Abstract
The 2023 annual Division of Translational Toxicology (DTT) Satellite Symposium, entitled “Pathology Potpourri,” was held in Summerlin, Nevada, at the Society of Toxicologic Pathology’s 41st annual meeting. The goal of this symposium was to present and discuss challenging diagnostic pathology and/or nomenclature issues. This article presents summaries of the speakers’ talks along with select images that were used by the audience for voting and discussion. Various lesions and topics covered during the symposium included induced and spontaneous neoplastic and nonneoplastic lesions in the mouse liver, infectious and proliferative lesions in nonhuman primates, interesting presentations of mononuclear cell infiltrates in various animal models and a complex oral tumor in a rat.
Keywords
Brought to You by the Letter “H”: Hepatocellular Carcinoma, Hepatoblastoma, and Hemangioma/Hemangiosarcoma
The symposium began with an introduction by Drs Robert Sills and Mark Cesta of the Division of Translational Toxicology, National Institute of Environmental Health Sciences (DTT/NIEHS) in which they presented a brief overview of the DTT’s mission and current initiatives. Dr Erin M. Quist (Charles River Laboratories, Inc., [CRL] Durham, North Carolina) then presented several interesting cases recently reviewed by a pathology working group (PWG) as part of a special review conducted by the DTT/NIEHS. The presented cases featured neoplastic lesions from B6C3F1/N mice that were part of a 2-year toxicity/carcinogenicity study in which Dr Michael Ryan (Battelle, Columbus, Ohio) was the study pathologist (SP), the Quality Assessment Pathologist (QAP) was Dr Crystal Johnson ([then] CRL, Durham, North Carolina), and the reviewing pathologist (RP) and PWG Coordinator (PWGC) for the special review was Erin Quist of CRL (Durham, North Carolina). Thanks to Dr Charan Ganta and the NIEHS Histology Core Laboratory for assistance with immunohistochemistry (IHC), Emily Singletary and Leslie Couch for materials acquisition, whole slide imaging and PWG technical support, and Beth Mahler for photographic editing.
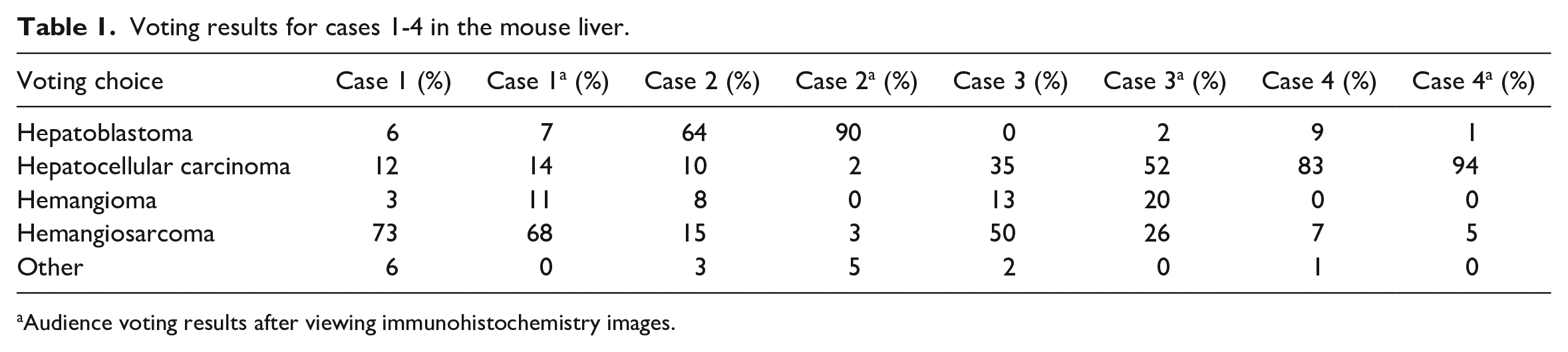
For all 4 cases, a series of photomicrographs were presented to the audience that included both low- and high-power magnifications of select sections of liver from the 2-year study (Figure 1). Dr Quist informed the audience that the diagnostic choices would be the same for all 4 cases and that the audience would be asked to revote after viewing IHC images for each case; the voting choices and results are presented in Table 1.
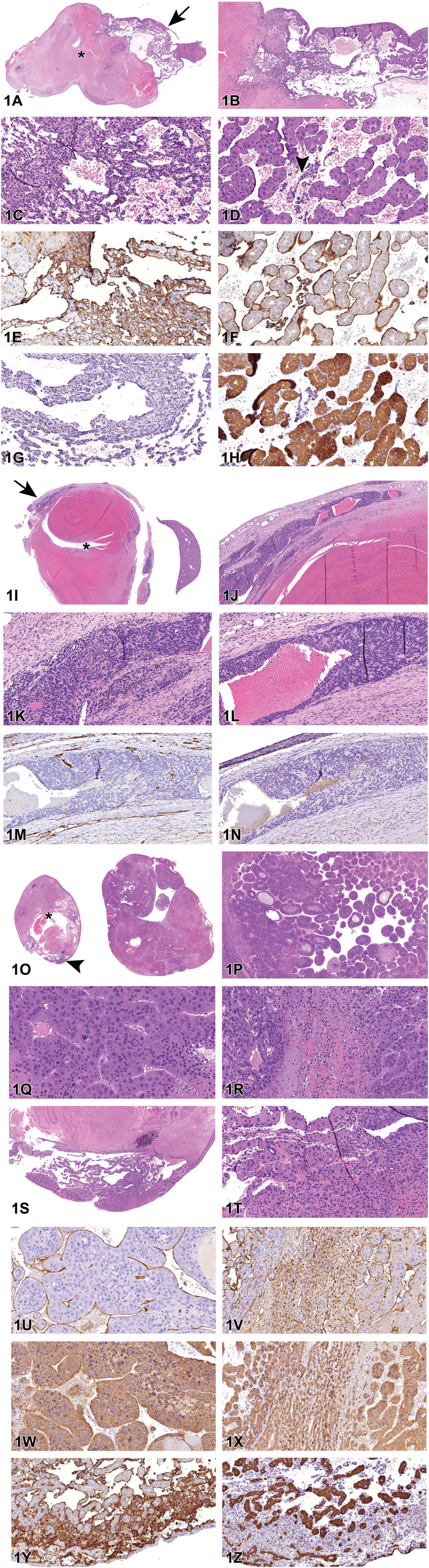
Mouse liver tumors, B6C3F1/N mouse. (A-H) Hemangiosarcoma, HE. (A) There is a poorly demarcated, unencapsulated neoplasm along the periphery of the liver (arrow). 4×. Note large area of thrombosis (*). 2×. (B and C) The neoplasm is characterized by poorly formed, haphazardly arranged, vascular channels lined by pleomorphic endothelial cells. 20×. (D) Vascular channels often expanded sinusoidal areas, disrupting the normal lobular architecture and creating large, blood-filled spaces in between the remaining hepatic cords; blind-ended trabeculae were occasionally observed (arrowhead). 20×. (E-H) Hemangiosarcoma, IHC. (E-H) Neoplastic cells exhibited positive CD31 staining (E and F, 20×) but were negative for CK18 (G and H, 20×); note that remaining hepatocytes stained intensely positive with CK18 (H, 20X). (I-N) Hepatoblastoma, HE.
Voting results for cases 1-4 in the mouse liver.
Audience voting results after viewing immunohistochemistry images.
Case 1 provided an example of liver—hemangiosarcoma (Figure 1A-H). The liver section was largely thrombosed with a poorly demarcated, unencapsulated neoplasm along the periphery that was characterized by poorly formed, haphazardly arranged, vascular channels lined by pleomorphic endothelial cells (Figure 1A-D). Vascular channels often expanded sinusoidal areas, disrupting the normal lobular architecture and creating large, blood-filled spaces in between the remaining hepatic cords (Figure 1C and D). Neoplastic cells occasionally formed blind-ended trabeculae that could be observed at a higher magnification (Figure D, arrowhead). Mitotic figures were rare. To help differentiate the neoplastic cell type and confirm the diagnosis of liver—hemangiosarcoma, the following IHC stains were used: cluster of differentiation (CD)31 (endothelial cell marker) and cytokeratin (CK)18 (hepatocellular marker). Neoplastic cells exhibited positive CD31 staining (Figure 1E and F) but were negative for CK18 (Figures 1G and H), while remaining hepatocytes stained intensely positive with CK18 which served as a great internal, positive control (Figure 1H).
Case 2 featured an example of liver—hepatoblastoma in another, largely thrombosed liver section (Figure 1I-N). The neoplasm was well-demarcated and confined to the periphery by a thick, fibrous connective tissue capsule that separated the neoplasm from the adjacent necrotic area (Figure 1J-L). Neoplastic cells formed what appeared to be an irregular rosette structure with a central, dilated, vascular channel surrounded by several cell layers of radially arranged, small, intensely basophilic, elongated cells (Figure 1J-L). Mitotic figures were frequently observed. On IHC, neoplastic cells were negative for both CD31 (Figure 1M) and CK18 (Figure 1N), the negative results being typical of hepatoblastoma, underscoring the major challenge of diagnosing hepatoblastoma with IHC.
Cases 3 and 4, unbeknownst to the audience, were two different liver tumors from the same animal: a hepatocellular carcinoma (Figure 1O-Q, 1U-X) and hemangiosarcoma (Figure 1O-T, Y and Z) with metastasis to the hepatocellular carcinoma section (Figure 1R and X). The hemangiosarcoma presented for Case 3, featured yet another thrombosed liver section containing a poorly demarcated, unencapsulated, neoplasm within the periphery of the tissue that was composed of haphazardly arranged vascular channels (Figure 1S-T). Neoplastic cells were pleomorphic endothelial cells similar in appearance to those presented in Case 1, and stained positive with the endothelial cell marker, CD31 (Figure 1Y); neoplastic endothelial cells were negative for CK18, but as in Case 1, remaining hepatocytes were intensely positive for this hepatocellular marker (Figure 1Z).
Case 4 was a beautiful example of hepatocellular carcinoma in the mouse and featured a liver section diffusely effaced and replaced by an infiltrative neoplasm composed of pleomorphic hepatocytes arranged in thick cords and trabeculae and containing large areas of hemorrhage and necrosis (Figure 1O-Q). Neoplastic cells exhibited a high mitotic index, hyperchromatic nuclei and marked anisocytosis/anisokaryosis (Figure 1Q). There was also a distinct focus of haphazardly arranged vascular channels coursing through the hepatocellular neoplasm and disrupting the continuity of the trabecular pattern within the carcinoma (Figure 1R). The diagnosis of hepatocellular carcinoma was further confirmed with negative CD31 (Figure 1U) and positive CK18 (Figure 1W) IHC staining. However, the focus of haphazardly arranged vascular channels within the hepatocellular carcinoma stained positive for CD31 (Figure 1V) and negative for CK18 (Figure 1X), confirming Dr Quist’s suspicions that this was a metastatic hemangiosarcoma that likely originated from the hemangiosarcoma presented as Case 3. After voting for Case 4 was completed, Dr Quist revealed that the reason why she believed that this focus was likely to be a metastatic hemangiosarcoma within the hepatocellular carcinoma was because she knew that Cases 3 and 4 were from the same animal, which she had deliberately not shared prior to voting.
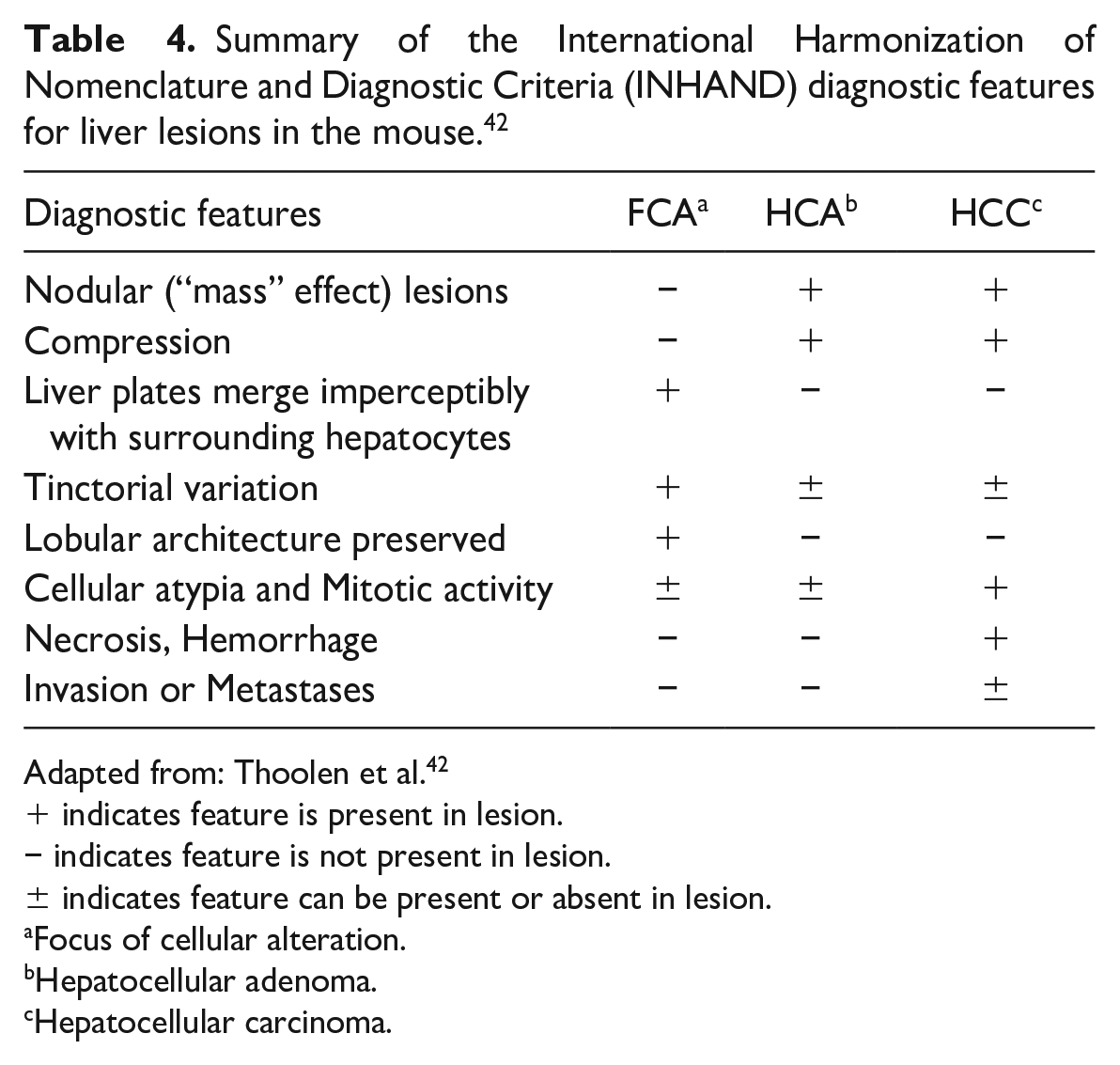
For the discussion, Dr Quist reviewed key diagnostic features of liver tumors in the mouse as summarized in the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) guidance document (Table 2). 42 Dr Quist further summarized diagnostic take home points by emphasizing the importance of recognizing growth pattern and behavior when differentiating hepatic neoplasms in the mouse. As demonstrated in the case examples, growth patterns of hemangioma/hemangiosarcoma are characterized by irregular vascular channels, hepatocellular carcinoma typically exhibits a growth pattern of thickened trabeculae, and hepatoblastoma is a neoplasm comprised of small, intensely basophilic and elongated cells radially arranged around a central vascular channel, forming rosettes, trabeculae, or pseudo-glandular structures. As for behavior, the location and appearance of metastasis can also be helpful in differentiating liver tumors in the mouse. Hepatic hemangiosarcomas readily metastasize to other parts of the liver or spleen and are typically well differentiated. Hepatocellular carcinomas and hepatoblastomas also frequently metastasize but most often to the lung and are poorly differentiated.
Key diagnostic features of liver neoplasms in the mouse. 42

Dr Quist also revisited the usefulness of IHC as a diagnostic tool for liver tumors in the mouse. For vascular neoplasms like hemangioma and hemangiosarcoma, CD31 and Factor 8 are helpful endothelial cell markers. For the liver, CK18 and CK19 are often used to confirm hepatocellular origin. However, hepatoblastoma is very elusive when it comes to IHC and does not stain positive with any known hepatocellular markers. A guidance document published by the American College of Pathologists 7 suggests using integrase interactor I (INI1), Glypican-3, and beta-catenin for hepatoblastoma but, as one audience member, and IHC expert, confirmed during the audience discussion, there are currently no reliable IHC markers for hepatoblastoma.
A Puzzling Primate Pulmonary Problem: Coccidioidomycosis in Two Rhesus Macaques
Dr Ronnie Chamanza (Janssen Pharmaceutical Companies of Johnson & Johnson, High Wycombe, UK) presented an interesting case of bronchopneumonia in two rhesus macaques (
The cases featured lung lesions from two outdoor-reared female animals that were 4 years of age at study start. The animals had been procured from the National Primate Center in Texas and were now part of an experimental severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) model development study for vaccine research, conducted at an Animal Biosafety Level 3 (ABSL-3) facility in Maryland. Standard quarantine policies were followed to ensure good health, and the animals were monitored for their health status prior to study start. On the first day of the study, both animals were inoculated with a SARS-CoV-2 virus strain by combination routes of intranasal (1 mL) and intratracheal (1 mL). Each animal received a total dose of 1.2 × 108 viral particles of the Wuhan SARS-CoV-2 strain (SARS-CoV-2 USA-WA1/2020 stock). Animals were monitored for clinical signs, including measuring body weight and temperature, and necropsy was scheduled at 14 days postinfection (dpi).
Dr Chamanza explained that other than some transient labored breathing and poor stool consistency recorded in one of the two animals, which went back to normal after a few days, no remarkable clinical findings were observed, and no expected SARS-CoV-2-related symptoms were observed in any of the inoculated animals on the study. Subsequently, all animals were euthanized as scheduled, 14 dpi, and the necropsy was performed according to the prevailing ABSL-3 practices and respiratory protection protocols. Only limited tissues, lungs, heart, liver, and kidneys, were collected from each animal. Dr Chamanza briefly outlined some of the practical challenges encountered by the necropsy team, which are associated with performing a necropsy in animals infected with highly contagious pathogens in an ABSL-3 facility. The limited access to animal rooms (in terms of both time and personnel) imposed for biosafety reasons, and the mandatory need to minimize creation of aerosols or splatters during the necropsy, precluded the necropsy team from conducting a more thorough macroscopic examination (e.g., cutting through lesions), or sampling more tissues. Nevertheless, the limited macroscopic examination of the lung showed consolidation and multifocal to coalescing areas of pale discoloration. For histology processing, 8 lung sections were obtained from the 6 to 7 lung lobes normally present in rhesus macaques, and the tissues were fixed, embedded, and processed routinely for histopathology evaluation.
Before presenting the histological images, Dr Chamanza informed the audience that the first set of slides was designed to help establish a morphological diagnosis, while the second set of slides would ask for the etiological diagnosis. For the morphological diagnosis, H & E stained lung sections taken at low (Figure 2A), medium (Figure 2B), and high (Figure 2C and D) magnifications were presented. The common feature in the images was multifocal to coalescing areas of mixed inflammatory cell infiltrates composed of varying populations of degenerate neutrophils, epithelioid macrophages, prominent multinucleated giant cells (MNGC), and fewer lymphocytes and plasma cells (Figure 2B-D). The lung architecture was in most cases completely obscured, and the bronchioles and alveoli were often dilated and filled with inflammatory cells, cellular debris, or edema, while moderate interstitial fibrosis was also present. Voting choices for round 1, with voting results were as follows: granulomatous pneumonia (68%), pyogranulomatous pneumonia (29%), interstitial pneumonia (1%), other (1%), and suppurative bronchopneumonia (0%).
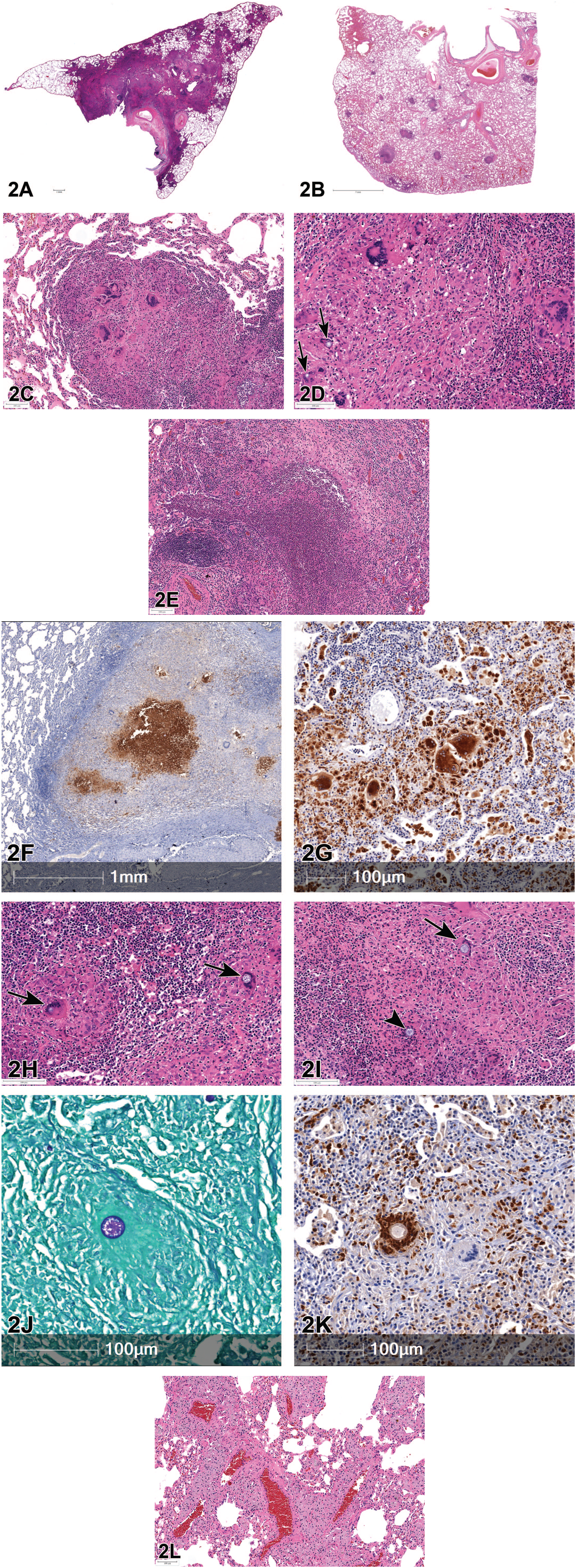
Lung from a rhesus macaque,
After presenting additional photomicrographs comprised of, high-magnification images showing an abundance of polymorphonuclear cells within a bronchus (Figure 2E), an IHC image of the same section showing positive myeloperoxidase (MPO) staining for neutrophils (Figure 2F), an H&E image showing coalescing granulomatous inflammation with abundant MNGCs (Figure 2D), and a corresponding IHC image showing positive staining for CD68 for macrophages (Figure 2G), the audience was asked to vote again. Voting choices for round 2, with voting results were as follows: pyogranulomatous pneumonia (92%), granulomatous pneumonia (8%), interstitial pneumonia (0%), other (0%), suppurative bronchopneumonia (0%). Dr Chamanza concurred with the audience that pyogranulomatous pneumonia may be considered the most appropriate diagnosis, but also recognized that the previously preferred diagnosis by the audience during round 1 of voting, “granulomatous pneumonia” is a valid alternative, and might be preferred by others.
Dr Chamanza then briefly discussed the possible causes for this lesion, and listed the differential diagnoses, acknowledging that this list was not exhaustive because several bacterial (eg,
For the etiological diagnosis, representative H & E sections obtained at high magnification (Figure 2H and I) were presented. These images showed that there were round, nonbudding, fungal yeast structures (spherules) of various sizes and stages of maturity, ranging from 15 to 50 μm in size scattered throughout the pyogranulomatous inflammatory lesions. The yeast structures (spherules) were surrounded by an unstained double wall, and the larger mature forms contained numerous 2- to 5-μm basophilic internal structures (endospores). Degenerate or ruptured spherules were frequently observed to be surrounded by MNGC or contained within the macrophage cytoplasm (phagocytosed), while smaller and more immature intact spherules were more likely to be found among cellular debris or surrounded by granulocytic infiltrates (Figure 2H and I). Voting choices for the cause and results of round 3 of voting were as follows: coccidioidomycosis (70%),
Before the next round of voting, additional IHC and histochemical stains (courtesy of Dr Martinot) were presented to reach a more definitive diagnosis, and a tabulated summary of all evaluations performed was shown. The additional sections presented included, negative IHC stain for SARS-CoV-2 nucleocapsid protein, a periodic acid-Schiff (PAS) stain (Figure 2J) and a negative acid-fast stain for mycobacterium. The PAS stain demonstrated the selective magenta staining of the cell walls (due to high polysaccharide moieties) and the granule-containing endospores (Figure 2J). An additional IHC stain for MPO demonstrated the nonbudding nature of an intact spherule surrounded by neutrophils (Figure 2K). Before the final voting was initiated, Dr Chamanza reminded the audience that while the study was conducted at an ABSL-3 facility on the east coast of the United States, the animals were obtained from a primate center in Texas, where they had been housed outdoors, emphasizing that the place of origin and husbandry might help in reaching the final diagnosis. Voting choices and results of round 4 of voting were as follows: coccidioidomycosis (97%),
Since most of the audience (97%) agreed with the diagnosis of coccidioidomycosis, Dr Chamanza proceeded to provide brief background information on the etiologic agents, epidemiology, pathogenesis, and the factors influencing the occurrence of coccidioidomycosis in laboratory NHP and humans. He reiterated the significance of geographical origin in the diagnosis of the disease, including in the identification of the specific species. Coccidioidomycosis (also known as Valley fever, California fever, or San Joaquin Valley fever) is a fungal disease that affects many species of mammals, including dogs, laboratory NHP, and humans, and occurs only in the western hemisphere.19,24 It is endemic in the semiarid parts of the southwestern United States known as the “Lower Sonoran Life Zone” (southern parts of Texas, Arizona, New Mexico, and central and southern California), and in certain semiarid regions of Mexico and Central and South America, where the environment is conducive to the survival of the fungus in the soil.11,19,24 The disease is caused by two nearly identical soil-borne, dimorphic fungal species,
The mycelial phase (saprophytic form), which is composed of hyphae and the infective arthroconidia, grows in the soil, and inhalation of the arthroconidia from disturbed soil is the typical mode of infection.19,24 Once inhaled, the arthroconidia transforms to yeast spherules (parasitic form) within infected tissue, which enlarge and form endospores. 24 In immune-competent animals and humans, the resultant disease is often clinically inapparent (although coughing and dyspnea may be experienced) and hence, coccidioidomycosis in laboratory NHPs is generally diagnosed at postmortem.24,25 However, unlike in humans, skin or subcutaneous lesions are rarely observed in NHPs, and the disease is primarily respiratory or pulmonary with limited widespread dissemination.24,25 The most commonly observed gross lesions in the lungs are multifocal to coalescing areas of consolidation affecting several or all lobes, which may exude purulent material on cut surface in the most affected regions. 25 On microscopy, a pyogranulomatous bronchopneumonia characterized by mixed inflammatory infiltrates, including neutrophils, epithelioid macrophages, MNGC, and lymphocytes and plasma cells, is often observed. It is commonly understood that early fungal phases (arthroconidia and newly formed endospores) are associated with a more neutrophilic inflammation, while the tissue phase (mature or endosporulated spherules) is usually associated with granulomatous inflammation. 39 Since infection is primarily by inhalation of arthroconidia, and the spherule is generally not considered to be infectious, animal-to-animal or animal-to-human transmission does not occur, although direct inoculation of arthroconidia into skin, bone, or other tissues by penetrating objects might cause infection. 35 Laboratory-acquired infections have reportedly occurred either through parenteral inoculation with tissues or cultures containing yeast forms, or inhalation of spores that have developed (sporulated) in infected materials.3,35,43
In the final segment of the presentation, Dr Chamanza reviewed the diagnosis and differential diagnoses of coccidioidomycosis. The diagnosis of coccidioidomycosis is usually made based on visualization of the organism in routine histologic sections supported by the use of histochemical stains such as the PAS reaction (Figure 2J), consideration of the animal’s epidemiological history (geographical origin and possible exposure to contaminated soil), and the application of other confirmatory tests such as serology, tissue culture, species-specific IHC, in situ hybridization, or polymerase chain reaction.24,25 No confirmatory tests were carried out in this case, but a diagnosis of coccidioidomycosis was made based on the identification of characteristic spherules on PAS and Grocott’s methenamine silver stains, and the geographical origin of the animals. The nonbudding morphology of the spherule differentiates it from that of
Dr Chamanza concluded by stressing that diagnosis of pulmonary lesions in NHP can be complicated by the epidemiological history of the animal (age, source, and origin), husbandry and housing conditions (outdoor vs barrier-reared), experimental procedures, or health status. He reiterated the importance of the epidemiological history or geographical origin by illustrating how knowledge of the origin of the animals in this study (Texas) played a key role in formulating a diagnosis. Consideration of the epidemiological history of animals should include their present geographical location at the test facility and its local ecosystem, the geographical place of origin or breeding facility, the supplier, and any other holding facility used while the animals were in transit.
Mouse Liver Lollapalooza: Neoplastic and Nonneoplastic Lesions in the Mouse Liver
Dr Allison Boone (Experimental Pathology Laboratories [EPL], Inc., Durham, North Carolina) presented 3 cases of liver lesions observed in B6C3F1/N mice. Cases were derived from two, 2-year toxicology/carcinogenesis bioassays conducted by the DTT/NIEHS. The SPs for the bioassays were Drs. Abraham Nyska and the late J.C. Peckham. Dr Boone would like to thank Drs. Arun Pandiri and Mark Cesta at NTP/NIEHS, as well as Michael Carden, Leslie Couch, Emily Singletary, and Lorri Ezedin at EPL, Inc. for their assistance and support with the presentation.
Attendees were shown a series of both low- and high-magnification photomicrographs of the liver lesions for all 3 cases (Figure 3). The diagnostic choices were similar for all cases, and Table 3 includes the voting choices and results.
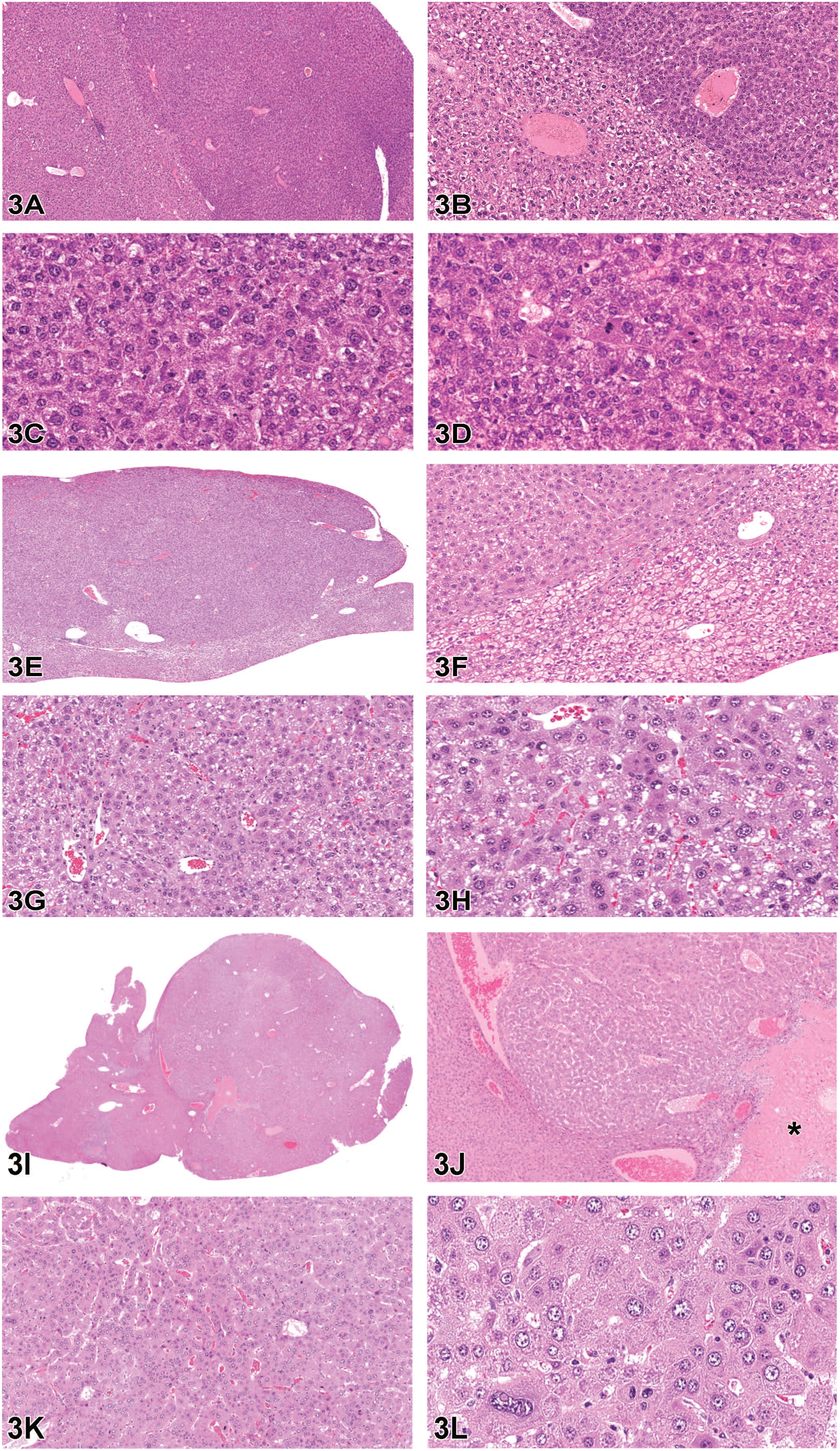
Mouse liver lesions, B6C3F1/N mouse. (A-D) Focus of cellular alteration. (A) The liver contains a well-demarcated basophilic focus. HE. (B) On higher magnification of the junction between the focus and normal liver, hepatocytes are observed to merge imperceptibly with adjacent hepatocytes. HE. (C) The focus is composed of hepatocytes that exhibit minimal cellular atypia. HE. (D) Mitotic activity is captured in the focus. HE. (E-H) Hepatocellular adenoma. (E) The liver contains a well-demarcated mass. HE. (F) On higher magnification of the junction between the mass and normal liver, the mass is observed compressing the adjacent hepatocytes. HE. (G) In the mass, lobular architecture is not preserved. HE. (H) Neoplastic hepatocytes exhibit mild to moderate cellular atypia. HE.
Voting results for mouse liver lesions, cases 1 to 3.
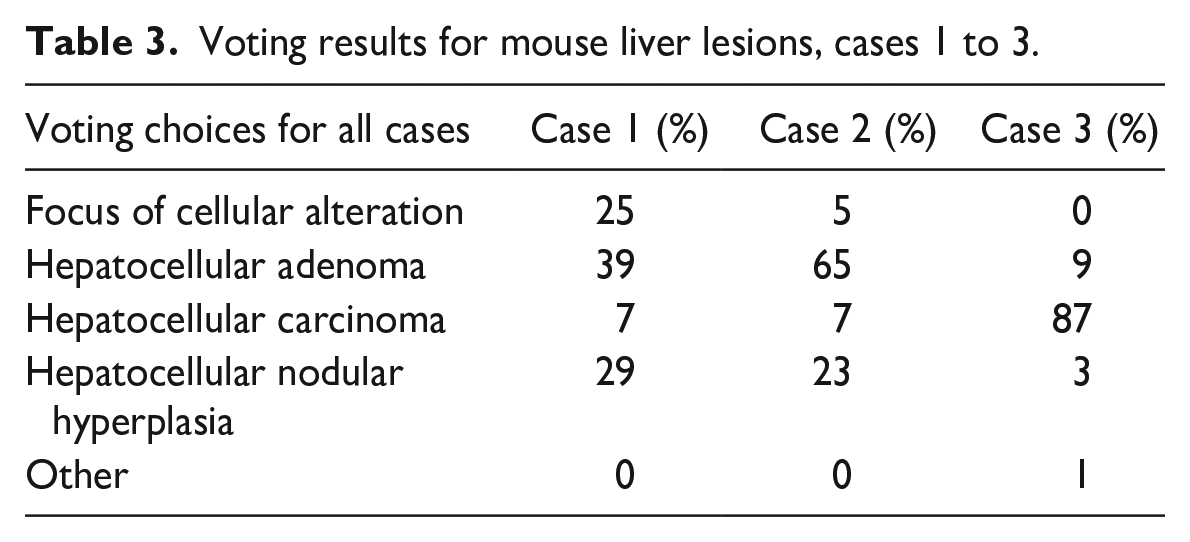
Case 1 comprised photomicrographs from a focus of cellular alteration (Figure 3A-D). The example consisted of a well-demarcated basophilic focus with hepatic cords that merged imperceptibly with adjacent hepatocytes (Figure 3A and B). In addition, the focus exhibited minimal cellular atypia (Figure 3C) and low mitotic activity was observed (Figure 3D); compression/invasion of the hepatic parenchyma, necrosis, and hemorrhage was not observed. Only 25% of attendees selected the diagnosis of focus of cellular alteration, and 29% and 39% of attendees chose hepatocellular nodular hyperplasia and hepatocellular adenoma, respectively, as their voting choices. These results demonstrate the challenge in differentiating neoplastic and nonneoplastic hepatic lesions.
For cases 2 and 3, Dr Boone presented photomicrographs from a hepatocellular adenoma (Figure 3E-H) and hepatocellular carcinoma (Figure 3I-L), respectively. The hepatocellular adenoma consisted of a well-demarcated mass (Figure 3E) that compressed adjacent hepatic cords (Figure 3F). The mass lacked retention of lobular architecture (Figure 3G) and mild-to-moderate cellular atypia was observed in the neoplastic population (Figure 3H). The hepatocellular carcinoma replaced greater than 50% of the liver section (Figure 3I), compressed adjacent hepatic cords, and contained a small area of necrosis (Figure 3J). Neoplastic hepatic plates were up to 6 cells thick, lacked normal lobular architecture (Figure 3K), and exhibited marked cellular atypia (Figure 3L). The most prevalent voting results for cases 2 and 3 agreed with the NTP diagnoses of hepatocellular adenoma and carcinoma, respectively.
Following voting, Dr Boone reviewed the features specific for each diagnosis with a collage of the photomicrographs as seen in Figure 3 and a summary of the diagnostic features specific to each lesion based on the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) guidance document (Table 4). 42
Summary of the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) diagnostic features for liver lesions in the mouse. 42
Adapted from: Thoolen et al. 42
indicates feature is present in lesion.
indicates feature is not present in lesion.
indicates feature can be present or absent in lesion.
Focus of cellular alteration.
Hepatocellular adenoma.
Hepatocellular carcinoma.
During the beginning of the discussion, Dr Boone reminded attendees that foci of alteration and hepatocellular tumors can occur spontaneously or secondary to chemical exposure with differential species/strain susceptibility. For the remaining component of the discussion, Dr Boone focused on the significance of the foci of cellular alteration in rats and mice. She mentioned how all foci may not be viewed as a preneoplastic change16,17,34,40; however, there are reports that have associated the foci as precursors to neoplasia.2,15 Given this dilemma, Dr Boone mentioned ways to further characterize the relevance of the foci in rodent studies for hazard assessment, which included reversibility of the lesion after cessation of exposure during a recovery period, consideration of enzyme markers of preneoplasia (e.g., gamma-glutamyl transpeptidase [GGT]), 41 and the mode of action of the associated toxicant. Furthermore, she mentioned newer technological advances that may enable further characterization of the foci including spatial omics, single cell sequencing, and error-corrected duplex sequencing.
Dr Boone concluded the presentation with an update on the current activities in the DTT, Comparative and Molecular Pathogenesis Branch, molecular pathology group (MPG), which may assist with further characterization of the foci. She mentioned that the MPG is currently embarking on a project that involves whole genome sequencing of rodent tumors/nontumors resulting from various exposures 36 and generating a database of mutation signatures and cancer driver genes. The studies will not only assist in the development of targeted sequencing panels to investigate preneoplasia but will also aid in the comparison of human and rodent cancers induced by similar exposures and thus establish translational relevance with human cancers.
A Mononuclear Mystery
Dr Gregory A. Krane (Moderna, Cambridge, Massachusetts) presented three studies with the goal of suggesting appropriate terminology for accumulations of round cells. All of the studies were for development of lipid nanoparticle-encapsulated (LNP), messenger RNA (mRNA) therapeutics, although the mRNA product and specific indication for each study were de-identified for the purposes of the symposium. For each study, Dr Krane provided relevant background information, presented a series of photomicrographs, and then queried the audience with a series of voting choices, followed by explanation of the diagnosis chosen by Moderna’s pathology team. Dr Krane would like to acknowledge Franny Ambrose and Dr Annie Golding of Moderna, who performed the IHC work described in this manuscript, and Dr Youmna Kfoury of Moderna, who provided the gross photographs of the spleen for study three (Figure 4S-T). Dr Krane also thanks Beth Mahler of EPL for support with figures, Dr Martha Hensel of the MD Anderson Cancer Center for her peer-review of the manuscript, and Dr Erin Quist of CRL and the pathology team at Moderna (especially his manager Dr Eric Jacquinet) for their feedback with regards to the development of this presentation and manuscript.
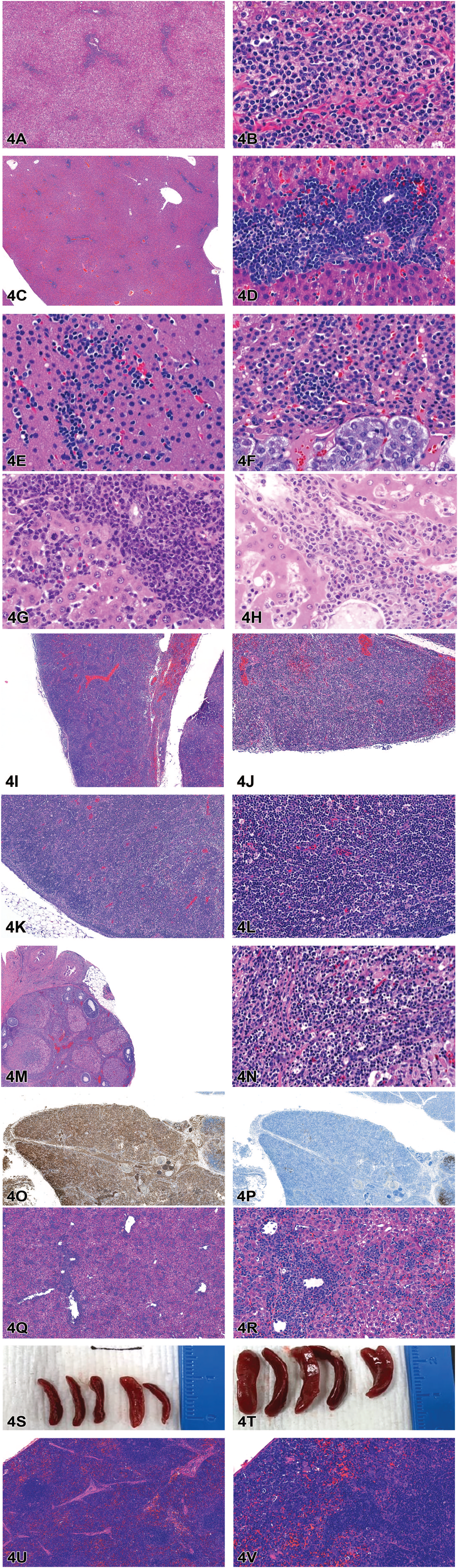
(A) Liver, NHP, study one, HE. A low magnification image of the liver demonstrates a multifocal infiltration of round cells in these rats treated with an LNP-encapsulated mRNA-encoding cytokine intended for oncology indications. (B) Liver, NHP, study one, HE. Higher magnification of the liver reveals this monomorphic population of cells to expand periportal regions and sinusoids. They have a round cell morphology but are not consistent with any particular mature leukocyte lineage. They were therefore originally diagnosed as “infiltration, myeloid cell” due to their lack of obvious differentiation and resemblance to myeloid precursors in normal bone marrow. Follow-up immunohistochemistry (not shown) characterized these cells predominantly as cytotoxic T lymphocytes, at which point the diagnosis was amended to “infiltration, lymphoid.” (C and D) Liver, rat, study one, HE. Findings in the rat liver are similar to those shown for NHP. (E and F) Adrenal gland, rat, study one, HE. Myeloid infiltration was also present in the adrenal gland in the cortex (E) and at the cortico-medullary junction (F).
Study one featured two dose range finding studies for a cytokine intended for oncology indications, with one study being performed in 2.5- to 3-year-old male and female cynomolgus monkeys (NHP), and the other study being performed in 9- to 10-week-old male and female Sprague Dawley rats. Animals were administered the test article once weekly for 3 weeks followed by terminal necropsy 24 hours after the final administration. Dr Krane showed photomicrographs of histologic sections from NHP liver (Figure 4A and B), rat liver (Figure 4C and D), rat adrenal gland (Figure 4E and F), and rat and NHP livers from a prior study with a similar test article (Figure 4G and H).
All images shared a common finding of a monomorphic population of densely packed, medium-sized round cells with a prominent nucleus with open chromatin and small amounts of cytoplasm. This expansile population of cells compressed and sometimes infiltrated and replaced the surrounding parenchyma. In the liver, although predominantly in portal and peri-portal areas, these cells were also randomly present amongst hepatocytes and infiltrated sinusoids, were increased in number in the vasculature, and were associated with single cell necrosis of hepatocytes. In the rat, this finding was diagnosed in the liver, adrenal gland, pancreas, lung, epididymis, and Harderian Gland, and in the NHP in the liver, adrenal gland, pancreas, thyroid gland, uterus, and choroid plexus of the brain (although images from each affected organ were not presented to the audience, the audience was shown a list of affected organs). Relevant findings shared with the audience (without presented photomicrographs) were increased myeloid cellularity in the bone marrow of the rat, a shift of myeloid precursors toward early stages of proliferative cells (bone marrow cytology) in the rat, increased macrophage cellularity in the red pulp and increased lymphocyte cellularity in the white pulp of the rat spleen, and increased macrophage cellularity of lymph nodes in the rat and NHP.
Voting choices for Study one, with voting results were as follows: infiltration, lymphocytic (6%), increased cellularity, lymphocytic (1%), infiltration, mononuclear cells (59%), increased cellularity, mononuclear cells (13%), inflammation, mononuclear cells (4%), infiltration, mixed cells (9%), inflammation, mixed cells (0%), lymphoma (0%), and infiltration, myeloid (9%). The diagnosis chosen by Moderna’s pathology team was infiltration, myeloid. The Moderna pathology team found themselves in a difficult situation for this study, as there was not an INHAND or SEND diagnosis compatible with the features of this finding. This monomorphic population of densely packed round cells exhibited a leukocyte morphology, but it was not consistent with any particular mature leukocyte lineage. The morphology and biologically aggressive behavior differed from mononuclear or mixed cell infiltration as described in INHAND guidelines. The finding was not inflammatory in nature, as there were no tissue responses consistent with inflammation, nor did it efface tissue architecture to the point of being able to justify it as a neoplasm. The team felt this was an infiltration (versus a diagnosis of increased cellularity), as the cells were in aggregates and not throughout an entire tissue compartment. The team was concerned that the use of the term “infiltration, mononuclear” would not capture the aggressive behavior of the cells infiltrating, compressing, and remodeling architecture of the host tissue parenchyma. Given the resemblance of the cells to myeloid precursors in bone marrow, combined with the increased cellularity of cells with myeloid lineage in some hematolymphoid tissues and the presence of histiocytic cells in this infiltrate in a prior study, the Moderna pathology team carried forward the diagnosis of “infiltration, myeloid” that had been used in the prior study.
Given the team’s concern for neoplastic potential of this myeloid infiltration exhibiting biologically aggressive behavior, the investigative pathology group followed with IHC to characterize the cell type and better evaluate risk. Dr Krane presented IHC images from NHP liver for CD3, CD8, NKG2α, and CD20, which immunolabel T cells, cytotoxic T cells, NK cells, and B cells, respectively, and rat liver for CD3 and CD8. These images demonstrated the cellular infiltrate to predominantly immunolabel for CD3 and CD8, which characterized them as a population of cytotoxic T cells. In the NHP, there were fewer cells immunolabeled for NKG2α (NK cells) and CD20 (B cells). Dr Krane presented quantitative image analysis data that reinforced the interpretations of the IHC. Given the characterization of these infiltrating cells as predominantly cytotoxic T lymphocytes, the finding was amended to “infiltration, lymphoid,” as lymphocytes are not derived from the myeloid lineage. 33 Although the interpretation of the cells’ behavior was unchanged, their characterization was useful in interpreting the finding to be a result of exaggerated pharmacology, with stimulation of both lymphocytes in peripheral tissues and myeloid cells in bone marrow and hematolymphoid tissues in the same mechanism as the intended pharmacology, but to an amplified extent as compared to the desired clinical effect. This was influential in the decision to use lower doses for follow-up GLP studies. In summarizing this study, Dr Krane mentioned that it is important to use INHAND and SEND terms whenever possible, but this example represents a unique circumstance where pathologists must use their scientific judgment to create a new term (while justifying the terminology in their narrative) that best captures the features of the finding.
Study two showed findings from a 9- to 10-week-old female Sprague Dawley rat in a dose range finding study with an unidentified test article. This rat was found dead on study with no prior clinical observations, and the only gross finding at necropsy was bilaterally enlarged iliac lymph nodes. Dr Krane showed photomicrographs of histologic sections of the thymus (Figure 4I and J), lymph node (Figure 4K and L), and ovary (Figure 4M and N), with corresponding images from a control animal on the same study for comparison. In all of the images shown, there are sheets of monomorphic, small to medium round cells with hyperchromatic nuclei with small amounts of cytoplasm that variably effaced and replaced pre-existing architecture of the tissues. Large aggregates of similarly appearing lymphocytes were also present in the vasculature of various tissues. Other organs affected by this finding, but not shown in the presentation, were the lung, kidney, uterus, cervix, and vagina.
Voting choices for Study two, with voting results were as follows: infiltration, lymphocytic (14%), increased cellularity, lymphocytic (24%), infiltration, mononuclear cells (20%), increased cellularity, mononuclear cells (16%), inflammation, mononuclear cells (0%), infiltration, mixed cells (0%), inflammation, mixed cells (2%), and lymphoma (24%). Owing to the multiorgan effacement of pre-existing architecture by a monomorphic round cell population, the correct diagnosis was lymphoma. After votes were cast, Dr Krane showed photomicrographs demonstrating IHC for CD3 (diffuse immunolabeling of neoplastic cells, T cell marker, Figure 4O) and CD20 (no immunolabeling of neoplastic cells, B cell marker, Figure 4P), further justifying the diagnosis of lymphoma plus additionally characterizing it as a malignancy of T cells.
Study three showed findings from 10-week-old, female C57Bl6 mice that were intravenously administered a test article intended to be part of a bone marrow transplantation preparation regimen three times during 1 week immediately followed by terminal necropsy. Relevant findings were increased liver enzymes, grossly enlarged spleens at necropsy, and increased spleen weights. Only the liver and spleen were collected at necropsy for histologic examination by the principal investigator. Histopathology from the liver and spleen shown in the presentation demonstrated in both tissues a population of monomorphic, medium to large round cells with prominent nuclei and variable amounts of cytoplasm, which sometimes were multinucleated or exhibited mitoses. In the liver (Figure 4Q and R), this population of cells filled and expanded hepatic sinusoids and compressed the adjacent parenchyma, with occasional concurrent hepatocellular degeneration/necrosis in these neighboring regions. In the spleen (Figure 4S-V), this population of cells filled the red pulp and obscured but did not efface background architecture. This population of cells correlated with the grossly enlarged spleens and increased spleen weights.
Voting choices for study three, with voting results were as follows: infiltration, lymphocytic (4%), increased cellularity, lymphocytic (9%), infiltration, mononuclear cells (13%), increased cellularity, mononuclear cells (7%), inflammation, mononuclear cells (7%), infiltration, mixed cells (4%), inflammation, mixed cells (2%), and lymphoma (55%). The diagnosis chosen by the Moderna pathology team for study three was increased cellularity, mononuclear. Dr Krane acknowledged that most of the audience favored the diagnosis of lymphoma, though the Moderna pathology team did not feel there was enough remodeling of tissue architecture by the cellular population to justify a diagnosis of neoplasia. However, the principal investigator of this study was cautioned that this diagnosis was made solely on the evaluation of the two tissues submitted. Should other tissues from the animals have been collected and made available for microscopic examination, depending on the extent of change in tissue architecture in other tissues, it is possible that the diagnosis for this study may have been lymphoma instead of increased cellularity, mononuclear.
Dr Krane discussed that evaluation of limited tissue sets is not an uncommon situation for pathologists, especially for discovery studies where they become involved after most of the project has already been completed. He discussed the role of pathologists in team science and encouraged attendees to educate principal investigators to involve pathologists early on in study design meetings to avoid scenarios where additional information could be helpful to optimize study conclusions and recommendations but is not possible to ascertain due to deficiencies in study design such as inadequate sample collection for clinical pathology, histopathology, organ weights, or other endpoints. An audience member personally discussed with Dr Krane that perhaps for these scenarios, it is more appropriate that a pathologist use the terminology of “impression” or “opinion” instead of “diagnosis,” as is the practice of some health care providers in diagnostic radiology or pathology settings, given that such language characterizes the limitations of the interpretation based on the available information.
The theme of this presentation was terminology of findings characterized by accumulations of round cells, with a focus on infiltration, increased cellularity, and round cell malignancy. Dr Krane provided a summary table (Table 5) comparing features of these findings, based on a combination of anecdotal experience and available literature. 44 Important points to consider when considering these diagnoses are distribution of the cells, percentage of the tissue affected, maintenance of tissue architecture, if the cells have heterogenous or monomorphic appearance, if the cells are in abnormal anatomic locations, the number of tissues affected, if concurrent inflammation is present, and the mitotic rate. Although one audience member postulated that infiltration and increased cellularity may be the same finding and should have a unified term, Dr Krane did feel that they appear distinct enough visually to be diagnosed as separate findings, even if the underlying mechanism causing the finding is the same. A representative from the INHAND oversight committee agreed with Dr Krane’s response in personal discussions. Primarily, distinctions between infiltration and increased cellularity are that infiltration tends to represent multifocal aggregates of cells, whereas increased cellularity tends to represent cells occupying an entire tissue compartment (and sometimes expanding that compartment). Although the percentage of tissue affected may be greater with increased cellularity as compared to infiltration, otherwise the criteria for these findings are quite similar. 44 However, round cell malignancies differ from both infiltration and increased cellularity, as they tend to affect larger amounts of tissue, efface, and/or replace pre-existing tissue architecture, are composed of monomorphic cell populations, occupy abnormal anatomic location, affect greater numbers of organs, may have secondary inflammation, and exhibit a higher mitotic rate. 44
A Comparison of Microscopic Features of Cellular Infiltration, Increased Cellularity, and Round Cell Malignant Tumors.
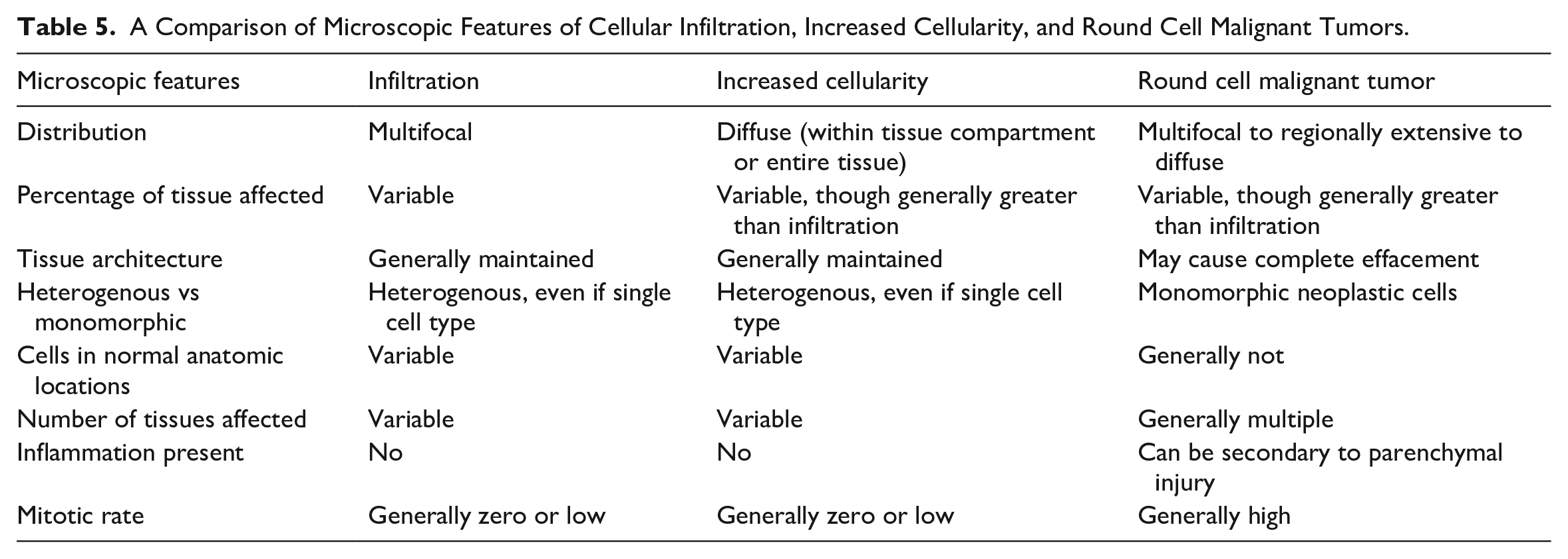
Ultimately, the primary take home message of Dr Krane’s presentation is that the diagnoses of infiltration, increased cellularity, round cell tumor, or inflammation must be carefully weighted, as they represent different histopathologic presentations, even if they may share some underlying mechanisms. Although utilization of SEND and INHAND compatible terminology is recommended whenever possible, sometimes it is necessary to create alternative scientifically justifiable terminology if there is not a SEND or INHAND finding that best conveys the appearance, mechanism, and/or implications of the finding. Identification of such safety findings can help drive partnerships with investigative studies that involve scientists in other disciplines that provide further context to such findings, and it is imperative that pathologists build relationships with such scientists to design studies that can provide the most impactful and useful data possible. Ultimately, training both as veterinarians and pathologists provides comparative biomedical science knowledge from the subcellular to the multi-animal level that can create opportunities to improve the value delivered by scientific collaborations with a variety of stakeholders who depend on us to provide safe, effective, and novel diagnostics and therapies to patients with unmet need.
Proliferative Pathology of NHPs
Dr Martha Hensel (University of Texas MD Anderson Cancer Center [MDACC], Bastrop, Texas) presented two interesting cases of spontaneous tumors from NHP colonies at the Michael E Keeling Center for Comparative Medicine and Research. The animals were experimentally naïve and part of an inhouse breeding program. Dr Carolyn Hodo (MDACC) was the primary pathologist on both cases and contributed to the presentation. The author would like to acknowledge Drs Stanton Gray and Stephanie Buchl for providing the clinical perspective for the cases.
The cases presented included colon from a rhesus macaque (
Case 1 featured colon from a 22-year-old, multiparous, female rhesus macaque (
A series of low- and high-magnification H&E-stained sections of the mass in the colon were presented to the audience for their diagnosis (Figure 5A-D). The low magnification image (Figure 5A) provided orientation of the lesion and highlighted the invasive nature of the proliferative glandular tissue that extended from the serosa to the submucosa. Higher magnification images of the region of interest in the muscular wall of the colon demonstrated that the lesion had degrees of morphological variation (Figure 5B). The predominant lesion was composed of characteristic invasive endometrial glands lined by cuboidal to tall columnar endometrial cells supported by stroma; glands were surrounded by and contained blood (Figure 5C). A second lesion was composed of cuboidal cells arranged in nests and tubules on a desmoplastic stroma (Figure 5D). Nuclei were irregularly round with finely stippled chromatin and 1 small nucleolus. Anisocytosis was moderate; no mitotic figures were noted in the section available for review.
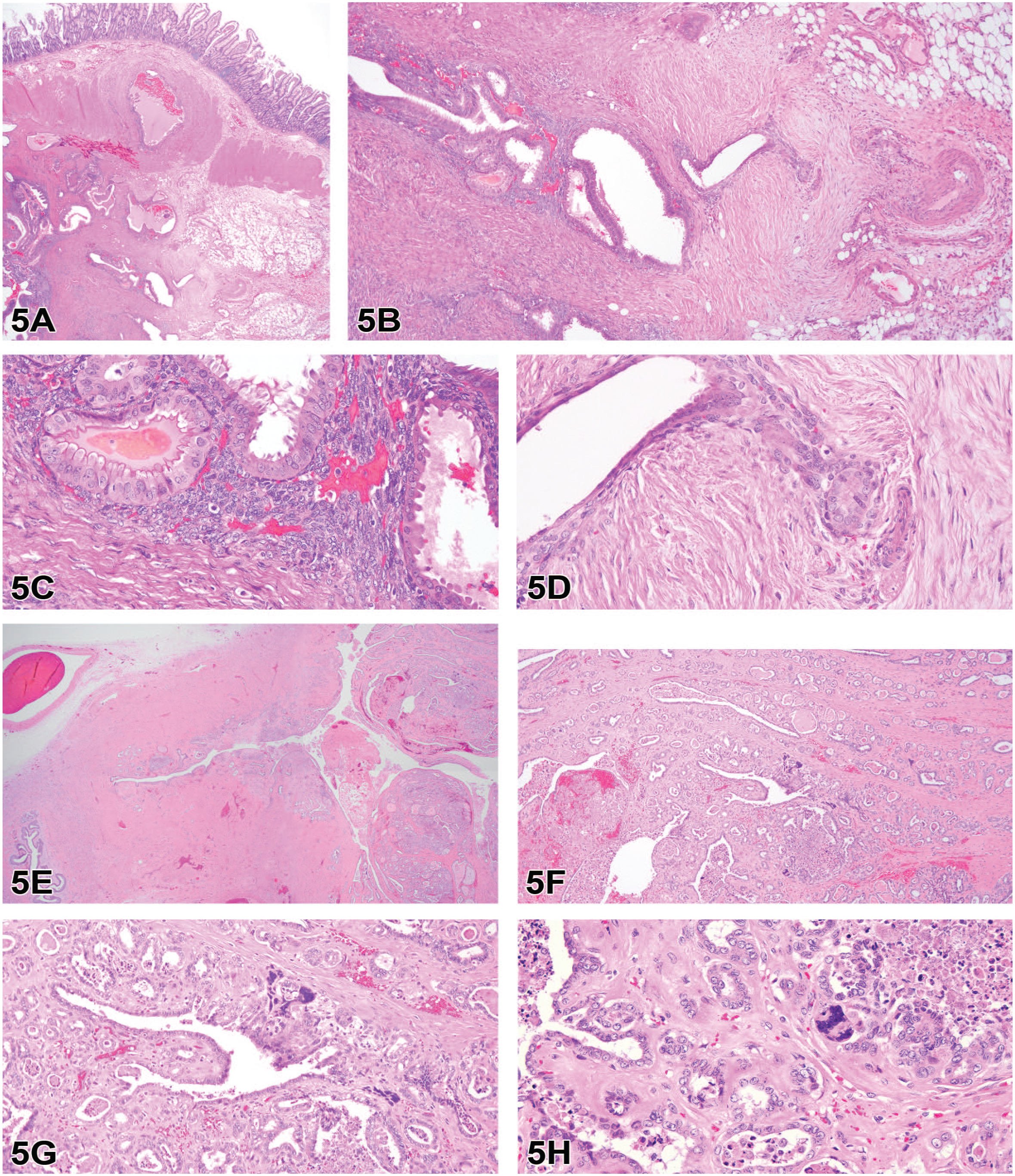
Proliferative lesions in a rhesus macaque (A-D) and owl monkey (E-H) from experimentally naïve diagnostic necropsy cases. (A) Low magnification of colon demonstrates the infiltrative nature of the glandular epithelium, which at higher magnification (B) demonstrated that the lesion had degrees of morphological variation. The predominant lesion (C) was composed of characteristic invasive endometrial glands lined by cuboidal to tall columnar endometrial cells supported by stroma; glands were surrounded by and contained hemorrhage. A second morphology was composed of cuboidal cells arranged in nests and tubules on a desmoplastic stroma (D). (E) Overview of the cervix with a proliferative, multinodular invasive mass that extends from the mucosa into the muscular layer. (F) Higher power magnification highlighting the invasive nature of irregular tubules on a moderate fibrovascular stroma with scattered areas of hemorrhage. (G) Most neoplastic cells were cuboidal to columnar with variably distinct cell borders, a small amount of eosinophilic cytoplasm, and central oval nuclei with coarsely stippled chromatin and one prominent basophilic nucleolus. Multifocally throughout the neoplasm was a second population of large multinucleated syncytial cells with hyperchromatic nuclei, which resembled syncytiotrophoblasts (G and H).
The audience was polled for their preferred diagnosis. Voting choices with voting results were as follows: endometrial carcinoma (10%), colon adenocarcinoma (29%), endometriosis (49%), retroperitoneal fibromatosis (11%), or other (1%). The audience concurred with the diagnosis preferred by the speaker: endometriosis. However, this case was selected because it was not considered a straightforward diagnosis. At necropsy, the featured section of colon was originally diagnosed as a colon carcinoma due to the gross appearance and supporting evidence of a known genetic predisposition to developing colorectal cancer plus a positive fecal occult blood test. Endometriosis was diagnosed based on histologic examination, but the second morphologic population with the scirrhous stroma supporting nests and tubules of cuboidal epithelium suggested an additional diagnosis of either colon carcinoma or endometrial carcinoma.
Endometriosis is a common reproductive tract lesion in aged female rhesus macaques with a prevalence approaching 30% in some populations.6,47 The incidence may increase in colonies with a high rate of Cesarean section deliveries as one of the proposed pathogeneses is explantation of endometrial tissue during surgery. 1 Endometriosis is also diagnosed in up to 10% of reproductive aged women, and rhesus macaques can serve as an animal model for the condition. Characteristic histologic features include glandular epithelium and stroma that mimic endometrium. The glands may be filled with blood and/or degenerate and viable macrophages. Hemorrhage may also extend into the stroma and be accompanied by macrophages with erythrocytes or hemosiderin pigment. The lesion can wax and wane with the estrous cycle and induce hemoabdomen if the cysts rupture.
Endometriosis is most common in the ovary, but ectopic endometrial tissue can be found throughout the abdomen including the colon. Endometriosis often appears as variably sized, brown, nodular lesions along the omentum, mesentery, or serosal surface of the organs and are known colloquially as “chocolate cysts.” Colon carcinomas may arise from the mucosa of the ascending, transverse, or descending colon as solitary or multifocal nodular masses with an accompanying scirrhous response that often causes constriction of the lumen. 23 A comparison of the typical appearance of colon carcinoma and endometriosis was presented to highlight the gross features of each condition.
As part of the signalment, the presenter offered the information that the animal was heterozygous for
Case 2 was tissue from a 4-year-old female owl monkey (
A series of H&E-stained sections of the cervix were presented to the audience for their diagnosis (Figure 5E-H). The low magnification image provided the anatomical orientation for the lesion as the location was not provided for the audience to increase the difficulty of the exercise (Figure 5E). However, Dr Hensel did point out the normal tissue on the left side of the image to help orient the viewers and offered the useful adage that if you are unsure of what you are looking at then you are probably in the reproductive tract. The second image highlighted the invasive nature of irregular tubules on a moderate fibrovascular stroma with scattered areas of hemorrhage (Figure 5F). Most neoplastic cells were cuboidal to columnar with variably distinct cell borders, a small amount of eosinophilic cytoplasm, and central oval nuclei with coarsely stippled chromatin and one prominent basophilic nucleolus (Figure 5G). Multifocally throughout the neoplasm was a second population of large multinucleated syncytial cells with hyperchromatic nuclei, which resembled syncytiotrophoblasts (Figure 5G-H). Neoplastic tubules often contained karyorrhectic and proteinaceous debris, and the stroma was multifocally hyalinized.
After the images were reviewed, the audience was polled for their diagnosis. Voting choices with voting results were as follows: urothelial carcinoma (15%), gestational choriocarcinoma (20%), cervical adenocarcinoma (20%), epithelioid trophoblastic tumor (42%), or other (3%). Perhaps reflecting the difficulty of this case or the uncertainty of the anatomical location, fewer respondents weighed in with their selections (60 responses for case 2 compared to 68 for case 1). Nonetheless, 42% of respondents selected epithelioid trophoblastic tumor, which was the diagnosis preferred by Dr Hensel.
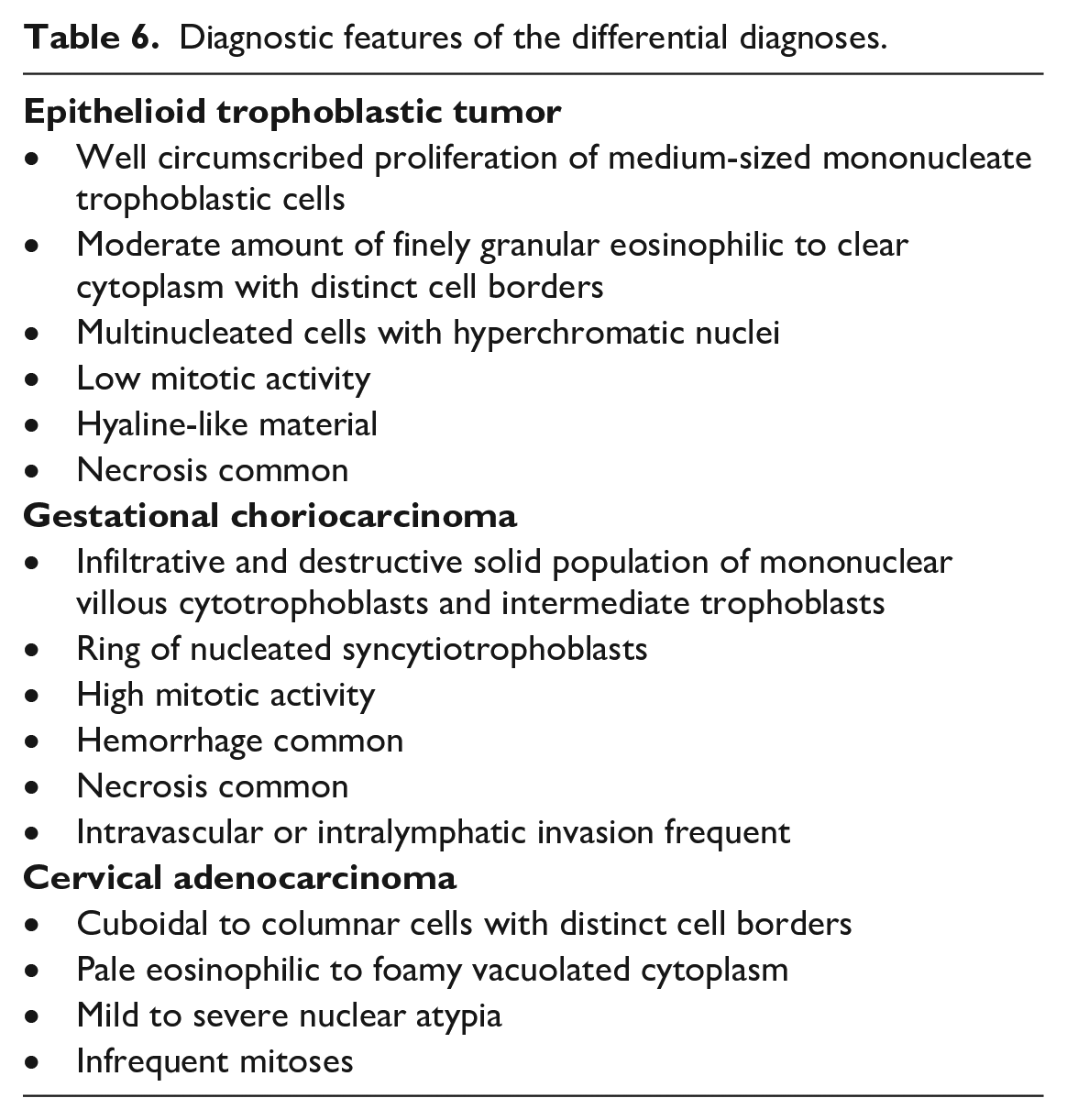
The differentials considered included: epithelioid trophoblastic tumor, gestational choriocarcinoma, or cervical adenocarcinoma. Diagnostic features of each differential are presented in Table 6. An epithelioid trophoblastic tumor was considered the most likely diagnosis due to the bland appearance of the cuboidal population of neoplastic cells and the presence of multinucleated cells with hyperchromatic nuclei. A gestational choriocarcinoma was considered less likely because these tumors reportedly have features of malignancy such as high mitotic rate, infiltrative and destructive growth pattern, large areas of necrosis, and intravascular invasion. None of these features were noted in the tumor presented. Cervical adenocarcinoma was ruled out due to the presence of the multinucleated cells, which are not a feature of this tumor. To confound diagnostic confidence, there is overlap between the trophoblastic tumor types, and epithelioid trophoblastic tumors may be part of a mixed trophoblastic tumor.
21
However, our diagnostic confidence was increased by the concurrence of our fellow pathologists at the meeting. This case was an interesting example of an epithelioid trophoblastic tumor, which is a rare diagnosis in NHPs; our literature search turned up a single case report in the ovary of a cynomolgus macaque (
Diagnostic features of the differential diagnoses.
Audience members questioned why the animal could not urinate following delivery. Compression of the urethra by the neoplastic tissues possibly exacerbated by swelling associated with labor prior to C-section is considered the most likely explanation for the inability to urinate. After the presentation was concluded, an audience member offered a hypothesis for why the clinical signs only appeared after parturition: perhaps the weight of the gravid uterus exerted enough tension to keep the urethra open despite the space occupying nature of the cervical mass. Once the tension was alleviated (i.e., the infant was delivered), the counterbalance was removed, and the mass effect was observed.
This Bites: Oral Tumor in a Rat
Dr Shawn Lennix (formerly of Labcorp Early Development Laboratories Inc., Madison, Wisconsin) presented a case featuring an oral mass in a rat from a 2-year toxicity/carcinogenesis study for which Dr Molly Boyle (Labcorp, Somerset, New Jersey) was the study pathologist. Dr Lennix thanked Drs. Victoria Laast, Melissa Behr, and Jennifer Dreyfus (Labcorp, Madison, WI) for their support; Steve Van Adestine and Jasmine Holland for providing photography and digital imaging assistance; and Susan Lynk and Nate Sendelbach for performing IHC (Labcorp, Madison, WI).
The case presentation featured a 20-week-old male, Wistar Han Rat (RccHan: WIST) control animal receiving deionized water on a 2-year toxicity/carcinogenesis study. On Day 92/Week 14 of the study, a 29 × 20 × 20 mm soft, dark red mass was noted on the mandible and the animal was euthanized for welfare reasons. At necropsy, the mandibular lymph nodes were grossly unremarkable. Upon histopathologic examination, the axillary and mesenteric lymph nodes had no microscopic findings.
Dr Lennix showed the audience images of the mass
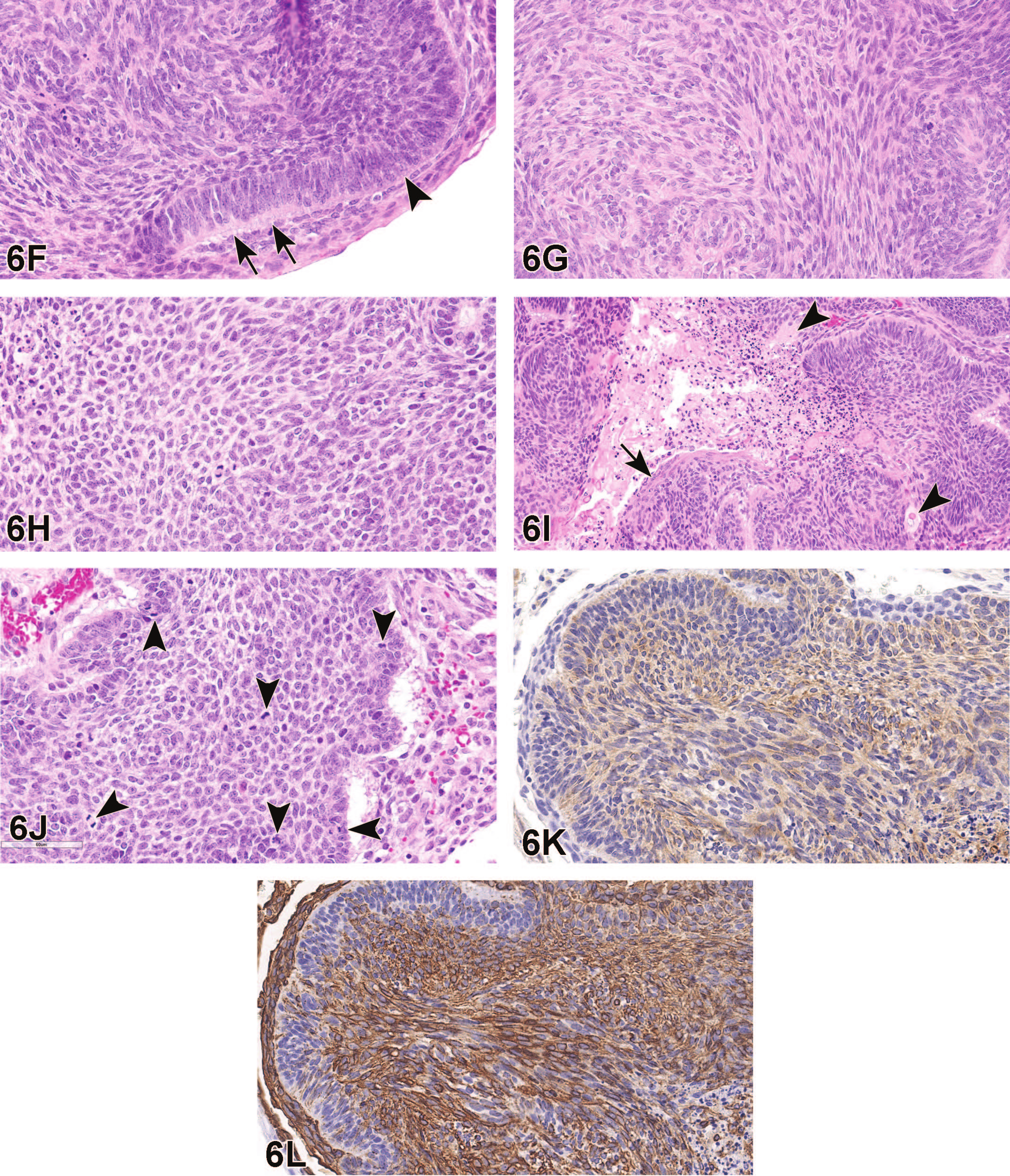
An oral tumor from a 20-week-old male Wistar Han Rat (RccHan: WIST) that was a control animal receiving deionized water on a 2-year toxicity/carcinogenesis study. A dark red to tan mass expands the tissues of the left mandibular region (A). The cut surface of the mass in the mid-sagittal plane reveals a multiloculated mass that partially replaces the mandible (B). A low magnification photomicrograph of hematoxylin and eosin (H&E) stained tissue depicts islands and trabeculae of epithelial cells and multiple cystic spaces (C). Two cysts lined by parakeratinized stratified squamous epithelium contain degenerate epithelial cells, erythrocytes, hemosiderophages, and neutrophils (D). Neoplastic cells are arranged in highly cellular aggregates that have irregular (bosselated) margins (arrowheads) and are supported by a variably dense fibrovascular stroma (E). At the periphery of islands, odontogenic epithelial cells with nuclei in antibasilar locations (ameloblastic morphology) transition from loosely palisading (arrows) to a jumbled arrangement (arrowhead).
The audience was asked to diagnose the mass and the voting choices and results were as follows: ameloblastoma (39%), ameloblastic odontoma (32%), ameloblastic carcinoma (28%), odontoma (1%), squamous cell carcinoma (0%), and melanoma (0%). Dr Lennix was visibly pleased some participants agreed with her diagnosis of ameloblastic carcinoma and a variety of other answers were selected by participants.
Dr Lennix walked through features of the tumor in this case. She pointed out odontogenic epithelial cells arranged in islands and trabeculae that surrounded a large cystic space, replaced bone, and compressed adjacent skeletal muscle (Figure 6C and D). The neoplastic cells were arranged in highly cellular aggregates with irregular margins supported by variably dense fibrovascular stroma (Figure 6E). At the periphery of the islands, cuboidal to columnar neoplastic odontogenic epithelial cells with nuclei occasionally in antibasilar locations were arranged in undulating or bosselated margins forming “ink drop” patterns (Figure 6E). At the center of islands, neoplastic cells were spindle shaped and formed streams and bundles (“sarcoma-like”) or were polygonal and reminiscent of stellate reticulum with variably distinct fine intercellular processes and polygonal nuclei (Figure 6G and H); in some regions, the neoplasm exhibited cystic degeneration (Figure 6I). Multiple cysts lined by parakeratinized stratified squamous epithelium contained free erythrocytes, hemosiderophages, degenerate epithelial cells, and neutrophils. In a few areas, cells at the center of islands showed squamous differentiation and some small aggregates of were keratinized (Figure 6I). There were18 mitoses per 2.37 mm2 and, interestingly, mitotic figures were observed both in basilar cells and those cells more centrally located within islands. Cells demonstrated mild anisocytosis and anisokaryosis (Figure 6J). Vascular and neural invasion were not observed. The neoplastic cells had positive immunoreactivity to both CK and vimentin with moderate and strong cytoplasmic staining, respectively (Figure 6K and L).
Next, Dr Lennix discussed the diagnostic features of proliferative lesions associated with teeth and referenced a number of sources4,8,10,12,14,20,22,26,28,30,31 as summarized in Table 7. She noted the specific features important in diagnosis of the tumor which include (1) identification of odontogenic epithelium, (2) stellate reticulum, (3) a determination on the presence of dental hard tissues, and (4) mitotic figures. Typically, odontogenic epithelial cells demonstrate ameloblastic morphology characterized by palisading cuboidal to columnar epithelial cells with antibasilar nuclei (i.e., reversed polarity) and clear space in the basilar cytoplasm. Stellate reticulum is composed of polygonal-shaped cells separated by faintly staining matrix or clear space with delicate cell-to-cell cytoplasmic processes. One or both of these tissue arrangements should be present in odontogenic tumors and are variably distinct. In this particular neoplasm, the odontogenic epithelium was loosely ameloblastic with many areas of jumbled epithelial cells with nuclei that failed to regularly demonstrate the characteristic palisading of ameloblastoma. A well-defined stellate-like reticulum was only present in a few regions. Dental hard tissues were not seen in the neoplasm and thus ruled-out odontoma and ameloblastic odontoma. Regarding odontogenic tumors, Dr Lennix mentioned her experience in diagnosing canine ameloblastoma has included the observation of low mitotic counts that are frequently 0 to 1 mitotic figures in 10 high-power (400×) fields. As a result, the more robust mitotic count in this case raised her suspicion that a diagnosis other than ameloblastoma should be considered. Dr Lennix briefly mentioned the presence of squamous differentiation and keratinization are not features specific to oral squamous cell carcinoma and have been observed in ameloblastic carcinoma. 32
Dr Lennix briefly discussed the diagnostic criteria for dental tumors in INHAND 10 and compared the features of odontoma, ameloblastic odontoma, and ameloblastoma to ameloblastic carcinoma. Dr Lennix reiterated that odontogenic epithelium and stellate reticulum, while reliably distinct in ameloblastoma, were not easily identifiable in this case, thus adding greater suspicion to her hunch that another diagnosis should be considered. Dr Lennix then gave an overview of publications related to select odontogenic tumors in the veterinary and toxicologic pathology literature (Table 8). Odontogenic tumors have been induced in mice by polyoma virus. 14 Experimentally induced odontogenic carcinoma has been reported in Wistar rats. 28 Spontaneous ameloblastoma has been described in rats and has been considered rare10,26 and, similarly, spontaneous ameloblastic carcinoma has been reported in a dog, 22 a horse, 8 and a domestic brown rat. 32 In Fischer rats, ameloblastic carcinoma has been induced by methlynitrosourea. 4 In dogs, a diagnosis of ameloblastic carcinoma relies on microscopic features such as increased mitotic rate, loss of odontogenic features, cellular pleomorphism, and biologic behavior (i.e., metastatic disease).30,31 Descriptions of ameloblastic carcinoma have included the features of high nuclear to cytoplasmic ratio, dark eosinophilic or vacuolated cytoplasm, nuclear pleomorphism with large to giant and pyknotic or small, and hyperchromatic nuclei.8,22 Commentary within the veterinary literature is sparse regarding mitotic activity with mitoses of 6 to 9/hpf and 5 to 6/hpf reported in ameloblastic carcinoma in a dog and horse, respectively.8,22
Overview of publications related to select odontogenic tumors in the veterinary and toxicologic pathology literature.
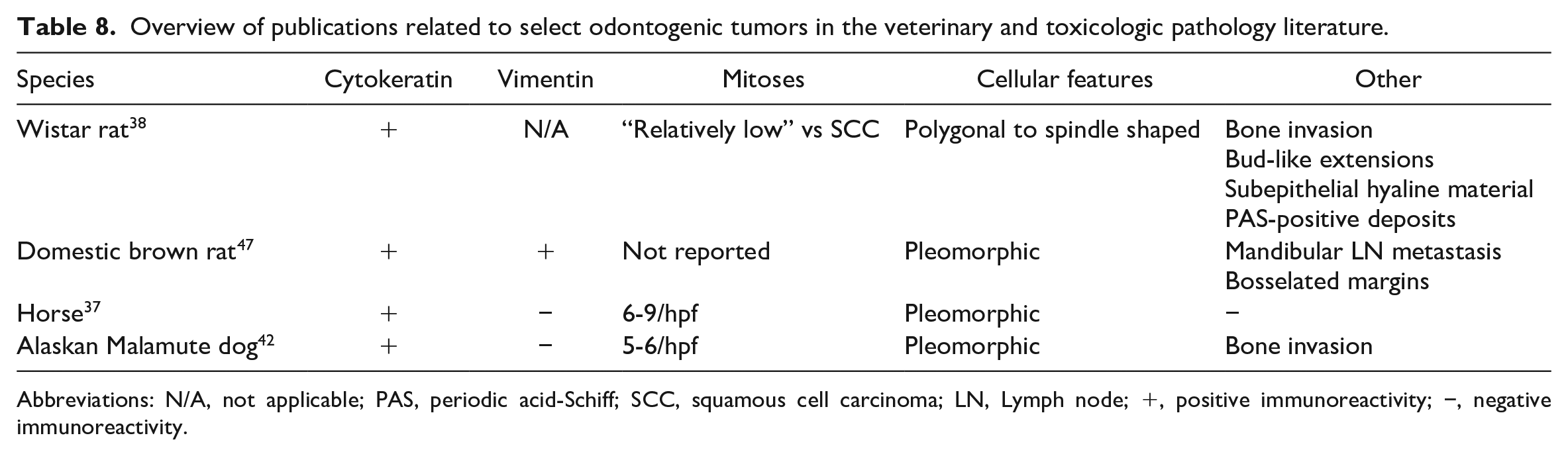
Abbreviations: N/A, not applicable; PAS, periodic acid-Schiff; SCC, squamous cell carcinoma; LN, Lymph node; +, positive immunoreactivity; −, negative immunoreactivity.
Dr Lennix then concluded with a summary of the case and take home points. The histopathologic distinction between ameloblastoma and ameloblastic carcinoma can be a difficult one to make. Despite having some features relatable to ameloblastoma, a diagnosis of ameloblastic carcinoma was favored based on the presence of odontogenic epithelium, stellate-like reticulum, cellular pleomorphism, and increased mitotic figures. Cellular pleomorphism was characterized by a combination of neoplastic cells exhibiting polygonal and spindle-shaped morphology in a pattern of streaming bundles that gave it a “sarcomatous” appearance. The co-expression of vimentin and CK has been reported in ameloblastic carcinoma in a domestic brown rat 32 and has been described in human ameloblastoma and fetal tooth germ. 20 In addition, in humans, ameloblastic carcinoma reliably expresses CK18, MMP-2, and MMP-9 and has an increased Ki67 index which distinguishes it from ameloblastoma. 46
Footnotes
Acknowledgements
The authors thank the DTT/NIEHS and the DTT pathologists for their guidance and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported [in part] by the NIH, National Institute of Environmental Health Sciences.

