Abstract
For toxicology testing of (agro)chemicals, different study types are being performed with general and/or reproductive toxicity endpoints (see Organisation for Economic Co-operation and Development guidelines). In most of these rat studies, vaginal cytology is performed on serial samples (collected by lavage) for evaluation of cycle regularity and evidence of mating, and/or on a single sample collected on the day of necropsy for information on the estrous cycle stage and allowing correlation with histopathology. In the latter case, the utility of vaginal cytology can be argued. In this article, estrous cycle stages based on vaginal cytology of samples taken on the day of necropsy and histopathology of ovaries, uterus, and vagina (gold standard for estrous cycle stage assessment) were compared. The agreement was generally low. Disagreement between the two methods could be explained by time differences between lavage and necropsy, by manipulation of vaginal epithelium during lavage which may impact epithelial morphology on histology, and by misinterpretation of vaginal cytology during or shortly after lactation. Based on the results of estrous staging within different study types, we strongly discourage vaginal cytology from samples collected on the day of necropsy since there is no added value, vaginal manipulation can be stressful and may complicate the histologic diagnosis.
Keywords
Introduction
Toxicology testing of (agro)chemicals may include an extensive battery of studies in rats with both general toxicology and reproductive toxicology endpoints. In several guidelines of the Organisation for Economic Co-Operation and Development (OECD guidelines), there is a requirement to perform vaginal cytology on samples taken on the day of necropsy. The 90-day general toxicology study is described in the OECD guideline OECD 408,
1
whereas reproductive toxicity studies (or a combination of general toxicity and reproductive toxicity studies in one or more generations) are described in the OECD guidelines 421,
2
422,
3
and 443.
4
The studies with reproductive endpoints all require a complex study design in which males and females are mated in order to evaluate developmental toxicity and effects on fertility. For these purposes, knowledge of the estrous cycle on an individual basis is needed, thus preparing serial vaginal samples (by using a cotton-tipped swab or by vaginal lavages) for cytologic assessment of a regular estrous cycle is an absolute requirement.
5
This enables a check on normality and duration of the different stages, to decide if individual female rats should be included in the study or not (pretest). During the mating period, vaginal samples are prepared to evaluate whether sperm is present as evidence of mating. In the cases where histopathology of the female reproductive organs is also required, it can be questioned whether the evaluation of the estrous cycle on the day of necropsy by vaginal cytology is of any added value. In the guidelines it is stated that At necropsy, the estrous cycle of all females should be determined by taking vaginal smears. These observations will provide information regarding the stage of estrous cycle at the time of humane killing and assist in histological evaluation of estrogen sensitive tissues.
1
A vaginal smear is examined on the day of necropsy to determine the stage of the oestrous cycle and allow correlation with histopathology in ovaries/reproductive organs.2-4
In the present article, vaginal cytology results of samples taken on the day of necropsy and histopathology of female reproductive organs from control animals subjected to different study types were evaluated and compared. The usefulness of evaluation of vaginal lavage samples for the cytologic determination of estrous cycle stage in these studies is discussed and includes a critical review of the validity of arguments stated in the guidelines. The most important arguments that were taken into account were (1) avoidance of unnecessary handling to minimize discomfort of the animals, (2) the influence on the vaginal morphology at histologic evaluation shortly after lavage, (3) the incorrect staging of vaginal cytology samples taken on the day of necropsy (diestrus) suggesting a normal estrous cycle which is not the case in female rats during or shortly after lactation (lactating animals are per definition not cycling), and (4) the superior reliability of histopathology of multiple reproductive organs for the evaluation of the cycle stage at a single time point. Additional consequences might be a reduction of time and costs.
Material and Methods
Study types (according to OECD guidelines)
Studies from the following OECD guidelines are included in the present evaluation:
OECD 408: Repeated Dose 90-Day Oral Toxicity Study in Rodents (90-Day study)
OECD 421: Reproduction/Developmental Toxicity Screening Test (Repro-Screening study)
OECD 422: Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test (28-Day Combi-study)
OECD 443: Extended One-Generation Reproductive Toxicity Study (EOGRT study)
These different general and reproductive toxicity study types, described in the Guidelines for Chemical Testing which include the recommendation of vaginal cytology, are summarized in Table 1. The table also includes the number of studies per study type that were evaluated in the present review and some relevant details of the study design. In the Repro-Screening studies (OECD 421), 2 28-Day Combi-studies (OECD 422), 3 and the EOGRT studies (OECD 443) 4 vaginal cytology was performed on samples collected daily by lavage for 2 weeks before mating to document normal estrous cyclicity, during the mating period for proof of mating and on the day of necropsy. In the 90-Day studies (OECD 408), 1 a vaginal cytology sample was only collected on the day of necropsy.
Different study types according to OECD guidelines (Testing of Chemicals Guidelines) with evaluation of vaginal cytology during the in-life phase and/or as reproductive endpoint at necropsy.

Abbreviations: LD = lactation day; n = number of control females evaluated per study; PND = postnatal day.
Only 5 females were selected for full histopathology.
For the F1-generation only the 20 females that were selected for histopathology (cohort 1A) were included in this review.
Animals
For all studies reviewed (in-life period between May 2016 and September 2019), nulliparous and non-pregnant female Crl: WI(Han) rats obtained from Charles River Deutschland, Sulzfeld, Germany, were used. At initiation of the study, the females were 6 to 7 weeks old (OECD 408 and 443 studies)1,4 or 12 to 14 weeks old (OECD 421 and 422 studies).2,3 Only data from control group females surviving until the end of their assigned study period were included in the evaluation. This also included females with total litter loss, non-pregnant females, or females that did not mate.
Husbandry
Female rats were group housed (up to 5 animals) in polycarbonate cages (Macrolon, MIV type, height 18 cm) for the complete treatment period (90-Day studies; OECD 408) 1 or during the pretest and premating period (studies including reproduction). During the mating phase, males and females were cohabitated on a 1:1 basis and during the postmating phase, females were individually housed in Macrolon plastic cages (MIII type, height 18 cm). During the lactation phase, pups were housed with the dam. The cages contained appropriate bedding (Lignocel S 8-15, JRS—J. Rettenmaier & Söhne GmbH + CO. KG, Rosenberg, Germany) and were equipped with water bottles. Target temperatures of 18°C to 24°C with a relative target humidity of 40% to 70% were maintained. A 12-hour light/12-hour dark cycle was maintained, except when interrupted for designated procedures. Ten or greater air changes per hour with 100% fresh air (no air recirculation) were maintained in the animal rooms. Pelleted rodent diet (SM R/M-Z from SSNIFF Spezialdiäten GmbH, Soest, Germany) was provided ad libitum throughout the study. The feed was analyzed by the supplier for nutritional components and environmental contaminants. Municipal tap water was freely available to each animal via water bottles.
For psychological/environmental enrichment and nesting material, animals were provided with paper (Enviro-dri, Wm. Lilico & Son (Wonham Mill Ltd), Surrey, United Kingdom)
Each study plan was reviewed and agreed by the Laboratory Animal Welfare Officer and the Ethical Committee of Charles River Den Bosch as required by the Dutch Act on Animal Experimentation. 5
Vaginal Lavage, Preparation of Samples, and Cytology Evaluation
In the different study types, samples were collected by vaginal lavage (between 7:00 and 10:00
Insertion of 0.1 to 0.2 ml saline into the vagina using a disposable pipette (VWR International, Inc., 405 Heron Drive, Bridgeport NY, 08014 USA) and flushing 3 times.
Spreading the content of the pipette on a glass slide and air drying.
Fixation (at least 10 minutes) in methanol and staining with Giemsa (Merck, Darmstadt, Germany).
Evaluation of the vaginal cells6,7 and assignment to one of the 4 estrous cycle stages as follows: estrus: mainly cornified epithelial cells; metestrus: in general many cells present, containing mainly granulocytes that surround nucleated cells and some cornified epithelial cells; diestrus: in general less cells and mucus present, containing equal numbers of granulocytes, nucleated cells and cornified epithelial cells; proestrus: mainly nucleated epithelial cells). After evaluation of serial lavages, the estrous cycle was classified as regular, irregular, acyclic, extended estrus, or unable to determine.
Evaluation of the slides was performed by laboratory technicians.
Necropsy, Histology, and Histopathology
Complete postmortem examinations were performed on all animals. Animals were anesthetized using isoflurane and subsequently exsanguinated. At the time of necropsy, the tissues and organs were collected, weighed (if applicable), and placed in 10% neutral-buffered formalin fixative (neutral phosphate buffered 4% formaldehyde solution, Klinipath, Duiven, The Netherlands).
Tissues were embedded in paraffin (Klinipath, Duiven, The Netherlands), sectioned, mounted on glass slides, and stained with hematoxylin and eosin (Klinipath, Duiven, The Netherlands). All tissues were examined by a board-certified toxicological pathologist with training and experience in laboratory animal pathology. For the purpose of the present data review, only the histopathology results of ovaries, uterus/cervix, and vagina were evaluated, including the determination of the estrous cycle stage.8-10
Data Evaluation
The original evaluation of vaginal cytology and histopathology of the ovaries, uterus, and vagina were used as data set for the present article.
Results
Estrous Stages in Pretest and (Pre)Mating Period
Serial (daily) vaginal samples in the pretest and (pre)mating period of studies with reproductive endpoints
In Table 2, two examples of the estrous cycle stages based on serial (daily) vaginal samples are given. These example studies included two different 28-Day Combi-studies (OECD 422). In Study 1, the daily vaginal samples were staged as estrus (labeled green) every 4 days, showing a very regular pattern indicating that all 10 control females had a regular 4-day cycle and were all suitable for use in the study. The regularity was similar in the pretest (the first 14 days) and premating period (days 15-28), ending in proof of copulation (evidence of mating) when sperm cells were present in the vaginal sample (labeled orange). The green (and orange) labeling increases the recognition of regularity (or absence of regularity) of the cycle.
Two study examples of estrous cycle staging by vaginal cytology (OECD 422 studies).
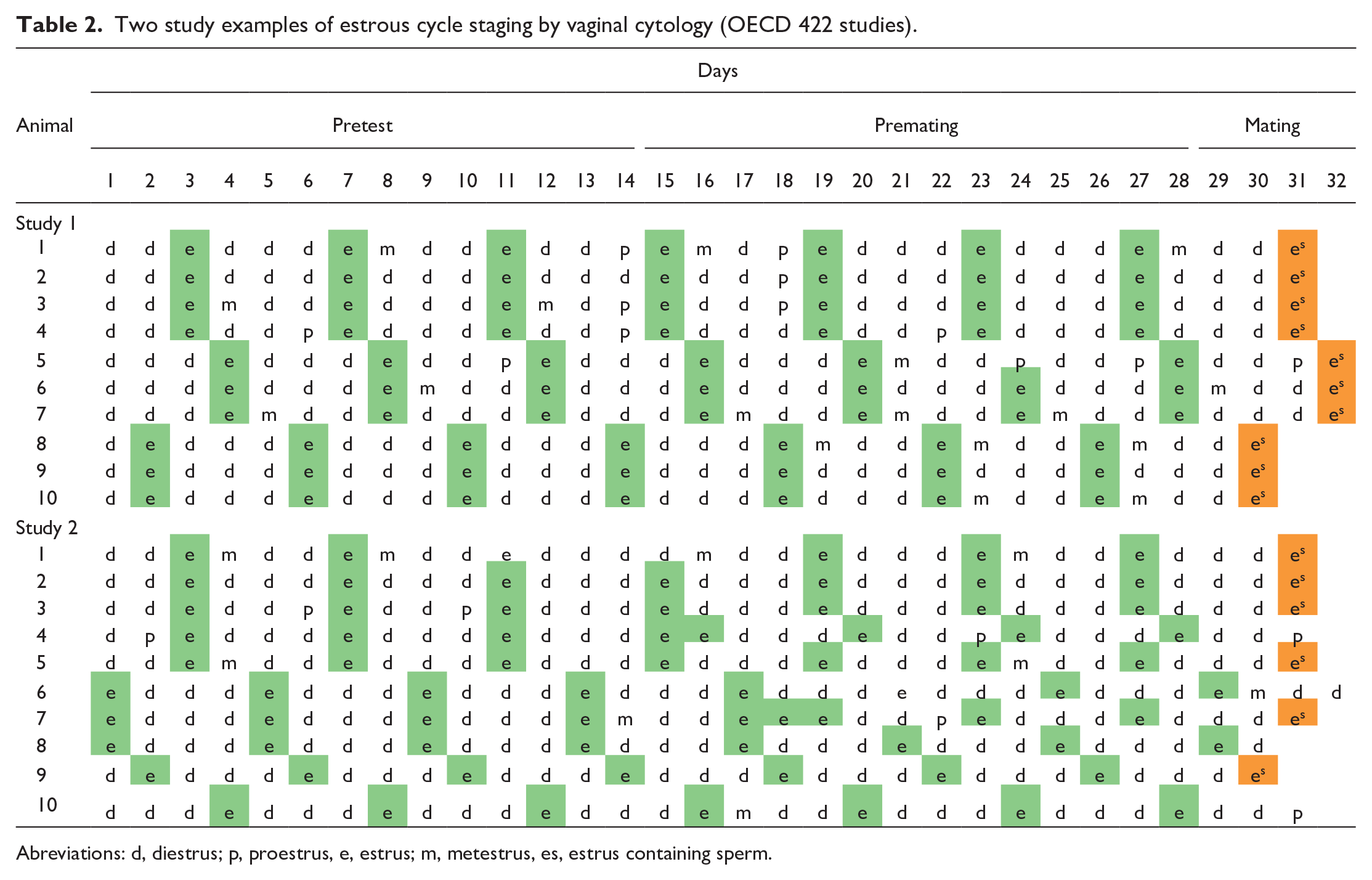
Abreviations: d, diestrus; p, proestrus, e, estrus; m, metestrus, es, estrus containing sperm.
In Study 2, some irregularities were observed in the estrous cycle stages of 3 females during the premating period (female nos. 1, 4, and 7), either missing an estrus (female no. 1) or showing slight shifts in the estrous stages (female nos. 4 and 7). These small irregularities were considered within the normal range of 4- to 5-day cycles. In all three females, the cyclicity was restored within the premating period and two females showed proof of mating (sperm present in vaginal smear). Some females (female nos. 6, 8, and 10) did not show sperm in the vaginal smear after the mating period and therefore proof of mating is lacking for these animals. Although only two examples of a repeated dose toxicity study with reproduction/developmental toxicity screening test (OECD 422) are given, similar results were seen for all other reproduction studies that are evaluated in this article.
Estrous Stages on the Day of Necropsy
Repeated dose 90-day oral toxicity study in rodents (OECD 408)
In Table 3, a comparison of the estrous cycle stages is given for all control females of six 90-Day studies (OECD 408) based on cytologic evaluation of vaginal lavage samples taken on the day of necropsy and based on the histology (morphology of ovaries, uterus, cervix, and vagina). Agreement of estrous stages between the two methods is shaded in gray. The mean percentage of agreement of the estrous stages between cytology and histology of the studies investigated was 45%, with a range of 20% to 70% per study.
Comparison of the estrous cycle stages based on vaginal cytology and on histology of multiple reproductive organs of 90-Day studies at the day of necropsy (OECD 408 studies).

Matches are shaded gray.
Abbreviations: Cyto, cytology; Histo, histology; ncs, no cycle stage: morphology did not match a specific estrous stage; –, no material available; d, diestrus; p, proestrus, e, estrus; m, metestrus.
Reproduction/developmental toxicity screening test (OECD 421)
The reproduction/developmental toxicity screening studies include cytologic evaluation of vaginal lavage samples taken on the day of necropsy. Histopathologic examination is performed on ovaries only, in accordance with OECD 421 guidelines. Only for females that did not mate or for which mating did not result in the delivery of healthy pups all reproductive organs were evaluated, allowing the estrous stage determination by histologic examination. This does not occur very often. From 10 different OECD 421 studies with 10 control females per study (total n = 100), the percentage of control females that did not mate or for which mating did not result in healthy pups was only 7%, and thus on average, there was less than 1 female in the control group per study for which estrous cycle stage could be determined histologically. Therefore, this study type was not used for the comparison of vaginal cytology and histopathology.
Combined repeated dose toxicity study with the reproduction/developmental toxicity screening test (OECD 422)
Determination of the estrous cycle stages in the Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test (OECD 422) was performed on the cytology of lavage samples from 10 females taken on the day of necropsy and based on the histopathology of the reproductive organs of 5 of these females that were selected for the histopathology evaluation and a few females that did not produce healthy offspring (see Table 4). There was no agreement in cycle stages between the two methods in the females that had normal healthy offspring (lactating females). Almost all females were in diestrus according to the vaginal lavage samples taken on the day of necropsy (at lactation day 14-16). Histopathology assessment of ovaries, uterus, and vagina of the 5 selected females confirmed the lactating status of the animal which, as expected, did not correspond to a diestrus stage since lactating females are per definition not cycling. For a few females, findings in the vagina were recorded (e.g., mucification, infiltration inflammatory cells, luminal debris). In several studies (7 from the 10 studies), one or two control females did not have offspring and thus were non-lactating on the day of necropsy (indicated by animal numbers 6 and/or 7 in Table 4). For most of these females, a normal cycle could be assigned based on the histopathology of the ovaries, uterus, and vagina.
Comparison of the estrous cycle stages based on vaginal cytology and on histology of multiple reproductive organs of 28-Day Combi-studies at the day of necropsy (OECD 422 studies).
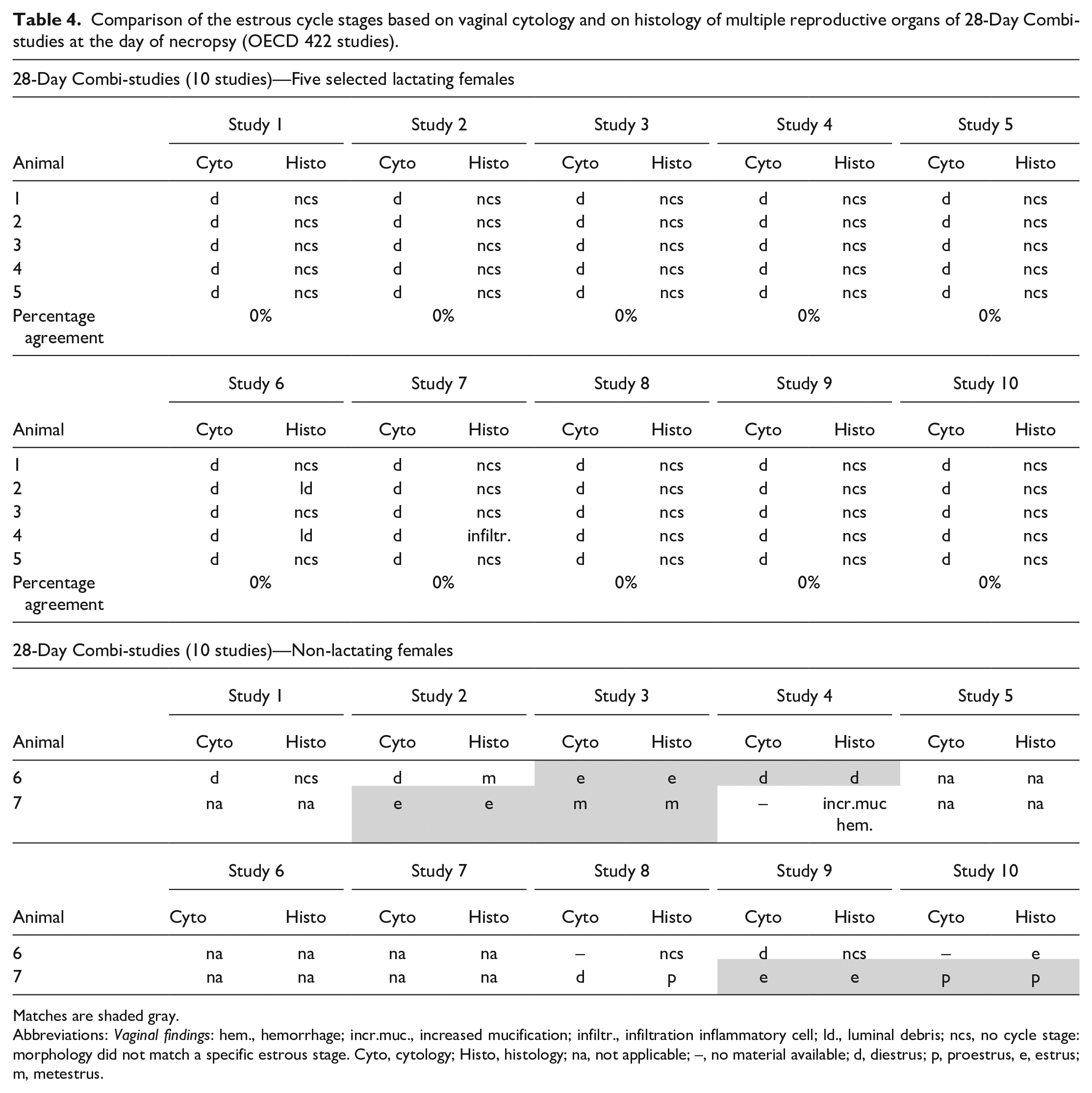
Matches are shaded gray.
Abbreviations: Vaginal findings: hem., hemorrhage; incr.muc., increased mucification; infiltr., infiltration inflammatory cell; ld., luminal debris; ncs, no cycle stage: morphology did not match a specific estrous stage. Cyto, cytology; Histo, histology; na, not applicable; –, no material available; d, diestrus; p, proestrus, e, estrus; m, metestrus.
Histopathology of ovary, uterus, and vagina at lactation day 14-16 of 28-Day Combi-studies showed morphologic characteristics that did not fit within a certain stage in the estrous cycle but represent all features typical of a recent pregnancy. Ovaries contained large corpora lutea, consisting of hypertrophic cells, and a considerable number of small follicles (Figure 1A). Large antral follicles were lacking. In almost all uterus sections, a remnant of an implantation site was found at the mesometrial triangle just outside the myometrial layer (Figure 1B). The uterus was rather small (Figure 1B), consisting of a compact stroma and cuboidal luminal and glandular epithelium (Figure 1C). The luminal and glandular epithelial cells were generally inactive with rare mitotic activity. The vaginal epithelium of individual females had different morphological appearances (Figure 1D and E). In many cases, the epithelium consisted of 2 to 3 layers, the basal layer(s) consisting of flat basal cells and the luminal layer of slightly mucified cells (Figure 1D). In some females, the epithelium had vacuoles with or without additional granulocytes (or debris) in the lumen (Figure 1E) and granulocytes could also be observed within the epithelium. Females that were necropsied earlier than lactation day 14-16, e.g., in case of total litter loss, generally showed a multilayered vaginal epithelium with marked mucification, which is comparable to the vaginal epithelium of a normal pregnant rat (Figure 1F). The normal morphological characteristics of vaginal epithelium at late diestrus from a sexual mature nulliparous female rat is demonstrated in Figure 2A. The epithelium of this female was 5 cells thick, and some intraepithelial neutrophils were present. In Figure 2D, a normal vaginal cytology sample interpreted as diestrus is shown and consisted of a mixture of isolated or small clusters of nucleated cells with few isolated anucleate cells. Neutrophils were dispersed and trapped in the pink-stained mucous. In Figure 2B, vaginal epithelium of a control female at lactation day 14-16 is shown with an increase in neutrophilic infiltration. This vaginal morphology was found in several control females in a few studies. In Figure 2C, a typical vaginal cytology sample of a normal diestrus (upper slide) of a female in the premating period and of an abnormal diestrus at lactation day 14-16 (lower slide) is shown. In most cases, the females at lactation day 14-16 typically show such large thick blue-stained mass which is absent in the normal diestrus vaginal samples. Vaginal cytology of a normal diestrus as demonstrated in Figure 2D shows a mixture of cells (nucleated or cornified epithelial cells and neutrophils). In the female presented here, the number of cells was relatively low and some mucus was present. The vaginal cytology samples of many females taken at lactation day 14-16 showed thick aggregates of mucus in which abundant cells (epithelial cells and inflammatory cells) were trapped (Figure 2E). Although all cell types were present, neutrophils were dominant. Also near the mucus aggregate, neutrophils were the predominant cell type and much more prominent than the normal diestrus based on vaginal cytology (Figure 2F compared with Figure 2D). The vaginal cytology sample at day 14-16 of lactation did not meet all the typical characteristics of a normal diestrus. However, the presence of the different cell types and mucus in smears from lactating females could easily be confused with a diestrus stage.
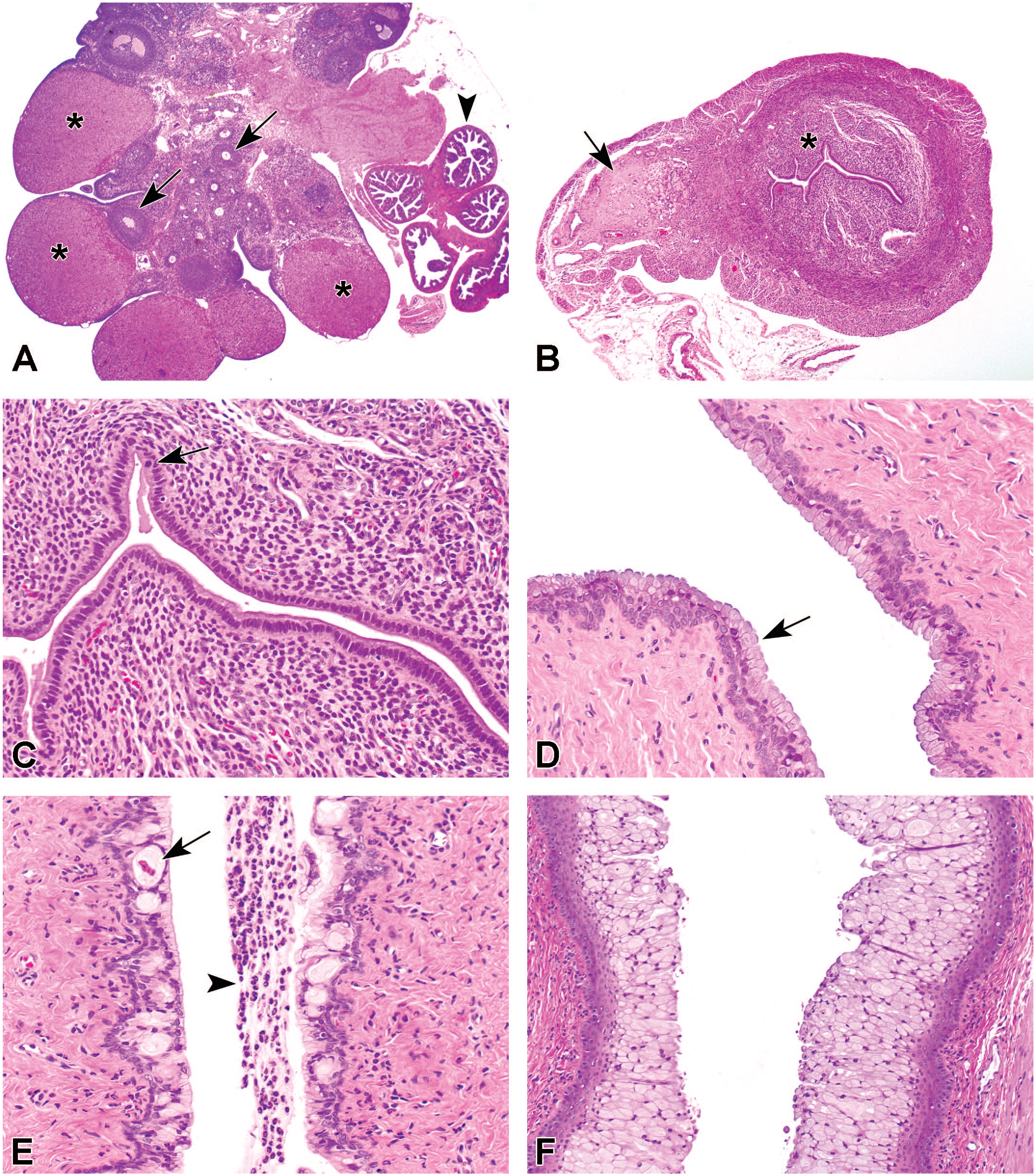
Female reproductive organs, typically for control females of a combined study (OECD 422) at lactation day 14-16. (A) Ovary contains many large corpora lutea with large luteal cells (asterisks) and small follicles (arrows). Arrowhead indicates the oviduct; OM objective 2X. (B) Uterus is relatively small, showing remnants of implantation sites (arrow) and a compact stroma (asterisk); OM objective 4X. (C) Higher magnification of uterus showing cuboidal luminal and glandular epithelium, a single cell may show mitotic activity (arrow); OM objective 20X. (D) Vaginal epithelium consisting of a basal layer of flat basal cells and a luminal layer of mucified cells; OM objective 20X. (E) Vaginal epithelium at lactation day 14-16 containing vacuolated cells (arrow) and granulocytes (arrowhead) in the lumen; OM objective 20X. (F) Marked mucification of the vaginal epithelium of a control female necropsied shortly after delivery with total litter loss; OM 20X. OM indicates original magnification.
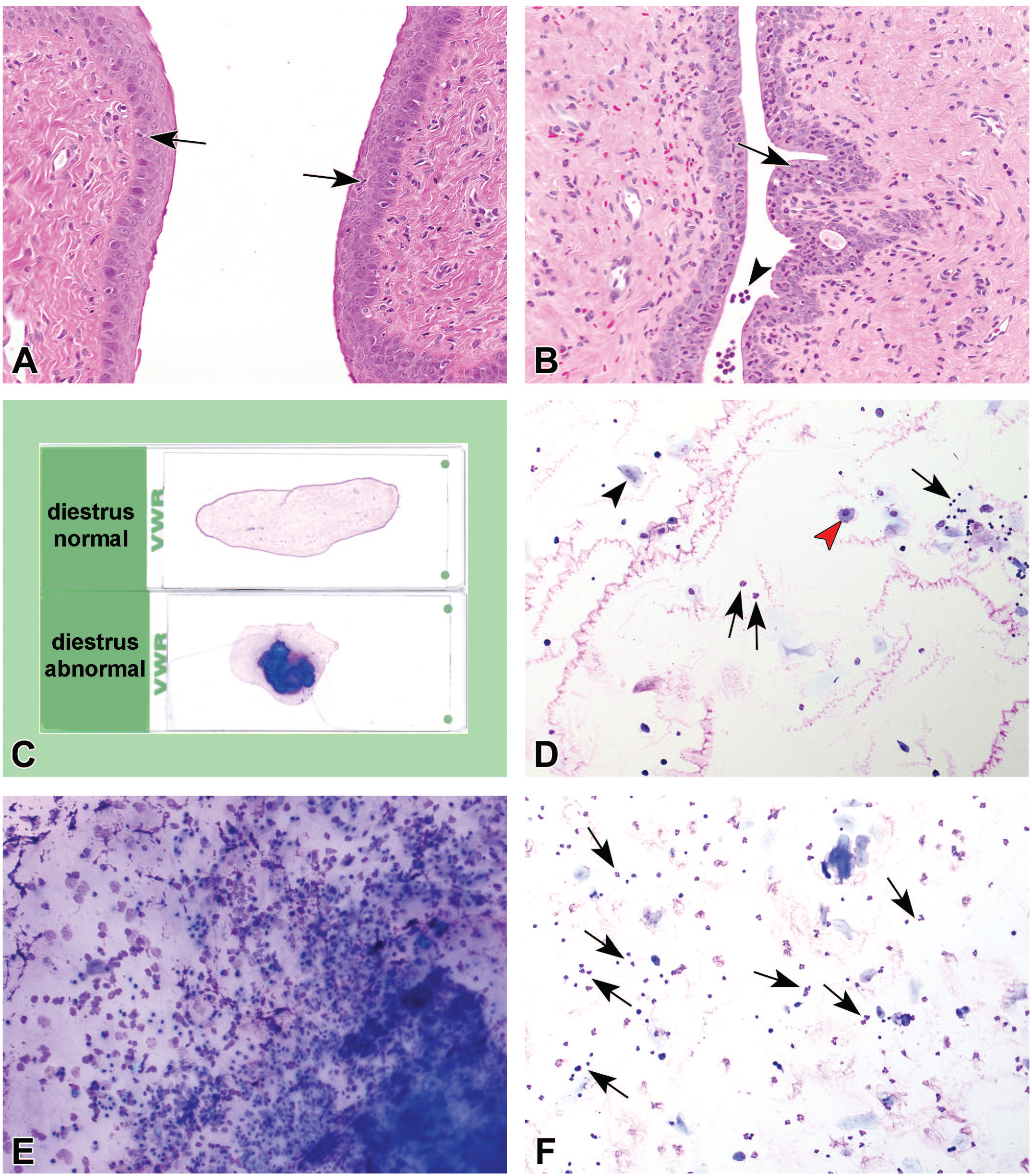
Comparison of vaginal morphology of a normal late diestrus (of a nulliparous control female) and of a “diestrus” at lactation day 14-16 (A, B), and cytology samples obtained by lavage of a normal diestrus (C-upper slide, D) and of a female necropsied at lactation day 14-16 (C-lower slide, E, F). (A) A 5 layered vaginal epithelium of a normal diestrus with some intraepithelial neutrophils (arrows). OM objective 20X. (B) A 2 to 3 layered vaginal epithelium with abnormal numbers of neutrophils in the vaginal epithelium (arrows) which are also present in the lumen (arrowhead); OM objective 20X. (C) A vaginal cytology sample of a female taken in the pretesting phase and staged as diestrus (upper slide) and of an abnormal diestrus at necropsy day of females at lactation day 14-16 (lower slide) with a thick mucus aggregate stained blue. (D) Normal diestrus from premating phase with generally less cells and mucus and equal numbers of cornified epithelial cells (black arrowhead), nucleated cells (red arrowhead), and neutrophils (arrows); OM objective 10X. (E) Vaginal cytology sample of a female taken at necropsy at lactation day 14-16 and staged as diestrus in the area of the thick aggregate containing mucous material in which abundant cells were trapped; OM objective 10X. (F) Vaginal cytology sample of a female taken at necropsy at lactation day 14-16 just near the blue-stained mucus aggregate consisting of different cell types but with prominent numbers of neutrophils (arrows); OM objective 10X. OM indicates original magnification.
Extended one-generation reproductive toxicity study (OECD 443)
F0-females were killed on lactation day 23-25, a few days after weaning of their offspring (postnatal day 21). In contrast to the females of the combined studies (which were killed on lactation day 14-16), vaginal samples at lactation day 23-25 were not all staged as diestrus and 15% to 30% of the females were in a different estrous stage. Similar to the selected females of the combined studies (killed at lactation day 14-16), ovaries, uterus, and vagina of the F0-females (killed at lactation day 23-25) were subjected to histopathologic evaluation, but in general no estrous stage could be assigned. This was only possible for the few control females that did not mate or failed to deliver healthy offspring. As only in the latter category (very few females), the estrous stage could be determined based on histopathology; the agreement between vaginal cytology and histology for F0-females in EOGRT studies was very low (less than 1%).
Females of the F1-generation (Cohort 1A) selected for histopathologic evaluation were generally killed at postnatal day 87-100 and were not part of the reproduction phase. In contrast to the F0-females, for all females of the F1-generation it was possible to assign a stage of the estrous cycle histologically. In Table 5, the agreement between estrous cycle stages determined by vaginal cytology and by histopathology of the reproductive organs are marked in gray. The mean agreement of 5 studies (n = 20 for each study) was 60% with a range of 47% to 70%.
Comparison of the estrous cycle stages based on vaginal cytology and on histology of multiple reproductive organs of the F1-generation of EOGRT studies (OECD 443 studies).
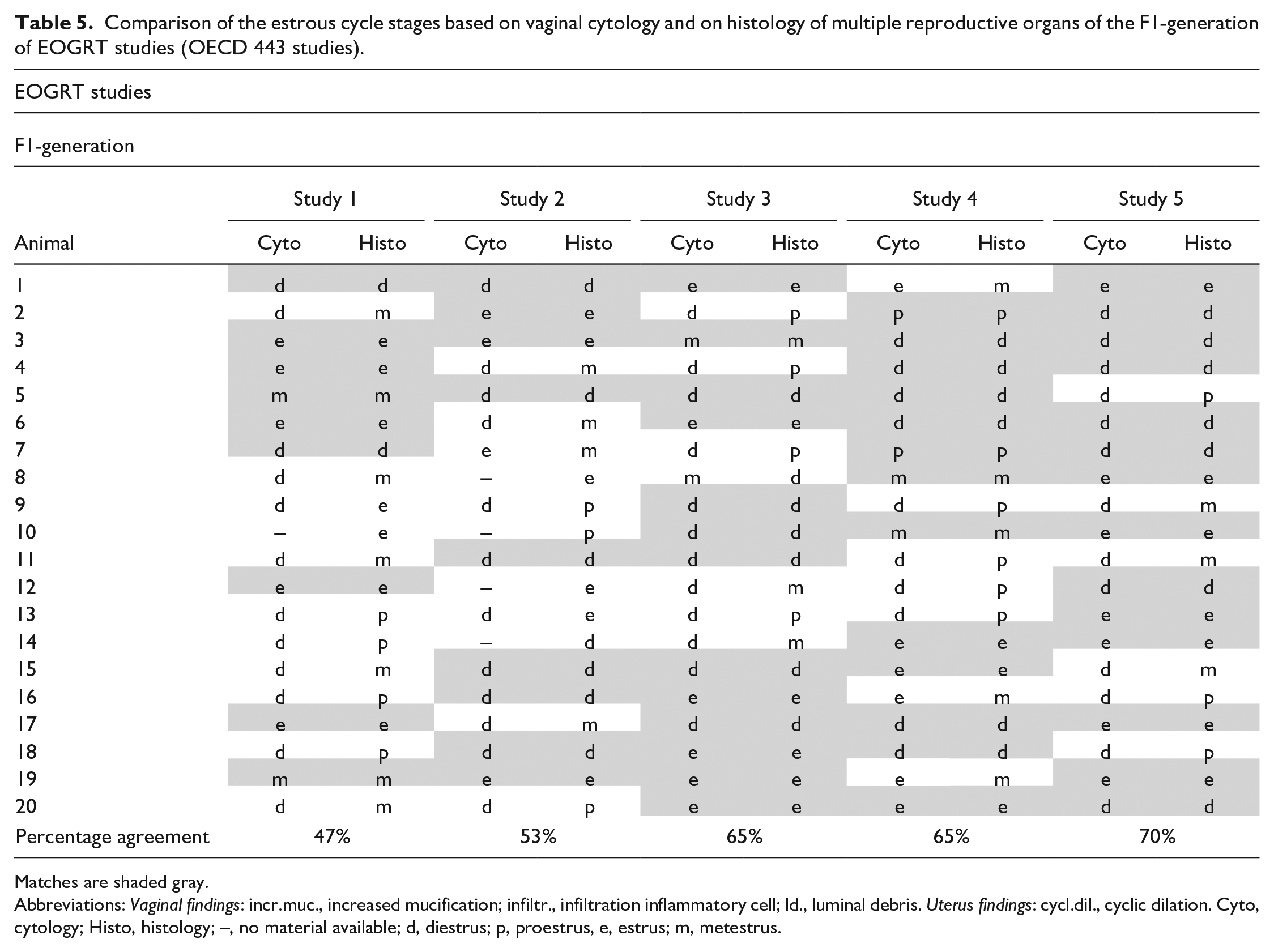
Matches are shaded gray.
Abbreviations: Vaginal findings: incr.muc., increased mucification; infiltr., infiltration inflammatory cell; ld., luminal debris. Uterus findings: cycl.dil., cyclic dilation. Cyto, cytology; Histo, histology; –, no material available; d, diestrus; p, proestrus, e, estrus; m, metestrus.
In Figure 3, the summary results of mean percentage agreement of estrous cycle stages between vaginal cytology and histology of multiple reproductive organs are given for the different study types. Agreement between the two methods was highest in 90-Day studies (45%) and the F1-generation of the EOGRT studies (60%) but these stages were still unexpectedly low. From the few non-lactating females of the 28-Day Combi-studies, Repro-Screening studies, and the F0-generation of the EOGRT studies, a normal cycle could be expected resulting in a possible agreement between vaginal cytology and histology. In the studies reviewed, this resulted in a mean agreement of 9% in combined studies, and less than 1% agreement in the F0-generation of the EOGRT studies.
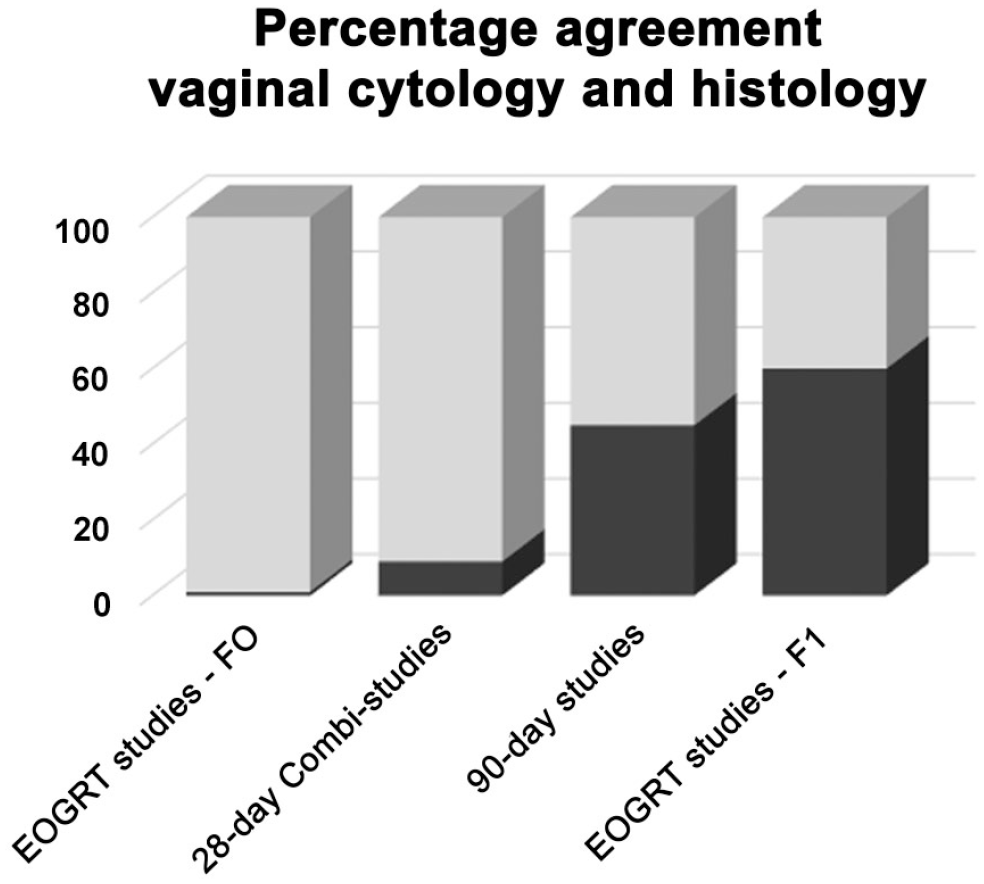
The summary results of the mean percentage agreement (black) per study type between the estrous cycle stages based on vaginal cytology and histology of multiple reproductive organs.
Discussion
The main goal of evaluating vaginal cytology is to obtain information on normal or abnormal estrous cycles. This is important in order to (1) include only females with regular cycles in a reproduction study, (2) prove that mating occurred, and (3) detect alterations in normal cycling of individual animals over time, particularly if treatment related.
Detecting changes in the estrous cycle can only be achieved when series of data are available and the pattern of the different cycle stages shows whether the estrous cycle of individual females is normal or abnormal (as shown in Table 2). Even when an estrous cycle stage based on vaginal cytology sometimes does not fit in the series, the regularity of an estrous stage every 4 days (in the case of a 4-day cycling rat) is a good indicator for a normal estrous cycle. 7
Different methods are available for the preparation of vaginal samples, and it is left to individual laboratories to decide which to use according to the skills, experience, and preferences of the staff. A sample can be obtained using a cotton-tipped swab or, alternatively, a vaginal lavage can be performed by gently flushing water or saline using a pipette. 5 It is generally accepted that vaginal lavage is less invasive, more gentle, and less stressful for the animals 11 and yields a higher cellularity sample. Therefore, it is considered to be the preferred method 12 and the authors would recommend to specify this in the OECD guidelines. The daily lavage procedure prior and during mating, together with vaginal cytology evaluation, is time-consuming and despite the fact that it is gentler than using a cotton swab, it still should be performed with care to avoid pseudopregnancy 13 and damage of the vaginal epithelium. 7 Although there are other methods to determine the estrous stage, such as visual assessments, vaginal wall impedance, and urine biochemistry, vaginal cytology remains the gold standard in live animals. 14
In most of the toxicity tests described in Table 1, evaluation of vaginal cytology is (also) required on the day of necropsy (i.e., OECD 408, 421, 422, 443).1-4 Arguments stated in the guidelines are (1) “Determination of the estrous cycle by taking vaginal smears will provide information regarding the stage of estrous cycle at the time of killing and assist in histological evaluation of estrogen sensitive tissues (OECD 408)” 1 and (2) “Vaginal smears should be examined in the morning on the day of necropsy to determine the stage of the estrous cycle and allow correlation with histopathology of ovaries (OECD 421) 2 or female reproductive organs (OECD 422 and 443).”3,4 A single vaginal cytology sample taken at the day of necropsy is only of limited value 7 and may help in defining a change in endocrine status by showing less or no variety in estrous cycle stages in treated rats compared with the control group which normally show a high variety in estrous cycle stages. However, these estrous cycle stages alone have limited value and should be interpreted together with other endpoints such as histopathology or hormonal changes. In contrast to estrous cycle staging based on vaginal cytology which rely only on exfoliated cells from the superficial vaginal mucosa, it should be noted that estrous staging based on histology incorporate the evaluation of the entire structure of all organs of the reproductive tract (i.e., ovary, uterus with cervix and vagina) with careful attention to the appropriate synchronicity across those organs specific to each stage and gives more reliable information on estrous cycle. Therefore, the histology of the entire female reproductive tract should be considered the gold standard for a single time point evaluation of the estrous cycle. 9 Vaginal cytology does not add any additional information to the histologic assessment and can in fact detract from the quality of the histology assessment.
In the present article, the utility of estrous cycle determination by vaginal cytology and histopathology was evaluated by comparing the two methods for the following study types: 90-Day study (OECD 408), 28-Day Combi-study (OECD 422), and EOGRT (OECD 443). Such a comparison was not possible for the Repro-Screening study (OECD 421). In this study type, only ovaries are selected for histopathologic evaluation; therefore, only abnormalities of the ovaries will be recorded by the pathologist. For a reliable staging of the estrous cycle in the rat based on histology, ovaries, uterus, and vagina should be examined and interpreted as a whole. 9 As stated in the guideline of Repro-Screening studies, vaginal cytology evaluation on the day of necropsy should be performed “to determine the stage of the estrous cycle and allow correlation with histopathology of ovaries.” Since no reliable estrous stage can be given based on histology of the ovaries alone and due to the fact that the females in this study type are necropsied at lactation day 14-16, they have not yet resumed a normal estrous cycle. Therefore, vaginal cytology of these animals will not represent a particular stage of the estrous cycle and evaluation does not have any value. Besides the Repro-Screening studies, also in combined studies and the F0-generation of the EOGRT studies, a correspondence between the estrous cycle stages of vaginal cytology and histopathology was lacking. This was mainly based on the lactating status of the females and the erroneously evaluation of “diestrus” cytology which in fact was lactational anestrus. The rare cases of agreement between vaginal cytology and histology were from females that were not lactating. From these non-lactating females, all reproductive organs were evaluated (even when they were not one of the five selected animals for histopathology as in combined studies). Necropsies of the females with healthy offspring from the combined studies and the F0-generation of the EOGRT studies were performed at lactation day 14-16 and lactation day 23-25, respectively. In most rat reproductive toxicity studies, pups are weaned at postnatal day 21, making the duration of lactation 21 days. 15 This means that in combined studies and in the F0-generation of the EOGRT studies, the females were necropsied within the lactation period (lactation day 14-16) or just after weaning (lactation day 23-25). It is well known that pregnancy followed by lactation includes mammary gland development, onset of lactation, and maintenance of milk secretion, which is regulated by many hormones of the endocrine system and influenced by the hypothalamus-pituitary axis. 16 Due to this enormous change in the normal hormonal pattern, the morphological features of ovaries, uterus, and vagina during lactation are completely different from the morphology seen in normal estrous cycle stages. The morphological characteristics of the reproductive tract of females during the lactation period were clearly visible in the example of females in combined studies, showing ovaries with many large corpora lutea, an inactive uterus with low cuboidal epithelium, and vaginal epithelium not corresponding with the known features of nulliparous animals.
Females killed at lactation day 23-25 (i.e., the F0-generation of the extended one generation studies) showed a high variability in morphology of the reproductive organs. Some females had normal cycle features, others not. This variability can be expected just after weaning of the pups as the stimulus for milk production is removed and the females begin to cycle again. At this time point, some females just had their first postpregnancy/lactation ovulation. This strongly indicates that in combined studies and F0-Generation in EOGRT studies, there is no added value to determine the estrous stage at the day of necropsy in females with offspring.
Both 90-Day studies and the F1-generation of EOGRT studies showed a much higher percentage agreement of estrous cycle stages determined by vaginal cytology and histopathology. In contrast to the studies discussed above, the female rats of the 90-Day studies and the F1-generation of EOGRT studies are nulliparous, sexually mature, and generally have a normal regular cycle. Therefore, a higher percentage of agreement between cytology and histopathology would be expected. However, this was still lower than anticipated (45% in 90-Day studies and 60% in F1-generation of EOGRT studies). This low agreement is in accordance with similar evaluations in mice of vaginal cytology and vaginal histology at the day of necropsy. 17 A comparison between these methods resulted in a percentage agreement of 55% for diestrus, 77% for proestrus, 46% for estrus, and 31% for metestrus. It has well been described that some estrous stages have a short duration 12 (e.g., metestrus of approximately 6 hours) and dependent on the time difference between the lavage procedure and necropsy, the female may already be in the next stage. Another possible explanation for discordance is that manipulation of vaginal epithelium during lavage may impact the epithelial morphology (e.g., increased inflammatory cells and in rare cases erosions of the epithelium or removal of the superficial cornified layer characteristics of estrus) 11 which may result in misinterpretation of the histopathology. Disagreement of vaginal cytology and histology may also occur due to the fact that vaginal epithelial morphologic changes during the estrous cycle is a continuum. The cytological classification of the estrous stages is based on differences in the proportions of anucleate and nucleated epithelium cells, and neutrophils.7,12,18 and the transition between two consecutive estrous stages is continuous and not abrupt.12,19 It would therefore not be surprising to find higher rate of discordance between successive estrous cycle stages, as was the case for many disagreements in the 90-Day studies.
As mentioned earlier, frequent vaginal manipulation can induce pseudopregnancy 13 with subsequent mucification of the vaginal epithelium 20 and if not performed accurately vaginal lavage or swabbing may cause damage to the vagina and/or incite an inflammatory response. 21 The handling that is needed for taking vaginal cytology samples is potentially stressful for the animals and should be avoided when unnecessary. Therefore, one must make a well-considered decision as to what study type and at which time points vaginal cytology has essential added value.
Evaluation of a single vaginal cytology sample collected on the day of necropsy is just a snapshot in the estrous cycle. Based on such a single estrous cycle stage, it is impossible to say whether a female rat has a normal cycle (i.e., appropriate sequence and duration of stages) and this single vaginal cytology sample does not give any more information than the histologic evaluation. Moreover, histologic examination of the complete female reproductive system (ovaries, uterus/cervix, and vagina) is much more accurate in staging of the estrous cycle in the rat than vaginal cytology and can also distinguish morphologic abnormalities in the reproductive organs and asynchrony between the various organs. This is also confirmed by the document provided by the OECD 5 stating that diestrus is very difficult to recognize by vaginal cytology as this stage is highly variable and is also diagnosed as “resting-stage” in pseudopregnant and acyclic females. So, a diestrus based on vaginal cytology can be either diestrus of a normal cycle, absence of a normal cycle (e.g., as is the case in lactating females), or a sign of an abnormal estrous cycle. This strengthens the opinion of the authors that a single time point vaginal cytology evaluation does not give reliable information on the status of female cyclicity, lactating status, or abnormalities in the female reproductive organs. In contrast, histopathologic evaluation of the reproductive organs sampled at the day of necropsy gives significantly more valuable information. Finally, it should also be taken into account that handling the animal is needed to take vaginal cytology samples and can be stressful, it takes time and effort from the technicians and it by its nature (removal of cells, physical manipulation) changes the vaginal morphology and thus also possibly the histopathological diagnosis. Therefore, the necessity to perform vaginal cytology on a single time point (on the day of necropsy) for studies with histopathology of the female reproductive tract is of no added value. In addition, a single time point evaluation of vaginal cytology does not allow recognition of lactating females and will generally be diagnosed as diestrus which suggests a normal cycle.
In conclusion, vaginal cytology evaluation in a study is important for the evaluation of normal estrous cyclicity during the in-life phase and is necessary in studies with a reproduction phase as part of the study design. The evaluation of the estrous cycle is only possible if serial daily samples are taken. A single isolated estrous stage based on vaginal cytology is unreliable and of no added value: it does not give any additional information on the estrous cyclicity or abnormal findings in the female reproductive tract and increases handling stress and should therefore not be recommended, especially when the reproductive tract is also evaluated histologically at the same time point. In addition, trying to stage a cycle by vaginal cytology when there is not yet a normal cycle expected, as is the case in lactating rats, results in inaccurate estrous staging. Based on these arguments, the authors recommend to adapt the Guidelines by omitting the requirement for the evaluation of vaginal cytology taken on the day of necropsy in the study types discussed in the present article (i.e., OECD 408, 421, 422, and 443).
Footnotes
Acknowledgements
The authors thank Inge de Ronde for her support in evaluating and selecting the vaginal cytology samples prepared from the vaginal lavages and the colleagues of the histology laboratory for preparing the slides for histopathology evaluation and the colleagues of Charles River Laboratories for their contribution in the studies evaluated in the present article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
