Abstract
Osteoarthritis (OA) can lead to a significant functional disability. Poly[2-(methacryloyloxy)ethyl phosphorylcholine] (pMPC) liposomes are a novel treatment modality for OA, intended to restore the natural lubrication properties of articular cartilage. Here, we report on two studies aimed to assess the local and systemic safety and toxicity of pMPCylated liposomes in comparison with physiological saline, in Sprague-Dawley (SD) rats and in sheep after a single intra-articular (IA) injection. The animals were sacrificed after 1 and 6 weeks (rats) and 3 and 6 weeks (sheep). No signs of toxicity or abnormal clinical findings were observed. Histopathological evaluation revealed no signs of reactivity or abnormal findings in the injected joints or in any other organs. In conclusion, a single IA injection of the pMPCylated liposomes demonstrated an excellent safety profile and did not result in local reactivity or systemic toxicity, thus supporting its further development for use in humans.
The major human joints represent mechanically highly stressed, biological environments that under physiological conditions maintain their efficient functionality due to the self-lubricating nature of the articular cartilage that covers the sliding surfaces of the joints. Osteoarthritis (OA) results when articular cartilage degrades, the underlying subchondral bone is exposed, and consequently the joints’ natural lubrication and articulation ability breaks down. The clinical outcome of OA manifests in the form of gradual development of joint pain, stiffness and crepitus with motion, and limitation of movement, hence significantly limiting an active lifestyle. 1
Although OA is a very common condition, which affects 7% of the global population, available treatment options are limited and there is no generally accepted therapy that alters the course of OA. Significant challenges that persist with drug delivery and treatment for OA are (1) a lack of vasculature within synovial joints and (2) the rapid clearance of locally delivered therapeutics due to synovial fluid exchange. 2 Unlike other joint diseases, OA is locally restricted to one or a few joints, which provides a unique opportunity for local intra-articular (IA) treatment. Hence, there is a clear unmet need to develop long-lasting therapeutic strategies to overcome the hurdle of rapid clearance.
Currently, the IA local injectable is the treatment option of choice after conservative pain relief measures have failed and before total knee replacement surgery. Among the local IA injectables, the viscosupplement hyaluronic acid (HA) is a popular option. HA is an important natural component of the joint and IA-HA has been proposed to provide shock-absorptive, chondroprotective, and anti-inflammatory effects within the knee. 3 In osteoarthritic joints, the molecular weight and concentration of HA are diminished. The concept of IA viscosupplementation suggests that the injection of HA could help restore the viscoelasticity of the synovial fluid. HA is approved by the US Food and Drug Administration and European Medicines Agency for the treatment of knee OA and marketed by multiple companies. However, the efficacy of IA-HA treatment for knee OA has been a topic for controversy, particularly among the orthopedic community, as the current literature provides inconsistent results and conclusions regarding this treatment method. 4
Although IA-HA injections are usually considered safe and well tolerated, there are numerous reports of potential adverse events following their use, some of them can be systemic and severe. Most of the reported adverse events are local inflammatory reactions at the injection site, characterized clinically as knee effusion and pain. 5 Sometimes, the IA injection can result in flares of crystalline arthropathy. Although these adverse reactions are temporary and resolve quickly, there are reports on a few more severe inflammatory responses. 6
As HA primarily enhances the viscosity of the synovial fluid and does not act on the cartilage surface, it is rapidly metabolized as part of the turnover of the synovial fluid, a factor that diminishes its therapeutic role. Surface-active boundary lubricants that coat the articular surface of the joint might be a promising strategy for the treatment of OA and a potential alternative to IA-HA injections. Liposomes have been commonly studied and commercialized in the context of drug delivery, also for IA applications, 7 The mode of action of these liposomal boundary lubricants is based on the hydration lubrication mechanism 8 : Hydration layers can sustain high loads while exhibiting a fluid-like response to shear with low friction. In the past, the tendency of liposomes to aggregate in solution was a challenge for liposomal commercial applications. A widely used approach to overcome this obstacle is to use steric stabilization via the incorporation of polyethylene glycol (PEG) chains that provide an entropic barrier to aggregation. PEGylation, however, diminishes the efficient lubrication of the bare, unfunctionalized, phosphocholine-exposing liposomes, attributed to the much lower level of hydration of the PEG chains compared with the phosphocholine surface of the liposomes. 9
A novel approach for stabilizing phosphatidylcholine liposomes and for maintaining the ultralow friction properties is by incorporating a lipid/poly-phosphocholine conjugate. 9 Poly[2-(methacryloyloxy)ethyl phosphorylcholine] (pMPC) is easily polymerized, biocompatible, and possesses antifouling properties due to its highly hydrated nature that is based on the formation of a thick hydration shell around the phosphocholine groups. The pMPC as a potential alternative to PEG has gained increasing attention and poly-phosphocholinated liposomes have recently been reported to dramatically increase the retention time following IA injection in mice joints. 10 The use of pMPCylated liposomes as biolubricants to mechanically restore the natural lubrication properties of articular cartilage is an encouraging route for the treatment of OA.
To ensure that new biolubricants do not lead to adverse events, and specifically to local inflammatory reactions that can result in chondrocyte death, it is inevitable to perform well-planned preclinical Good Laboratory Practice studies in animals. Here, we report the full in vivo biocompatibility in terms of local and systemic effects of pMPCylated liposomes in two animal studies—in rats and in sheep—to pave the path for commercial product development. The pMPCylated liposomes were subjected to biocompatibility testing according to ISO 10993 standards. 11
Sprague-Dawley Rat Study
A total of 60 Sprague-Dawley rats (30 males and 30 females) at the age of 9 weeks at treatment initiation were obtained, housed, and treated at Envigo CRS LTD (Israel). The study was performed after receiving approval (No. IL-2106-102-3) from the National Council for Animal Experimentation and in compliance with ISO regulations. The rat model is considered as an appropriate animal species for toxicity evaluation following IA injection as it is relatively easy to inject into the joint and the rat has reasonable joint volume and very little spontaneous cartilage degeneration to complicate histopathology interpretation of toxicity findings. Moreover, the rat is easy to monitor clinically for swelling and gait abnormalities post IA injection.
The animals were randomized into two groups: The animals in group 2 were subjected to a single IA injection of pMPCylated liposomes to both knee joints at a constant dose volume of 50 µl/joint. The animals in group 1 were injected with physiological saline under identical experimental conditions, serving as the control group. Two termination time points were scheduled: 1 week post single IA injection and 6 weeks post single IA injection (Table 1). The IA injection was carried out to both knee joints/animal using 1 ml syringe, connected to a 27G needle. All animals were subjected to a detailed clinical examination. At termination, blood was taken for clinical pathology examination and all the animals in the study were subjected to a full necropsy and gross pathological examination. Histopathology examination was performed on tissues from 10 animals (5 males and 5 females)/group/termination time point, a total of 40 animals. The bilateral knee joints (i.e. injection sites; 2/animal) were decalcified and sectioned in the coronal plane to sample a level consistent with the methods described by Bolon et al. 12 The trimmed joints were processed and embedded in paraffin, sectioned and stained with H&E. Incidence of hemorrhage and mineralization was recorded and graded according to a 5-point scale (0 = none, 1 = minimal, 2 = mild, 3 = moderate, 4 = severe). Histopathological changes in the knee joints were evaluated and scored using the parameters and grading scales in Supplemental Tables 1 to 3, adapted from Bolon et al., 12 Schafer et al., 13 and Silva et al. 14 Total reactivity grade was calculated as follows: For each treatment and control injection site, scoring for all parameters was totalled, after multiplying by a factor of 2 the score for inflammatory cell infiltrates and necrosis, and then averaged per group. The average score for the control treatment was subtracted from the test treatment average to determine a reactivity grade based on the following scale: 0-2.9, minimal or no reaction; 3-8.9, slight reaction; 9-15; moderate reaction; >15, severe reaction. The amount of residual material was evaluated and scored in injection sites according to the following grading scale: 0 = no residual material is detected, 1 = minimal amount of residual material is detected, 2 = medium amount of residual material is detected, 3 = large amount of residual material is detected, 4 = test material appears intact. Any other histopathological changes were described and scored using a semi-quantitative generic grading of five grades (0-4). Statistical calculations were performed using MeanSDRelative_01.2.Rnw and Microsoft Excel 365. A normality test was performed, which considered Gaussian distribution (e.g. Shapiro-Wilk normality test; P < .01). If the normality test passed for all groups, a recommendation for analysis using parametric method was specified: An equal-variance test was performed (e.g. Bartlett test; P < .01). If the Bartlett test passed, an analysis using parametric method was performed using one-way analysis of variance (ANOVA) with Dunnett posttest. If the Bartlett test did not pass, an analysis using nonparametric method was performed using Kruskal-Wallis test with Mann-Whitney U test. If the normality test did not pass for all groups, an analysis using nonparametric method was performed using Kruskal-Wallis test with Mann-Whitney U test.
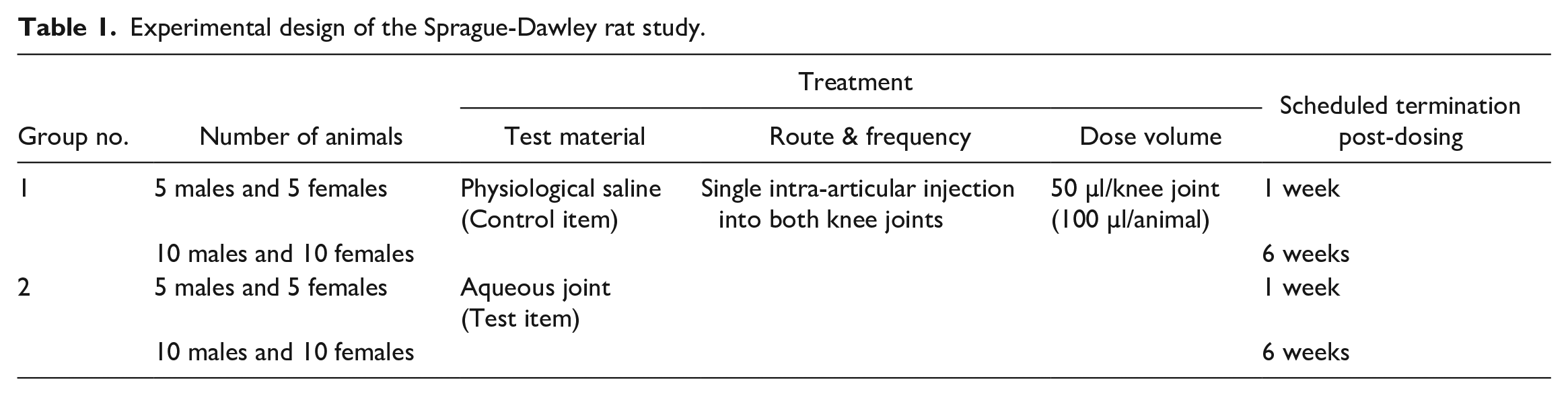
Experimental design of the Sprague-Dawley rat study.
No mortality or obvious adverse reactions were observed among all animals throughout the entire observation period and no treatment-related changes were found in body weight. No treatment-related effects were evident in any of the hematology, biochemistry, and coagulation parameters of the pMPC liposome–treated group vs the control group at both termination time points.
A few statistically significant differences vs the control group were noted in organ weight and organ-weight-to-body-weight ratio in the pMPC liposome–treated males and were considered incidental as no similar changes were evident in the pMPC liposome–treated females and no abnormal findings were observed in the histopathological evaluation. The statistically significant differences were limited to increased group mean weight and ratio values (P < .01 and P < .05, respectively, one-way ANOVA, Dunnett test) of the heart and increased group mean ratio value (P < .05, one-way ANOVA, Dunnett test) of the liver in the males assigned to the 1-week termination time point. A statistically significant decrease in group mean ratio value (P < .05, one-way ANOVA, Dunnett test) of the kidney was noted in the pMPC liposome–treated males assigned to the 6-week termination time point.
No gross lesions were macroscopically observed among all animals at the scheduled termination time points. No tissue reaction was observed during microscopic evaluation at the injection site of the pMPC liposomes at both termination time points (1- and 6-week observation periods) or in the knee joints injected with the physiological saline (control group) (Figure 1). No treatment-related findings were evident in any of the other examined organs.
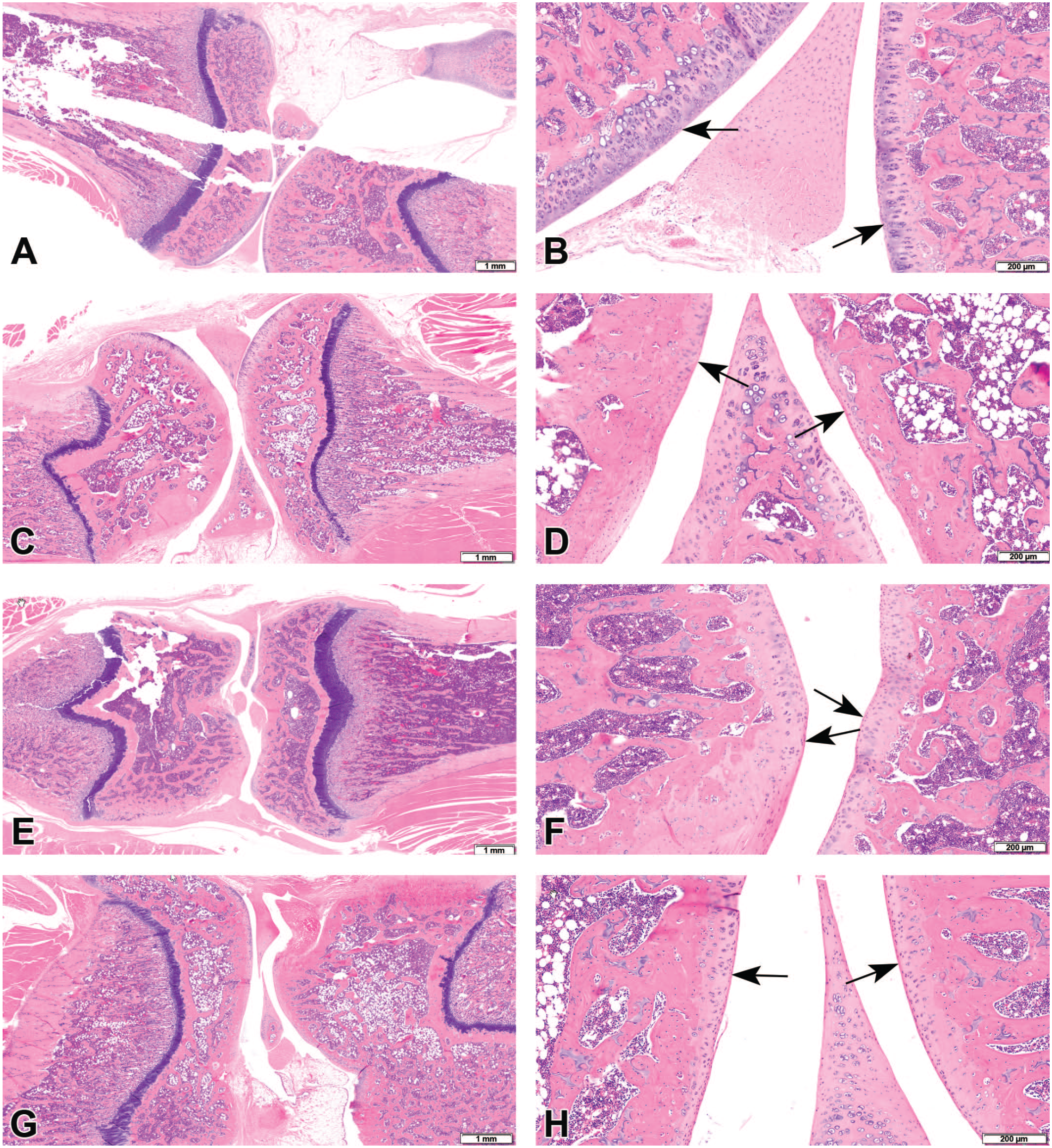
(A and B) Histopathological section of the left knee from a control group rat, 1 week post intra-articular injection. No abnormalities are detected. (A) Low magnification, (B) higher magnification. Arrows indicate articular cartilage. (C and D) Histopathological section of the left knee from a control group rat, 6 weeks post intra-articular injection. No abnormalities are detected. (C) Low magnification, (D) higher magnification. Arrows indicate articular cartilage. (E and F) Histopathological section of the left knee from a poly[2-(methacryloyloxy)ethyl phosphorylcholine] liposome group rat, 1 week post intra-articular injection. No abnormalities are detected. (E) Low magnification, (F) higher magnification. Arrows indicate articular cartilage. (G and H) Histopathological section of the left knee from a poly[2-(methacryloyloxy)ethyl phosphorylcholine] liposome group rat, 6 weeks post intra-articular injection. No abnormalities are detected. (G) original objective 4X, (H) original objective 10X. Arrows indicate articular cartilage.
Sheep Study
A total of 8 female sheeps, 4 months to 1.5 years at study initiation, were obtained from Shikmim farm, Israel, and housed and treated in Kibbutz Lahav, Israel. This study was performed after receiving approval from the National Council of Animal Experimentation (IL-21-03-112).
Ovines have been selected for this study as a large amount of research has been done using the ovine stifle joint, as it has similar cruciate ligaments to humans and can therefore be used in surgical training. In addition, bone mineral density is similar between ovine and humans, and contact pressures generated in ovine are largely comparable with those in humans.
In each sheep, IA injection was applied in both hind legs on day 0. The right knee was administered with undiluted pMPCylated liposomes (3 ml). The left knee was administered with 0.9% (w/v) sodium chloride (3 ml). Animals were sacrificed either on day 21 (group 1) or on day 42 (group 2) (Table 2).
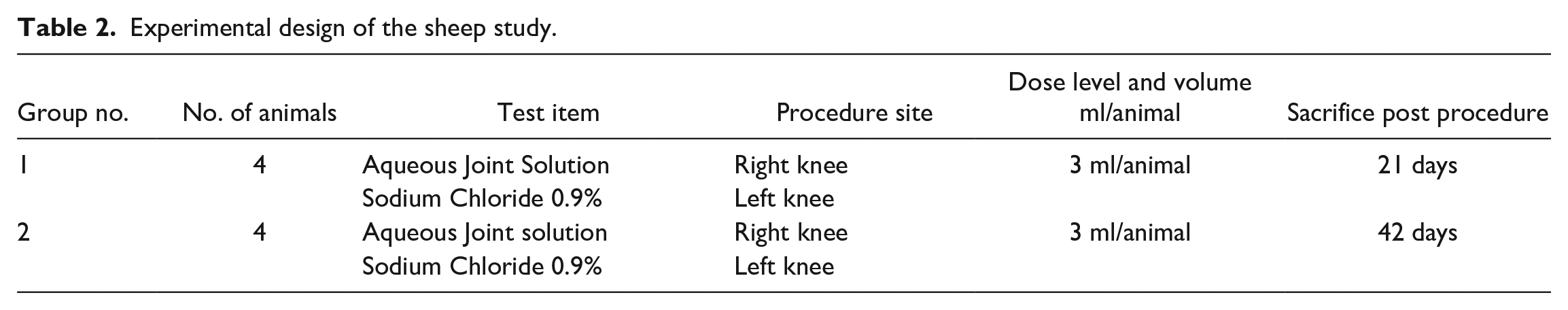
Experimental design of the sheep study.
Animals were observed at least twice daily and a detailed clinical examination was carried out at least once weekly, or more frequently when indicated by the response of the animals to the procedure. At each scheduled necropsy, all animals were subjected to gross pathological examination. The left and right knees and the popliteal lymph nodes were collected from all animals and fixed in 10% neutral buffered formalin. Tissues were trimmed, embedded in paraffin, sectioned at ~5 µm thickness, and stained with hematoxylin and eosin (H&E). Histopathological examination of the joints was described and scored in a similar manner to the SD rats’ study (see above).12 -14 Statistical calculations were performed using Excel (Microsoft Ltd., Redmond, Washington).
No mortality occurred throughout the entire observation period and all animals gained weight normally during the observation period. In addition, no clinical abnormality was observed, including any lameness or knee swelling. No gross pathological findings were observed during necropsy. In addition, no tissue reaction or any other abnormality was observed in the knees or lymph nodes of either the treatment or control group (Supplemental Figure 1). No residual test material was identified within the joint.
Here, we report the toxicological evaluation of pMPCylated liposomes, after a single IA injection in SD rats and sheep. No treatment-related changes were noted in any of the animals, either systemically or locally.
Animal models are essential for the understanding of the efficacy and safety of new treatments for OA, and include mice, guinea pigs, rats, dogs, rabbits, pigs, sheep, horses, and goats. 15 These animal models are also widely used for preclinical safety assessments, and therefore reports on the normal histopathological appearance of the joints following IA injections, in addition to providing information on the experimental design of such studies, are of great importance. Here, we report the findings of two such experiments, performed in SD rats and sheep, and provide information on the histopathological findings at different time points after injection.
Although lipid-base formulations are being used and developed for IA injections to treat OA, very little information is available on their local and systemic toxicity in preclinical animal models. Furthermore, to the best of our knowledge, preclinical toxicity information for IA injection of pMPCylated liposomes has not been reported before, and therefore the information provided in this report is of special importance.
The most valuable evaluation to perform while assessing IA toxicity testing is the histopathological assessment of the injected joint, with particular emphasis on the presence (or absence) of irreversible changes, such as chondrocyte death or loss of collagen matrix due to the loss of matrix-producing cells. 16 Death of chondrocytes can be the result of direct toxicity to the chondrocytes, or secondary to an inflammatory reaction. Potential histopathological changes that can be observed include acute or chronic synovitis, subchondral bone resorption, or loss of cartilage matrix proteoglycan. 16 These changes can be observed at different time points after injection and may resolve spontaneously, emphasizing the importance of assigning several sacrifice times when performing the toxicity studies. Our studies indicated the absence of any damage to the chondrocytes, adjacent bone, or the presence of inflammatory cells within the joint or synovial membranes, suggesting excellent tolerability of pMPCylated liposomes.
In summary, in view of the reported findings and under the conditions of this study, it can be concluded that a single IA injection of pMPCylated liposomes is safe and does not result in local reactivity or systemic toxicity in SD rats and in sheep.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221105393 – Supplemental material for Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis
Supplemental material, sj-docx-1-tpx-10.1177_01926233221105393 for Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis by Yuval Ramot, Oleg Dolkart, Michal Steiner, Sabrina Jahn, Ronit Goldberg, Orna Cacical, Yossi Lavie, Nati Ezov, Gabi Agar and Abraham Nyska in Toxicologic Pathology
Supplemental Material
sj-docx-2-tpx-10.1177_01926233221105393 – Supplemental material for Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis
Supplemental material, sj-docx-2-tpx-10.1177_01926233221105393 for Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis by Yuval Ramot, Oleg Dolkart, Michal Steiner, Sabrina Jahn, Ronit Goldberg, Orna Cacical, Yossi Lavie, Nati Ezov, Gabi Agar and Abraham Nyska in Toxicologic Pathology
Supplemental Material
sj-tif-3-tpx-10.1177_01926233221105393 – Supplemental material for Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis
Supplemental material, sj-tif-3-tpx-10.1177_01926233221105393 for Preclinical In Vivo Safety of Poly-Phosphorylated Superlubrication Vectors for the Treatment of Osteoarthritis by Yuval Ramot, Oleg Dolkart, Michal Steiner, Sabrina Jahn, Ronit Goldberg, Orna Cacical, Yossi Lavie, Nati Ezov, Gabi Agar and Abraham Nyska in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: OD, SJ, RG, and GA are employees of Liposphere Ltd. These authors have a potential conflict of interest as they may use the drug for commercial use.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Liposphere Ltd.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
