Abstract
Emerging urinary kidney safety biomarkers have been evaluated in recent years and have been shown to be superior to the serum parameters blood urea nitrogen (BUN) and creatinine (sCr) for monitoring kidney injury in the proximal tubule. However, their potential application in differentiating the location of the initial kidney injury (eg, glomerulus vs tubule) has not been fully explored. Here, we assessed the performance of two algorithms that were constructed using either an empirical or a mathematical model to predict the site of kidney injury using a data set consisting of 22 rat kidney toxicity studies with known urine biomarker and histopathologic outcomes. Two kidney safety biomarkers used in both models, kidney injury molecule 1 (KIM-1) and albumin (ALB), were the best performers to differentiate glomerular injury from tubular injury. The performance of algorithms using these two biomarkers against the gold standard of kidney histopathologic examination showed high sensitivity in differentiating the location of the kidney damage to either the glomerulus or the proximal tubules. These data support the exploration of such an approach for use in clinical settings, leveraging urinary biomarker data to aid in the diagnosis of either glomerular or tubular injury where histopathologic assessments are not conducted.
Introduction
Drug-induced kidney injury (DIKI) can target different regions of the kidney nephron, most notably in the glomerulus and/or proximal tubules. A set of emerging urinary kidney safety protein biomarkers including kidney injury molecule 1 (KIM-1) and albumin (ALB) was endorsed by regulatory agencies more than a decade ago due to their ability to outperform serum biomarkers in monitoring kidney toxicity rats.3,12,16 However, no translational glomerulus-specific injury response biomarkers have been identified. Histopathologic examination has been the gold standard to differentiate glomerular from tubular injury, but it is complicated by the fact that glomerular injury is challenging to detect by standard hematoxylin and eosin (H&E) staining in the earliest phases of injury and that glomerular injury often leads to secondary tubular injury. In addition, glomerular damage is often confirmed by transmission electron microscopy (TEM), which requires specific tissue preparation at the time of necropsy and is too laborious to be used routinely.
Rat kidney toxicity studies were conducted under the kidney working package of Translational Safety Biomarker Pipeline (TransBioLine) with the mission of enabling development and implementation of emerging safety biomarkers in clinical trials and diagnosis of disease. The studies were funded through the European Union’s Innovative Medicines Initiative (IMI), which is a public-private partnership between the European Union and the European pharmaceutical industry. The goal of these studies was to identify novel translational glomerulus-specific response biomarkers to enable glomerular toxicity monitoring in both preclinical species and in the clinic. Data for urinary kidney toxicity biomarkers such as KIM-1 and ALB were also generated in these studies.
Although KIM-1 and ALB have been widely used as kidney toxicity biomarkers, their potential use in differentiating glomerular from tubular injury has not been fully explored. KIM-1 is a type 1 transmembrane protein whose expression is markedly upregulated in the proximal tubule in post-ischemic rat kidneys or upon treatment with nephrotoxicants. Increased urinary KIM-1 level signals tubular injury.7,8 ALB is a 69 kDa negatively charged globular protein and is the most abundant plasma protein. Limited amounts of ALB are filtered and then almost entirely reabsorbed by proximal tubular epithelial cells under normal physiological conditions. Increased levels of ALB could be present in urine when there is glomerular injury that allows large amounts of albumin to enter the glomerular filtrate, or when there is tubular injury that limits ALB reabsorption by compromised tubules.11,14 Therefore, we hypothesized that it should be possible to use the magnitude of relative changes of KIM-1 and ALB to differentiate between glomerular and tubular injury, with no change in either biomarkers indicating no kidney injury; a low magnitude of ALB increase without significant KIM-1 increase indicating glomerular injury; a low magnitude increase of ALB in the presence of significant KIM-1 increase indicating tubular injury; and a high magnitude of ALB increase in the presence of significant KIM-1 increase indicating kidney injury to both glomeruli and tubules.
To explore whether the magnitude of changes in urinary KIM-1 and ALB can be established for supporting such conclusions, we applied a mathematical model and an empirical method to two different data sets: a training set based on 448 individual rats derived from 12 rat kidney toxicity biomarker sensitivity studies, and then a test set from an additional 252 individual rats derived from another 10 rat kidney toxicity studies. Thresholds were developed using the training set, and the performance determined based on the test set. The results of the performance of these algorithms are presented here.
Materials and Methods
Rat Kidney Toxicology Studies
The studies were approved by the Institutional Animal Care and Use Committee (IACUC) of Merck & Co, Inc, Rahway, New Jersey, and were conducted in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) in compliance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals and the Animal Welfare Act.
The design and results of 12 rat kidney biomarker sensitivity studies using nephrotoxicants N-phenylanthranilic acid (NPAA), Cisplatin, Carbapenem A, Cyclosporin A, Gentamicin, Propyleneimine, Hexachloro 1,3 butadiene (HCB), D-Serine, Bacitracin, Puromycin (PAN), Doxorubicin and Thy1.1, as well as 6 rat kidney biomarker specificity studies using non-nephrotoxicants Norethindrone, 17a-Ethynylestradiol, Allylamine, Minoxidil, Bromobenzene, and MSD compound B were previously published and well-described. 16 The experimental designs for the 12 rat kidney biomarker sensitivity studies and 6 rat kidney biomarker specificity studies are summarized in Tables 1 and 2, respectively.
Rat kidney toxicology sensitivity studies used to establish ALB and KIM-1 thresholds.
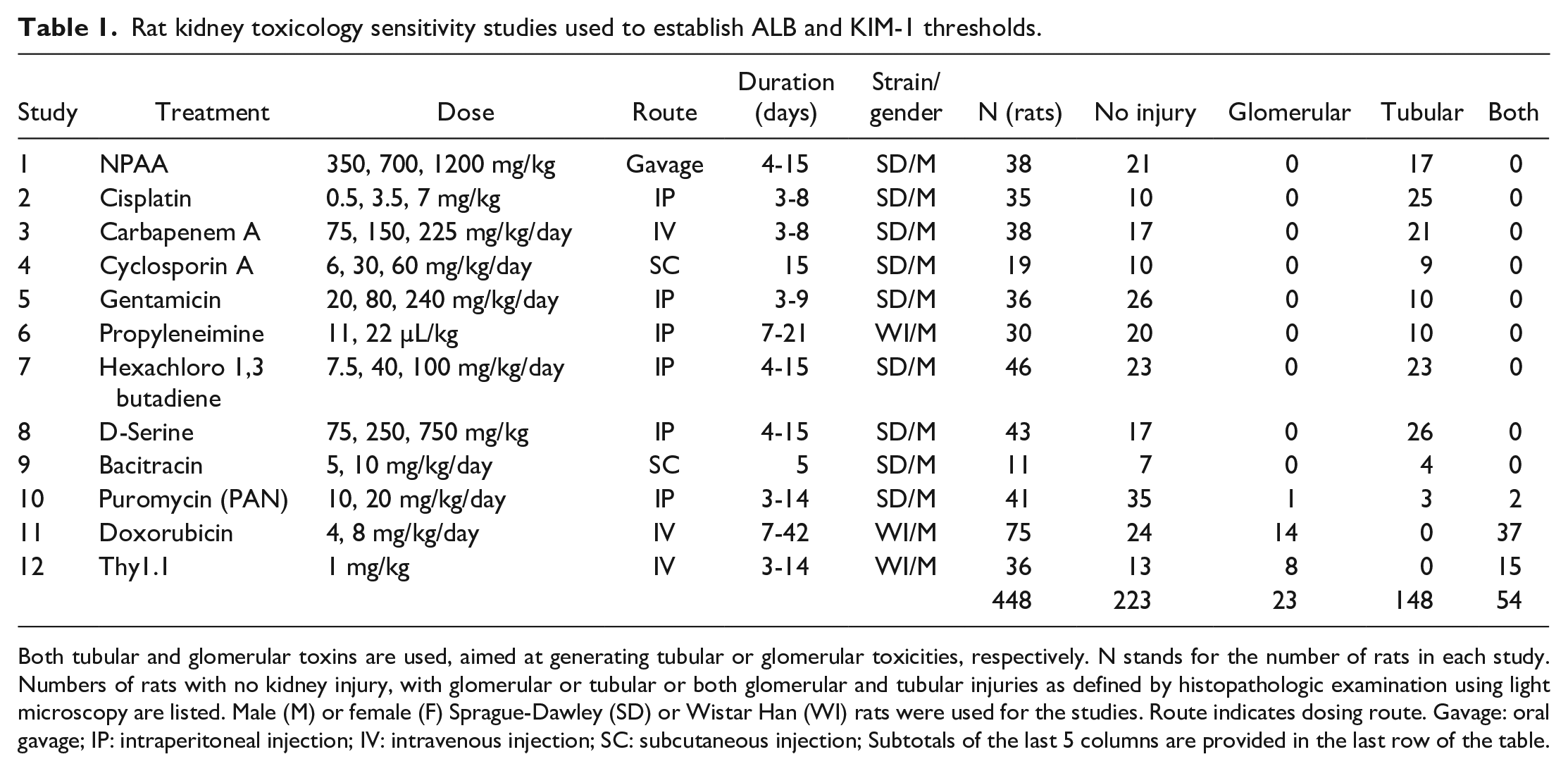
Both tubular and glomerular toxins are used, aimed at generating tubular or glomerular toxicities, respectively. N stands for the number of rats in each study. Numbers of rats with no kidney injury, with glomerular or tubular or both glomerular and tubular injuries as defined by histopathologic examination using light microscopy are listed. Male (M) or female (F) Sprague-Dawley (SD) or Wistar Han (WI) rats were used for the studies. Route indicates dosing route. Gavage: oral gavage; IP: intraperitoneal injection; IV: intravenous injection; SC: subcutaneous injection; Subtotals of the last 5 columns are provided in the last row of the table.
Rat kidney toxicology specificity studies for predicting glomerular and/or tubular toxicity.
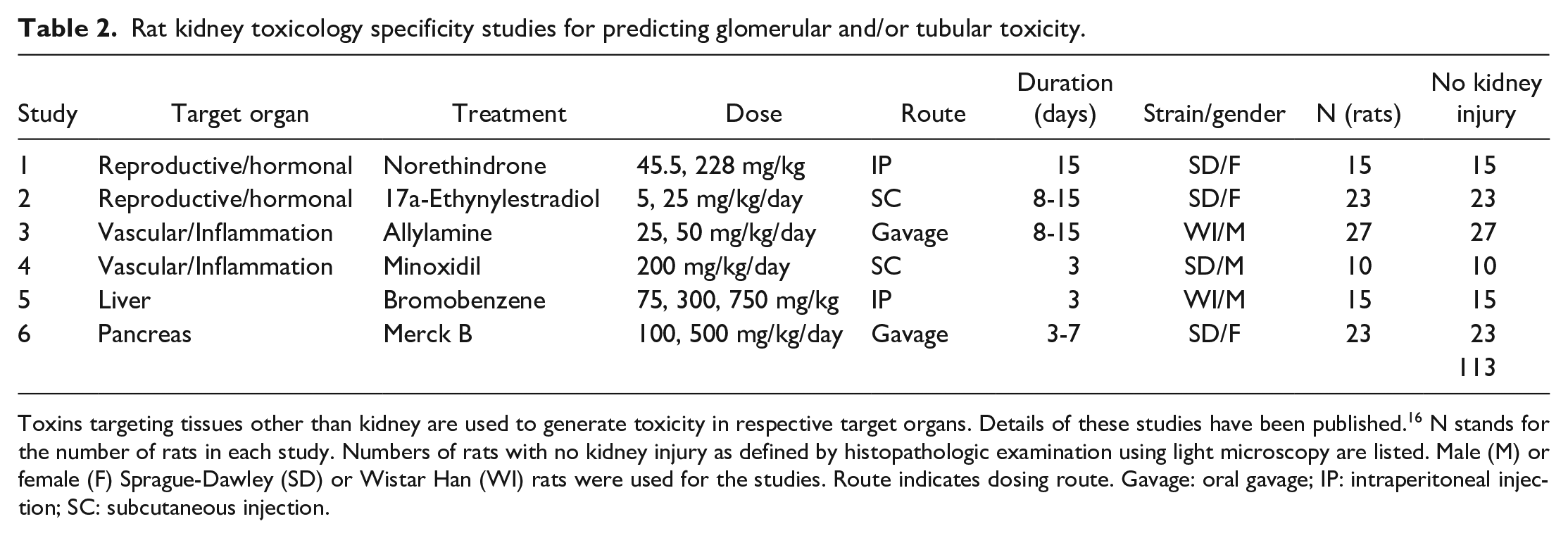
Toxins targeting tissues other than kidney are used to generate toxicity in respective target organs. Details of these studies have been published. 16 N stands for the number of rats in each study. Numbers of rats with no kidney injury as defined by histopathologic examination using light microscopy are listed. Male (M) or female (F) Sprague-Dawley (SD) or Wistar Han (WI) rats were used for the studies. Route indicates dosing route. Gavage: oral gavage; IP: intraperitoneal injection; SC: subcutaneous injection.
Four additional rat kidney toxicity studies using glomerular toxicant MSD compound MK-8511, puromycin, doxorubicin, and tubular toxicant cisplatin were conducted within the framework of TransBioLine IMI. The designs of these studies are summarized in Table 3 and described below. For all 4 studies, rats were euthanized by exsanguination via the vena cava under isoflurane-induced anesthesia on the day of necropsy. Blood for routine serum chemistry was collected from the vena cava into a serum separator tube, and urine was collected overnight (16-18 hours) on wet ice. Kidney tissue was collected from each study for histomorphologic examination. Additional kidney specimens from selected studies were collected from rats at necropsy for ultrastructural evaluation by TEM as described previously. 5
Rat kidney toxicology studies conducted within IMI framework.
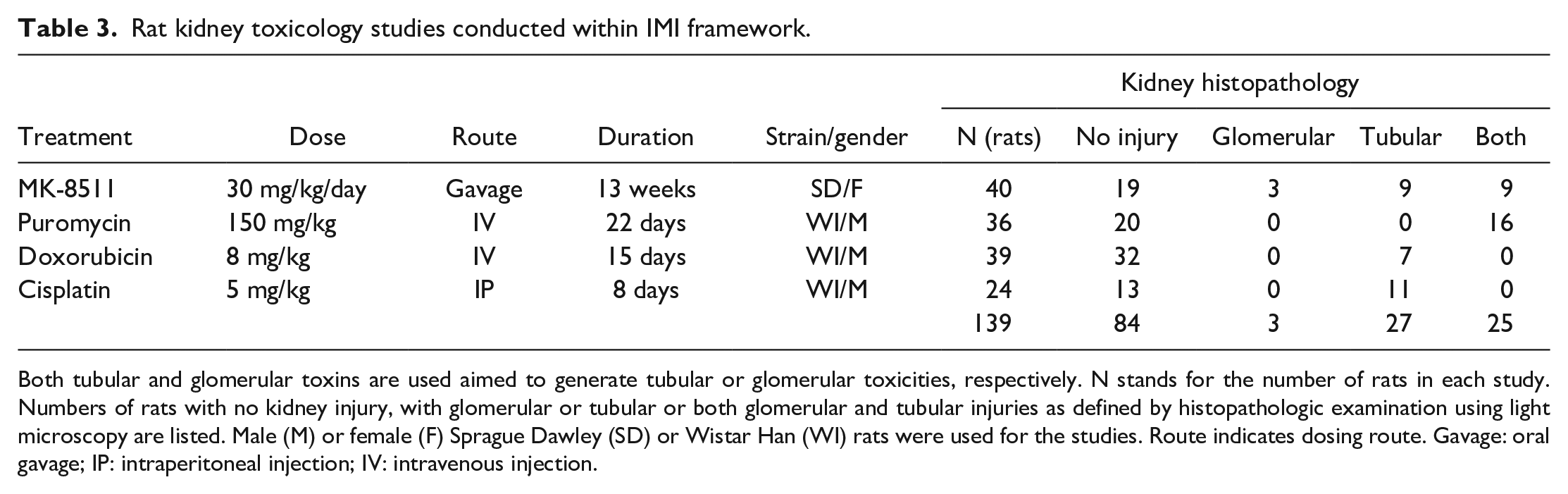
Both tubular and glomerular toxins are used aimed to generate tubular or glomerular toxicities, respectively. N stands for the number of rats in each study. Numbers of rats with no kidney injury, with glomerular or tubular or both glomerular and tubular injuries as defined by histopathologic examination using light microscopy are listed. Male (M) or female (F) Sprague Dawley (SD) or Wistar Han (WI) rats were used for the studies. Route indicates dosing route. Gavage: oral gavage; IP: intraperitoneal injection; IV: intravenous injection.
MK-8511 study
A total of 40 female Sprague Dawley rats at approximately 6 weeks of age (8 controls and 32 treated) were dosed orally with 0.4% (w/v) methylcellulose (vehicle) or MK-8511 (Merck & Co, Inc) at 30 mg/kg/day for up to 13 weeks. Compound MK-8511 was an MSD drug candidate targeting dementia and exhibited glomerular toxicity in long-term rat studies. Albumin levels in urine were monitored biweekly starting from study week 3. The threshold for meaningful changes of urine ALB was set at 300 μg/mg uCr (albumin concentrations were normalized to urine creatinine), a threshold used in clinical settings for macroalbuminuria. 10 The first 6 rats in the treatment group that exceeded the predetermined urine ALB threshold of 300 μg/mg uCr were placed into the recovery group. These 6 animals were dosed for an additional 7 days following their last interim urine collection with urinary ALB above the threshold and then placed on recovery (discontinuation of treatment) with biweekly urine monitoring. These 6 rats were selected for necropsy when their urine ALB levels returned to below 300 μg/mg uCr in urine assessments or at the end of the study (week 13). The remaining animals in the treatment group that exceeded the urine ALB threshold of 300 μg/mg uCr during urine monitoring were selected for interim necropsies. For every 3 rats in the treatment group designated for interim necropsy, 1 control rat with the highest albumin level was designated for concomitant interim necropsy. Any rats remaining after study week 12 were designated for the terminal necropsy. Urine was collected over a 16 to 18 hour period on wet ice at pretest and then biweekly starting at study week 3 from rats fasted overnight. Blood and urine were collected at the time of necropsy.
Puromycin study
A total of 36 male Wistar rats at approximately 8 weeks of age (12 controls and 24 treated) were dosed intravenously with a single dose of 0.9% (w/v) sodium chloride (vehicle) or puromycin (puromycin aminonucleoside, Sigma-Aldrich, Inc, St Louis, Missouri) at 150 mg/kg, respectively. Necropsies were conducted on study days 4, 8, and 22 for 4 control and 8 treated rats per time point. Urine was collected over 16 to 18 hours on wet ice at pretest, study days 4, 8, 15, and 22 from rats fasted overnight, while blood was collected only at necropsy.
Doxorubicin study
A total of 39 male Wistar rats at approximately 8 weeks of age (15 controls and 24 treated) were dosed intravenously with a single dose of 0.9% (w/v) sodium chloride (vehicle) or doxorubicin (doxorubicin hydrochloride, Sigma-Aldrich, Inc) at 8 mg/kg. Necropsies were conducted on study days 4, 8, and 15 for 5 control and 8 treated rats per time point. Urine was collected over 16 to 18 hours on wet ice at pretest, study days 4, 8, and 15 from rats fasted overnight, while blood was collected only at necropsy.
Cisplatin study
A total of 24 male Wistar rats at approximately 8 weeks of age (8 controls and 16 treated) were dosed intraperitoneally with a single dose of 0.9% (w/v) Sodium Chloride (vehicle) or cisplatin (cisplatin, Sigma-Aldrich, Inc) at 5 mg/kg respectively. Necropsies were conducted on study days 4 and 8 for 4 control and 8 treated rats per time point. Urine was collected over 16 to 18 hours on wet ice at pretest, study days 4 and 8 from rats fasted overnight, while blood was collected only at necropsy.
End-Point Measurements
For histopathologic examination, hematoxylin and eosin stained tissue sections were examined, and severity grades were assigned using a 0-5 grading scale: 0 (no observable pathology), 1 (minimal), 2 (mild), 3 (moderate), 4 (marked), and 5 (severe). For the purpose of statistical analysis, the kidney histopathologic findings for both glomerular and tubular injuries were simplified to a binary indication of “yes” or “no.” The TEM was conducted for the MK-8511, puromycin and doxorubicin studies, and only for several selected rats per study.
A panel of routine serum chemistry parameters including BUN, sCr, and urinary creatinine were determined using a Beckman AU5800 Chemistry Analyzer according to manufacturer’s instructions. The collected urine was centrifuged and the supernatant frozen and stored at −70°C for subsequent analyses of urinary biomarkers. Urinary concentrations of rat kidney injury biomarkers KIM-1 and ALB were determined using a custom 3-plex assay (KIM-1, ALB, and clusterin) from Meso Scale Discovery (Rockville, Maryland) according to the manufacturer instructions. Urine creatinine concentration was used to normalize values of each urinary biomarker.
Data Analysis
The urine concentration of KIM-1 and ALB were normalized against the urine creatinine concentrations. The baseline levels of urine biomarkers KIM-1 and ALB were determined as the average of the normalized analyte concentrations of all time points of the control group animals and the normalized pretest analyte concentrations of the treatment group animals. The fold changes were calculated as the normalized analyte concentration at the time of necropsy over the baseline. Data were divided into a training set and a test set, and a tree was built with a classification model using the training set comprised of 448 individual rat data derived from 12 rat kidney toxicity biomarker sensitivity studies (Table 1), The effectiveness of the tree was assessed using the test set comprised of an additional 252 individual rat data derived from another 10 rat kidney toxicity studies (Tables 2 and 3).
Threshold Setting Using the Empirical Method
The threshold for KIM-1 was empirically set at 3-fold according to our previously published work and from internal experience in rat and nonhuman primates.4,6,16 This threshold has worked well for the past studies and is unchanged for this analysis. Of the 448 rats used in the 12 rat kidney biomarker sensitivity studies (Table 1), 114 rats were study controls with no changes in urinary ALB levels. 223 out of the 448 rats (including the 114 controls) had no kidney histopathologic findings with the average urinary ALB fold change to the baseline at 4.8-fold (standard deviation [STDEV] = 25) and the 95% confidence levels between 1.6-fold and 8.0-fold. The rats with urinary ALB changes without renal histopathologic findings were observed primarily in the NPAA and the bacitracin studies. Based on these data, we set the urine ALB threshold at 10-fold above baseline, meaning that when KIM-1 increase is less than 3-fold over the baseline, urinary ALB fold change below 10 would indicate no kidney injury, and above 10 would indicate glomerular injury only. Of the 448 rats, 23 had glomerular injury only and 54 had both glomerular and tubular injury according to the standard histopathologic examination. The urinary ALB fold change from baseline in the glomerular injury only subgroup was 92-fold (STDEV = 100) with the 95% confidence level between 51-fold and 133-fold. Furthermore, the ALB fold change in the rats with both glomerular and tubular injury averaged at 1550-fold with STDEV at 1147-fold (Table 1). Based on these data, we set the urine ALB threshold at 300 fold above baseline (approximately the mean of ALB plus 2 times of STDEV), meaning that when KIM-1 increase is greater than 3-fold above the baseline, urinary ALB fold change below 300 would indicate tubular injury only, and above 300 would indicate both glomerular and tubular injury (Figure 1).
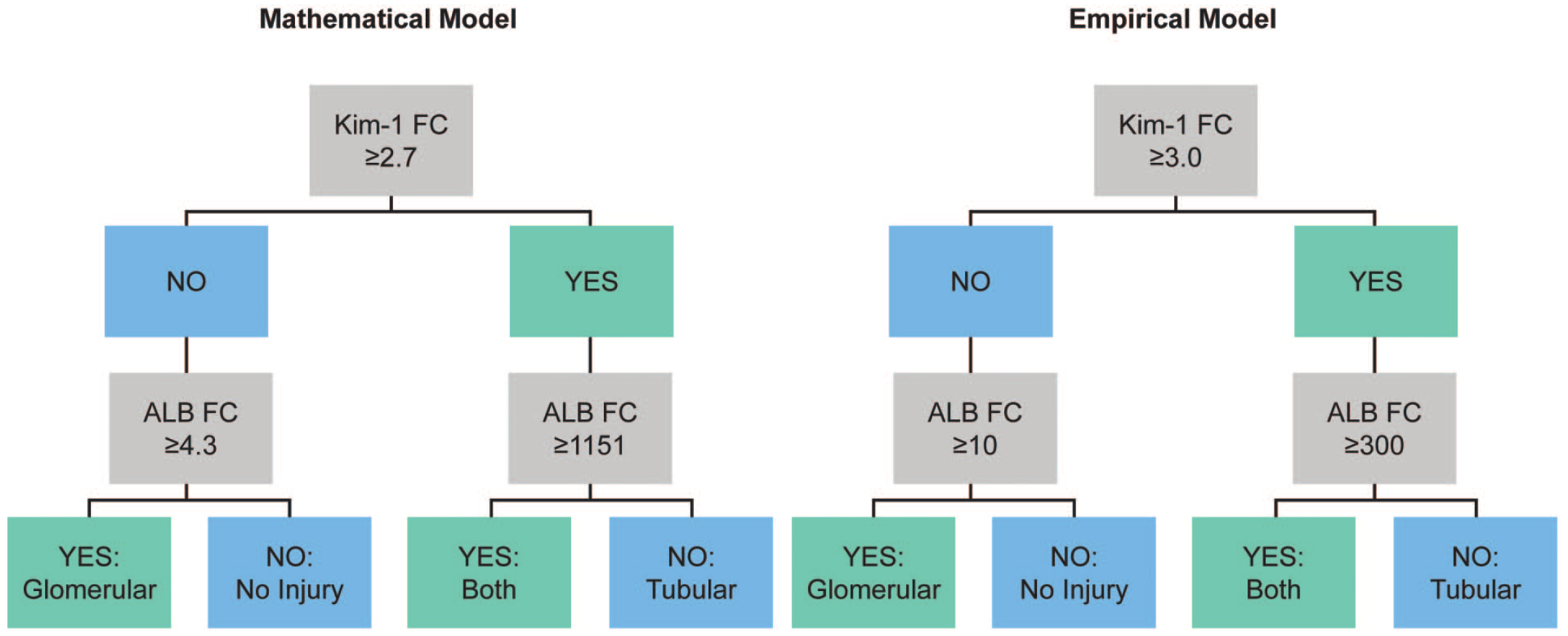
Thresholds of KIM-1 and ALB set by the mathematical and empirical models and the algorithm for kidney toxicity prediction. Both prediction algorithms use urine KIM-1 fold change (FC) as the first level threshold and urine ALB FC as the second level threshold. Glomerular: glomerular injury only; Tubular: tubular injury only; Both: both glomerular and tubular findings.
Threshold Setting Using a Mathematical Model
To classify subgroups based on ALB and KIM-1 fold changes, a mathematical model-based pruning procedure was used on data from 448 rats involved in 12 rat kidney biomarker studies.1,9 The aim was to categorize the rats into four pre-defined subgroups: no kidney injury, glomerular injury only, tubular injury only, or both glomerular and tubular injuries. Pruning is an effective technique for reducing network size while maintaining optimum performance, making it more practical for real-world applications. This technique simplifies the decision tree model by removing unnecessary branches or nodes and involves identifying the parts of the tree that are overfitting and then pruning them to reduce model complexity. The process starts by creating a decision tree model with default parameters and a complexity parameter (CP) to control tree growth. If the cost of adding a variable is higher than the value of CP, tree growth stops.
For the calculation, the rpart package in R 4.1.0 (R Core Team, 2021) was used. This package offers convenient tools for decision tree pruning, providing functions and methods that streamline the pruning process and enable easy adjustment of the CP. The rpart package develops classification or regression models using a two-stage method, 13 resulting in models represented as binary trees. In this analysis, the tree was built by the following steps: (1) the single variable was found which best splits the data into two and (2) based on the separated data, this process is applied to each subgroup until no improvement can be made or reach a minimum size defined by the user. The CP plays a role in determining which splits should be pruned. For specific parameter specifications, refer to the R code in Supplement.
Based on the pruning method, the threshold for KIM-1 fold change is established at 2.7-fold. When the KIM-1 fold change is less than 2.7-fold, the model predicts that there is no tubular injury. The tree is further separated using ALB fold change at 4.3-fold, meaning the model predicts there is glomerular injury if ALB fold change is greater than 4.3-fold, otherwise the model predicts there is no injury. On the contrary, if the KIM-1 fold change is greater than 2.7-fold, the model predicts there is tubular injury. The tree is further separated using ALB fold change at 1151-fold, meaning that both glomerular and tubular injuries are predicted if ALB fold change is greater than 1151-fold, otherwise the model predicts tubular injury only (Figure 1).
Prediction of Kidney Injury
To check whether the classification model performs well, we used a confusion matrix, which calculates the performance of a classifier and presents a table design of the different consequences of the prediction and provides the result of a classification problem that helps to visualize the data. We obtained four different combinations from the predicted and actual values of a classifier using the performance matrix. (1) True Positive (TP); (2) False Positive (FP); (3) True Negative (TN); and (4) False Negative (FN). Using these measures, the overall accuracy of the model was calculated using the formula: Accuracy = (TP + TN)/(TP + TN + FP + FN). The four other measures were (1) Sensitivity = TP/(TP+FN); (2) Specificity = TN/(TN+FP); (3) Positive predictive value (PPV) = TP/(TP + FP); and (4) Negative predictive value (NPV) = TN/(TN + FN).
Results
The kidney histopathologic findings and biomarker results, including KIM-1 and ALB of the 12 rat kidney biomarker sensitivity studies and 6 rat kidney biomarker specificity studies, have been described previously. 16 The total numbers of rats used in those studies, and the numbers of rats with no kidney findings, glomerular findings only, tubular findings only or both glomerular and tubular findings are summarized in Table 1. There were no kidney histopathologic findings in the 6 rat kidney biomarker specificity studies (Table 2). The ALB and KIM-1 fold changes over baseline in these rat studies are summarized in Figure 2.
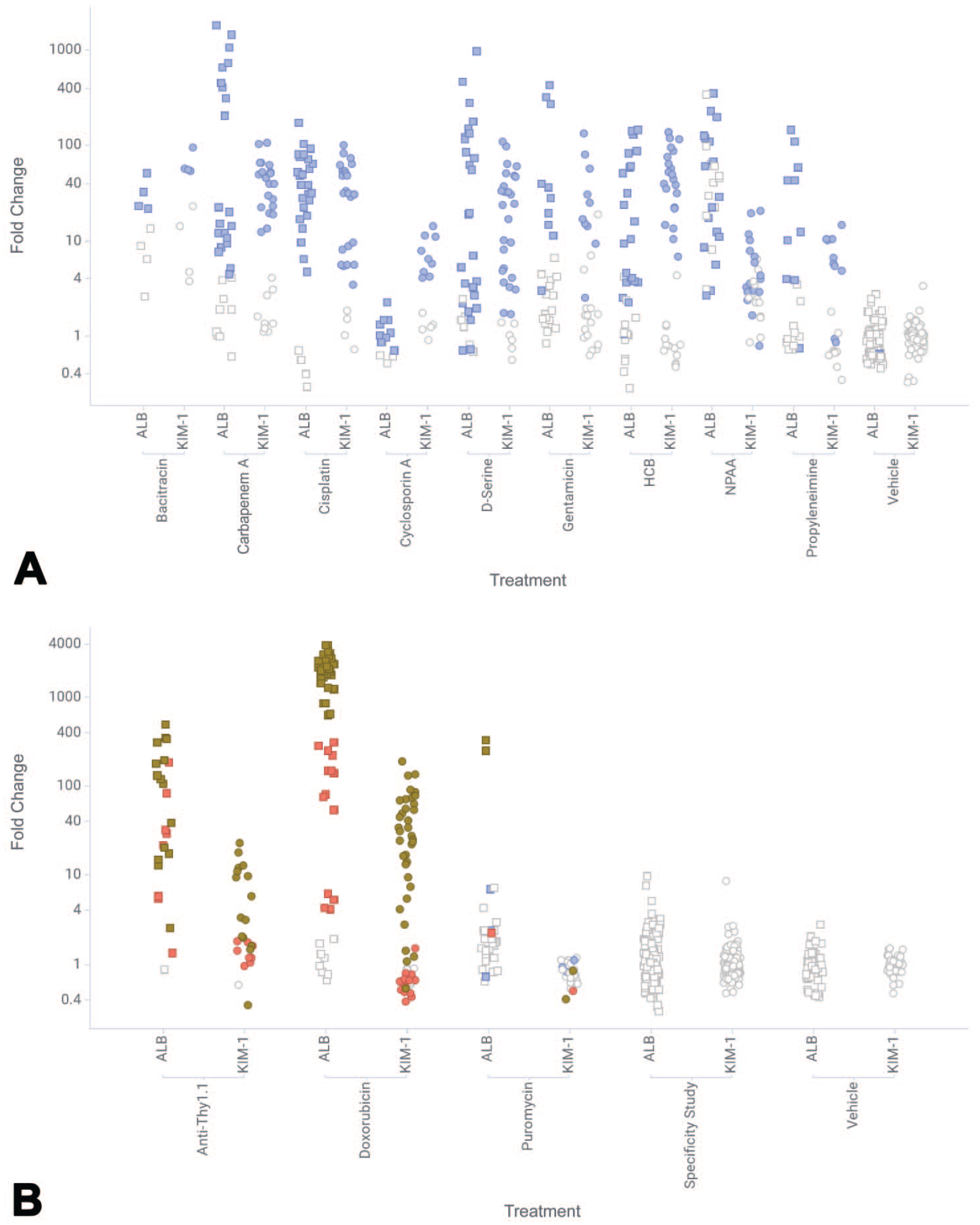
Fold changes of KIM-1 and ALB of the 12 kidney biomarker sensitivity studies and 6 specificity studies. (A) Tubular toxicant studies; (B) Glomerular toxicant studies and specificity studies. Fold changes of ALB (square) and KIM-1 (circle) over baseline are plotted for each rat study. The vehicle controls of all sensitivity studies are binned together. All rats in the specificity studies are binned together as well, due to no kidney findings in these studies. The colors indicate the histopathologic findings. White: no kidney injury; Blue: tubular injury only; Red: glomerular injury only; Brown: both tubular and glomerular findings.
Our aim for the 4 rat kidney biomarker studies conducted within the framework of TransBioLine was to induce minimal kidney injury targeting glomeruli or tubules. The total numbers of rats included in these studies and the numbers of rats with no kidney findings, glomerular findings only, tubular findings only or both glomerular and tubular findings are summarized in Table 3. The urinary ALB and KIM-1 fold changes over baseline in these 4 rat studies are summarized in Figure 3.
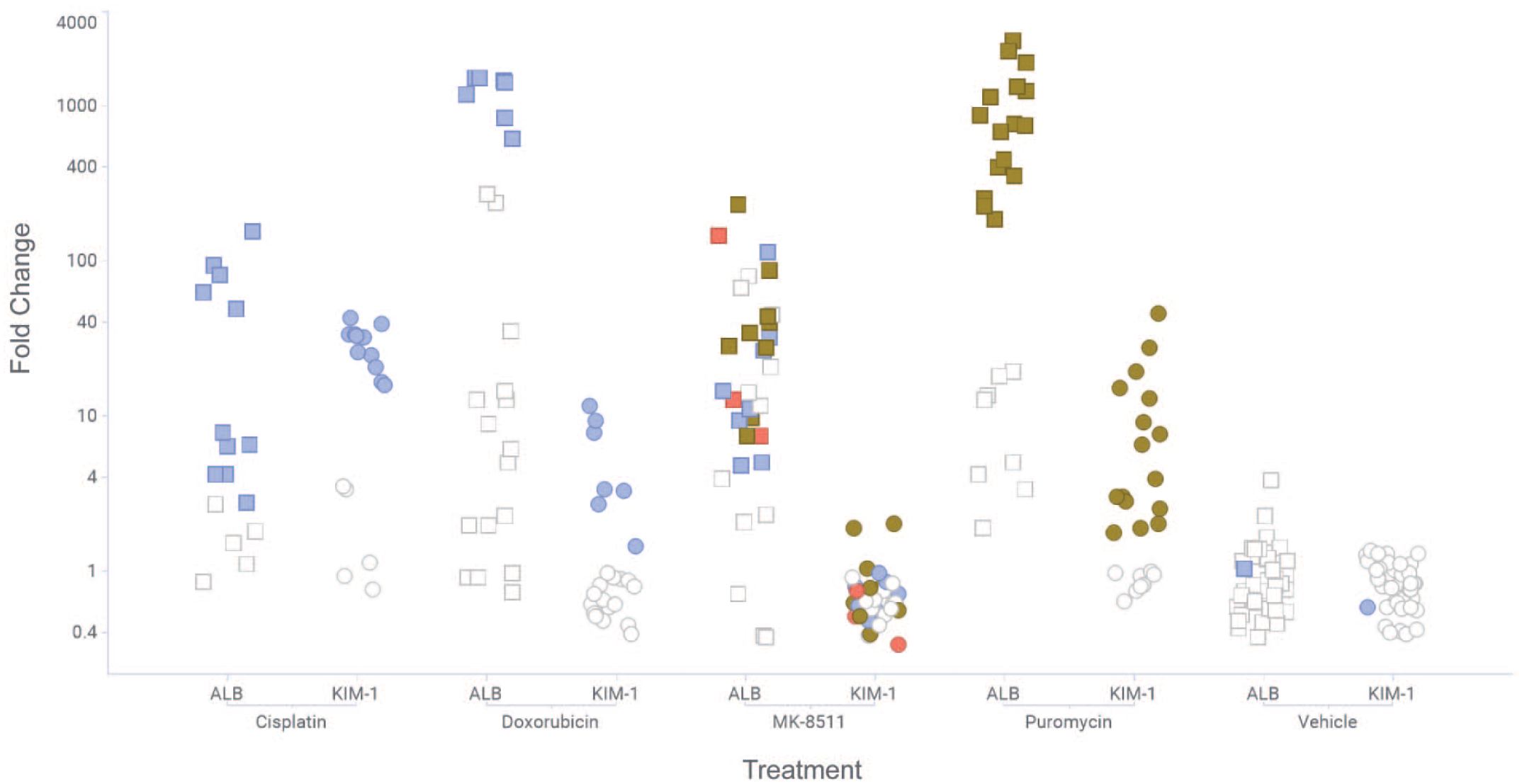
Fold changes of KIM-1 and ALB in the 4 rat studies conducted within the IMI framework. Fold changes of ALB (square) and KIM-1 (circle) over baseline are plotted for each rat study. The controls of the 4 studies are binned together. The colors indicate the histopathologic findings. White: no kidney injury; Blue: tubular; Red: glomerular; Brown: both tubular and glomerular findings.
MK-8511 Study
The intent of this study is to induce low-grade glomerular injury in rats. Administration of MK-8511 at 30 mg/kg/day by daily oral gavage to female Sprague-Dawley rats for up to 13 weeks resulted in histomorphologic changes in the kidney including tubular casts, glomerulopathy, tubular epithelial basophilia, and increased tubular epithelial pigment. Ultrastructurally, effacement (giving the appearance of fusion) of foot processes in glomerular podocytes and/or increased quantities of protein droplets in the cytoplasm of podocytes were observed in 2 of the 3 test-article–treated animals evaluated by TEM. Kidney histomorphologic changes persisted following the 4-week to 7-week recovery phase with some animals showing trends toward recovery with reduced incidence and/or severity of tubular basophilia.
There were no changes in serum biochemistry parameters in the 13-week MK-8511 rat study. MK-8511 treatment resulted in increased urinary albumin levels over the course of the study. At the time of necropsy, 18 rats still exhibited elevated urine ALB levels above the threshold of 300 µg/mg uCr, with fold changes from control baseline ranging from 13-fold to 231-fold. Increases of both KIM-1 and CLU were not observed at the time of necropsy, but 2 treated rats showed a transient increase of both KIM-1 and CLU levels during the course of the study and they are among the 13 rats that showed tubular basophilia at necropsy.
All MK-8511–treated rats with renal findings showed increased urinary ALB levels, most with urine ALB levels greater than 300 µg/mg uCr. However, there were several rats with high albumin levels that did not show glomerulopathy on light microscopy. The rats that underwent the recovery phase generally had reduced urinary ALB levels.
Puromycin Study
The intent of this study is to induce low-grade glomerular injury in rats. Mild renal glomerulopathy was evident under light microscopic examination in puromycin-treated rats on study days 8 and 22. The glomerulopathy was characterized by eosinophilic thickening within the glomerular tufts. In affected rats, there was a marked accumulation of acellular eosinophilic material within the lumen of tubules (hyaline casts) extending from the cortex into the papilla on study day 8, and a mild accumulation of hyaline casts primarily in the papilla on study day 22. Associated with the accumulation in the tubular lumen on study day 8, there was mild tubular epithelial cell degeneration, accumulation of eosinophilic droplets in the tubular epithelial cytoplasm, and occasional exfoliation of the tubular epithelial cells. On study day 22, tubular degeneration was not evident, and there was mild tubular basophilia (consistent with a regenerative process) accompanied by a focal increase in interstitial lymphocytes. Ultrastructural evaluation of glomeruli by TEM showed alterations in glomerular podocyte foot processes such as shortening and effacement in specimens from treated rats collected on study days 4 and 8. These findings correlated with the glomerulopathy observed by light microscopic examination on study day 8.
There was a very slight increase in BUN on study days 4 and 8. Urinary ALB increases were observed at study day 4 in 7 out of 8 animals. This increase was approximately 4-fold to 20-fold above the baseline. Urinary ALB levels further increased on study days 8 to approximately 750-fold to 2600-fold over baseline. Urinary ALB levels decreased from peak values to approximately 250-fold to 850-fold over baseline on study day 22. Urinary KIM-1 showed no increase on study day 4. However, KIM-1 levels increased in all treated animals to approximately 7-fold to 46-fold over baseline on study day 8, followed by a decline on study day 22, when only three rats had marginally elevated KIM-1 levels at approximately 3-fold to 4-fold over baseline.
Doxorubicin Study
The intent of this study was to induce low-grade glomerular injury in rats. There were no kidney histopathologic findings in treated rats on study days 4 and 8. On study day 15, there was minimal to moderate renal tubular degeneration characterized by an increase in homogeneous eosinophilic material within dilated tubules, occasional exfoliated cells within the tubules, accumulation of eosinophilic globules within tubular epithelial cells, and in some rats, segmental renal tubular basophilia. Bowman’s space in some glomeruli was expanded and filled with similar eosinophilic material, but histologic changes were not evident in glomerular tufts by light microscopy. Transmission electron microscopy was performed on specimens collected from the renal cortex on study days 4, 8, and 15. Doxorubicin-treated rats exhibited blunting, effacement, and loss of glomerular podocyte foot processes, evident starting on study day 8. Effacement of the foot processes continued on study day 15.
There were no notable changes in BUN or sCr in this study. Doxorubicin treatment resulted in increases of urinary ALB starting from study day 8, with a group average fold change of treated over control on study day 8 and study day 15 of 50-fold and 1332-fold, respectively. Urinary KIM-1 was elevated starting on study day 15, with 5-fold average changes of KIM-1 in treated rats relative to controls.
Cisplatin Study
The intent of this study was to induce low-grade tubular injury in rats. Cisplatin-induced minimal to moderate renal tubular degeneration was present in the kidneys of 5 out of the 8 treated rats on study day 4. Moderate renal tubular degeneration was also present in 6 out of the 8 treated rats on study day 8.
There were no test article-related changes in sCr or BUN. Increased urinary levels of the kidney safety biomarkers KIM-1 and ALB were observed on both study day 4 and study day 8. The elevations of these urinary biomarkers matched very well with the tubular findings in kidneys of the treated rats.
Prediction accuracy
The overall prediction accuracy for the 448 rat training set was at 86% and 84% using the KIM-1 and ALB thresholds set by the mathematical model and the empirical method, respectively (Table 4).
Performance of the mathematical model and the empirical model on the training data set.
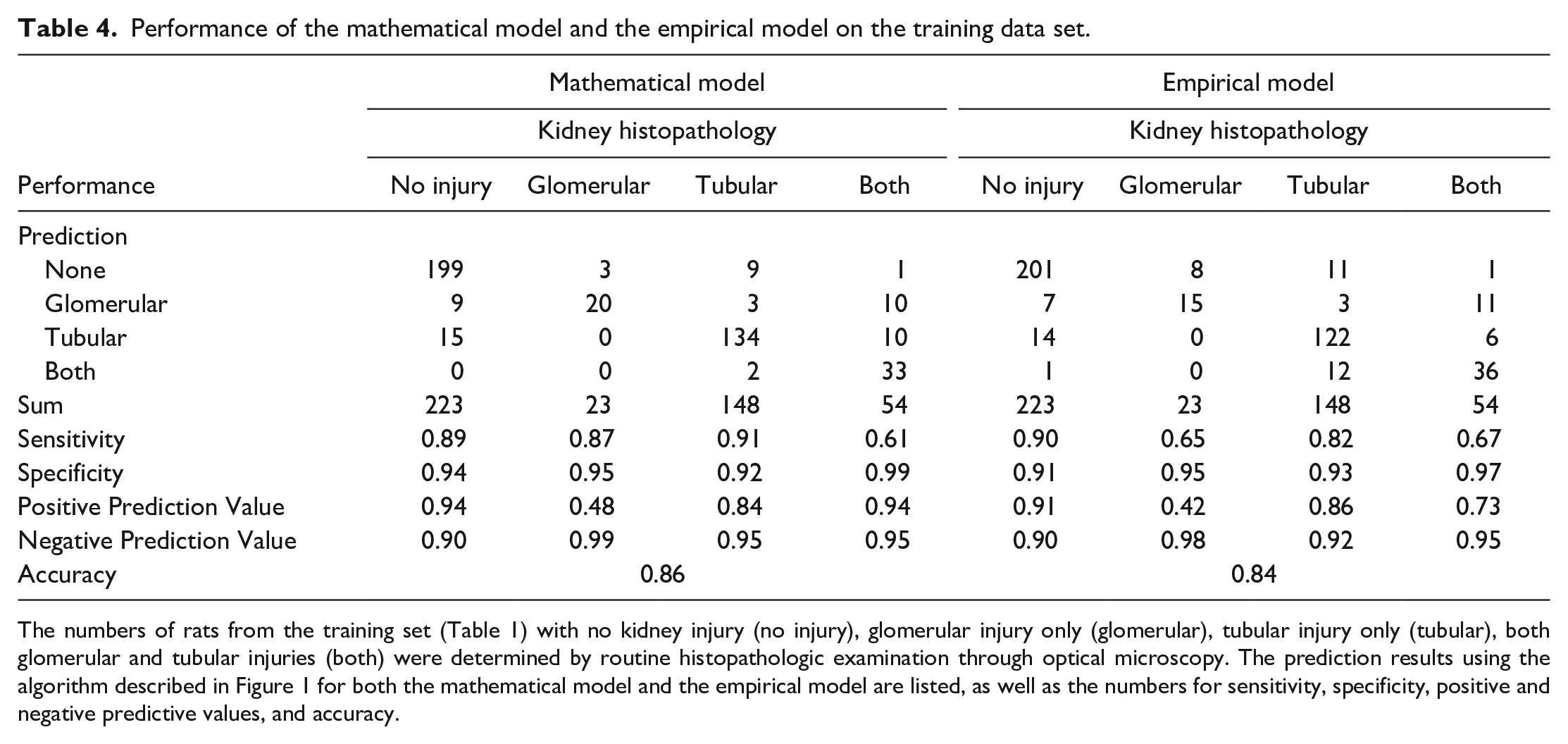
The numbers of rats from the training set (Table 1) with no kidney injury (no injury), glomerular injury only (glomerular), tubular injury only (tubular), both glomerular and tubular injuries (both) were determined by routine histopathologic examination through optical microscopy. The prediction results using the algorithm described in Figure 1 for both the mathematical model and the empirical model are listed, as well as the numbers for sensitivity, specificity, positive and negative predictive values, and accuracy.
The thresholds of KIM-1 and ALB set by either the mathematical model or the empirical method were tested with a data set of 252 rats including the 6 kidney biomarker specificity rat studies (Table 2) and the 4 rat kidney toxicity studies conducted under the IMI TransBioLine framework (Table 3). The performances of the 2 sets of cutoffs including the sensitivity, the specificity, and the positive and negative prediction values are shown in Table 5. The overall predictive accuracy for the 252 rat test set was 75% for the mathematical model and 80% for the empirical method.
Performance of the mathematical model and the empirical model on the test data set.
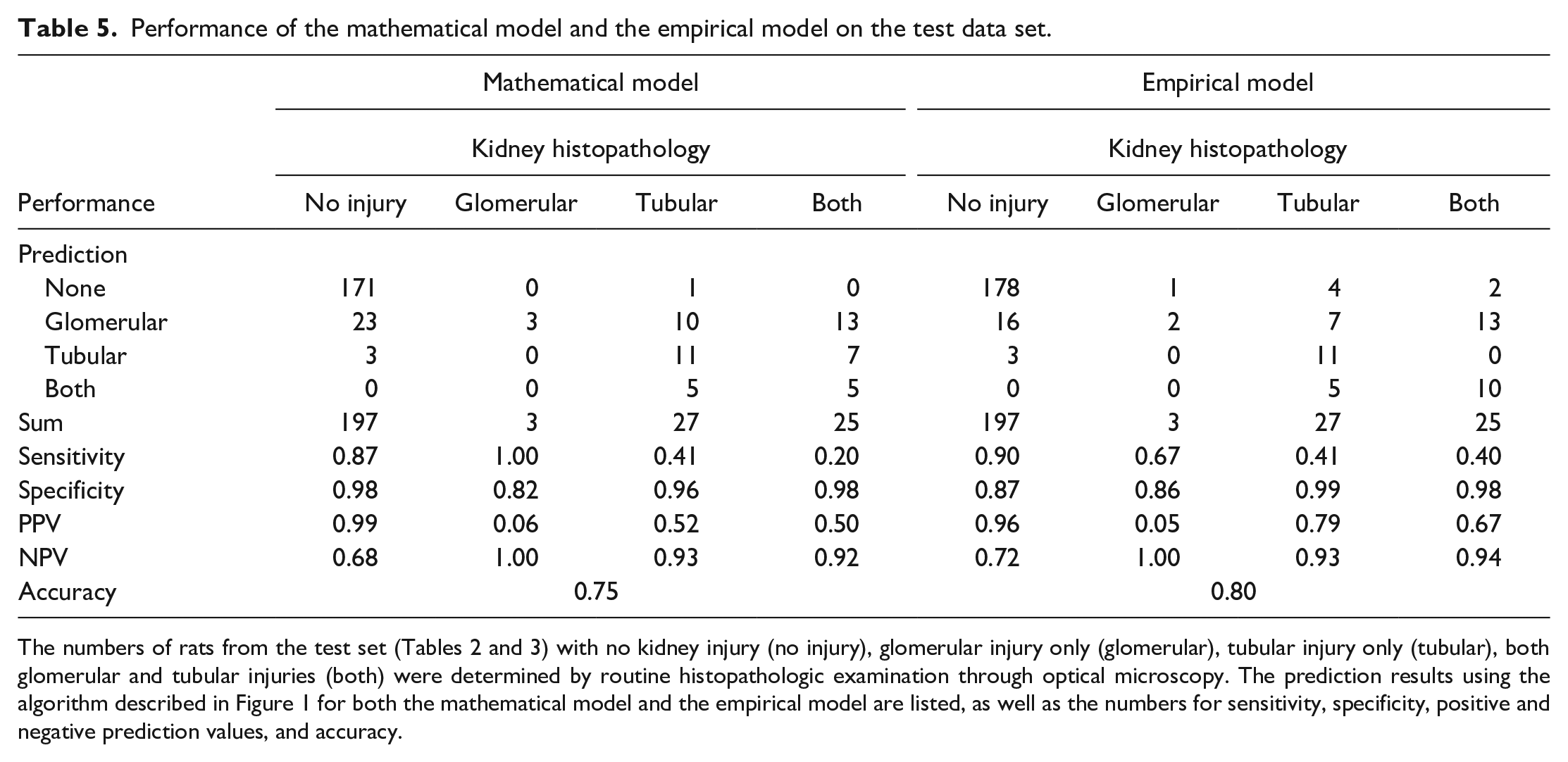
The numbers of rats from the test set (Tables 2 and 3) with no kidney injury (no injury), glomerular injury only (glomerular), tubular injury only (tubular), both glomerular and tubular injuries (both) were determined by routine histopathologic examination through optical microscopy. The prediction results using the algorithm described in Figure 1 for both the mathematical model and the empirical model are listed, as well as the numbers for sensitivity, specificity, positive and negative prediction values, and accuracy.
Discussion
The aim of this assessment was to test the feasibility of differentiating glomerular versus tubular toxicity in rat kidneys by examining only the urinary KIM-1 and ALB fold changes above the baseline. We used a 448 rat data set to establish the KIM-1 and ALB thresholds using either the mathematical model or the empirical method, and then applied these thresholds to predict the kidney injury outcome using a data set of 252 rats from 10 rat kidney toxicity studies. The overall prediction accuracy for both models exceeds 75%, indicating that this approach is encouraging and provocative. To our knowledge, this is the first such effort to predict origins of kidney injury using the magnitude of fold changes of only two urinary kidney safety biomarkers in rat.
Unlike tubular toxicity, the initial changes of glomerular injury cannot be definitively identified with the standard optical microscopy used in routine histopathologic examination of H&E-stained tissues. This point is well illustrated in the puromycin and the doxorubicin studies. In the puromycin study, the increase in urinary ALB on study day 4 without the concomitant increase in KIM-1 was indicative of glomerular changes, even though those changes were not yet observable by light microscopy. The presence of ultrastructural alterations of glomerular foot processes on study day 4 observed by TEM, before glomerulopathy was evident by light microscopy, was consistent with the observed increase in ALB on study day 4. In the doxorubicin study, the increase in urinary ALB on study day 8 was indicative of glomerular changes that were identified by TEM but were not yet observable by light microscopy at that time point. Furthermore, this pattern was also seen in the Long Evans Hooded (LEH) leucine-rich repeat kinase 2 (LRRK2) knockout rats. 5 In these studies, the glomerular injury in the rats was not detectable by routine histopathologic examinations but was predicted by both the mathematical model and the empirical algorithms. Therefore, the histopathologic predictions using these algorithms were not only feasible, but they were also complementary to routine histopathology gold standards.
The fold changes of urinary ALB over baseline were used for threshold setting instead of an absolute value in this study. This decision is made due to variation in absolute urinary ALB values of the control rats observed across the studies, which were conducted over a time span of 2 decades. The variations could be due to several factors, such as rat strain variations and the use different lots of ALB measurement kits. In the more recently conducted four rat studies under the IMI TransBioLine framework, the average baseline of urinary ALB is approximately 30 μg/mgCr, thus placing the 10-fold threshold from the empirical method at an absolute value close to 300 μg/mgCr. In clinical practice, urinary ALB values of 30 μg/mgCr and 300 μg/mgCr are designated as the thresholds for microalbuminuria and macroalbuminuria, respectively. 10 Urinary ALB was one of the eight kidney safety biomarkers measured in a kidney safety biomarker qualification phase 1 trial. 2 Recently, urinary ALB level normalized to creatinine was found to be one of the parameters associated with the histological diagnosis of glomerular disease in a human in a clinical cohort with kidney disease. 15 It is conceivable that in the clinic setting, urinary ALB threshold can either be a fold change or an absolute value depending on the outcomes of clinical research.
The PPVs for glomerular injury seemed very low at 6% and 5% for the mathematical model or the empirical method, respectively (Table 5). This is due to the low number of rats with glomerular injury only, and the high number of FPs for glomerular injury, with a large portion of the FPs from the no injury subgroup. In the puromycin study, increased urine ALB levels in 5 rats exceeded the 4.3-fold threshold on study day 4, with 4 of the 5 rats exceeding the 10-fold ALB threshold, consistent with ultrastructure changes observed with electron microscopy, whereas those changes were not observed within routine histopathologic examination. Similarly in the doxorubicin study, urinary ALB levels in all 8 rats on study day 8 exceeded the 4.3-fold threshold, with 5 of the 8 rats exceeding the 10-fold ALB threshold, indicating glomerular injury on study day 8 that was observable by TEM but not by routine histopathologic examination. Considering the increase of urinary ALB and the presence of glomerular injury observable by TEM at those time points, as well as the fact that the rats in these studies did develop glomerular injury observable under optical microscopy at later time points in these studies, it is reasonable to assume that the rats with increased ALB levels were true positives for glomerular injury at those early time points. If we re-calculate the PPVs according to this assumption, then the PPVs for glomerular injury would have improved to 31% and 29% for the mathematical model and the empirical models, respectively.
Although the principal of this approach seems to work, there are limitations to the analyses presented here. Although the 448 rat data set used to set the thresholds for KIM-1 and ALB is a sizable data set, the fact the ALB thresholds determined by the mathematical model and the empirical method were not very close was at least partly due to the limited size of the training data set. In particular, the number of rats with only glomerular injury was small. The performance of these models is expected to improve upon the addition of more existing and future rat kidney toxicity study data, to improve the quality and size of the training data set.
The prediction accuracy for the training set were 86% and 84% for the mathematical model and the empirical model, respectively (Table 4). It is expected that the mathematical model would generate better prediction accuracy for the training set although the performance of the two models are comparable in this instance. The prediction accuracy for the test set was 75% and 80% for the mathematical and the empirical models, respectively (Table 5). The prediction errors were mainly from the relatively poor sensitivities for the tubular injury only subgroup and the tubular and glomerular injury subgroup. The intention of creating low-grade kidney injury in the 4 IMI TransBioLine rat studies in the test set contributed to an overall lower magnitude of biomarker fold changes compared with the training set and the lower sensitivities observed. Although the empirical thresholds performed a little better for the test set, as we continue to expand the size of the training and test data sets, we believe that the mathematical model would eventually outperform the empirical model. At the present time, we think the kidney histopathologic predictions should be applied at the level of treatment groups rather than at the individual animals due to the current prediction accuracy.
To simplify the model, we treated both high-grade and low-grade tubular injury the same way and just labeled them being positive for tubular injury. However, there were far more cases with grade 1 and 2 injury than grade 3 and 4 injury. This could potentially create a bias in setting ALB at a lower threshold. Rats with high-grade tubular-only injury could exhibit magnitude of fold changes of KIM-1 above 3-fold and ALB above 300-fold, as observed in the Carbapenem A study, were classified into the “both glomerular and tubular injury” category in the empirical model instead of being binned into the tubular injury only category. Again, we expect the future expansion of the training data set would help to set better thresholds.
In summary, through statistical analysis of the results of a set of rat kidney toxicity studies, we showed that it is possible to differentiate glomerular injury from tubular injury using the magnitude of urinary KIM-1 and ALB changes in rats (Figure 1). Although there are rooms for improvement for both models, we deduce that such an approach could be applicable in clinical settings as well, given the ever increasing volume of kidney biomarker data in human. It would be highly significant to fill in the current void in medical practice if the accessible biomarkers can be used to predict the status of kidney injury to help the monitoring and clinical intervention of kidney diseases.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233241248656 – Supplemental material for Magnitude of Urine Albumin and KIM-1 Changes Can be Used to Differentiate Glomerular Injury From Tubular Injury in Rats
Supplemental material, sj-docx-1-tpx-10.1177_01926233241248656 for Magnitude of Urine Albumin and KIM-1 Changes Can be Used to Differentiate Glomerular Injury From Tubular Injury in Rats by Yi-Zhong Gu, Erina Paul, Katerina Vlasakova, Sean P. Troth, Frank D. Sistare, Lila Ramaiah, Oliver Potz, Santosh Sutradhar and Warren E. Glaab in Toxicologic Pathology
Footnotes
Acknowledgements
The authors thank colleagues in Department of Nonclinical Drug Safety, Merck & Co, Inc, Rahway, New Jersey, for conducting the rat studies, generating data and providing data interpretation, as well as Dr Sophia Samodelov for her critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The TransBioLine project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No. 821283. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation program and the European Federation of Pharmaceutical Industries and Associations (EFPIA). This communication reflects the author’s view and neither IMI nor the European Union or EFPIA are responsible for any use that may be made of the information contained therein.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
