Abstract
To investigate the influence of geographical origin, age, and sex on toxicologically relevant spontaneous histopathology findings in cynomolgus macaques (Macaca fascicularis), we performed a comparative analysis of historical control data (HCD) from 13 test sites that included 3351 animals (1645 females and 1706 males) sourced from Mauritius, China, Vietnam, and Cambodia, aged from 2 to 9.5 years, and from 446 toxicology studies evaluated between 2016 and 2021. The most common findings were mononuclear infiltrates in the kidney, liver, brain, and lung, which showed highest incidences in Mauritian macaques, and heart, salivary glands, and gastrointestinal tract (GIT), which showed highest incidences of mononuclear infiltrates in mainland Asian macaques. Developmental and degenerative findings were more common in Mauritian macaques, while lymphoid hyperplasia and lung pigment showed higher incidences in Asian macaques. Various sex and age-related differences were also present. Despite origin-related differences, the similarities in the nature and distribution of background lesions indicate that macaques from all geographical regions are suitable for toxicity testing and show comparable lesion spectrum. However, in a toxicity study, it is strongly recommended to use animals from a single geographical origin and to follow published guidelines when using HCD to evaluate and interpretate commonly diagnosed spontaneous lesions.
Keywords
Introduction
The laboratory nonhuman primates (NHP) play a crucial role in the nonclinical safety assessment of pharmaceuticals and vaccines, as the most phylogenetically proximate animal model to humans.1,2 The cynomolgus macaque, which is the most common laboratory NHP species used in toxicology studies that shares many anatomic, physiological, and immunological similarities to humans, is often used as a pharmacologically relevant laboratory animal species in biopharmaceutical drug development and some vaccine research models. 3 However various factors, such as ethical considerations and responsible and efficient use of animals, 4 the increase in biotherapeutic development, and supply shortages in the face of increased demand, have sometimes resulted in the use of lower numbers of cynomolgus macaques per group in a study relative to rodent studies.2,5 For example, dose-escalation studies with monoclonal antibodies often use only one animal per group. 4 In addition, the current demand for laboratory macaques has also led to the increased use of animals sourced from different and more diverse geographical regions and/or suppliers, including various breeding colonies and multiple holding facilities. 5 Some of the issues are also pertinent to dog studies. Other direct or indirect consequences of the current demand for NHP has been the mixing of animals from different geographic origins or suppliers or animals from the same source but of a wider age range, within a development program, or even within a study, or the placement of studies at geographically diverse test facilities which may have varying animal husbandry practices. 5 These practices have resulted in the potential introduction of multiple sources of variability within development programs or studies, raising concerns that the results and interpretation of the study data and subsequently, toxicology study outcomes might be affected.3,6 The concerns are based on the fact that, in nonclinical safety testing, the origin and genetic background of the animal are some of the experimental variables that may affect the response to other study variables, such as the pharmacological responses or the presence of spontaneous pathology findings, which may in turn affect the interpretation of study results. Laboratory-reared cynomolgus macaques are sourced from a number of different populations with inevitable genetic variability. 7 However, while a wealth of genetic, genomic, and bioinformatics information has been developed for rhesus macaques, there is less information on cynomolgus macaques.3,7 It is known, though, that captive stocks of cynomolgus macaques originate from at least one of the three following populations: (1) native Indochinese or mainland macaques, which originate from Thailand, Cambodia, or Vietnam, and are frequently captive-bred in China, and have a higher genetic variation; (2) macaques from the southern region of the Isthmus of Kra into insular Asia and Philippines which are more homogenous compared to Indochinese macaques; and (3) non-native cynomolgus macaques on the Island of Mauritius, which have a very low genetic diversity when compared to native Asian macaques, presumably due to the founding effects (Mauritian macaques were derived from an estimation of around 20 founding animals), and total isolation.8-10 Due to these geographical origins, Indochinese or mainland macaques are alternatively referred as “continental,” while those from the Philippines, Indonesia, and Mauritius are often referred to as “insular” or in the case of those from Mauritius referred to as “Island” macaques. The comparatively poor genetic polymorphism in macaques from Mauritius is believed to represent a more inbred population of macaques, while the high genetic diversity in Indochinese or mainland macaques is believed to be at least in part, due to the gene flow from rhesus to cynomolgus macaques. Hence, cynomolgus macaques from China, Vietnam, or Cambodia share genetic similarities with rhesus macaques, while cynomolgus macaques derived from insular Malaysia and Philippines, and Mauritian macaques are free of the genetic influx from rhesus macaques.8-10 As a result of these differences in genetic variation, animals derived from each of these geographical origins have been reported to demonstrate distinct differences in clinical pathology parameters,11-13 response to pathogens, 14 response to vaccines, 10 differences in microbial flora, 6 and differences in spontaneous pathology findings.5,11,15
Although, as mentioned above, a few reports on the influence of geographical origin on spontaneous pathology findings have been published,11,15,16 the majority of the presently available literature on the background lesions in laboratory macaques has in general paid very little consideration on the influence of these factors on the nature and incidences of background or induced pathology. 17 In addition, the available literature on this subject is based on a limited number of animals and studies, usually from a single site, and with little to no effort to minimize other potential sources of bias, such as, study type, age, animal husbandry, or pathologist’s bias, which can be institutional. Furthermore, the translational relevance of spontaneous pathology findings and the significance of the observed origin-related differences are seldom addressed.
While the genetics and origin of macaques are key factors that should be controlled for when comparing studies within programs, environmental factors, such as age, diet, water quality, animal husbandry practices and housing, types of studies and dose routes, the geographical location of the test facility and its local ecosystem, and exposure to pathogens at the supplier, or holding facility, should also be taken into consideration. For instance, environmental stressors, such as group-housing of incompatible animals, outdoor housing, diet, or exposure to environmental pathogens, might play a larger role in the incidence and severity of inflammatory infiltrates in the GIT than the animal’s origin and genetics,6,18-21 while social stress and exposure to the protozoan parasite Trypanosoma cruzi (Chagas disease) might influence the incidence of heart lesions.22-24 Therefore, a more accurate comparison of the differences in spontaneous pathological findings in NHP from diverse geographic origins should strive to minimize the effects of environmental factors, harmonize the terminology, and attempt to reduce the effects of pathologist’s bias by conducting a multisite and global-reaching review that captures a much wider database.
The main objective of this retrospective study was to investigate, characterize, and report the possible relationship of geographical origin on the incidences of spontaneous histopathology findings in captive-bred cynomolgus macaques by carrying out a comprehensive analysis of historical histopathology incidences from animals sourced from four different geographical regions. Further ancillary objectives were to investigate and highlight the effects of other animal and environment-related factors that can modulate the incidences of spontaneous histopathology findings, such as age, sex, and local microenvironment. The ultimate objective was to provide readily accessible information on the origin-related differences of toxicological relevance in cynomolgus macaques, and some recommendations on the most efficient utilization of animals from different geographical origins, in the current environment of supply shortage, thereby contributing to a more efficient and responsible utilization of laboratory NHP. This study represents the largest bicontinental (Europe, North America, and one site in China), multisite retrospective study to date, to document the incidences and prevalence of spontaneous histopathology findings in laboratory cynomolgus macaques, in the context of geographical origin, age, sex, and animal husbandry.
Materials and Methods
Animals and Geographical Origin
A total of 3351 control cynomolgus macaques (1645 females and 1706 males) from 446 repeat-dose preclinical toxicology studies conducted and evaluated between June 2016 and June 2021, at Charles River Laboratories (CRL) and Labcorp Drug Development (formerly Covance) facilities in North America (CRL: Ashland, Reno, Shrewsbury, Senneville, and Sherbrooke; and Labcorp: Madison, Somerset and Greenfield), and Europe (CRL: Edinburgh, UK and Lyon, France; Labcorp: Harrogate, UK, Huntingdon, UK, and Labcorp Early Development Services Munster, Germany). Study durations ranged from single doses, followed by a 2-week recovery period to 39 weeks.
All animals were purpose-bred for laboratory use, from various suppliers approved by the relevant test facility ethical committees. The majority originated from breeders located in one of the following three most common geographical regions from which to source macaques for laboratory use: China (1338 animals), Vietnam (503 animals), and Mauritius (1340 animals). The number of animals of each geographical origin from each facility is shown in Table 1. These data also included low numbers of animals from other sources, such as Cambodia, but were not included in the analysis of geographical origin (but included in the analysis of sex and age) due to insufficient sample size. All animals were controls that had been vehicle-dosed using various dose routes (oral gavage, oral capsule, intravenous bolus or continuous injection, intramuscular, subcutaneous, intradermal, intrathecal, intravitreal injection, or ocular instillation), and the number of animals dosed by each route is shown in Table 2. To rule out the influence of study procedure- or dose route-related changes, data were analyzed with and without some specific dose routes, such as the intrathecal (for brain and spinal cord) and inhalation (for lung) routes. The influence of other dose routes on other tissues was not assessed. Animals were dosed with an appropriate vehicle that included empty or gelatin capsules for the oral capsule route, carboxymethylcellulose or drinking water for the oral gavage route, and phosphate-buffered saline, physiological saline, and acetic or citrate vehicles for intravenous or subcutaneous injection routes.
Number of control cynomolgus monkeys (Macaca fascicularis) contributed by each test facility separated by source.
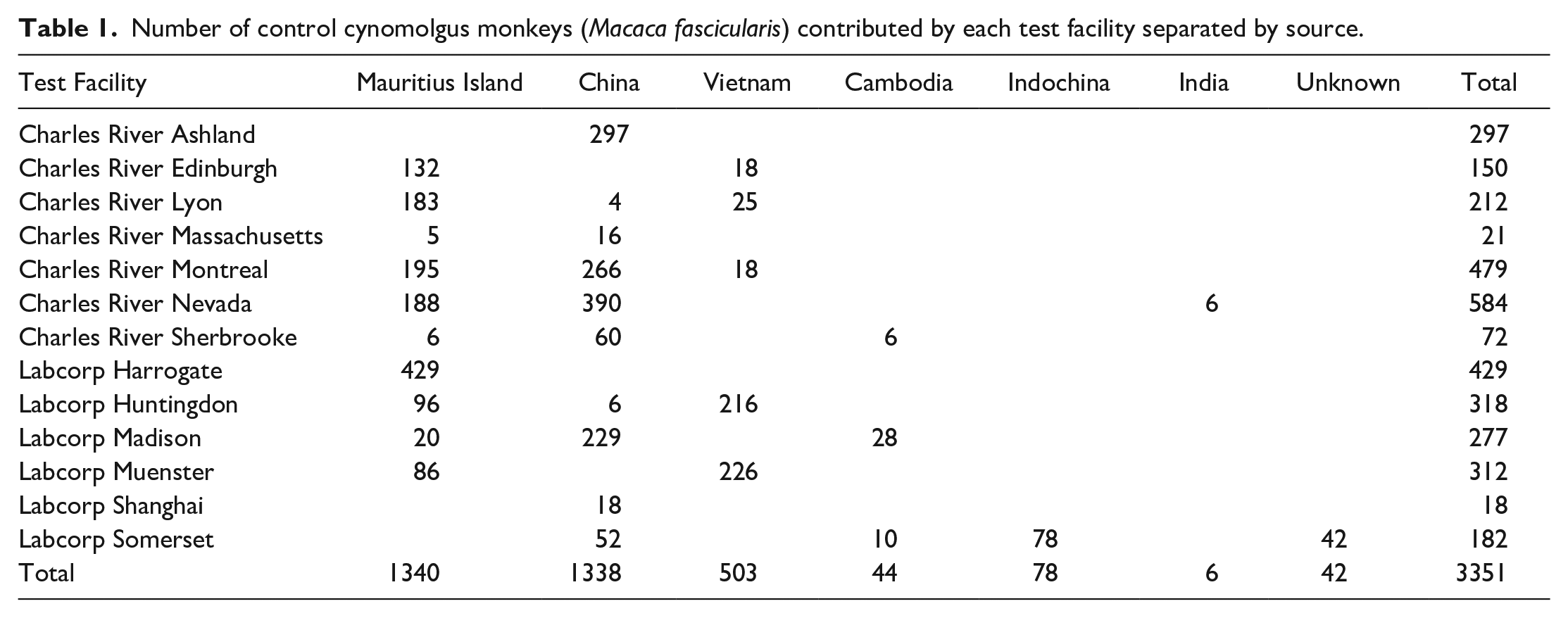
Number of control cynomolgus monkeys (Macaca fascicularis) dosed by different routes.
The age range of animals at the commencement of their respective toxicity studies was 2 to 9.5 years. To determine the potential effects of age on the incidence and occurrence of spontaneous histopathology findings, animals included in this review were allocated into two broad age-groups, based on their age at the commencement of the individual studies: (1) sexually mature animals (mean age 4 years and over) and (2) young adolescent macaques (mean age of 4 years or less), hereafter referred as “mature” and “juvenile” animals, respectively. Studies with an age range that was judged to overlap excessively between these categories were excluded from the age comparisons.
Prior to each toxicity study, animals received an appropriate veterinary examination and screened for a range of infectious agents according to the local standard operating procedures. Animals were group-housed in socially compatible groups of two or three animals of the same sex and dose group in appropriate custom-designed cages. Studies performed in North American facilities were conducted at American Association for the Accreditation of Laboratory Animal Care (AAALAC International)-accredited contract research organizations (CROs), and in compliance with all federal regulations and recommendations detailed in The Guide for the Care and Use of Laboratory Animals and in accordance with Charles River or Labcorp Drug Development policies on the care, welfare, and treatment of laboratory animals. In addition, all procedures involving the animals were reviewed and approved by the respective CRO’s Institutional Animal Care and Use Committees (IACUC) prior to dose administration. Studies performed in the United Kingdom and European Union were also conducted at AAALAC International-accredited CROs, in accordance with the UK Animals (Scientific Procedures) Act 1986, and the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes and European Directive 2010/63/EU on the protection of animals used for scientific purposes (Strasbourg, Council of Europe), respectively, in addition to institutional ethical committees. The temperature and humidity were automatically controlled (a minimum of eight air changes/hour and temperature of 19°C to 26°C, and 30% to 70% relative humidity), and an automatic 12-hour light cycle was maintained. All animals were offered certified commercial primate diets (that varied by site, country, or region) and food supplements (fruits and vegetables) and various cage enrichment devices, while water was provided ad libitum.
Pathology
Animals were humanely euthanized according to institutional standard operating procedures, detailed necropsies performed, and tissues collected and preserved in 10% neutral buffered formalin. The scope of tissues collected was generally similar across sites. Histology processing involved embedding of tissues in paraffin wax, sectioning to a 3 to 5 µm thickness, and staining with hematoxylin and eosin (H&E). In rare cases, immunohistochemistry (IHC) was applied at the study pathologist’s discretion. Details of the IHC methods are not included. Tissues were examined microscopically, and the findings entered directly into a computerized data recording application that varied according to the facility. Histopathology findings generated from studies conducted within the past 5 years (2016-2021) were collected retrospectively from control groups of cynomolgus macaques. From this pool of data, studies to be incorporated into the present retrospective investigation were selected based on the criteria of having at least one control or untreated group and being Good Laboratory Practice (GLP)-compliant. Histopathology findings were collected either from dedicated HCD databases or compiled manually and converted into a standard format on a single database in Microsoft Excel for Microsoft 365. Similar findings were merged into single terms using International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) terminology whenever possible. There were several reasons for doing this, and examples included the use of synonymous terms, for example, “Glycogen, increased” and “Glycogen, vacuolation” merged to “Accumulation, glycogen,” and the inconsistent use of modifiers to indicate sublocations or distribution, for example, multifocal, submucosal, and periportal. Selected pairs of incidences were analyzed using Fisher’s exact test (two-tailed). Tables of findings for each tissue/sex comparing the incidences by origin and age are available as supplementary material. In addition to tabulated incidence, micrographs of selected lesions are provided for illustrative purposes. These included IHC to characterize representative examples of specific lesions in control animals.
Results
Most Common Spontaneous Histopathology Findings Irrespective of Geographical Origin
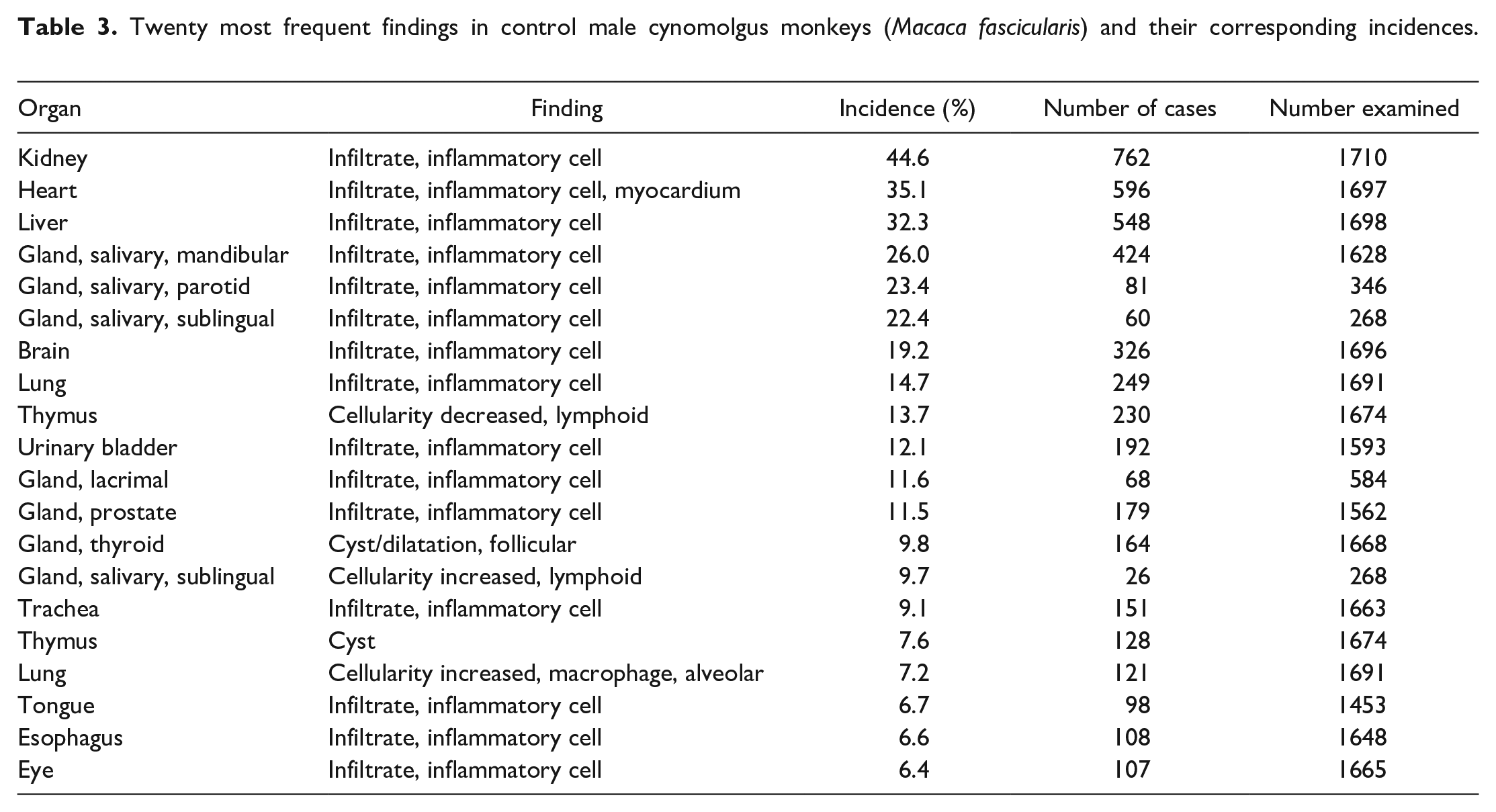
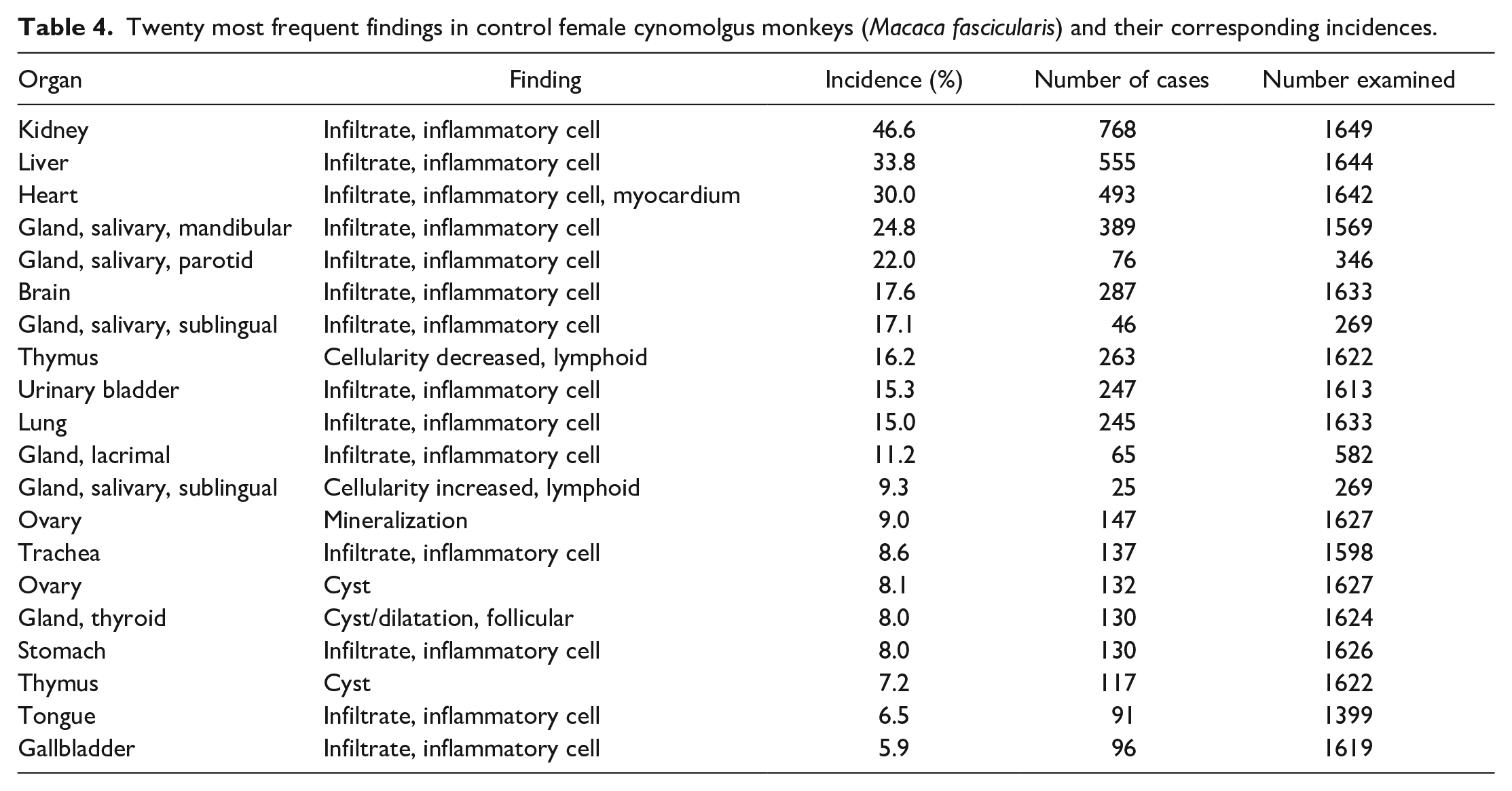
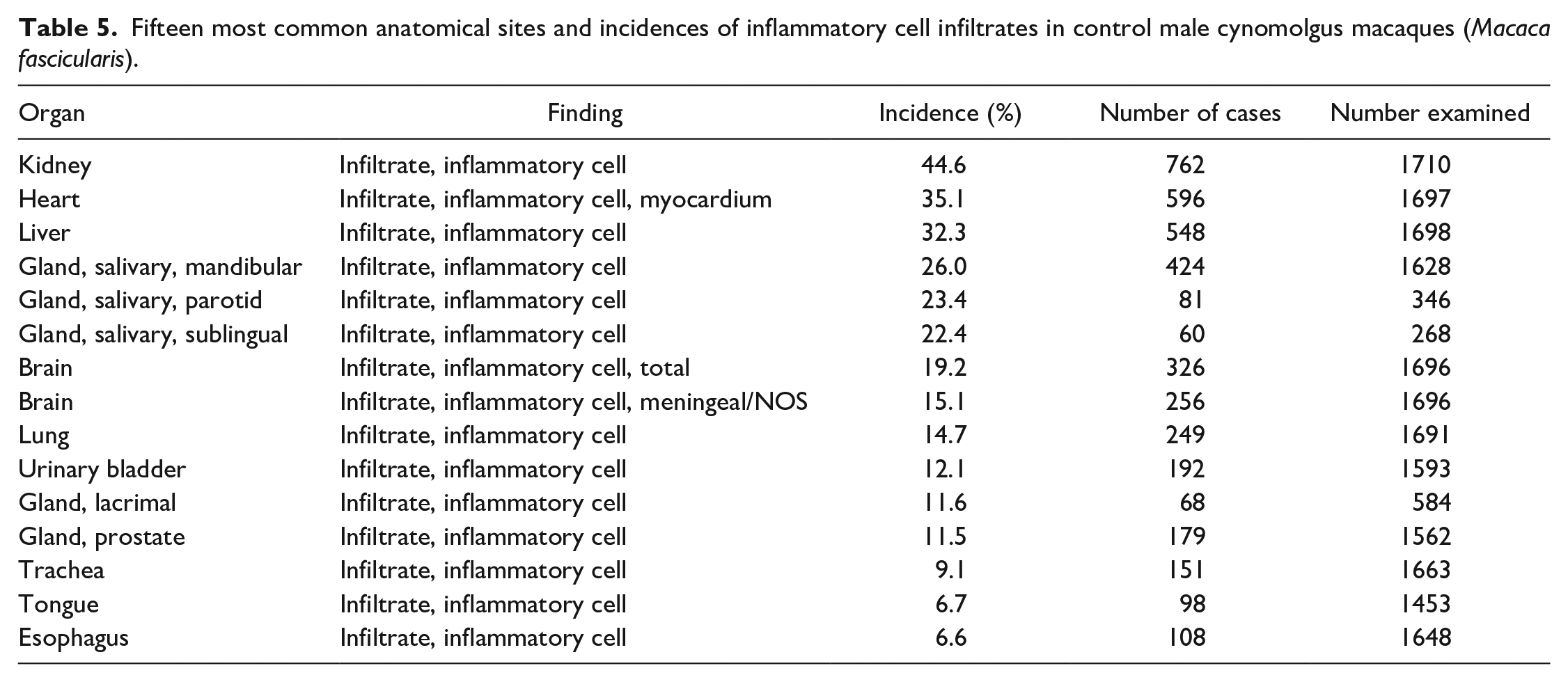
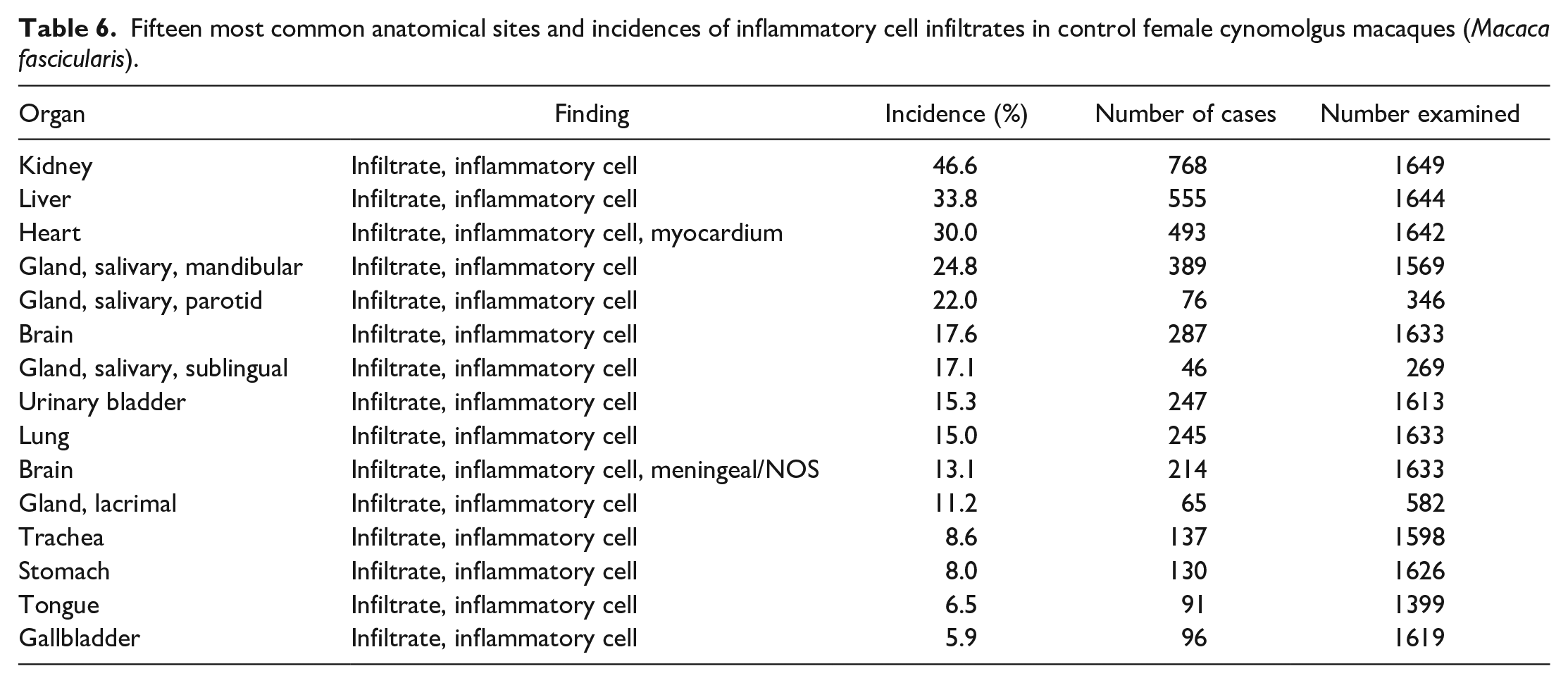
A summary of the incidences of the most common spontaneous histopathology findings in animals from all geographical regions and age, grouped together, for each sex (Tables 3 and 4) shows that the most commonly observed histopathology findings were mononuclear or lymphoplasmacytic inflammatory cell infiltrates in various organs (Figures 1 and 2), with a preponderance for a perivascular distribution in some tissues. The tissues most commonly affected with inflammatory infiltrates were the kidney (44.6% in males and 46.6% in females), heart (35.1% and 30.0%), liver (32.3% and 33.8%), salivary glands (for mandibular 26.0% and 24.8%), brain (19.2% and 17.6%), lung (14.7% and 15.0%), urinary bladder (12.1% and 15.3%), lacrimal gland (11.6% and 11.2%), prostate (11.5%), trachea (9.1% and 8.6%), and stomach (6.0% and 8.0%) (Tables 5 and 6). The most commonly used term or qualifier to describe the inflammatory infiltrates was by far “mononuclear” although other qualifiers, such as “lymphoplasmacytic” or “lymphocytic,” were occasionally used, particularly in those organs with a propensity to developing lymphoid aggregates or follicles, such as the salivary glands, lacrimal glands, or the uvea of the eye. The nature and morphological presentation of mononuclear inflammatory cell infiltrates was as previously described15,22,24,25 and consisted of interstitial accumulations of mainly lymphocytes and plasma cells in varying proportions, with occasional histiocytes, and associated with negligible or no evidence of injury to parenchymal tissues. The latter characteristic feature (absence of substantial tissue injury) was the key differentiating feature between inflammatory infiltrates and inflammation. Inflammation was recorded when there was evidence of substantial degeneration and necrosis of parenchymal tissue, such as renal tubules, alveolar pneumocytes, gastric glands, cardiac myocytes, or hepatocytes, in addition to inflammatory cell infiltrates, usually of a mixed nature, or when other obvious signs of inflammation, such as edema and fibrin depositions, were present in association with inflammatory infiltrates, within the interstitium.
Twenty most frequent findings in control male cynomolgus monkeys (Macaca fascicularis) and their corresponding incidences.
Twenty most frequent findings in control female cynomolgus monkeys (Macaca fascicularis) and their corresponding incidences.
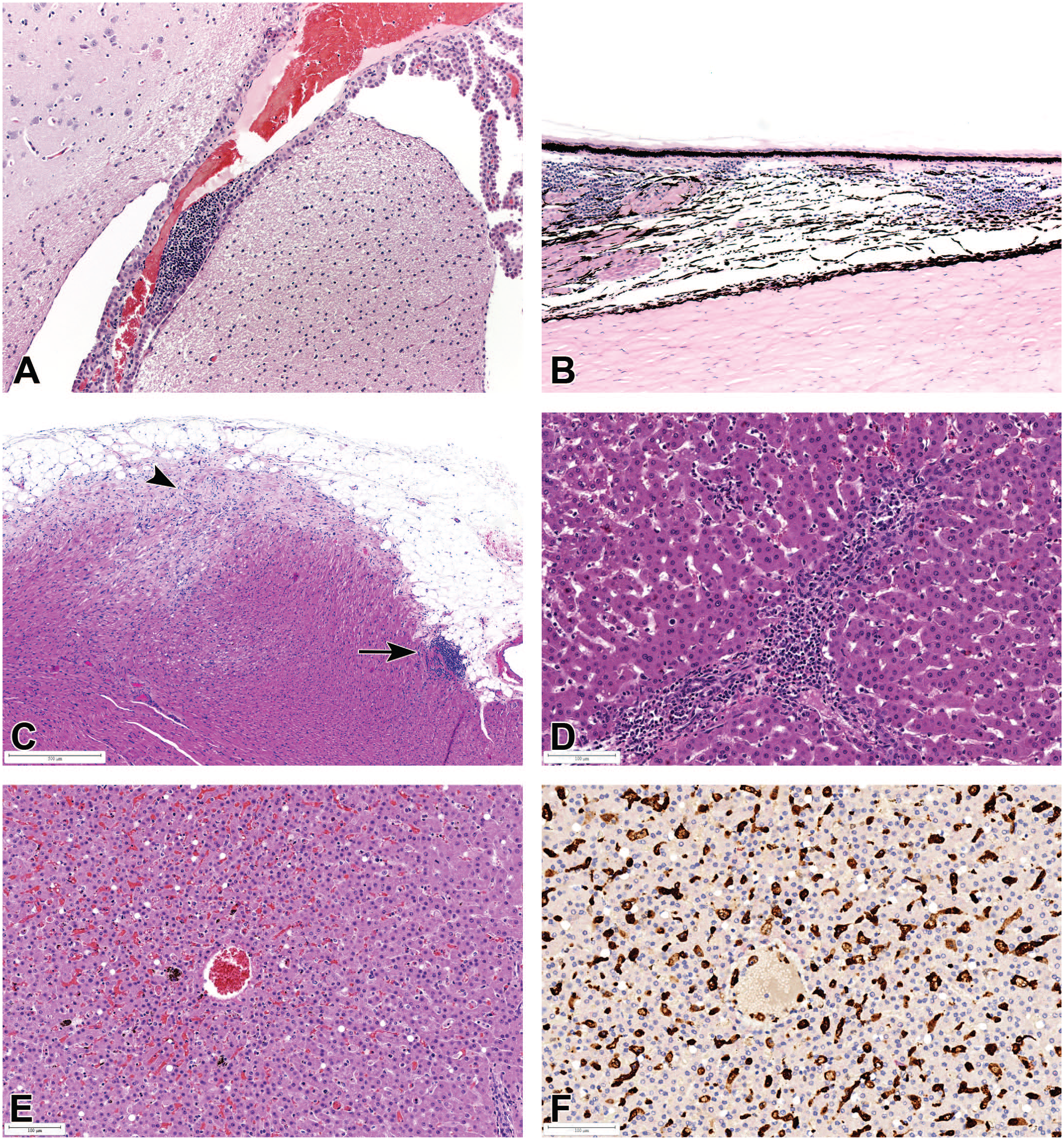
Mononuclear inflammatory cell infiltrates in (A) the choroid plexus of the brain; (B) brain, ciliary body, and choroid of the eye; (C) the apex of the heart (arrow), with myofiber vacuolation and degeneration (arrowhead), and karyomegaly; and (D) sinusoidal and periportal areas of the liver. All H&E. (E) Liver showing sinusoidal leukocytosis, associated with Kupffer cell hyperplasia, sinusoidal dilatation, congestion, hemosiderin pigment, and minimal hepatocyte vacuolation, (H&E); and (F) positive Iba1 immunostaining (brown) demonstrating an increase in sinusoidal-lining Kupffer cells.
Mononuclear inflammatory cell infiltrates in (A) the lung, showing mostly a mixture of alveolar macrophages and perivascular lymphocytic infiltrates, (B) urinary bladder lamina propria, showing a perivascular distribution, (C) kidney interstitia and (D) submucosa of the gall bladder. All H&E.
Fifteen most common anatomical sites and incidences of inflammatory cell infiltrates in control male cynomolgus macaques (Macaca fascicularis).
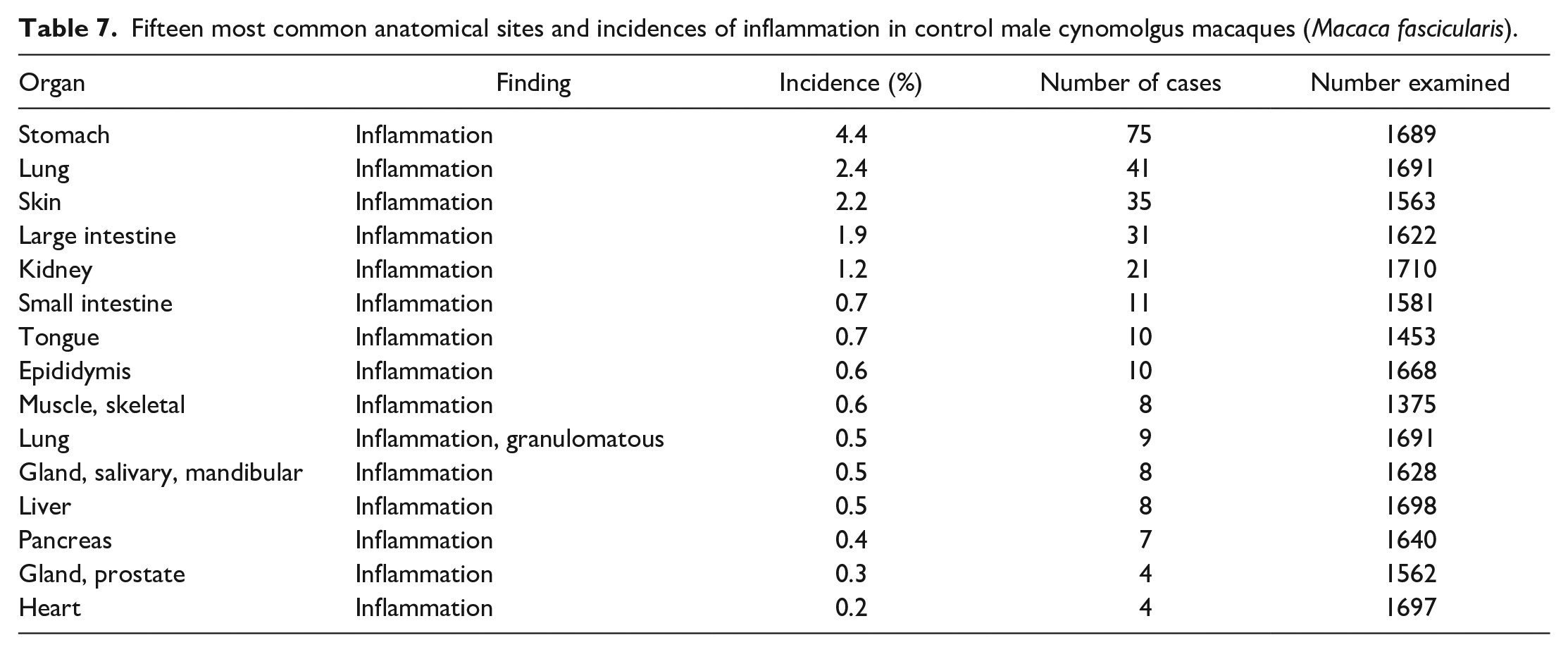
Fifteen most common anatomical sites and incidences of inflammatory cell infiltrates in control female cynomolgus macaques (Macaca fascicularis).
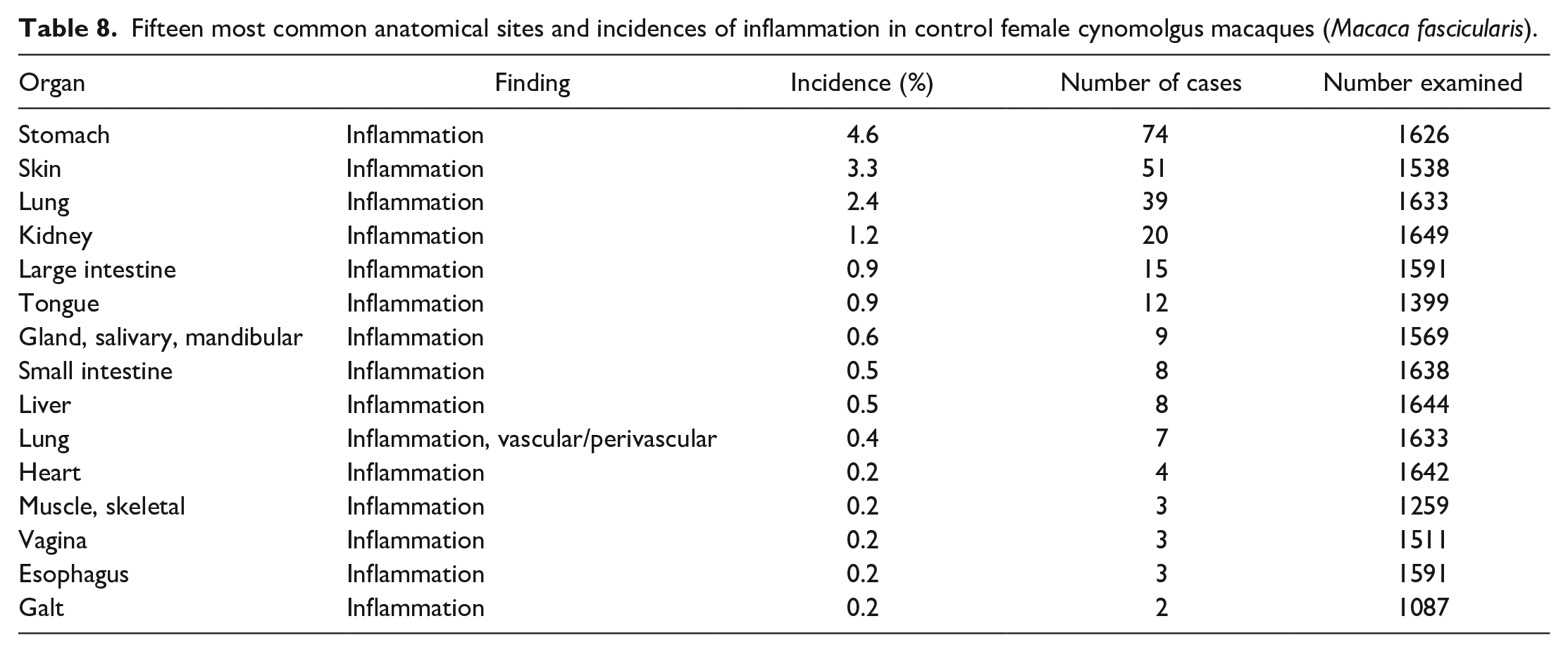
Findings other than inflammatory infiltrates occurred at a much lower incidence and included degenerative and congenital lesions, such as cysts (ovary, thyroid gland, pituitary gland, thymus), ectopic tissue (thymus in the thyroid gland, squamous cysts in the heart), atrophy/lymphoid depletion in the thymus, mineralization (brain, adrenal gland, ovary), pigment deposits (lung), or vacuolation (liver). Decrease lymphoid cellularity or atrophy in the thymus (13.7% and 16.2%) was the most commonly recorded of these second-tier group of findings, followed by mineralization in the ovary (9.0%) and cysts in the ovary (8.1%) and thymus (7.6% and 7.2%). Inflammation and other findings associated with chronic inflammation, such as fibrosis or adhesions, were relatively common in certain tissues, such as the stomach (4.4% and 4.6%), skin (2.2% and 3.3%), lung (2.4%), large intestine (1.9% and 0.9%), and kidneys (both 1.2%) (Tables 7 and 8). In addition to inflammation, relatively high incidences of epidermal ulcer/erosion and pleural fibrosis were recorded in the skin and lung, respectively.
Fifteen most common anatomical sites and incidences of inflammation in control male cynomolgus macaques (Macaca fascicularis).
Fifteen most common anatomical sites and incidences of inflammation in control female cynomolgus macaques (Macaca fascicularis).
Comparison of Incidences of Common Spontaneous Histopathology Findings Based on Geographical Origin
A comparison of the incidences of common background findings in cynomolgus macaques from China, Mauritius, and Vietnam revealed statistically significant and/or numerical differences in the following findings: inflammatory infiltrates in the brain, eye and lacrimal glands, liver and gallbladder, lung, salivary glands, stomach, large intestine, urinary bladder, and vagina; inflammation in the skin, stomach, and large intestine; lymphoid hyperplasia and hyalinosis in the spleen; increased adipocytes and increased cellularity in the bone marrow; lung pigment; and developmental or degenerative cysts in the ovary, thymus, thyroid gland, and heart.
Mononuclear inflammatory infiltrates and inflammation
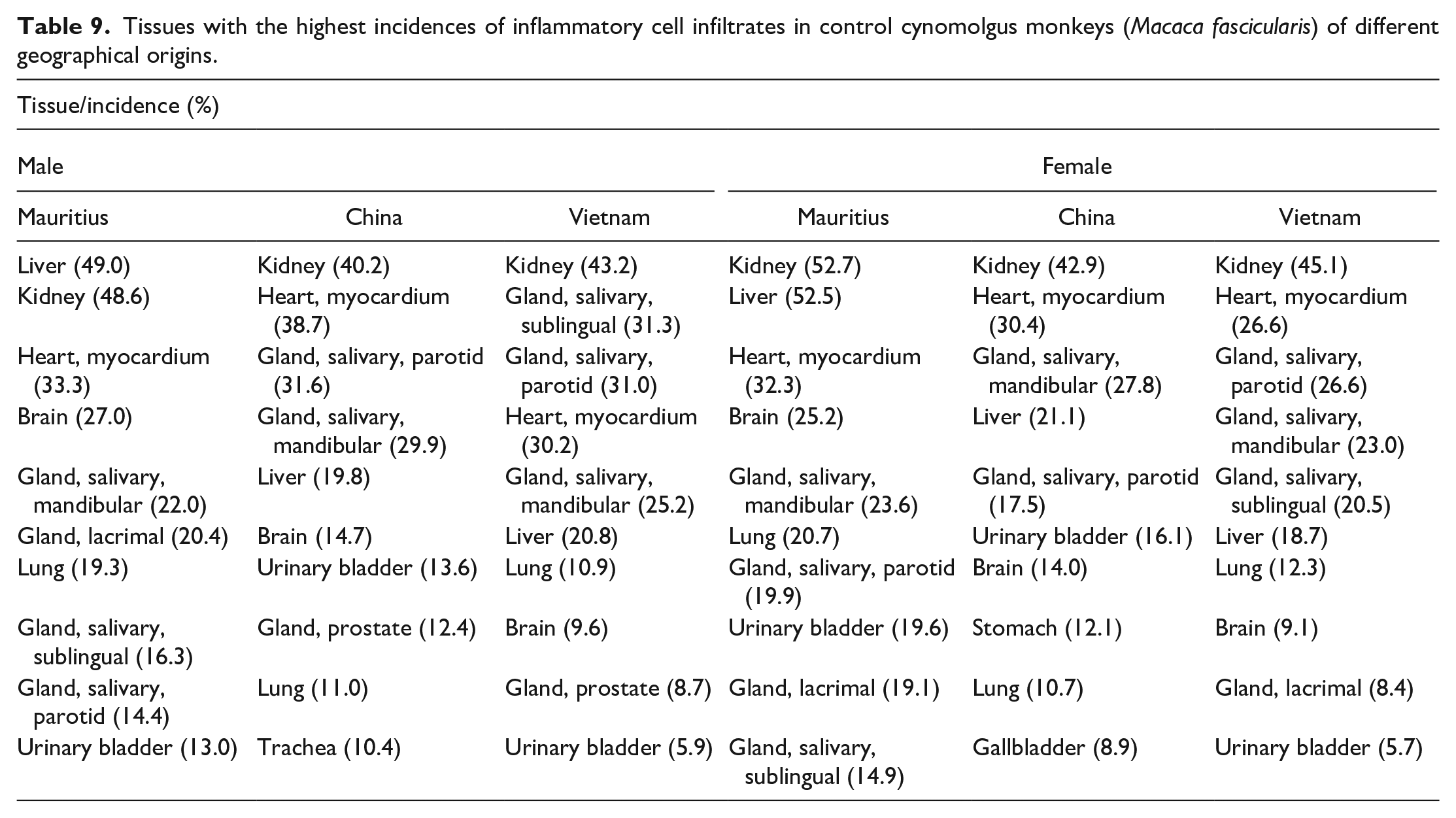
When animals were grouped according to the geographical origin, the list of most frequently affected tissues were generally similar to those observed in the combined analysis of animals from all geographical origins, and inflammatory cell infiltrate was still the most commonly observed finding. However, the incidences of inflammatory infiltrates in each of the most commonly affected tissues and the order in which the topmost affected organs appeared varied according to the sex, geographical origin (Table 9), or age (Table 10) of the animals. For instance, the kidney had the highest or second highest incidence of inflammatory infiltrates in all groups, whereas liver infiltrates in Mauritian males occurred at an incidence of more than twice that recorded in Chinese and Vietnamese males, with the incidences in Chinese and Vietnamese macaques being comparable (49.0% compared with 19.8% and 20%, respectively—both P < .001). Similarly, the incidence of liver infiltrates in female Mauritian macaques (52.5%) also occurred at more than twice those in Chinese (21.1%) and Vietnamese (18.7%) females (both P < .001). However, the reverse was true for gallbladder infiltrates, with higher incidences being recorded in Chinese males and females (8.0% and 8.9%), when compared to male and female Mauritian (4.3% and 4.6%) macaques (P = .006 and P = .003, respectively). Liver infiltrates occurred predominantly in the periportal regions or focally within the sinusoids (Figure 1D), with or without associated single-cell necrosis. Diffuse sinusoidal infiltrates (sinusoidal leukocytosis) and Kupffer cell hyperplasia (Figure 1E and F), with or without sinusoidal dilatation or erythrocyte sequestration, were a common nonspecific observation in animals with clinical disease, such as intestinal lesions associated with diarrhea.
Tissues with the highest incidences of inflammatory cell infiltrates in control cynomolgus monkeys (Macaca fascicularis) of different geographical origins.
Tissues with highest incidences of inflammatory cell infiltrates within each age category of control cynomolgus macaques (Macaca fascicularis).
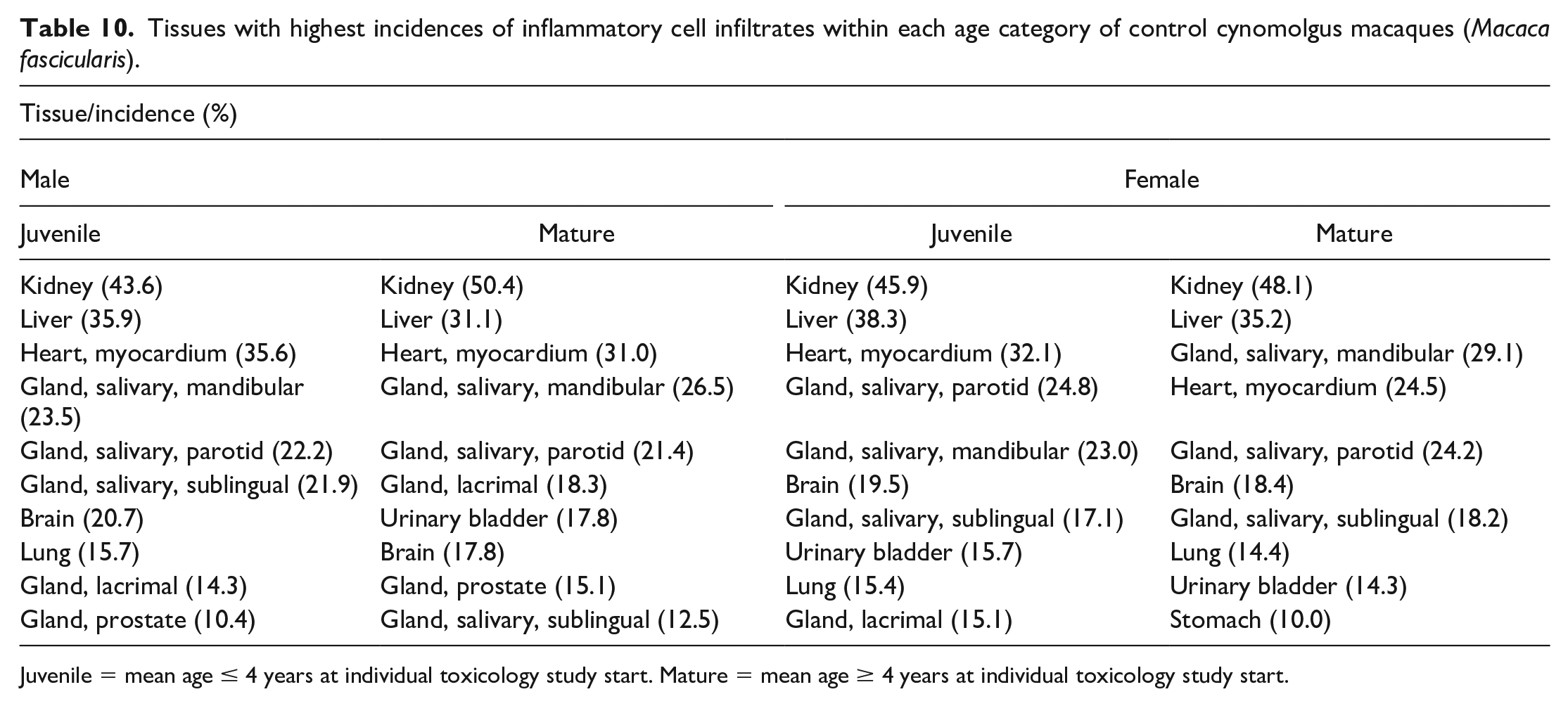
Juvenile = mean age ≤ 4 years at individual toxicology study start. Mature = mean age ≥ 4 years at individual toxicology study start.
Another striking difference in the incidence of mononuclear inflammatory infiltrates was observed in the brain, where the incidences in male and female Mauritian macaque (27.0% and 25.2%) were nearly twice that in Chinese macaques (14.7% and 14.0%; both Ps < .001) and more than twice that in Vietnamese macaques (9.6% and 9.1%). To rule out the influence of study procedures and dose route-related changes on the brain findings, studies with the intrathecal and intracerebral dosing, and continuous intravenous infusion were excluded in the analysis. Lung infiltrates in Mauritian male and female macaques (19.3% and 20.7%; both Ps < .001) were also nearly twice those in Chinese (11.0% and 10.7%) and Vietnamese (10.9% and 12.3%) macaques. Similarly, eye (predominantly uveal) and lacrimal gland infiltrates were more frequently observed in Mauritian when compared to Chinese or Vietnamese macaques. The incidence of inflammatory infiltrates in male and female Mauritian macaques in the eye (9.0% and 9.6%) and lacrimal glands (20.4% and 19.1%) was approximately twice those observed in the eye (5.6% and 3.5%; P = .02 and P < .001) and lacrimal glands (9.2% and 7.5%; both Ps < .001) from Chinese macaques. In Vietnamese macaques, the incidences of this finding in the eye and lacrimal glands were either comparable to or slightly lower than those observed in Chinese macaques. Skin inflammatory findings were also observed at higher incidences in Mauritian macaques when compared to Chinese or Vietnamese macaques. Skin and subcutaneous inflammation in male and female Mauritian macaques (5.0% and 7.1%) were several magnitudes higher than those in Chinese macaques (0.5% and 1.3%; both Ps < .001), while higher incidences of ulcer/erosions were observed in male Mauritian macaque (3.2%) when compared to Chinese (1.1%, P = .010) and Vietnamese (0.4%, P = .021) macaques. Edema of the skin was observed at higher incidences in female Mauritian macaques (7.6%) when compared to those from China (0.5%) or Vietnam (0.0%), both Ps < .001.
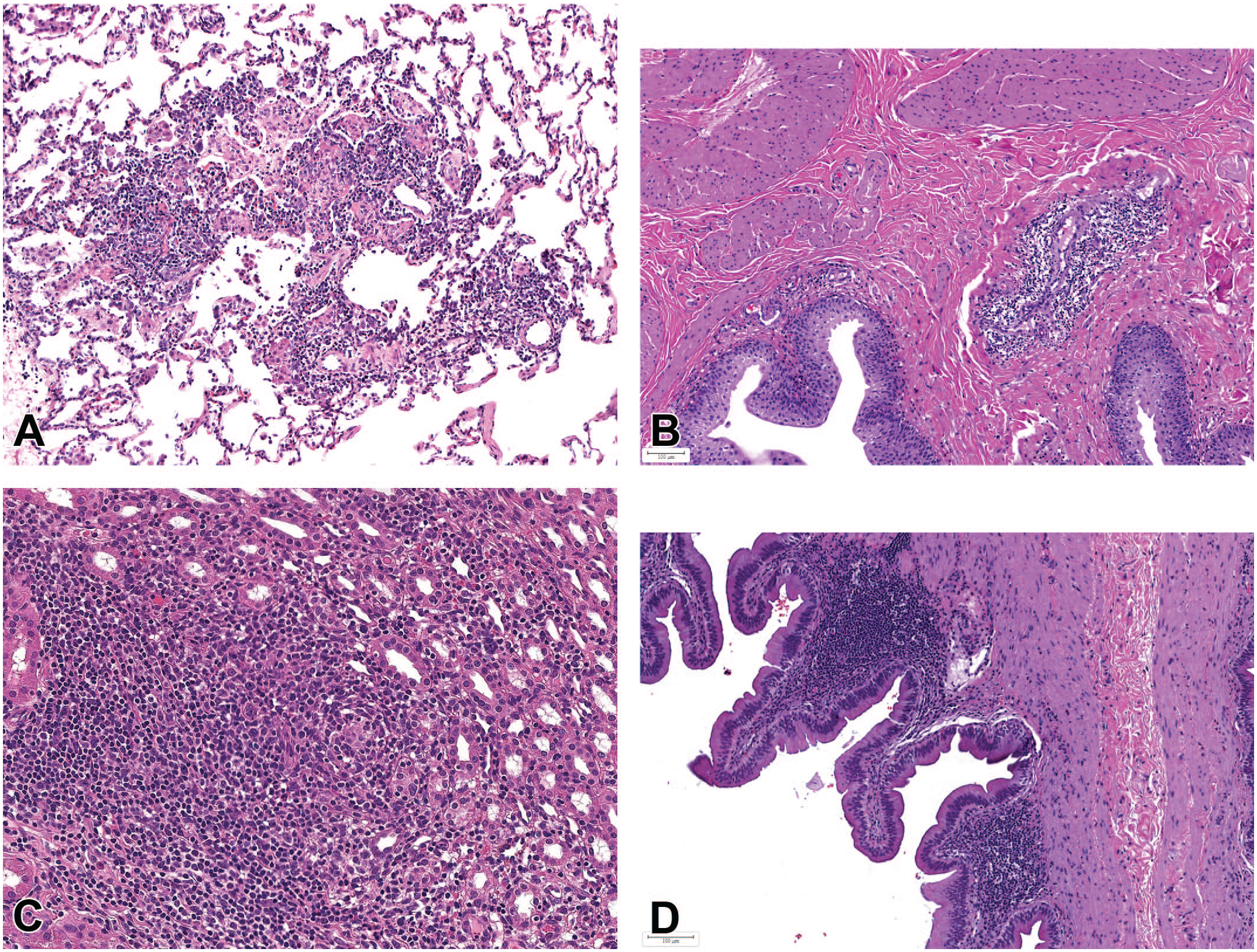
Mononuclear inflammatory infiltrates and inflammation in the GIT also revealed some notable differences between animals from different geographical regions, and in these tissues, macaques from Asia tended to have higher incidences of either one or both individual inflammatory conditions (mononuclear infiltrates and inflammation) than those from Mauritius (Table 11). Mononuclear infiltrates in the stomach were observed at higher incidences in male and female Chinese macaques (9.0% and 12.1%), when compared to males and females from both Mauritius (2.8% and 5.3%; both Ps < .001) and Vietnam (4.6% and 3.7%; P = .028 and P < .001). As with inflammatory infiltrates, the incidence of inflammation in the stomach was higher in male and female Chinese macaques (4.2% and 4.7%) when compared to those from Mauritius (2.9% and 2.3%; P = .187 and .022), although the incidence of gastric inflammation in Vietnamese male and female macaques (9.3% and 9.4%) occurred at higher incidences than in both Chinese and Mauritian macaques. However, when incidences of both inflammatory infiltrates and inflammation which constitute a pathology continuum were combined, the incidences in Chinese and Vietnamese macaques were similar but remained higher than those in Mauritian macaques. In the representative tissues examined in this survey, gastric inflammation was mostly subchronic to chronic in nature, affecting both the pylorus and the fundic/body region, and was characterized by moderate to marked lymphoplasmacytic cell infiltration in the mucosa and submucosa, dilatation or atrophy of mucosal glands with attenuation of glandular epithelium, neutrophilic infiltrates within gastric glands (glandular microabscesses), separation and scarcity of glands accompanied with an increase in the lamina propria and occasional mucosal erosion (Figure 3). Spiral bacteria or Helicobacter pylori were rarely observed in these lesions. In some chronic stomach lesions, frequently observed in decedent animals that had protracted idiopathic chronic diarrhea (ICD), in addition to the lesions described above, chronic gastritis was associated with infiltration by pale or foamy histiocytes in the superficial layers of the mucosa, with frequent mucosal erosions (Figure 3C and D).
Number and incidences of inflammation/inflammatory cell infiltrates in the GIT tissues in control cynomolgus monkeys (Macaca fascicularis) compared between sources.
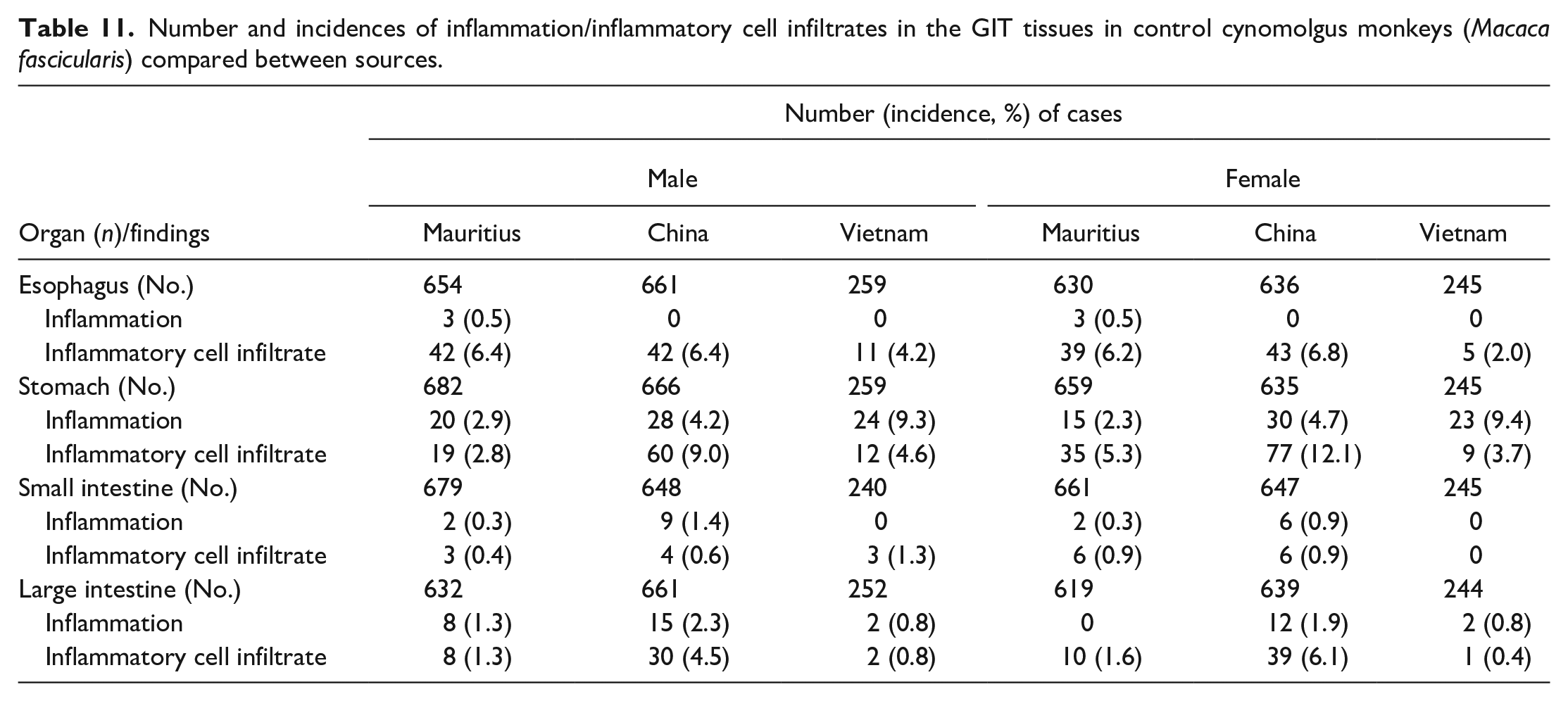
Stomach. Mononuclear inflammatory cell infiltrates and inflammation: (A) lymphoplasmacytic infiltrates showing plasma cells with Russell bodies (arrows) in the cardia, (B) inflammation in the fundus with glandular separation, and (C and insert) chronic gastritis with erosions, glandular microabscesses (arrowheads), and infiltration by vacuolated histiocytes (arrows) in the subepithelial mucosa. (D) Chronic inflammation with erosions and vacuolated histiocytes (arrows) in the pylorus. All H&E.
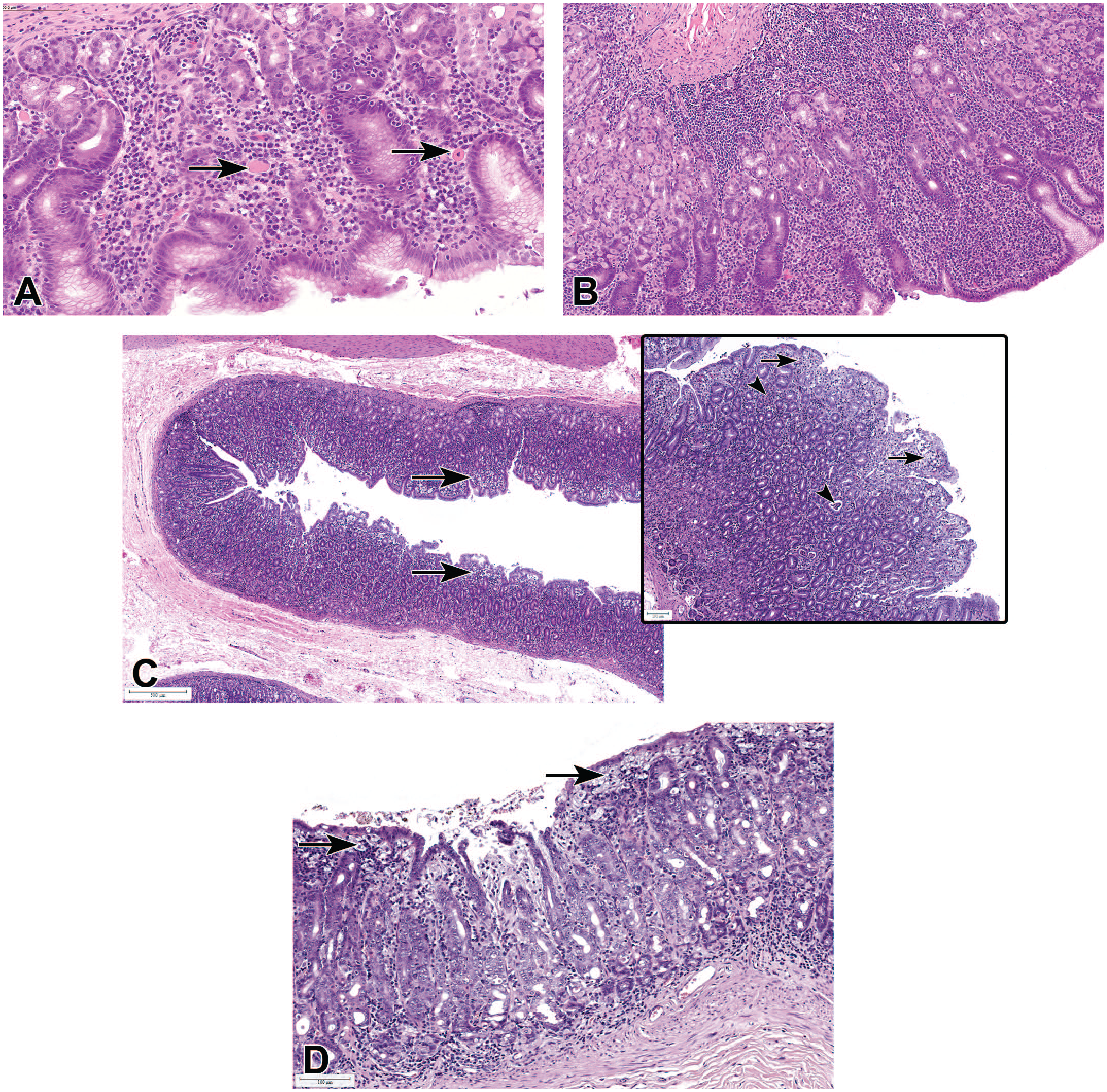
A comparison of the incidences of inflammation and inflammatory infiltrates in the large intestine between Chinese and Mauritian macaques generally followed a similar pattern to that observed in the stomach. In Chinese males and females, higher incidences of inflammatory infiltrates (4.5% and 6.1%) and inflammation (2.3% and 1.9%) were recorded when compared to the same findings in Mauritian macaques (1.3% and 1.6% for infiltrates, and 1.3% and 0.0% for inflammation). Although most asymptomatic infiltrates in the intestines were predominantly comprised plasma cells, cases associated with chronic diarrhea and hypoproteinemia often showed an increase in CD3 + T-cells in the lamina propria (Figure 4).
Intestinal mononuclear cell infiltrates: (A) lymphoplasmacytic infiltrates with villous atrophy and blunting in the duodenum, associated with clinical malabsorption and hypoproteinemia. Duodenum is identified by the presence of Brunner’s gland (B). H&E. (B and C) IHC staining of the small intestine showing that most lymphocytic infiltrates are CD3-positive for T-cells (brown), but CD19 negative for B cells (D).
Some minor differences in incidences of inflammatory infiltrates were observed only in one sex, and these included higher incidences in the heart in males from China (37.8%) compared to those from Mauritius (33.3%; P = .042) and Vietnam (30.2%; P = .018), and higher incidences in the mandibular (29.9%) and parotid (31.6%) salivary glands from Chinese males, compared to those from Mauritius (22.0% mandibular and 14.4% parotids; P = .001 and P = 0.009), while those from Vietnam (25.2% mandibular and 31.0% parotids) were comparable to those from China. In females, much higher incidences of inflammatory infiltrates in the pancreas were observed in Mauritian macaques (6.1%) when compared to Chinese (1.7%; P < .001) and Vietnamese macaques (0.8%; P < .001).
Hemolymphoreticular system
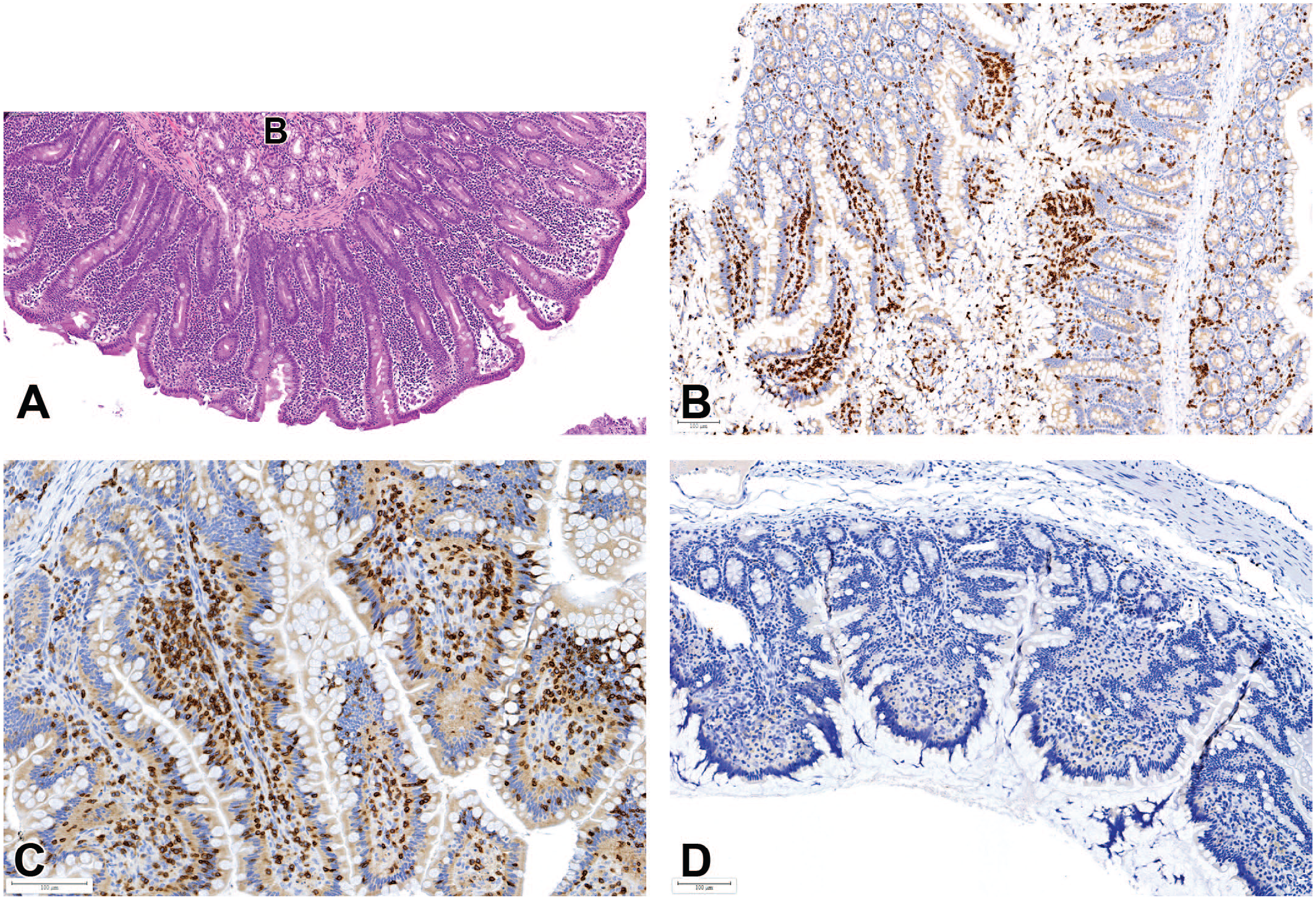
In the spleen, a higher incidence of lymphoid hyperplasia or increased lymphoid cellularity (Figure 5) was observed in male and female animals from China (6.3% and 5.5%) compared to those from Mauritius (2.5% and 3.6%; P < .001 and P = .142) and Vietnam (3.9% and 4.1%; P = .202 and P = .495). In addition, hyalinosis of the splenic germinal centers was also present at higher incidences in Chinese male and female macaques (1.7% and 1.6%) when compared to Mauritian (0.3% and 0.0%; P = .012 and P < .001) and Vietnamese (0.0% and 0.4%; P = .040 and P = .306). Germinal center hyalinization was frequently recorded in association with the abnormally large, nodular, and often misshapen and coalescing follicles, which were generally considered by to represent lymphoid hyperplasia (Figure 5). The hyperplastic lymphoid foci were sometimes observed at necropsy as raised pale foci causing compression of adjacent tissue or bulging of the splenic capsule microscopically. Prominent lymphoid follicles in other hematopoietic and/or lymphoid organs including bone marrow and lymph nodes sometimes occurred concomitantly. In the bone marrow, increased cellularity was recorded at higher incidences in female Mauritian macaques (2.6%) compared to Chinese females (0.6%; P = .003). Higher incidences of vacuolated macrophages in the lymph nodes and spleen were recorded in macaques from Mauritius when compared to those from China and Vietnam.
Increased lymphoid cellularity and abnormal germinal centers in the spleen: (A) enlarged (B) and coalescing (A) germinal centers with hyalinosis (arrows) and compression of adjacent tissues. All H&E. (C) Positive (brown) CD19 immunostaining for B cells showing expansion of lymphoid follicles and germinal centers. (D) Positive CD3 immunostaining for T-cells (brown) showing compression of T-cell areas.
Cardiovascular System and Lungs
In addition to the differences in the incidence of myocardial inflammatory cell infiltrates between Mauritian and Chinese males, there were a few other minor differences in the incidences of cardiac lesions. Overall, there were slightly more findings recorded in the heart in cynomolgus macaques from Mauritius than were recorded in macaques from China or Vietnam, and some findings, such as squamous cyst or plaques, ectopic thyroid, pigment, and mineralization, were reported only in Mauritian macaques. In addition, minor increases in the incidences of karyomegaly in the heart were observed in Mauritian macaques when compared to those from China. In contrast, though, myocardial degeneration/necrosis was recorded at higher incidences in Chinese when compared to Mauritian and Vietnamese macaques, with Vietnamese macaques having the lowest incidences of this finding. Other findings of toxicological significance in the heart, such as granulomatous inflammation in the pericardium or pericardial fat (Figure 6A and B), myocardial fibrosis, cardiomyocyte vacuolation (Figure 1C), and vascular/perivascular inflammation, occurred at relatively low but comparable incidences among animals from all geographical origins. Pericardial microgranuloma or granulomatous inflammation (Figure 6), comprised epithelioid or multinucleated macrophages surrounded by other inflammatory cells, were most commonly observed in the adipose tissue at the base of the heart and most commonly occurred in animals with an underlying disease associated with weight loss or fat necrosis, such as chronic diarrhea.
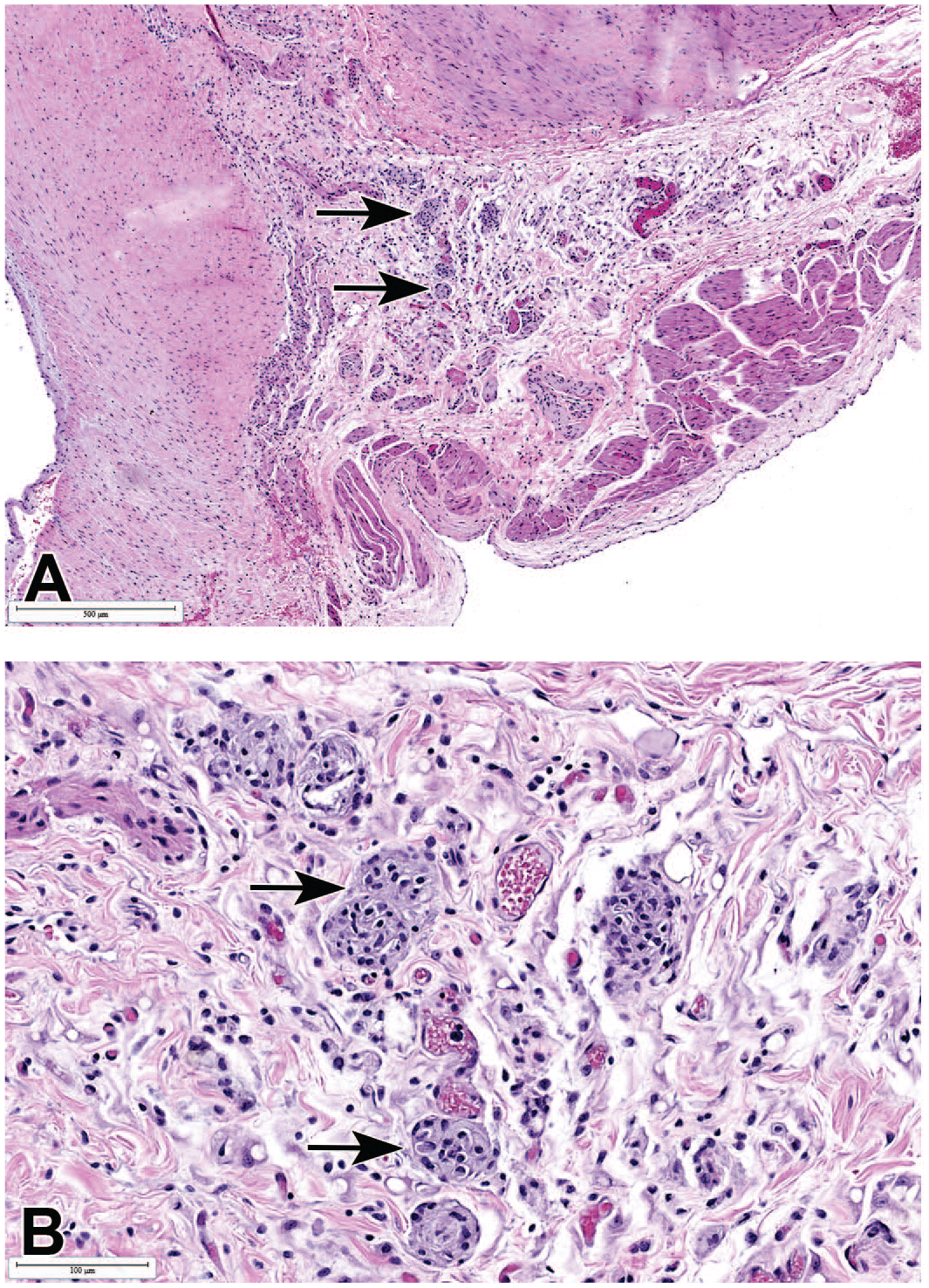
Granulomatous inflammation or microgranuloma (arrows) in the pericardial adipose tissue at the base of the heart. (A) Low magnification. (B) Higher magnification. All H&E.
As in the heart, there were only a few minor differences in the incidences of lung findings other than the inflammatory findings already discussed above. Considerably higher incidences of exogenous pigment (anthracosis) were observed in Vietnamese (12.4% and 9.8%) and Chinese (3.6% and 4.8%) male and female macaques, when compared to Mauritian macaques (0.4% and 0.6%), (all Ps < .001 except female Chinese versus Vietnamese for which P = .008), with Vietnamese macaques having much higher incidences than the other two.
Degenerative and Developmental Lesions
Cysts were more commonly recorded in Mauritian macaques when compared to Chinese and Vietnamese macaques. Higher incidences of cysts or cystic dilation were recorded in the thymus in male (12.8%) and female (12.0%) Mauritian macaques, when compared to male and female Chinese (3.8% and 4.4%), or Vietnamese macaque (5.1% and 2.1%; P < .001 for all). Similarly, there were higher incidences of cysts in the thyroid (15.8% and 11.2%) and parathyroid (3.7% and 3.3%) glands of male and female Mauritian macaques, when compared to Chinese (5.7% and 5.3% for thyroid and 0.9% and 1.1% for parathyroid), or Vietnamese (4.8% and 7.4% for thyroid and 1.9% and 2.3% for parathyroid) male and female macaques. In the thyroid gland, incidences of ectopic thymus were also higher in Mauritian macaques relative to Chinese or Vietnamese macaques (except in males which were comparable to Mauritian). Higher incidences of cysts in Mauritian macaques relative to the Chinese macaques were also observed in the ovary, oviduct, and uterus. Other origin-related differences were observed in the incidences of tension lipidosis in the liver, axonal degeneration in the spinal cord, increased fibrous connective tissue (likely developmental) and tubular hypoplasia in the testes, and degree of testes or prostate maturity at a given age. The incidences of mineralization and perivascular pigment in the brain and tension lipidosis in the liver were also higher in Mauritian when compared to Chinese and Vietnamese macaques. In contrast, increased incidences of axonal degeneration in the spinal cord, increased fibrous connective tissue and tubular hypoplasia in the testes, and decreased sperm in the epididymis were higher in the Chinese and Vietnamese macaques, when compared to those from Mauritius.
Sex-Related Differences in Incidences of Spontaneous Histopathology Findings
Some minor sex-related differences were observed in the urinary bladder, stomach, adrenal gland, bone marrow, skin, bone, brain, spleen, kidney, heart, liver, and lung. Findings that showed an increase in incidence in females relative to males included inflammation and inflammatory infiltrates in the urinary bladder, glandular dilatation and inflammatory infiltrates in the stomach, increased hematopoiesis and myelopoiesis in the bone marrow, edema and subcutaneous inflammation in the skin, mineralization in the brain, extramedullary hematopoiesis in the lymph nodes, hepatocyte vacuolation and inflammatory infiltrates in the liver, inflammatory infiltrates, basophilic tubules and hyaline casts in the kidney, and pituitary gland vacuolation. Findings that showed an increase in males relative to females included myocardial inflammation/degeneration, myocardial infiltrates, cardiac myocyte karyomegaly and cyst/plaques in the heart, pigment in the brain, infiltrates in the eye, capsular fibrosis in the spleen, glycogen accumulation, hepatocyte degeneration/necrosis and fibrosis in the liver, and macrophage accumulation, granuloma, and alveolar type 2 pneumocyte hyperplasia in the lung.
Age Group-Related Differences in Incidences of Spontaneous Histopathology Findings
To determine the potential influence of age on the incidence and occurrence of spontaneous histopathology findings, study animals were split into the following two age-groups, based on their age at the study start in the individual toxicology study: (1) mature group, containing animals of mean age 4 years and above, and (2) juvenile group, with animals of mean age of 4 years or below, further referred to as “mature” and “juvenile,” respectively. As previously stated, studies containing animals of an excessively wide age range, which was described as one that considerably overlapped the two broad age categories described above, were excluded from the age comparisons. A number of findings with age-related differences in incidence with statistical significance were present in the data, with higher incidences in mature animals (tubular dilatation in male kidneys, hyaline casts in female kidneys, vacuolation in female adrenal glands, inflammatory cell infiltrates in the thyroid glands, prostate gland, epididymis and spinal cord in males, cyst in the cervix, Balantidium coli in the large intestine in both sexes, inflammation, epithelial degeneration/necrosis and crypt dilatation in the large intestine of females, degeneration/necrosis in the heart of males, decreased lymphoid cellularity of the thymus in both sexes, increased adipocytes in the bone marrow in females, tension lipidosis of the liver in males, decreased sperm and inflammation in the epididymis, mineralization in the seminal vesicle, hypospermatogenesis in the testis) and higher incidences in juvenile animals (edema of the skin in females, parasitism in the stomach of males, inflammatory cell infiltrates in the large intestine and lungs in males and the myocardium in both sexes, increased lymphoid cellularity in the spleen in males and the mandibular lymph node in both sexes, and mineralization in the brain of males). Tubular dilatation in the kidneys was commonly observed in macaques that had presented with diarrhea with large intestinal inflammation or infiltrates, and secondary dehydration.
Discussion
We set out to investigate and document the possible relationship of geographical origin, and other animal-related factors on the incidences of spontaneous histopathology findings in laboratory-raised cynomolgus macaques, based on an extensive analysis of contemporary HCD of animals from different geographical origins. Among the strengths of this article is that, to the best of our knowledge, this survey represents the largest (in terms of numbers of animals) and most comprehensive, contemporaneous retrospective study on the spontaneous pathology of NHP carried out to date. It is also the first compilation of HCD from multiple sites comprising the largest CROs in Europe and North America and therefore involves the largest pool of qualified pathologists in the advent of global harmonization of pathology terminology. The article is also among the few to document the incidences and prevalence of spontaneous histopathology findings in laboratory cynomolgus macaques, in the context of geographical origin, age, sex, and animal husbandry. Among the limitations of this article are the inherent shortcomings of retrospective studies, which include the unequal distribution of numbers of animals and their geographical origin across the sites, which can lead to potential laboratory bias. In addition, a multisite analysis carries with it the potential for interpathologist or interlaboratory variability or bias in terms of recording thresholds and terminology. However, global harmonization of toxicology and pathology protocols and procedures, pathologists’ training, nomenclature (INHAND), multisite sharing of pathology slides through digital pathology, and the performance of rigorous pathology peer reviews of NHP studies by most Sponsors have greatly contributed to the minimization of these potential sources of bias. Several geographical origin-related differences in the incidence of spontaneous histopathology findings were observed in this survey, in addition to a few minor sex- and age-related differences. Contrary to some previous reports that describe lower overall incidences and severity of spontaneous histopathology findings in Mauritian macaques relative to mainland Asian macaques,11,24 our findings show origin-related differences that depended on the type of finding and tissue or organ system. In addition, our investigation revealed that the combined incidences of all findings and the list of most common findings and most commonly affected tissues were generally comparable between animals from different geographical regions. These findings also show a high level of similarity in the incidence of background histopathology findings between cynomolgus macaques from China and Vietnam, further supporting the notion that these subpopulations share similar genetic background and could be interchangeably used for biomedical research. Possible origin-related differences were, however, observed in Mauritian macaques when compared to mainland Asian macaques as detailed below.
Mononuclear Inflammatory Cell Infiltrates
Contrary to previous reports,5,11,24 the incidences of mononuclear inflammatory cell infiltrates in most of the commonly affected tissues, or tissues of toxicological significance, such as the liver, brain, lung, eye, and skin, were nearly twice as high in Mauritian than in continental Asian macaques. Contrastingly, and in agreement with previously published reports,5,11,15 higher incidences of inflammatory infiltrates in the GIT, gall bladder, and some salivary glands were present in Asian in comparison to Mauritian macaques. In addition, lymphoid hyperplasia and hyalinosis of germinal centers in the spleen occurred at higher incidences in Asian macaques, as has also been previously reported. 11 Therefore, this study demonstrated that differences in the incidences of inflammatory infiltrates between Mauritian and Asian macaques are tissue- or organ-dependent and might be further influenced by other extraneous factors, such as exposure to pathogens or lack thereof, the microbiome, and animal husbandry and local environment at the holding facility, in addition to the animal genetics. 5
In most tissues, mononuclear or lymphoplasmacytic inflammatory infiltrates occurred without concomitant damage to parenchymal organs, as has been previously reported,22,26 and these simple infiltrates are frequently not associated with changes in clinical chemistry.22,24 Spontaneous mononuclear infiltrates without inflammation are believed to represent the trafficking of activated immune cells (B and T lymphocytes) into nonlymphoid, antigen-rich tissues, either after injury or in the steady state, in response to antigenic or cytokine signals. 27 Unlike cellular components of the innate immune system, such as granulocytes, which only appear at sites of inflammation, primed T-cells and activated regulatory T-cells (Tregs) traffic to the antigen-rich nonlymphoid tissue to exert effector and regulatory responses, respectively, whether or not there is an active inflammatory process,27,28 and communicate through short-range cytokines and/or direct cell-cell contact. 29 Therefore, most of the inflammatory infiltrates observed in a variety of tissues under noninflammatory conditions in this study represent trafficking T-cells, 28 although aggregation of some innate immune cells, such as macrophages, may also be present. 29
Having a working knowledge of the incidence, nature, severity grades, and distribution of spontaneous mononuclear inflammatory infiltrates in several key organs has gained increasing significance in the advent of targeted therapy, particularly in oncology, where on-target off-tumor findings may present as inflammatory reactions in subanatomic locations where the target is known to be expressed. The inflammatory response associated with target engagement may mimic some spontaneous inflammatory changes in test-article-treated macaques. Conversely, a pathologist’s knowledge of the target expression profile may lead to an over-interpretation of spontaneously occurring findings in tissues where the target is known to be expressed, due to the underlying bias toward finding treatment-related effects in tissues concerned, particularly in studies with a limited number of animals and no control animals.
In addition to target-related lesions, a number of biopharmaceutical modalities, such as bispecific T-cell engagers—described elsewhere in this Special Issue, can lead to a nonspecific and immunostimulatory increase in inflammatory cell infiltrates in various tissues. 30 The immunostimulatory effects of these cancer biotherapeutics may also be mediated by changes in cytokine levels that are secondary to activation of macrophages, natural killer (NK) cells, and neutrophils through ligation of their IgG Fc-receptors. Other immunostimulatory modalities, such as RNA-based therapies, for example, toll-like receptor (TLR) agonists can also increase inflammatory cells infiltrates through stimulation of the innate immune system by inducing phagocytosis and secretion of inflammatory cytokines, such as IL-1, IL-6, and TNF-α.31,32 In general, cytokine release stimulates the activation of vascular endothelial cells, which results in a recruitment of leukocytes into the affected tissue in which inflammation and inflammatory infiltrates can occur. 33 In the liver, in particular, sinusoidal leukocytosis and Kupffer cell activation, with or without sinusoid dilatation, platelets or erythrocyte sequestration, or hemosiderin pigment in Kupffer cells may be observed as a nonspecific innate liver response to general cytokine release, 34 in addition to immune complex disease associated with monoclonal antibody therapeutic agents, 35 certain antibody drug conjugates,36,37 or bispecific T-cell engagers. 38 Many aspects of these liver lesions (sinusoidal leukocytosis, sinusoidal dilation, Kupffer cell activation, and congestion with hemosiderosis) frequently occur spontaneously in macaques with some opportunistic infections, particularly in the GIT (Figures 3 and 4); and diarrhea and soft feces are generally the most common clinical observation observed in these animals. An in-depth discussion on diarrhea and GIT pathology in macaques is discussed elsewhere in this issue (Johnson et al. 2022) 39 .
Lung and Cardiovascular System
The incidences of spontaneous findings of toxicological relevance in the lung and cardiovascular system were largely in agreement with the previously reported incidences.15,22,40 In the lung, the overall incidence of commonly observed findings, increased alveolar macrophages and mononuclear inflammatory cell infiltrate, is in agreement with those previously reported.22,40 However, in a previous study comparing incidences of spontaneous lung histopathology findings between inhalation and noninhalation studies, considerably higher incidences were observed in inhalation studies, 40 providing evidence that study type can modulate incidences of spontaneous lung histopathology findings. Our results showed a similar trend (data not shown) and comparable incidences to this previous publication. 40 The authors suggested several reasons for the higher incidences of spontaneous lung histopathology in inhalation studies. The first is a possible unconscious bias or over-sensitization of the pathologists when evaluating the lung as a target tissue in inhalation studies, which might lead to an “over-recording” of spontaneous findings (lowering the threshold). The second possible reason is the increased number of lung (lobes) sections (up to eight sections) sampled in inhalation studies compared to oral/gavage studies, which might lead to an increased chance of encountering focal findings. Another cause of increased incidences in inhalation studies could be linked to some nebulized or powdered vehicle-inducing phagocytic clearance by alveolar macrophages. 40 Overall, these data from the literature show that there are several confounding factors that can modulate the incidences of spontaneous lung histopathology, which include, the dose route, sampling, and fixation methods (number of lung lobes and trimming orientation, inflation, or immersion fixation), and other study procedures, such as bronchioloalveolar lavage. However, considering these confounding factors were comparably distributed among animals from different geographical origins in our study, the observed differences between animals from different geographical origins may represent true origin-related differences. Therefore, geographical origin should be considered when interpreting lung findings or requesting HCD of the lungs in cynomolgus macaques. This is of particular importance in studies with inhaled biologics. In these studies, the characteristic pattern of perivascular/peribronchiolar mononuclear inflammatory cell infiltrates, which is considered to occur secondary to immunogenicity and increased alveolar macrophages secondary to clearance mechanisms, 41 may be difficult to differentiate from spontaneous histopathology findings.
The lower incidences of lung pigment in Mauritian compared to Asian macaques has been previously reported 11 and is considered to represent lower incidences of pneumoconiosis or anthracosis in the island macaques. Pneumoconiosis, the inhalation of mineral dust particles, such as carbon (anthracosis) or silica (silicosis), is considered to result from the inhalation of carbon at the breeder or supplier’s facility as a consequence of the local environmental air quality. 40 Pneumoconiosis pigment tends to asymptomatically accumulate in macrophages with perivascular or peribronchial distribution and should be differentiated from pigment associated with the lung mite, Pneumonyssus simicola, which is usually associated with pathological changes in the lung, such as dilated, thickened bronchioles with epithelial erosion or ulceration, and mixed infiltrates comprised lymphocytes, eosinophils, and macrophages. All types of lung pigments may extend to local draining lymph nodes, such as the bronchial or mediastinal lymph nodes.
Incidences of pleural fibrosis/adhesions were higher in Mauritian macaques compared to Chinese and Vietnamese macaques, indicating some possible origin-related differences of unknown pathogenesis but of little or no clinical significance. 22 Toxicologically, lung adhesions/fibrosis and other lung inflammatory findings should be differentiated from target-related lung injury, or viral-induced lesions in Covid-19 vaccine efficacy studies. In these cases, pretrial screening of animals by various imaging techniques can be useful, particularly in studies where there are no control animals.42,43
Although a slightly wider range of heart findings were observed in Mauritian when compared to Asian macaques, contrary to previously published reports, 16 there was no increase in the incidences of myocardial degeneration in Mauritian macaques when compared to those from Asia. In addition, the incidences of background cardiovascular findings in animals from most geographical origins in this study were generally much lower than those previously reported,22,44 indicating an overall trend toward animals with less cardiovascular lesions. Since stress and the repeated bouts of endogenous catecholamine release associated with transportation, captivity, and routine manipulation during toxicology studies are believed to be at least in part, responsible for myocardial degeneration/necrosis,16,21,22,45 the improvements in the standards of animal husbandry, enrichment, and housing (e.g., socially compatible cage mates) could be responsible for this decrease in the incidence of heart lesions. Along the same lines, the increase in cardiac lesions in mature males may be a related to increased aggression in this group resulting in more social stress.
Brain, Spinal Cord, Eye, and Lacrimal Glands
Inflammatory infiltrates in the brain frequently presented as focal or multifocal perivascular mononuclear cells infiltrations affecting the parenchyma, the meninges, or the choroid plexus. 46 As previously stated, the incidence of this finding in Mauritian macaques was approximately twice that in Chinese- and Vietnamese-origin macaques. The increase in the incidence of mononuclear infiltrates in the brain in Mauritian macaques was determined not to be related to study procedures or dosing since there were no macaques of Mauritian origin that were dosed by the intrathecal route (therefore, results were the same after the few animals dosed by this route were removed), and the distribution of other procedures and dosing routes was comparable between Asian and Mauritian. Therefore, the increase in the incidence of mononuclear inflammatory cell infiltrates in the brain of macaques of Mauritian origin is also likely to be origin-related, although the likely cause of these inflammatory infiltrates in macaques is not known. Axonal degeneration was observed in the spinal cord in our retrospective study, which is contrary to the general absence of histopathologic findings in the spinal cord in the previous retrospective studies.22,46 The presence of spinal cord findings in our study is likely a result of the much larger sample size, scope, and the diverse source of animals in this study. Therefore, the increased incidences of axonal degeneration of the spinal cord in Chinese and Vietnamese macaques when compared to those from Mauritius most likely reflect a true geographical origin-related difference. Mononuclear cell infiltrates in the uvea of the eye, which showed higher incidences in Mauritian macaques, have previously been reported,47,48 and the incidences reported for Mauritian macaques 47 are comparable to those observed in our study. In general, these infiltrates are not visible during the ophthalmoscopic examination. Some minor sex-related differences were observed in the brain and eye. For example, mineralization in the brain showed a higher incidence in females relative to males, and pigment in the brain and infiltrates in the eye were observed at a higher incidence in males than females. These sex-related differences have not been reported before.
Hematopoietic and Lymphoid Systems
Compared to macaques of Asian origin, macaques from Mauritius have been reported to have lower incidence and/or severity of spontaneous pathological findings in the hemolymphoreticular system. 11 Increased lymphoid cellularity was a common finding in the hematopoietic and lymphoid systems in animals from all geographical origin. Although this finding has previously been associated with subclinical type D retroviral infection, 21 most macaques used in nonclinical studies are routinely tested for simian retroviruses. Given the rigid screening processes for infectious disease and clean laboratory environment,49,50 it is most likely that increased lymphoid follicles observed in the hemolymphoreticular system of these animals represent heightened immunosurveillance and response to a nonspecific stimulus. However, it should be acknowledged that the diagnosis of increased cellularity in lymphoid tissue in particular may be subject to interpathologist variability in the recording thresholds for this finding in control groups. In the spleen, the terms lymphoid hyperplasia or increased lymphoid cellularity are also used to describe the large, misshapen, and sometimes coalescing lymphoid follicles that often result in compression of the surrounding tissue and have irregular and bizarre germinal centers that often exhibit hyalinization. Various theories have been advanced as to their cause or etiology.22,51-53 The first and most espoused theory is that they represent nodular hyperplasia of the white pulp, and to support this theory are the frequently present hyalinized germinal centers in the same follicles or spleens, which are considered to represent antigen-antibody complexes deposition, possibly as a result of persistent antigenic stimulation.22,51 However, others consider these to be developmental in origin and have thus proposed to use the term “compound follicles” to describe this common observation in the macaque spleen. 54 This background lesion and other spontaneous presentations of increased lymphoid cellularity in the lymph node, spleen, or bone marrow should be differentiated from lesions induced by immunostimulatory small molecules, such as oligonucleotides TLR agonists,31,55 or immunostimulatory immuno-oncology biotherapeutics, such as immune checkpoint inhibitors, 54 or compounds that co-stimulate T-cells, such as the anti-CD134 (anti-OX40) antibody. 56 In such cases, evidence of widespread mononuclear infiltrates (lymphocytes and macrophages) in various organs associated with test-article-induced T-cell proliferation and increased spleen weights may help in differentiating test-article effects from spontaneous background findings. Lymphocytic infiltrates in the bone marrow associated with immunostimulatory compounds can be differentiated from spontaneously occurring lymphoid follicles in the bone marrow by the absence of clearly formed follicles with germinal centers in the case of treatment-related immunostimulation, and the presence of similar multiorgan infiltrates. Therefore, a careful comparison of test-article-treated animals to controls is necessary to distinguish spontaneous background findings from those that may be related to an administered pharmacologic potentially causing immunostimulation.
Hyalinization of germinal centers was often associated with lymphoid hyperplasia and sometimes with Russell bodies, fibrosis, and histiocytic accumulation. Other findings recorded in the spleen commonly included focal capsular fibrosis, histiocytic infiltration, vacuolated macrophages, and pigment. Histiocytic vacuolation has been observed after treatment with a fully human mAb with a silent Fc 57 and was considered to represent a nonspecific response to antigen clearance or impaired antibody-antigen complex clearance. Histiocytic vacuolation should also be differentiated from polyethylene glycol (PEG)-related vacuolation or phospholipidosis, which usually involve multiple organs and other type of cells. 58 Lymphoid atrophy, potentially resulting from stress or inanition was observed in lymphoid tissues. In the thymus, atrophy should be differentiated from involution. Lymphoid atrophy, potentially resulting from handling or nutritional stress or inanition was observed in lymphoid tissues. In the thymus, atrophy should be differentiated from age-related or physiological involution (or atrophy), which is usually a gradual and progressive process that generally starts at the onset of sexual maturity in macaques, although females are known to involute more slowly or later than males. However, because age-related thymic involution can be influenced by other factors, such as stress, pregnancy, steroid hormone status, antigenic load, and nutrition, there is an inherent variability in the morphological appearance of the macaque thymus that must be considered when evaluating the organ.59,60 In addition, due to these modifying factors, involution may occur much earlier in some individual animals within a study. The thymus can also undergo acute atrophy under conditions of physiological stress, such as animal handling, study procedures, infection, or treatment with therapeutic agents.
We also set out to investigate the possible influence of age and sex on the incidences of spontaneous histopathology findings, such as inflammatory infiltrates and inflammation. Unsurprisingly, inflammatory infiltrates and inflammation in the urinary bladder were higher in females than in males since females generally have higher incidences of cystitis than males 61 (shorter urethra), while the observed bone marrow changes of increased hematopoiesis could be related to menses-related anemia. 62 However, the reasons behind other sex-related differences observed in the stomach, (increased glandular dilatation and inflammatory infiltrates in females), skin (edema and subcutaneous inflammation), adrenal gland (cortical vacuolation and mineralization), brain (mineralization), lymph nodes (extramedullary hematopoiesis), liver (hepatocyte vacuolation and inflammatory infiltrates), kidney (basophilic tubules and hyaline casts), and pituitary gland (vacuolation) are not known. While the likely reasons behind many of the age-related differences were unclear, physiological processes associated with growth could explain some of the age-related differences, such as the age-related decrease in lymphoid cellularity of the thymus (involution).
The difference in the incidences of spontaneous lesions between Asian and Mauritian macaques is considered likely to be due to a combination of both environmental and genetic factors.5,17 Environmental factors, such as early or late exposure to infectious agents, the microbiome, housing and social dynamics, diet and water source at the breeding or holding facilities, and air quality, may modulate the rate and occurrence of spontaneous histopathology findings. 5 With the modern implementation of very high standards of NHP housing and husbandry by most breeders and test facilities in recent years, 1 it is likely that the animal genetics and the microbiome, including early or lack of exposure to pathogens, play a bigger role in the differences observed between Asian and Mauritian macaques. Due to their total isolation for centuries, Mauritian macaques have developed different immune responses related to their major histocompatibility complex allele concentrations. 63 Coupled with the lack of exposure to certain pathogens, such as Plasmodium spp., compared with macaques obtained from Asian sources, 64 differences in the incidence of inflammatory infiltrates in certain tissues are unsurprising.
Conclusions and Recommendations
In conclusion, while there are very little differences in the spectrum of spontaneous histopathology findings between animals from different geographical origins, there are significant origin-related differences in the incidences of some of the more commonly observed spontaneous findings between cynomolgus macaques (of the same age range) from mainland Asia and Mauritius. Since some spontaneous findings may resemble test-article-related findings, the mixing of animals from different geographical origins within a study can present an interpretative challenge due to the significant differences in the incidences of spontaneous findings, and this can potentially affect the outcome of a study and the fate of a drug development program. It can also be concluded from the results of this survey that the incidences of spontaneous findings between cynomolgus macaques of different origins, but within mainland Asia (e.g., Chinese and Vietnamese), appear to be comparable and therefore not as different as that between animals from mainland Asia and Mauritius. Considering the above, our recommendations are that, for consistency within a drug development program and to generate reliable and reproducible data that can be compared between toxicological studies, it is recommended to use cynomolgus macaques from one source on a program. 5 If this is not possible, Indochinese (China, Vietnam, and Cambodia) macaques of the similar age range may be used interchangeably on a drug development program as has been previously proposed (Charles River White paper). Most importantly, however, it is strongly recommended to avoid mixing animals from multiple sources in a single toxicology study, due to the reasons given above. In addition, it is also strongly recommended to use geographic origin-based HCD or published literature to gain baseline knowledge about macaques from different geographical origins.5,65
Supplemental Material
sj-pdf-1-tpx-10.1177_01926233221096424 – Supplemental material for The Influence of Geographical Origin, Age, Sex, and Animal Husbandry on the Spontaneous Histopathology of Laboratory Cynomolgus Macaques (Macaca Fascicularis): A Contemporary Global and Multisite Review of Historical Control Data
Supplemental material, sj-pdf-1-tpx-10.1177_01926233221096424 for The Influence of Geographical Origin, Age, Sex, and Animal Husbandry on the Spontaneous Histopathology of Laboratory Cynomolgus Macaques (Macaca Fascicularis): A Contemporary Global and Multisite Review of Historical Control Data by Ronnie Chamanza, Stuart W. Naylor, Michela Gregori, Molly Boyle, Marcia E. Pereira Bacares, Elodie Drevon-Gaillot, Annette Romeike, Cynthia Courtney, Kelsey Johnson, Julie Turner, Nadine Swierzawski and Alok K. Sharma in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
