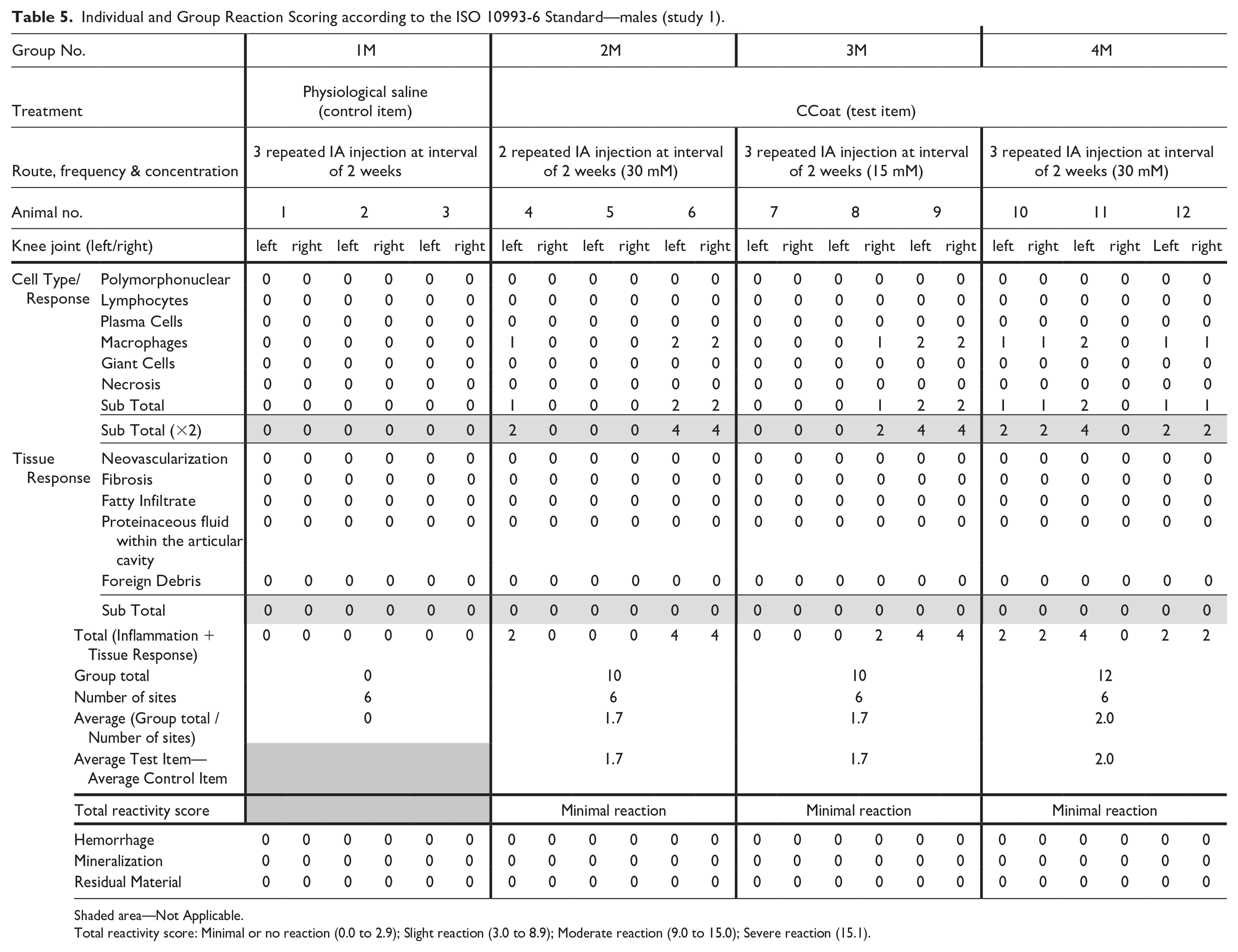
Shaded area—Not Applicable.
Total reactivity score: Minimal or no reaction (0.0 to 2.9); Slight reaction (3.0 to 8.9); Moderate reaction (9.0 to 15.0); Severe reaction (15.1).
Individual and Group Reaction Scoring according to the ISO 10993-6 Standard—females (study 1).
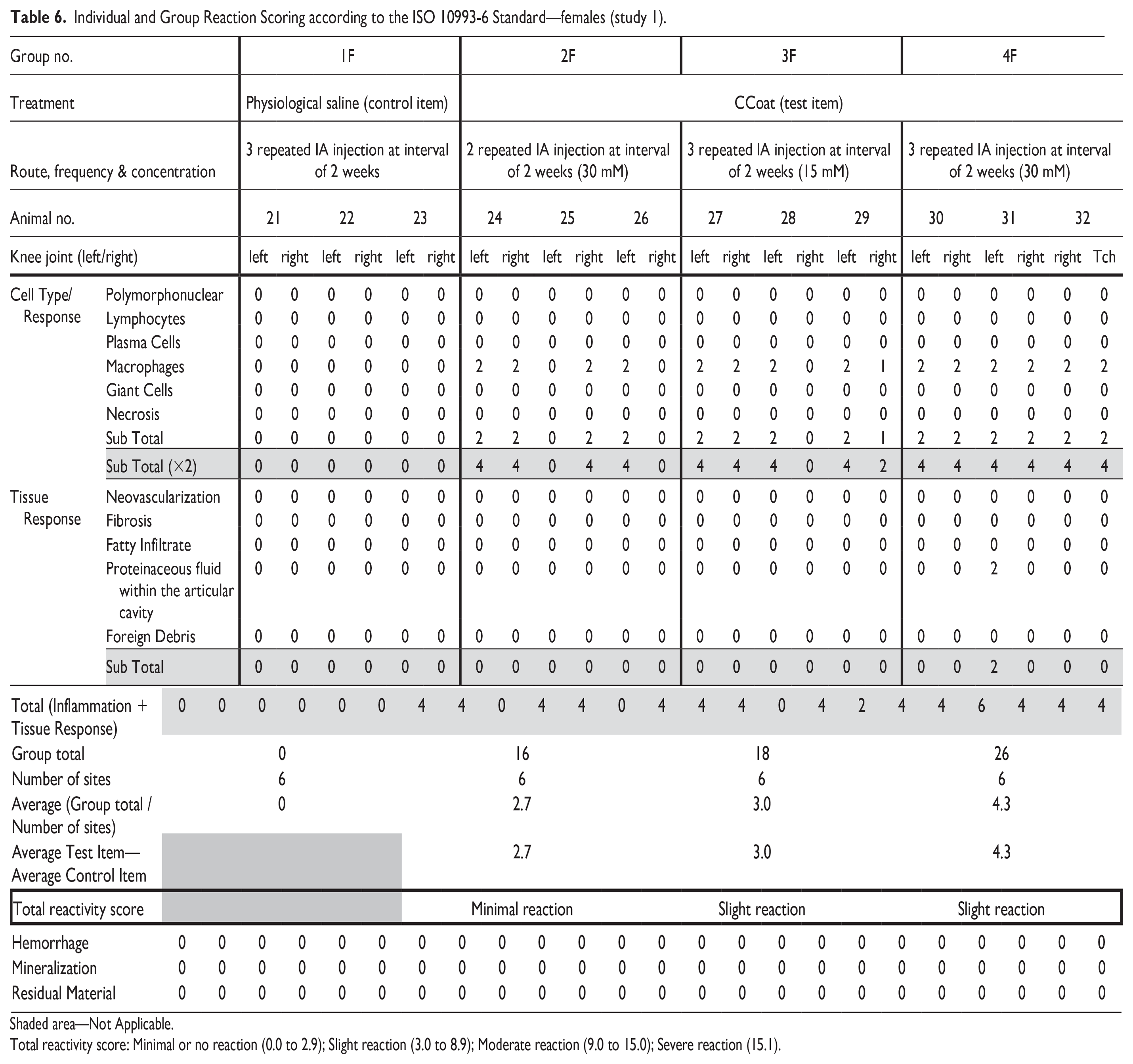
Shaded area—Not Applicable.
Total reactivity score: Minimal or no reaction (0.0 to 2.9); Slight reaction (3.0 to 8.9); Moderate reaction (9.0 to 15.0); Severe reaction (15.1).
No treatment-related changes were observed in any other organ examined.
Immunofluorescence evaluation
Immunofluorescence evaluation was limited to study 1 and was performed to one male and one female from each group. The knee joint sections were stained using a CD68+CD163/P-Stat1/C-Maf triple-label immunofluorescent (IF) assay and the findings were compared with those noted in the H&E staining. In the Control animals (Group 1), no immunofluorescence staining was observed within the joint space or at the joint capsule margin in either case, indicating that no macrophages were found. However, in all Test Item treated groups (Groups 2-4), there were low numbers of macrophages lining the joint capsule/synovial membrane. Almost all macrophages, with only rare exceptions, exhibited dual nuclear labeling with both C-Maf (M2-specific macrophage marker) and P-Stat1 (M1-specific macrophage marker) signals. The C-Maf nuclear signal was notably strong and diffuse, while the P-Stat1 nuclear signal was generally reduced and manifested as either a single condensed spot or a diffuse weak-to-mild signal (Figure 2). Due to the preponderance of the C-Maf compared with the P-Stat1 nuclear signal, these findings suggest the macrophages identified in the tissue sections are M2 macrophages.
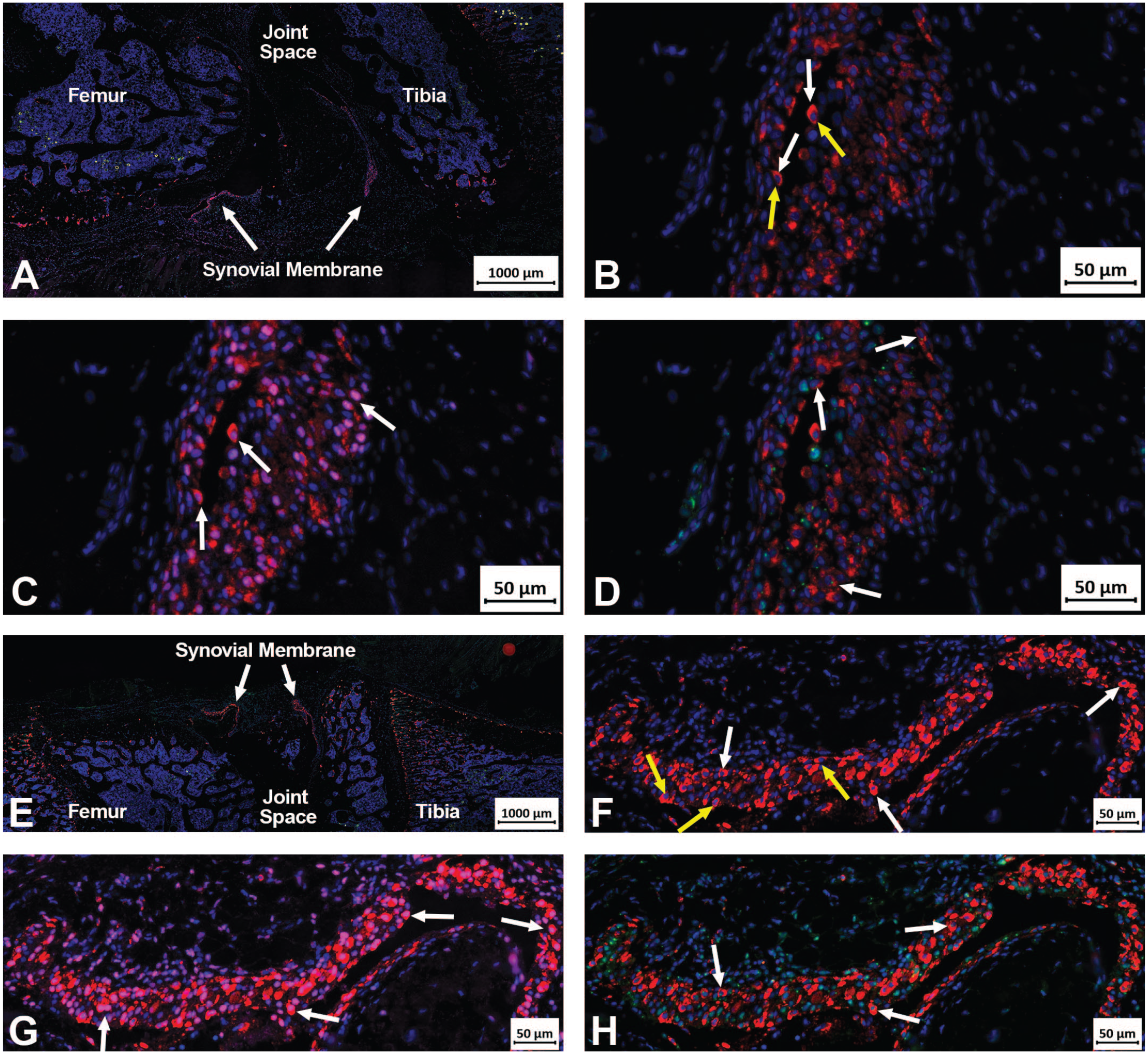
Immunofluorescence images of the knee joint in study 1. (A-D) These immunofluorescence (IF) images capture the right knee joint of an animal from Group 3M, which received three repeated IA injections of 15 mM CCoat. Positive cells for CD68 and CD163 cocktail are indicated by red cytoplasm, C-Maf signal is indicated by pink/purple nucleus, Pstat1 by green nuclear signal and DAPI by the blue nucleus. Autofluorescence is present within the tibia and femur, which presents as yellow color. A. The white arrows highlight the synovial membranes with IF-positive macrophages. B. IF-positive cells expressing CD68 and CD163 within the synovial membrane are observed in this image. The red cytoplasm is indicated by white arrows, while the nucleus, highlighted in blue and positive for DAPI, is marked by yellow arrows. C. These are the same cells as depicted in Figure B. The C-Maf signal is represented by the pink/purple coloration in the nucleus (white arrows), signifying the presence of M2 macrophages. D. These cells correspond to those shown in Figure B. The green nuclear signal, denoting positive P-Stat1 binding, is marked by white arrows. However, it is noteworthy that the signal is diminished both in area and intensity. (E-H) These IF images capture the right knee joint of an animal from Group 4F, which received three repeated IA injections of 30 mM CCoat. Positive cells for CD68 and CD163 cocktail are indicated by red cytoplasm, C-Maf signal is indicated by pink/purple nucleus, Pstat1 signal by green nuclear signal and DAPI by the blue nucleus. E. The white arrows point to synovial membranes with immunofluorescence-positive macrophages. F. Cells within the synovial membrane exhibit positive IF for CD68 and CD163. The red cytoplasm is indicated by white arrows, while the nucleus, highlighted in blue (yellow arrows), confirms DAPI positivity. G. These cells correspond to those in Figure F. The C-Maf signal is represented by the pink/purple coloration in the nucleus (white arrows), indicating the presence of M2 macrophages. H. These are the same cells as shown in Figure F. The green nuclear signal, indicative of positive P-Stat1 binding, is observed, but notably reduced in both area and intensity.
Study 2
Mortality, clinical signs, local reactions, and body weights
No mortality was observed in any of the animals during the entire study period. Throughout the study, no abnormal clinical signs were noted, and there were no local reactions at the injection sites. The gait of all animals was consistently scored as normal (score 0) throughout the entire study period.
All groups exhibited comparable mean group body weight gain at each termination time point. The food consumption of the Test Item-treated group, for both males and females, was within the normal range throughout the study period. No ophthalmologic-related findings were noted in either the Test Item-treated group or the control group.
Clinical pathology
There were no Test Item related changes in any of the clinical pathology parameters compared with control values.
Organ weight and organ-weight-to-body-weight ratio
Organ weights included both absolute values and relative to terminal body weight values. There were no Test Item related changes in organ weights and organ weights to body weight ratio compared with control values.
Macroscopic observations
No gross pathological findings were observed in any of the Test Item treated animals during their scheduled necropsy. In the Control group, one female assigned to the first termination time point, exhibited swelling in the left knee region and a small hemorrhage-like lesion.
Histopathological findings
Histopathological evaluation 1-week post-last dosing session
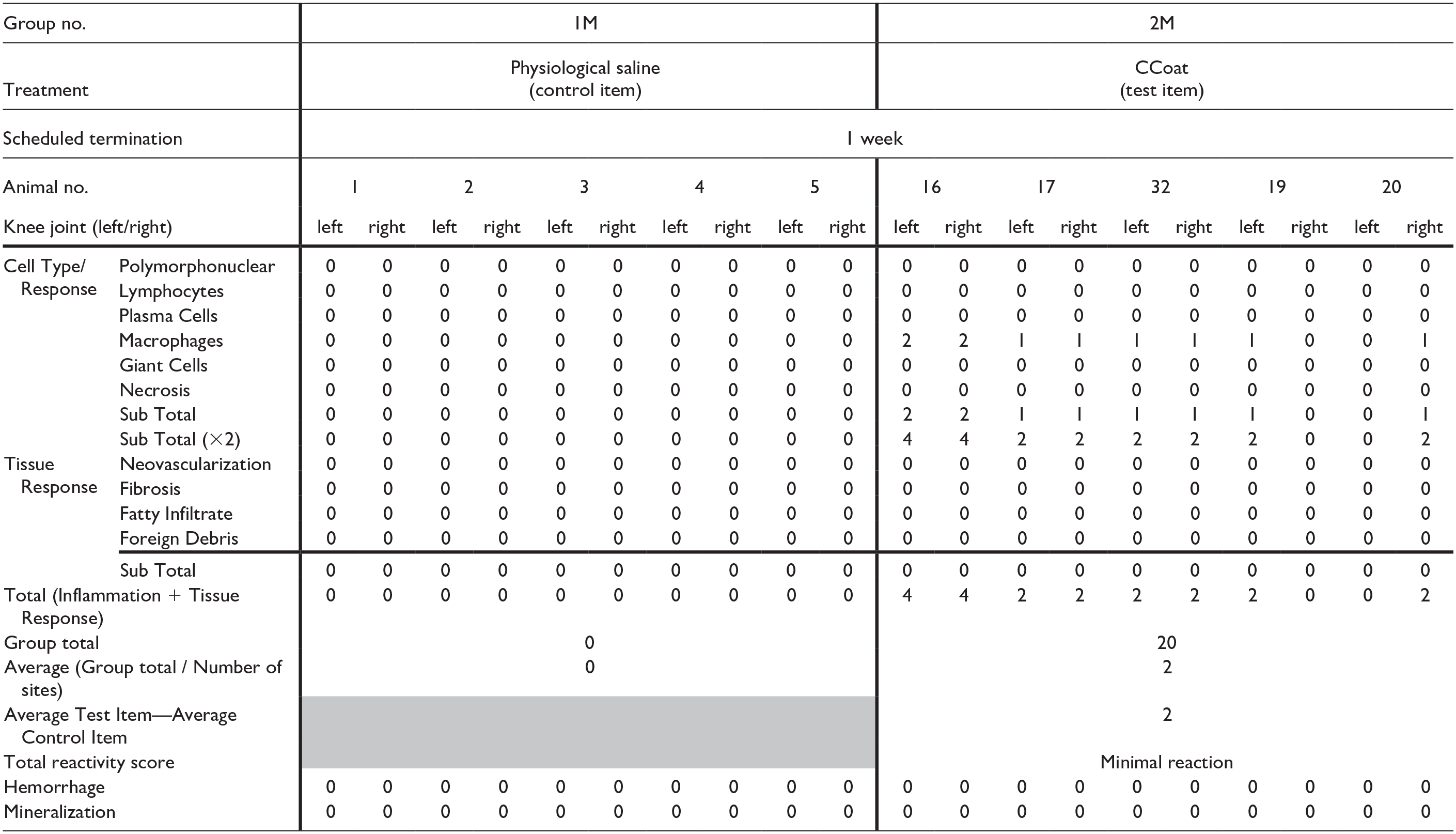
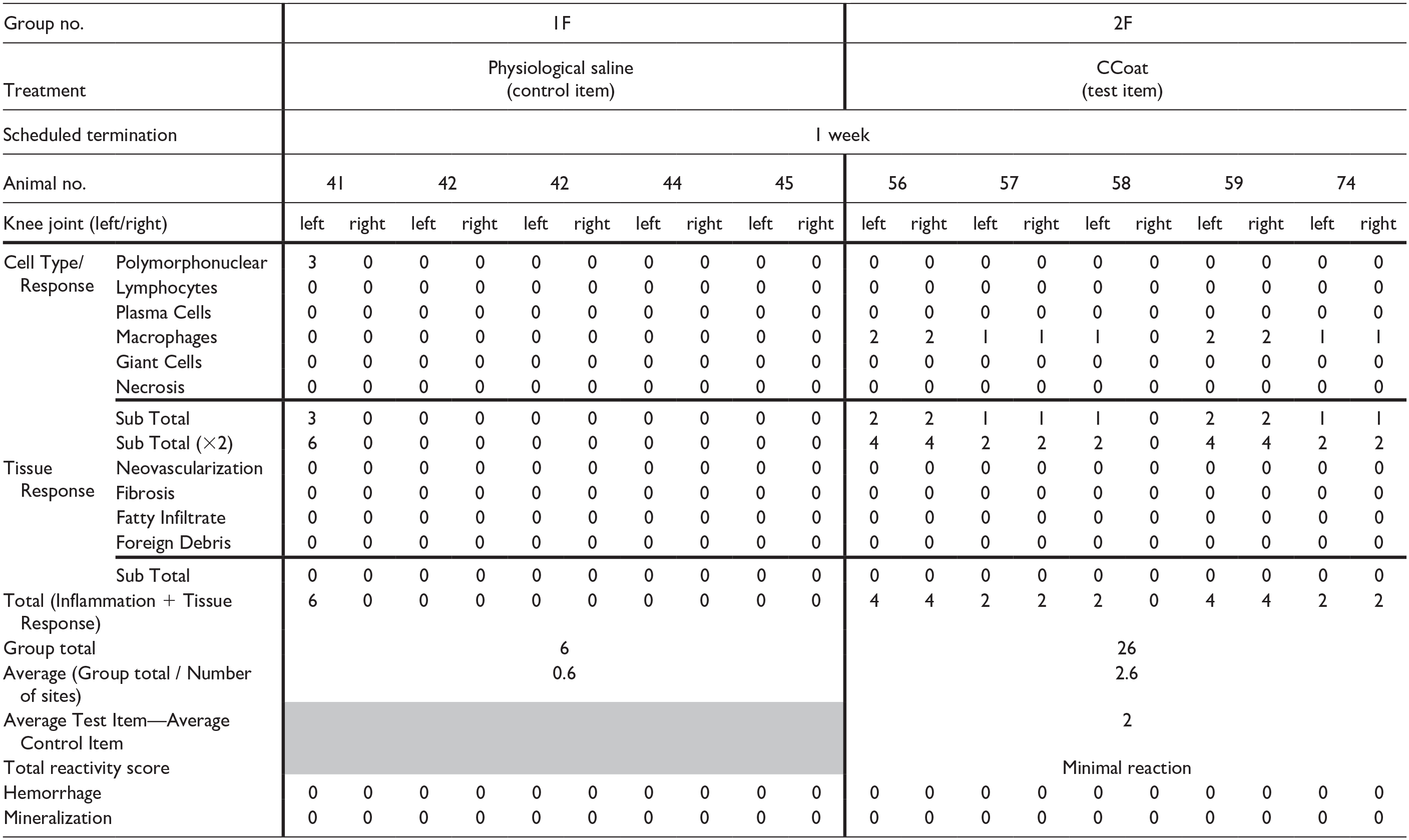
Treatment-related changes were observed in the injected knee joints of the Test Item treated group (Group 2) in both sexes. These changes involved minimal to mild presence/accumulation of vacuolated macrophages beneath the synovial membrane, without the presence of any inflammatory cells (i.e., polymorphonuclear cells or lymphocytes). No pathology was observed in the articular cartilage or adjacent bone. The total reactivity of the Test Item was scored as minimal in Group 2 for both males and females compared with the Control group (Tables 7 and 8). No additional changes were noted in the injected knee joints of all Test Item groups in both sexes.
Individual and Group Reaction Scoring according to the ISO 10993-6 Standard—males, 1 week post last dosing session (study 2).
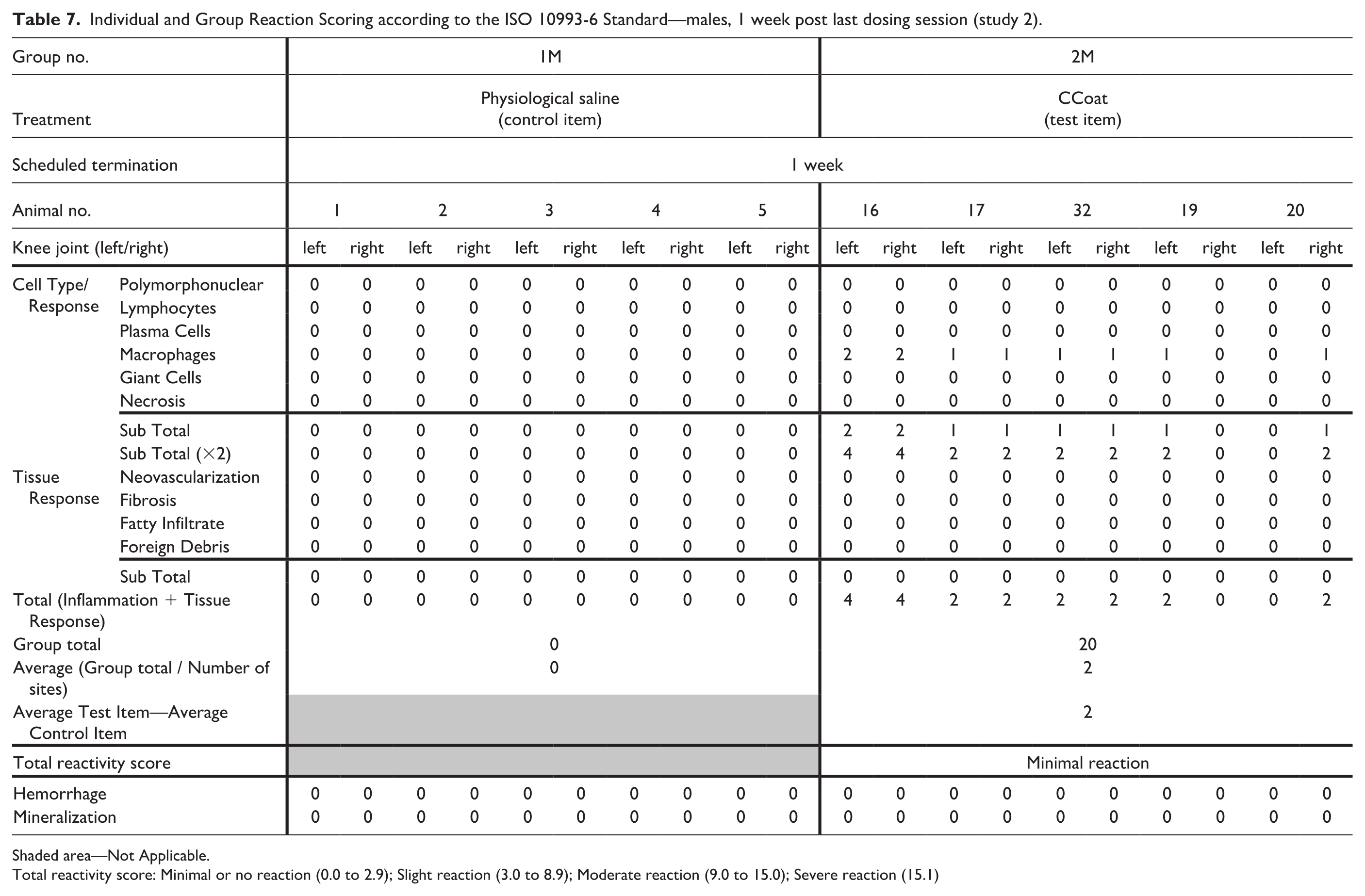
Shaded area—Not Applicable.
Total reactivity score: Minimal or no reaction (0.0 to 2.9); Slight reaction (3.0 to 8.9); Moderate reaction (9.0 to 15.0); Severe reaction (15.1)
Individual and Group Reaction Scoring according to the ISO 10993-6 Standard—females, 1 week post last dosing session (study 2).
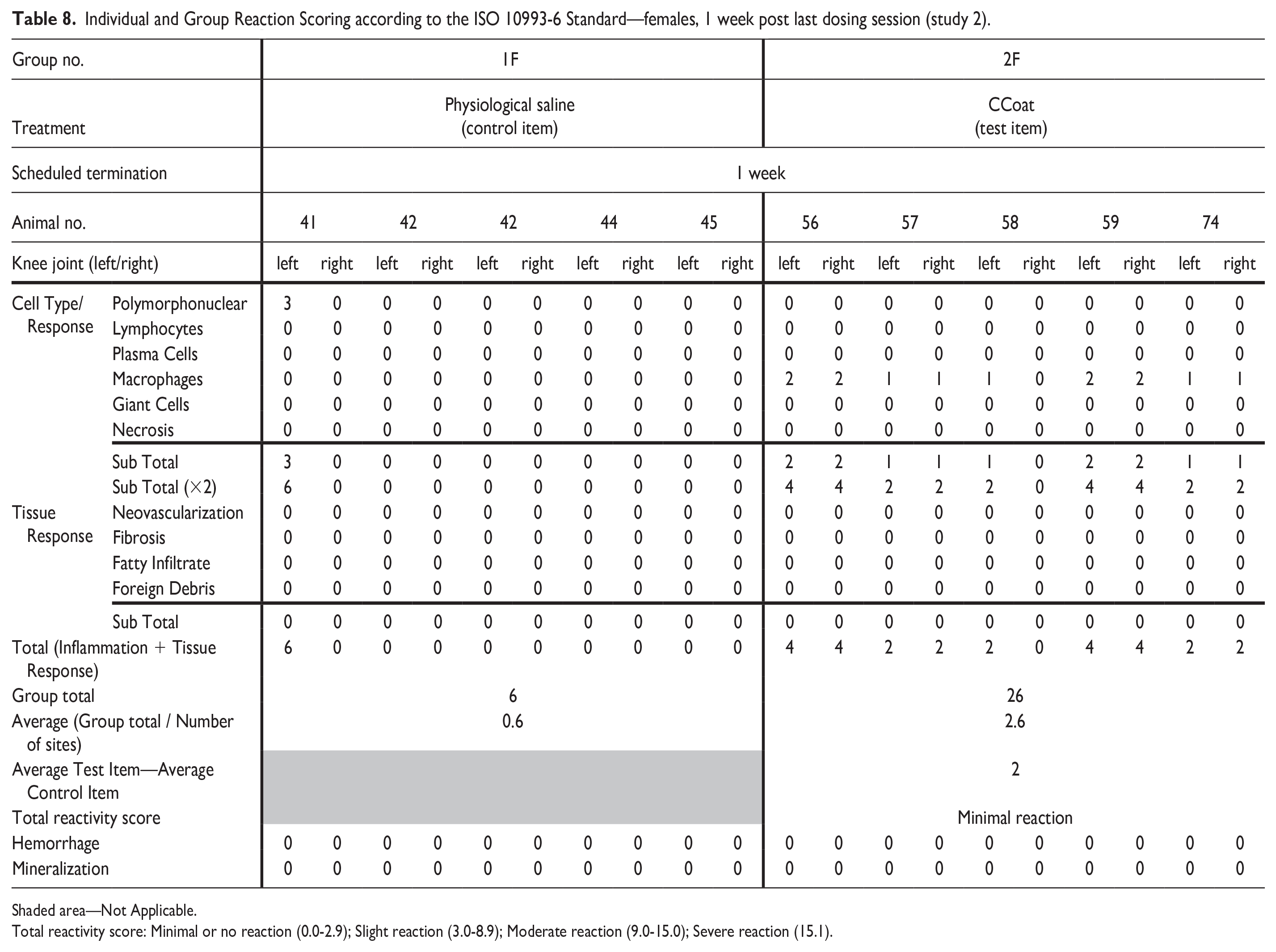
Shaded area—Not Applicable.
Total reactivity score: Minimal or no reaction (0.0-2.9); Slight reaction (3.0-8.9); Moderate reaction (9.0-15.0); Severe reaction (15.1).
No Test Item related changes were noted in any other examined organ. A solitary case in an animal assigned to the Control group exhibited moderate acute inflammation (synovitis) in the knee joint that was compatible to the macroscopical findings.
Histopathological evaluation 13-week post-last dosing session
No Test Item related changes were observed in any examined organ. Specifically, the treatment-related changes observed in the injected knee joints of the Test Item treated group (Group 2) in both sexes, 1 week post the last injection, were completely recovered by the 13th week post-injection. The Test Item exhibited no reaction, with a total reactivity score of 0 in Group 2 for both males and females, as compared with the Control group.
Discussion
Here, we present the findings from two rat studies designed to assess the local and systemic effects of pMPCylated liposomes administered via repeated IA injections. While no systemic effects were identified, in both studies we observed minimal to mild accumulation of monomorphic vacuolated macrophages beneath the synovial membrane in the injected joints, without the presence of any inflammatory cells such as polymorphonuclear cells or lymphocytes. The findings were similar in animals injected with either the low- or high-dose of the Test Item or administered two or three repeated IA injections.
Histopathological evaluation was based on the scoring systems recommended by the ISO 10993-6:2016 Annex E, 14 however it was not limited to these grading systems. The basic suggested descriptors specified in the ISO 10993-6:2016, Annex E are limited to specific cell types and fixed numerical grades and are often not suited to the robust descriptions needed to fully characterize the variety of local tissue responses in medical device implant studies. Furthermore, this grading system is less useful for devices intended to be injected into anatomic sites that are not mentioned in this standard (e.g., articular cavity). The approach to microscopic evaluation must be crafted on a case-by-case basis due to the wide variation in types of devices and study complexities, but certain design elements are still common in analytical schemes for implanted devices. 30
Macrophages, predominant immune cells in the synovium, are primarily situated in the synovial lining layer alongside fibroblasts and play an essential role in maintaining the homeostasis of healthy synovial tissues. These macrophages include tissue-resident synovial macrophages, which are embryonically derived, and nontissue resident synovial macrophages, such as those derived from bone marrow. Biomaterial scaffolds are extensively utilized to facilitate cartilage repair. 45 Implantation of biomaterials invariably triggers an inflammatory reaction characterized by the recruitment of inflammatory cells. This includes neutrophils, which contribute to the acute response, and macrophages, which are involved in both acute and chronic responses. These cells act to isolate and eliminate the foreign material while initiating the repair process. The local tissue microenvironment and signaling mediators critically influence macrophage differentiation toward either a pro-inflammatory type I (M1) phenotype or an anti-inflammatory type II (M2) phenotype. M2 macrophages play a pivotal role in tissue repair, promoting neovascularization, phagocytosis, and fibrosis. Notably, the macrophage population responding to absorbable implants often exhibits an M2-like phenotype, characterized by an abundant cytoplasm indicative of active phagocytosis, and may include cytoplasmic vacuolation. 35
Previous research has demonstrated that macrophage responses to particulate biomaterials hinge on the average particle size of the materials. Biomaterials with particle sizes smaller than a single-nucleated macrophage (approximately 10 μm in diameter) are readily engulfed through phagocytosis by macrophages. 37 Noteworthy advancements in the preclinical safety evaluation of bioabsorbable implants suggest that the phagocytic response to such implants often involves large and foamy macrophages containing phagocytized products.10,28,35
In study 1, IF analysis was performed using a CD68+CD163/P-Stat1/C-Maf triple-label IF assay to verify the types of macrophages observed. Immunofluorescence evaluation was limited to study 1 and was performed to one male and one female from each group. A cocktail of CD68 and CD163 was employed to identify macrophages in tissue sections, although this combination alone is insufficient for characterizing polarized macrophages. CD163 serves as a specific marker for macrophages, whereas CD68 may also detect certain dendritic cell subsets. Macrophages are known to form a heterogeneous cell population with functional variability that largely depends on their polarization status. The polarization of macrophages is influenced by specific transcription factors, with p-Stat1 and RBP-J associated with M1 polarization, and C-Maf with M2. The combination of CD68 and CD163 with these transcription factors proved effective for characterizing macrophage polarization. 1
Importantly, in the absence of a concurrent mixed inflammatory process, this response is characterized as very monomorphic and monotypic, distinguishing it from an inflammatory response that is more pleomorphic. Inflammatory responses involve macrophages in various stages and forms of stimulation and reactivity, and are polytypic, involving multiple cell types acting in concert, including lymphocytes, polymorphonuclear cells, and macrophages. It is worth noting that the M1 and M2 macrophage populations exhibit distinct morphological phenotypes recognizable through imaging techniques using a florescent microscope. 34 Specifically, M1 macrophages appear compact (smaller) and spindloid, while M2 macrophages are more spread-out with larger cytoplasm and, not infrequently, multinucleated, as observed by a fluorescent microscope.
The Medical Device Implant Site Evaluation working group, as appointed by the Society of Toxicologic Pathology (STP) Scientific and Regulatory Policy Committee, emphasizes the importance of histopathology evaluation. The primary goal is to distinguish between an inflammatory response signaling a potentially problematic (adverse) immunological reaction and a predominantly phagocytic response that is a normal and temporary biological reaction.18,30
In the current studies, we observed minimal-to-mild accumulation of vacuolated macrophages beneath the synovial membrane, with no presence of inflammatory cells such as polymorphonuclear cells or lymphocytes. In the absence of a concurrent mixed inflammatory process, this response is very monomorphic and monotypic, that distinguishes it from an inflammatory response which is more pleomorphic (i.e., involves macrophages in various stages and forms of stimulation and reactivity) and polytypic, involving multiple cells types acting in concert (to include lymphocytes, polymorphonuclear cells, and macrophages). 35 The morphology of these macrophages aligns with the characteristics of M2-like macrophages, displaying a monomorphic and vacuolated appearance indicative of their absorptive role rather than a pro-inflammatory response. These findings align with the IF evaluations, that demonstrated that almost all macrophages, with few exceptions, had predominately C-Maf nuclear labeling, suggesting an M2 macrophage.1,7,11,43 Therefore, together with the fact that there is lack of impact on joint structure or function, the reaction in the articulation is deemed nonadverse. This aligns with the distinction made by the STP working group, reinforcing the importance of differentiating between immunologically problematic responses and normal, transient biological reactions in the evaluation of histopathological findings.
In study 2, a singular case from the Control group presented moderate acute inflammation (synovitis) in the knee joint. This occurrence is attributed to an incidental, procedure-related contamination that took place during the IA injection. It is important to recognize and differentiate these incidental findings from treatment-related effects, contributing to a comprehensive understanding of the study outcomes.
The findings of the two reported studies align with the findings reported after a single injection in rats and sheep, that also showed no signs of reactivity or abnormal findings in the injected joints or in any other organs. 33 In view of the reported findings and under the conditions of these studies, it can be concluded that 2 or 3 repeated IA injections of CCoat into the knee joint, at an interval of 2 or 3 weeks between successive injections, and at the maximal feasible volume of 50 microliters to male and female SD rats, is not associated with major short-term or long-term (i.e., up to 3-month recovery duration) systemic or local microscopic adverse effects, and the Test Item is considered safe.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233241271400 – Supplemental material for Biocompatible Solutions: Evaluating the Safety of Repeated Intra-Articular Injections of pMPCylated Liposomes for Knee Osteoarthritis Therapy in Rat Models
Supplemental material, sj-docx-1-tpx-10.1177_01926233241271400 for Biocompatible Solutions: Evaluating the Safety of Repeated Intra-Articular Injections of pMPCylated Liposomes for Knee Osteoarthritis Therapy in Rat Models by Yuval Ramot, Noam Kronfeld, Michal Steiner, Eric D. Lee, Ronit Goldberg, Sabrina Jahn and Abraham Nyska in Toxicologic Pathology
Footnotes
Correction (October 2024):
Article updated online to correct the term “drug” to “medical device” in the Declaration of Conflicting Interests.
Abbreviations
IA, intra-articular; IF, immunofluorescence; OA, osteoarthritis; PC, phosphatidylcholine; pMPC, poly(2-[methacryloyloxy]ethyl phosphorylcholine; SD, Sprague-Dawley.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SJ and RG are employed by Liposphere Ltd. They acknowledge a potential conflict of interest as they may have commercial involvement with the medical device.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Liposphere Ltd., Givat-Shmuel, Israel.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.