Abstract
Toxicology studies in nonhuman primates were conducted to evaluate selective, brain penetrant inhibitors of LRRK2. GNE 7915 was limited to 7-day administration in cynomolgus monkeys at 65 mg/kg/day or limited to 14 days in rhesus at 22.5 mg/kg b.i.d. due to physical signs. Compound 25 demonstrated acceptable tolerability at 50 and 225 mg/kg b.i.d. for 7 days in rhesus monkeys. MK-1468 was tolerated during 7-day administration at 100, 200 or 800 mg/kg/day or for 30-day administration at 30, 100, or 500 mg/kg b.i.d. in rhesus monkeys. The lungs revealed hypertrophy of type 2 pneumocytes, with accumulation of intra-alveolar macrophages. Transmission electron microscopy confirmed increased lamellar structures within hypertrophic type 2 pneumocytes. Hypertrophy and hyperplasia of type 2 pneumocytes with accumulation of intra-alveolar macrophages admixed with neutrophils were prominent at peripheral lungs of animals receiving compound 25 or MK-1468. Affected type 2 pneumocytes were immuno-positive for pro-surfactant C, but negative for CD11c, a marker for intra-alveolar macrophages. Accumulation of collagen within alveolar walls, confirmed by histochemical trichrome stain, accompanied changes described for compound 25 and MK-1468. Following a 12-week treatment-free interval, animals previously receiving MK-1468 for 30 days exhibited remodeling of alveolar structure and interstitial components that did not demonstrate reversibility.
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disease for which no disease-modifying interventions exist, while current therapies primarily address symptomatic features. Genome-wide association studies and recent epidemiologic studies implicate both genetic components and environmental factors related to the incidence of PD. Activating mutations of leucine-rich repeat kinase 2 (LRRK2) have been identified, which are associated with dominantly inherited PD, while variants of LRRK2 have also been identified in sporadic forms of PD.10,28,42,43 Current interest in the biology of LRRK2 has prompted development of LRRK2 inhibitors as potential treatment for PD,3,8,31,52 particularly given that kinase activity of LRRK2 is increased in microglia and dopaminergic neurons of the substantia nigra of Parkinson’s patients with sporadic (idiopathic) disease.16,41,43
LRRK2 is a 286 kDa protein comprising 2527 amino acids, which structurally has multiple scaffold domains and distinct catalytic domains (Figure 1). The central catalytic core of LRRK2 contains a Ras of Complex (Roc) GTPase domain adjacent to the C-terminus of Roc (COR), followed by a kinase domain. The central GTPase domain (Roc) and its C-terminus (COR) are the structural basis for classification as a Roco protein. 50 This central catalytic core is surrounded by the N-terminal protein domains named armadillo (ARM), ankyrin (ARK), and leucine-rich repeat (LRR) domains, and a C-terminal WD40 domain, scaffold domains that mediate protein to protein interactions. The GTPase Roc domain also serves as a molecular switch controlling LRRK2 monomer-to-dimer equilibrium mediated through the COR domain and serves as an interface for interactions with other binding partners.15,21,45

Linear structure of LRRK2 monomer. ARM indicates armadillo; ANK indicates ankyrin; COR, C-terminus of Roc; KIN indicates kinase; LRR, leucine-rich repeat; ROC, Ras of complex.
LRRK2 facilitates a wide range of protein interactions and exhibits a relatively wide tissue expression in mammals (Figure 2). Physiological activities of LRRK2 are implicated in numerous cellular functions related to vesicular activity (“traffickin,”) and processes related to inflammation, although LRRK2 likely exhibits distinct roles in various cell types. Transport of cytoplasmic vesicles plays a major role in formation and function of lamellar structures in type 2 pneumocytes of the lungs.33,44,48 Loss of LRKK2 expression in transgenic rodent models have demonstrated changes in morphology and exocytosis of lamellar structures in the type 2 pneumocytes.24,34
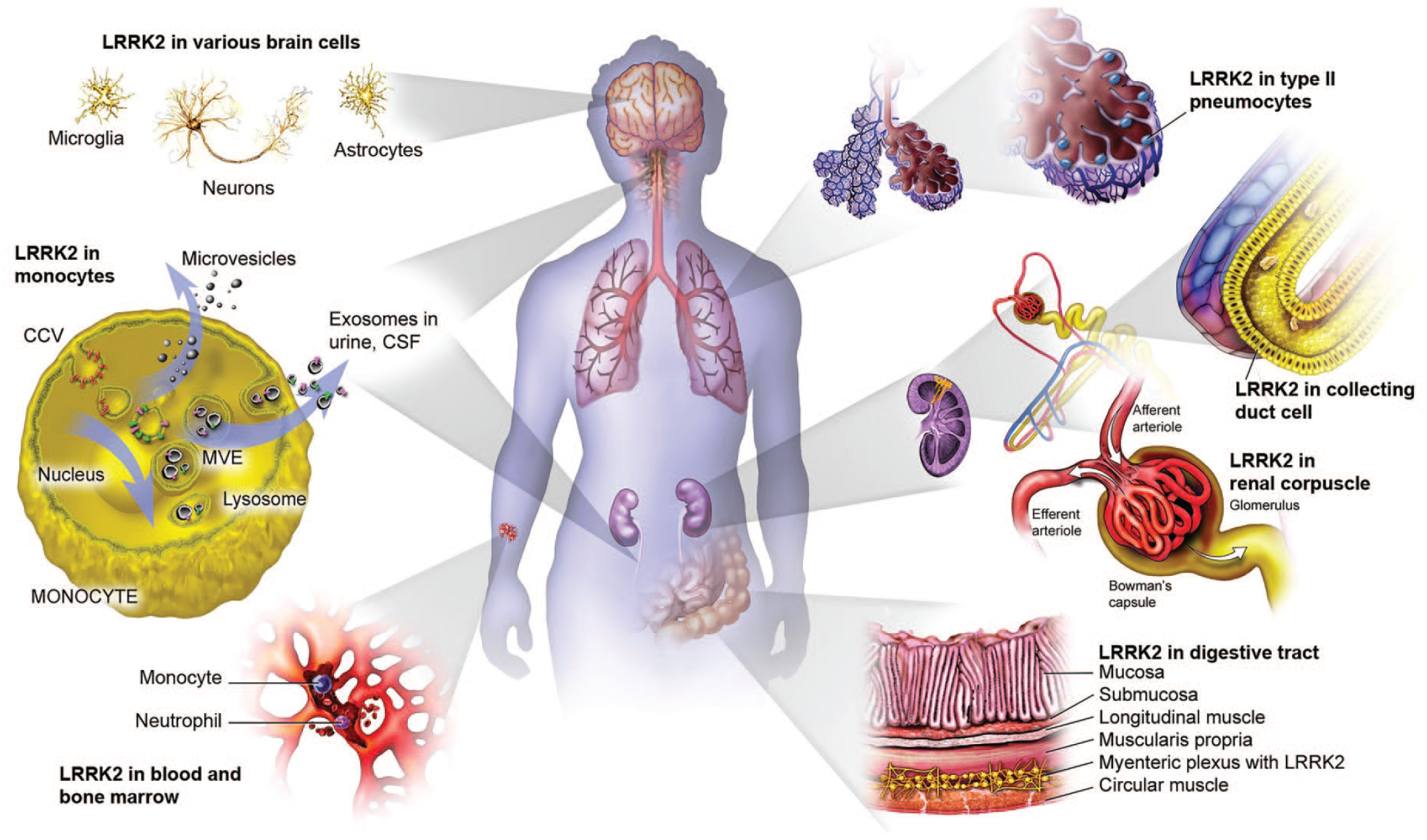
Distribution and tissue expression of LRRK2. CSF indicates cerebral spinal fluid. CCV indicates clathrin coated vesicles; MVE indicates multivesicular endosomes.
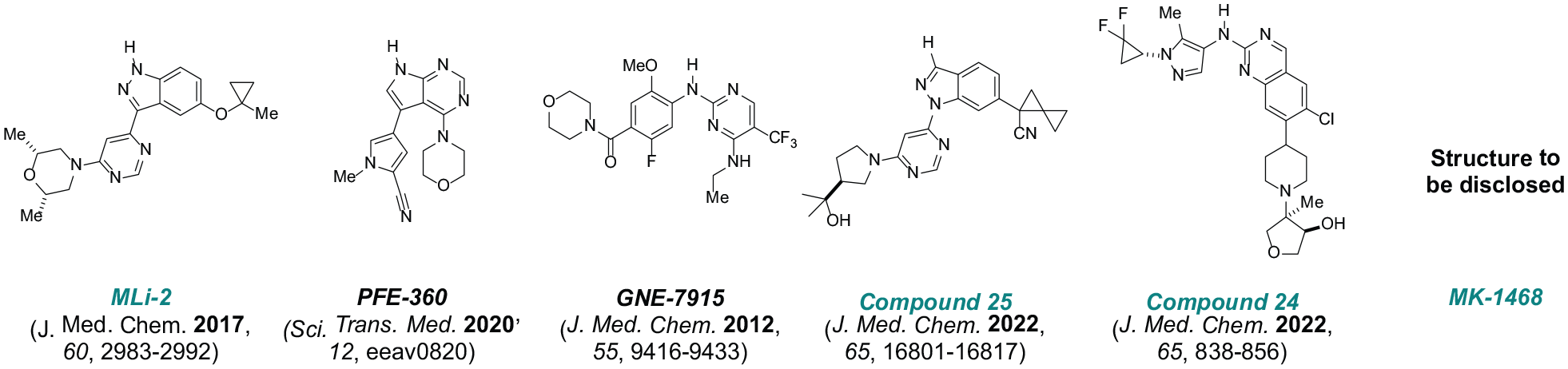
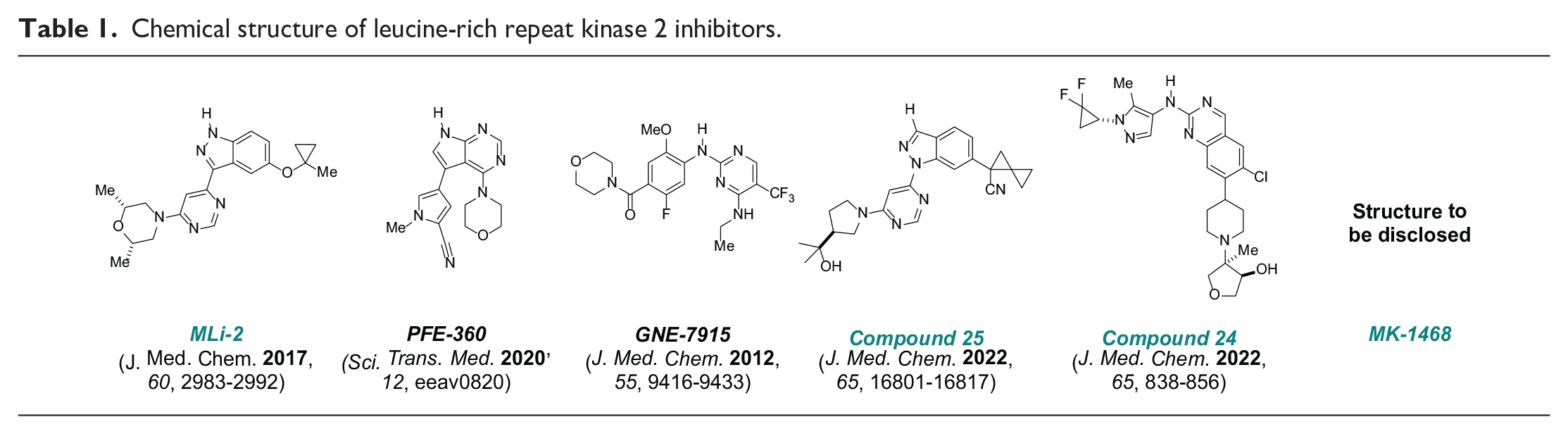
The key objective of an internal discovery program is to identify type I (ATP-competitive) LRRK2 inhibitors, leveraging high-throughput screening and knowledge-based approaches to select novel, tractable chemical series with enhanced levels of kinome selectivity and physiochemical properties facilitating central nervous system (CNS) penetration. 14 Subsequent disclosure of structurally distinct, potent, and selective molecules (Table 1) includes a widely adopted tool compound, MLi-2, evaluated in comparison with PFE360 and GNE7915 sponsored by the Michael J. Fox Foundation LRRK2 Safety Initiative.5,14,17,27,47 The results of recent studies evaluating GNE 7915, compound 25 (previously disclosed as MLi-030), and MK-1468 are presented herein.
Chemical structure of leucine-rich repeat kinase 2 inhibitors.
Early studies with LRRK2 inhibitors describe hypertrophy of type 2 pneumocytes within alveolar walls of the distal lung, or cytoplasmic vacuolation of type 2 pneumocytes, in the lungs of mice or nonhuman primates, features that initially were judged to be reversible and without effect on respiratory function.5,13,18,20 Results of subsequent repeat dose studies in nonhuman primates (rhesus or cynomolgus monkeys) are presented following administration of structurally diverse, selective, and potent inhibitors of LRRK2. The design of these studies involves comprehensive sampling and evaluations of the lungs to ascertain the scope and distribution of potential pulmonary pathology.
Materials and Methods
Test Articles and Formulation
GNE 7915 and MK-1468 were formulated for oral dosing in 10% (w/w) polysorbate 80 in deionized water. The oral dosing formulation for compound 25 was prepared as an amorphous dispersion comprised 33% base form and 67% hydroxypropyl methylcellulose in vehicle comprised 0.5% (w/v) methylcellulose/0.25% sodium lauryl sulfate in deionized water.
Animals and Study Management
Cynomolgus monkeys (Macaca fascicularis), 3 to 12 years of age or rhesus monkeys (Macaca mulatta), 1 to 3 years of age weighing between 2 and 7.5 kg at initiation of dosing were obtained from purpose-bred sources maintained by Merck & Co., Inc., Rahway, NJ, USA. The studies were conducted in facilities of Merck & Co., Inc., Rahway, NJ, USA accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC International). All procedures performed for these studies were approved by the Institutional Animal Care and Use Committee. The Purina Mills International (PMI)-Certified Primate Diet and daily food enrichments were provided, and water was available ad libitum.
Experimental Design
Exploratory repeat dose toxicology studies were conducted with the following test articles, GNE 7915, compound 25, and MK-1468 for a duration of 7 to 15 days in rhesus or cynomolgus monkeys (1-4 animals per dose group). Animals were orally dosed daily with vehicle control or one of the test articles at doses ranging between 30 and 1000 mg/kg/day total dose (see Summary in Table 2).
Experimental designs.
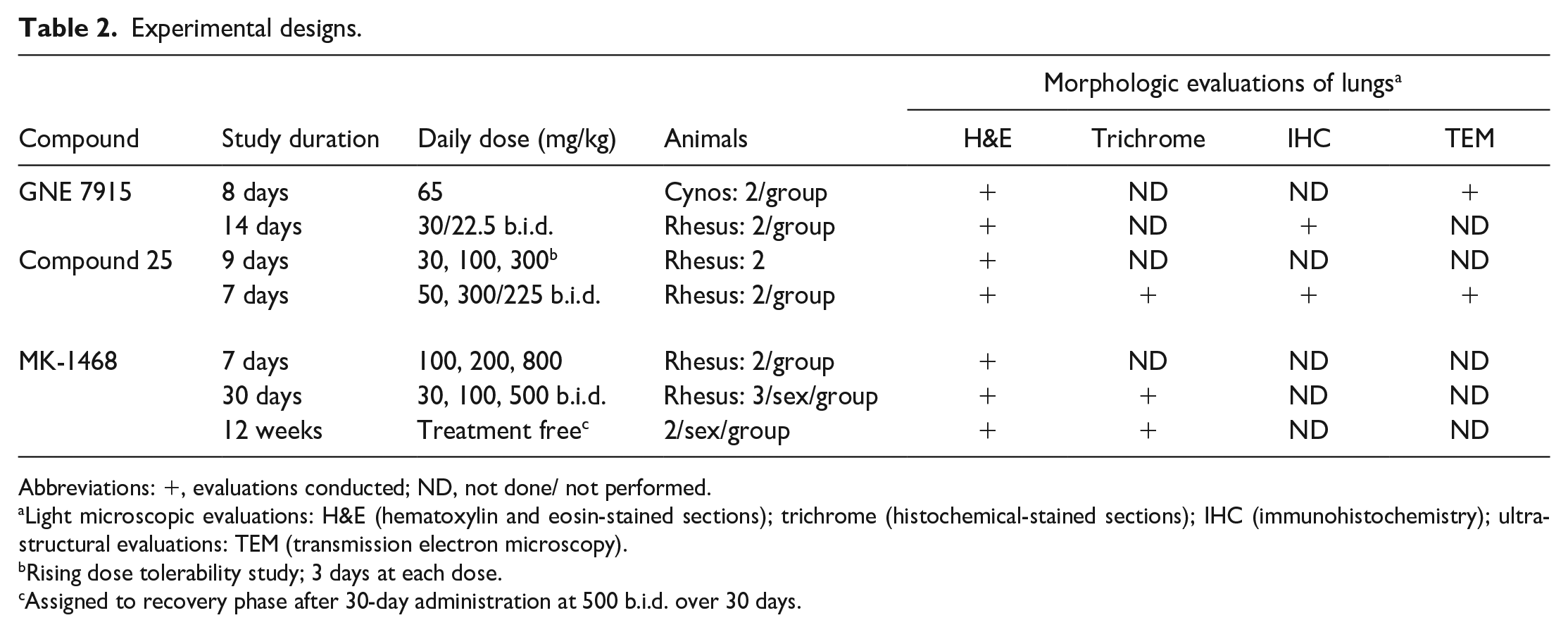
Abbreviations: +, evaluations conducted; ND, not done/ not performed.
Light microscopic evaluations: H&E (hematoxylin and eosin-stained sections); trichrome (histochemical-stained sections); IHC (immunohistochemistry); ultrastructural evaluations: TEM (transmission electron microscopy).
Rising dose tolerability study; 3 days at each dose.
Assigned to recovery phase after 30-day administration at 500 b.i.d. over 30 days.
A 1-month repeat dose toxicology study was conducted in rhesus monkeys (5/sex/dose group) receiving oral administration of either vehicle or MK-1468 at 30, 100, or 500 mg/kg bid. Two animals/sex at completion of the dosing phase after having received 500 mg/kg b.i.d. were assigned to a 12-week treatment-free interval (see summary in Table 2).
The vehicle or test article formulations were orally administered at a dose volume of 5 mL/kg in the exploratory and one-month repeat dose toxicology studies. Antemortem observations and measurements were conducted, including periodic cage side observations for any clinical signs, measurement of body weights, and estimation of food consumption. Quantitative assessments of respiratory function were not included for any of the studies. Additional parameters related to electrocardiography, and ophthalmic evaluations were incorporated for specific studies. Scheduled necropsy was conducted at the end of the dosing phase of each study and at completion of a 12-week treatment-free interval for 2/sex from the 500 mg/kg b.i.d. group previously receiving MK-1468 for 1 month.
Clinical Pathology
All animals were fasted overnight, followed by blood collection from femoral artery or other suitable sites. Peripheral blood was collected in ethylenediaminetetraacetic acid (EDTA) tubes for hematology assessment, or in sodium citrate tubes for coagulation assessment, and in serum separator tubes for determination of biochemistry parameters.
Postmortem Procedures
At study termination, cerebrospinal fluid obtained by aseptic technique from the foramen magna and fresh specimens of brain from prefrontal cortex or from temporal cortex were collected and frozen for analyses of test article concentration. A comprehensive series of tissues were obtained for each monkey at study termination and fixed in 10% neutral-buffered formalin (NBF). The intact lungs were gently infiltrated with 10% NBF via the trachea to expand the lungs followed by immersion of the lungs in 10% NBF for a minimum of 24 hours. Collection of lung specimens for ultrastructural evaluations is explained for procedures involving transmission electron microscopy.
The extent and orientation applicable to evaluations of lungs related to these current studies substantially exceed sampling procedures typically conducted for routine evaluations of lung by light microscopy. The initial study conducted with GNE 7915 over 8 days, and the rising dose tolerability study with compound 25, employ sampling of the lungs representing cross-sections of the hilar regions and cross-sections of the distal regions from each lobe of both lungs. Given the nature and distribution of changes in the lungs, the extent of pulmonary tissue evaluated for subsequent studies utilizes approximately 5 mm thick, full plane-fixed specimens oriented parallel to the respective main stem bronchus and thereby incorporating the hilar region contiguous with peripheral and distal margins for each of the 6 major lobes (Figure 3).
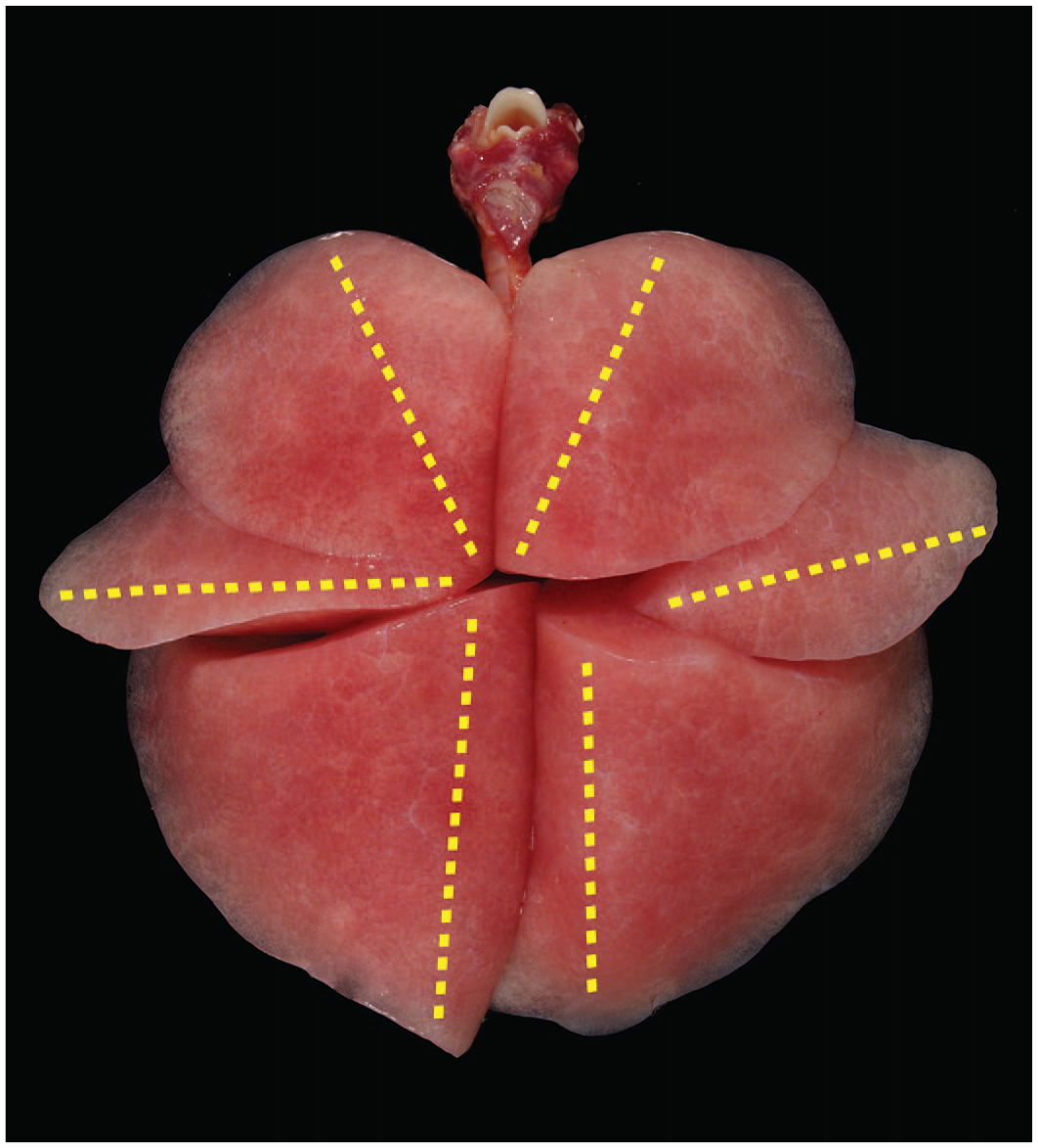
Image of primate lungs immediately following placement of fixative demonstrating plane of trim for each major lobe as shown in yellow font. Orientation of trim captures the main stem bronchus with associated vasculature inclusive of branching airways, terminal bronchioles, and distal alveolar structure for each of 6 lobes of the lungs. Hemotoxylin and eosin-stained sections for each lobe were placed on large format slides for light microscopic evaluation.
Following processing, each full plane specimen of lung was embedded in paraffin using mega-cassettes (Cell-Path) and 5-µm thick sections were obtained and stained with hematoxylin and eosin. This approach afforded optimal quality tissues for histomorphologic evaluation via light microscopy of each individual lobe of both lungs with appropriate orientation and presentation of major and secondary airways, preservation of associated pulmonary vasculature, and comprehensive representation of distal bronchioles, terminal alveolar structure, and peripheral regions of the lungs, including pleural surfaces.
Ultrastructural Evaluations of Lung by Transmission Electron Microscopy
Lungs for ultrastructural evaluations were collected either by direct placement of 4% paraformaldehyde and 1% glutaraldehyde in 0.1 M phosphate buffer (4F:1G) into the main stem bronchus of the left cranial lobe, or by placement of 10% neutral-buffered formalin via the trachea and subsequent transfer of selected specimens of lung into 4F:1G fixative. Approximately, 1 mm3 lung specimens were post-fixed in 2% osmium tetroxide, then processed and infiltrated with epoxy resin on a Lynx II (Electron Microscopy Sciences) automated tissue processor. Specimens were subsequently embedded in LX-112 epoxy resin (LADD Research Industries) and ultrathin sections were obtained on a Leica EM UC6 ultramicrotome. The sections were stained with 2% uranyl acetate and Reynold’s lead citrate and examined on a 120 kV FEI Tecnai Spirit Biotwin transmission electron microscope (Thermo Fisher Scientific, Inc.). Images were obtained using the Gatan Orius SC1000 Digital CCD camera (Gatan, Inc.) integrated with the transmission electron microscope.
Immunohistochemistry and Histochemistry
Five micrometer thick formalin-fixed, paraffin-embedded sections of lung tissue were subjected to heat-induced epitope retrieval prior to staining with the following primary antibodies: rabbit polyclonal antibody to surfactant protein B at 1:400 dilution (LifeSpan Biosciences, Inc. catalog no. LS-B8081), rabbit polyclonal antibody to Pre-ProSurfactant C at 1:2000 dilution (Abcam, catalog no. ab90716) or mouse monoclonal antibody to CD11c at 1:100 dilution (Novocastra, catalog no. NCL-L-CD11c-563). The immunoreactivity of lung sections with the primary antibodies was visualized using respective biotinylated secondary antibodies (donkey anti-rabbit or donkey anti-mouse) and avidin-biotin complex with 3,3′-Diaminobenzidine (DAB; Dako K3468) chromogen followed by counter staining with Gill’s hematoxylin no. 1 (Ricca 3535).
Parallel sections (3-5 µm thickness) of representative lung were obtained from paraffin-embedded tissue. Lung sections were subsequently stained with modified Masson’s trichrome (with Goldner-Foot modification) to characterize distribution and prominence of collagen staining within the lungs.
Postmortem Evaluations
Histomorphologic evaluations by light microscopy, ultrastructural evaluations via transmission electron microscopy, and evaluations of immunohistochemistry and histochemistry by light microscopy were conducted by board-certified pathologists. Independent contemporaneous peer review was also conducted by experienced board-certified pathologists for each study. Consultations among pathologists were utilized to compare findings, facilitate consistent application of terminology across various studies, and during review of tissues from internal historical control animals.
H&E-stained sections of lung obtained from cynomolgus monkeys receiving MLi-2 at 15 or 50 mg/kg/day for 15 days conducted under Protocol FY15-009, sponsored by the Michael J. Fox Foundation LRRK2 Safety Initiative, were provided for ad-hoc review but results thereof are not included.
Sampling for Toxicokinetics
At predetermined timepoints, whole blood (350-500 μL per sample) was collected from the saphenous vein (or femoral artery/vein) of male and female monkeys into EDTA-treated tubes and processed to plasma for analysis. Approximately, 5 to 6 blood samples were serially collected from each animal on each toxicokinetic day. Plasma samples were stored at ≤ −10°C until analyzed. Plasma analyte concentrations were determined using liquid chromatography/tandem mass spectrometry methods for each test article.
Toxicokinetic Calculations
Toxicokinetic parameters (ie, AUC0-24 h, and trough [C24h]) were calculated with noncompartmental methods using Phoenix WinNonlin (Certara, Princeton, NJ). The area under the plasma concentration versus time curve from the time of dosing to the time of the last observation (AUC0-24h) was calculated as a measure of systemic exposure using the linear trapezoidal rule. After repeated doses, plasma test article concentrations at time zero were assigned a value equal to the minimum observed plasma concentrations during the dose interval for each animal. The plasma analyte concentration at 24 hours (C24h) post first daily dose was assessed as the trough concentration. Unbound plasma AUC0-24h and C24h values were calculated from total values using free fractions of 0.053, 0.0055, and 0.124, respectively for GNE 7915, compound 25, and MK-1468. Mean and values for toxicokinetic parameters were calculated from the individual values, as appropriate. Due to apparent lack of sex differences in plasma exposures for each given test article, the toxicokinetic data were sex-combined for subsequent analyses.
Results
The results of light microscopic findings and ultrastructural findings in the lungs are provided for each compound in the following narratives and summarized in tabular format (Table 3). More detailed descriptions of findings for each compound are presented in the following paragraphs.
Incidence of light microscopic findings in lungs.
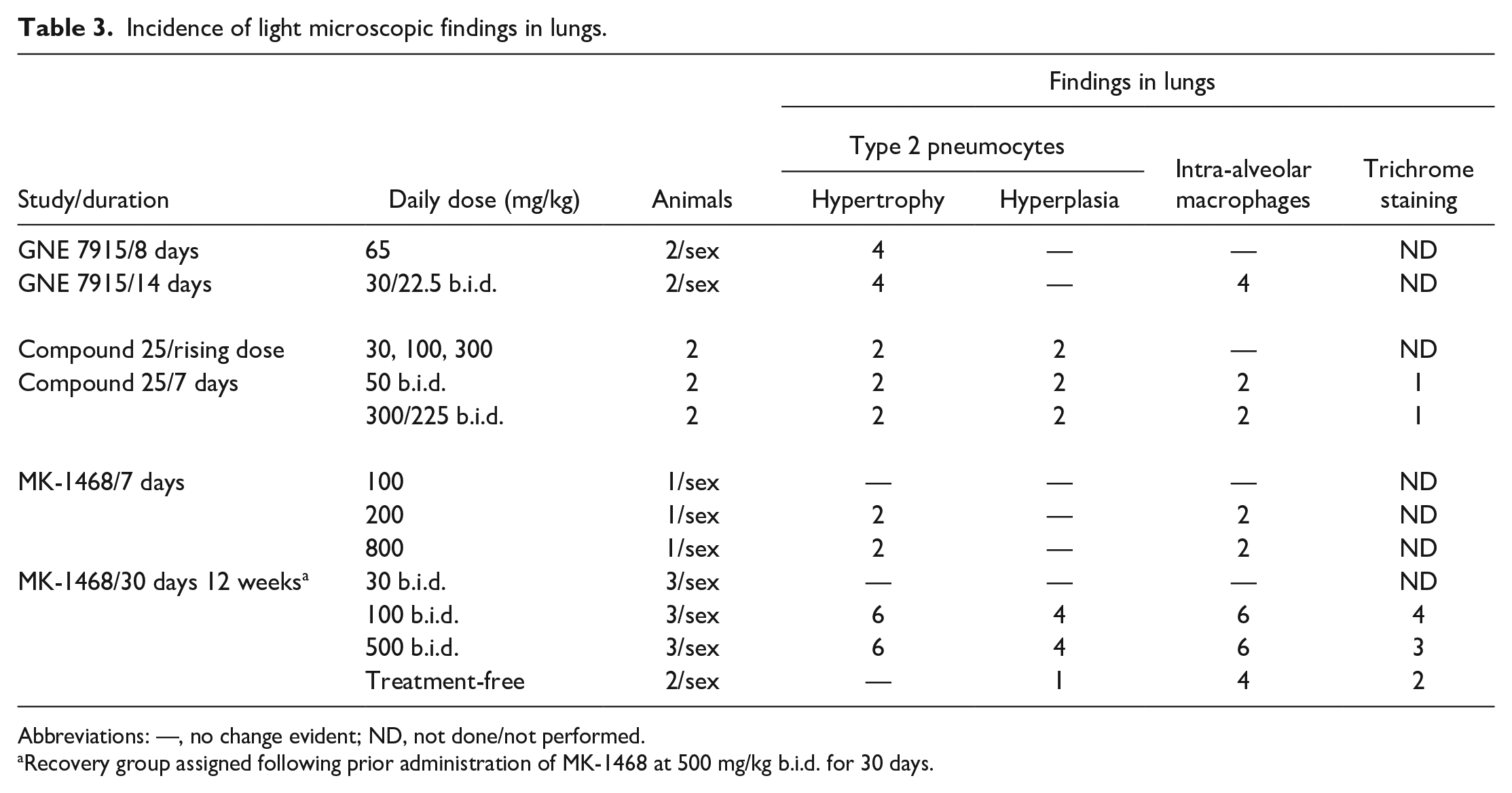
Abbreviations: —, no change evident; ND, not done/not performed.
Recovery group assigned following prior administration of MK-1468 at 500 mg/kg b.i.d. for 30 days.
GNE 7915
Daily oral administration of GNE 7915 was limited to 65 mg/kg/day over 8 days in cynomolgus monkeys due to physical signs of somnolence, reduced activity, trembling motions, atypical chewing motions, emesis, and/or reduced food consumption. These signs were exhibited by treated monkeys over the study duration. One cynomolgus monkey was humanely terminated early on study day 6 due to more prominent physical signs. Slight decrements in erythron parameters (red blood cell count, hematocrit, hemoglobin), lower white blood cell counts, and minimally increased alanine transaminase (ALT), aspartate transaminase (AST), and total bilirubin were noted among treated monkeys on study day 8. Mean systemic exposures to GNE 7915 as measured by AUC(0-24 h) were 44.7 µM•hours on day 1 or 121 µM•hours at study day 7, while mean concentration of GNE 7915 in cerebral spinal fluid (CSF) reached 0.46 µM at 4 hours post-dose on study day 8. Levels of GNE 7915 in CSF reached 0.45 µM for the early termination animal on study day 6.
The lungs exhibited hypertrophy of type 2 pneumocytes, characterized by distension of their cytoplasm due to numerous, poorly circumscribed vacuoles evident by light microscopy. Hypertrophy of type 2 pneumocytes, evident within hilar and peripheral regions of the lungs, was present by light microscopy in all lobes. Hypertrophy was attributed to increased prominence of secretory vesicles, as confirmed by ultrastructural evaluations demonstrating increased size and number of lamellar structures consistent with secretory granules containing phospholipid rich material (surfactant) within hypertrophic type 2 pneumocytes (Figure 4A and B). These features in the lungs were noted for each of 4 treated animals.
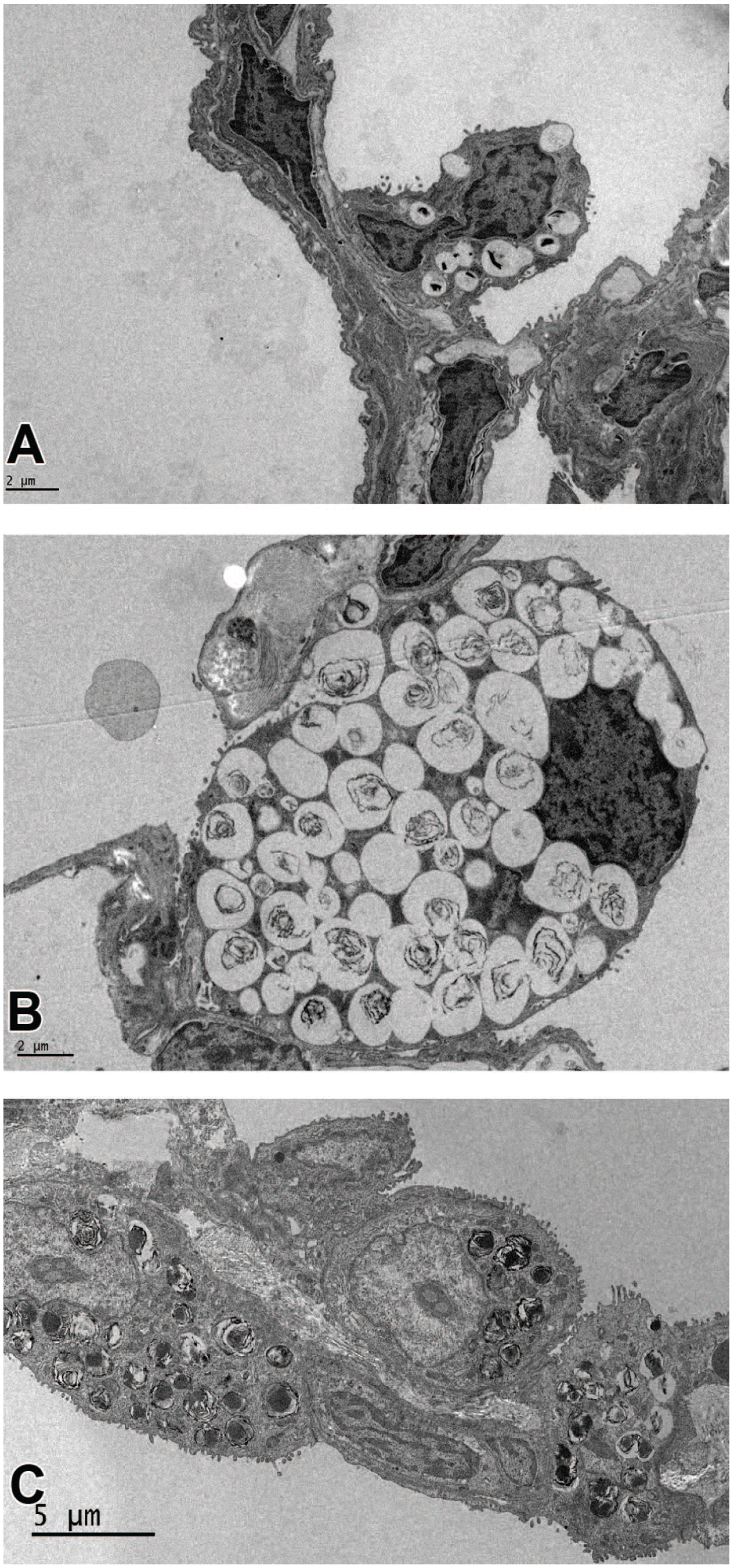
(A) Ultrastructural features of the alveolar wall of a control animal illustrating normal features of the type 2 pneumocyte projecting toward the alveolar lumen and positioned adjacent to type 1 pneumocytes and alveolar vasculature. Several but few lamellar structures are present within the type 2 pneumocyte. (B) Hypertrophy of a type 2 pneumocyte is associated with greatly increased number of lamellar structures that fill the cytoplasm following 8-day administration of GNE 7915. (C) Adjacent hypertrophic type 2 pneumocytes lining the alveolar wall following 7-day oral administration of compound 25, exhibit variation in numbers of lamellar structures within type 2 pneumocytes, consistent with findings noted by light microscopy.
Daily oral administration of GNE 7915 was limited to 22.5 mg/kg b.i.d., over 14 days in rhesus monkeys, due to physical signs similar to those described in cynomolgus monkeys (see above) and due to body weight loss during study week 2 ranging between −0.1 and −0.4 kg for individual animals. Unsatisfactory food consumption was generally observed. Slight to moderate decrements in erythron parameters (red blood cell count, reticulocytes, hematocrit, hemoglobin, and mean corpuscular volume) were noted. Mean systemic exposure to parent compound as measured by AUC(0-24h) was 88.7 µM•hours on study day 13. Levels of pSer935 in peripheral blood mononuclear cells (PBMCs) as a measure of pharmacodynamic target engagement on study day 14 at 24 hours post-dose were 37% of baseline values. Similarly, pSer935 levels in the lungs of treated animals on day 15 after receiving 22.5 mg/kg b.i.d. for 14 days, were 27.9% of levels measured in control lung.
Hypertrophy of type 2 pneumocytes had multifocal distribution within each lobe of both lungs of each treated animal, variably accompanied by increased numbers of macrophages within alveolar spaces (Figures 5-7). Although hypertrophy of type 2 pneumocytes was widely distributed in each lobe, focal regions at peripheral locations exhibited more prominent changes. Hypertrophy of type 2 pneumocytes was variably associated with intra-alveolar accumulations of both neutrophils and macrophages. Hypertrophic type 2 pneumocytes stained immuno-positive for pro-surfactant protein and immuno-positive for surfactant protein B, while staining by surfactant protein B was also evident in alveolar macrophages. Intra-alveolar macrophages stained immuno-positive for CD11c and exhibited distinct membrane staining, whereas alveolar epithelium, both type 1 and type 2 pneumocytes, were immuno-negative.
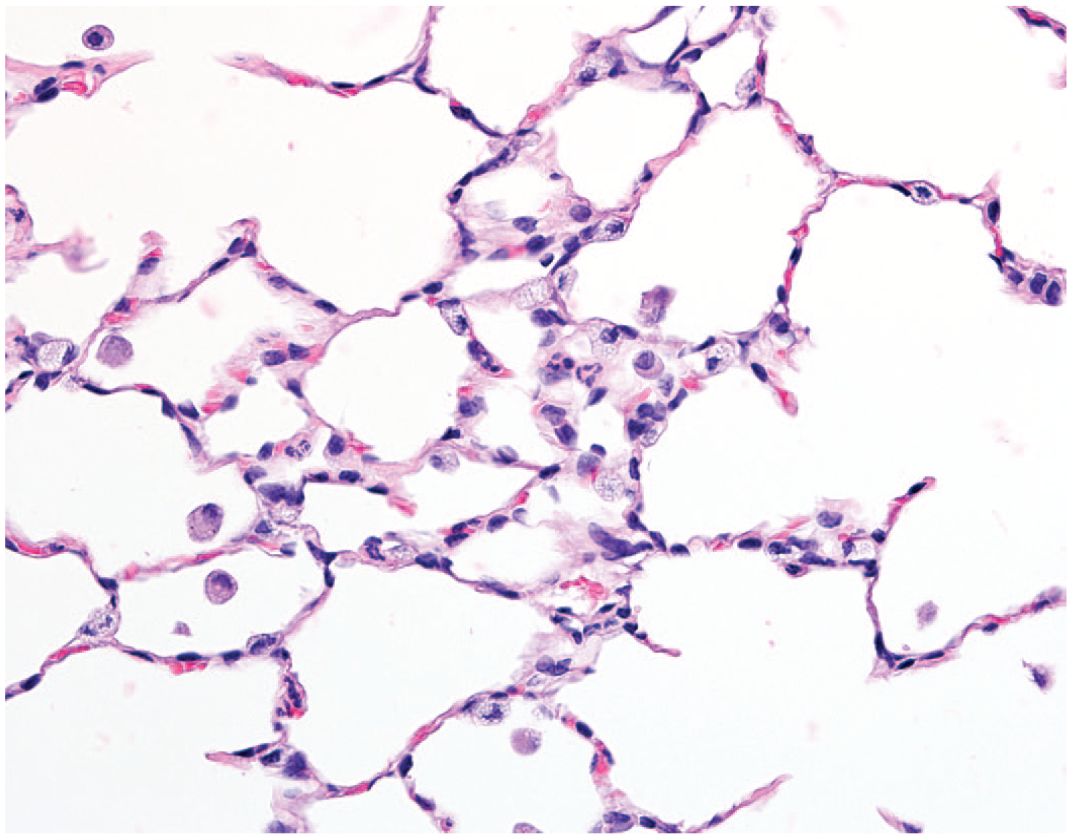
The distal lung exhibits hypertrophy of type 2 pneumocytes associated with occasional intra-alveolar macrophages or neutrophils following 14-day administration of GNE 7915 at 22.5 mg/kg b.i.d., original objective 40X.
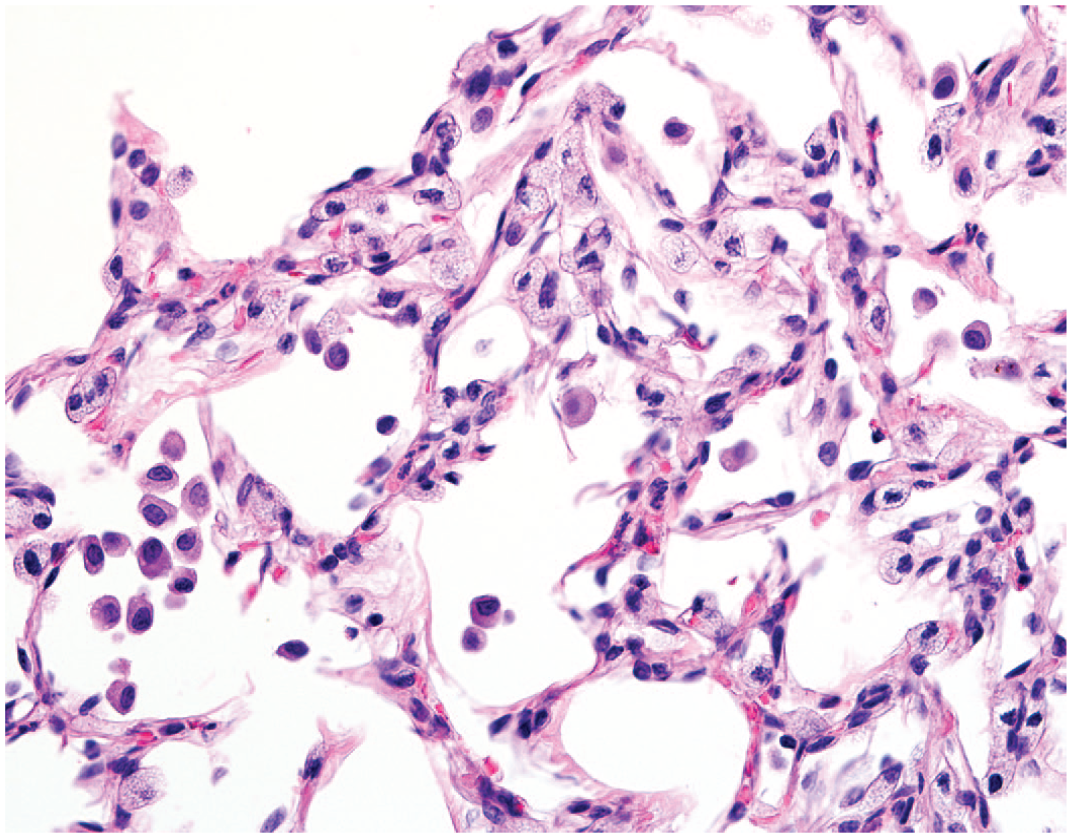
Image of the distal lung exhibiting prominent hypertrophy of type 2 pneumocytes, variable accumulation of intra-alveolar macrophages, and irregular distortion of alveolar structure following 14-day administration of GNE 7915 at 22.5 mg/kg b.i.d., original objective 40X.
An image of distal lung exhibiting hypertrophy of type 2 pneumocytes, increased cellularity of alveolar walls and accumulation of intra-alveolar macrophages admixed with neutrophils following 14-day administration of GNE 7915 at 22.5 mg/kg b.i.d., original objective 40X.
Compound 25
Compound 25 demonstrated suitable tolerability during a rising dose study in rhesus monkeys following oral administration at 30, 100, or 300 mg/kg/day for 3 days at each respective dose (without washout between dosage levels). There were no relevant effects on body weights, food consumption, or hematologic and serum biochemistry parameters. Physical signs were limited to emesis following administration at 300 mg/kg/day. Systemic exposures to parent compound as measured by AUC(0-24h) were 30.7 µM•hours on day 3 after 30 mg/kg/day; 136 µM•h on study day 6 after 100 mg/kg/day; or 181 µM•hours on study day 9 after 300 mg/kg/day.
Hypertrophy of type 2 pneumocytes exhibiting features resembling those described in monkeys receiving GNE 7915, was distributed throughout multiple lobes of lung while hyperplasia of type 2 pneumocytes was readily identified in peripheral locations but confined to the caudal lobes of the lungs.
Subsequently, compound 25 was administered at 50 and 300 mg/kg b.i.d. on study day 1 to rhesus monkeys. Decreased activity, somnolence, and trembling motion were prominent at 7 hours post-dose on day 1 for monkeys receiving 300 mg/kg b.i.d., prompting reduction of the high dose to 225 mg/kg b.i.d. for administration on study days 2 through 7. Decreased activity and/or somnolence persisted in one animal on study days 2 to 4, following dose level reduction. Slight increases of total bilirubin were noted for animals at 225 mg/kg/b.i.d., while no relevant differences for hematology or other serum biochemistry parameters were evident. Systemic exposures (AUC[0-24h]) at 50 mg/kg b.i.d. were 84.8 µM•hours and 99.0 µM•hours on study days 1 and 7, respectively; and 391 µM•h on study day 1 at 300 mg/kg b.i.d. and 329 µM•hours on study day 7 at 225 mg/kg b.i.d. Mean concentration of compound 25 in CSF reached 0.080 µM at 24 hours post-dose on study day 8 for animals receiving 225 mg/kg b.i.d. while drug levels in CSF were below limit of quantification for animals at 50 mg/kg b.i.d.
Hypertrophy of type 2 pneumocytes was widespread in lobes of both lungs and interspersed with areas of hyperplasia of type 2 pneumocytes for each monkey receiving 50 or 225 mg/kg b.i.d. Hyperplasia of type 2 pneumocytes was more evident in peripheral regions of multiple lobes of the lungs, but also exhibited involvement of central locations of affected lobes among monkeys receiving 50 or 225 mg/kg b.i.d. (Figures 8-10). Accumulation of macrophages within alveoli, variably accompanied by neutrophils, was evident at sites of more prominent thickening of alveolar walls (due to hyperplasia of type 2 pneumocytes) and with occasional accumulations of mononuclear cells within interstitial tissue adjacent to small vessels in centri-acinar regions or adjacent to terminal bronchioles (Figure 9). Pleural surfaces were not remarkable. Affected type 2 pneumocytes were immuno-positive for pro-surfactant C, but immuno-negative for CD11c, a marker for intra-alveolar macrophages (Figures 11 and 12). Ultrastructural features of representative lung were similar to those described for GNE 7915 regarding presence of hypertrophic type II alveolar pneumocytes containing more prominent and variably sized lamellar structures and exhibiting typical microvillar projections of the plasma membrane exposed to the alveolar space (Figure 4C). Crowding of type II pneumocytes immediately adjacent to each other resulted in irregular thickening of alveolar walls, occasionally associated with focal accumulation of eosinophilic fibrillary material within alveolar walls. Light microscopy identified increased collagen staining as demonstrated by histochemical trichrome for animals receiving 50 or 225 mg/kg b.i.d., respectively (illustrated in Figure 13, as compared with control in Figure 14).
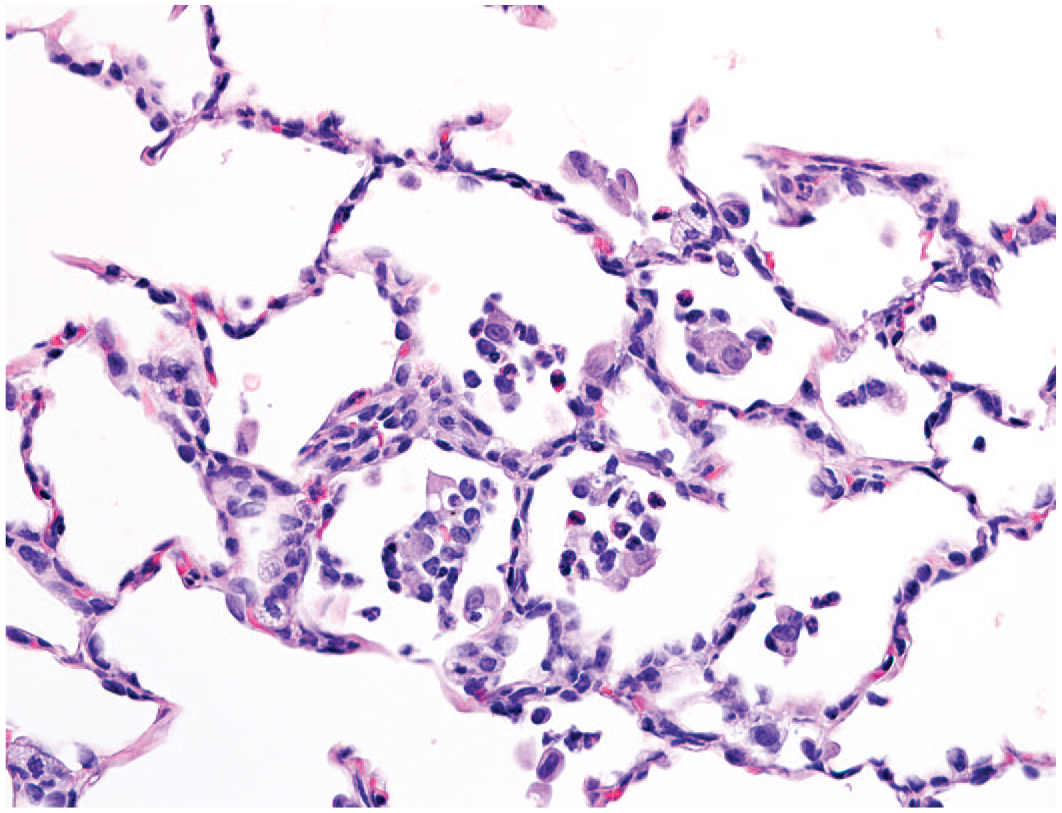
Irregular interstitial thickening of alveolar walls (remodeling) is associated with prominent hypertrophy and hyperplasia of type 2 pneumocytes, with variable accumulation of intra-alveolar macrophages and/or neutrophils. Similar foci are extensively distributed in of both lungs following 7-day administration of compound 25 at 50 or 225 mg/kg b.i.d., original objective 20X.
Prominent hypertrophy and hyperplasia of type 2 pneumocytes are associated with intra-alveolar and interstitial cellular accumulations. Distortion of alveolar walls is evident following 7-day administration of compound 25 at 50 or 225 mg/kg b.i.d., original objective 20X.
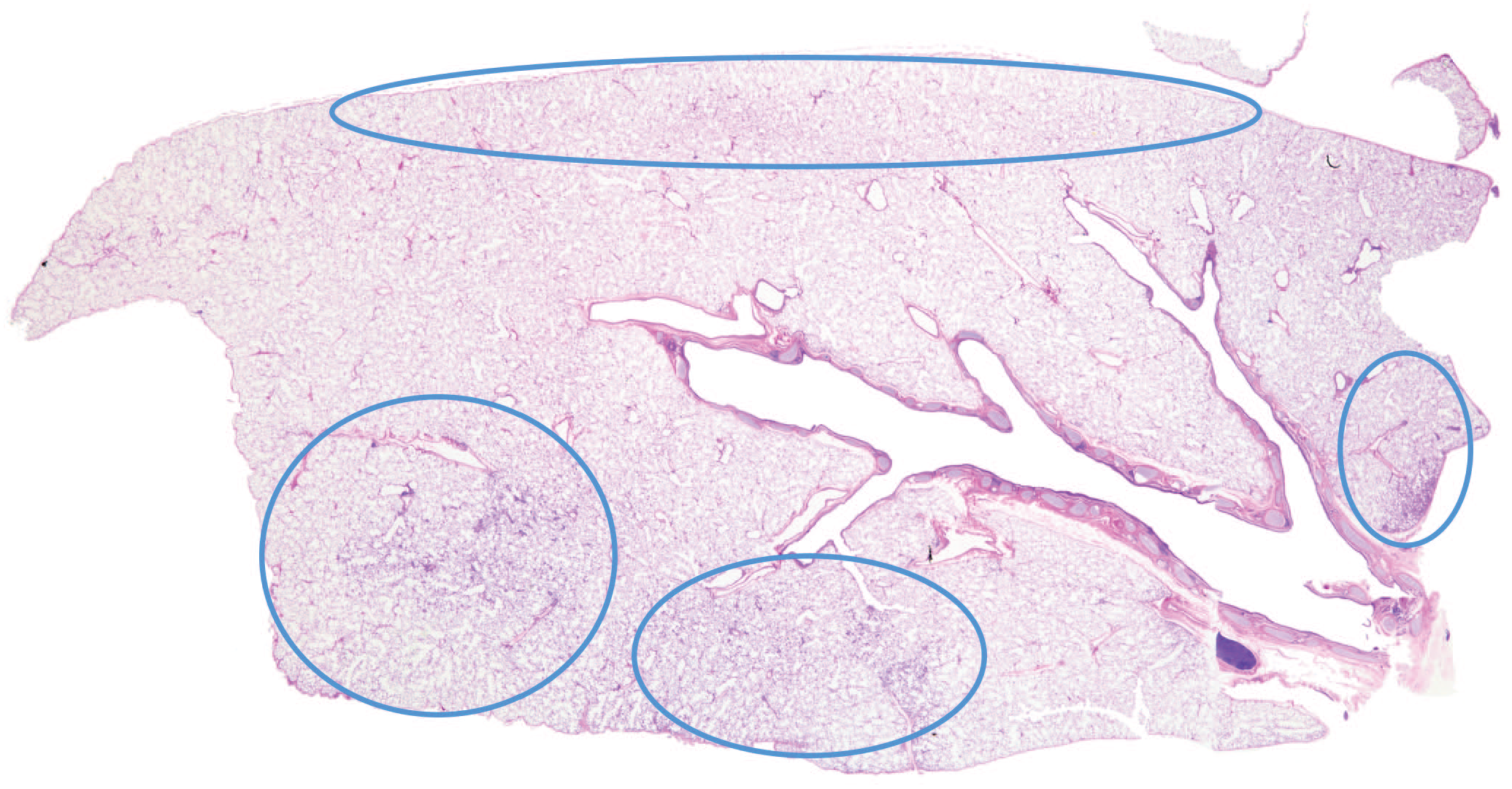
The extent and distribution of hyperplasia of type 2 pneumocytes within peripheral and subpleural locations with extension to central regions are illustrated for a representative lobe of lung. Involvement is typical of findings associated with administration of compound 25 or MK-1468.
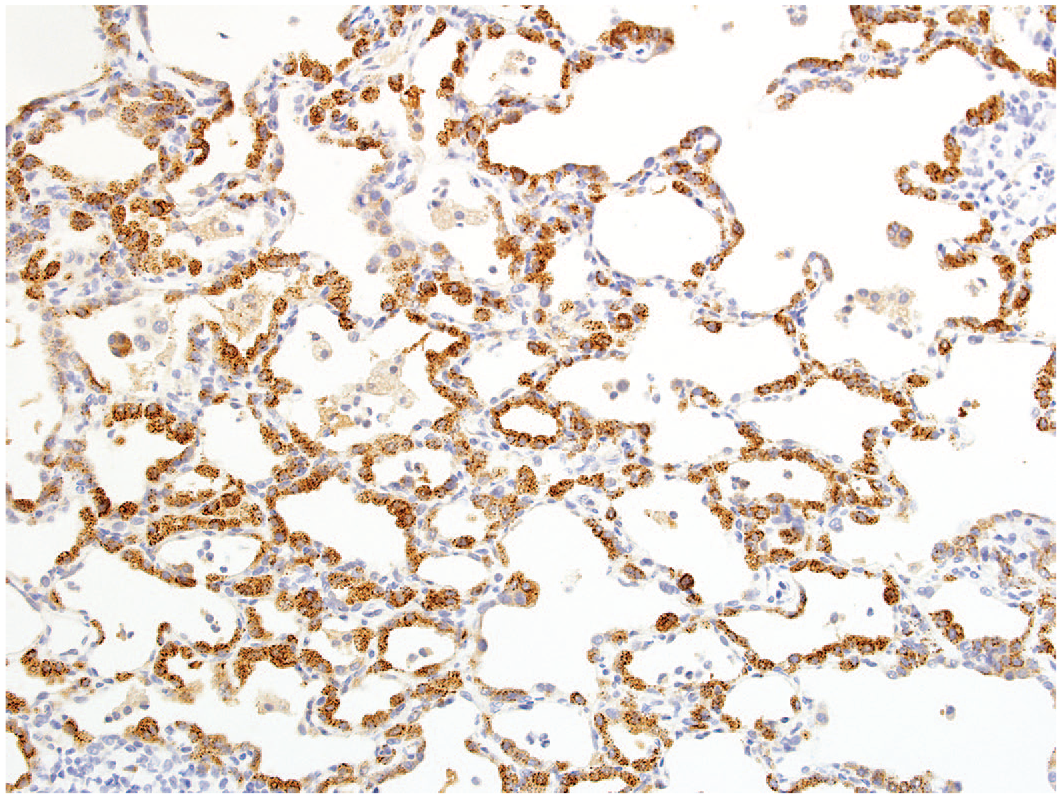
Representative image exhibiting widespread immuno-positive staining for pro-surfactant C among hyperplastic type 2 pneumocytes following 7-day administration of compound 25, original objective 20X.
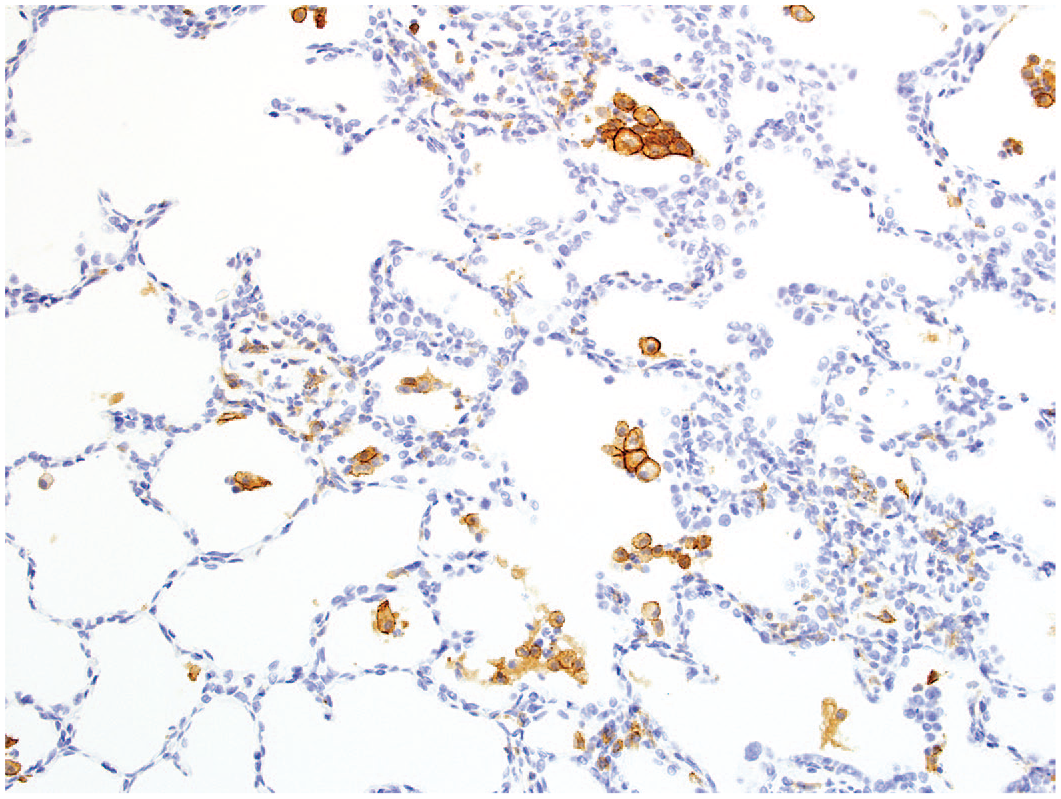
Positive immuno-staining of intra-alveolar macrophages for CD11c following 7-day administration of compound 25, original objective 20X.
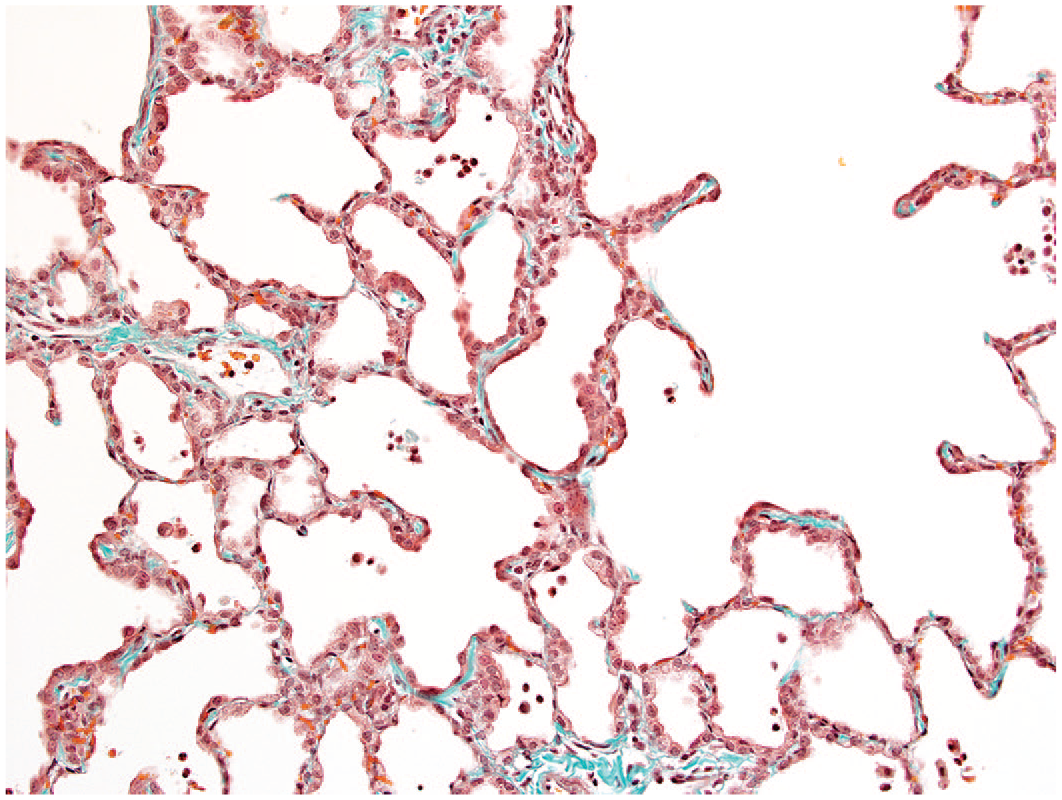
Prominent and irregular pattern of increased collagen staining of alveolar walls and associated interstitial tissue evident via trichrome staining, original objective 20X.

Trichrome of normal lung from a vehicle control illustrating sparse, focal staining of alveolar walls consistent with expected features and distribution, original objective 20X.
MK-1468
MK-1468 was well tolerated during 7-day oral administration at 100, 200, or 800 mg/kg/day to rhesus monkeys, although sporadic emesis among males occurred during the study. Systemic exposures to parent compound expressed as AUC(0-24 hr) on day 7 were 14.1, 25.2, and 65.2 µM•hours respectively for the low, mid, and high dose animals. Concentration of MK-1468 in CSF at 24 hours post-dose on day 8 ranged between 0.006 to 0.005, 0.006 to 0.010, or 0.03 to 0.19 µM respectively, for females and males at the low dose, mid dose, and high dose. Similarly, concentrations of MK-1468 in brain on day 8 at 24 hours post-dose ranged between 0.43 and 0.05, 0.25 and 0.10, or 0.49 and 2.42 µM, respectively, for females and males. Minimally decreased triglycerides for animals receiving 200 and 800 mg/kg/day, and minimally increased total and direct bilirubin for males only at 800 mg/kg/day were noted on study day 6. Histomorphologic changes in the lungs of animals at 200 or 800 mg/kg/day were characterized by increased numbers of intra-alveolar macrophages variably scattered in each lobe in association with hypertrophy of type 2 pneumocytes. Hypertrophic type 2 pneumocytes exhibited greater cellular dimensions due to prominent, finely vacuolated cytoplasm and generally involved peripheral regions of individual lobes with less involvement of more central regions. Intra-alveolar macrophages exhibited finely vacuolated cytoplasm, sometimes associated with accumulation of pale-staining, amorphous material adjacent to alveolar walls. The lungs of animals receiving 100 mg/kg/day were not remarkable.
MK-1468 was administered by daily oral gavage to rhesus monkeys for approximately 30 days at 30, 100, or 500 mg/kg b.i.d. No relevant clinical signs and no evidence of unusual respiratory signs were noted over the dosing interval. There were no effects on food consumption or body weights. There were no ophthalmic findings and no electrocardiographic changes among treated animals at completion of the dosing phase. No relevant differences in hematology or serum biochemistry parameters were evident at study weeks 2 or 4.
Mean sex-combined systemic exposures to MK-1468 expressed as AUC(0-24 hr) in week 5 ranged between 15.0, 31.6, and 67.2 µM•hours, respectively, for the low-dose, mid-dose, and high-dose animals. Mean concentration of MK-1468 in CSF at 24 hours post-dose (after completion of the 30-day dosing phase) ranged between 0.002 and 0.003, 0.003 an 0.028, or 0.022 and 0.021 µM, respectively, for the low-dose, mid-dose, and high-dose females and males. Levels of MK-1468 in the brain were 0.06 to 0.15, 0.16 to 1.0, and 0.57 to 1.2 µM, respectively, for the low-dose, mid-dose, and high-dose females and males.
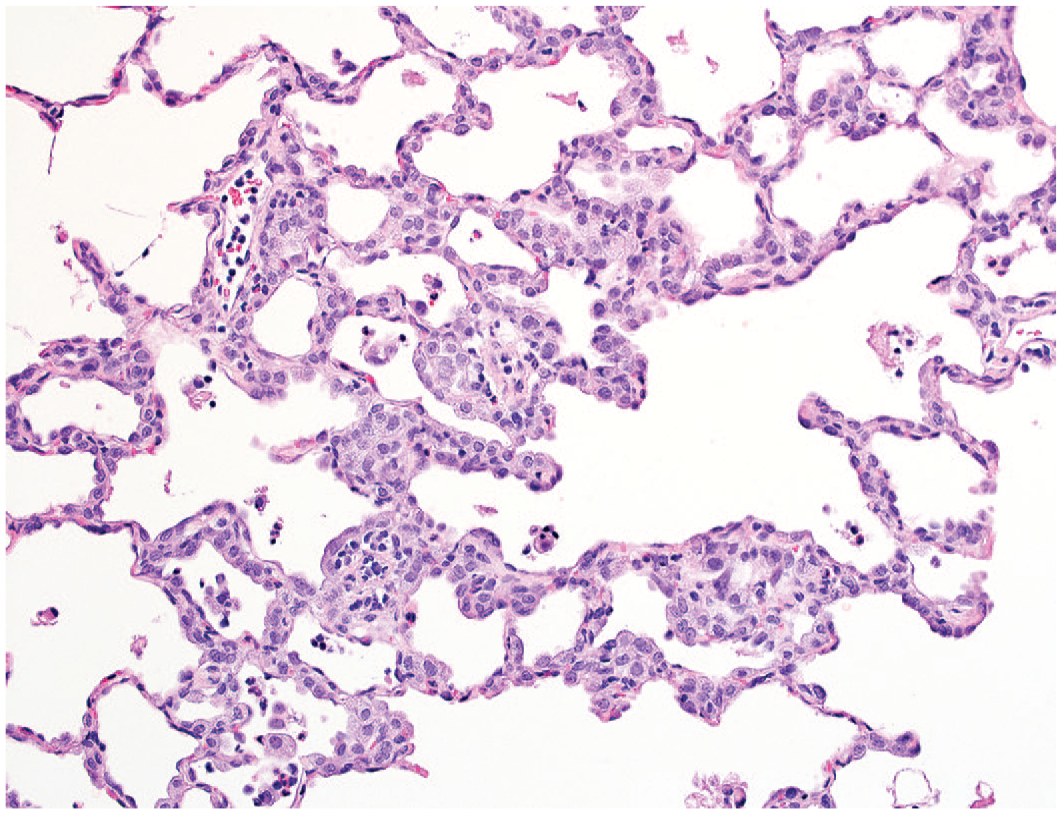
Hypertrophy of type 2 pneumocytes, characterized by individual or small clusters of large type 2 pneumocytes with finely vacuolated cytoplasm that variably covered the luminal aspect of alveolar walls, diffusely affected multiple lobes of the lungs for each female and each male receiving 100 or 500 mg/kg b.i.d. for 30 days. Hypertrophy was associated with hyperplasia of type 2 pneumocytes, distributed as multifocally confluent regions where alveolar lumens were lined by closely aligned, plumb or low cuboidal type 2 pneumocytes. At those locations, alveolar walls were thickened and somewhat distorted, while alveolar lumens sometimes contained accumulations of macrophages variably admixed with neutrophils (Figure 15). Histochemical trichrome staining identified increased collagen within alveolar walls at sites associated with type 2 pneumocyte hyperplasia among 4 animals and 3 animals receiving 100 or 500 mg/kg b.i.d., respectively (Figure 16). The extent and severity of changes described for the lungs were similar for affected animals receiving 100 or 500 mg/kg b.i.d. at completion of the 30-day dosing phase.
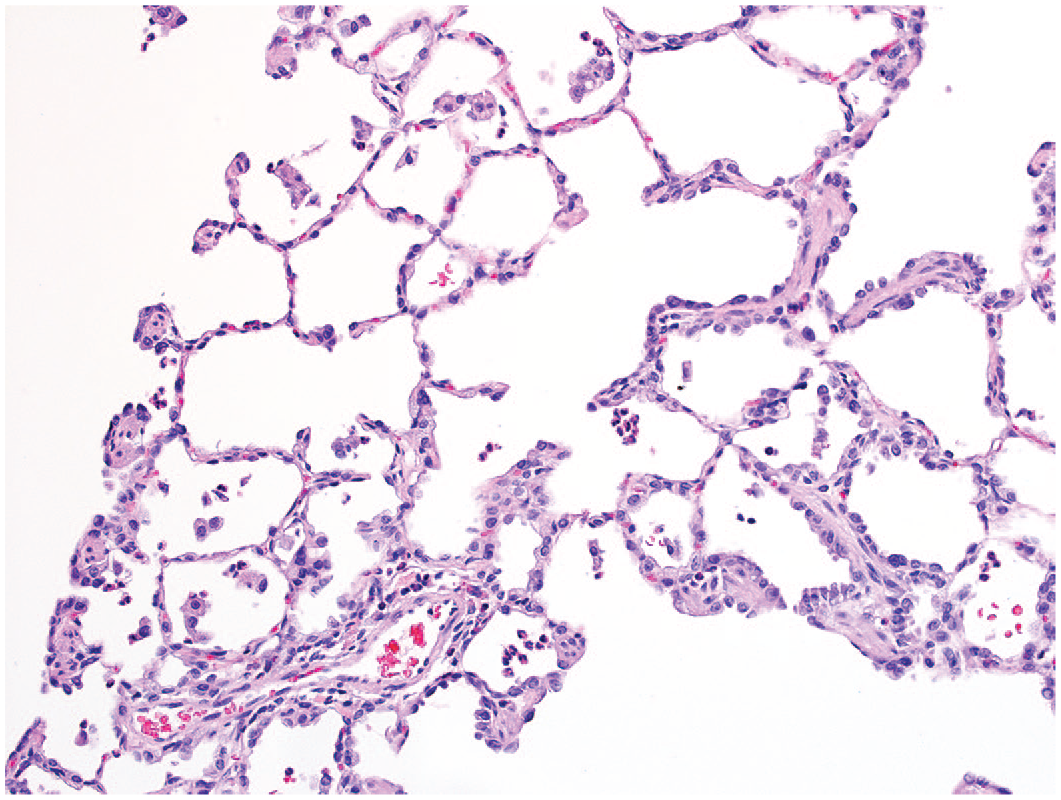
Representative image of lung following 30-day administration of MK-1468 illustrating hyperplasia of type 2 pneumocytes variably associated with thickening of alveolar walls and intra-alveolar presence of neutrophils or macrophages. These features were similar for animals receiving 100 or 500 mg/kg b.i.d., original objective 20X.
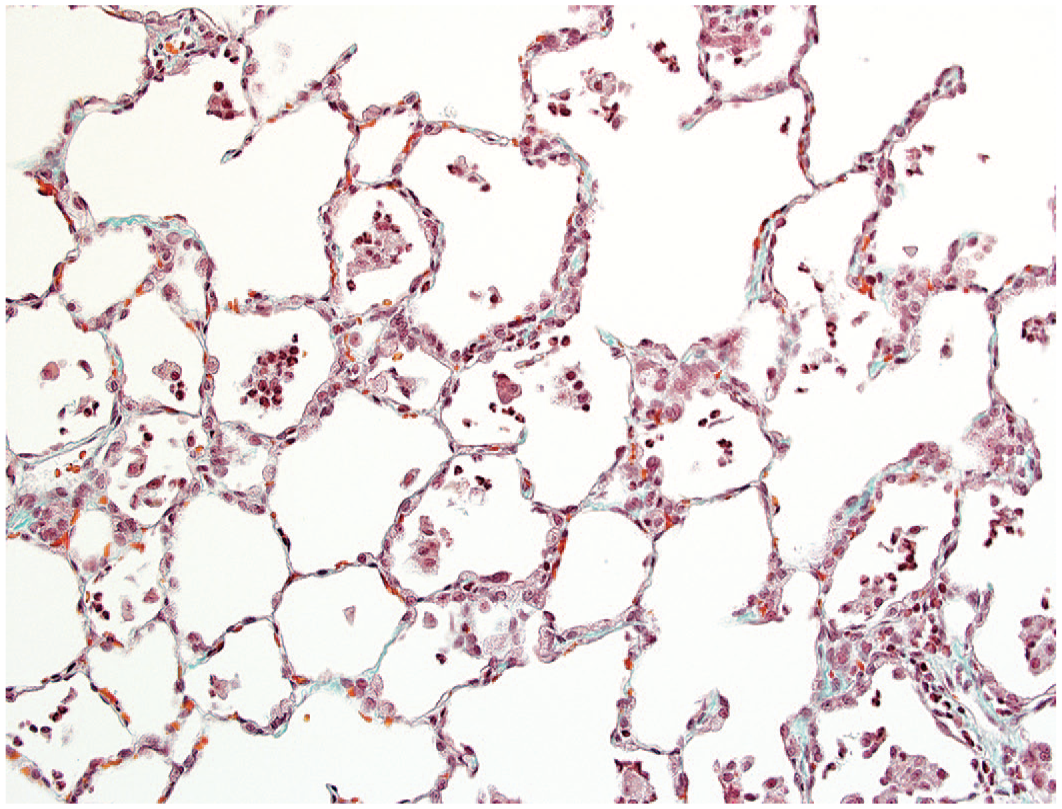
Increased prominence of trichrome staining within alveolar walls is associated with hypertrophy and hyperplasia of type 2 pneumocytes at completion of 4-weeks of administration of MK-1468 at 100 or 500 mg/kg b.i.d. Cellular accumulations are evident within alveolar spaces and within adjacent interstitial locations, original objective 20X.
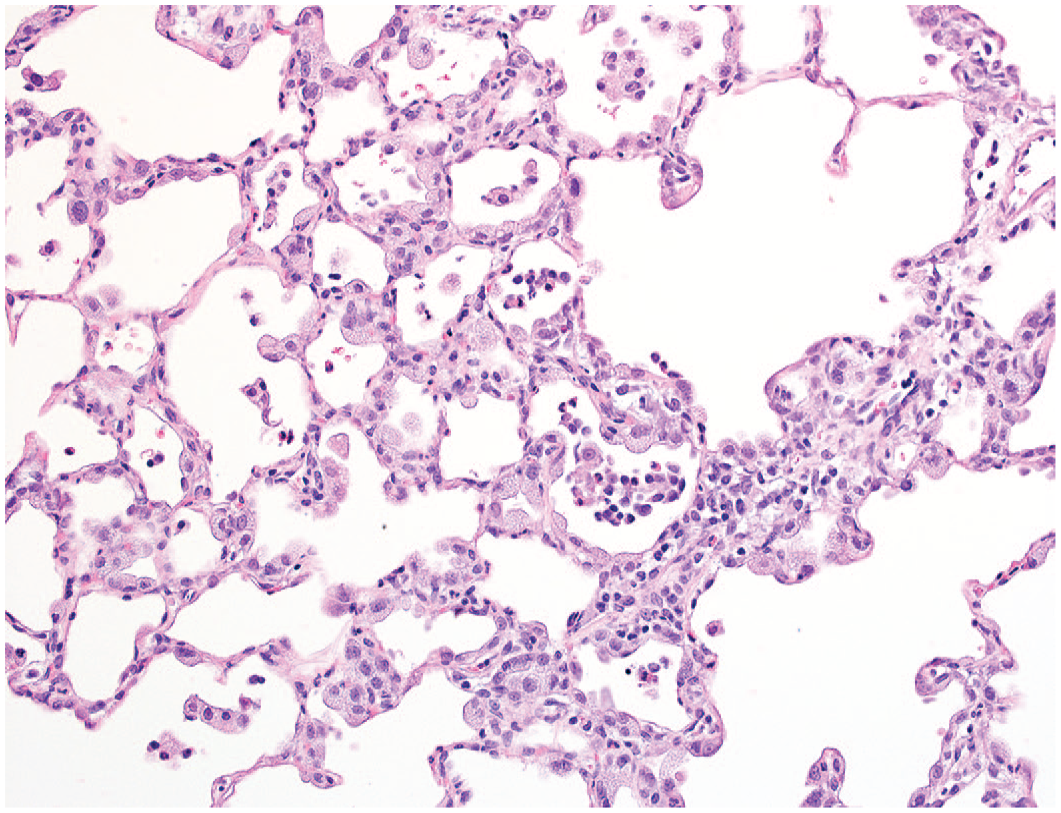
Following a 12-week treatment-free interval, several animals previously receiving 500 mg/kg b.i.d. exhibited changes in the lungs comparable with those encountered among animals evaluated at completion of the 30-day dosing interval; although diffuse simple hypertrophy of type 2 pneumocytes was not present as compared with the extensive pattern evident throughout the lungs as noted for animals evaluated at completion of the dosing phase. However, foci of hyperplasia of type 2 pneumocytes associated with intra-alveolar accumulation of macrophages were readily apparent (Figure 17). Trichrome stains demonstrated increased collagen staining within alveolar interstitial tissue in 2 of 4 animals and exhibited a more generalized distribution of increased collagen within alveolar walls of the lungs as compared with affected animals evaluated at completion of the dosing phase (Figure 18).
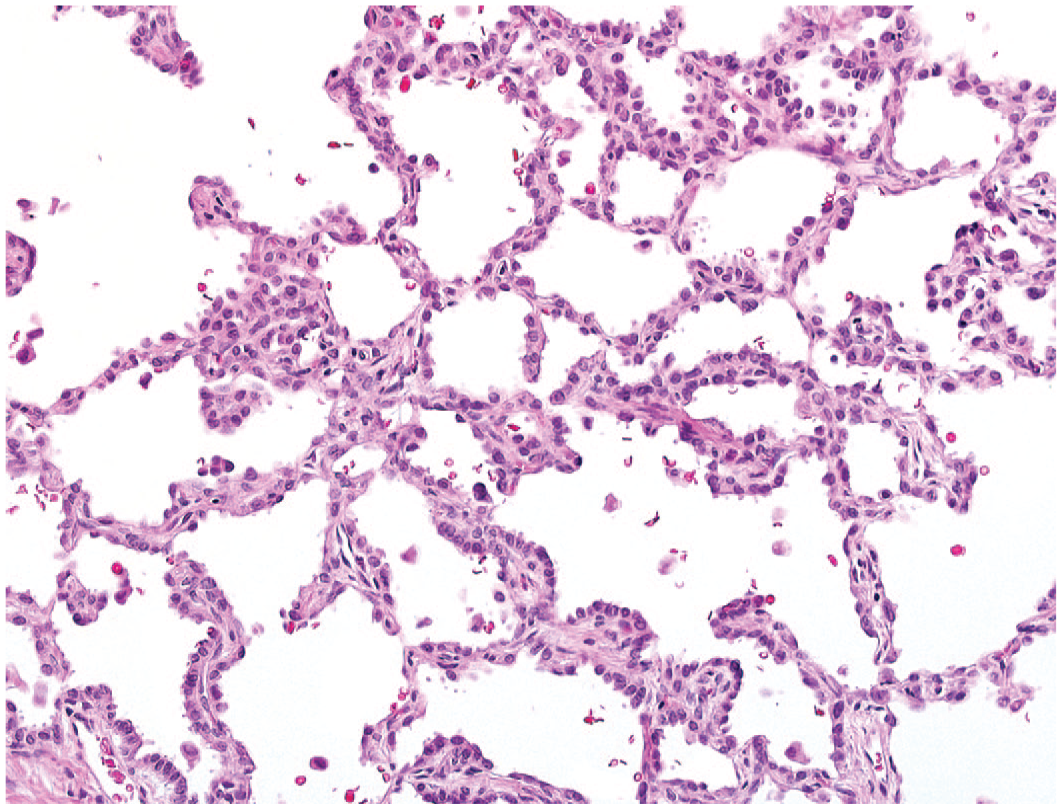
Image of lung at completion of a 12-week treatment-free interval following prior administration of MK-1468 at 500 mg/kg b.i.d. for 30 days. Prominent hyperplasia of type 2 pneumocytes is associated with irregular thickening of alveolar walls, original objective 20X.
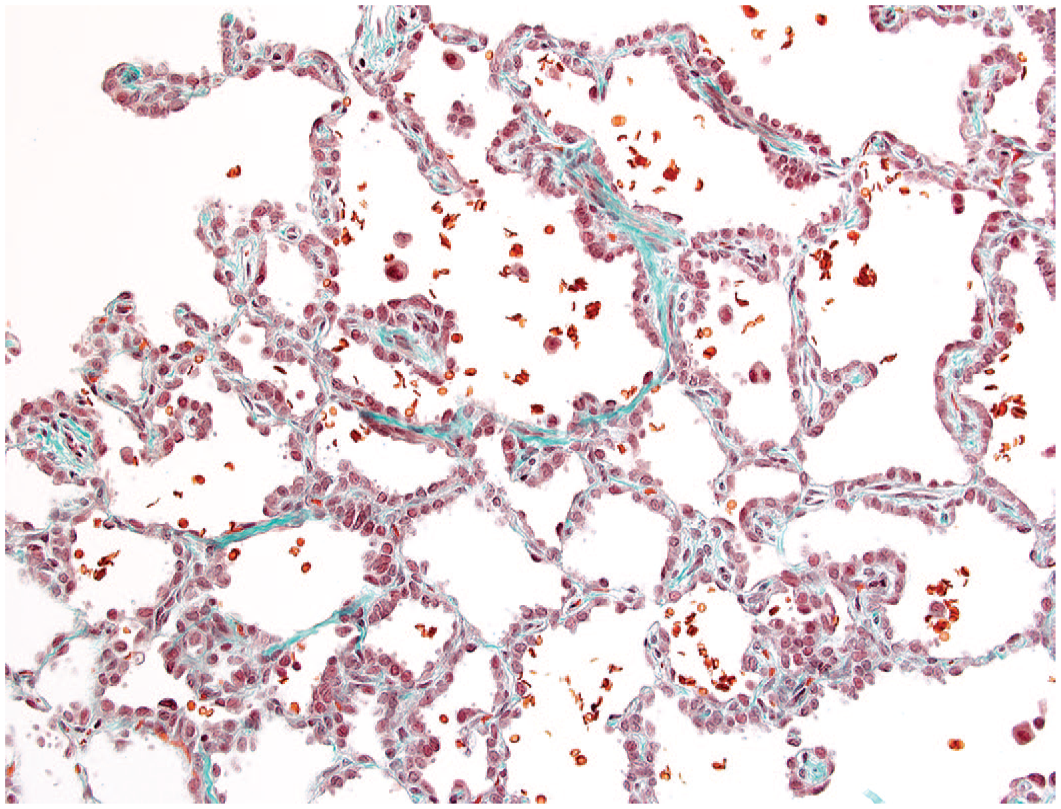
More prominent accumulation of collagen within alveolar walls is evident by trichrome staining for animals following a 12-week treatment-free interval after prior administration of MK-1468 for 30 days at 500 mg/kg b.i.d., original objective 20X.
Toxicokinetic Results
Comparison of unbound plasma exposures to PBMCs IC50s for each compound was determined as a measure of peripheral target engagement (summarized in Table 4). Unbound plasma concentrations of parent compound at trough (Trough or C24hr) were calculated from the total Trough using free unbound plasma fractions (Fu) determined in cynomolgus or rhesus monkey plasma. The ratio of the unbound trough to the pSer935 IC50 in PBMCs was calculated as a measure of peripheral target engagement.
Mean plasma toxicokinetic parameters.
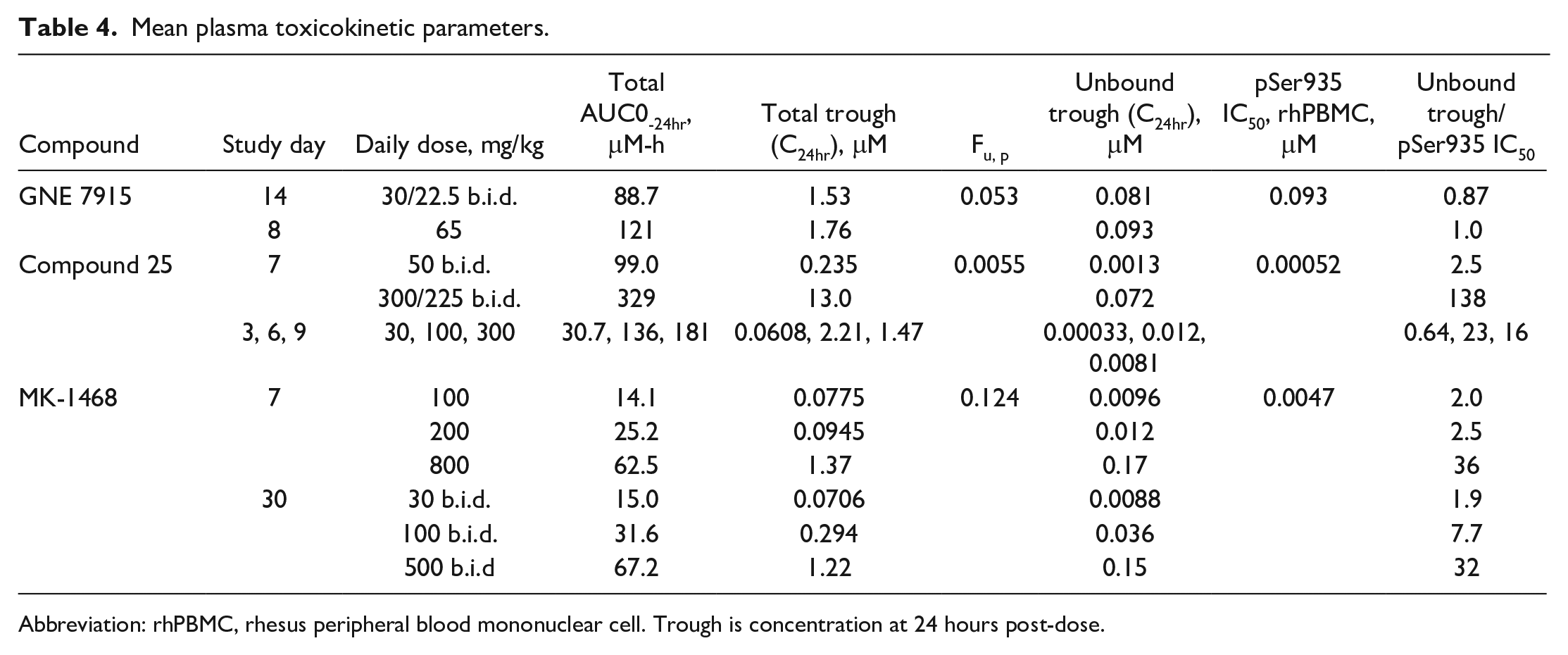
Abbreviation: rhPBMC, rhesus peripheral blood mononuclear cell. Trough is concentration at 24 hours post-dose.
All 3 compounds were brain penetrant as meaningful concentrations in CSF were obtained for each compound as presented in the preceding paragraphs. Peripheral unbound exposures were maintained at or above the pSer935 IC50 with higher levels of peripheral target engagement being associated with compound 25 and MK-1468, as compared with GNE 7915. While each compound may have different toxicokinetic and pharmacodynamic profiles, in general, maintaining an unbound plasma concentration more than 2-fold above the pSer935 IC50 was associated with changes described for type 2 pneumocytes of the lung.
Discussion
The constellation of morphologic changes described within the lungs of nonhuman primates represents a continuous spectrum, with similar features elicited by three structurally different LRRK2 inhibitors. These changes consistently center upon type 2 pneumocytes lining alveolar walls that exhibit greater prominence with more abundant vacuolated cytoplasm. These features are characterized as hypertrophy, the distribution of which is widely scattered in multiple, if not all lobes of the lungs, sometimes in diffuse patterns. Hypertrophy of type 2 pneumocytes resemble the description by prior authors5,20 and is similar to description of lysosome-related organelles within type 2 pneumocytes associated with inhibition of LRRK2 or as described in various LRRK2 knock-out models.2,22,37 Lamellar structures exhibit distinctive morphology identified within type 2 pneumocytes of the distal lung,44,51,53 which are involved with formation, storage, and secretion of surfactant. Type 2 pneumocytes also contribute to reuptake of spent surfactant that is either processed into new surfactant or degraded, while alveolar macrophages have the capability to endocytose and completely catabolize surfactant.
Studies demonstrate that the GTPase Rab38, which facilitates surfactant homeostasis in type 2 pneumocytes, is selectively positioned at the limiting membrane of lamellar structures of type 2 pneumocytes, although Rab7 and Rab4b also exert related functions in type 2 pneumocytes. 54 Various Rab38 knock-out models in rodents exhibit alterations of type 2 pneumocytes associated with enlargement of lamellar structures,34,38,54 bearing resemblance to findings currently described for nonhuman primates. Greater numbers of type 2 pneumocytes are evident by densely arranged and contiguous lining of alveolar walls, representing obvious hyperplasia. Distribution of type 2 pneumocyte hyperplasia tends to occur more prominently at peripheral locations of the lungs but also involves regions deeper within individual lobes (illustrated in Figure 10) and involves multiple if not each lobe of the lungs following administration of Compound 25 or MK-1468. Hyperplastic type 2 pneumocytes are invariably hypertrophic but less frequently exhibit vacuolated cytoplasm. Identity of type 2 pneumocytes was confirmed by ultrastructural evaluations and by immuno-staining by pro-surfactant C, a unique and specific phenotypic marker for type 2 pneumocytes of the lung, and by immuno-positive staining for surfactant protein B.
Hypertrophy and hyperplasia of type 2 pneumocytes are associated with increased cellularity within alveolar lumens, predominantly by accumulations of macrophages, sometimes admixed with neutrophils, within alveolar spaces. The identity of intra-alveolar macrophages was confirmed by immuno-staining by CD11c. Cellular accumulations within alveolar spaces in regions associated with type 2 pneumocyte hyperplasia are variably associated with irregular thickening (remodeling) of alveolar walls, including the presence of increased collagen deposition within alveolar walls as confirmed by increased staining by histochemical trichrome stains. Increased cellularity and thickening of the alveolar interstitium are sometimes present adjacent to small blood vessels within alveolar walls or adjacent to terminal bronchioles. There is no evidence to suggest that intra-alveolar accumulations of macrophages and neutrophils were accompanied by infiltrating or sloughed type 2 pneumocytes, as previously proposed. 28 Furthermore, the current findings draw attention to the fact that the insult to type 2 pneumocytes have downstream consequences leading to remodeling of alveolar structure in the distal lung.6,7 The spectrum of findings elicited in the lungs by each compound aligned with systemic potency uniquely exhibited by each molecule, given that Compound 25 and MK-1468 were associated with potentially greater peripheral engagement as determined by sustained unbound plasma concentrations above the concentration associated with the respective IC50 in PBMCs. However, duration of dosing also likely impacts the onset and progression of changes described in the lungs.
The relevance of extensive sampling and specific orientation utilized for evaluation of each lobe of the lungs became essential for a more comprehensive approach suitable for identifying potential findings that differ or exhibit variations of distribution in affected lung among treated animals receiving a given compound. Prior citations describing effects of LRRK2 inhibitors in nonhuman primates do not disclose details regarding the extent of sampling or extent of evaluation of lungs.5,20 We are unable to draw direct comparisons with the results presented by those authors, relative to findings presented currently. Effects in lungs of rodents have been described for LRRK2 knock-out models, although changes appear to be limited to type 2 pneumocyte hypertrophy24,34,49 or associated with increased size of lamellar structures in type 2 pneumocytes.22,34,54 Regardless, the extensive sampling approaches utilized for our current studies afforded more comprehensive evaluations that identified both distribution and character of tissue responses for each lobe of the lungs.
The current findings described for the lungs of nonhuman primates represent an on-target effect of structurally distinct, selective LRRK2 inhibitors. The functions of LRRK2 are linked to intracellular trafficking in the endosome-lysosome pathway encompassing complex vesicular functions.1,19 Expression of LRRK2 in the lungs is substantial (see Fuji et al 20 ; supplemental information), and mounting evidence implicates LRRK2 as having critical roles for function of type 2 pneumocytes related to surfactant production and for maintenance of alveolar integrity by renewal of type 1 pneumocytes by differentiation from dividing type 2 pneumocytes.26,46 LRRK2 appears to function as a master regulator of vesicular trafficking through interactions with Rab GTPases. 43 Rab38, a GTPase that exhibits high expression in lung-restricted to type 2 pneumocytes, 54 is a likely substrate for LRRK2. Furthermore, deficiencies of Rab38 are associated with pulmonary disorders in rodents and humans, which for humans eventually lead to complications related to pulmonary fibrosis.12,30,38
Although impact to type 2 pneumocytes is characterized for the distal lungs, it is important to acknowledge that inhibition of LRRK2 also carries potential implications for function and integrity of alveolar macrophages, for both normal resident, sessile alveolar macrophages and intra-alveolar macrophages, the latter that invariably accompany changes described for type 2 pneumocytes. Constitutive expression of LRRK2 in myeloid cells including those of macrophage lineage is essential to intact cellular immune responses.11,29,30,36,40 The current studies do not exclude potential direct effect upon intra-alveolar macrophages following administration of a LRRK2 inhibitor.
Changes described in lungs that persist after a treatment-free interval following withdrawal of test article, suggest that a remodeling process in the lungs is associated with inhibition of LRRK2, particularly given that intact function of type 2 pneumocytes is essential to repair processes and integrity of alveolar structure. Increased numbers of intra-alveolar macrophages admixed with neutrophils, in association with changes described for type 2 pneumocytes, implicates interactions among these components leading to re-modeling of alveolar structure. Type 2 pneumocytes have essential roles in surfactant dynamics and in response to pulmonary insult, thereby providing key roles in repair mechanisms and maintenance of integrity of distal lung structure and function. Insult to type 2 pneumocytes is recently regarded to be associated with pulmonary disorders impacting surfactant production26,35,46 or during response to compromised alveolar integrity. As originally described by Haschek and Witschi 23 and Osanai et al, 39 intact type 2 pneumocytes are critical to orderly repair processes of the lung. More recent studies implicate type 2 pneumocytes that exert pivotal roles during pulmonary fibrosis.26,46 However, interactions among alveolar type 1 and type 2 pneumocytes with resident alveolar macrophages, and with interstitial macrophages, represent a complex dynamic within the local environment of the distal lung.4,9,25,32,35 The character and extent of changes detected in the lungs following administration of LRRK2 inhibitors and particularly following a substantial drug-free interval, differ from prior findings and interpretations that effects of LRRK2 inhibition in nonhuman primates could be reversible. The current results indicate that pulmonary effects associated with repeated oral administration of potent, selective LRRK2 inhibitors in nonhuman primates are associated with remodeling of the distal lung, which do not exhibit reversibility. The potential long-term consequences in the lungs related to greater duration studies following administration of potent LRRK2 inhibitors are unknown.
Footnotes
Author Contributions
The analyses and interpretations expressed in this publication are solely those of the authors. Each author contributed to the composition of the manuscript and contributed to discussions leading to content and organization of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.