Abstract
The cynomolgus macaque is the most commonly used nonhuman primate in nonclinical toxicity testing, but the impact of the geographic source of cynomolgus macaque on differences in spontaneous pathology and response to xenobiotics has only recently been explored. Previous work from the authors' facility has described spontaneous cardiac findings in predominantly Indonesian-source animals; however, the authors have recently observed a novel spectrum of cardiac findings in Mauritian-source animals. This review evaluated the spontaneous macroscopic and microscopic cardiac findings in vehicle control Mauritian-source macaques used for routine toxicity testing. When compared to the prior review in predominantly Indonesian macaques, a higher incidence of myocardial degeneration was observed with additional novel findings including macroscopic and microscopic subendocardial hemorrhage with hemosiderin, myocardial fibrosis, and arterial medial degeneration/hemorrhage. Other findings including inflammatory cell infiltrates, anisokaryosis, and squamous plaques were observed with a comparable incidence as previously reported in Indonesian macaques. Myocardial degeneration, subendocardial hemorrhage, and myocardial fibrosis can mimic test-article-related cardiac toxicity, and a thorough understanding of the incidence and severity of these spontaneous findings is necessary to prevent misidentifying test-article-related cardiac findings in this genetic source of cynomolgus macaque in nonclinical safety testing.
Introduction
Morphologic evaluation of the heart in nonclinical safety testing is a key component in identifying cardiovascular toxicity and understanding the potential risk of cardiac effects when administering drug candidates to humans. The cynomolgus macaque (Macaca fascicularis) is the most commonly used nonhuman primate in toxicity testing, and captive stocks are available from three geographically separated and genetically distinct populations: the mainland Indochinese animals (Vietnam, Cambodia, and Thailand), which are commonly bred in Chinese facilities; the insular Malaysian and Philippine animals; and the introduced population on the island of Mauritius (Stevison and Kohn 2008). These three sources of cynomolgus macaques display distinct differences ranging from physical characteristics, hematology parameters, immunologic response to organ transplantation, to spontaneous pathology findings (Menninger et al. 2002; Drevon-Gaillot et al. 2006; Schillaci et al. 2007). A thorough understanding of spontaneous findings in the cynomolgus macaque must take into consideration the genetic background of the animals used in nonclinical toxicity testing. Previous work from our facility detailed the spontaneous cardiac findings in cynomolgus macaques from Chinese, Indonesian, and Mauritian breeders (Keenan and Vidal 2006); however, more than 90% of the animals in the review were of Indonesian origin, and a spectrum of unreported cardiac findings have been observed in control Mauritian cynomolgus macaques at GlaxoSmithKline (Vidal, personal observation). To better characterize the spontaneous cardiac pathology of Mauritian source animals, we conducted a review of the macroscopic and microscopic findings in exclusively Mauritian animals.
Materials and Methods
All procedures and care of animals were in accordance with the principles for humane care outlined by the Institute of Laboratory Animal Resources' Guide for the Care and Use of Laboratory Animals, the USDA Animal Welfare Act, and were reviewed and approved by the GlaxoSmithKline Institutional Animal Care and Use Committee.
Cynomolgus macaques (29 males and 42 females) aged 2.5–7 years from general toxicity studies with durations of four days to three months were used for this study. These macaques received vehicle only by either the oral, intravenous, or subcutaneous routes of administration. The macaques were obtained from Covance Research Products, Inc. (Alice, TX, USA) and Charles River Laboratories (Houston, TX, USA). Macaques originated from Mauritius, and all were captive-bred and raised in groups outdoors in their country of origin before shipment to the United States. In our facility macaques were singly housed in climate-controlled rooms at 64–84°F, with a relative humidity of 30–70%. The rooms had approximately ten to fifteen air changes per hour and a photoperiod cycle of twelve hours light/twelve hours dark. Macaques were fed with a commercially available complete diet and generally received eight (male) or six (female) biscuits/day. A daily allotment of fresh fruit was also provided. Water was provided ad libitum.
Prior to necropsy, each macaque was pretreated with an intramuscular or subcutaneous injection of ketamine HCl (approximately 10 mg/kg), anesthetized with an intravenous injection of sodium pentobarbital (starting at approximately 30 mg/kg), and then exsanguinated. All necropsies were conducted under the supervision of a veterinary pathologist. The heart was carefully removed from the thoracic cavity, rinsed with saline, and all four chambers were opened to allow for visual inspection of the valves and the endocardial surface. Photographs of selected hearts were taken, and the heart was then immersed in 10% neutral buffered formalin and fixed for twenty-four hours at room temperature. The time from exsanguination to immersion fixation was generally less than thirty minutes.
Following fixation, the heart was trimmed to include the left ventricle with papillary muscle, left atrium, and mitral valve; interventricular septum with aortic outflow tract and aortic valve; and right ventricle and atrium with tricuspid valve. Tissues were processed to paraffin and 5 micron sections were cut, placed on Superfrost Plus slides (VWR Scientific, West Chester, PA, USA), and stained with hematoxylin and eosin (H&E). Perl’s iron stain and Masson’s trichrome were performed on a subset of animals to confirm the presence of hemosiderin and fibrosis, respectively (Prophet et al. 1994). All slides were evaluated by a single veterinary pathologist (Vidal), and a peer review was conducted to ensure that microscopic changes were recorded consistently.
Results
Macroscopic Evaluation
Red discoloration was observed within the left ventricle of 13% of the hearts examined at necropsy. This change ranged from focal to multifocal and was most prominent within the interventricular septum below the aortic valve (Figure 1 ) and within the papillary muscles adjacent to the insertion of the chordae tendineae.
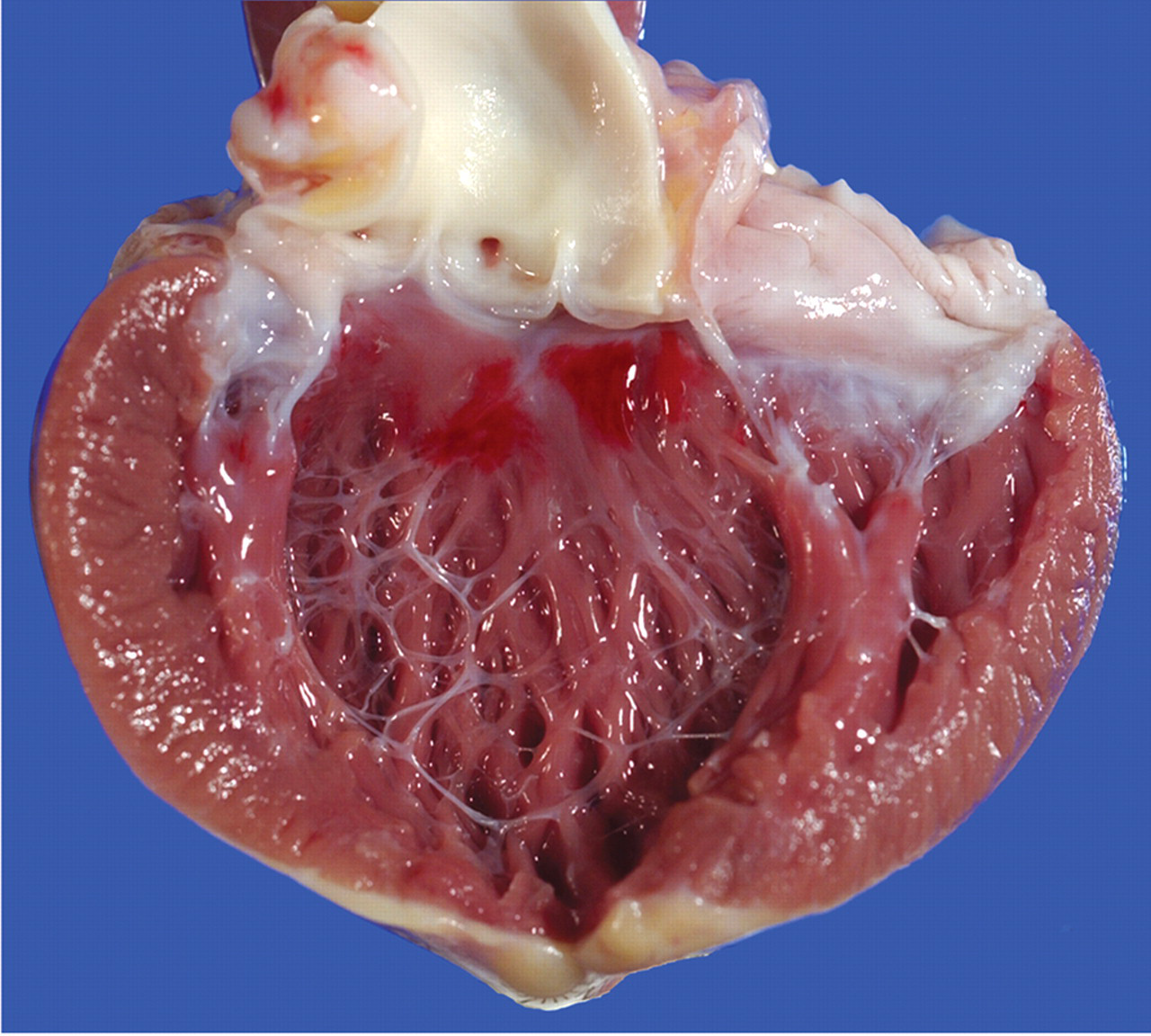
Subendocardial hemorrhage in the heart of a Mauritian-origin cynomolgus macaque. The left ventricle has been opened and the left AV valve transected to demonstrate multiple red foci within the interventricular septum below the aortic valve.
Microscopic Evaluation
Microscopic observations for all animals are summarized in Table 1 .
Incidence of microscopic findings in control male and female Mauritian-origin macaques.

Myocardial degeneration/necrosis was observed with a total incidence of 13% and was most commonly present within the papillary muscles of the left ventricle and occasionally within the free wall of the left ventricle or the interventricular septum. These findings varied from individual degenerate myofibers surrounded by a small number of inflammatory cells (Figure 2A) to multiple variably coalescing foci of necrosis (Figure 2B and 2C).
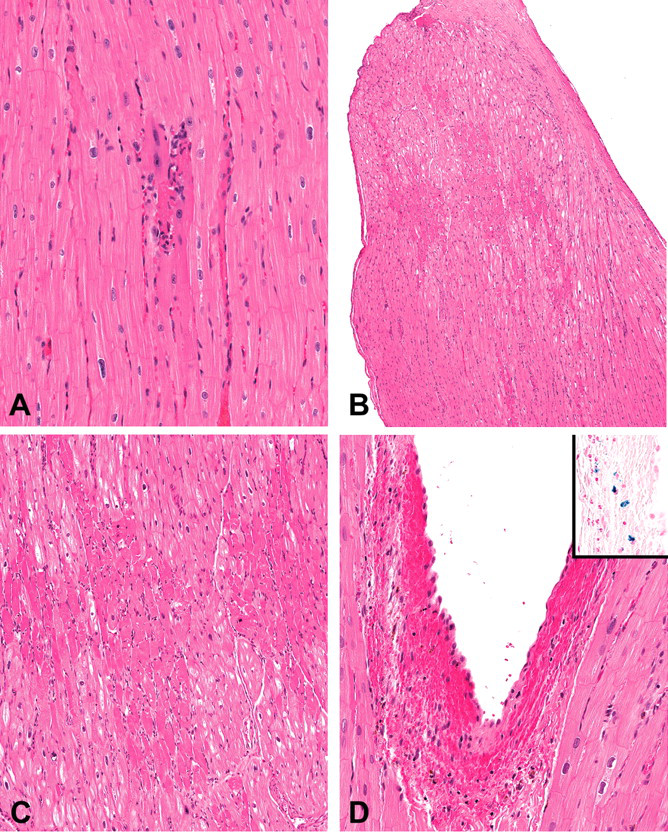
Representative photomicrographs of various spontaneous findings within the heart of Mauritian-origin cynomolgus macaques. (A) Individual degenerate myofiber surrounded by a small number of inflammatory cells within the papillary muscle. (B and C) Multiple variably coalescing foci of necrosis within the distal half of the papillary muscle. (D) Subendocardial hemorrhage within the papillary muscle of the left ventricle. Inset: Perl’s iron stain demonstrating the presence of hemosiderin within the focus of subendocardial hemorrhage. (E) Focal fibrosis within the papillary muscle. (F) Masson’s trichrome on an adjacent section of (E) demonstrates multiple variably coalescing foci of collagenous connective tissue that separate and replace myofibers. (G) Medial degeneration of an intramural coronary artery with cellular debris and medial hemorrhage.
Subendocardial hemorrhage was observed with a total incidence of 30% and consisted of variable amounts of free hemorrhage between the endocardium and underlying cardiac muscle of the left ventricle and was most commonly observed within the papillary muscles of the left ventricle and the interventricular septum below the aortic valve (Figure 2D). This finding varied from a small focus of subendocardial erythrocytes to larger foci of free hemorrhage that separated underlying cardiac myofibers. In addition to the subendocardial hemorrhage, a single macaque displayed hemorrhage underlying the epicardium of the left atrium.
A tan to golden brown pigment was observed with a total incidence of 17% and was most commonly observed within the papillary muscles of the left ventricle and the interventricular septum. This finding varied from a small focus of individual histiocytes with intracellular pigment to larger foci of free pigment. Often the pigment was associated with hemorrhage and occasionally with erythrophagacytosis. A Perl’s iron stain was performed on three animals, and the above described pigment was confirmed to be hemosiderin (Figure 2E inset).
Fibrosis of the papillary muscle or the free wall of the left ventricle was observed with a total incidence of 4%. This finding consisted of the replacement of myofibers with dense collagenous connective tissue with embedded multiple variably sized blood vessels, and the surrounding myofibers were often enlarged, disorganized, and displayed anisokaryosis (Figure 2E). A Masson’s trichrome stain was performed, and the collagenous connective tissue displayed an intense staining reaction (Figure 2F).
Medial degeneration and/or hemorrhage of the coronary arteries was observed with a total incidence of 6% and was present in intramural vessels of either the right or left ventricles. The finding was characterized by cellular debris, hemorrhage, and/or regeneration within a focal area of the vascular smooth muscle (Figure 2G). One of the four affected animals displayed only hemorrhage within the media.
Inflammatory cell infiltrates were observed with a total incidence of 40% and were randomly distributed throughout all portions of the heart and pericardial fat. The cellular composition varied but was most often lymphoplasmacytic or lymphohistiocytic and rarely eosinophilic.
Anisokaryosis and karyomegaly were observed with a total incidence of 27% and were most commonly observed within the left ventricle and papillary muscle.
Squamous plaques/cysts were observed with a total incidence of 10% and were present on the epicardial surface of the atria and/or coronary grooves.
Pericardial reactivity was observed with a total incidence of 3% and consisted of enlarged, reactive epicardium and opposing pericardium (present attached to thymus).
A single macaque displayed abundant adipose present throughout the heart but most prominently within the right ventricle.
Discussion
Previous work from our facility suggested a low incidence of spontaneous myocardial degeneration and did not identify subendocardial hemorrhage in cynomolgus macaques (Keenan and Vidal 2006); however, in recent years these findings have been observed with an apparent increase in incidence and severity in both control and test article treated macaques in our facility. A thorough understanding of the type and incidence of spontaneous background findings is critical to differentiate these from test-article-induced pathology. Differences in the geographic origin of cynomolgus macaques has been associated with differing incidence and type of various spontaneous findings (Drevon-Gaillot et al. 2006), and due to a shift in the source of animals used at GlaxoSmithKline, a current review of spontaneous cardiac findings was performed in Mauritian-source animals.
A spectrum of unique findings was observed including myocardial degeneration/necrosis, subendocardial hemorrhage, and accumulation of pigment (consistent with hemosiderin) and less frequently myocardial fibrosis. Myocardial degeneration/necrosis was observed with an overall incidence of 13%. The previously published review of spontaneous background findings in the heart of macaques (primarily Indonesian in origin) used in our facility demonstrated an incidence of myocardial degeneration/necrosis of 4–5% (Keenan and Vidal 2006). The myocardial degeneration/necrosis observed in Indonesian animals in the previously published review involved individual to small groups of myofibers with or without a surrounding inflammatory response. In contrast, the myocardial degeneration/necrosis observed in Mauritian-origin macaques was increased in incidence and severity and ranged from individual myofibers to large variably coalescing foci of necrosis. In addition, three Mauritian-source macaques demonstrated remodeling and replacement of portions of the papillary muscle or the left ventricular free wall with fibrous connective tissue, which likely represents the chronic outcome of the more extensive examples of degeneration/necrosis observed in this source of macaques.
Subendocardial hemorrhage was observed at necropsy with an incidence of 13% and an overall microscopic incidence of 30% in the current study, but it was not identified as a finding in the previously published review from our facility. In addition, aggregates of intrahistiocytic and/or free hemosiderin were observed with or without the hemorrhage. Subendocardial hemorrhage can be considered to represent an agonal change in some species; however, the presence of hemosiderin suggests that hemorrhage occurred at least several days prior to necropsy (Epstein et al. 2001). In addition, the presence of acute free hemorrhage, erythrophagacytosis, and hemosiderin-laden histiocytes in the same section of heart suggests that multiple episodes of hemorrhage may have occurred.
Myocardial degeneration/necrosis and subendocardial hemorrhage are commonly reported findings in a variety of species and associated with elevations in catecholamines (Cowan, Giddens, and Reichenbach 1983; Samuels 2007). In humans, subendocardial hemorrhage and myocardial degeneration are commonly observed following cerebral injury secondary to abrupt elevations of catecholamines (Lewis and Silver 2001; Samuels 2007). In rhesus macaques, infusion of norepinephrine has been shown to induce subendocardial/subepicardial hemorrhage and/or myocardial necrosis with subsequent remodeling and fibrosis (Khullar et al. 1989). In addition, subendocardial hemorrhage has been observed in rhesus macaques as a sequela to experimental surgical manipulation (Melville et al. 1969), and myocardial degeneration has been observed in squirrel monkeys following environmental stress (Corley et al. 1973). The findings observed in this review are consistent with those reported secondary to elevations in catecholamines and document the higher incidence of these findings in macaques of Mauritian origin than that observed in a published review from our facility using primarily Indonesian-source macaques. An increased proportion of macaques used being of Mauritian origin correlates with the increased incidence of these findings observed at our facility. It is unclear why these animals display a propensity to develop these lesions, but Mauritian-origin cynomolgus macaques are a genetically distinct population derived from a small founding group that has been isolated from other cynomolgus macaques for several hundred years and may respond differently to stress than their mainland counterparts (Bonhomme et al. 2008; Krebs et al. 2005).
Four of the macaques in this study had evidence of medial degeneration and/or hemorrhage within the coronary arteries. This finding was not observed in the prior review of Indonesian source macaques; however, the low incidence and focal nature of the lesions make comparisons difficult. Other findings including inflammatory cell infiltrates, anisokaryosis, and squamous plaques were observed with a comparable incidence as previously reported in Indonesian macaques.
When evaluating the incidence of spontaneous findings in cynomolgus macaques, the geographic origin of the animals must be taken into consideration and can have significant impact on the type and incidence of a variety of findings (Drevon-Gaillot et al. 2006). While the defined genetic status and Herpes B–, SIV-, and SRV-virus-free status of Mauritian origin animals are attractive characteristics for a variety of experimental studies (Burwitz et al. 2009; Krebs et al. 2005), the relatively high incidence of spontaneous cardiac findings (myocardial degeneration and subendocardial hemorrhage), which can mimic test-article-related cardiac toxicity, could potentially complicate the use of this genetic source of cynomolgus macaque in nonclinical safety testing without a thorough understanding and recognition of these spontaneous findings.
Footnotes
Acknowledgments
The authors thank Dr. Patrick Wier and Dr. Michael Ringenberg for conducting the pathology peer review, Beverly Maleeff and Terra Lutz for photographic support, and Kenneth Walker and Donna Haile for technical support.
