Abstract
Removal of the core fucose from the Fc region of humanized monoclonal antibodies (afucosylated antibodies) enhances their antibody-dependent cell cytotoxicity activities in killing cancer cells. Based on the authors’ experience and literature, administrations of afucosylated antibodies have been associated with neutropenia in cynomolgus monkeys. However, in a recent general toxicology study conducted with an afucosylated antibody in cynomolgus monkeys, transient neutropenia was observed and correlated with the emergence of anti-drug antibodies (ADAs) in the affected animals. To further explore the relationship between neutropenia, afucosylated antibodies, and ADAs in cynomolgus monkeys, we performed an investigational retrospective meta-analysis of data from general toxicology studies conducted with Genentech’s therapeutic antibodies administered to cynomolgus monkeys between 2005 and 2021. In this analysis, transient neutropenia strongly correlated with ADA-induced inflammation in cynomolgus monkeys administered afucosylated antibodies. This may reflect the simultaneous occurrence of two distinct processes of neutrophil elimination and utilization, thus overwhelming bone marrow reserve capacity leading to transient neutropenia. The integrated analysis of immunogenicity, and anatomic and clinical pathology results from these studies highlights the correlation of transient neutropenia in cynomolgus monkeys with ADA-related inflammation, potentially exacerbated by enhanced effector function of afucosylated antibodies.
Keywords
Introduction
Humanized monoclonal antibodies (mAbs) are increasingly used to treat several human diseases, thereby representing the fastest growing group of biotherapeutics.1-7 While some of these mAbs may function without utilizing antibody effector mechanisms (e.g. binding to a receptor or ligand and thereby blocking ligand-receptor interactions), many of these therapeutic antibodies (Abs) trigger their effector functions which involve the recruitment of immune cells (e.g. antibody-dependent cellular cytotoxicity [ADCC]/antibody-dependent cellular phagocytosis [ADCP]) to eliminate the target cells such as cancer cells, virus-infected cells, or invading pathogens.8-10 Using different mechanisms, the Fc region of these mAbs plays a critical role in the elimination of the target cells. These include (1) activation of the complement system via interaction with C1, (2) recruitment of phagocytes via the Fc receptors and the subsequent activation of the phagocytic pathway (i.e. ADCP), and (3) activation of the ADCC pathway mediated by natural killer (NK) cells.11-15 Among these mechanisms, ADCC is considered the key mechanism of action to kill cancer cells, and the binding affinity of the Fc region of the anti-cancer therapeutic antibodies (Ab) to the respective Fc receptors on the effector cells is an important component of their intended effector functions.13,16,17
One group of IgG Fc receptors, FcγR, are expressed on leukocytes and comprise three separate classes: FcγRI (CD64), FcγRII (CD32), and FcγRIII (CD16). FcγRI is constitutively expressed on monocytes and macrophages and can also be induced on neutrophils with interferon gamma (IFN-γ) and granulocyte colony-stimulating factor (G-CSF). 18
FcγRII can be divided into FcγRIIA, FcγRIIB, and FcγRIIC. However, macaques do not express FcγRIIC.19,20 FcγRIIA and FcγRIIC have activating functions, whereas FcγRIIB has inhibitory functions. All members of the FcγRII family are integral membrane glycoproteins and contain conserved extracellular domains, exhibiting 85% to 92% of amino acid identity between the FcγRIIa and the inhibitory FcγRIIb receptors in humans, and even more so in macaques with 95% amino acid homology between the two subreceptors.21,22 This high degree of amino acid identity has presented challenges in discriminating between the FcγRII subreceptors, and as a result, much of the literature published to date does not mention the specific form of the receptor expressed by a given cell type, reporting instead only the presence or absence of FcγRII. 23 Moreover, it has been shown that cynomolgus and pigtail macaque neutrophils may express 3-fold to 5-fold higher total FcγRII than their human counterparts,23,24 which are known to express only the activating form of the receptor. 25 That said, FcγRII is generally expressed on granulocytes and phagocytes in humans and macaques. In addition, FcγRII is expressed on B cells and NK cells in macaques.24,26
FcγRIII is also divided into FcγRIIIA and FcγRIIIB. The FcγRIIIA is expressed on NK cells and monocytes/macrophages, while FcγRIIIB is expressed on neutrophils and eosinophils.8,11,24,27-30 In macaques, only one FcγRIII is present. It is homologous to the human FcγRIIIA and is only expressed on NK cells and monocytes/macrophages. Macaques do not have FcγRIIIB. 31 Binding affinity of FcγRIIIA seems to be particularly sensitive to core fucosylation of the N-linked glycan at N297 of the Fc tail of IgG, as its binding affinity can be enhanced up to 50 times—with corresponding increase in effector function—if the Fc tail of the IgG is not fucosylated.32,33 As such, as illustrated in Figure 1, removal of the core fucose from N-glycans of the Fc region of IgG represents the most effective approach to increase ADCC activity through enhanced NK cells’ FcγRIII-IgG Fc interactions.34,35 These Abs lacking the core fucose in their Fc regions are often referred to as afucosylated or nonfucosylated Abs (Figure 1). However, fucose content does not seem to affect binding to the other FcγRs (FcγRI and FcγRIIA/B) in humans.35-39 Some marketed afucosylated therapeutic antibodies, including obinutuzumab and mogamulizumab, are currently being used for the treatment of neoplastic diseases in humans.40-42
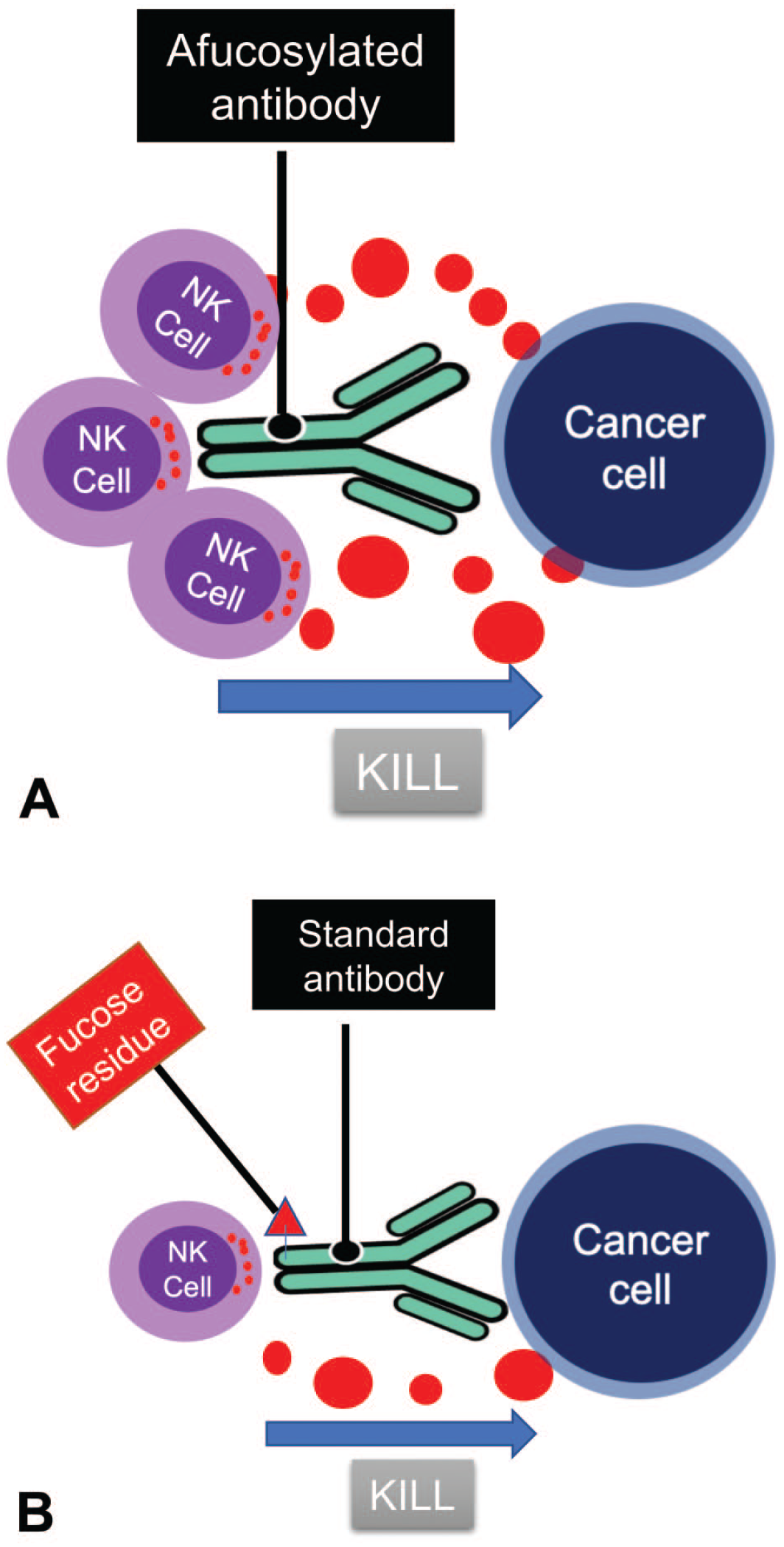
Schematic representation of the differences in ADCC mechanism between (A) an afucosylated antibody that lacks its core fucose and (B) a standard antibody that still contains its core fucose. As shown in (A), the lack of core fucose at the Fc region of the IgG enhances the binding affinity of the afucosylated antibody to FcγRIIIA (or CD16a) on NK cells leading to corresponding increase in effector function. In contrast, as shown in (B), the presence of core fucose at the Fc region of the IgG limits the binding affinity of the antibody to NK cells, with relatively lower effector function and as a result, a weaker ADCC. ADCC indicates antibody-dependent cell cytotoxicity; NK, natural killer.
Before the administration of any new investigational drug in humans for clinical trials, the safety and efficacy of these molecules are assessed in several nonclinical in vitro, in vivo, and/or ex vivo studies. While taking the pharmacological relevance of the target into consideration, many Ab in vivo toxicology studies are often performed in macaques. Biotherapeutics administered to animals or humans usually have some differences in structure compared with the native, endogenous molecules. Therefore, they can be perceived as foreign and elicit an immune response leading to the production of anti-drug antibodies (ADAs). These ADAs can form various types of immune complex (IC) with the therapeutic Ab which can occasionally lead to systemic and localized tissue effects due to type III hypersensitivity reactions. 43
During the course of a general toxicology study with an afucosylated mAb in cynomolgus monkeys, we observed transient neutropenia along with other clinical pathology changes consistent with systemic inflammation. We also noticed that the timing of these findings coincided with the emergence of ADA in the affected monkeys. While possible, based on the authors’ experience, neutropenia is not a common occurrence with ADA-associated systemic effects in macaques. However, it has been shown that afucosylated mAbs may directly cause neutropenia in cynomolgus monkeys. 44 To understand the relationship between neutropenia, afucosylated mAbs, and ADA associated with afucosylated mAb administration in cynomolgus monkeys, we conducted an investigational retrospective analysis of all outsourced large molecule general toxicology cynomolgus monkey studies between 2005 and 2021. In the studies we reviewed, we report that (1) ADA-associated pathology with respect to large molecules is an uncommon phenomenon; (2) afucosylated mAbs have a much stronger correlation with ADA-related pathology changes compared with the standard therapeutic Abs; (3) ADA-induced systemic effects associated with administration of standard Abs in cynomolgus monkeys are usually not accompanied by any changes in neutrophils; and (4) neutropenia strongly correlated with ADA-induced inflammation in cynomolgus monkeys administered afucosylated mAbs relative to cynomolgus monkeys administered standard Abs.
The integrated analysis of immunogenicity, and anatomic and clinical pathology results from these studies in aggregate highlights the correlation of transient neutropenia in cynomolgus monkeys with ADA-related inflammation, potentially exacerbated by enhanced effector function of afucosylated mAbs.
Materials and Methods
The outsourced study database in Safety Assessment of Genentech, Inc., was searched for all the cynomolgus monkey studies conducted with large molecules from January 1, 2005, to December 31, 2021. We applied the following exclusion criteria: efficacy studies; pilot single-dose pharmacokinetic or pharmacodynamic studies; imaging studies; exploratory biomarker studies; exclusive immunogenicity studies; exclusive safety pharmacology studies; reproductive or development and reproductive toxicology (DART) studies in segment I, II, and III; and juvenile toxicity studies. We also excluded antibody-drug conjugate (ADC) studies due to drug payload’s inherent and direct mechanism on bone marrow toxicity and/or neutrophils. In addition, studies with large molecules that have a direct effect on neutrophils due to their mechanisms of action were excluded. Studies without a complete set of finalized reports were excluded as well. After the exclusion criteria, a total of 59 repeat-dose general toxicology studies of unconjugated antibodies administered to cynomolgus monkeys between January 1, 2005, to December 31, 2021, were included in this analysis. All studies had been finalized and included complete sets of final toxicology reports, ADA reports, clinical pathology reports, and anatomic pathology reports. These 59 studies were performed on 2280 cynomolgus monkeys with 45 large molecules across 43 distinct programs. Among these 59 studies, 10 studies were conducted with afucosylated mAbs on 362 cynomolgus monkeys and 49 studies with standard Abs on 1918 cynomolgus monkeys. The 10 afucosylated mAb studies consisted of 7 molecules across 6 programs, while the 49 standard Ab studies consisted of 38 molecules across 37 programs. The breakdown of these studies can also be seen in Table 1.
Breakdown of the numbers of studies, programs, and molecules analyzed in the meta-analysis.
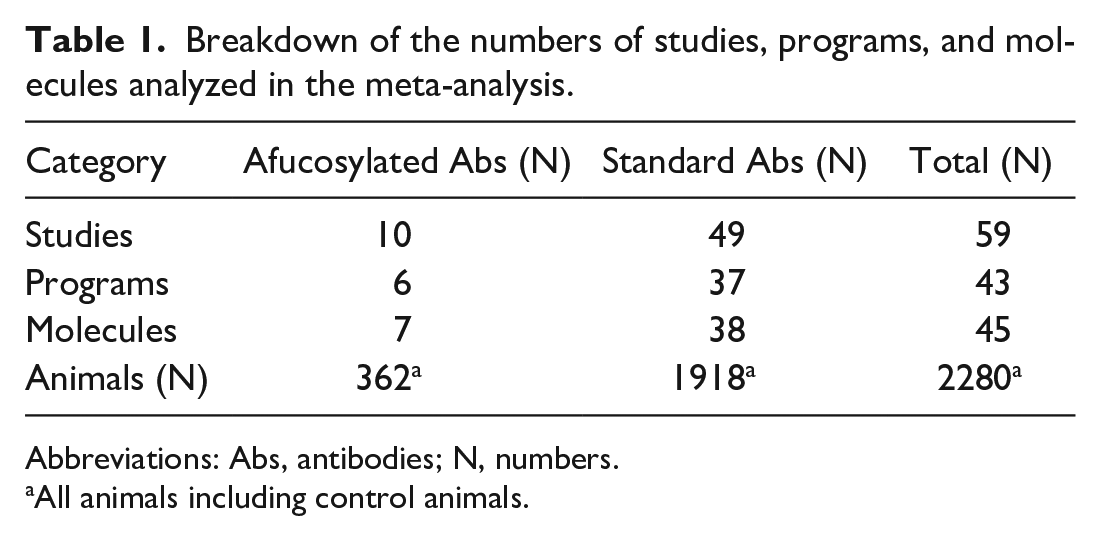
Abbreviations: Abs, antibodies; N, numbers.
All animals including control animals.
The cynomolgus monkeys included in this meta-analysis were ~2 to 5 years old at the beginning of dosing, and were all purpose-bred and came from a mixture of Cambodian and Chinese breeding facilities.
Ethical Statement
All in vivo animal procedures were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)-accredited facility under an Institutional Animal Care and Use Committee–approved protocol at Genentech Inc. Except where noted, standard procedures and conditions for animal care, housing, access to water and food, environment, and room maintenance were used. All other procedures were performed in accordance with laboratory standard operating procedures and/or established laboratory best practices.
Results
ADA-Related Pathology is an Uncommon Phenomenon
We first established the total number of studies (afucosylated Abs and standard Abs combined) with ADAs, followed by investigating how many of the studies with ADAs resulted in ADA-related pathology (i.e., clinical and anatomic pathology combined).
As shown in Table 2, 100% (59/59) of the studies had animals with ADAs. However, only ~20% (12/59) of these studies showed ADA-related pathology findings with only ~7% (4/59) ADA-related fatality. We also matched the ADA positivity, ADA-related pathology positivity, and ADA-related fatality positivity rates at the individual animal level across all studies. As shown in Table 2, the ADA positivity rate in the individual animals across all studies was ~33% (559/1705), while the ADA-related pathology positivity and ADA-related fatality positivity rates were ~14% (78/559) and ~2% (10/559), respectively. These results showed that ADA-related pathology is not a common phenomenon when cynomolgus monkeys developed ADA in response to biotherapeutic administration.
Numbers and percentages of studies and individual animals with ADA, ADA-related pathology, and ADA-related fatality with afucosylated Abs versus standard Abs.
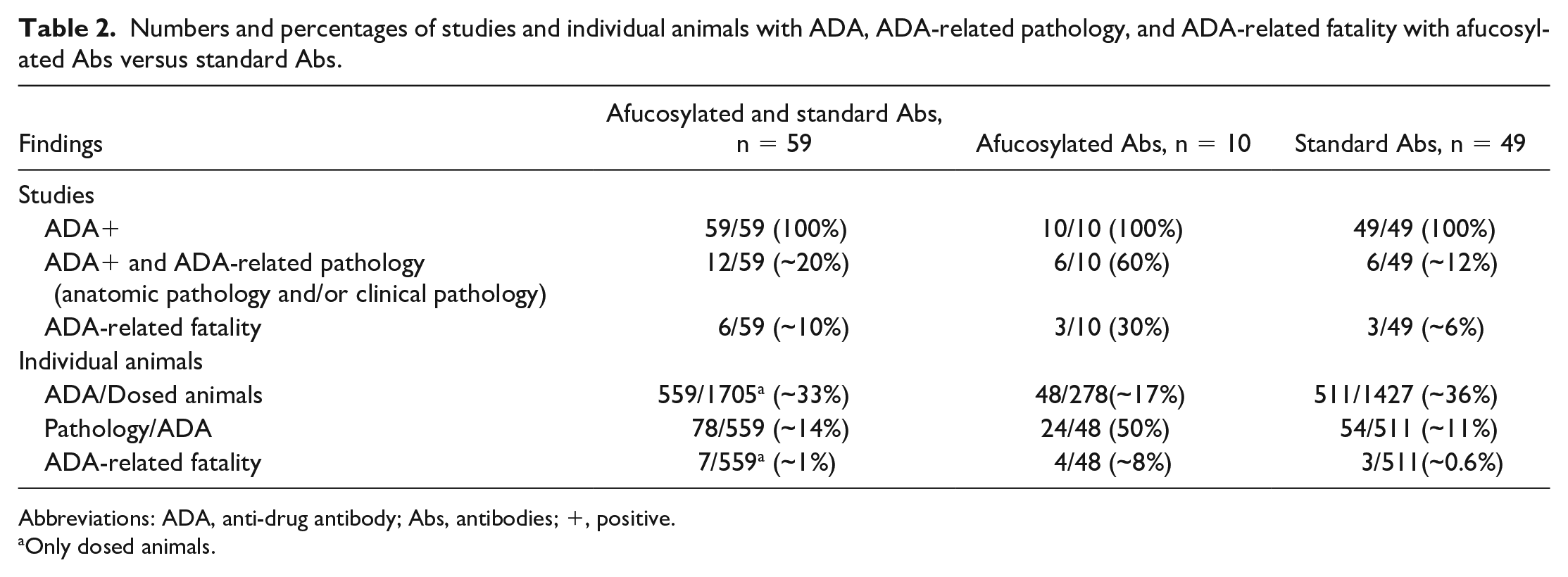
Abbreviations: ADA, anti-drug antibody; Abs, antibodies; +, positive.
Only dosed animals.
Afucosylated Abs Were Associated With Much Higher ADA-Related Pathology Changes Relative to the Standard Abs
To sort out the contributions of each Ab type to the ADA-related pathology observed, we separated the pathology findings by Ab type. As shown in Table 2, there were a total of 10 toxicology studies conducted with afucosylated Abs and 49 toxicology studies with the standard Abs. Regardless of the Ab type, all studies demonstrated ADA positivity (100%); 60% (6/10) of the afucosylated Ab studies had ADA-related pathology findings, whereas only ~12% (6/49) of the standard Ab studies showed ADA-related pathology changes. Moreover, 30% (3/10) of the afucosylated Ab studies had ADA-related fatality, while ~6% (3/49) of the standard Ab studies had ADA-related fatality. At the individual animal level, across all the 10 studies evaluated for the afucosylated Abs, the ADA, ADA-related pathology, and the ADA-related fatality positivity rates were ~17% (48/278), 50% (24/48), and ~8% (4/48), respectively. Across all the 49 studies evaluated for the standard Abs, the ADA, ADA-related pathology, and ADA-related fatality positivity rates were ~36% (511/1427), ~11% (54/511), and ~0.6% (3/511), respectively, at the individual animal level. Overall, these results suggested that afucosylated Abs have a much stronger correlation with ADA-related pathology and fatality changes relative to the standard Abs.
Clinical Pathology Changes (Excluding Neutrophils) Associated With ADA-Related Systemic Effects
As shown in Table 3, in hematology, the clinical pathology changes associated with ADA-related systemic effects across studies included decreases in red cell mass and platelet counts, along with increases in total white blood cell counts. Clinical pathology changes associated with coagulation included increases in prothrombin and partial thromboplastin times along with increases and decreases in fibrinogen. In clinical chemistry, changes included increases in C-reactive protein (CRP), blood urea nitrogen (BUN), creatinine, and globulin along with decreases in albumin, albumin/globulin ratio (A:G), and total protein.
Clinical pathology changes (excluding neutrophils) associated with ADA-related systemic effects.

Abbreviation: ADA, anti-drug antibody.
CRP was only measured in 7 of the studies with pathology.
Of all these clinical pathology changes, decreases in albumin, A:G, and platelet counts along with increases in globulin, fibrinogen, and CRP were the most consistent clinical pathology findings across most studies with ADA-related systemic effects. These findings are consistent with systemic inflammation resulting from ADA-related tissue injury. Given the importance of neutrophils in this study, a separate section has been dedicated to neutrophilic changes that accompanied ADA-related systemic effects.
Histopathology Changes Associated With ADA-Related Systemic Injury
As shown in Table 4, and illustrated in Figure 2, histopathology changes attributed to ADA-related systemic injury included multi-organ mononuclear cell infiltration and vasculitis/perivasculitis. These were occasionally accompanied by hemorrhage and fibrin thrombi, glomerulonephritis, increased lymphoid organ cellularity, and pulmonary and hepatocellular changes. These changes were consistent with findings associated with ADA-induced type III hypersensitivity reaction 45 and correlated with the clinical pathology findings described above. The increased lymphoid cellularity was often observed in the mandibular and mesenteric lymph nodes, along with increased follicular cellularity in the splenic white pulp. Although perivasculitis/vasculitis and increased lymphoid organ cellularity can occur spontaneously in young cynomolgus monkeys, 46 due to the association with high ADA titers, and the presence of the clinical pathology changes described above, these changes were considered to be secondary to an ADA-associated immune response. The pulmonary and hepatocellular changes were observed in conjunction with other ADA-related changes mentioned above and were only observed in animals with ADA or high ADA titers in those instances. The pulmonary injuries were characterized by lung and airway hemorrhage, pulmonary alveolar and periarterial hemorrhage with neutrophilic inflammatory cellular infiltration in the alveolar wall, or lung changes associated with diffuse edema, congestion, perivascular hemorrhage, and alveolar fibrin thrombi. The hepatocellular changes were characterized by hepatocellular necrosis or Kupffer cell hyperplasia.
Histopathology changes associated with ADA-related systemic injury.
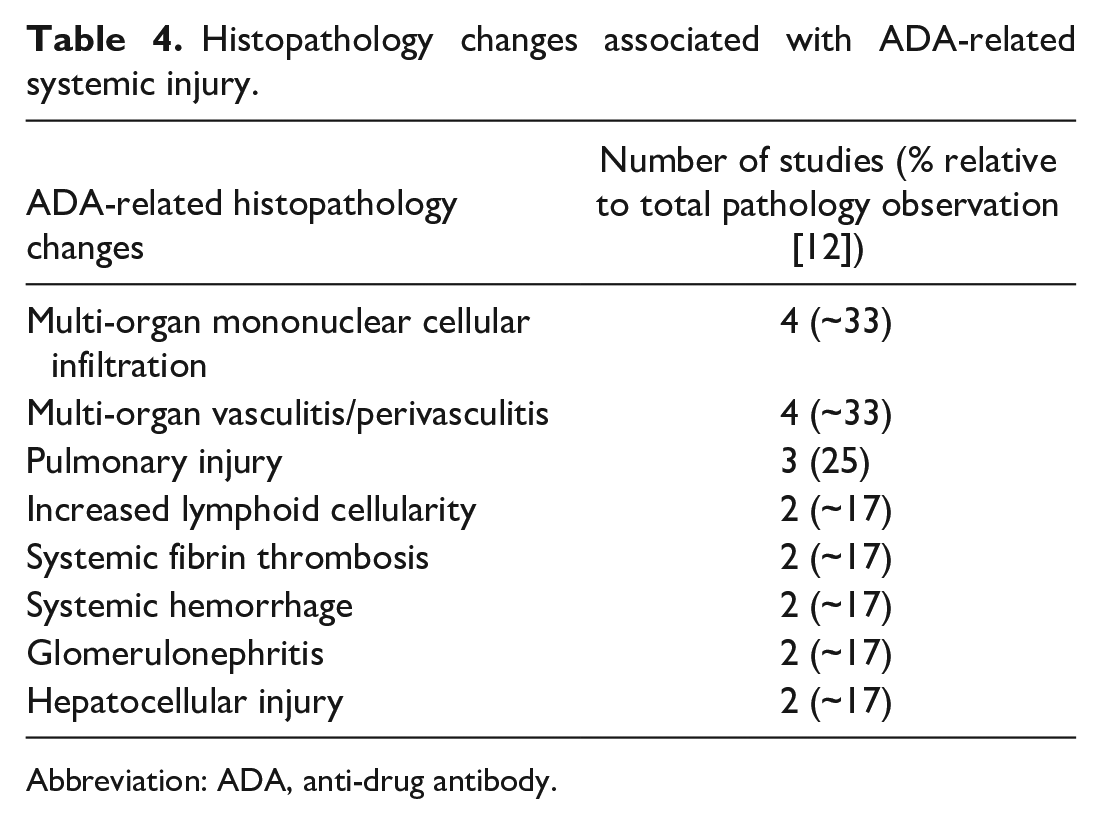
Abbreviation: ADA, anti-drug antibody.
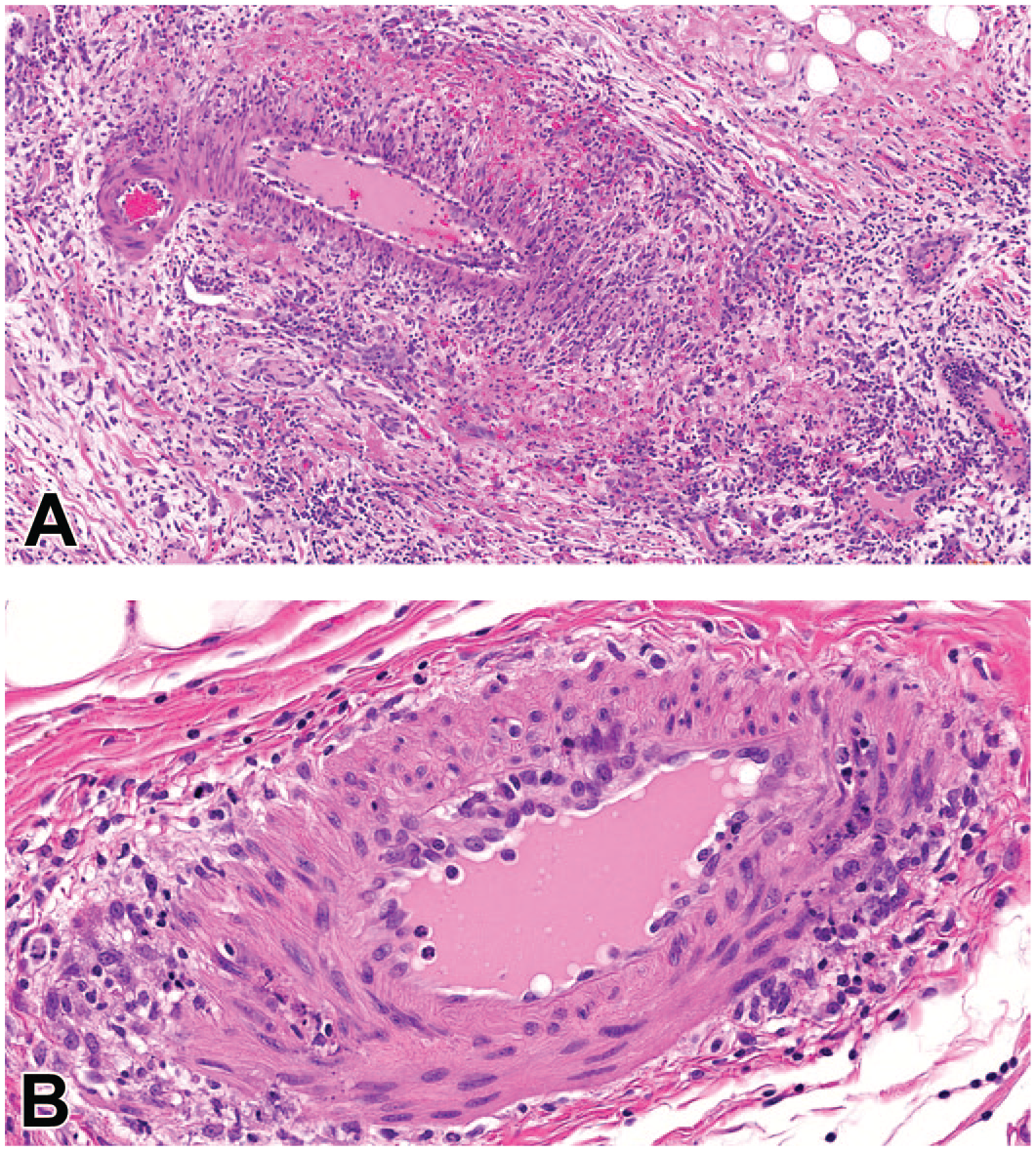
(A) Vasculitis and perivasculitis in the submucosa of the gallbladder of a cynomolgus monkey. Note mixed mononuclear cells surrounding a large vein and several smaller vessels; the endothelium is plump and reactive, and there are areas of hemorrhage and fibrin deposition extending into the adventitia. Hematoxylin & Eosin. (B) Arteritis and periarteritis in the right coronary artery of a cynomolgus monkey. Note the plump, reactive endothelium, expansion of the tunica intima, and the mixed inflammatory cells, some of which are degenerating, disrupting the tunica media, and extending into the adventitia. Both examples are from a macaque that had been administered an afucosylated antibody weekly by intravenous administration for approximately 6 weeks. This macaque had developed both a decrease in circulating neutrophils and a positive titer for anti-drug antibodies during the fourth week of the study. Hematoxylin & Eosin.
The Frequency of ADA-Related Clinical Pathology Findings Was Slightly Higher Than the Incidence of Anatomic Pathology Changes
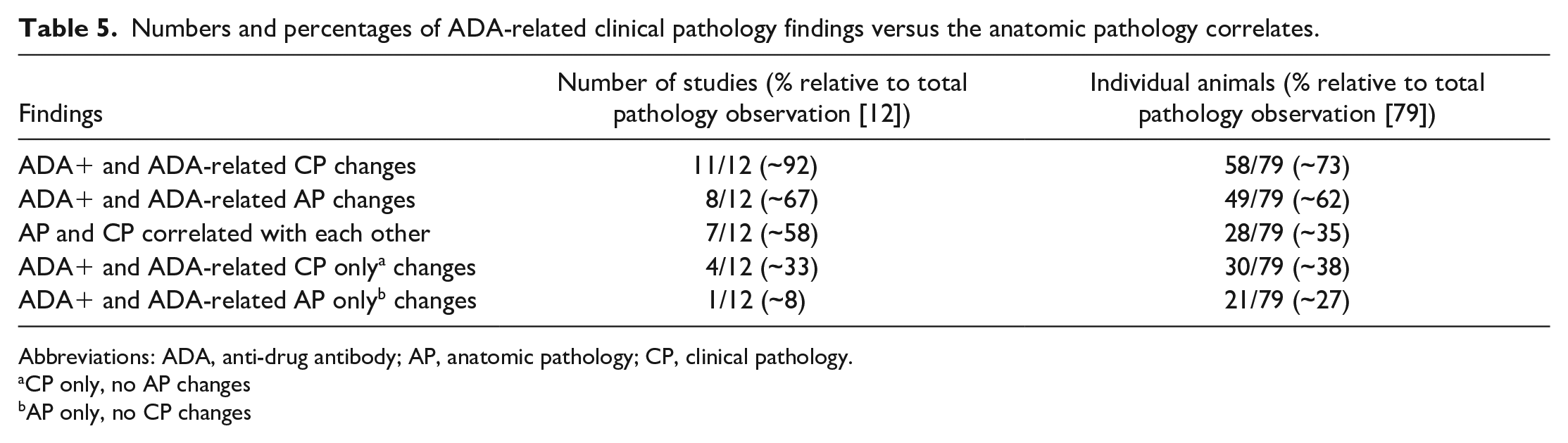
Given the transient nature of some of the ADA-related clinical pathology findings observed in some of the general toxicology studies included in this retrospective study, we decided to investigate the degree of correlation between the ADA-associated clinical pathology findings and the anatomic pathology correlates. As shown in Table 5, ~92% (11/12) of the studies with ADA-related pathology findings had clinical pathology correlates, while ~67% (8/12) had anatomic pathology correlates; ~58% (7/12) of the studies had both anatomic and clinical pathology correlates; ~33% (4/12) of the studies had only transient clinical pathology correlates with no anatomic pathology correlates, and ~8% (1/12) of the studies had only anatomic pathology correlates with no clinical pathology correlates.
Numbers and percentages of ADA-related clinical pathology findings versus the anatomic pathology correlates.
Abbreviations: ADA, anti-drug antibody; AP, anatomic pathology; CP, clinical pathology.aCP only, no AP changesbAP only, no CP changes
At the individual animal level, ~73% (58/79) of the animals with ADA-related pathology findings had clinical pathology correlates, while ~62% (49/79) had anatomic pathology correlates; ~35% (28/79) of the animals had both anatomic and clinical pathology correlates; ~38% (30/79) of the animals had only transient clinical pathology correlates with no anatomic pathology correlates, and ~27% (21/79) of the animals had only anatomic pathology correlates with no clinical pathology correlates.
These findings showed that the frequency of ADA-related clinical pathology findings was slightly higher than the incidence of the anatomic pathology changes. This further confirms that ADA-related pathology may occasionally be transient and may wane prior to conducting terminal necropsy at the end of the study. The clinical pathology samples were from both interim and terminal collections. In scenarios where the clinical pathology changes were transient with no histopathology correlates at terminal necropsy, this was because the clinical pathology changes were observed during the interim collections and may have subsided at the end of the study.
Neutropenia Strongly Correlated With ADA-Induced Inflammation in Cynomolgus Monkeys Administered Afucosylated Abs
To understand the relationship between neutropenia, afucosylated mAbs, and ADA associated with afucosylated mAb administration in cynomolgus monkeys, we first evaluated the type of neutrophilic changes that were commonly observed with ADA-induced inflammation regardless of the antibody type. Based on Genentech’s historical control data generated from the outsourced studies, neutrophil counts in cynomolgus monkeys ranged from 1.46-12.48 × 103/µl. Values below this range were classified as neutropenia, and values above this range were classified as neutrophilia.
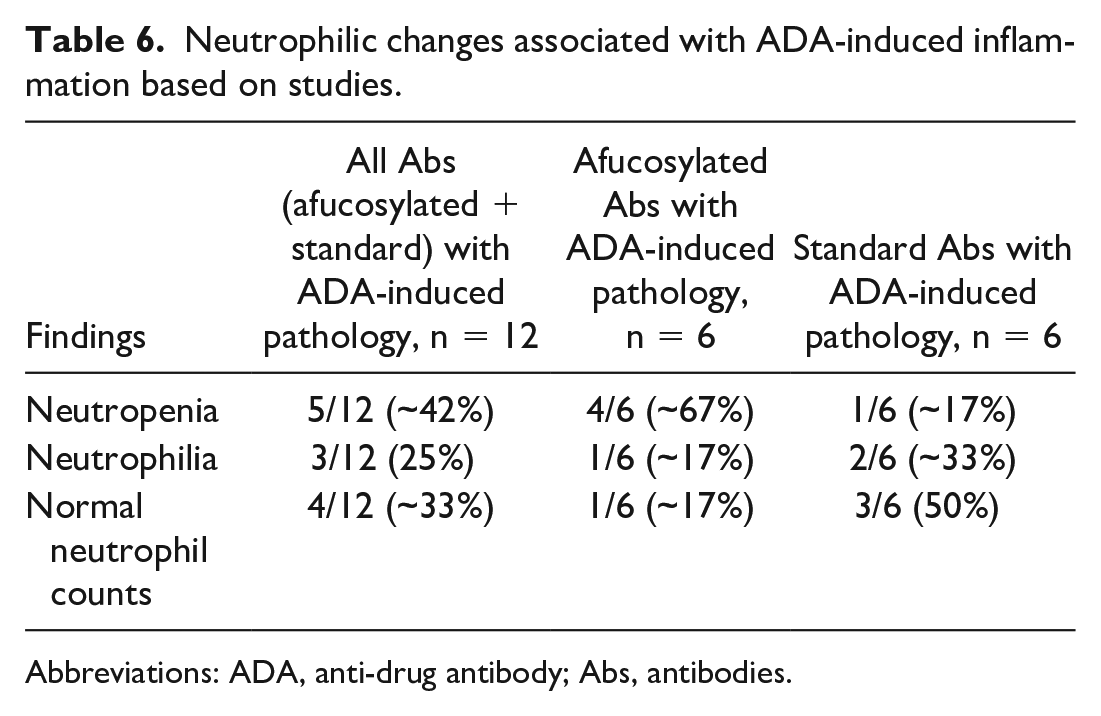
As shown in Table 6, ~42% of studies with confirmed pathology changes had neutropenia, ~33% had normal neutrophil counts, and 25% of the studies had neutrophilia. These results suggested that neutropenia was the most common neutrophilic change that accompanied ADA-related inflammation.
Neutrophilic changes associated with ADA-induced inflammation based on studies.
Abbreviations: ADA, anti-drug antibody; Abs, antibodies.
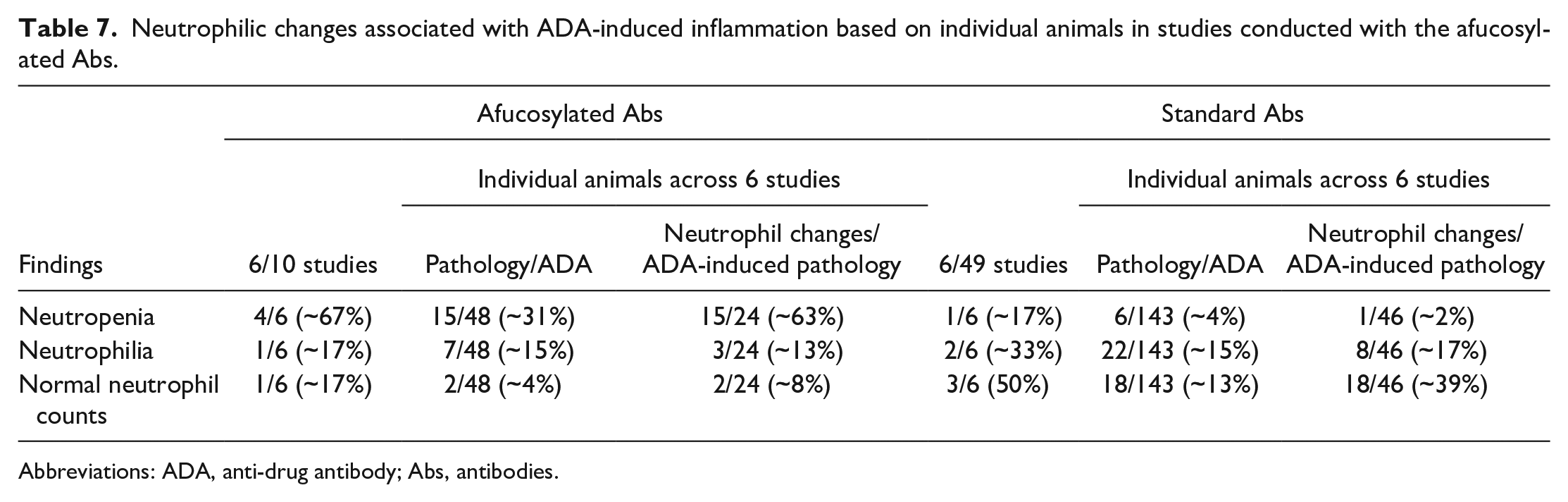
To sort out the contributions of each Ab type to the ADA-related neutrophilic changes described, we separated the neutrophilic findings by Ab type. As shown in Table 6, ~67% (4/6) of the afucosylated Ab studies with confirmed pathology changes had neutropenia, whereas only 17% (1/6) had neutrophilia and another 17% (1/6) showed no changes in neutrophils. At the individual animal level, as shown in Table 7 for afucosylated Ab studies, ~63% of the animals (15/24) with ADA-related pathology had neutropenia, whereas only ~13% (3/24) had neutrophilia and another ~8% (2/24) showed no changes in neutrophil counts. These results suggested that neutropenia was the primary neutrophilic change that accompanied ADA-induced inflammation in cynomolgus monkeys administered afucosylated Abs.
Neutrophilic changes associated with ADA-induced inflammation based on individual animals in studies conducted with the afucosylated Abs.
Abbreviations: ADA, anti-drug antibody; Abs, antibodies.
In contrast, as shown in Table 6 for the standard Ab studies, only 17% (1/6) of the standard Ab studies with confirmed pathology changes showed neutropenia, whereas 33% (2/6) had neutrophilia and 50% (3/6) had normal neutrophil counts. At the individual animal level, as shown in Table 7 for standard Ab studies, ~2% of the animals (1/46) with ADA-related pathology had neutropenia, while ~17% (8/46) had neutrophilia and ~39% (18/46) showed no changes in neutrophil counts suggesting that ADA-induced inflammation in cynomolgus monkeys administered standard Abs does not usually elicit any neutrophilic change. Overall, these results suggested that neutropenia strongly correlated with ADA-induced inflammation in cynomolgus monkeys administered afucosylated Abs.
Afucosylated Abs Were Not Associated With Neutropenia in the Absence of ADA-Induced Inflammation
Given the strong correlation between neutropenia and ADA-induced inflammation due to afucosylated Ab administration, we investigated whether afucosylated Abs can cause neutropenia in the absence of ADA-induced inflammation. As shown in Table 8, despite the presence of ADA, 40% (4/10) of the studies conducted with afucosylated Abs did not have any ADA-related pathology changes. Moreover, none of these studies displayed neutropenia or any other form of neutrophilic change, suggesting that afucosylated Abs may not directly cause neutropenia in all the studies evaluated.
Neutrophilic changes associated with afucosylated Abs in the absence of ADA-induced inflammation.
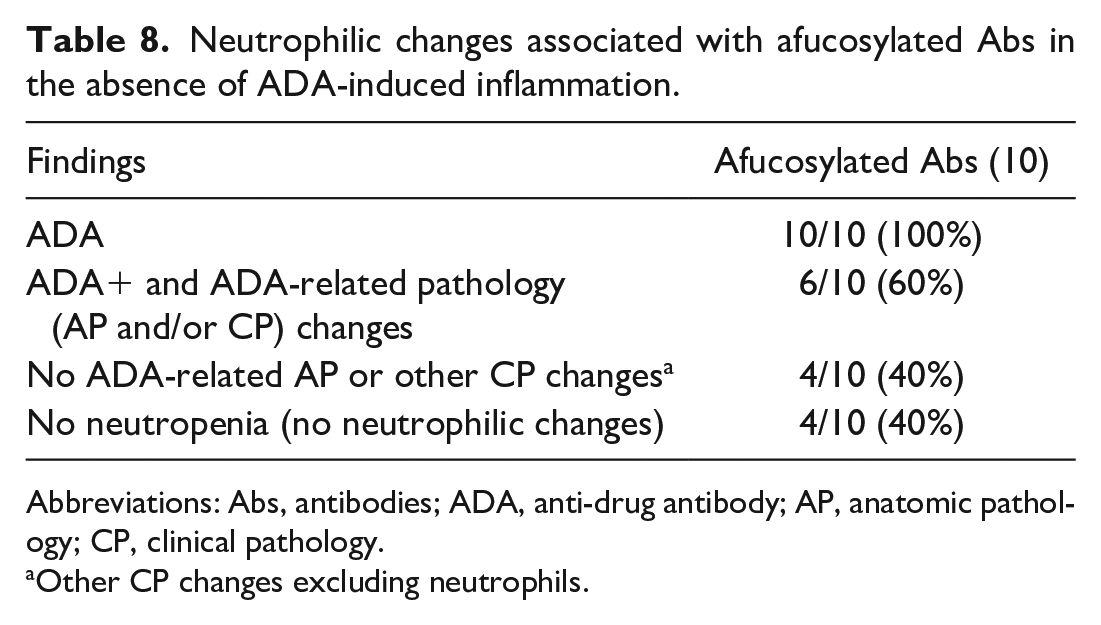
Abbreviations: Abs, antibodies; ADA, anti-drug antibody; AP, anatomic pathology; CP, clinical pathology.
Other CP changes excluding neutrophils.
Discussion
This retrospective analysis was conducted due to the observation of transient neutropenia in a recently conducted outsourced general toxicology study. The study was conducted to assess the safety of an afucosylated therapeutic antibody administered at 30 and 100 mg/kg, q1w × 7 in cynomolgus monkeys in a 6-week general toxicology study. During the course of the study, neutropenia, along with other inflammatory clinical pathology changes, was observed in one animal in the high-dose group on day 9, and by day 22, five animals in the high-dose group (5/8), along with 2 animals in the mid-dose group (2/8), had developed similar changes. On day 35 of the study, despite continued dosing, there was a trend toward normal neutrophil counts in 2 of the affected animals in the high-dose group. By day 45 (end of the study), the neutrophil counts had reversed toward the normal range in almost all affected animals except for 2 animals (one each in the mid-dose and high-dose groups). Histopathologic findings consistent with ADA-related pathology, including vasculitis in the gall bladder, heart, and kidney, were observed in one of the animals with sustained neutropenia (see Figure 2). The advent of the neutropenia coincided with the emergence of ADA in these animals, and likewise, the affected animals were the only ones that developed ADA in this study. Based on this correlation, we inferred that the neutropenia in this study was likely to be ADA related. All things considered, based on data from other programs, we knew that neutropenia had been observed in cynomolgus monkeys administered afucosylated therapeutic Abs. As such, there was interest in understanding whether afucosylated Abs caused neutropenia in cynomolgus monkeys. This notion was further supported by one of the case studies that were presented in a workshop report on “Biotherapeutics in non-clinical development” organized by BioSafe. BioSafe is the nonclinical safety expert group of the Biotechnology Innovation Organization (BIO), whose mission is to identify and respond to key scientific and regulatory issues and challenges related to the nonclinical safety evaluation of biopharmaceuticals. 44 The case study in question showed that an afucosylated therapeutic antibody (NVS123) caused neutropenia in cynomolgus monkeys due to the cross-linking of the target cells and neutrophils by the afucosylated antibody that consequently caused activation-induced elimination of neutrophils. 44 That said, it is also generally recognized that neutropenia is an uncommon occurrence with ADA-associated systemic effects in macaques. This was further confirmed by the results in Tables 6 and 7 showing that normal neutrophil counts and to a lesser degree neutrophilia were the common neutrophilic changes associated with ADA-induced pathology in cynomolgus monkeys administered standard Abs which represented the majority of the studies assessed in this retrospective analysis (Table 1).
We initially embarked on this retrospective analysis to better understand the relationship between neutropenia, afucosylated Abs, and ADA associated with afucosylated Ab administration in cynomolgus monkeys. One of the most important observations in this analysis was the strong correlation between neutropenia and ADA-induced systemic effects in cynomolgus monkeys given afucosylated Abs (Tables 6 and 7). Results in Table 8 also showed that despite the presence of ADA, afucosylated Abs were not associated with neutropenia in the absence of ADA-induced inflammation, further suggesting that neutropenia only ensued in the presence of ADA-induced inflammation caused by the administration of afucosylated Abs in the toxicology studies assessed in this retrospective analysis. Nonetheless, the case study from the workshop mentioned above also suggested that afucosylated antibodies may also possess the intrinsic ability to cause neutropenia by activation-induced mechanism when neutrophils are cross-linked with the immune target cells. 44 Based on this, we theorized that there were two distinct simultaneous processes responsible for the neutropenia observed in cynomolgus monkeys with ADA-induced pathology in response to the administration of afucosylated antibodies: (1) we hypothesized that an underlying low-grade activation-induced elimination of neutrophils may be present in all these studies. However, neutrophils appeared not to be affected in the absence of ADA-related inflammation (Table 8) because the rate of elimination of neutrophils (or demand for neutrophils) typically remains less than or equal to the rate of bone marrow response (supply of neutrophils), and (2) ADA-induced inflammation also caused neutrophil utilization mostly at a rate that is less than or equal to the rate of bone marrow response. We believed this was the reason why there were higher number of animals with no neutrophilic changes associated with ADA-induced systemic effects in monkeys administered standard therapeutic antibodies (Table 7). When these 2 mechanisms occur simultaneously in monkeys administered afucosylated Abs, then the rate of neutrophil utilization/elimination can become greater than the rate of bone marrow response (demand > supply) leading to neutropenia. This illustration for this proposed mechanism can also be found in Figure 3.
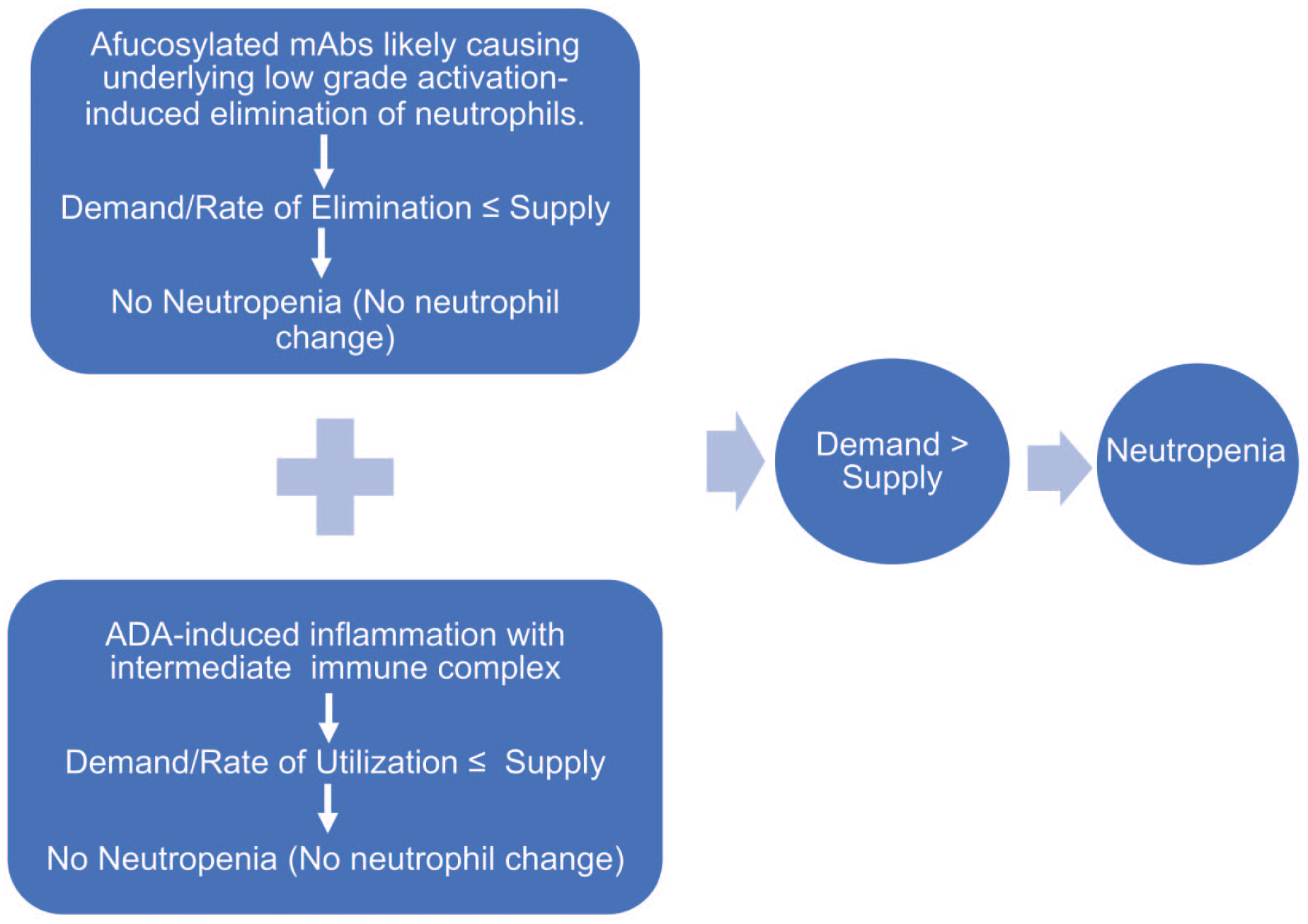
Proposed mechanism for the neutropenia observed with afucosylated antibodies. Simultaneous occurrence of two distinct mechanisms of neutrophil elimination and utilization may have caused neutropenia in cynomolgus monkeys administered afucosylated Abs as the rate of neutrophil utilization/elimination becomes greater than the rate of bone marrow response (demand > supply). Abs indicates antibodies; ADA, anti-drug antibody; mAbs, monoclonal antibodies.
Based on the first proposed mechanism and the case study that showed an afucosylated antibody (NVS123) cross-linking cynomolgus monkeys’ neutrophils with the target cells, 44 there is a possibility that afucosylated antibodies may have an enhanced binding affinity to macaques’ neutrophils. However, the exact mechanism by which this may be happening is unknown at the moment, as macaques’ neutrophils do not express FcγRIIIB unlike in humans 31 and the fucose content of the IgG Fc does not seem to affect binding to the other FcγRs (FcγRI and FcγRIIA/B) expressed on neutrophils in humans.35-39 Nevertheless, given the fact that little is known about Fc receptors in macaques due to their lesser characterizations compared with those of humans, there is a possibility that the afucosylated antibodies may have an enhanced binding affinity to macaques’ neutrophils relative to humans. Further studies will be required to confirm this hypothesis.
Another important observation in this retrospective analysis was the higher frequency of ADA-related clinical pathology findings relative to the incidence of the anatomic pathology changes. This was likely due to the dynamic nature of the large soluble ICs that are deposited in tissues and activate the complement pathway with subsequent tissue damage. Over time, with continued animal dosing and ADA production, the types and sizes of the IC change, the antigen/antibody ratios change, and the immunoglobulin isotype and clearance mechanisms change. 47 This likely explained the reasons why in some of the toxicology studies analyzed in this retrospective analysis, the clinical pathology changes that correlated with the presence of ADA were transient in nature and lacked any significant anatomic pathology correlates at terminal necropsy. This might also be the reason why ADA-related pathology is not a common phenomenon as shown in Table 2, and consistent with what has been reported in the literature. 45
Additional important observations in this meta-analysis were the higher frequency of ADA-related pathology and fatality associated with the administration of afucosylated mAbs relative to standard Abs. The reason for these observations is not well understood. While individual animal variability/sensitivity may have played a role, we do not believe that was the primary cause for these observations. During this meta-analysis, we also noticed that the ADA-related pathology was mostly observed at the mid-dose to high-dose groups for both the afucosylated and standard Abs. However, there were other studies across both the afucosylated and standard Abs with no ADA-related pathology at relatively similar doses; there were also instances where ADA-related pathology was observed at lower doses with both antibody types. Notwithstanding, it seemed the afucosylated Abs have a higher tendency to form the large soluble ICs that were largely responsible for the ADA-related pathology 47 relative to the standard Abs. As such, further studies will be required to unravel the mechanisms behind these observations.
Finally, this analysis involved 2280 cynomolgus monkeys, 362 and 1918 of which were from studies conducted with afucosylated antibodies and standard Abs, respectively (Table 1). Across the 10 studies conducted with the afucosylated Abs, only 48 of the 278 animals (~17%) that were dosed had ADA, 24 of the 48 animals (50%) with ADA had ADA-related pathology (Table 2), and 15 of the 24 animals (~63%) with ADA-related pathology had neutropenia (Table 7). While the overall number of animals affected was relatively small, the strong correlation of neutropenia with ADA-related pathology across different studies conducted with distinct afucosylated molecules, along with the intra-study and inter-study individual animal variabilities, makes this observation convincing.
In conclusion, we have conducted a retrospective analysis to better understand the relationship between neutropenia, afucosylated mAbs, and ADA associated with afucosylated mAb administration in cynomolgus monkeys. The integrated analysis of immunogenicity, and anatomic and clinical pathology results from these studies in aggregate highlights the correlation of transient neutropenia in cynomolgus monkeys with ADA-related inflammation, potentially exacerbated by enhanced effector function of afucosylated mAbs.
Footnotes
Acknowledgements
The authors would like to thank the following colleagues who were consulted during the course of this analysis: Noel Dybdal, Gary Cain, Christopher Davies, and Robert Hendricks.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All work was supported by Genentech Inc., a member of the Roche group.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
