Abstract
The toxicity of ATR-107, a human anti-interleukin-21 receptor (IL-21R) monoclonal antibody (mAb), was evaluated in CD-1 mice and cynomolgus monkeys after single-dose intravenous (IV) administration, and in Sprague-Dawley (SD) rats and cynomolgus monkeys after weekly IV and subcutaneous (SC) administration in 13-week toxicity studies that included recovery. Adverse liver necrosis, diffuse bridging fibrosis, and higher liver enzymes occurred in rats in the low-dose IV group (10 mg/kg), but not at 50 or 250 mg/kg IV, and not following SC administration despite overlapping systemic ATR-107 exposures. Similar findings were not seen in mice or cynomolgus monkeys. A series of investigative rat toxicity studies showed liver findings only occurred after administration of at least 3 weekly doses, only occurred in rats that developed anti-drug antibodies (ADAs), and the incidence was associated with higher ADAs titers. However, the presence of ADAs did not always result in liver injury. Liver findings did not occur in nude rats, which had high ATR-107 exposures and no ADAs. These findings suggest an adaptive immune response with formation of ADAs was necessary for development of ATR-107-related liver findings, and that liver injury can occur in rats secondary to development of ADAs following repeated administration of a human therapeutic mAb.
Keywords
Introduction
Interleukin (IL) 21 is a type 1 cytokine with sequence homology to other type 1 cytokines, including IL-2, IL-4, and IL-15. 23 Similar to other cytokines in this family, IL-21 signals through a receptor complex consisting of a shared common gamma chain with specificity for IL-21 provided by the IL-21R chain. 27 IL-21 is produced by CD4+ T cells and natural killer (NK) T cells that are activated during an immune response. 8 The receptor for IL-21 is expressed in many lymphoid and hematopoietic cells and its expression is up-regulated by IL-21. 10 IL-21 plays a role in the differentiation and growth of B cells and T cells, and is associated with chronic inflammation and autoimmunity, including playing a role in inflammatory bowel disease and systemic lupus erythematosus (SLE) in humans. 9 IL-21 is also an essential growth factor for the development of the Th17 cell lineage, most commonly associated with chronic inflammation. 15
ATR-107 is an antagonizing anti-human IL-21R immunoglobulin (Ig) G1 monoclonal antibody (mAb) that potently neutralizes IL-21 bioactivity by preventing the binding of IL-21 to IL-21R. ATR-107 contains mutations in the Fc binding portion of the molecule to reduce effector function and does not mediate cytotoxic effector function based on an in vitro cell-based cytotoxicity assay. In tissue cross-reactivity studies, methods development was problematic and staining of negative control material could not be eliminated. The staining pattern in the mouse, rat, cynomolgus monkey, and human were generally similar and involved staining in all tissues tested. Similar binding of ATR-107 to human and cynomolgus monkey IL-21R (2-4 nM affinity Kd) was observed, whereas binding to mouse or rat IL-21R was lower (16 and 71 nM Kd, respectively) 31 consistent with the lower (60%) amino acid sequence homology between human and mouse/rat IL-21R. ATR-107 had reduced IL-21 neutralization activity in rodents using cell-based assays, but in vivo studies still demonstrated functional activity at doses as low as 1 mg/kg in the mouse and rat, including studies in the MRL-Faslpr mouse model of SLE and in a rat model of air pouch inflammation induced by injection of mouse IL-21 into the air pouch. 31 Pharmacodynamic activity was also demonstrated in cynomolgus monkeys based on the ability of ATR-107 to block rhuIL-21-induced expression of the IL-2RA gene in cynomolgus monkey whole blood samples ex vivo. 30
The nonclinical safety of ATR-107 was evaluated in CD-1 mice and cynomolgus monkeys after intravenous (IV) administration in single-dose studies followed by an 8-week observation period, and in Sprague-Dawley (SD) rats and cynomolgus monkeys after IV and subcutaneous (SC) administration in 13-week repeat-dose (1 dose/week) toxicity studies followed by a 13-week recovery phase. Unexpected liver effects were observed after IV (but not SC) dosing in rats without significant findings in other tissues; similar liver findings were not seen in cynomolgus monkeys or mice. A series of investigative toxicity studies were conducted in CD-1 mice, SD rats, and nude rats. Results from these studies suggested the liver effects only occurred in rats and were related to an immune response associated with anti-drug antibody (ADAs) formation against ATR-107 when administered IV. High-level results of the liver findings have been presented previously in a blinded manner.17,19 Together, the data demonstrate an uncommon manifestation of liver effects related to an immune response to a therapeutic human mAb administered in rats.
Materials and Methods
Test and Control Articles
ATR-107 (WYE-400107, PF-05230900, VL6) was supplied as frozen bulk liquid stocks, then thawed and diluted with vehicle (20 mM
Study Animals and Care
The SD (Crl:CD[SD]) rats were 7 to 9 weeks old and at least 170 g in body weight at the initiation of dosing (Charles River Canada, Quebec, Canada). Female nude (Crl:NIH-Foxn1rnu) rats were approximately 6 weeks old and at least 150 g in body weight at the initiation of dosing (Charles River Wilmington, MA, USA). Crl:CD-1 (ICR) mice were 7 to 9 weeks old and approximately 22 g in body weight at the initiation of dosing (Charles River Canada). Cynomolgus monkeys were approximately 3 to 5 years old and approximately 2 to 6 kg in body weight at the initiation of dosing (Covance, Alice, Texas).
Studies were conducted in accordance with Good Laboratory Practice (GLP) Regulations of the US Food and Drug Administration (21 CFR Part 58) or following the spirit of GLP. All procedures performed on animals were in accordance with regulations and established guidelines and were reviewed and approved by Wyeth Institutional Animal Care and Use Committee. All animals were considered within acceptable health limits at the beginning of the studies. Housing conditions met or exceeded those required or recommended by the Animal Welfare Act or the “Guide for the Care and Use of Laboratory Animals.” Mice and rats were housed individually in plastic solid bottom cages with contact bedding. Cynomolgus monkeys were socially housed in stainless steel enclosures. Mice and rats were fed Teklad Certified Global Rodent Diet (#2016C—pellets) ad libitum; for the nude rat study, the food was autoclaved. Monkeys were fed Teklad Certified Global Primate Diet (#2050C) ad libitum, as well as biscuits, and daily fruit and vitamins supplements. Animals were fasted overnight prior to scheduled necropsies; monkeys were also fasted overnight prior to clinical pathology collections. In all studies, water was available ad libitum, and alternating 12-hour periods of light and dark were used. Rat studies were maintained at 72°F or 22°C and 50% relative humidity, while monkey studies were maintained at 74°F and 40% relative humidity.
Determination of ATR-107 Concentrations in Serum
The concentration of ATR-107 in serum samples was determined using a validated enzyme-linked immunosorbent assay (ELISA). Briefly, the assay used a soluble form of the human IL-21R, histidine-tagged (his-tagged) recombinant human IL-21R (rhuIL-21R), to capture ATR-107. Captured ATR-107 was detected with a mouse anti-human IgG mAb conjugated to horseradish peroxidase and a substrate, 3,3’, 5,5’-tetramethylbenzidine, which generated a colored product that was measured at 450 nm. The serum samples were analyzed along with quality control (QC) samples at three ATR-107 concentrations. Sample concentrations were determined by interpolation from the calibration curve that was fit using a 4-parameter logistic equation. The assay had a quantitative range of 45.0 to 768 ng/mL of ATR-107 in 100% serum.
Determination of Anti-Drug Antibodies in Serum
The presence of ADAs in serum samples was determined using a validated BioVeris ligand-binding bead-based assay. In this assay, samples were co-incubated with biotinylated ATR-107 and ruthenylated ATR-107 overnight (biotinylation and ruthenylation are commonly used methods for labeling reagents for this type of assay). 14 Streptavidin-coated paramagnetic beads were then incubated with the mixture. The plate was then placed in the M-Series 384 Analyzer (BioVeris) and a magnet was applied to capture the paramagnetic beads onto a surface electrode. Unbound reactants were washed away and the ruthenylated ATR-107, captured on the beads, was electrically excited resulting in the production of light. The light was measured by photo detectors with the read-out in response units (RUs). A positive control (goat anti-human IgG) was used to monitor the performance of the assay. Normal CD-1 mouse, SD rat, or cynomolgus monkey serum was used as the negative control and also to determine the assay cutoff RU value in each species, which was defined as twice the mean of the RU value of the negative control for each plate. Samples were initially tested in a screening format at dilutions of 1:25 and 1:75. Samples generating an RU less than the cutoff RU were considered negative. Samples generating an RU greater than or equal to the cutoff RU were considered positive and reanalyzed in a full dilution series to confirm the positive result and determine the titer. The titer of a positive sample was defined as the reciprocal dilution of the sample that would have generated an RU equal to the cutoff RU value. Titers were calculated in the Watson LIMS and were reported as log titer values. The minimum required dilution was 1:25, and the limit of detection was 1.40 (the log of 25). Therefore, negative samples were designated as < 1.40.
Overview of In Vivo Toxicology Program
An overview of the in vivo toxicology program relevant to the liver findings is shown in Table 1, including study objectives, study design, and parameters assessed in each study. Single-dose IV tolerability studies were conducted in CD-1 mice and cynomolgus monkeys with 8-week postdose observation periods. Repeat-dose (1 dose/week) 13-week GLP toxicity studies with 13-week recovery phases were conducted in SD rats and cynomolgus monkeys; both the IV and SC routes were evaluated. Based on the liver findings in the 13-week GLP toxicity study in rats, a series of repeat-dose (1 dose/week) investigative toxicity studies were conducted: three 4-week IV studies were conducted in SD rats, one 4-week IV and SC study was conducted in CD-1 mice, and one 4-week IV study was conducted in nude rats. In most of these studies, evaluated parameters consisted of mortality, clinical observations, body weight, food consumption, clinical pathology (hematology and/or clinical chemistry collected at necropsy unless otherwise specified below), organ weights, and macroscopic and microscopic examinations (unless otherwise specified in Table 1). Blood samples were collected at various time points (specified below) after dosing and serum samples were analyzed for the quantitation of ATR-107 and the detection of anti-ATR-107 antibodies (i.e. ADAs) using the methods described above. In addition to these in vivo studies, non-GLP and GLP tissue cross-reactivity studies were conducted using mouse (non-GLP only), rat, cynomolgus monkey, and human tissues; a full tissue list was examined in all species.
Summary of study objectives and design for nonclinical toxicity studies with ATR-107.
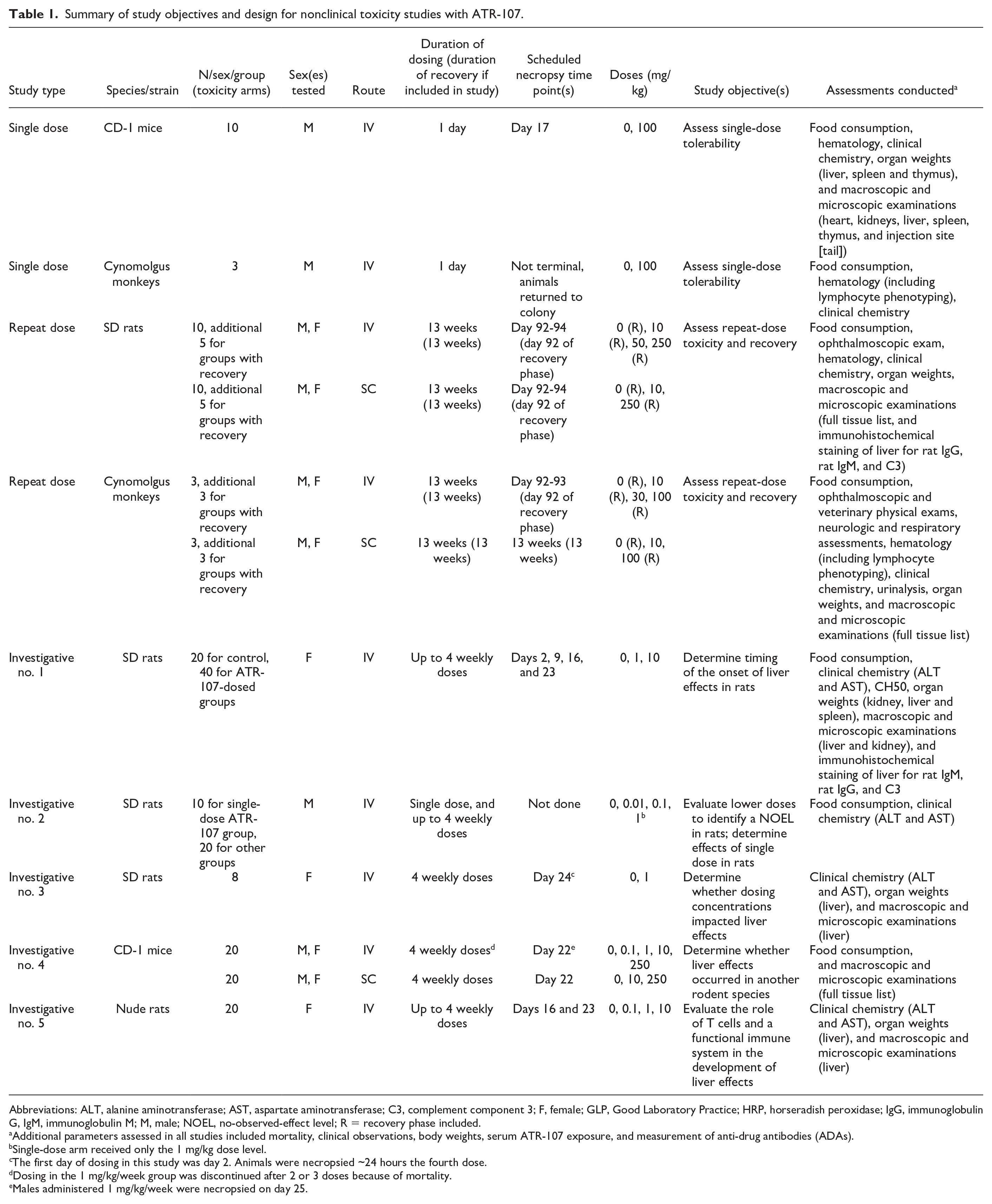
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; C3, complement component 3; F, female; GLP, Good Laboratory Practice; HRP, horseradish peroxidase; IgG, immunoglobulin G, IgM, immunoglobulin M; M, male; NOEL, no-observed-effect level; R = recovery phase included.
Additional parameters assessed in all studies included mortality, clinical observations, body weights, serum ATR-107 exposure, and measurement of anti-drug antibodies (ADAs).
Single-dose arm received only the 1 mg/kg dose level.
The first day of dosing in this study was day 2. Animals were necropsied ~24 hours the fourth dose.
Dosing in the 1 mg/kg/week group was discontinued after 2 or 3 doses because of mortality.
Males administered 1 mg/kg/week were necropsied on day 25.
Single-dose tolerability studies
The single-dose tolerability of ATR-107 was evaluated in male CD-1 mice (10/group) and in male cynomolgus monkeys (3/group) via IV bolus administration at doses of 0 (vehicle) or 100 mg/kg followed by an 8-week observation period. ATR-107 dosing concentrations were 50 mg/mL. A subset of mice was necropsied on day 17. The study in cynomolgus monkeys was non-terminal, with lymphocyte phenotyping conducted in addition to standard hematology parameters.
13-Week repeat-dose toxicity studies
ATR-107 was administered once weekly for 13 weeks as an IV bolus or SC injection to male and female SD rats and cynomolgus monkeys. ATR-107 was administered at 10, 50, or 250 mg/kg/week (IV), or 10 or 250 mg/kg/week (SC), to SD rats (10/sex/group). Control animals (0 mg/kg/week, 10/sex/group) received vehicle by both routes. Additional animals (5/sex/group) from the 0, 10 (IV), 250 (IV), and 250 (SC) mg/kg/week groups underwent a 13-week recovery phase. A separate group of rats was used for determination of serum ATR-107 concentrations and the presence of ADAs, targeting 3 samples per time point. ATR-107 dosing concentrations were 2, 10, and 50 mg/mL for the 10, 50, and 250 mg/kg dose levels, respectively, in rats. ATR-107 was administered at 10, 30, or 100 mg/kg/week (IV), and 10 or 100 mg/kg/week (SC) to cynomolgus monkeys (3/sex/group). Additional animals (3/sex/group) from the 0, 10 (IV), 100 (IV), and 100 (SC) mg/kg/week groups underwent a 13-week recovery phase. ATR-107 dosing concentrations were 5, 15, and 50 mg/mL for the 10, 30, and 100 mg/kg dose levels, respectively, in cynomolgus monkeys. A full tissue list was collected for microscopic examination from each study; tissues were formalin fixed, embedded, sectioned, and stained with hematoxylin and eosin. Selected rat livers were stained with Masson’s trichrome, or underwent immunohistochemical staining for rat IgG, rat IgM, and complement component 3 (C3); details of the antibodies and methods used in immunohistochemical staining are provided in Table 2. Microscopic findings were recorded using a standard semi-quantitative 1 to 5 severity scale of minimal, mild, moderate, marked, or severe. A pathology peer review was conducted. Serum toxicokinetics (TK) of ATR-107 were evaluated after the first and 13th doses (satellite animals were used in the rat study). Blood was also collected prior to the first dose, and prior to dosing on days 8 (in rats only in the 250 mg/kg/week IV and SC groups) and 92 of the dosing phase, and day 91 during the recovery phase to determine the presence of ADAs (satellite animals were used in the rat study).
Immunohistochemistry targets, antibody sources, working concentrations, and detection systems used to stain selected rat livers.
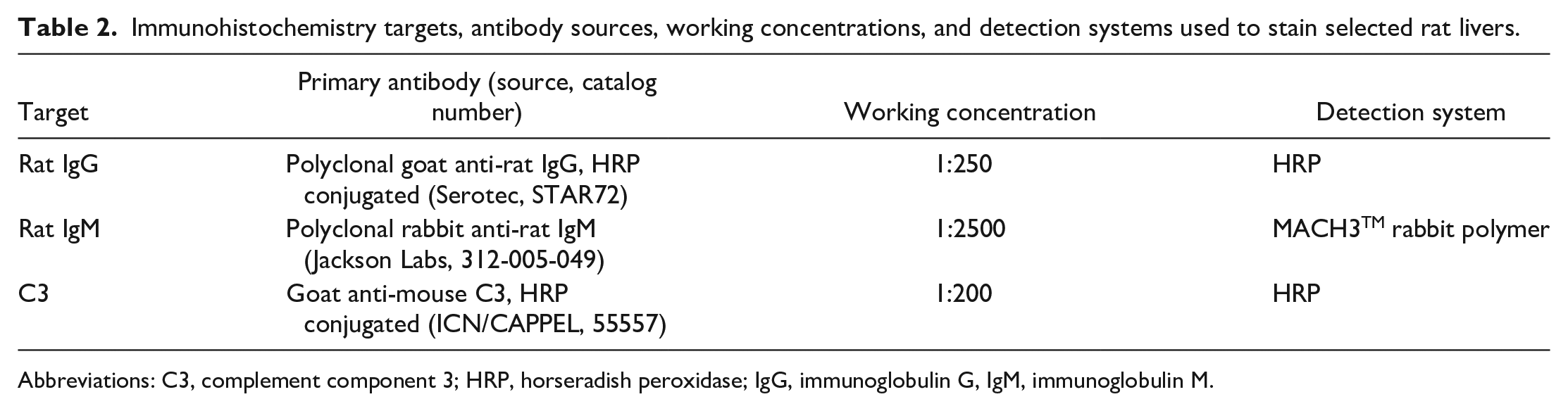
Abbreviations: C3, complement component 3; HRP, horseradish peroxidase; IgG, immunoglobulin G, IgM, immunoglobulin M.
Investigative toxicity studies
Number 1 investigative study in Sprague-Dawley rats to determine the time course of the onset of liver effects
The objective of this study was to further investigate the liver findings noted with ATR-107 in the initial 13-week rat study and determine the timing of the onset of the liver findings. ATR-107 was administered once weekly by IV bolus injection at 1 or 10 mg/kg/week for up to 4 doses to female SD rats (40/group) at concentrations of 0.2 or 2 mg/mL, respectively. Control animals (0 mg/kg/week, 20 animals) received vehicle. Only females were used because it was thought that adequate data could be obtained from one sex. In addition to in-life observations, clinical chemistry (alanine aminotransferase [ALT] and aspartate aminotransferase [AST]), organ weights (kidney, liver, and spleen), and macroscopic and microscopic examinations (liver and kidney) were assessed in this study. Clinical chemistry assessments were limited to ALT and AST because these were the enzymes affected the most in the 13-week study. To evaluate the time course of the liver enzyme changes, animals were bled 6 hours after each dose and 48 hours prior to subsequent doses for measurement of ALT and AST serum enzyme activities. The 6-hour postdose time point was selected based on the acute nature of the liver findings, and the 48-hour predose time point was selected to determine whether any alterations in ALT and AST were transient. A subset of 9 to 10 ATR-107-dosed animals was necropsied 24 hours following each dose administration for microscopic evaluation of the liver and kidneys and serum samples were obtained for measurement of CH50 to assess for complement activation. Blood samples for determination of serum ATR-107 concentrations and the presence of ADAs were collected prior to dosing (ADAs only) and on days 2, 9, 16, and 23 (one day after dosing).
Number 2 investigative study in Sprague-Dawley rats to determine the no-observed-effect level of liver effects
The objective of this study was to test ATR-107 at lower doses in rats to determine the no-observed-effect level (NOEL) for liver findings and also to determine whether any effects would occur in rats after a single dose, which had not been previously evaluated. ATR-107 was administered as a single 1 mg/kg IV bolus injection (10/group), and as once weekly IV bolus injections at 0.01, 0.1, or 1 mg/kg/week (20/group) for up to 4 doses to male SD rats at concentrations of 0.002, 0.02, and 0.2 mg/mL, respectively. Control animals (0 mg/kg/week, 20 animals) received vehicle. Satellite animals were used to assess the TK of ATR-107. Only males were used because it was thought that adequate data could be obtained from one sex. Animals receiving the single dose were terminated on day 16 following collection of blood samples for measurement of ALT and AST serum enzyme activities, and determination of serum ATR-107 concentrations and the presence of ADAs. Clinical chemistry assessments were limited to ALT and AST because these were the enzymes affected the most in the 13-week study. Animals receiving repeat doses were bled 6 hours after the third (20/group) and/or fourth (10/group) doses for measurement of ALT and AST serum enzyme activities; a subset of animals (10/group) was terminated on day 17 (2 days after the third dose), and the remaining 10 animals/group received a fourth dose on day 22 and were terminated on day 24. Blood samples for determination of serum ATR-107 concentrations and the presence of ADAs were collected from repeat-dose toxicity animals prior to dosing and at termination (10/group, days 17 or 24).
Number 3 investigative study in Sprague-Dawley rats to determine whether test article concentration impacted liver effects
ATR-107 was administered by IV bolus at 1 mg/kg once weekly at concentrations of 0.2 or 1.5 mg/mL to female SD rats (8/group) for four doses to investigate whether the delivered concentration of ATR-107 affected the liver findings. Control animals (0 mg/kg/week, 8 animals) received vehicle. Only females were used because it was thought that adequate data could be obtained from one sex. ALT and AST serum enzyme activities were evaluated 24 hours prior to each dose and 6 hours after dosing. Clinical chemistry assessments were limited to ALT and AST because these were the enzymes affected the most in the 13-week study. Animals were necropsied 24 hours after the fourth dose for microscopic evaluation of the liver. Blood samples for determination of serum ATR-107 concentrations were collected at necropsy. Blood samples for determination of the presence of ADAs were collected on day 1 prior to dosing and at necropsy.
Number 4 investigative study in CD-1 mice to determine whether the liver effects occured in another rodent species
Because ATR-107 was only administered in mice as a single dose in the initial tolerability study, the objective of this study was to determine whether liver effects would occur in a rodent species other than rats following repeat dosing at a sufficient duration to allow induction of ADAs. ATR-107 was administered once weekly to male and female CD-1 mice (20/sex/ main study group and an additional 18/sex/group in TK groups giving a total of 76 mice/group) at 0.1, 1, 10, or 250 mg/kg/week (IV bolus), or at 10 or 250 mg/kg/week (SC) for up to 4 doses. Because of mortality, dosing was discontinued after 2 or 3 doses in the 1 mg/kg/week group. ATR-107 dose concentrations were 0.02, 0.2, 2, and 50 mg/mL for the 0.1, 1, 10, and 250 mg/kg dose levels, respectively. Control animals (0 mg/kg/week, 20/sex) received vehicle. A full set of tissues was collected for microscopic evaluation on the last day of dosing, except for males administered 1 mg/kg/week, which were necropsied 3 days after the last dose. Clinical pathology evaluations were originally scheduled but were not performed during the study because of the clinical condition of the animals. Blood samples for determination of serum ATR-107 concentrations and the presence of ADAs were collected terminally from surviving animals at necropsy (1 mg/kg/week IV) or prior to administration of the fourth dose (all other groups) on day 22.
Number 5 investigative study in nude rats to determine the role of T cells in the liver effects
The objective of this study was to determine whether the immune system, and in particular T cells, played a role in the liver findings observed in rats. Nude rats, which are athymic, lack mature T cells, and are unable to mount a T cell-dependent antibody response were used. 25 Female nude rats (20/group) were administered ATR-107 via IV bolus at 0.1, 1, or 10 mg/kg once a week for 3 or 4 doses at concentrations of 0.02, 0.2, and 2 mg/mL, respectively. Control animals (0 mg/kg/week, 20 animals) received vehicle. Only females were used because it was thought that adequate data could be obtained from one sex. Half of the animals from each group were administered ATR-107 for 3 doses and the remaining animals were administered ATR-107 for 4 doses. Clinical chemistry (ALT and AST, approximately 6 hours after the third and/or fourth dose), organ weight (liver only), and macroscopic and microscopic assessments (liver only) were conducted. Clinical chemistry assessments were limited to ALT and AST because these were the enzymes affected the most in the 13-week study. Blood samples were collected terminally at necropsy on days 16 or 23 for determination of ATR-107 concentrations and the presence of ADAs.
Results
Single-Dose Tolerability
In the single-dose tolerability studies in mice or cynomolgus monkeys, ATR-107 was well tolerated at an IV dose of 100 mg/kg, with no impact on mortality, clinical observations, body weight, or clinical pathology parameters. There were no test article-related findings from macroscopic and microscopic evaluations in mice, and no findings from lymphocyte phenotyping in monkeys (necropsy, macroscopic, or microscopic evaluations were not conducted in monkeys).
Thirteen-Week Intravenous and Subcutaneous Administration With 13-Week Recovery
ATR-107 was administered IV (10, 50, or 250 mg/kg) or SC (10 or 250 mg/kg) once weekly for 13 weeks to SD rats. A summary of the end of dosing phase findings is presented in Table 3. One female at 10 mg/kg IV was found dead on day 16 (1 day after the third dose). The cause of death was marked liver necrosis, which was midzonal to diffuse, bridging, and accompanied by little inflammation (Figure 1A-D). Viable hepatocytes around necrotic areas sometimes contained variable numbers of small to large eosinophilic cytoplasmic droplets (Figure 1D). Areas of necrosis were immunopositive for rat IgG and rat IgM, and faintly stained for C3; cytoplasmic droplets were immunopositive for rat IgG (Figure 2A), rat IgM (Figure 2B), and C3.
Summary of ATR-107-related mortality and incidence of liver findings, serum ATR-107 exposure, and incidence of ADAs following IV or SC ATR-107 administration at up to 250 mg/kg/week for 13 weeks in rats: End of dosing phase data (collected between days 85 and 92 depending on the assessment).
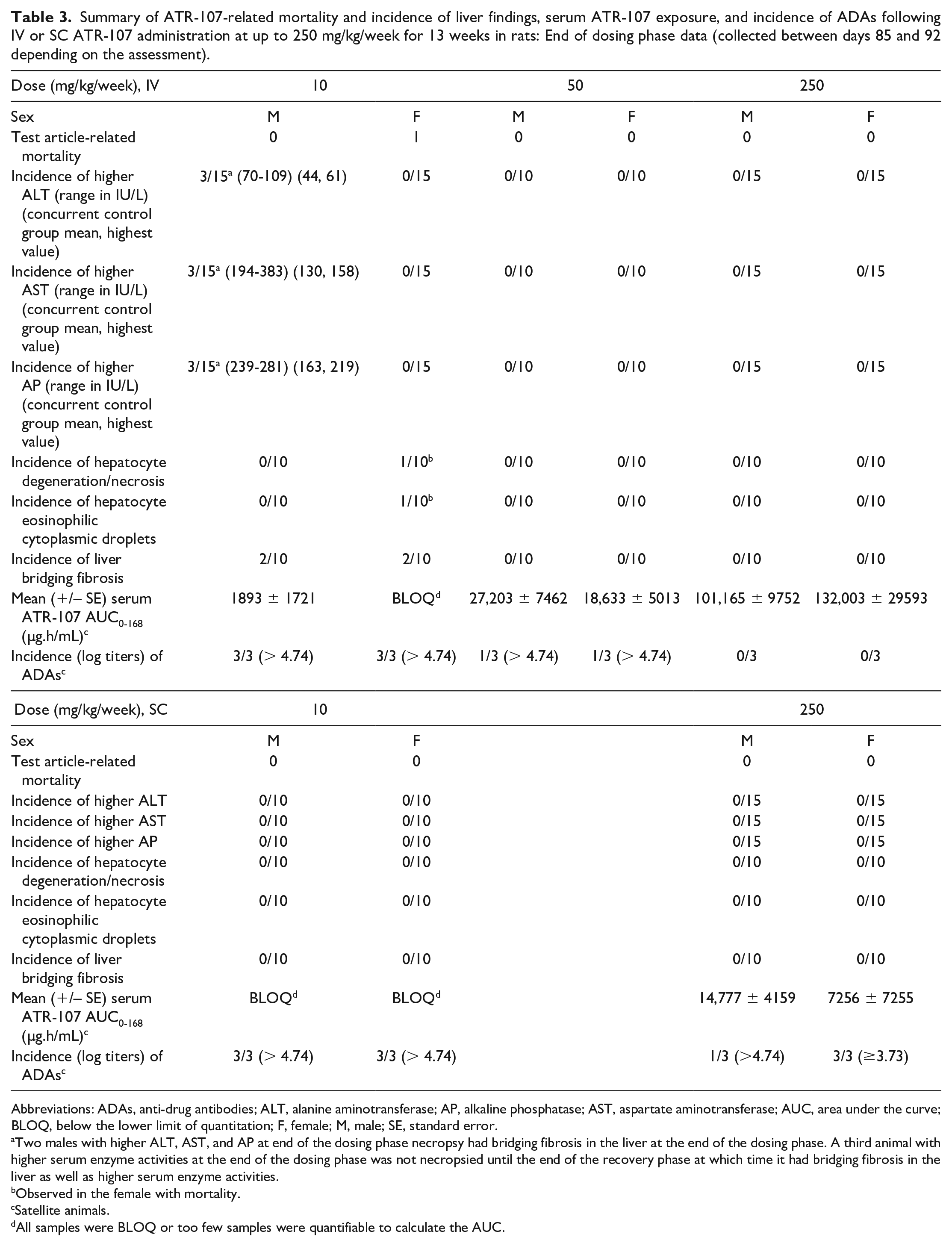
Abbreviations: ADAs, anti-drug antibodies; ALT, alanine aminotransferase; AP, alkaline phosphatase; AST, aspartate aminotransferase; AUC, area under the curve; BLOQ, below the lower limit of quantitation; F, female; M, male; SE, standard error.
Two males with higher ALT, AST, and AP at end of the dosing phase necropsy had bridging fibrosis in the liver at the end of the dosing phase. A third animal with higher serum enzyme activities at the end of the dosing phase was not necropsied until the end of the recovery phase at which time it had bridging fibrosis in the liver as well as higher serum enzyme activities.
Observed in the female with mortality.
Satellite animals.
All samples were BLOQ or too few samples were quantifiable to calculate the AUC.
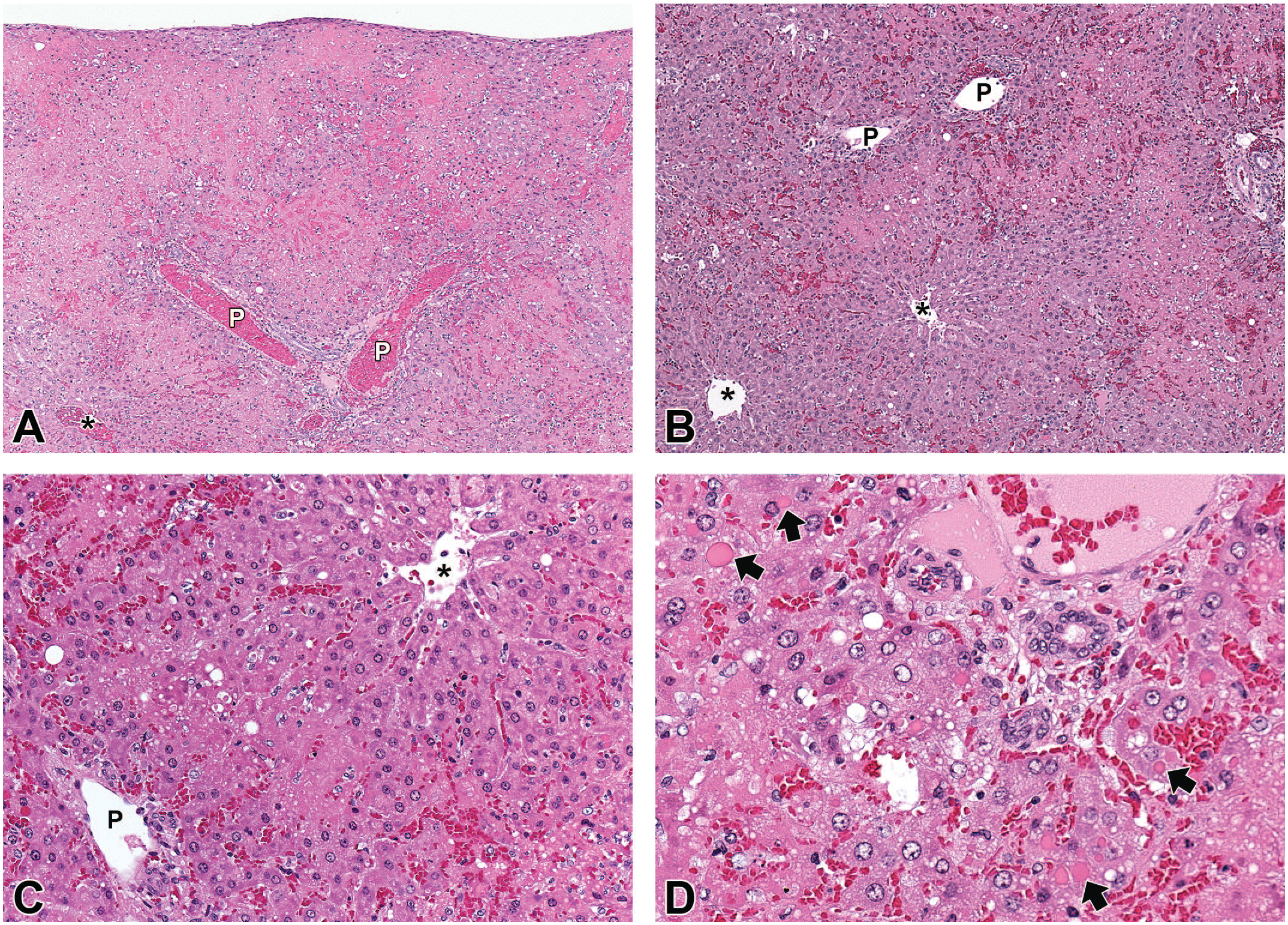
Microscopic liver findings from a rat administered three doses of 10 mg/kg/week ATR-107 IV and found dead on Day 16, the day after the 3rd dose. A, Marked hepatic necrosis with some sparing of hepatocytes adjacent to portal (P) or centrilobular (*) regions. B, Higher magnification showing midzonal necrosis with sparing of hepatocytes adjacent to portal (P) or centrilobular (*) regions. C, Higher magnification showing midzonal necrosis between portal (P) and centrilobular (*) regions and a lack of an inflammatory cell infiltrate. D, Hepatocytes adjacent to a portal region sometimes contain variable numbers of small to large eosinophilic cytoplasmic droplets (some shown by arrows). There is no associated inflammatory cell infiltrate.
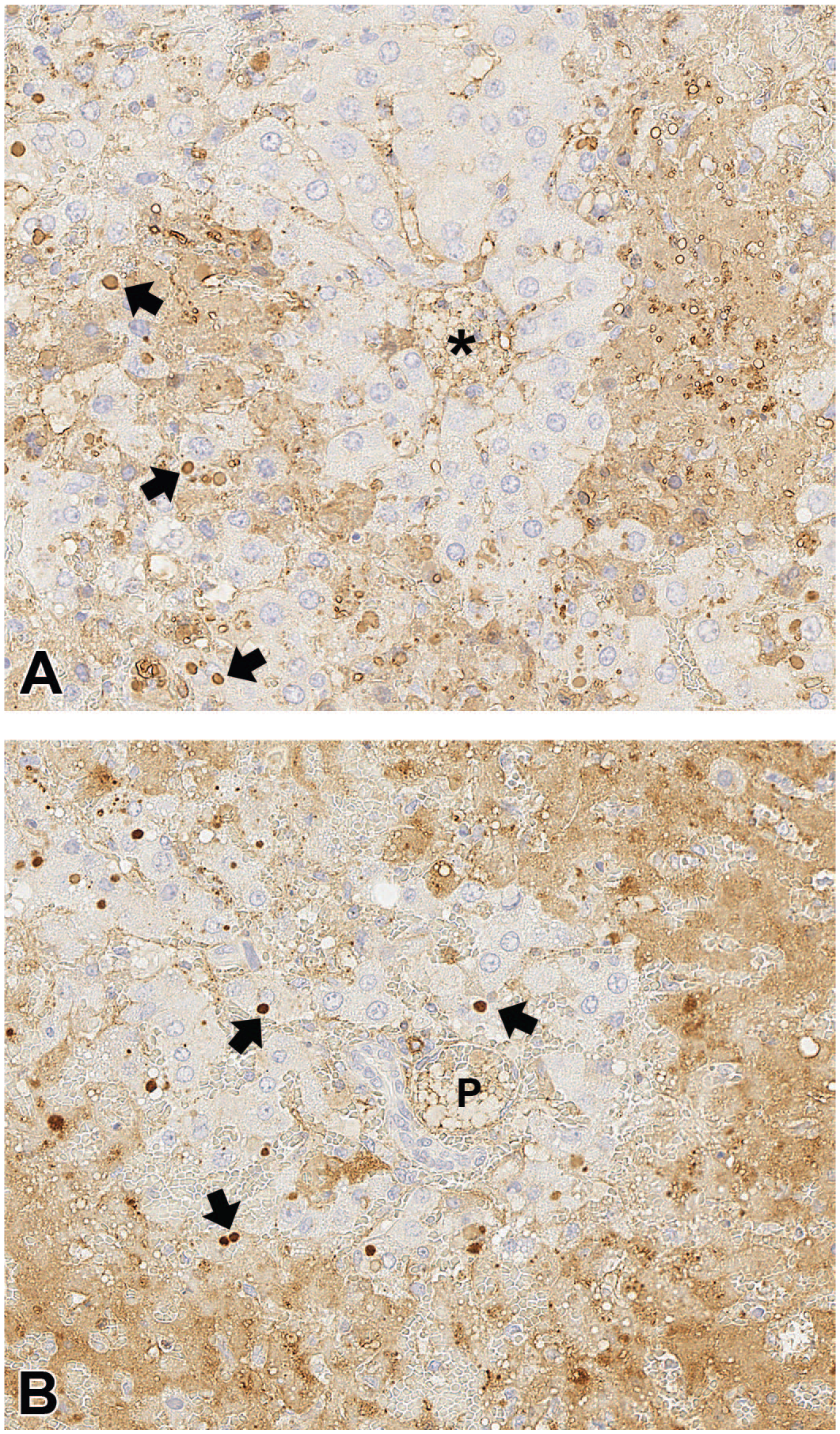
Immunohistochemical staining of liver from a rat administered three doses of 10 mg/kg/week ATR-107 IV and found dead on Day 16, the day after the 3rd dose. A, Immunostaining for rat IgG. A rim of non-necrotic hepatocytes is between the central vein (*) and areas of necrosis. The areas of necrosis are immunopositive for rat IgG (diffuse brown staining). Hepatocytes sometimes contain variable numbers of small to large cytoplasmic droplets (some shown by arrows) which are immunopositive for rat IgG. B, Immunostaining for rat IgM. A rim of non-necrotic hepatocytes is between the portal region (P) and areas of necrosis. The areas of necrosis are immunopositive for rat IgM (diffuse brown staining). Hepatocytes sometimes contain variable numbers of small to large cytoplasmic droplets (some shown by arrows) which are immunopositive for rat IgM.
At the end of dosing phase necropsy, 2 males and 2 females administered 10 mg/kg IV had moderate to marked diffuse bridging liver fibrosis characterized by the presence of fibrous septae that disrupted the normal liver architecture and bridged portal and centrilobular regions with the formation of discrete parenchymal nodules (Figure 3). These nodules contained areas of single-cell hepatocyte necrosis and cytoplasmic vacuolation. Fibrotic areas contained pigment-laden macrophages; bile duct hyperplasia, degeneration, and necrosis; and multifocal areas of eosinophil-rich mixed cell inflammation. Higher serum ALT (70-109 IU/L), AST (194-383 IU/L), and alkaline phosphatase (AP, 239-281 IU/L) values in three males were associated with the liver bridging fibrosis (Table 3); two males were necropsied at the end of the dosing phase and one at the end of the recovery phase. No evidence of immune-complex disease was seen microscopically in the kidney. Rats in the SC and all other IV groups did not have similar findings.
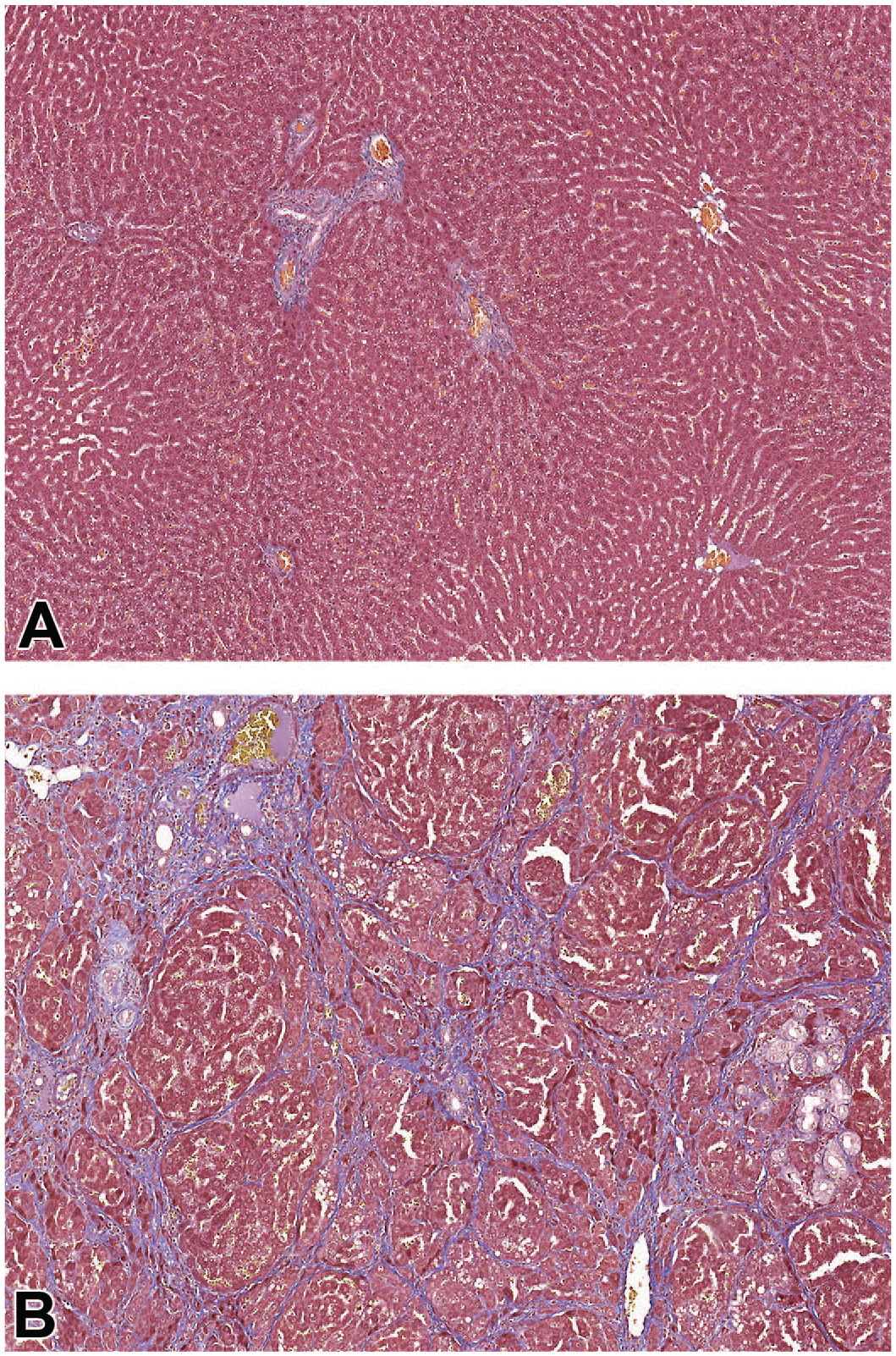
Masson’s trichrome staining of the liver from a rat administered vehicle control (A) or ATR-107 (B) for 13 weeks. A, Normal amounts of collagen (blue staining) in a rat administered the vehicle control IV and SC. B, Fibrosis in a rat administered 10 mg/kg/week ATR-107 IV, with a substantially greater amount of collagen disrupting the normal architecture of the liver with bridging of portal and centrilobular areas and the formation of parenchymal nodules.
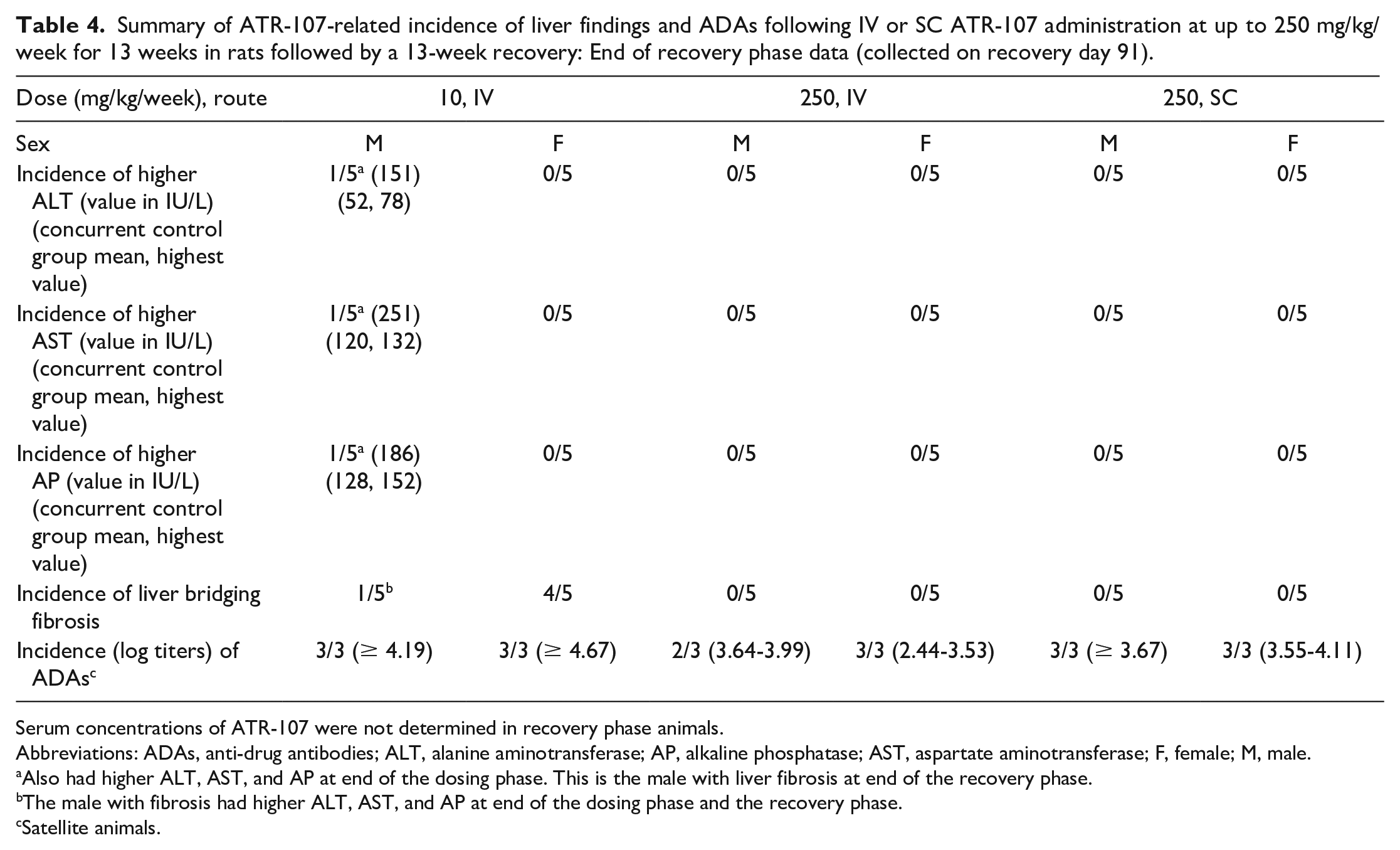
A summary of the end of recovery phase findings is presented in Table 4. At the end of a 13-week recovery phase, minimal to moderate diffuse bridging fibrosis similar to that present at the end of dosing necropsy was present in 1/5 males and 4/5 females at 10 mg/kg IV. In the male, this finding was associated with higher serum ALT (115 IU/L), AST (251 IU/L), and AP (186 IU/L), and this male had higher ALT, AST, and AP at the end of the dosing phase.
Summary of ATR-107-related incidence of liver findings and ADAs following IV or SC ATR-107 administration at up to 250 mg/kg/week for 13 weeks in rats followed by a 13-week recovery: End of recovery phase data (collected on recovery day 91).
Serum concentrations of ATR-107 were not determined in recovery phase animals.
Abbreviations: ADAs, anti-drug antibodies; ALT, alanine aminotransferase; AP, alkaline phosphatase; AST, aspartate aminotransferase; F, female; M, male.
Also had higher ALT, AST, and AP at end of the dosing phase. This is the male with liver fibrosis at end of the recovery phase.
The male with fibrosis had higher ALT, AST, and AP at end of the dosing phase and the recovery phase.
Satellite animals.
Both ATR-107 exposure and ADAs were measured in a separate group of rats. Exposure for 10 mg/kg/week dose IV (in females) or SC (in both sexes) could not be calculated because ATR-107 concentrations were below the lower limit of quantitation (BLOQ). ATR-107 exposures increased with increasing dose in the IV groups. ATR-107 exposure at 250 mg/kg/week SC was lower than what was observed at 50 mg/kg/week IV. ATR-107 exposures at 50 and 250 mg/kg/week IV, and 250 mg/kg/week SC, were higher than the exposure at 10 mg/kg/week IV where liver effects were seen. Values were variable within each group, possibly related to effects from ADAs. No rats had detectable ADAs on Day 8 (prior to the second-dose administration, only 250 mg/kg/week evaluated), while most rats in ATR-107-dosed groups had detectable ADAs by day 92 (end of dosing phase). The incidence of ADAs induction in rats (combined sexes) on day 92 was 100% in the 10 mg/kg/week group (IV), 33.3% in the 50 mg/kg/week group (IV), 0% in the 250 mg/kg/week group (IV), 100% in the 10 mg/kg/week group (SC), and 66.7% in the 250 mg/kg/week group (SC); samples were collected from the control group (IV/SC) on day 1 and did not have detectable ADAs. Animals that were positive for ADAs at the end of the dosing phase had log titers of 3.73 to > 4.74. The presence of detectable ADAs correlated with lower serum ATR-107 exposure; the lower ATR-107 concentrations were interpreted as being related to ADAs-mediated increased clearance or interference with detection and quantitation in the case of neutralizing antibodies. At the end of recovery, serum ATR-107 concentrations were BLOQ of the assay in all but one rat, at 250 mg/kg IV, which had a serum concentration of 187 ng/mL (single time point collection). The incidence of ADAs induction in rats (combined sexes) at the end of the recovery phase was 100% in the 10 mg/kg/week group (IV), 83.3% in the 250 mg/kg/week group (IV), and 100% in the 250 mg/kg/week group (SC). Thus, ADAs were detected at a higher incidence at the end of the recovery phase in the 250 mg/kg/week IV and SC groups compared with the end of the dosing phase.
In cynomolgus monkeys, there were no test article-related findings after IV (10, 30, or 100 mg/kg) or SC (10 or 100 mg/kg) administration of ATR-107 once weekly for 13 weeks. The incidence of ADAs on day 92 of the dosing phase was low in cynomolgus monkeys and was inversely dose related (8.3% and 16.7% at the lowest IV and SC doses, respectively; and 0% at all other IV and SC doses). The NOEL for the IV and SC routes of administration in this study was 100 mg/kg/week, the highest dose administered.
Investigative Studies
Based on the liver findings observed in rats during the 13-week repeat-dose toxicity study (liver necrosis, bridging fibrosis, and higher liver enzymes), a series of investigative toxicity studies were conducted to address specific questions related to the mechanism of the findings. It was considered unlikely that the liver effects resulted from the pharmacologic activity of ATR-107 because: (1) there were high titers of ADAs at 10 mg/kg/week IV while serum ATR-107 concentrations were mostly BLOQ, (2) there were no similar liver findings in animals given ≥50 mg/kg/week IV, or in any SC dose group, and (3) measurable serum ATR-107 concentrations were detected in animals given ≥ 50 mg/kg/week IV, and in the 250 mg/kg/week SC dose group. Instead, it was hypothesized that the findings resulted from consequences of the antibody response to ATR-107 coupled with the IV dosing route, and the investigative studies were designed with this in mind.
Number 1 investigative study in Sprague-Dawley rats to determine the time course of the onset of liver effects
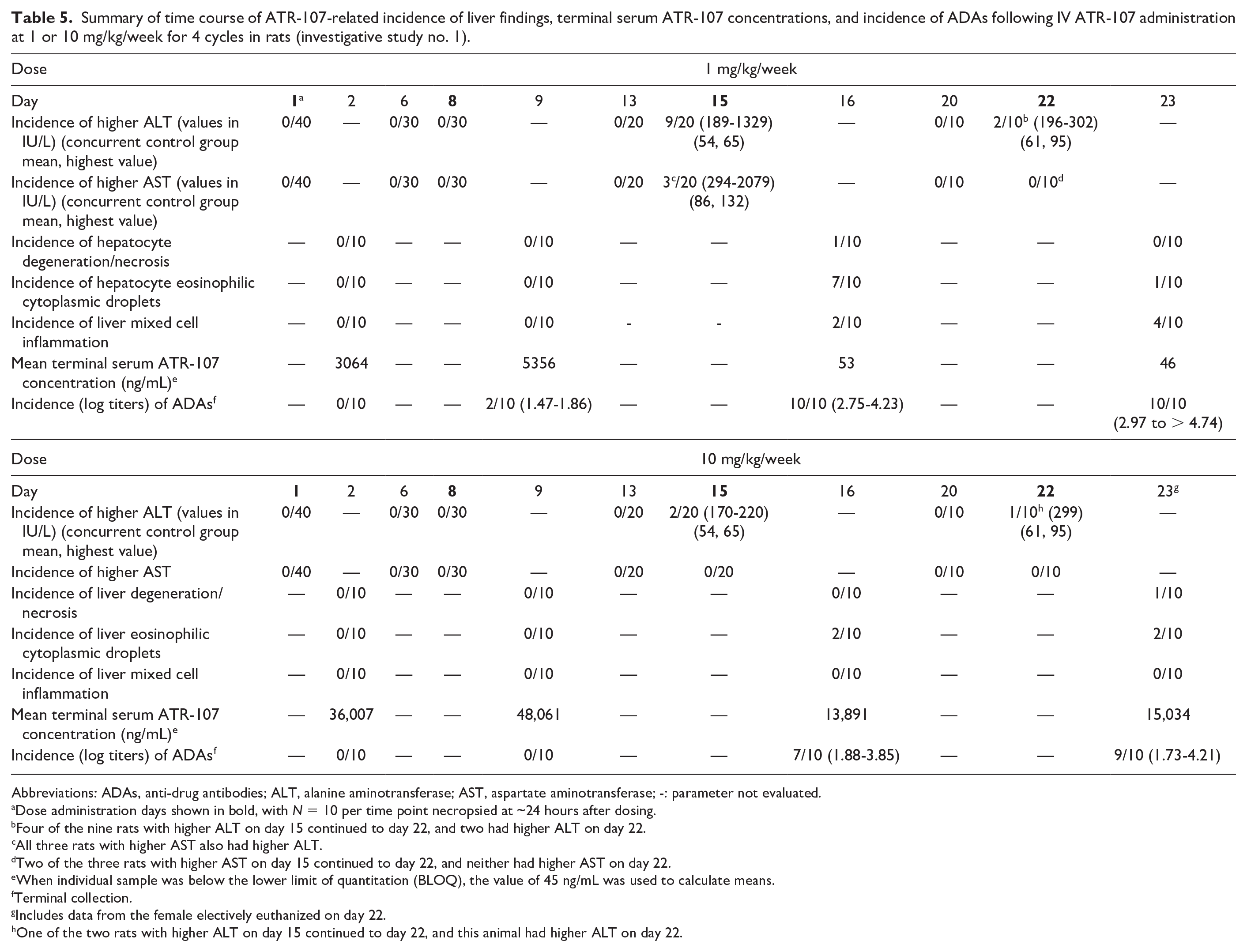
The objective of this investigative study was to determine the timing of onset of the liver findings. A summary of the findings from this study is shown in Table 5. There was 1 ATR-107-related unscheduled death: one 10 mg/kg/week animal was electively euthanized on day 22 approximately 6 hours after administration of the fourth dose; this animal did not have test article-related ALT, AST, or CH50 findings prior to the fourth dose, and blood samples were not taken prior to elective euthanasia from this animal. Clinical signs in this animal included decreased motor activity (recumbency), dyspnea, cool to touch, and pale appearance. Microscopic findings consisted of marked midzonal hepatocellular degeneration/necrosis and eosinophilic cytoplasmic droplets in hepatocytes, similar to what had been seen in the 13-week study in rats.
Summary of time course of ATR-107-related incidence of liver findings, terminal serum ATR-107 concentrations, and incidence of ADAs following IV ATR-107 administration at 1 or 10 mg/kg/week for 4 cycles in rats (investigative study no. 1).
Abbreviations: ADAs, anti-drug antibodies; ALT, alanine aminotransferase; AST, aspartate aminotransferase; -: parameter not evaluated.
Dose administration days shown in bold, with N = 10 per time point necropsied at ~24 hours after dosing.
Four of the nine rats with higher ALT on day 15 continued to day 22, and two had higher ALT on day 22.
All three rats with higher AST also had higher ALT.
Two of the three rats with higher AST on day 15 continued to day 22, and neither had higher AST on day 22.
When individual sample was below the lower limit of quantitation (BLOQ), the value of 45 ng/mL was used to calculate means.
Terminal collection.
Includes data from the female electively euthanized on day 22.
One of the two rats with higher ALT on day 15 continued to day 22, and this animal had higher ALT on day 22.
There were no ATR-107-related CH50 changes at any time, or ALT or AST changes through day 13 (48 hours prior to the third-dose administration), or microscopic liver findings through day 9 (1 day after the second-dose administration, Table 5). ATR-107-related higher ALT was first noted on day 15, after the third dose of 1 or 10 mg/kg, with a higher incidence at 1 mg/kg; a subset of rats with higher ALT also had higher AST. On day 20 (2 days prior to the fourth-dose administration), ALT and AST values in all remaining rats that received ATR-107, including those with higher values on day 15, were comparable with controls, suggesting the higher ALT and AST were transient (Table 5). On day 22, after the fourth-dose administration, ALT and/or AST were again higher in a subset of rats. Higher ALT and/or AST were associated with liver microscopic findings of hepatocellular degeneration, necrosis, and eosinophilic cytoplasmic droplets, and/or mixed cell inflammation in some, but not all, rats (Table 5). In addition, a few rats had microscopic changes in the liver but no changes in liver enzymes. The liver findings were morphologically similar to those seen in the 13-week rat study. The rat with the highest ALT (1329 IU/L and AST (2079 IU/L) on day 15 was the one with hepatocyte degeneration/necrosis on day 16. The liver sections were stained with antibodies for rat IgG, IgM, and C3. The necrotic areas were immunopositive for rat IgG and IgM, and faintly stained for C3. Cytoplasmic droplets were immunopositive for rat IgG, rat IgM, and C3. Mixed cell inflammation in the liver consisted primarily of lymphocytes and macrophages with fewer numbers of eosinophils and neutrophils and occasional mast cells; this finding was sometimes accompanied by slight bile duct hyperplasia. There were no ATR-107-related organ weight changes in the liver, kidney or spleen, and no ATR-107-related microscopic findings in the kidney (spleen was not examined microscopically).
Serum ATR-107 concentrations increased with increasing dose following the first and second doses; however, concentrations after the third dose were generally much lower. Prior to the third dose on day 15, a few animals had low titers of ADAs, whereas after the third (day 15) and fourth (day 22) doses almost all animals had ADAs with high titers. In most samples, the presence of ADAs correlated with substantially lower serum ATR-107 concentrations; the lower ATR-107 concentrations were interpreted as being related to ADAs-mediated increased clearance or interference with detection and quantitation in the case of neutralizing antibodies. In conclusion, induction of ATR-107-related liver findings required at least 3 weekly doses of ATR-107 while induction of ADAs required at least 2 weekly doses with a marked increase in incidence after 3 weekly doses.
Number 2 investigative study in Sprague-Dawley rats to determine the no-observed-effect level of liver effects
The objectives of this investigative study were to identify the NOEL for the liver findings in rats, and to determine whether any liver effects occurred in rats after a single dose. There were no changes in ALT or AST on day 16 after a single dose of ATR-107. The incidences of ATR-107-related liver enzyme changes in this study after repeat weekly dosing are summarized in Table 6. There were no ATR-107-related ALT or AST changes at 0.01 mg/kg/week. At 0.1 or 1 mg/kg/week, higher ALT was first noted on day 15, after the third-dose administration with a higher incidence at 1 mg/kg/week, and a subset of rats with higher ALT also had higher AST. Similar findings were seen on day 22. Quantifiable concentrations of ATR-107 were detected after the first dose; the exposure increased with increasing doses between 0.01 and 1 mg/kg. After the third or fourth doses, ATR-107 concentrations decreased and were BLOQ for all animals at 0.01 and 0.1 mg/kg/week, and most animals at 1 mg/kg/week; because of lack of quantifiable ATR-107 concentrations in most animals at most time points, the AUC was not calculated. The lower ATR-107 concentrations were interpreted as being related to ADAs-mediated increased clearance or interference with detection and quantitation in the case of neutralizing antibodies. ADAs were detected in 70% of animals administered a single dose of ATR-107 on day 16. ADAs were detected in many animals administered three weekly doses of 0.01 mg/kg/week, and in most animals administered 4 weekly doses of 0.01 mg/kg/week. ADAs were detected in all animals administered 0.1 or 1 mg/kg/week after either 3 or 4 doses. In conclusion, ATR-107-related higher serum ALT and AST serum enzyme activities occurred after 3 weekly IV doses of ATR-107, and the NOEL was 0.01 mg/kg/week; no changes in ALT or AST were observed on day 16 after a single IV dose of 1 mg/kg ATR-107 despite induction of ADAs.
Summary of ATR-107-related incidence of ALT and AST changes, serum ATR-107 concentrations, and incidence of ADAs following IV ATR-107 administration at 0.01, 0.1, or 1 mg/kg/week for 3 or 4 weeks in rats (investigative study no. 2).

Abbreviations: ADAs, anti-drug antibodies; ALT, alanine aminotransferase; AST, aspartate aminotransferase; AUC, area under the curve; BLOQ, below the lower limit of quantitation.
The rat with higher ALT on day 15 did not continue to day 22; this was a different rat.
Two of the five rats with higher ALT on day 15 continued to day 22, and both had higher ALT on day 22.
Animals with higher AST also had higher ALT.
All samples were BLOQ or had too few quantifiable samples to calculate the AUC.
Terminal collection from 10/sex/group.
Number 3 investigative study in Sprague-Dawley rats to determine whether test article concentration impacted liver effects
ATR-107 was administered at 1 mg/kg/week for 4 weeks at concentrations of 0.2 or 1.5 mg/mL to investigate whether the delivered concentration affected the liver effects. There were no changes in ALT or AST after the first or second weekly dose. After the third and fourth weekly doses, ATR-107-related higher ALT was observed (Table 7). A total of 3 of the 8 animals administered ATR-107 at 0.2 mg/mL were affected, and a total of 2 of the 8 animals administered ATR-107 at 1.5 mg/mL were affected (6 hours after dosing). Higher ALT was not seen in any animal at 24 hours before the fourth dose, indicating recovery in the 6 days after the third dose. Higher AST values were not observed in any ATR-107-dosed animals. All animals with higher ALT after the third and/or fourth dose had ATR-107-related microscopic findings of slight periportal mixed inflammation and slight eosinophilic cytoplasmic droplets in hepatocytes at necropsy 24 hours after the fourth dose, and in addition, six other animals that did not have higher ALT had these microscopic findings. There were no differences in the microscopic findings in these six animals compared with those that had higher ALT, including severity, to suggest why only some animals had higher ALT. The incidences of higher ALT values and microscopic findings in the liver were similar at both ATR-107 dosing concentrations. Serum concentrations of ATR-107 on day 24 in most of the rats dosed with ATR-107 were BLOQ, which was interpreted as being related to the clearance of ATR-107 by ADAs or interference with detection and quantitation in the case of neutralizing antibodies; the incidence of ADAs was 100% (8/8) with both dose concentrations.
Summary of incidence of ATR-107-related ALT changes, microscopic findings, and ADAs following IV ATR-107 administration of 1 mg/kg/week for 4 weeks at two different concentrations in rats (investigative study no. 3).
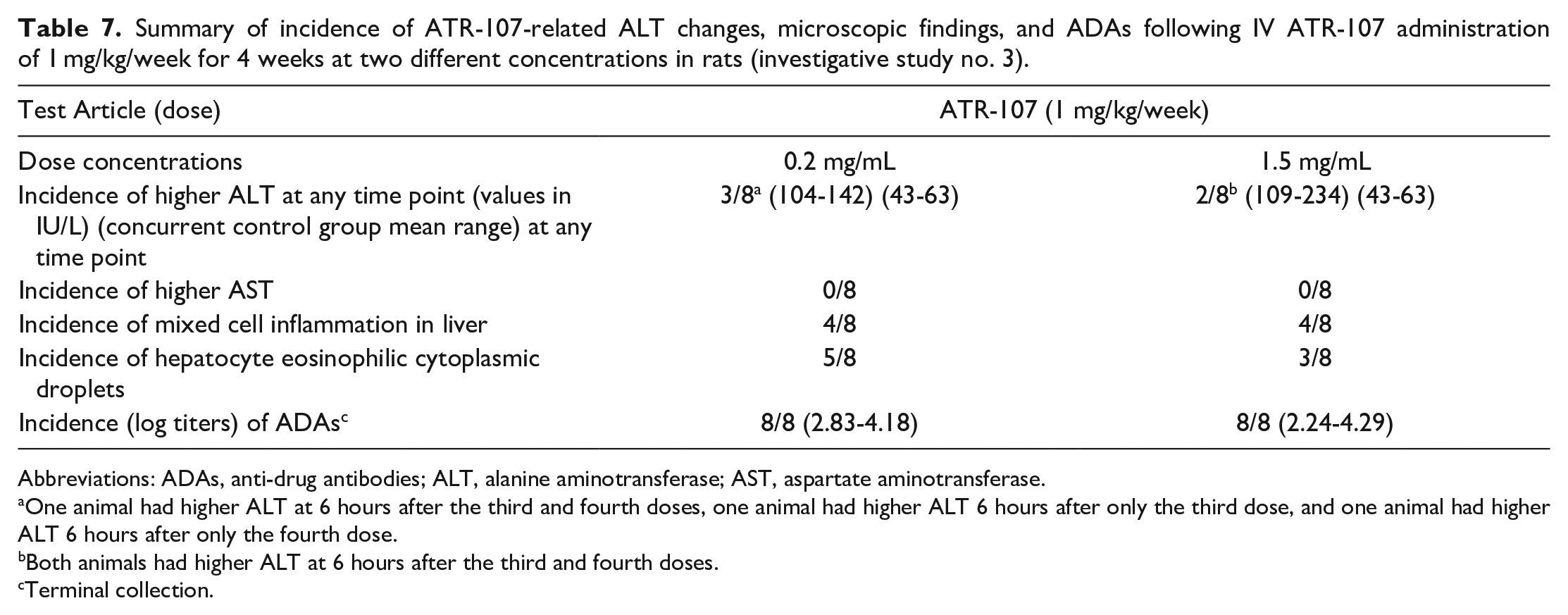
Abbreviations: ADAs, anti-drug antibodies; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
One animal had higher ALT at 6 hours after the third and fourth doses, one animal had higher ALT 6 hours after only the third dose, and one animal had higher ALT 6 hours after only the fourth dose.
Both animals had higher ALT at 6 hours after the third and fourth doses.
Terminal collection.
Number 4 investigative study in CD-1 mice to determine whether the liver effects occured in another rodent species
ATR-107 was administered to CD-1 mice at doses and routes similar to those in previous rat studies. Beginning after the second weekly dose, and typically within 4 minutes to 1 hour of ATR-107 administration, mortality occurred in animals administered ATR-107 by the IV route (Table 8). Of the 76 mice in each group (including TK animals), the highest incidence of mortality was at 1 mg/kg IV (20/76), with lower incidence at 0.1 (5/76), 10 (4/76), and 250 (1/76) mg/kg/week IV, thus forming a bell-shaped curve centered at 1 mg/kg/week IV (Figure 4); there was no mortality in the groups administered 10 or 250 mg/kg/week SC. In unscheduled deaths, the rapid onset of clinical signs (including decreased motor activity, convulsions, ataxia, dyspnea, hunched posture, cool to touch, ptosis, tachypnea, and bradypnea) and/or death after a minimum of 2 doses, and the inability to sample blood from most affected animals (suggesting collapse of vascular tone) was consistent with anaphylaxis from an immune response to ATR-107 (type I hypersensitivity). In some animals administered 1 mg/kg/week with mortality, ATR-107-related mild to moderate thrombi were seen in the lungs with increased intravascular neutrophils, although other animals with mortality at this dose, and animals at other doses, did not have these findings. In the IV-dosed animals that survived to the final necropsy, there were no ATR-107-related effects on body weight, food consumption, or on macroscopic or microscopic findings. However, surviving animals administered 0.1 or 1 mg/kg/week did have clinical signs after the second and third doses that were similar to those observed in animals with mortality. In contrast to the findings in IV-dosed groups, there were no deaths or ATR-107-related clinical signs or effects on body weight, food consumption, injection sites, or macroscopic and microscopic findings in the 10 or 250 mg/kg/week SC dose groups. ADAs were detected in 54/70 animals (77%) at 0.1 mg/kg/week IV, in 41/56 animals (73%) at 1 mg/kg/week IV, in 5/71 animals (7%) at 10 mg/kg/week IV, and in 0/74 animals (0%) at 250 mg/kg/week IV; and in 16/74 animals (22%) at 10 mg/kg/week SC and in 0/70 animals (0%) at 250 mg/kg/week SC. In summary, ATR-107 was immunogenic in mice and multiple IV doses at ≥ 0.1 mg/kg/week resulted in apparent anaphylaxis with a bell-shaped dose-response curve centered at 1 mg/kg/week IV.
Summary of incidence of unscheduled deaths and ADAs following IV and SC ATR-107 administration at up to 250 mg/kg/week in mice (investigative study no. 4).

Abbreviation: ADAs, anti-drug antibodies.
Collected terminally at necropsy (1 mg/kg/week IV) or prior to administration of the fourth dose (all other groups).
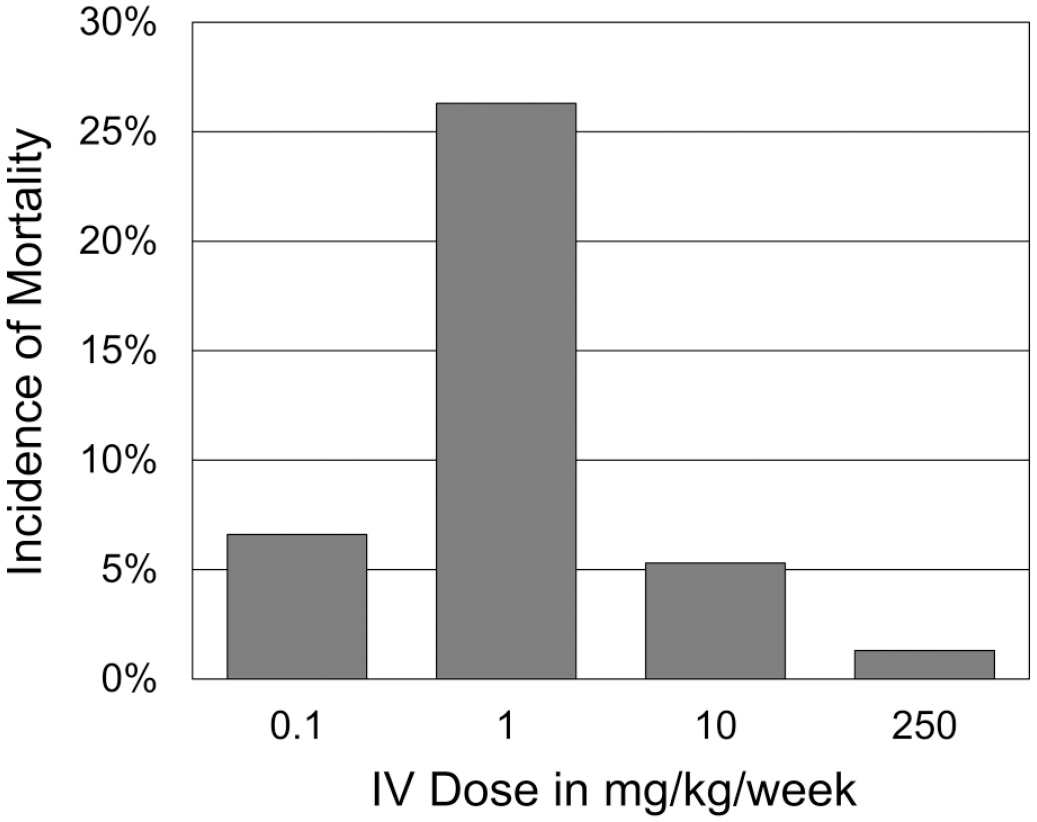
Incidence of ATR-107-related mortality in mice administered ATR-107 at 0.1, 1. 10, or 250 mg/kg/week IV in a 4-week toxicity study. The highest incidence of mortality was at 1 mg/kg/week, with lower incidences at lower and higher doses, thus forming a bell-shaped curve centered around 1 mg/kg/week. The mortality began after the second weekly dose, and typically occurred within 4 minutes to 1 hour of ATR‑107 administration. Mortality was consistent with anaphylaxis from an immune response to ATR‑107 (type I hypersensitivity). No mortality was observed after SC dosing at 10 or 250 mg/kg/week.
Number 5 investigative study in nude rats to determine the role of T cells in the liver effects
ATR-107 was administered via weekly IV bolus to female nude rats at 0, 0.1, 1, or 10 mg/kg/week (doses at which ATR-107-related liver effects were seen in SD rat studies) for 3 or 4 weeks. There were no ATR-107-related clinical observations, effects on body weight, or alterations in ALT and AST. At the 3-week and 4-week necropsies, there were no ATR-107-related microscopic liver findings (negative data not shown). Serum ATR-107 concentrations increased with dose (individual concentration data included in Figure 5, further described below). No ADAs were detected. In conclusion, ATR-107 did not elicit liver findings in nude rats, unlike the results in wild-type SD rats, indicating that a functional immune system, and in particular functional T cells, were necessary for development of the effects. The lack of ADAs in nude rats suggests that the development of these antibodies requires functional T cells. Taken together, these data suggest that functional T cells and an adaptive immune response with development of ADAs were necessary for development of ATR-107-related liver findings.
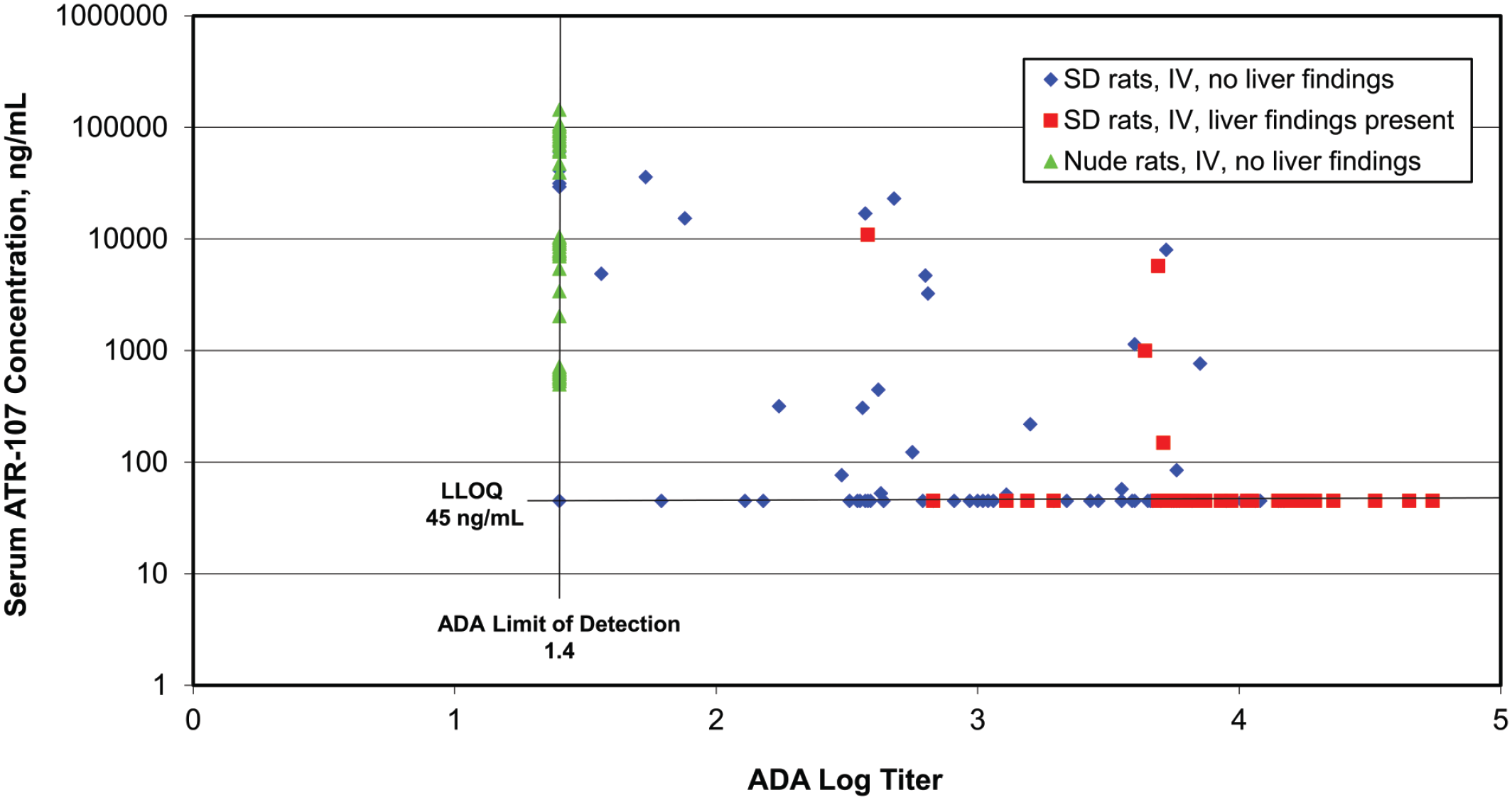
Association of liver findings, ATR-107 exposure, and anti-drug antibody (ADA) titers in SD rats from investigative studies #1, #2, and #3 and nude rats from #5 investigative study administered ATR‑107 at 0.01 to 10 mg/kg/week IV for 3 or 4 doses across multiple studies. Liver findings (liver enzyme elevations or microscopic findings) were observed only in Sprague Dawley (SD) rats with ADA titers at or above 2.57 (red squares), and most of these rats had serum ATR-107 concentrations at or below the lower limit of quantitation (LLOQ); concentrations below the LLOQ are represented as being at the LLOQ of 45 ng/mL on the graph. Nude rats (green triangles) had no detectable ADAs, high serum ATR‑107 exposures, and lacked liver findings. Many SD rats without liver findings (blue diamonds) had detectable serum ATR‑107 concentrations and lower titers of ADAs.
Incidence of ATR-107-Related Liver Findings in Sprague-Dawley Rats by Dose Across Studies
The incidence of SD rats with ATR-107-related liver findings, based on the presence of ATR-107-related clinical chemistry or microscopic findings at any time point, was summarized across studies as shown in Figure 6. Only rats administered 3 or more weekly doses were included because there were no ATR-107-related liver findings prior to this. The number of rats for analysis (males and females combined) at each dose level was 20 (0.01 mg/kg/week, all from no. 2 investigative study), 20 (0.1 mg/kg/week, all from no. 2 investigative study), 56 (1 mg/kg/week, from no. 1, 2, and 3 investigative studies), 50 (10 mg/kg/week, from 13-week and no. 1 investigative studies), 20 (50 mg/kg/week, all from 13-week study), and 30 (250 mg/kg/week, all from 13-week study). As shown in Figure 6, the incidence formed a bell-shaped curve centered at 1 mg/kg/week. This curve is similar to that formed by the incidence of mortality seen in mice administered ATR-107 IV for up to 4 weeks (Figure 4).
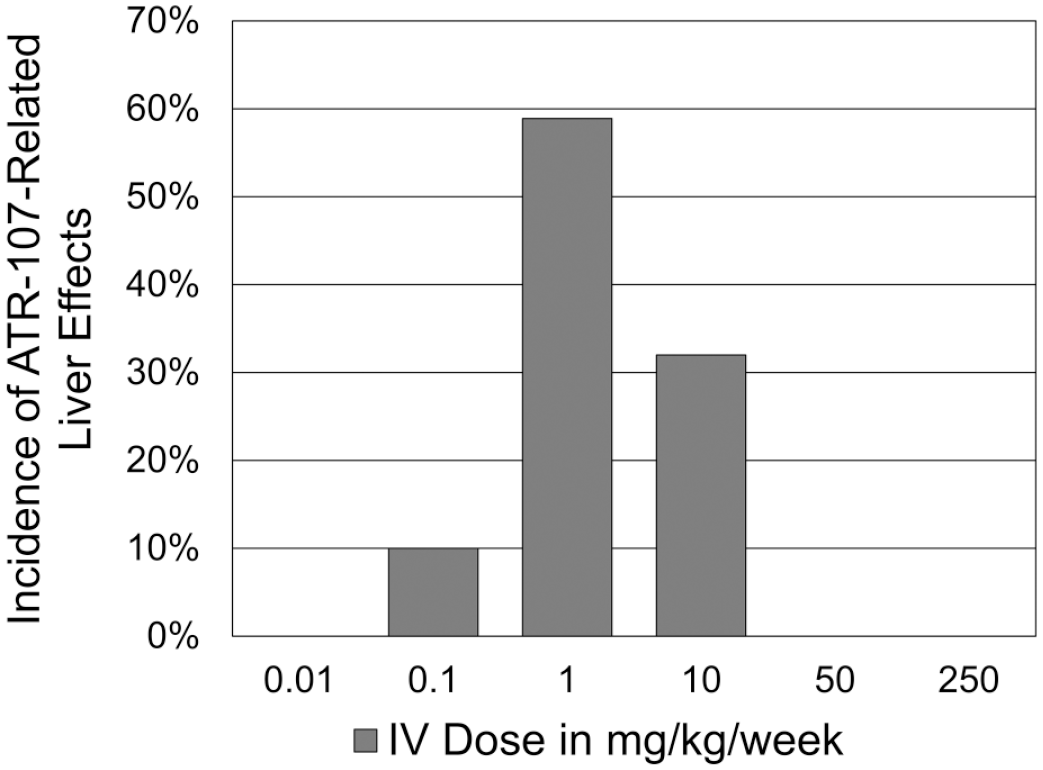
Incidence of ATR-107-related liver findings based on the presence of ATR 107-related clinical chemistry or microscopic findings at any time point. Data were summarized across all rat studies. Only rats administered 3 or more weekly doses were included. The incidence forms a bell-shaped curve centered at 1 mg/kg/week. There were no ATR-107-related effects when administered by the SC route.
Impact of ATR-107 Exposure and Anti-Drug Antibodies on Liver Effects in Sprague-Dawley and Nude Rats
An analysis of the data generated across the investigative studies in SD and nude rats-dosed IV was conducted to determine whether there was a relationship between the liver effects and the ATR-107 exposure or ADAs titers from individual animals where all this information was available (Figure 5). This analysis revealed several important points. The incidence of ATR-107-related liver findings (which were observed only in SD rats administered ATR-107 after at least 3 weekly doses, and only by the IV route) increased with increasing ADAs titers but not ATR-107 exposure, suggesting that the liver findings were more directly related to ADAs titers and not serum ATR-107 concentrations. All rats with ATR-107-related liver findings were SD and had ADAs log titers > 2.5 (i.e. higher antibody titers against ATR-107), whereas no rats (SD or nude) with non-detectable or lower ADAs titers (log titers < 2.5) had ATR-107-related liver findings. In most rats with ATR-107-related liver findings, serum ATR-107 concentrations were at or below the lower limit of quantitation (BLOQ), whereas in rats with no ATR-107-related liver findings, serum concentrations of ATR-107 ranged from BLOQ to > 10 000 ng/mL. Thus, higher ADAs titers were necessary for development of ATR-107-related liver findings, whereas measurable ATR-107 serum concentrations were not.
Tissue Cross-Reactivity Results
Tissue cross-reactivity studies were conducted using mouse, rat, cynomolgus monkey, and human tissues; mouse tissues were only evaluated in a non-GLP study. Overall, the tissue cross-reactivity assessment was problematic. Staining of negative control material could not be eliminated; such issues with tissue cross-reactivity studies are well known. 18 Staining with ATR-107 was observed in all tissues (i.e. ≥ 34 tissues/species), and was seen in the cytoplasm, in cytoplasmic granules, and on the cell membrane; in the liver staining was seen in hepatocytes and leukocytes.
Discussion
Weekly administration of the fully human anti-human IL-21R mAb ATR-107 to rats for up to 13 weeks resulted in adverse liver necrosis, diffuse bridging fibrosis, and higher liver enzyme activity in some rats in the low-dose IV group (10 mg/kg), but not in the 50 or 250 mg/kg IV groups, and not following SC administration despite overlapping systemic concentrations of test article. Similar findings were not seen in the 13-week study in cynomolgus monkeys. A series of investigative studies were conducted in rats and mice to better understand the time course and the lowest effect level of the liver findings, the potential impact of dose concentrations, and the role of an intact immune system in the development of the liver effects. In SD rats, adverse ATR-107-related findings in the liver occurred only after at least 3 weekly doses, occurred in a bell-shaped dose-response centered around 1 mg/kg, and required the presence of high ADAs titers. The findings were not impacted by different ATR-107 dose concentrations. When nude rats were administered ATR-107 IV, there were no detectable ADAs, and liver effects were not observed despite achieving high serum ATR-107 exposures. An analysis of the data generated across the rat studies showed higher ADAs titers were necessary for development of ATR-107-related liver findings, whereas measurable ATR-107 serum concentrations were not. A wide range of doses of ATR-107 were also evaluated in a repeat-dose study in another rodent species (CD-1 mice). ATR-107-related mortality in mice occurred in a bell-shaped curve centered at 1 mg/kg/week, occurred after at least 2 weekly IV doses, was consistent with anaphylaxis, and was observed at all IV doses but not at either SC dose tested. ADAs were observed in the mice by the IV route at high titers, and less frequently by the SC route.
In the 13-week study, in the 10 mg/kg/week IV group, the incidence of microscopic changes in the liver at the end of the dosing phase was 25% (males and females combined), whereas at the end of the recovery phase the incidence in this group was 50% (males and females combined). We do not believe that the higher incidence at the end of the recovery phase represents a delayed effect on the liver because clinical chemistry changes suggestive of liver injury (higher ALT, AST, and/or AP) appeared to occur just after dosing. However, additional experiments would be necessary to further evaluate this.
Taken together, the results indicate several important points regarding the mechanism of ATR-107-related liver findings in rats. (1) The liver findings in rats do not appear to be directly related to ATR-107 inhibiting IL-21R, because the effects were not seen after SC administration at any dose, including those that provided equivalent or higher systemic exposures compared with IV doses that resulted in liver effects, and effects were not seen in nude rats despite high systemic ATR-107 exposures. (2) The liver findings in rats dosed by the IV route appear to be related to an adaptive immune response against the human antibody ATR-107, because the findings were only evident after multiple weekly doses, the findings occurred in a bell-shaped curve as related to dose, a strong ADAs response was detected in affected rats, and there was an absence of ATR-107-related liver findings in nude rats, which did not develop ADAs. (3) The presence of ADAs was not always associated with ATR-107-related liver effects. Many rats with ADAs did not develop liver findings after multiple IV or SC doses, although, all rats with liver findings that were also tested for ADAs had high log titers (>2.57). These results indicate that ADAs were necessary, but not alone sufficient, to cause the liver effects observed in rats by the IV route. The ADAs in rats were IgG and IgM, with IgG predominating (data not shown). (4) Liver effects were observed only after the IV route of administration and within a specific dose range. Very low or very high IV doses did not result in liver effects in rats, and there were no liver effects in rats following ATR-107 administration by the SC route. (5) Based on liver enzymes being transiently higher just after repeat IV dosing, effects on the liver appear to likely be associated with the bolus IV delivery of ATR-107 into the circulation of sensitized rats. (6). ALT was more consistently observed being higher compared with AST. Overall, we hypothesize that the IV bolus delivery of ATR-107 (i.e. the antigen) allowed rapid interaction with existing ADAs and formation of immune complexes. We believe that the large number of immune complexes formed by the IV bolus overwhelmed immune-complex clearance mechanisms and led to the effects observed when present in the correct stoichiometric ratio, given that lower or higher ATR-107 doses did not cause liver effects. As the immune complexes were cleared, serum ATR-107 concentrations were reduced and ongoing liver injury ceased, allowing liver enzymes to return to lower or normal values. Unfortunately, we did not determine whether circulating immune complexes were present on our studies.
While liver necrosis and fibrosis has not been reported very often following administration of therapeutic proteins in the context of efficacy or toxicity studies, similar morphologic findings have been described in several species following administration of various nontherapeutic foreign proteins to sensitized animals using a variety of conditions and species. 28 In experiments somewhat analogous to IV injection of a therapeutic protein, IV injection of human serum albumin (HSA) into sensitized rats resulted hepatocyte necrosis4,5,12 which was sometimes described as midzonal and periportal, 12 or midzonal and sparing hepatocytes adjacent to portal tracts and central veins. 5 Antigen, rat gamma globulin, and complement were detected in the liver, concentrated in damaged areas.11,12 Vacuolation and eosinophilic bodies were seen in the cytoplasm of parenchymal cells, and were interpreted to be enlarged lysosomes. 5 It was thought that liver damage was the result of an antigen-antibody reaction.4,5,11,12,28 Repeated administration of HSA resulted in scarring of the liver with lobular distortion and fibrosis bridging portal and centrilobular areas.5,11 The necrosis was thought to precede fibrosis. 5 Other investigators have also shown liver fibrosis following intraperitoneal administration of heterologous serum, such as porcine serum, in sensitized rats, although hepatocyte necrosis was not considered to play a major role in the porcine serum model.1,2,22,26 The development of an immune response was necessary for the development of liver fibrosis in the porcine serum model, as rats tolerized to porcine serum did not develop liver fibrosis, although the authors were not able to further determine the roles of cellular versus humoral responses. 3 Overall, these findings are similar to those seen in rats administered ATR-107 which had liver necrosis; fibrosis with disruption of the normal hepatic architecture; eosinophilic cytoplasmic bodies in hepatocytes; and rat IgG, rat IgM, and C3 in necrotic areas. These findings in our studies were associated with development of an immune response to ATR-107 based on the lack of similar findings in immunodeficient nude rats.
Other species have also been reported to have similar findings in the liver following injection of foreign proteins, although again not in the context of administering a therapeutic protein. Injection into the portal circulation of soluble complexes of HSA and anti-HSA into normal rabbits, or injection of HSA in hyperimmunized rabbits both resulted in midzonal liver necrosis within 24 hours with scanty cellular infiltrates. 28 The necrosis ranged from small foci to large diffuse areas involving several lobules, but cells closest to the portal areas or central veins were almost invariably spared. Both experiments were considered to represent a condition of antigen excess. The morphologic findings described in these rabbits are very similar to those seen in the rats in our studies, in that the necrosis ranged from small foci to larger regions, and that cells near portal and centrilobular regions were spared. It is also possible that IV injection of ATR-107 would result in a short-term condition of antigen excess, whereas SC administration would not, although the ADAs response of individual animals would impact the relative excess. However, if antigen excess is necessary to cause the liver injury, it is unclear why the findings were inversely related to dose, as higher IV doses of ATR-107 would be expected to result in greater levels of antigen excess. It is possible that the stoichiometric ratio of antigen (ATR-107) and ADAs may play a role, as the stoichiometric ratio of antigen and antibody is known to play a role in deposition of antigen:antibody complexes.21,24,29
In addition to liver injury associated with injection of foreign nontherapeutic proteins, hepatotoxicity has occasionally been described following administration of therapeutic proteins in monkeys. Liver enzyme elevations, hepatocellular necrosis and vacuolation, and associated inflammation were reported following administration of the rhuApoL/TRAIL trimeric antibody to cynomolgus monkeys. 33 Liver toxicity was only observed in monkeys that developed ADAs and the incidence of rhuApoL/TRAIL-induced hepatotoxicity rose with as the number of dosing cycles increased. Investigative studies suggested that cynomolgus anti-rhuApoL/TRAIL antibodies promoted ligand crosslinking and subsequent aggregation of death receptors, thereby leading to apoptosis of cynomolgus hepatocytes.
IL-21 receptor, the target of ATR-107, is not present in hepatocytes. Overall, the tissue cross-reactivity assessment with ATR-107 was problematic. Staining of negative control material could not be eliminated; such issues with tissue cross-reactivity studies are well known. 18 Staining with ATR-107 was observed in all tissues; in the liver, staining was seen in hepatocytes and leukocytes. However, because findings in rats occurred only after repeated weekly doses and were not associated with exposure, and because there were no test article-related findings in the numerous other tissues that showed staining, we believe that it is unlikely that direct binding of ATR-107 to hepatocytes or other cells, such as leukocytes, led to hepatocyte necrosis in rats. Similarly, we believe that the findings in mice were consistent with anaphylaxis and not an event secondary to direct binding of ATR-107 to a specific tissue element or elements based on the mortality not occurring until after the second dose, occurring at a lower incidence at higher IV doses, not occurring after SC doses, and being associated with development of ADAs. In addition, there was not any evidence of specific consistent tissue or organ damage microscopically in mice. Similar staining of all tissues was also observed in the cynomolgus monkey and human, and no effects suggestive of specific binding leading to cell damage were seen in these species. Thus, we believe that the tissue cross-reactivity results represented false-positive findings that were not associated with deleterious in vivo effects. False-positive results are known to occur with some regularity with tissue cross-reactivity studies. 7
Several other cases of liver injury following administration of protein therapeutics to cynomolgus monkeys in the context of toxicity studies have also been reported as blinded case studies. IV administration of a therapeutic antibody resulted in high ADAs titers and severe hepatocyte degeneration with eosinophilic globular inclusions in hepatocytes, as described by Rojko et al 24 in their case 2. Cytoplasmic globules containing endogenous IgG, IgM, IgA, and C3 have been reported in the liver of cynomolgus monkeys with ADAs and multiorgan immune-complex disease as described by Rojko et al 24 in their case 4, although hepatocyte degeneration or necrosis were not noted. Hepatocyte globules containing mAb test article, but not endogenous antibodies or complement, have also been reported by Rojko et al 24 in cynomolgus monkeys with immune-complex disease in their Case 5. The eosinophilic/cytoplasmic globules reported in these various cynomolgus monkey studies seem morphologically similar to those seen in the rats in the current studies where rat IgG, rat IgM, and C3 were detected (the presence of IgA was not assessed in our studies).
The nonclinical investigative studies with ATR-107 allowed the program to progress into a Phase 1 single-dose human clinical trial based on the understanding that the findings were related to an immune response, and that immune responses in animals are not predictive that immunogenicity will also occur in humans.6,16,19,20 The AUC exposure in rats at the highest IV dose tested in the 13-week study at the end of the study was ~50× over the predicted human AUC exposure at the highest planned IV dose of 100 mg at the time of the calculations. ATR-107 was administered IV in humans at single doses up to 120 mg, 13 and exposure multiples remained above 30×. ATR-107 elicited ADAs in 76% of human subjects, with a similar incidence by the IV or SC routes, although evidence of liver effects was not observed. 13 Investigations in human cells suggested that the immunogenicity in humans might be related to enhanced engagement of antigen presenting machinery by dendritic cells. 32 The program was terminated, and further mechanistic investigations into the liver effects observed in rats and assessment of potential relevance to humans were not conducted.
We are also aware of three other programs within the Pfizer portfolio where liver necrosis or fibrosis, apparently associated with ADAs and immunogenicity, have been seen in toxicity studies in rats after repeat dosing with protein therapeutics intended for human use, but not targeting IL-21R; in all cases, the therapeutics were based on antibody structures (M Leach, B Buetow, personal observations). Thus, liver necrosis or fibrosis, while rare, can occur in rats administered protein therapeutics intended for human use under circumstances that may result in immunogenicity.
In summary, ATR-107 was evaluated in toxicity studies in mice, rats, and cynomolgus monkeys by IV and SC routes of administration. Adverse ATR-107-related findings were only observed after multiple IV doses and only in mice and rats. The liver findings in rats and presumed anaphylaxis in mice appear to be related to an adaptive immune response against the human antibody ATR-107 and had no correlation with ATR-107 systemic exposure, suggesting that the findings most likely were not related to ATR-107 blocking the IL-21 pathway. Unlike rodents, in cynomolgus monkeys there were no immune-mediated ATR-107-related findings after multiple IV doses, although ADAs were only seen in two animals in the 13-week toxicity study, both in the lowest dose tested (10 mg/kg/week, one IV and one SC).
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of the many scientists involved in conducting and reporting the individual studies described in this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have been employed by Wyeth and/or Pfizer. Wenyue Hu is currently an employee of Vividion Therapeutics, Karuna Sachdeva is currently an employee of Jazz Pharmaceuticals, and Michael Leach is currently an employee of Trident Toxicology, Inc. Bernard Buetow is currently employed by Pfizer. The authors declare no other potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies presented in this report were sponsored and funded by Wyeth, and continuing support for data interpretation and publishing was provided by Pfizer following its acquisition of Wyeth.
