Abstract
The 2024 annual Division of Translational Toxicology (DTT) Satellite Symposium, entitled “Pathology Potpourri,” was held in Baltimore, Maryland, at the Society of Toxicologic Pathology’s 42nd annual meeting. The goal of this symposium was to present and discuss challenging diagnostic pathology and/or nomenclature issues. This article presents summaries of the speakers’ talks along with select images that were used by the audience for voting and discussion. Various lesions and topics covered during the symposium included induced nonneoplastic lesions in the mouse kidney, induced and spontaneous neoplastic lesions in the mouse lung, infectious and proliferative lesions in nonhuman primates, an interesting inflammatory lesion in a transgenic mouse strain, and a lesson on artifact recognition.
Keywords
A Glomerular Conundrum
Dr Shambhunath Choudhary (Pfizer Inc., Pearl River, New York) presented test article-related lesions in the glomeruli of the kidneys in male and female CD-1 mice from a 26-week repeated dose oral toxicity study. At necropsy there were no significant gross pathology observations or any kidney specific clinical pathology findings. Histopathologic evaluation revealed characteristic glomerular lesions (Figure 1A-B) in mid- and high-dose animals.
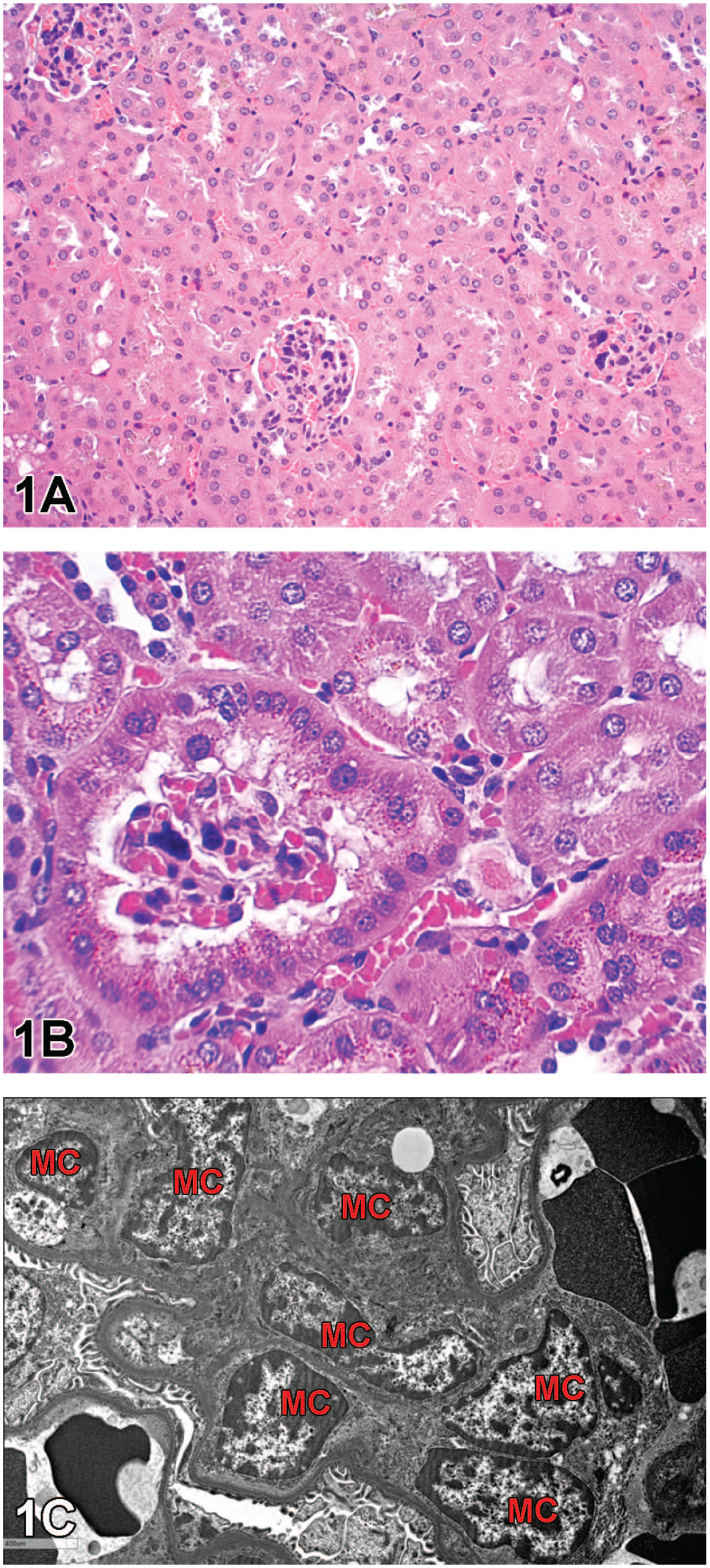
A-C, Representative images of kidney findings from CD-1 mice. (A) Low magnification showing cell aggregates in the glomerular tufts. H&E. (B) Higher magnification of (A) showing cell aggregation within the glomerulus and pigment accumulation in the parietal and tubular epithelial cells. H&E. (C) TEM image of glomerulus showing proliferating mesangial cells (MC) with multi-lobulated nuclei.
Low- and high-magnification photomicrographs of H&E-stained kidney sections with representative lesions observed in both sexes were shown to the audience for voting (Figure 1A-B). The most prominent histological feature included aggregation of cells within the glomerular tufts; these cells appeared as one to multiple, round to elongated, globular basophilic structures (Figure 1A-B). Multifocally, golden brown pigments were observed within the parietal epithelium of glomeruli and adjacent tubular epithelium (Figure 1B). Voting choices and audience responses were (1) glomerulonephritis (0%); (2) increased cellularity, mesangial cells (13%); (3) hypertrophy, mesangial cells (44%); (4) increased cellularity, podocytes (2%); (5) hypertrophy, podocytes (16%); (6) increased cellularity, glomerular endothelial cells (2%); (7) hypertrophy, glomerular endothelial cells (16%); and (8) 1 & 2 (7%).
Dr Choudhary then presented a couple of Transmission Electron Microscope (TEM) images of the glomerular lesion shown previously in light microscopic images (Figure 1C). The voting choices shared previously were shared again, and the audience was asked to vote. On this second occasion, responses were (1) glomerulonephritis (2%); (2) increased cellularity, mesangial cells (13%); (3) hypertrophy, mesangial cells (38%); (4) increased cellularity, podocytes (6%); (5) hypertrophy, podocytes (21%); (6) increased cellularity, glomerular endothelial cells (6%); (7) hypertrophy, glomerular endothelial cells (12%); and (8) 1 & 2 (2%). Based on TEM images (Figure 1C), the proliferating cells in glomeruli were identified as mesangial cells (with bi- or multilobulate nuclei and expanded mesangial matrix); therefore, the diagnosis was increased cellularity of mesangial cells. This set the stage for a quick review and discussion on the mesangial cells of the glomerulus.
The mesangial cells together with the podocytes and the glomerular endothelial cells form the glomerular capillary tuft responsible for the kidney ultrafiltration function. Parietal epithelial cells lining the Bowman’s capsule are the fourth cell type within the glomerulus. 15 Mesangial cells make up about 30 to 40% of the glomerular cell population and produce a mesangial matrix. The mesangium (mesangial cells together with matrix) forms a stalk that hold and organize the multiple capillary loops within the glomerulus.3,15 Accordingly, the injury or loss of mesangial cells, known as mesangiolysis, leads to loss or simplification of capillary loops. Mesangial cells are considered glomerular stromal cells and in addition to supporting glomerular capillaries, they have homeostatic functions and phagocytic activity. As such, they are involved in the glomerular development, the production of glomerular basement membrane matrix, and may facilitate the glomerular basement membrane turnover. 3 Once activated, mesangial cells may also regulate immune responses via recruitment of immune cells (through secretion of cytokines/chemokines or expression of Toll-like/ intracellular pattern recognition receptors), by controlling inflammation or mediating the reparative response to injury. 3
Increased number of mesangial cell nuclei with or without concomitant increase in cell size by light microscopic examination is considered the main diagnostic feature lesion a diagnosis of increased cellularity of mesangial cells. 22 A confirmative diagnosis should be made with electron microscopy to further characterize the cell type(s) affected within the glomerular tuft. Hyperglycemia, hypertension, and/or increased glomerular filtration rate have been linked to mesangium expansion in rats and humans.46,51 Over time and with further injury, increased cellularity of mesangial cells may be associated with an increased production of mesangial matrix proteins and marked proteinuria, where the lesion can then be more properly labeled as mesangioproliferative glomerulonephritis. 22 This hyperplastic change, however, is not considered a preneoplastic lesion and has not been associated with renal tumors in toxicity studies. 22
Dr Choudhary then briefly presented the study design and briefly explained the likely pathogenesis of the lesion. An additional finding in these kidneys was the presence of pigment within the parietal and tubular epithelium, which was consistent with increased blood turnover, as suggested by a hematology finding of higher red blood cell indices (pharmacological effect of the test article). Mesangial cell proliferation was likely a compensatory response to eliminate accumulated blood pigments in the glomerular filtrate which would be consistent with a phagocytic activity of mesangial cells. This change was not considered adverse as it was not associated with any degenerative and/or inflammatory changes in the glomeruli and partial recovery of this finding occurred after the recovery period.
Dr Choudhary’s presentation ended with a lively audience discussion. This was a novel lesion to many attendees. Major part of discussion was focused on the types of glomerular-associated proliferative and/or neoplastic changes that can be observed in regulatory toxicity studies or in humans. Attendees agreed that although glomerular lesions are observed in several malignant diseases, primary tumors of glomeruli are rare and not well documented.
The main purpose of this presentation was to raise awareness among toxicologic pathologists about the type of glomerular changes commonly observed in regulatory toxicity studies. Pathologists should also pay attention to cell types or components of the glomerulus (eg, mesangium, podocytes, endothelial cells, parietal cells, basement membrane, or Bowman’s capsule), factors that can affect their function and/or morphology, and molecular pathology tools (TEM, special stains) that can help confirming the diagnosis.
Eternal Sunshine of the Unspotless Mind
Dr Typhaine Lejeune (Charles River Montreal) presented 2 cases of a similar microscopic mass found in the brain of a mouse and a rat from 2 different studies. Dr Lejeune wanted to acknowledge the work of the Charles River Montreal immunohistochemistry team on these cases, as well as the sponsor for both studies for authorizing this publication.
The first case featured a 12-week-old CD1 mouse (Crl:CD1[ICR]) from a 7-day intravenous (single dose at study start) study (case 1). Upon slide reading, a mass was observed on one of the brain sections of the animal. A series of low—and high-magnification H&E-stained sections of the mass were presented to the audience for their diagnosis (Figure 2A-D). The low-magnification image (Figure 2A) provided an overview of the location of the mass which was in the forebrain, adjacent to the right lateral ventricle. A higher magnification image demonstrated the compressive nature of the mass on surrounding brain parenchyma, as well as an apparent deviation of the adjacent ventricle (Figure 2B). The highest magnifications (Figure 2C-D) showed a homogeneous population of large cells with abundant eosinophilic cytoplasm and a central, oval nucleus with finely stippled chromatin and a nucleolus. Necrosis nor mitosis were observed. The audience was polled for their diagnosis and voting choices with results were as follows: hamartoma (0%), choristoma (4%), benign ependymoma (18%), malignant (low-grade) oligodendroglioma (12%), benign granular cell tumor (43%), secondary tumor (metastasis; 12%), and none of the above (10%) (Table 1).
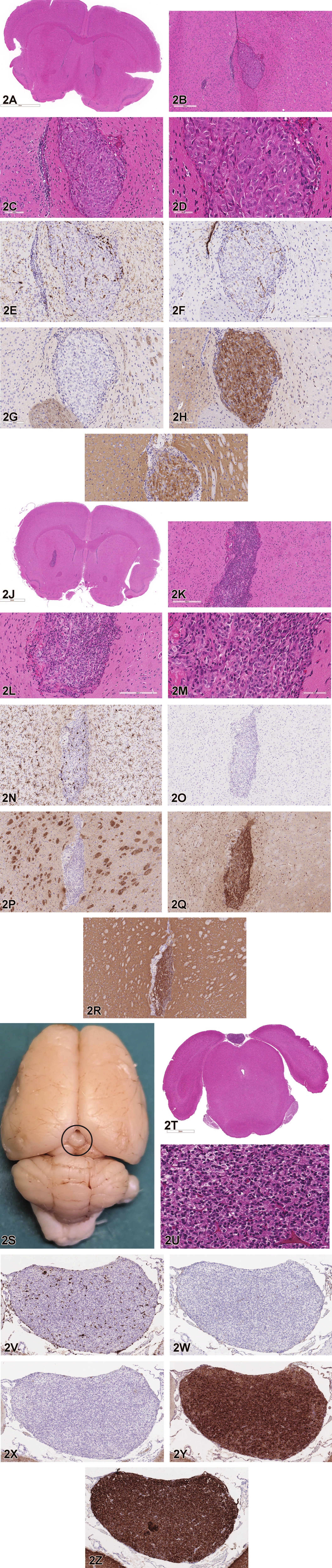
(A-D) Mass from a CD1 mouse (case 1). (A) A small mass was found in the forebrain section, next to the right lateral ventricle. HE. (B) The mass appeared to compress the surrounding parenchyma and slightly deviate the ventricle. HE. (C-D) On higher magnification, it was delineated by a thin fibrous capsule and composed of a homogeneous population of large cells with abundant eosinophilic cytoplasm and a central, oval nucleus with finely stippled chromatin and a nucleolus. HE. (E-I) Immunohistochemical stains performed on case 1. Cells were diffusely negative for IBA1 (E; except for a few individual cells), nestin (F) and neurofilament 200 (G), excluding a neuronal origin. Cells were diffusely positive for chromogranin A (H) and synaptophysin (I), demonstrating a neuroendocrine origin. (J-M) Mass from a Sprague-Dawley rat (case 2). (J) Similar to case 1, a small mass was found in the forebrain section, next to the left ventricle. HE. (K) It appeared to compress the surrounding parenchyma. HE. (L-M) At higher magnification, it was delineated by a thin fibrovascular capsule and composed of a homogeneous population of large cells with abundant eosinophilic cytoplasm and a central, oval nucleus with finely stippled chromatin. HE. (N-R) Immunohistochemical stains performed on case 2. Results were comparable to case 1 and as follows: cells were diffusely negative for IBA1 (N; except for a few individual cells), nestin (O) and neurofilament 200 (P); and diffusely positive for chromogranin A (Q) and synaptophysin (R). (S-U) Normal rat pineal gland. (S) Macroscopic view. In rodents, the pineal gland sits between brain hemispheres and the superior colliculi (circle). (T) Microscopic view of the pineal gland demonstrating how loosely it is attached and therefore, can be easily displaced. HE. (U) On higher magnification, the pineal gland is mainly composed of pinealocytes which are large cells with an abundant eosinophilic cytoplasm and an oval, central nucleus with finely stippled chromatin and a nucleolus. HE. (V-Z) Immunohistochemical stains performed on a normal rat pineal gland which stained similar to both cases presented: negative for IBA1 (V; except for a few individual cells which are microglial cells found in the normal pineal gland), nestin (W) and neurofilament 200 (X); and positive for chromogranin A (Y) and synaptophysin (Z).
Voting Results for Cases 1-2 in the Brain of Rodents.

Audience voting results after viewing immunohistochemistry images.
A recut deeper in the mass was requested but first, because of the risk of lesion exhaustion, 5 consecutive sections were cut (and kept unstained) and the recut was taken 200 µm deeper in the block. Unfortunately, there was no visible lesion on the recut as it was exhausted during sectioning. However, the unstained sections were used to investigate the cell of origin of the mass by immunohistochemistry. As per study protocol, IBA1 staining (for microglia) was already available and the following markers were requested: neurofilament 200 (NF200) and nestin for a possible neuronal origin and chromogranin A and synaptophysin for a neuroendocrine origin. As shown on Figure 2E-I, IBA 1 (Figure 2E), nestin (Figure 2F), and NF200 (Figure 2G) were negative, except for a few individual IBA1 or nestin positive cells scattered in the mass; and chromogranin A (Figure 2H) and synaptophysin (Figure 2I) were strongly and diffusely positive (cytoplasmic). The audience was polled a second time with the same list of diagnostic choices. Based on immunohistochemistry results, the poll outcome was as follows: hamartoma (0%), choristoma (9%), benign ependymoma (35%), malignant (low-grade) oligodendroglioma (2%), benign granular cell tumor (31%), secondary tumor (metastasis; 11%), and none of the above (but what happened to the fifth unstained slide?; 13%).
Dr Lejeune mentioned that a few weeks passed by when she came across a 7.5-month-old Sprague-Dawley rat (Crl:CD[SD]) in a 6-month oral gavage study (case 2) in which a microscopic mass was found in the brain. A series of low- and high-magnification H&E stained sections of the mass (Figure 2J-M) were presented and featured the same characteristics as the first case: a compressive mass was observed in the forebrain section, adjacent to the left ventricle and composed of a homogeneous population of large cells with abundant eosinophilic cytoplasm and a central, oval nucleus with finely stippled chromatin. The same immunohistochemistry panel was run on this second mass (Figure 2N-R) and results were also similar to the first case: IBA1 (Figure 2N), nestin (Figure 2O) and NF200 (Figure 2P) were negative, except for a few individual IBA1 positive cells scattered in the mass; and chromogranin A (Figure 2Q) and synaptophysin (Figure 2R) were strongly and diffusely positive (cytoplasmic). The audience was polled one last time in light of this new case and results were as follows: hamartoma (5%), choristoma (0%), benign ependymoma (10%), malignant (low-grade) oligodendroglioma (7%), benign granular cell tumor (21%), secondary tumor (metastasis; 7%), none of the above, this just an artifact (50%).
Dr Lejeune revealed that this was an artifact in both cases and explained why she came to that conclusion. On H&E sections (Figure 2C and 2L), the compressed parenchyma surrounding each mass was exclusively occupied by dark neurons which are a known handling artifact of brain, demonstrating that surrounding tissues had been pushed aside rather than truly compressed in-life. Furthermore, in case No. 2 (Figure 2K), medium sized vessels observed on the margin of the mass were too large to be from the brain parenchyma or from tumoral neovascularization and IBA1 staining (Figure 2G and 2P) showed a lack of microglial reaction in the surrounding brain parenchyma. In addition, both animals did not have any clinical signs, gross findings or any other microscopic finding.
A review of the list of proposed diagnostics for these 2 masses was proposed with their respective features described. Hamartomas and choristomas 52 are both developmental anomalies featuring a nonneoplastic mass of mature tissue which is either derived from a resident cell type for hamartomas or histologically normal for a part of the body other than the one in which it is located for choristomas (eg, a corneal dermoid). Benign ependymomas 49 feature cells arranged in rows and rosettes (with perivascular pseudo-rosettes) and their immunohistochemical characteristics include positive staining for GFAP, S100, NCAM, and nestin; the latter being negative in both masses presented. There are reports of ependymomas with a neuronal differentiation 39 with occasional synaptophysin or chromogranin A positive staining. However, such reports have not been found in animals. A malignant oligodendroglioma of low grade would have a characteristic “honeycomb” pattern which features cells with distinct borders and a clear (or lightly stained) cytoplasm.25,49 A benign granular cell tumor was included in the proposed list as cytoplasmic granules are not always visible on H&E sections and the general cell morphology (large cells with abundant cytoplasm and a central large nucleus) of the 2 masses were similar to that of a granular cell tumor. However, benign granular cell tumors in the brain of rodents are confined to meninges. They are the most common meningeal tumor in rats and are considered a meningioma variant.25,49 Granular cell tumors will stain positive with a histochemical stain such as PAS and positive for S100 immunohistochemical stain, among other markers. Finally, as there were no other microscopic findings in the animals, a secondary tumor was ruled out.
The next question raised was: which artifact could this be? As shown in Figure 2S-U, the pineal gland, which is a nearby structure, was a likely option. Indeed, in rodents, the pineal gland sits between brain hemispheres and the superior colliculi (Figure 2S) and is loosely attached; such that it is usually pulled apart from the brain at necropsy upon removal of meninges. In some cases, it does remain attached to the brain but can be easy to disturb from its normal location during histology processes. According to Dr Lejeune, it proved difficult to find consistent immunohistochemical features of the normal rodent pineal gland in the literature, so she decided to run the same immunohistochemical panel on a rat pineal gland. The staining results, as shown in Figure 2V-Z, were the same as both presented cases allowing to conclude that, in both cases, this artifact was in fact a misplaced pineal gland.
An overview of the pineal gland features, function and lesions was undertaken. 23 The pineal gland was discovered by Galen of Pergamon in the before-Christ era. It is a neuroendocrine gland, part of the circumventricular organs which means it does not have a blood-brain barrier. The majority of the gland is composed of modified neurons, the pinealocytes, and remaining cells are glial cells (astrocytes and microglia). The pineal gland is the circadian oscillator of the body through the nocturnal production and secretion of melatonin. Pinealocytes synthetize melatonin from the amino acid tryptophan during darkness, which is secreted directly as pinealocytes do not have storage capabilities. In higher vertebrates, the pineal gland is activated through the photosensitive retina. However, retinal photoreceptors and their opsin pigments are not directly involved in this process. Instead, there exists a subpopulation of specialized photosensitive ganglion cells (ipRGC) which bear a specific pigment, the melanopsin. Upon activation of ipRGC, the photic information is sent to the suprachiasmatic nucleus (SCN) of the brain through the retino-hypothalamic tract (RHT). The path continues with 3 other synapses in the paraventricular nucleus in the hypothalamus, the intermediolateral nucleus in the thoracic spinal cord and the superior cervical ganglion (SCG), respectively. Post-synaptic fibers from the SCG release norepinephrine which, in turn, activates pinealocytes. Under light conditions, the SCN secretes γ-amino butyric acid (GABA) which inhibits melatonin synthesis whereas during darkness, the secretion of glutamate by the SCN activates melatonin synthesis. Interestingly, shorter wavelengths of light (in the blue-violet) are more efficient at activating the RTH and thus, preventing sleep, as do screens emitting blue light. In addition, ipRGC are also responsible for the pupillary light reflex through a different pathway.
According to the published INHAND nomenclature, 9 nonproliferative lesions of the rat and mouse pineal gland include fibrosis, inflammatory cell infiltration, mineralization, vacuolation and the presence of striated muscle fibers. Proliferative lesions are benign or malignant pinealomas which are distinguished from one another based on growth pattern (noninvasive vs invasive) and cytological features of malignancy such as high mitotic rate, pleomorphism and necrosis. Regardless of malignancy, pinealoma cells can be positive for synaptophysin, neuron-specific enolase or neurofilament markers.
In the light of the 2024 STP symposium about innovative therapies, it is interesting to note that researchers have found that AAV vectors accumulate in the pineal gland of transgenic mice after injections into the brain or spinal cord. 44 The main take home point emphasized was the fact that wherever a pathologist is in their career, humility remains a fundamental quality to sustain.
What’s the Skin-ny?
Dr Emily Mackey (Anatomic pathology resident at Wake Forest University School of Medicine, Winston-Salem, NC) presented a case of genital squamous cell carcinoma in situ in a cynomolgus macaque (Macaca fascicularis). Dr Mackey would like to acknowledge Dr Cline for his mentorship and assistance with the presentation. She would also like to thank Drs Nancy Kock (WFU), Gayathriy Balamayooran (WFU), David Caudell (WFU), and Richard Lang (Experimental Pathology Laboratories, Inc., Durham, North Carolina) for additional case review and Cathy Mathis (WFU) and Lisa O’Donnell (WFU) for histology and immunohistochemistry assistance. Dr Mackey would also like to acknowledge, Dr Omar Sangueza (WFU) for providing dermatopathology expertise.
The case presentation featured a male cynomolgus macaque, that was estimated to be 32 years old. This animal was imported from Indonesia in 2003, and at that time was approximated to be 20 years old. The animal was utilized in several different experimental protocols over 12 years at Wake Forest University, most involving diabetes mellitus and glucose regulation. Clinical euthanasia was elected for this animal due to declining clinical condition and inappetence following an episode of sedation. Prior to the death, the animal had an approximately 3-year history of scrotal and penile lesions.
The audience was first shown a gross photograph of the scrotal and penile lesions (Figure 3A) and asked to choose the best diagnosis. Voting choices and results were as follows: (1) Maculopapular rash: measles virus (0%), (2) Vesicular dermatitis: macacine herpesvirus 1 (“Herpes B” virus) (8%), (3) Papillomas: papillomavirus (32%), (4) Pustular dermatitis: Staphylococcus aureus (8%), (5) Granulomatous dermatitis: Mycobacterium leprae (11%), (6) 1 and 3 (22%), (7) 2 and 3 (8%), and (9) 3 and 5 (11%). The audience was then shown four sequentially magnified photomicrographs of H&E stained sections taken from an ulcerated scrotal lesion present on the gross photo (Figure 3B-E). The lesion was characterized by irregular epidermal hyperplasia that extended deep into the dermis, which was infiltrated by lymphocytes (Figure 3B). Figure 3C highlighted erosion of the overlying epidermis with replacement by a serocellular crust. Figure 3D demonstrated full thickness dysplasia of the epidermis characterized by loss of cell polarity and normal stratification of the keratinocytes. Some keratinocytes had abundant clear cytoplasm with hyperchromatic nuclei (koilocytic change), shown in Figure 3E. The audience was then asked to choose the best diagnosis for this lesion. The voting choices and results were as follows (1) epidermal hamartoma (0%), (2) chronic hyperplastic dermatitis (34%), (3) actinic keratosis (11%), (4) papilloma (6%), (5) squamous cell carcinoma in situ (21%), (6) basal cell carcinoma (23%), and (7) squamous cell carcinoma (4%).
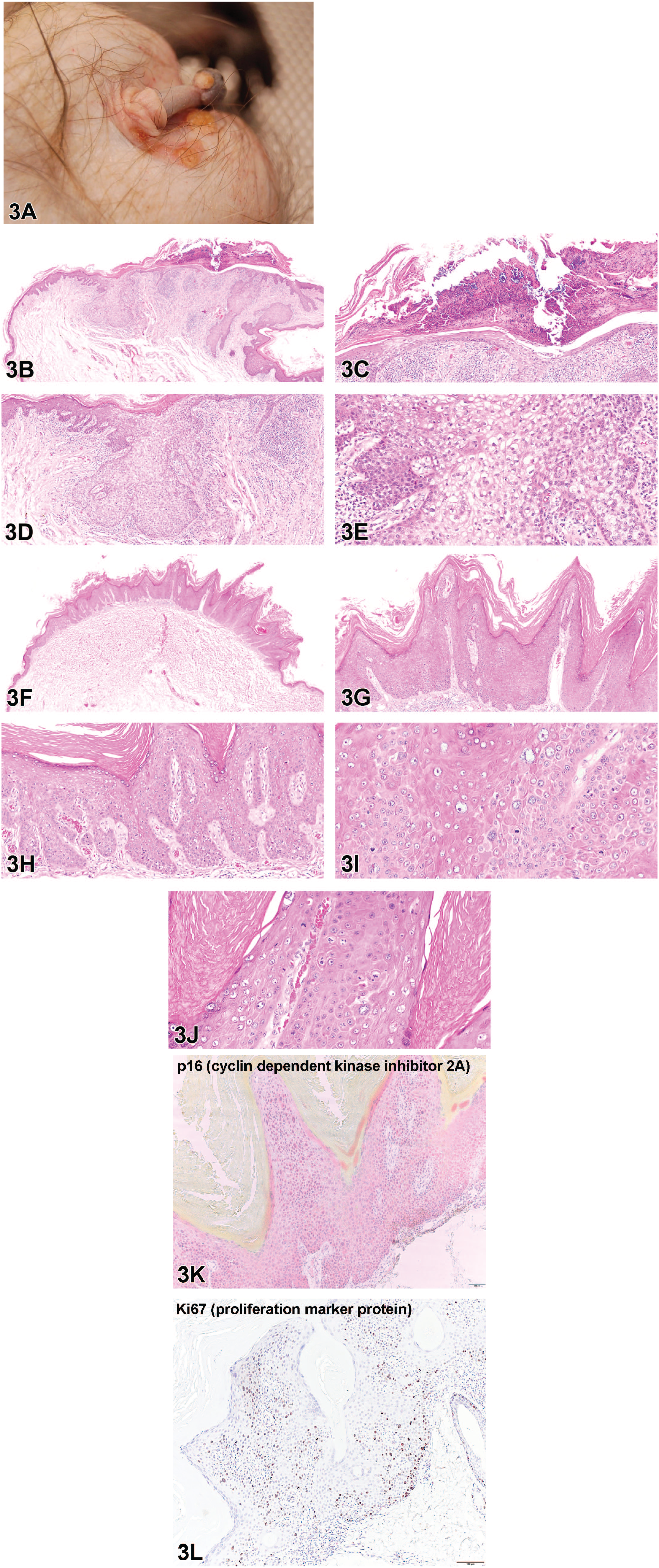
(A) A gross photograph demonstrates several about 5-10mm in diameter proliferative and erosive lesions on the scrotum and penis from a 32-year-old male cynomolgus macaque (Macaca fascicularis). (B-E): Histologic images from an erosive scrotal lesion. (B) A low magnification H&E-stained photomicrograph from the scrotum demonstrates marked irregular epidermal hyperplasia with lymphocytic dermatitis. (C) A serocellular crust composed of keratin, abundant neutrophils, proteinaceous fluid, and colonies of bacterial cocci is present over the eroded epithelial surface. (D) Higher magnification H&E-stained photomicrograph of the lesion demonstrates dysplasia of the epidermis characterized by loss of cell polarity and normal stratification of the keratinocytes. (E) On higher magnification of (D), keratinocytes have abundant clear cytoplasm and hyperchromatic nuclei, characteristic of koilocytic change. H&E indicates hematoxylin and eosin. (F-J): Histologic images from a proliferative scrotal lesion. (F) A low magnification H&E-stained photomicrograph from the scrotum demonstrates an exophytic epidermal lesion. (G-H) Higher magnification H&E-stained photomicrographs of the lesion demonstrate full-thickness epidermal dysplasia that is confined by a basement membrane. On higher magnification, there is marked anisokaryosis of keratinocytes throughout the epidermis (J) and atypical mitoses are present (I). H&E indicates hematoxylin and eosin. (K-L): Immunohistochemical-stained panels of the second scrotal lesion demonstrate (K) moderate full thickness and continuous cytoplasmic and nuclear p16 immunostaining of the lesion, excluding the hyperkeratotic region and (L) strong scattered nuclear Ki67 positivity throughout all layers of the epidermis.
Histologic images from a separate proliferative scrotal lesion from the original gross photograph were presented to the audience (Figure 3F-J). The lesion was characterized by an exophytic well-demarcated area of epidermal hyperplasia with overlying abundant orthokeratotic hyperkeratosis (Figure 3F). In this area, the epidermis was expanded by dysplastic keratinocytes that were confined by the basement membrane (Figure 3G). There was marked anisokaryosis of nuclei throughout the epidermis (Figure 3I) and abnormal mitotic figures were present (Figure 3J). The voting choices and results were as follows: (1) epidermal hamartoma (0%), (2) chronic hyperplastic dermatitis (27%), (3) actinic keratosis (4%), (4) papilloma (46%), (5) squamous cell carcinoma in situ (21%), (6) basal cell carcinoma (2%), and (7) squamous cell carcinoma (0%).
Prior to revealing the answer, the audience was shown p16 and Ki67 immunohistochemistry (IHC) of the second scrotal lesion (Figure 3K-L) and told that both lesions had similar IHC findings. P16 is a cyclin dependent kinase inhibitor that prevents progression into the S phase of the cell cycle. 48 P16 immunostaining had full thickness and continuous cytoplasmic and nuclear immunostaining of the lesion, excluding the hyperkeratotic region, and therefore was considered positive. 11 Ki67 is a marker of proliferation, and the lesion had strong scattered nuclear positivity throughout all layers of the epidermis, which was considered patchy, yet positive immunostaining. 11 Given the gross, histologic, and IHC findings in this case the audience was then asked what was the most likely cause of the lesions. The voting choices and results were as follows: (1) ultraviolet light exposure (12%), (2) papillomavirus infection (70%), (3) chronic inflammation (9%), (4) poxvirus infection (3%), (5) epidermal toxin exposure (0%), and (6) herpesvirus infection (3%). Dr Mackey agreed with the majority of the audience that the findings were consistent with papillomavirus-induced lesions.
Dr Mackey then revealed that both histologic lesions presented in this case were given the morphologic diagnosis of squamous cell carcinoma in situ. Carcinoma in situ is characterized by irregular epidermal hyperplasia with full-thickness dysplasia manifested by loss of nuclear polarity and disruption of normal epithelial stratification, 50 as highlighted in the photomicrographs shown in both cases. Keratinocyte size and appearance are highly variable in carcinoma in situ and a papillomaviral cytopathic effect, manifested by koilocytosis in which keratinocytes have abundant clear cytoplasm and hyperchromatic nuclei, may be detected. 50 This feature was more prominent in the first histologic lesion presented (Figure 3E). Other major keratinocyte features of carcinoma in situ include nuclear hyperchromasia, anisokaryosis (Figure 3I), pleomorphism, cellular crowding, and abnormal mitotic figures (Figure 3J). Hyperkeratosis with or without serocellular crusting may also be observed, 22 (Figure 3B). A key feature used to differentiate this entity from squamous cell carcinoma is the presence of an intact basement membrane. 22
Carcinoma in situ of male genitalia is referred to as penile intraepithelial neoplasia in the human literature and is classified as nonhuman papillomavirus (HPV)-related or HPV-unrelated. 51 HPV-related penile intraepithelial neoplasms are much more common in humans, especially in North America. 24 Dr Mackey then provided background information on papillomaviruses, which are small, nonenveloped, double-stranded DNA viruses of the family Papillomaviridae that infect epithelial cells and replicate in keratinocyte nuclei. 30 These viruses infect keratinocytes at mucosal and cutaneous sites and are highly host-specific. 30 Papillomaviruses gain access through defects in the epithelium and infect cells of the stratum basale which leads to the activation of a cascade of viral gene expression that induces basal cell proliferation and terminal keratinocyte differentiation. 30 This cascade also leads to the production of the oncoprotein E7, which binds to retinoblastoma protein and signals it for degradation. 30 Retinoblastoma is a key protein in the cell cycle that binds and prevents the transcription factor E2F from entering the nucleus to initiate cell cycle progression. 30 Degradation of retinoblastoma protein by the papillomaviral oncoprotein E7 leads to constitutive release of E2F into the nucleus and initiation of cell cycle progression. 30 This uncontrolled cell proliferation initiates a negative feedback loop that signals p16 upregulation. Therefore, loss of retinoblastoma due to papillomaviral oncoprotein E7 results in an immunohistochemically detectable increase in cell p16, as was present in this case (Figure 3K). Upregulation of p16 is such a consistent finding that IHC is used in many studies to predict papillomavirus etiology and p16 expression is considered an optimal surrogate for papillomaviral-induced neoplasms.24,28,41,48
Dr Mackey then reviewed the key features of the other proposed diagnoses for the histologic lesions in this case. Epidermal hamartomas are plaques that are histologically characterized by irregular acanthosis with normal epidermal maturation and marked, laminated hyperkeratosis. 50 The plaques do not have epidermal dysplasia or p16 expression, which was demonstrated in the presented case. 50 Carcinoma in situ can be difficult to differentiate from chronic hyperplastic dermatitis. 50 Chronic hyperplastic dermatitis is characterized by irregular acanthosis with patchy spongiosis, hyperkeratosis, serocellular crusting, perivascular to interstitial dermal inflammation and sebaceous hyperplasia. 50 Dysplasia should not be a prominent feature and the epidermis would not diffusely express p16, as shown in the case presented. Actinic keratosis needs to be differentiated from squamous cell carcinoma in situ, as many features are similar. Actinic keratosis is characterized by irregular epidermal hyperplasia and atypia of keratinocytes with loss of polarization, crowding and overlapping, but a key feature is that the atypia is confined to the basal layers. 50 Dermal solar elastosis and fibrosis are also features that support actinic keratosis. 50 The full-thickness epidermal dysplasia and p16 positivity in the lesions shown support carcinoma in situ rather than actinic keratosis. Papillomas are benign, exophytic squamous epithelial proliferations that are composed of branching papillary fronds with central fibrovascular cores lined by hyperplastic nonkeratinizing squamous epithelium. 50 These are often associated with papillomavirus infection, however, the marked cellular pleomorphism and full-thickness mitotic activity with atypical mitoses in the case presented indicated malignant transformation and therefore carcinoma in situ. 50 Basal cell carcinoma arise from the follicular epithelium and essential features include nests of small basaloid cells with peripheral palisading and a fibromyxoid stroma, 50 which was not compatible with the observed lesions. Finally, squamous cell carcinoma is characterized by atypical keratinocytes that invade into the dermis and therefore breach basement membrane, 50 which was not observed in the lesions presented.
As the focus of the 2024 STP Symposium was innovative therapeutics, Dr Mackey ended the case presentation discussing the translational aspect of therapeutics for papillomavirus infection in macaques and humans. Human papillomaviruses (HPV) are medically important pathogens that are responsible for a diverse range of epithelial manifestations including malignant neoplasia, but there is still no approved effective cure for an ongoing human papillomavirus infection. 33 Therefore, there is a great need for animal models that accurately mimic papillomavirus infection in humans and allow for testing of therapeutic strategies. Nonhuman primates captured from wild are quite frequently infected with specific papillomaviruses, which can cause clinical manifestations of viral infection similar to HPV-associated lesions in humans. 12 At least 30 nonhuman primate papillomavirus types have been identified and some are very similar to human high-risk papillomaviruses. 12 For example, Macaca fascicularis papillomavirus type 3 is the most common macaque papillomavirus, and is genetically and phylogenetically similar to human papillomavirus 16, a common high-risk HPV strain in humans. 33 A 2021 study aimed to design a therapeutic vaccine against Macaca fascicularis papillomavirus type 3 that could be utilized in persistently papillomavirus-infected cynomolgus macaques. 33 The configuration of this vaccine may serve as template to generate novel vaccine candidates for therapeutic vaccination against high-risk HPV types, such as the analogous HPV 16 virus.
Dr Mackey concluded with several take home points regarding papillomavirus infection. Papillomavirus infection in macaques can cause similar lesions to those in humans, including inducing penile intraepithelial neoplasms, and p16 IHC expression can serve as a surrogate marker for infection. Furthermore, human papillomaviruses are prevalent and harmful pathogens that necessitate animal models able to accurately mimic infection and allow for testing of therapeutic strategies. Ultimately, nonhuman primates may be the best available model for developing effective treatments against human papillomavirus infection.
Trails of Ephemeral Ambiguity
Dr Priyanka Thakur (Charles River Laboratories, Inc., [CRL] Durham, North Carolina, contractor pathologist for DTT/NIEHS) presented three interesting cases of keratinizing and nonkeratinizing squamous lung lesions from Division of Translational Toxicology/National Institute of Environmental Health Sciences (DTT/NIEHS) rat inhalation studies.
The presented cases featured squamous lung lesions from female rats, which were part of a 2-year inhalation study of vanadium pentoxide and antimony trioxide from published technical reports (TR-590, antimony trioxide, TR-507, and vanadium pentoxide).31,32 Thanks to Dr Mark Cesta (DTT/NIEHS, North Carolina) and Dr Cynthia Willson (DTT/NIEHS [contractor pathologist from Inotiv], North Carolina), for their valuable feedback on the presentation and Emily Singletary (Experimental Pathology Laboratories, Inc., North Carolina) for scanning the whole slide images.
For all three cases, a series of photomicrographs were presented to the audience, including low- and high-power magnifications of select sections of lungs from the 2-year rat inhalation studies (Figure 4A-P). Dr Priyanka Thakur informed the audience that the diagnostic options would be the same for all three cases. The voting choices and results are presented in Table 2.
(A-D), Rat lung-cystic keratinizing epithelioma from a female Wistar Han Rat, HE stain. (A) A subgross section of the cystic keratinizing epithelioma shows a sharp demarcation between proliferating epithelioma cells and adjacent normal lung tissue. (B-C) Higher magnification images of the areas highlighted by red boxes in image A. These images show a central cavity filled with keratin (asterisks) and a thick complex and irregular cyst wall (arrows) resulting from local variations in proliferative activity, giving a rough or cobblestone appearance to the outer portion of the lesions. (D) Higher magnification image showing areas of active cellular proliferation where basal cells are disorganized (arrows). (E-J), Rat lung-squamous metaplasia from a female F344/N rat, HE stain. (E-I) The alveolar epithelium is replaced by squamous cells. The squamous metaplasia has more layers (indicated by arrows) and more keratin production (asterisk), along with greater proliferative activity, giving it a florid appearance. (J) An occasional focus shows nuclear pleomorphism and cellular atypia (arrow). (K-P), Rat lung-squamous cell carcinoma from a female Wistar Han Rat, HE. (K) A subgross section of the lung-squamous cell carcinoma features a large, predominantly solid mass with central keratin and necrotic tumor tissue. (L-O) The mass is composed of irregular nests or islands of disorganized pleomorphic squamous epithelial cells (L and O, arrows) that showed loss of polarity, cellular atypia, and mitotic figures (N and P, arrows). These islands of pleomorphic cells are separated by small to moderate amounts of fibrous stroma (L, arrowhead). A few of these islands contained central keratin pearls (L and M, asterisks).
Voting Results for Cases 1-3 in the Rat Lung Squamous Lesions.

Case 1, lung-cystic keratinizing epithelioma, was from a Wistar Han female rat exposed to antimony trioxide by inhalation for 2 years. The first image was a subgross section of the lesion (Figure 4A), and the audience was asked to focus on the highlighted regions inside the black boxes. High-magnification images of these regions were presented in the subsequent slides (Figure 4B-D). Pulmonary cystic keratinizing epithelioma, centrally filled with keratin, had a more complex, thicker, and irregular cyst wall due to local variations in proliferative activity. There was a sharp demarcation between proliferating epithelioma cells and adjacent normal lung tissue. The cyst wall often showed outgrowth of cells at the periphery and filled contiguous alveoli, giving a roughed or cobblestone appearance to the outer portion of the lesions (Figure 4C). In these areas, there were foci of active cellular proliferation where basal cells were disorganized, an important diagnostic feature of keratinizing epithelioma (Figure 4D).
Case 2 provided an example of lung-squamous metaplasia (Figure 4E-J) from a 2-year inhalation study of Vanadium Pentoxide in a female F344/N rat. This rat showed a significantly increased incidence of lung-squamous metaplasia. The alveolar epithelium in the affected area was replaced by squamous cells. In the areas of keratinization, there was an orderly progression from basal cells to keratinized surface epithelium, but the squamous metaplasia had more layers, more keratin production, and greater proliferative activity (Figure 4G and H). Occasional nuclear pleomorphism and cellular atypia were also observed (Figure 4J, arrow).
In case 3, a female Wistar Han rat exposed to antimony trioxide by inhalation for 2 years developed lung-squamous cell carcinoma. In the low-dose group, squamous metaplasia was observed, while in the high-dose group, one rat had two cystic keratinizing epitheliomas (case 1), and another rat had squamous cell carcinoma (case 3). The Keratinizing Squamous cell carcinoma presented for case 3 (Figure 4K-P) featured a large, predominately solid mass with central keratin and necrotic tumor tissue (Figure 4K). The mass was composed of irregular nests or islands of disorganized pleomorphic squamous epithelial cells (Figure 4L and O, arrow) that showed loss of polarity, cellular atypia, and mitotic activity (Figure 4N and P, arrows). These islands of pleomorphic cells were separated by small to moderate amounts of fibrous stroma (Figure 4L, arrowhead), infiltrating and obliterating the normal pulmonary architecture. A few of these islands contained central keratin pearls (Figure 4L and M).
During the discussion, Dr Thakur reviewed the historical incidences of squamous lung lesions. Squamous lung lesions, such as cystic keratinizing lesions, squamous metaplasia, and squamous cell carcinoma, are more commonly observed in rats than in mice. These lesions develop in response to the chronic inhalation of high-burden diverse particulate materials such as diesel exhaust, carbon black, talc, quartz, or titanium dioxide. 8 They are more likely to develop in rats with high exposure levels and typically appear late in the study. Furthermore, these lesions are more common in female than male rats but rarely occur spontaneously in control animals. 8
Dr Thakur shared with the audience that the most common chemically induced lung tumors in rats and mice are alveolar/bronchiolar (A/B) adenomas and carcinomas in DTT studies. The next two most common lung tumor types are squamous cell carcinoma and cystic keratinizing epithelioma. 14 Among DTT studies, the historical control incidence of cystic keratinizing epithelioma in rats (SD, F344/N) and mice (B6C3F1) is zero. The incidence of squamous cell carcinoma in control mice (B6C3F1) is zero and is less than 0.1% in control male and female F344/N rats. 14 Squamous metaplasia commonly occurs in inhalation studies in rats, usually related to the test article, but is not uncommon in vehicle-control inhalation studies.
Dr Thakur then shared a table (Table 3) showing squamous lung lesions induced by various chemical agents in DTT inhalation and gavage studies. Chemical exposures that led to more than one lung-squamous lesion were highlighted in bold text. It is interesting to note that both inhalation and gavage studies induced various lung squamous lesions, including squamous metaplasia, squamous cell carcinoma, and cystic keratinizing epithelioma. Factors such as enhanced local concentration and the physical and chemical characteristics of the inhaled material contribute significantly to inducing these squamous lesions in rodent chronic inhalation studies. In gavage studies, the lung may be exposed to unaltered chemicals or toxic metabolites through the bloodstream. 42 Another potential source of exposure could be through the reflux of materials given by gavage or inhalation during administration through gavage. 42
Squamous Lesions in Lungs in DTT Studies (Rats).

Dr Thakur provided an overview of the key diagnostic features of three types of lung squamous lesions: cystic keratinizing lesions, squamous metaplasia, and squamous cell carcinoma, with reference to the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND). 38 Keratinizing cysts are typically large, filled with keratin, and lined by well-differentiated squamous cells, forming a thin, uniform cyst wall. Dr Thakur emphasized that the thickness and nature of the cyst wall are important criteria for distinguishing between keratinizing cysts and keratinizing epithelioma. 38 It was discussed that while the range of squamous metaplasia observed in the case of the vanadium pentoxide female rat (case 2) was consistent with other studies, the lesion exhibited a higher level of proliferation and a greater degree of keratinization than expected with simple squamous metaplasia, leading to consideration of squamous cell carcinoma as a possible diagnosis. Although the lesions were quite florid, and several features suggested it was not squamous cell carcinoma. 32 The study also identified instances where keratinized material collected in lung alveoli formed keratinized cysts. In addition, the discussion highlighted that squamous metaplasia observed in this study was not associated with inflammation, in contrast to findings in other studies. 32
Dr Thakur concluded that while the precise nature of squamous cell proliferation in these lesions remains uncertain, multiple studies suggest a morphologic continuum from cystic metaplasia to squamous cell carcinoma. Although squamous metaplasia, keratinizing cyst, and cystic keratinizing epithelioma are common pathologic responses to a variety of inhaled particles in rats, they have also been observed in noninhalant rodent studies. For example, squamous metaplasia may also occur in rats fed vitamin A-deficient diets, in animals with subacute to chronic infectious pneumonias, and in lungs of rats chronically exposed to irritants in inhalation studies.45,47 Furthermore, epitheliomas that are more actively proliferating may progress to squamous cell carcinoma. Pathologists should be aware of other factors that can cause lesions commonly observed during rodent chronic studies.
Grizzly Paws
Dr Kristen Hobbie (Inotiv, Inc., RTP, NC) presented interesting examples of severe bone and inflammatory lesions in the hind paws of three female Ephx2tm1.1Dcz mice from a colony at the National Institute of Environmental Health Sciences (NIEHS) (RTP, NC). The study involving the three Ephx2tm1.1Dcz mice was initially evaluated by Dr Debabrata Mahapatra (Inotiv, Inc., RTP, NC), and subsequently presented and discussed at the Society of Toxicologic Pathology (STP) Division of Translational Toxicology (DTT) Satellite Symposium by Dr Kristen Hobbie. Dr Hobbie would like to acknowledge Dr Ron Herbert (DTT/NIEHS), Dr Charan Ganta (DTT/NIEHS/Inotiv, Inc.), Dr Debabrata Mahapatra (Inotiv, Inc.), Eli Ney (CMPB/NIEHS), and Molly Comins (NIEHS/Midnight Sun Technologies), as well as the NIEHS Pathology Core Lab (CMPB/NIEHS) and the Comparative Medicine Branch (NIEHS) for their contributions to the presentation.
All cases were presented to the audience as individual paws from the three affected female Ephx2tm1.1Dcz mice. Low, medium, and high magnifications of photomicrographs of hematoxylin & eosin (H&E)-stained paw slides from the three female Ephx2tm1.1Dcz mice were shared with the audience to aid in reaching diagnoses and a possible etiology. The voting choices and results for Paws 1-3 and for the possible etiology are shared in Table 4 and Table 5.
Voting Results for Paw 1-3.

Voting Results for Etiology.

Prior to sharing the photomicrographs of the histopathology in the hind paws, Dr Hobbie revealed the background of the Ephx2tm1.1Dcz mice. She informed the audience that Ephx2 mice have enabled epoxide hydrolase activity. 16 The Ephx2 gene participates in cholesterol metabolism and homeostasis, as well as other activities. 16 Ephx2tm1.1Dcz mice have an insertion mutation of the Ephx2 gene. 19 The Ephx2tm1.1Dcz mice with paw lesions in the study presented were female and approximately three months of age. They were part of a subset of male and female Ephx2tm1.1Dcz mice in the colony with paw lesions. All Ephx2tm1.1Dcz mice were specific pathogen-free and were untreated. All three female Ephx2tm1.1Dcz mice presented with swollen hind feet. They were housed in plastic, independently ventilated cages. Images taken with Faxitron® Core Specimen Radiography revealed bone lesions in the hind paws. Bone lesions included lytic areas with bony proliferation of the distal metatarsals, especially affecting digit 5. No other bony lesions were observed distal to the cervical vertebrae in these three mice.
Dr Hobbie first presented photomicrographs of Paw #1 to the audience to aid in the morphological diagnosis of the hind paw lesions in the three female Ephx2tm1.1Dcz mice. A low magnification photomicrograph of an unaffected forepaw from an Ephx2tm1.1Dcz mouse was included for comparison (Figure 5A). Photomicrographs included low (Figure 5B), medium (Figure 5C), and high (Figures 5D-5F) magnifications of images of H&E-stained slides from Paw #1. The majority of voters (49%) (Table 4) voted for osteolysis, osteomyelitis, and dermatitis, which was the correct answer. Dr Hobbie then presented low (Figure 5G), medium (Figure 5H), and high (Figures 5I and 5J) magnifications of photomicrographs of histopathologic lesions from Paw #2. The majority of voters (75%) (Table 4) voted for osteolysis, osteomyelitis, hyperostosis, and dermatitis, which was the correct answer. Finally, Dr Hobbie presented low (Figure 5K), medium (Figure 5L), and high (Figures 5M and N) magnifications of photomicrographs of histopathologic lesions from Paw #3. The majority of voters (47%) (Table 4) voted for osteolysis, osteomyelitis, and hyperostosis. The correct answer was osteolysis, osteomyelitis, hyperostosis, and dermatitis.
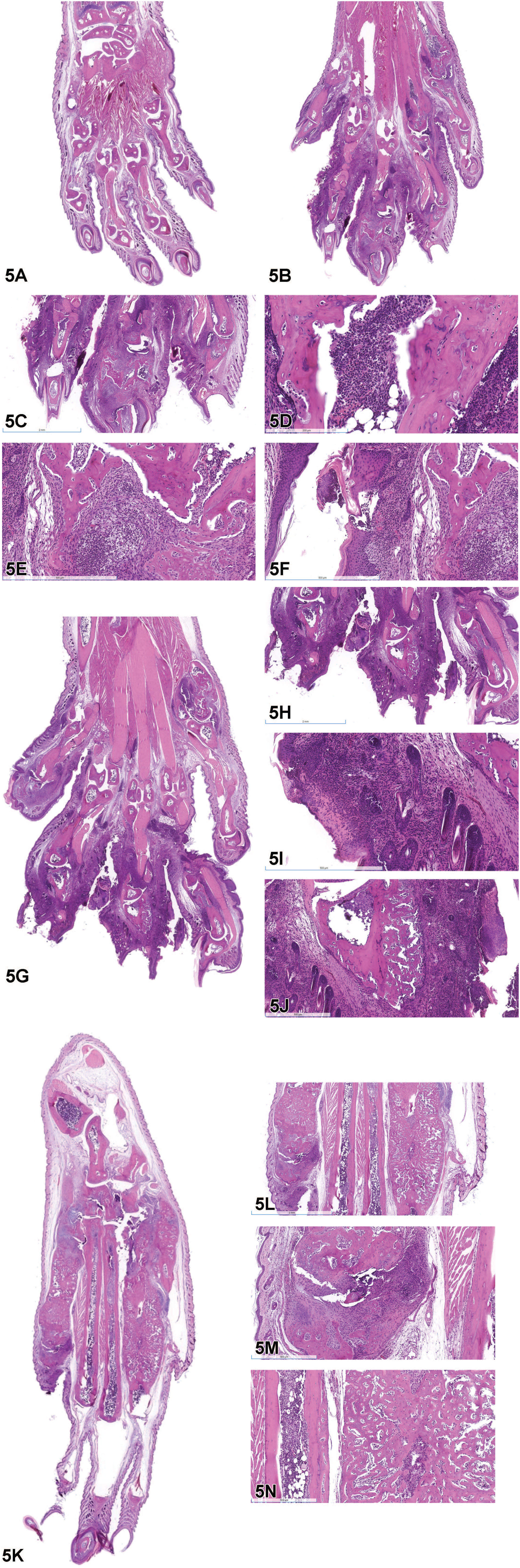
(A) Photomicrograph of a normal forepaw. (B) Hind paw from Ephx2tm1.1Dcz mouse. Note that the phalanges in Paw #1 are increased in size when compared to the normal forepaw in 5A. (C) The toes are greatly increased in size due to inflammation in the soft tissue associated with the bone. The bones of the third phalanx are markedly irregular due to bone loss and remodeling. (D) Neutrophils fill the medullary cavity replacing the hematopoietic cells and adipocytes. Neutrophils are also present adjacent to the bone. (E) Neutrophilic inflammation is associated with lysis and remodeling of the third phalanx. (F) The epidermis is hyperplastic and associated with a pustule. (G) Hind paw from Ephx2tm1.1Dcz mouse. Note that the phalanges in Paw #2 are increased in size when compared to the normal forepaw in 5A. (H) The dermis of the phalanges is markedly expanded by inflammation. (I) The dermis of the third phalanx is markedly infiltrated by neutrophils. The inflammation is associated with ulceration of the epidermis. (J) Irregular projections of woven bone extend from the surface of the phalangeal cortical bone. (K) Hind paw from Ephx2tm1.1Dcz mouse. Paw #3 is enlarged due to bone proliferation in the metatarsals. (L) The bone is proliferative and there is multifocal inflammation in the dermis. (M) Neutrophilic inflammation is associated with loss and remodeling of bone. (N) The cortical bone of the metatarsal on the right is replaced by irregular woven bone. The medullary cavity is decreased in size compared to the adjacent metatarsal bone. Neutrophilic inflammation is present within the medullary cavities.
The pathology of the hind paws in the three female Ephx2tm1.1Dcz mice included four key features: osteolysis, osteomyelitis, hyperostosis, and dermatitis. The phalanges and/ or metatarsal bones were predominantly affected. Osteolysis was characterized by loss (lysis) of bone and proliferation of new, irregularly shaped woven bone. Occasionally, osteoclasts in a Howship’s lacunae were observed at the bone margins. Osteomyelitis was characterized by abundant neutrophils in the medullary cavities and associated with the peripheral margins of the bone. Neutrophils in the medullary cavities often replaced the hematopoietic cells and adipocytes. Hyperostosis was characterized by proliferation of woven bone from the cortical bone surface or complete replacement of the cortical bone by woven bone. Hyperostosis was frequently associated with enlargement of the phalanges and/or metatarsal bones. Dermatitis was primarily neutrophilic and associated with epidermal hyperplasia, epidermal ulceration, and/or pustules.
Dr Hobbie then asked the audience to vote on a possible etiology for the bone and inflammatory lesions in the hind paws of the three female Ephx2tm1.1Dcz mice. She provided seven choices for voting that included viral, bacterial, fungal, immune-mediated, strain-related, unknown, or other. The voting results were as follows: viral (2%), bacterial (24%), fungal (2%), immune-mediated (22%), strain-related (41%), unknown (9%), and other (0%) (Table 5). Dr Hobbie explained that the cause of the paw lesions in the three female Ephx2tm1.1Dcz mice is currently unknown. A viral etiology seemed unlikely as the mice were specific pathogen-free and were free of many viruses, including the ectromelia virus, which can be associated with lesions of the extremities. Microscopic examination of H&E-stained tissue sections did not reveal the presence of bacteria or fungi. Bacterial culture and gram-stained slides of paw lesions were also negative for bacteria. The primary investigator had determined that the paw lesions were not associated with the Ephx2 gene. The paw phenotype was observed in mice that were homozygous and heterozygous for the Ephx2 mutation, as well as in wildtype Ephx2 mice. However, the paw phenotype was not observed in Ephx2 knockout mice. The laboratory is investigating a potential association of the paw lesions with a currently unidentified mutated gene.
Dr Hobbie mentioned that similar paw lesions have been observed in other mouse strains, including the TTP knock-in mouse. Tristetraprolin or TTP is a zinc finger transcription factor with anti-inflammatory activity. 36 TTP knock-in mice express mutated TTP. 27 A knocked-in point mutation is associated with loss of TTP function and subsequent increases in tumor necrosis factor (TNF) activity. 27 TNF is a proinflammatory cytokine. TTP knock-in mice have the same phenotype as TTP knock-out mice, which express complete deficiency. 27 Like the TTP knock-out mice, all TTP knock-in mice demonstrate failure to gain weight/fat loss, a severe peripheral polyarthritis, myeloid hyperplasia, and auto-immunity. 27
TTP knock-in mice displayed severe polyarthritis. 27 The paws of the mice were red and swollen and there was neutrophil accumulation in the soft tissue. There was also bone destruction as evidenced by increased osteoclastic activity seen with TRAP or tartrate resistant acid phosphatase stain and severe erosions and osteopenia observed with radiography and micro-computed tomography (micro-CT). 33 Dr Hobbie showed a photomicrograph of a paw histological section from a TTP knock-in mouse stained with TRAP, a cytochemical marker for osteoclasts. The TRAP stain showed increased osteoclastic activity demonstrated by red stippling in the histological section of the paw from the TTP knock-in mouse. Dr Hobbie also showed radiographs and micro-CT images of paws from the TTP knock-in mice demonstrating soft tissue swelling (radiograph) and bone loss or osteopenia with bony proliferation in the phalanges and metatarsals (radiograph and micro-CT). For comparison, Dr Hobbie showed a Faxitron® image and an H&E-stained slide of a hind paw from the three female Ephx2tm1.1Dcz mice. The Faxitron® image showed tissue swelling and areas of bone loss and bony proliferation involving the phalanges and metatarsals. The lesions in the Faxitron® image of the hind paw from the female Ephx2tm1.1Dcz mouse correlated well with the hind paw histopathology.
Despite the similarities in paw lesions between the Ephx2tm1.1Dcz mice and the TTP knock-in mice, Dr Hobbie pointed out differences in the mouse models. In the TTP mice, the paw phenotype is associated with knock-in or knock-out of the TTP gene, 27 whereas the paw phenotype in the Ephx2tm1.1Dcz mice is not associated with mutated or wildtype Ephx2 gene. TTP normally has anti-inflammatory activity and loss of TTP gene function is associated with increased TNF activity. 27 Ephx2 participates in cholesterol metabolism and homeostasis and mutation of the Ephx2 gene in humans is associated with familial hypercholesterolemia.17,18 All knock-in and knock-out TTP mice have the paw phenotype, whereas the paw phenotype was only observed in a subset of male and female Ephx2tm1.1Dcz mice. The paw lesions in the TTP knock-in mice involved the articular surfaces as well as bone and soft tissue. 33 The paw lesions in the Ephx2tm1.1Dcz mice involved bone and soft tissue, but generally spared the articular surfaces. There were systemic effects in the TTP knock-in and knock-out mice including cachexia, myeloid hyperplasia, splenomegaly, and auto-immunity. 27 There were no systemic effects in the Ephx2tm1.1Dcz mice. The lesions were restricted to the paws, specifically the hind paws, in the Ephx2tm1.1Dcz mice.
Dr Hobbie shared examples of comparative pathology with the audience including ulcerative pododermatitis, ectromelia virus and hypertrophic osteopathy. Ulcerative pododermatitis, also known as bumblefoot or sore hocks, is seen in companion birds, rabbits, rats, hamsters, and guinea pigs, as well as laboratory rats.4,6,7,53 It initially involves an injury, for example due to environmental conditions such as wire bottom cages. Injury is followed by secondary bacterial infection. Bacteria typically involved include Staphylococcus (eg, Staphylococcus aureus), as well as Pseudomonas or Escherichia. Ulcerative pododermatitis typically looks like an abscess-like swelling with a central, dark-colored scab. It is often seen in overweight animals, such as the diabetic rat phenotype. Ectromelia virus is in the family Poxviridae and the genus Orthopoxvirus.2,5,20,54 Ectromelia is typically found in mice used for research purposes. The natural route of infection is through skin abrasions. In the subacute form, the animals develop skin lesions. Edema, hyperemia, with small focal hemorrhages covered by dry crusts can be seen on the head, tail, and legs. Foci of necrosis covered by dark brown crusts can be seen on the fingers, ears, and tail. Eventually, there may be amputation of limbs, fingers, and/or the tail. Hypertrophic osteopathy is seen in dogs, cats, horses and humans.13,26,34,40 In humans, it is called hypertrophic osteoarthropathy. There is periosteal new bone formation and soft tissue swelling involving the limbs. These findings are usually confined to the limbs; the radius, ulna, tibia, metacarpals, and metatarsals are affected with relative sparing of the proximal limbs and phalanges. It is a paraneoplastic syndrome. In dogs, it is usually associated with primary or metastatic pulmonary neoplasia. It can also be associated with chronic inflammatory lesions in the thoracic cavity.
Dr Hobbie concluded with take-home points for the paw lesions in the three female Ephx2 9 mice. The bone and inflammatory lesions were isolated to the paws, specifically the hind paws. The cause of the paw lesions is currently unknown. A similar paw phenotype has been seen in other mouse strains. Key pathology features of the paw lesions include inflammation involving the bone and soft tissue, bone lysis and proliferation, as well as epidermal hyperplasia and ulceration. Dr Hobbie asked the audience if anyone had observed this change in another mouse strain or mutant and if anyone had insight into the disease’s pathogenesis. An audience member asked if Dr Hobbie had considered doing a periodic acid-Schiff (PAS) or Grocott’s methenamine silver (GMS) stains, as dogs with Malassezia fungi are associated with similar lesions in the paws. Dr Hobbie explained that these stains were not completed prior to the presentation. Another audience member commented on observing similar paw lesions in a cross-bred mouse strain and asked if the investigators had traced the lineage back to determine where the mutation occurred so that they could breed it out of the strain. Dr Hobbie stated that she believed the investigators managed their own colony but was unaware if the lineage had been traced. Subsequent information revealed that the mutation had not been traced back to a single breeding pair. A spontaneous mutation associated with inbreeding is suspected to be the cause of the paw phenotype in the Ephx2 9 mice.
A Hemato-Poetic Tragedy: A Case of Exuberant Erythroid Hyperplasia in a Rhesus Macaque
Dr Amanda Duggan (Anatomic pathology resident at Wake Forest University School of Medicine, Winston-Salem, NC) presented an interesting case of pancytopenia in a rhesus macaque (Macaca mulatta) featuring aspects of both clinical and anatomic pathology. The animal was part of a radiation late-effects study and was experimentally naïve. Dr Duggan would like to thank Dr David Caudell (WFU), the faculty pathologist on the case, for his guidance and mentorship, as well Drs. Nancy Kock (WFU), Mark Cline (WFU), and Gayathriy Balamayooran (WFU) for their contributions during case review. Dr Duggan would also like to acknowledge Dr Nancy Rosenthal (WFU), for her hematopathology expertise, and histotechnologists Cathy Mathis (WFU) and Lisa O’Donnell (WFU) for their assistance with histology and immunohistochemistry.
The case featured a 20-year-old male rhesus macaque that was imported from China in 2007. Antibody assays testing for simian T cell leukemia virus, simian immunodeficiency virus (SIV), simian retrovirus, and filovirus had been negative prior to transfer. The macaque was presented to the veterinary staff for a 24-hour history of lethargy and inappetence. Despite aggressive supportive care, he was found unresponsive during an evening check and pronounced deceased. The audience was shown selected red blood cell indices and platelet values from a complete blood count (CBC) taken a few hours prior to death. These values included an absolute red blood cell (RBC) count of 2.86M/uL (reference range:4.95-6.1M/uL), hematocrit of 21.6% (35-44%), hemoglobin of 7.2g/dL (11.7-14.3g/dL), mean corpuscular volume (MCV) of 76fL (67-74fL), mean corpuscular hemoglobin (MCH) of 25.2pg (18.7-26.0pg), mean corpuscular hemoglobin concentration (MCHC) of 33.3g/dL (25.7-36.9g/dL), and a nucleated red blood cell count of 202 per 100 white blood cells (WBCs). 55 Platelets were 11,000/uL (297,000-499,000). 55 The audience was informed that the CBC findings would be necessary to evaluate the upcoming blood smear and histologic sections of bone marrow.
A series of photomicrographs of a Diff-Quik stained blood smear was displayed to the audience with the CBC findings remaining within an inset. The audience was asked to evaluate all components of the blood smear but informed that the question would pertain to specific features of the red blood cells. When asked, 94% of the audience agreed that polychromasia, agglutination, and spherocytosis were present. Of those, 55% also recognized the presence of ghost cells. Eight percent suspected Auer rods were present, 2% saw only agglutination, 2% saw macroplatelets, and 2% saw only spherocytes. Dr Duggan revisited a few images to highlight the features present. She then described the abnormal red blood cell morphologies seen in this case and briefly explained how such abnormalities are formed and in what situations they are typically prompted to occur. This review also included an explanation of Auer rods, which are seen specifically in myeloid neoplasms but were not seen in this case. Dr Duggan then turned her attention to addressing the presence of dozens of immature erythroid precursors ranging from pronormoblasts to orthochromatic normoblasts (nucleated red blood cells) on the peripheral blood smear. The audience was asked to consider whether they thought these cells in circulation were part of an appropriate response or something neoplastic.
Photomicrographs of the H&E-stained sternal bone marrow at low- (Figure 6B) and high- (Figures 6C) magnification representing a densely cellular, pleomorphic cell population were shown. The audience was asked to consider these images in light of the CBC and blood smear findings and to provide a possible diagnosis. Voting choices and results regarding a possible diagnosis up to this point were as follows: polycythemia vera (6%), aplastic anemia (6%), pure red cell aplasia (4%), erythroid hyperplasia (31%), myelodysplastic syndrome (28%), erythroleukemia (22%), and pure erythroid leukemia (2%). After voting was completed, Dr Duggan admitted that additional information was necessary to discern between some of the voting choices and she proceeded to show a series of high-magnification photomicrographs of the bone marrow with additional immunohistochemical (IHC) stains. The following IHC stains were displayed (Figures 6D-: cluster of differentiation (CD)34 (blast cell marker), alpha-hemoglobin stabilizing protein (AHSP) (erythroid precursor marker), CD45 (leukocyte common antigen), and myeloperoxidase (MPO) (granulocyte marker). Bone marrow cells were negative for CD34 and showed about 50% immunoreactivity to AHSP and 50% immunoreactivity to CD45. Clusters of cells with AHSP positivity appeared negative for CD45, and vice versa. A subset of cells that stained faintly positive for CD45 showed intense immunoreactivity for MPO. With this additional information, Dr Duggan then posed the previous question again to the audience. Unfortunately, the voting application malfunctioned and failed to record this round of answers. Dr Duggan assured the audience that she was confident they had all changed their answers to reflect the most reasonable choice, erythroid hyperplasia.
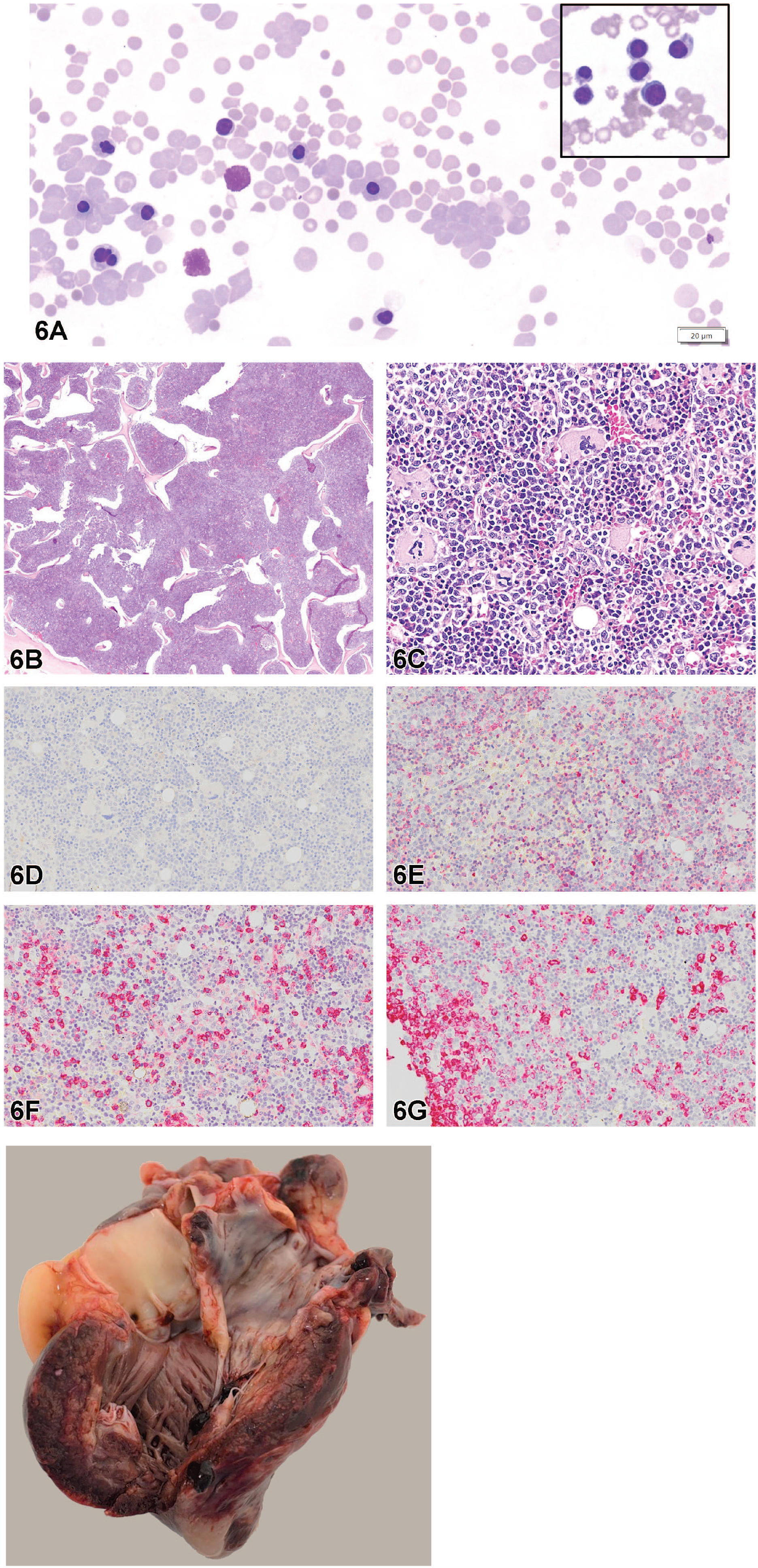
(A) Diff-Quik stained peripheral blood smear taken prior to death showing red blood cell agglutination, numerous nucleated red blood cells, polychromatophils, and spherocytes. The inset is a pronormoblast and several polychromatophilic normoblasts taken from a separate area. (B) A subgross H&E-stained section of sternal bone marrow showing diffuse hypercellularity. (C) H&E-stained section of sternal bone marrow showing hypercellular pleomorphic cell population consisting of all three hematopoietic lineages. (D-G) Immunohistochemistry from sternal bone marrow. (D) Section showing negative CD34 staining. Inset: Positive internal control (endothelial cells). (E) Section showing ~50% cytoplasmic positivity for AHSP staining. (F) Section showing ~50% light and intense cytoplasmic positivity for CD45 IHC staining. (G) Section showing positive IHC staining for MPO. (H) Gross image of the heart with acute myocardial hemorrhage and necrosis.
Taken together the CBC, blood smear findings, and histology with additional IHC best supported the presence of erythroid hyperplasia. Several immunohistochemical stains for erythroid precursors were available, including glycophorin A (CD235a) and CD71, however AHSP 37 was chosen as it had been previously validated for use by the comparative histology lab at WFU in rhesus macaques. An additional section of bone marrow was submitted to the human pathology lab at Atrium Wake Forest Baptist Health Hospital for CD71 staining and it displayed similar results to the AHSP IHC stain. The erythroid component of the bone marrow as displayed by AHSP staining was increased at ~50% when compared normal bone marrow. Normal bone marrow usually has an erythroid component between 15% and 20%. 10 During times of severe, sustained hypoxia, the erythroid component of the bone marrow can increase 5-7x basal levels. 21 This change was appreciated histologically by hyperplasia of bone marrow cells with large coalescing islands of erythroid precursors.
Each of the answer choices represented conditions that featured some component of red blood cell abnormality. These conditions were briefly discussed and explanations were provided as to why they were eliminated as possible causes. Polycythemia vera (PV), a neoplastic proliferation of erythroid progenitors, often shows increased bone marrow cellularity owing to an increase in the erythroid component. 43 Cases of polycythemia vera have an increase in the circulating red blood cell mass which appears diagnostically on a CBC as abnormally high hemoglobin and hematocrit values. 21 This condition was deemed unlikely based on the moderate anemia present on this animal’s CBC and other parameters denoting an overall low red blood cell mass. Next considered was aplastic anemia, which is a chronic condition of primary hematopoietic failure resulting in pancytopenia. At this point, the audience was aware that this case had an anemia and thrombocytopenia, however Dr Duggan would later share that this case also had a severe neutropenia. Bone marrow hyperplasia with circulating red blood cell precursors suggested that this bone marrow was still functional. Red blood cell aplasia, a condition in which only erythropoiesis fails, was also eliminated as a possibility based on the presence of erythroid precursors within bone marrow as demonstrated by AHSP IHC staining.
The remaining conditions of myelodysplastic syndrome, erythroleukemia, and pure erythroid leukemia, were similar in that they all may have appeared as a pancytopenia on CBC with a potential increase in circulation of “abnormal” cells. In the case of myelodysplastic syndrome (MDS), this disease of clonal expansion of hematopoietic cells displays >10% dysplasia within a single cell line. Dysplastic features of red blood cells, more formally known as dyserythropoietic changes, include the formation of megaloblasts, increased presence of nuclear budding, ring sideroblasts, and presence of large cells that contain multiple condensed nuclei (gigantoblasts). Examples of these changes were shown to the audience. Dysplasia was not a feature seen in this case within circulating cells or within the bone marrow. Furthermore, ring sideroblasts, red blood cell precursors with abnormal accumulations of iron, were not present on Prussian blue histochemical staining. When the discussion turned to erythroid leukemia, Dr Duggan admitted that this was not the current terminology for this entity. The 5th edition of the World Health Organization Classification of Hematolymphoid Tumors published in 2022 considered erythroid leukemia as a form of acute myeloid leukemia (AML), the presence of which is defined by >20% of blasts within the bone marrow. Negative CD34+ staining (Figure 6D) in conjunction with lack of dysplasia were used to refute the presence of AML. The final answer choice, pure erythroid leukemia, has retained its distinction from AML and is defined as a neoplastic proliferation of erythroblasts. The presence of this condition is defined by a bone marrow in which >80% of nucleated cells are erythroblasts. 29 The audience was shown a representative photomicrograph in which the bone marrow was almost entirely effaced by a monomorphic population of blast cells. In this case, the presence of a pleomorphic cell population visible on H&E and further confirmed by CD45, MPO, and AHSP IHC (Figures 6E-G) staining eliminated the possible presence of pure erythroid leukemia.
For discussion, Dr Duggan provided additional case details, including gross images of the heart (Figure 6H) showing acute myocardial hemorrhage and necrosis confirmed by histology as well as photomicrographs of the liver showing centrilobular hepatocellular necrosis. The cause of death for this animal was determined to be acute myocardial degeneration and hemorrhage secondary to severe thrombocytopenia, however the underlying etiology remained unknown. Dr Duggan went on to summarize potential causes of severe thrombocytopenia in nonhuman primates including drug-induced, neoplasia, infectious agents (Plasmodium spp., Babesia spp.), and immune-mediated. Recent drug administration to the animal had included meloxicam and omeprazole and there are reports within human literature of drug-induced thrombocytopenia secondary to meloxicam. 1 Immune-mediated thrombocytopenia with some concurrent targeting of red blood cells was supported by the presence of agglutination, spherocytosis, and ghost cells. The extent of the erythroid hyperplasia also remained a conundrum as it suggested a subacute nature to the clinical picture. Erythroid hyperplasia may have occurred in response to direct red blood cell destruction and subsequent hypoxia. Consideration was also given to the possible interplay involving megakaryocyte-erythroid progenitor (MEP) cells which are responsible for differentiation into megakaryocytes and red blood cells. Thrombopoietin, a hormone responsible for regulating platelet production, has been shown to stimulate hematopoiesis. 35 Dr Duggan lamented that at this time, the cause of this animal’s demise remained unknown.
Dr Duggan concluded by reviewing valuable ways to distinguish between neoplastic and nonneoplastic bone marrow conditions. In this case, CD34 IHC, Prussian blue histochemical staining, and lack of dysplasia were important in making that determination. Dr Duggan also added that a bone marrow cytology would have been beneficial, especially during consultation with a human hematopathologist. In people, most diagnoses are made antemortem using bone marrow biopsies and individual cell morphology is better evaluated on cytology. Flow cytometry, instead of IHC, would have also been an appropriate way to differentiate between types of cells present, however fresh tissue was no longer available when this option was considered. Finally, Dr Duggan reiterated that the intense erythroid hyperplasia was likely an appropriate response of the bone marrow in the face of hypoxia, and that such appearances of erythroid precursors on peripheral blood smears should not be assumed to be neoplastic in origin.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported [in part] by the NIH, National Institute of Environmental Health Sciences.
